Catalytic converters have expensive metals and can convert
1.
2.
3.
4. More than one option is correct
White spots seen on mustard leaves are due to a parasitic fungus known as
1. Puccinia graminis
2. Phytophthora infestans
3. Albugo candida
4. Mucor mucedo
During hypertrophy of the muscles which of the following will not increase?
1. Number of myofilaments
2. Number of mitochondria
3. Number of myofibrils
4. Number of muscle fibers
Which of the following amino acid is not coded by six codons?
1. Serine
2. Arginine
3. Alanine
4. leucine
Flexing of the arm is an example of
1. First-class lever system
2. The second class lever system
3. The third class lever system
4. This event can't be categorized as a lever system
Select an incorrect match with respect to resistant varieties of common crops
1. Pusa swarmim - Cauliflower
2. Pusa Komal - Cow-pea
3. Himgirl - Wheat
4. Pusa Gaurav - Brassica
Why hands and feet get swollen with accumulated fluid (ooedema) in persons suffering from dietary deficiency of proteins in kwashiorkor?
1. Plasma proteins serve as a source of proteins for tissue cells
2. Albumin and globulins retain water in the blood plasma by their osmotic effects
3. Albumin and globulins transport many substances such as thyroxine and in combination with them
4. Tissue cells plasma proteins for forming their cellular proteins
Myenteric nerve plexus is located in
1. Sub-mucosa
2. Muscularls mucosa
3. Muscularis externa
4. Mucosa
Which of the following does not occur during accommodation reflex with respect to near vision?
1. Contraction of circular ciliary muscles
2. Relaxation in suspensory ligaments
3. The lens becomes more spherical
4. Relaxation of circular ciliary muscles
Which of the following is not likely to happen in his neurohormonal control system, in response to the stress of any kind?
A. Hypothalamus activates the parasympathetic division of brain
B. The sympathetic nervous system is activated releasing epinephrine and norepinephrine from the adrenal cortex.
C. The craniosacral outflow is stimulated.
D. The sympathetic nervous system is activated releasing epinephrine and norepinephrine from the adrenal medulla.
1. A & B only
2. A & C only
3. A, B & C only
4. A, B, C & D
An unusual polysaccharide, formed of fructose residues and representing reserved food in tubers, is
1. Callose
2. Inulin
3. Glycogen
4. Hemicellulose
Trochlear is the thinnest and smallest cranial nerve originating from the floor of the midbrain. What type of nerve is it and which eyeball muscle does it innervate?
1. Motor, lateral rectus
2. Sensory, superior rectus
3. Motor, superior oblique
4. Sensory, median rectus
Read the following four statements A, B, C and D and select the right option having both statements alongwith one word scientific term correct for them
A. Free floating form of cnidaria : Medusa
B. Blood filled cavity in arthropods : Pseudocoel
C. Construction of an animal such as earthworm by repeated segments : Metamerism
D. Individual animals having both organs of sexes : Dioecious
(1) B & D
(2) A & B
(3) A & C
(4) C & B
For the enzyme phosphofructokinase which of the following is an allosteric inhibitor?
1. ADP
2. ATP
3. Glucose-6-phosphate
4. Isoleucine
Which of the following is a characteristic of chordates but it is not found in other animals?
(1) A notochord
(2) Jointed appendages
(3) An exoskeleton
(4) All of these
The metameric segmentation is present in
(1) Annelids only
(2) Annelida and Arthropoda
(3) Platyhelminthes and Annelida
(4) Earthworm and Taenia solium
All of the following are effects of hypothyroidism, except
1. Cretinism
2. Low intelligence quotient
3. Tachycardia
4. Irregular menstrual cycle
Which thyroid hormone is not stored as colloidal material in the thyroid follicles?
1. Calcitonin
2. Thyroxine
3. Triodothyronine
4.
Which of the following is not a derivative of tyrosine?
1. Epinephrine
2. Norepinephrine
3. Dopamine
4. Serotonin
Following are the reasons behind the explosive growth of population except
1. The decline in the maternal mortality rate
2. Decline in infant mortality rate
3. Decline in number of people in the reproductive age group
4. Decline in the death rate
The technique which uses amniotic fluid for detection of prenatal disorders is
1. Laparoscopy
2. Endoscopy
3. Amniocentesis
4. Ultrasound sonography
Mark the incorrect statement regarding the 23rd day of a 28-day menstrual cycle
1. High plasma estrogen level
2. Low plasma FSH and LH level
3. High plasma progesterone level
4. Spasm of spiral arteries of endometrium
Which of the following is an example of a three-horned herbivorous dinosaur that was common in the upper cretaceous period?
1. Brachiosaurus
2. Stegosaurus
3. Tyrannosaurus
4. Triceratops
There are five factors known to affect Hardy Weinberg equilibrium, which of the following cannot be included in the five factors?
1. Gene flow and genetic drift
2. Genetic recombination and natural selection
3. Mutation
4. Isolation
Following is a diagrammatic representation of an experiment. Study the diagram and answer the questions given
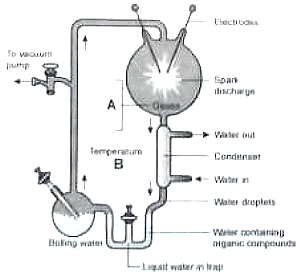
A. Name the gases present in the spark chamber
B. What was the temperature of the experiment?
C. Name the amino acid formed.
D. Name the scientist who conducted this experiment
A B C D
1. , , 800 Glycine, Alanine, Oparin and Haldane
Aspartic acid
2. , , , 800 Glycine, Miller
Aspartic acid
Alanine
3. , , 800 Glycine, Urey and Miller
Glutamate,
Alanine
4. , , 800 Glycine, Urey and Miller
Alanine,
Aspartic acid
Which of the following property of 3rd line of defense is the main basis of immunization?
