Which of the following statements is not true
1. DNA being more stable is preferred for storage of genetic information
2. DNA is the preferred biomolecule for transmission of genetic information
3. The protein synthesizing machinery has evolved around RNA
4. Both RNA and DNA can function as genetic material
In the given diagram of the nucleosome, the region marked as ‘x’ is rich in.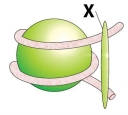
1. Glutamine.
2. Lysine.
3. Tryptophan.
4. Tyrosine.
Production of a human protein in bacteria by genetic engineering is possible because
1. Bacterial cell can carry out the RNA splicing.
2. The mechanism of gene regulation is identical in humans and bacteria.
3. The human chromosome can replicate in bacterium cell.
4. The genetic code is universal
Which of the following is not a hereditary disease?
1. Haemophilia.
2. Cretinism.
3. Cystic fibrosis.
4. Thalassemia.
Which of the following pairs of features is good example of polygenic inheritance.
1. Human height and skin colour.
2. ABO blood group in humans and flower colour in Mirabilis jalapa.
3. Hair pigmentation and tongue rolling in humans.
4. Human eye colour and sickle cell anaemia.
In replica-plate experiment some bacteria able to grow in penicillin containing medium due to
1. Natural selection
2. Reproductive isolation
3. Genetic drift
4. Crossing over
Which of the given below sexually transmitted disease in completely curable?
1. Trichomoniasis
2. Genital herpes
3. Hepatitis-B
4. HIV infection
Sickle cell anemia is an example of all except:-
1. Pleiotropy
2. Multiple allelism
3. Lethality
4. Gene mutation
Which of the following statement is correct?
1. The RNA is subjected to a process called splicing where the exons are removed
2. In capping an unusual nucleotide is added to the 3 ends of the RNA
3. Splicing represents the dominance of the DNA world
4. In trailing, adenylate residues are added in a template-independent manner
By digestion and pulverization detritus get fragmented. This fragmentation step of decomposition helps in
1. Increasing porosity of detritus
2. The increased surface area of detritus
3. The increasing rate of sedimentation
4. All the above
Which of the following