1. Neutralization
2. Memory
3. Discrimination between self & non-self
4. Specificity
Which human disease is associated with a social stigma and is also known as 'Kushtha Rog'?
1. T.B.
2. Leprosy
3. Syphilis
4. Gonorrhea
Conducting part of respiratory system extends from
1. Trachea to bronchi
2. External nostrils to trachea
3. External nostrils to terminal bronchioles
4. External nostrils to alveolar ducts
Which of the following laryngeal cartilages is paired and consist largely of hyaline cartilage with only the upper end made of elastic cartilage?
1. Thyroid cartilage
2. Cricoid cartilage
3. Arytenoid cartilage
4. Cuneiform cartilage
Which of the following statement is incorrect?
1. Hepatic portal system pours blood from digestive system into the liver
2. Renal portal system is well developed in fishes and amphibians
3. First heart sound is produced at the onset of ventricular diastole
4. Increased vagal tone decreases heart rate
Following is the diagrammatic representation of formed elements in the blood, Which of the following is the function of WBCs labeled as 'A'?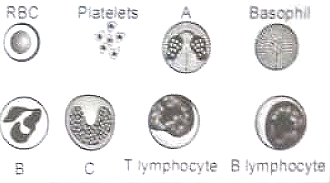
1. They are phagocytic cells which destroy foreign organisms entering the body
2. They secrete histamine serotonin heparin and are involved in inflammatory reactions
3. They resist infections associated with allergic reactions
4. They are responsible for immune responses of the body
Mark the incorrect match
1. Pashmina - Kashmiri goat - Cashmere shawis
2. Shahtoosh - Tibetian antelope - King of wools (Pantholops Hodgson)
3. Angoora Sheep - Superior - carpet wool
4. Merino - Exotic breed of sheep - Most popular fine wool breed
Surgical methods, also called sterilization, are used as terminal contraception methods to prevent any more pregnancies Surgical intervention
| 1. | Inhibits gamete release from gonads |
| 2. | Inhibits gamete formation |
| 3. | Blocks gamete transport |
| 4. | All of these |
Which of the following are not completely curable STDs?
1. AIDS and hepatitis-B
2. AIDS and genital herpes
3. Hepatitis-B and genital herpes
4. All of these
Which of the following is the direct method of gene transfer
1. Plasmid
2. Electroporation
3. BAC
4. Bacteriophage
Introduction of the plasmid into the host bacterium can be helped by
1. Treating the cells with a specific concentration of a divalent cation such as calcium
2. Enzyme B-galactosidase
3. Cellulase
4. Lysozyme
Select the incorrect match
1 Monascus - statins
2 Trichoderma - Thurioside
3 Aspergillus - citric acid
4 propionibacterium - swiss cheese
Which of the following was the first transgenic animal produced for food production?
1. Transgenic pig
2. Transgenic goat
3. Transgenic salmon
4. Transgenic cow
Production of Humulin by Eli Lilly company was done in
1. 1990
2. 1983
3. 2004
4. 2001
Which of the following conditions will not activate the JG cells to release renin so that glomerular blood flow and GFR come back to normal;?
1. Decrease in blood volume
2. Fall in ion concentration in the blood plasma
3. Rise in GFR
4 Fall in blood pressure
Following is a comprehensive list of excretory organs and the wastes they remove. Choose the incorrect pair
1. Lungs - Remove
2. Sebaceous glands - Sterols, Bilirubin, vitamins
3. Sweat - Water, urea. NaCI, lactic acid
4. Kidneys - Urea, uric acid, creatinine,
In a hospital 5 children are born on a particular day. What is the probability of three male children?
1. 1/2
2. 1/8
3. 3/5
4. 5/16
What ratio of coloured and white flower can be obtained by making cross in sweet pea with genotype CcppCcPp?
1. 6 : 10 respectively
2. 2 : 6 respectively
3. 5 : 3 respectively
4. 9 : 7 respectively
Eleven base pair per turn are generally present in which type of DNA?
1. B - DNA
2. C - DNA
3. D - DNA
4. A - DNA
Which of the following statement is not related to centriole?
1. Made up of nine evenly spaced peripheral fibrils of tubulin protein
2. Spindle fibres that give rise to spindle apparatus during cell division in plant cells
3. Central part of the proximal region is called the hub
4. It form the basal body of eukaryotic flagella
A number of organised flattened membranous sacs are present in the
1. Matrix of mitochondria
2. Nucleoplasm
3. Chloroplasts
4. Matrix of centriole
Eustachian tube, epiglottis and pinna of ear consist of
(1) Elastic cartilage
(2) Hyaline cartilage
(3) Calcified cartilage
(4) All of these
Who emphasized the need for initiation of work on artificial culture of immature embryos ?
1. B.M. Johri
2. S.C. Maheshwari
3. Panchanan Maheshwari
4. Shipra Guha
Which of the following statements is not correct ?
1. Development of flower is a random process
2. Theca in anther is often separated by longitudinal groove
3. Sporogenous tissue have a group of compactly arranged homogenous cells
4. Germpore is part of exine which lacks sporopollenin
One of the problems of hybrids is that hybrid seeds have to be produced every year. This problem can be overcome with the help of ?
1. Seed dormancy
2. Apomixis
3. Polyembryony
4. All the above
Statement -I- Transformation of ovule into seed and ovary into fruit proceeds simultaneously.
Statement - II- Development of endosperm and embryo is simultaneous
Options
1. Both I and II are correct
2. Only I is correct
3. Only II is correct
4. Neither I nor II is correct
How many of the following statements are correct ?
(1) Regulation of lac operon can be also viewed as regulation of enzyme synthesis by its substrate
(2) Transcription and translation is tightly regulated because it is energetically expensive process
(3) Translation is a process that has evolved around RNA, indicating that life began around RNA
(4) In bacteria, the transcribed mRNA is functional, hence can directly be translated
Options
1. 1,2 & 3
2. 1,2,3 & 4
3. 2,3 & 4
4. 1,3 & 4
DNA fingerprinting analysis have application in
1. Forensic science
2. Determining population diversities
3. Determining genetic diversities
4. All the above
During the process of translation ribosome moves from codon to codon along the mRNA, amino acids are added one by one, this process is dictated by DNA & represented by ?
1. mRNA
2. DNA
3. t-RNA
4. Polypeptide
Which of the following statements are correct ?
(1) Deforestation leads to enhanced green house effect
(2) Soil pollution can be due to agricultural chemicals and leachates from solid wastes deposited over it
1. Both 1 & 2 are correct
2. Only 1 is correct
3. Only 2 is correct
4. Neither 1 nor 2 is correct
Which of the following is not correctly matched ?
1. Histone - Lysine & Arginine
2. Euchromatin - Transcriptionally active
3. DNA genetic material - Radioactive phosphorus
4. Transformation - DNA thermally unstable
Process of reforestation can be speed up by
1. Planting alien species
2. With due consideration of earlier biodiversity
3. By planting cross-pollinated plants
4. With the help of greenhouse effect
Population density can be expressed in terms of :-
1. Numbers
2. Biomass
3. Per cent cover
4. All the above
Which of the following statement is not correct ?
1. Herbivores and plants are more adversely affected by competition than carnivores
2. Life cycle of endoparasite is more complex
3. Predation leads to decrease in species diversity
4. Selection pressure leads to most efficient reproductive strategy in organism
The technology of biogas production was developed in India by
1. IARI
2. KVIC
3. ICMR
4. Both 1 and 2
How many of the following is correct for phenylketonuria ?
(1) It is in born error of metabolism
(2) It is autosomal recessive disorder
(3) It is an example of pleiotropy
(4) There is accumulation of tyrosine
(5) It leads to mental retardation
(6) It leads to decrease pigmentation of hair & skin
1. All
2. 5
3. 4
4. 3
Which of the following statement is not correct ?
1. Chromosomal disorder can be easily studied with the help of Karyotypes
2. Inheritable mutation can be studied by generating pedigree of a family
3. Law of independent assortment always hold true for the genes that were located on same chromosome
4. Linkage maps corresponds to arrangement of genes on a chromosome
Which of the following is not correctly matched
| 1. | Honey bee - Haploidiploid sex determination |
| 2. | Birds - Male birds heterogametic |
| 3. | Grasshopper - XX-XO |
| 4. | Drosophila - XX-XY |
Polygenic inheritance
1. Are spread across the gradient
2. Are controlled by three or more genes
3. Are influenced by the environment
4. All the above
Mark the incorrect match
1. Tissue culture - Explant
2. Spirulina - Protein source
3. Atlas - 66 - Biofortification
4. Parbhani Kranti - Mutational breeding
Statement - I - Nearly 700 species have become extinct in recent times
Statement - II- More than 15,500 species are facing threat of extinction out of which more than 650 are from India
Options
1. Both I & II are correct
2. Only I is correct
3. Only II is correct
4. Both I & II is incorrect
How many species on planet earth waiting for to be discovered ?
1. 7 million
2. Nearly 6 million
3. 1.7 - 1.8 million
4. 8.5 million
Which of the following environmental factor/s regulate the rate of release of nutrients into the atmosphere ?
1. Soil moisture
2. pH
3. Temperature
4. All
Which of the following do not take place during successive seral stage ?
1. Increase in number of species
2. Increase in number of organism
3. Increase in total biomass
4. Increase in endemism
Which of the following statements is not correct?
1. Only human beings are self conscious
2. All living organism - present, past and future are linked to one another by the sharing of the common genetic material.
3. No non-living object is capable of self-replication
4. In living organisms there is no underlying interaction.
Which of the following statement is not correct?
1. Less than 21 elements are essential and benificial for plants out of known 105.
2. Nitrogen fixation requires strong reducing agent.
3. Most of the nitrogen fixation takes place during aerobic condition.
4. Absorption of mineral by root is either active or passive.
How many of the following is function of boron ?
i. Uptake and utilisation of
ii. Membrane functioning
iii. Pollen germination
iv. Cell elongation
v. Cell differentiation
vi. Carbohydrate translocation
Options
1. All
2. 5
3. 4
4. 3
Plant growth regulator's
| 1. | May act synergistically or antogonistically |
| 2. | Are intrinsic intercellular factor |
| 3. | Are simple, molecules of diverse chemical composition |
| 4. | All of the above |
Which of the following is not correctly matched
1. Viroids - Low molecular weight RNA
2. Prions - Abnormally folded protein
3. Lichen - Pollution indicator
4. Viruses - DNA and RNA as genetic material
Bladderwort and venus fly trap are :
1. Photoautotrophic
2. Partially heterotrophic
3. Parasitic
4. Saprophytic
Which of the following is not correct for blue green algae.
1. Have chlorophyll 'a' similar to higher plants.
2. Are unicellular, colonial or filamentous.
3. Fresh water/marine or terrestrial.
4. Generally biflagellated
Which tissue system forms the main bulk of the plant body ?
1. Ground tissue system
2. Vascular tissue system
3. Epidermal tissue system
4. Sub - cellular epidermal tissue system
Mark incorrect match :
| 1. | Phloem parenchyma - stores resin, latex and mucilage |
| 2. | Xylem parenchyma - stores starch or fat |
| 3. | Ray parenchyma - Radial conduction of water |
| 4. | Phloem fibres - composed of collenchyma |
Statement - I : Internal concentration of is major limiting factor for photosynthesis.
Statement - II: Effect of water as a factor is more through its effect on the plant, rather than directly on photosynthesis.
1. Both the statements are correct.
2. Only first statement is correct.
3. Only second statement is correct.
4. Both the statements are wrong.
How many of the following statements are true for respiration?
i. During aerobic respiration ultimate acceptor of electron is and it gets reduced to water.
ii. Respiration is catabolic pathway only.
iii. The initial stage of cellular respiration takes place in cytoplasm.
iv. Fermentation can take place during germination of seed.
Options :
1. All statements are correct
2. 3
3. 2
4. 1
Organisms belonging to the same taxa have a common ancestor, is related to which system of classification.
1. Natural system
2. Phylogenetic system
3. Artificial system
4. Numerical system
How many of the following statements are correct?
i. Meiosis in zygote in case of funaria is delayed.
ii. In gymnosperm female gametophyte is endosperm.
iii. Spirogyra have motile male gamete.
iv. Prothallus of pteris represents sporophyte.
Options :
1. All statements are correct
2. 3
3. 2
4. 1
Anisogamous reproduction is shown by :-
1. Ulothrix
2. Udorina
3. Oedogonium
4. All of the above
Which of the following is not characteristic of family Liliaceae ?
1. Perennial herbs
2. Exstipulate leaf
3. Non endospermic seed
4. Axile placentation.
Leaf develops
1. Exogeneously at node
2. Endogeneously at node
3. Mesogeneously at node
4. Schizogeneously at node
Stem tendrils are present in all, except
1. Pea
2. Cucumber
3. Grapevines
4. Watermelon
Most of the organelle duplication during cell division occurs at which phase
1. M - Phase
2. - Phase
3. S - Phase
4. - Phase
Sister chromatids separate from one another during:
1. Anaphase
2. Anaphase - I
3. Anaphase - II
4. Both 1 and 3
Interkinesis refers to :-
1. Phase between to interphase
2. Phase between meiosis - I and meiosis - II
3. Phase between two M - Phase
4. Phase between two - Phase
During mass phase flow of food which cell is responsible for development of pressure gradient in phloem.
1. Phloem parenchyma
2. Xylem parenchyma
3. Companion cell
4. Bast cell
An absorption line of the lowest frequency in the transition of hydrogen spectra is:
1. n=1 to n=2
2. n=3 to n=8
3. n=2 to n=1
4. n=8 to n=3
Which of the alkali metal chloride is having the highest m.p. ?
1. LiCl
2. NaCl
3. KCl
4. RbCl
When allylic alcohol (CH₂=CH-CH₂OH) is oxidized in the presence of bromine, which of the following products will be formed?
1. Oxalic acid
2. Formic acid
3. Succinic acid
4. Acrylic acid
Correct IUPAC name of the compound
1. 4-(Ethyl methanolyonxy)phenylpropanoate
2. Ethyl 4-propanoyloxybenzenecarboxylate
3. 4-(1-Oxo-2-oxabutyl)phenylpropanoate
4. 1-(1-Oxo-2-oxbutyl)-4-(1-oxopropoxy)benzene
2–Phenylcycloprop–2–en–1–one is allowed to react with phenylmagnesium bromide
and the reaction mixture is hydrolyzed with perchloric acid. The product formed is
| 1. |  |
| 2. |  |
| 3. |  |
| 4. | 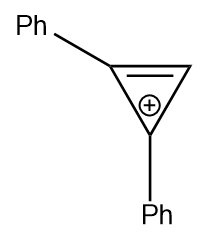 |
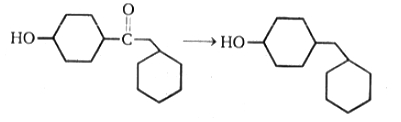
The above reduction can be best carried out by:
1. Clemmensen reduction
2. Wolff-Kishner reduction
3.
4. None of the above
A combination of nitrocellulose and nitroglycerine is a propellant of the type:
1. Hybrid propellant.
2. Mono propellant.
3. Double based propellant.
4. Biliquid propellant.
Assuming no change in volume, calculate the minimum mass of NaCl (approx.) necessary to dissolve 0.01 mol AgCl in 100 L solution.
[Given
1. 15 kg
2. 21 kg
3. 10 kg
4. 19kg
At identical temperature and pressure, the rate of diffusion of hydrogen gas is times that of a hydrocarbon having molecular formula . What is the value of n?
1. 1
2. 4
3. 3
4. 8
For one mole of van der Waal's gas when b=0 and T=300 K, the PV vs. 1/V plot is shown below.
The value of the van der Waal's constant 'a' (atm. ) is-
1. 1.0
2. 4.5
3. 1.5
4. 3.0
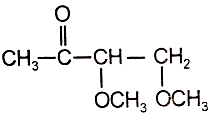
An organic compound (A) of the molecular formula gives a positive Iodoform test but does not reduce Tollen's reagent. When (A) is subjected to acid-catalyzed hydrolysis, another compound B is formed which gives positive Tollen's test. Identify the unknown compound (A).
1. 
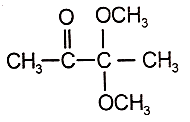
2. 
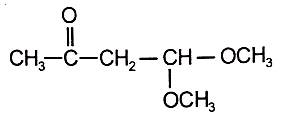
3. 
4. 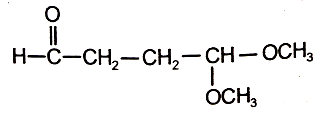
The Wolf Kishner's reduction cannot be applied to:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The major products in the reaction are
1. 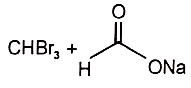 2.
2. 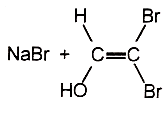
3. 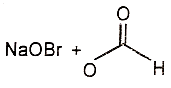 4.
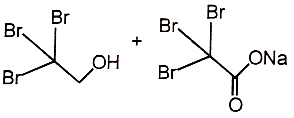
4. 
If =82 kJ/mol. Bond energy of NN. N=N, O=O and N=O bonds is 946, 418, 498 and 607 kJ/mol respectively. Then the resonance energy of will be
1.
2.
3.
4.
A compound (X) has the molecular formula . With and KOH. (X) gives (Y). Y responds to mustard on reaction, (V) upon treatment with evolves and gives alcohol (Z) which. gives iodoform test (X) is likely to be
1.
2.
3.
4.
For the reaction , the equivalent mass of is
1.
2.
3.
4. M
The entropy change in the isothermal reversible expansion of 2 moles of an ideal gas from 10 to 100 L at 300 K is
1.
2.
3.
4.
Given,
\(\begin{aligned}
& \mathrm{NO}(\mathrm{g})+\mathrm{O}_3(\mathrm{~g}) \rightarrow \mathrm{NO}_2(\mathrm{~g})+\mathrm{O}_2(\mathrm{~g}) \\
& \Delta \mathrm{H}=-198.9 \mathrm{~kJ} / \mathrm{mol} \\
& \mathrm{O}_3(\mathrm{~g}) \rightarrow 3 / 2 \mathrm{O}_2(\mathrm{~g}) ; \Delta \mathrm{H}=-142.3 \mathrm{~kJ} / \mathrm{mol} \\
& \mathrm{O}_2(\mathrm{~g}) \rightarrow 2 \mathrm{O}(\mathrm{g}) ; \Delta \mathrm{H}=+495.0 \mathrm{~kJ} / \mathrm{mol}
\end{aligned}\)
The enthalpy change (∆H) for the following reaction NO(g) + O(g) → NO2(g) is:
1. -304.1 kJ/mol
2. +304.1 kJ/mol
3. -403.1 kJ/mol
4. +403.1 kJ/mol
identify the product 'Z' in the following series of reactions
1. Hexanoic acid
2. -methyl caproicacid
3. Heptanoic acid
4. Heptane 1-7 diol
The Fisher presentation of D-glucose is given below:

The correct structure(s) of -L-glucopyranose is/(are):
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Photons of wavelength 4000 are used to break molecules. The percentage of energy converted
to the kinetic energy of atoms will be:
[Given: Bond dissociation energy of the molecule is 246.5 kJ/mol]
1. 12%
2. 8%
3. 26%
4. 17%
The second ionization potential of Be is 17.98 eV. If the outermost electron in is assumed to move in a spherical orbit with a central field of effective nuclear charge () consisting of nucleus and other electrons, by how many units of charge is the nucleus shielded by other electrons? [Energy of electron in the first orbit of H-atom = —13.6 eV]
1. 2.0 unit
2. 0.5 unit
3. 1.0 unit
4. 1.7 unit
Vulcanized rubber has
1. Thiol linkages
2. Thioether linkages
3. Sulphonic linkages
4. Alcohol linkages
The initial reaction in the formation of bakelite is an example of:
1. Aromatic electrophilic substitution
2. Aromatic nucleophilic substitution
3. Free radical reaction
4. Aldol reaction
The set of quantum numbers that can not be allotted to an electron in an atom is:
1. n = 3, l = 2, = +2, = -1/2
2. n = 2, l = 0, = +1, = +1/2
3. n = 1, l = 0, = 0, = +1/2
4. n = 4, l = 3, = 0, = -1/2
The geometry and the number of the unpaired electron(s) of \([MnBr_{4}]^{2-}\), respectively, are:
| 1. | Tetrahedral and 1 | 2. | Square planar and 1 |
| 3. | Tetrahedral and 5 | 4. | Square planar and 5 |
The geometry and magnetic property of , respectively, are
1. Tetrahedral, Paramagnetic
2. Tetrahedral, Diamagnetic
3. Square planar, Paramagnetic
4. Square planar, Diamagnetic
The IUPAC name of [Co(ONO)(NH3)5]Cl2 is:
1. Pentamminenitrocobalt(II)chloride
2. Pentamminenitrsoocobalt(III)chloride
3. Pentaamminenitrito-O-cobalt(III)chloride.
4. Pentammineoxo-nitrocobalt(III)chloride
1. NO3- acts as a monodentate ligand.
2. The Ce atom has a coordination number of 12.
3. The shape of the complex ion is an icosahedron.
4. The solution is used as an oxidizing agent.
A substance that gives a brick red flame and breaks down
on heating to give oxygen and a brown gas is:
1. Calcium carbonate
2. Magnesium carbonate
3. Calcium nitrate
4. None of the above
Identify the metal M, which has the following extraction principle.
1. Mg
2. Pb
3. Sn
4. Fe
Why is roasting important in the iron metallurgy process?
| 1. | FeO gets oxidized to Fe2O3 which does not combine with acidic SiO2 |
| 2. | SiO2 is an acidic oxide which can combine with metallic oxides i.e. FeO, and Fe2O3 |
| 3. | FeO when roasted get oxidized to more basic Fe2O3 |
| 4. | None of the above. |
The unpaired electron in Eu3+ will be the same in:
1. Yb2+
2. Ho3+
3. Sm2+
4. Gd3+
The correct statement is
1. Green vitriol and blue vitriol are isomorphus
2. KMnO4 and K2Cr2O7 are coloured due to d-d transitions
3. Cu2Cl2 and Ag2S are coloured
4. Upon strong heating paramagnetic gases are evolved by CuSO4 and AgNO3
The spin only magnetic moment of [ZCl4]2- is 3.87 BM where Z is
1. Mn
2. Ni
3. Co
4. Cu
The fusion of chromite ore (FeCr2O4) with Na2CO3 in air gives a yellow solution upon the addition of water. Subsequent treatment with H2SO4 produces an orange solution.
The yellow and orange colours, respectively, are due to the formation of:
1. Na2CrO4 and Na2Cr2O7
2. Cr(OH)3 and Na2Cr2O7
3. Cr2(CO3)3 and Fe2(SO4)3
4. Cr(OH)3 and Na2CrO4
Which of the following weighs less when weighed in magnetic field?
1. ScCl3
2. FeCl3
3. TiCl3
4. VCl3
For which of these electrodes will the reduction potential vary with pH?
l.
ll.
lll.
1. l only
2. ll only
3. l and ll only
4. l, ll and lll only
The number of stereoisomers obtained by bromination of trans-2-butene is:
1. 1
2. 2
3. 3
4. 4
How many dichloroisomers can be obtained from the following reaction?
1. 1
2. 2
3. 3
4. 4

The major product (X) in the above reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Which one of the following has maximum nucleophilicity?
1.
2.
3.
4.
By the action of concentrated hydrochloric acid on potassium chlorate we get this mixture of gases:
1.
2.
3.
4.
Which of the following molecules cannot show geometric isomerism?
1.
2.
3. HO - C = N - OH
4. 
Which salt is more hydrolysed ?
(Assume that Kb of all weak base is same)
1. NH4Cl
2. CuSO4
3. AlCl3
4. All are equally hydrolysed
The power generated in a circuit is given by \(P=i^2R,\) where \(i\) is current and \(R\) is resistance. The percentage errors in measuring \(i\) and \(R\) are \(0.3\) and \(0.5\) respectively. The maximum error in measuring power is
1. \(0.3\%\)
2. \(0.5\%\)
3. \(0.8\%\)
4. \(1.1\%\)
The position \(x\) of a particle moving along the \(x\)-axis varies with time \(t\) as \(x=20t-5t^2,\) where \(x\) is in meters and \(t\) is in seconds. The particle reverses its direction of motion at:
1. \(x=40~\text{m}\)
2. \(x=10~\text{m}\)
3. \(x=20~\text{m}\)
4. \(x=30~\text{m}\)
If represent radial and tangential acceleration, then the motion of particle will be uniformly circular for-
1. = 0, = 0
2. = 0, 0
3. 0, = 0
4. 0, 0
Path of a projectile with respect to another projectile as long as both remain in the air is
1. Circular
2. Parabolic
3. Straight
4. Hyperbolic
A swimmer swims a distance d upstream in \(4\) s and swims an equal distance downstream in \(2\) s. The ratio of swimmer's speed in still water to the speed of river water will be:
| 1. | \(\frac{6}{5} \) | 2. | \(\frac{3}{1} \) |
| 3. | \(\frac{5}{3} \) | 4. | \(\frac{4}{3}\) |
A man of mass \(60\) kg is standing on the ground and holding a string passing over a system of ideal pulleys. A mass of \(10\) kg is hanging over a light pulley such that the system is in equilibrium. The force exerted by the ground on the man is: (\(g=\) acceleration due to gravity)

1. \(20g\)
2. \(45g\)
3. \(40g\)
4. \(60g\)
A small coin is kept at a distance \(r\) from the centre of a gramophone disc rotating at an angular speed \(\omega\). The minimum coefficient of friction for which a coin will not slip is:
1. \(\dfrac{rω^{2}}{g}\)
2. \(\dfrac{g}{r\omega^2}\)
3. \(\dfrac{r^2ω^{2}}{g}\)
4. \(\dfrac{rω}{g}\)
The energy required to accelerate a car from rest to 30 m/s is E. The energy required to accelerate the car from 30 m/s to 60 m/s is
1. E
2. 2 E
3. 3 E
4. 4 E
A particle has been thrown up with double the escape speed from the surface of the earth. Its speed when it reaches interstellar space far away from the Earth's gravity is
1.
2.
3.
4. Zero
If \(E\) is the energy stored per unit volume in a wire having \(Y\) as Young's modulus of the material, then the stress applied is:
1. \(\sqrt{2EY}\)
2. \(2\sqrt{EY}\)
3. \(\frac{1}{2}\sqrt{EY}\)
4. \(\frac{3}{2}\sqrt{EY}\)
Water is filled in a container up to height h. A narrow hole is made at the bottom. The velocity with which water will come out through-hole is proportional to-
1.
2.
3.
4.
An ideal fluid is flowing in a steady-state from section \(A\) to \(B\) through a pipe in a vertical plane as shown. Select the incorrect statement.
| 1. | Total energy per unit volume is the same at both sections A and B. |
| 2. | The incoming flow rate at A is equal to the outgoing flow rate at B. |
| 3. | Loss in kinetic energy of the fluid is equal to gain in potential energy from section A to section B. |
| 4. | The flow of fluid from A to B is laminar flow. |
A mass of 30 g is attached with two springs having spring constant 100 N/m and 200 N/m and other ends of springs are attached to rigid walls as shown in the given figure. The angular frequency of oscillation will be
1.
2.
3. 100 rad/s
4. 200 rad/s
The motion of the particle is started at t = 0 and the equation of motion is given by x = 8 sin, where x is in cm and t is in seconds. When will the particle come to rest for the first time?
1.
2.
3.
4.
A policeman buzzes a whistle of frequency 420 Hz. A car with an audio recorder is moving towards the policeman with a speed of 72 km/h, recording a frequency of . If, after crossing, it records the frequency of , then - is: (Velocity of sound is 300 m/s)
1. Hz
2. 20 Hz
3. 28 Hz
4. 56 Hz
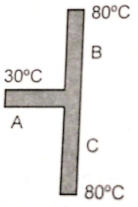
Three rods A, B, and C of the same material have the same cross-sectional area but have a length in the ratio 1:2:2 respectively. The left and right ends are at 30°C and 80°C. The temperature of the junction of the rods will be
1. 20°C
2. 55°C
3. 45°C
4. 10°C
The wavelength corresponding to maximum emitted energy is 4000 at 4000 K. If the temperature is decreased by 1000 K, then emitted wavelength becomes
1. 3000
2. 5333
3. 6000
4. 5000
In the \(P\text-V\) graph shown for an ideal diatomic gas, the change in the internal energy is:

| 1. | \(\frac{3}{2}P(V_2-V_1)\) | 2. | \(\frac{5}{2}P(V_2-V_1)\) |
| 3. | \(\frac{3}{2}P(V_1-V_2)\) | 4. | \(\frac{7}{2}P(V_1-V_2)\) |
Two Carnot engines x and y are working between the same source temperature \(T_1\) and the same sink temperature \(T_2\). If the temperature of the source in Carnot engine x is increased by \(\Delta T\), and in the Carnot engine y, the temperature of the sink is increased by\(\Delta T\), then the efficiency of x and y becomes \(\eta_\mathrm x\) and\(\eta_\mathrm y\). Then:
| 1. | \(\eta_{\mathrm{x}}=\eta_{\mathrm{y}}\) |
| 2. | \(\eta_{\mathrm{x}}<\eta_{\mathrm{y}}\) |
| 3. | \(\eta_{\mathrm{x}}>\eta_{\mathrm{y}}\) |
| 4. | The relation between \(\eta_{\mathrm{x}}\) and \(\eta_{\mathrm{y}}\) depends on the nature of the working substance |
A solid sphere is rolling without slipping such that the velocity of its centre of mass is v. Ratio of speeds of horizontal extreme points A and B is
1. 1:1
2. :1
3. 2:1
4. 1:
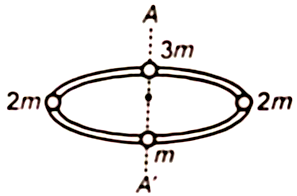
Four masses are joined to a light circular frame in the figure. The radius of gyration of this system about an axis passing through the center of the circular frame and perpendicular to its plane would be (where a is the radius of the circle)
1.
2.
3. a
4. 2a
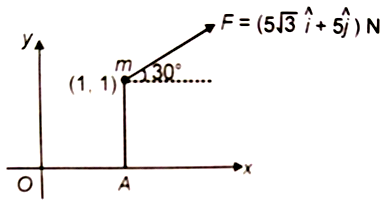
The torque of force F about origin O is
1.
2.
3.
4.
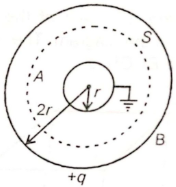
The conducting shells A and B are arranged as shown below. If the charge on the shell B is q, then electric flux linked with the spherical Gaussian surface S is-
1.
2.
3.
4.
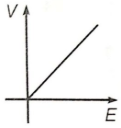
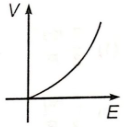
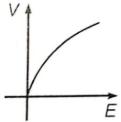
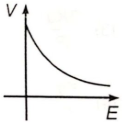
If E and V are electric field and electric potential respectively due to a point charge, then which of the following graphs best represent their variation?
1. 
2. 
3. 
4. 
The equivalent capacitance between points \(a\) and \(b\) in the network shown below is:
1. \(5~\text{C}\)
2. \(4~\text{C}\)
3. \(3~\text{C}\)
4. \(2~\text{C}\)
The maximum kinetic energy of an -particle coming out of a cyclotron accelerator is 20 MeV. The maximum kinetic energy of a proton that can be obtained from this accelerator is
1. 20 MeV
2. 40 MeV
3. 10 MeV
4. 5 MeV
If the instantaneous charge in the capacitor is 400 and current through the circuit is decreasing at the rate A/s, then the potential difference is equal to
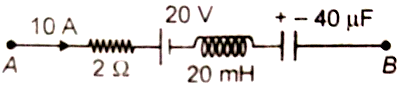
1. 30 V
2. Zero
3. 10 V
4. 70 V
A galvanometer coil has a resistance of 15 and it shows full deflection when 4 mA current flows through it. The value of shunt resistance needed to convert it into an ammeter which can read any current between 0 to 6 A is nearly equal to
1. 1
2. 0.1
3. 0.01
4. 0.001
| 1. | \(\frac{4}{A - 4}\) | 2. | \(\frac{A - 4}{4}\) |
| 3. | \(1\) | 4. | \(\frac{A + 4}{4}\) |
| 1. | \(4x+4y\) | 2. | \(4x-4y\) |
| 3. | \(4y-4x\) | 4. | \(y-x\) |
Let be the maximum frequency of Lyman series, be the frequency of the first line of Lyman series and be the frequency of the series limit of the Balmer series. Then which of the following is correct?
1.
2.
3.
4.
Light waves of intensities I and 9I interfere to produce a fringe pattern on a screen. The phase difference between the waves at a point P is and at other point Q. Then the ratio of intensities at P and Q is
1. 8:5
2. 1:4
3. 5:8
4. 9:1
In a YDSE, let X and Y be the upper and lower slits. A thin film of thickness t and refractive index is placed in front of X. Let = fringe width. The central maximum will shift
1. Downward
2. Upward
3. By
4. Both 2 & 3
The resistance between terminal A and B is
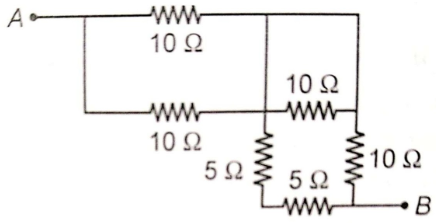
1. 5 Ω
2. 15Ω
3. 10 Ω
4. 20 Ω
The potentiometer wire PQ is 100 cm long and its resistance is 2r where r is the internal resistance of the battery. The balancing length PC is equal to
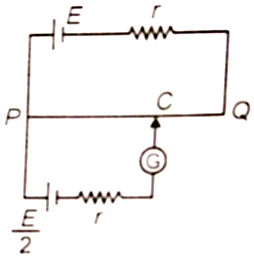
1. 25 cm
2. 75 cm
3. 50 cm
4. 40 cm
In the given graph of the photoelectric experiment, the maximum energy of the emitted photoelectron will be:
1. \(2~\text{eV}\)
2. \(0\)
3. \(4~\text{eV}\)
4. \(4~\text{J}\)
The de-Broglie wavelength associated with a proton accelerated from rest by 10 kV is . The de-Broglie wavelength associated with a 10 keV neutron is-
1.
2.
3. 2
4.
The circuit shown below is an electrical analog for which of the following logic gates?
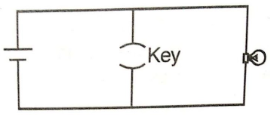
1. AND gate
2. OR gate
3. NOT gate
4. NOR gate
What is the equivalent resistance across the terminals of the battery, if the diodes are ideal?
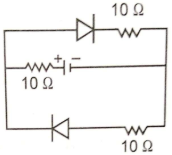
1. 10Ω
2. 20 Ω
3. 15 Ω
4.
A transistor circuit in common emitter configuration is shown. The current gain is 100. If , then is given by
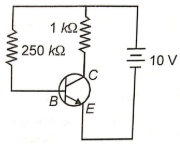
(1) 6 V
(2) 5 V
(3) 4 V
(4) 3 V
When an object is placed at 10 cm and 30 cm from a convex lens, images obtained are of the same magnitude of magnification. The focal length of the lens maybe
1. 10 cm
2. 15 cm
3. 20 cm
4. 25 cm
A liquid of refractive index is placed between two identical planoconvex lens touching each other at their spherical surface of radius R. If the refractive index of a planoconvex lens is 1.50, then the combination behaves as-
1. Convergent with power
2. Convergent with power
3. Divergent with power
4. Divergent with power
The minimum magnifying power of an astronomical telescope is 40. If the length of the telescope is 205 cm, then the focal length of its field lens is
1. 5 cm
2. 200 cm
3. 40 cm
4. 140 cm
When a magnetic material is subjected to a very small magnetizing force H, the intensity of magnetization is proportional to
1.
2.
3. H
4.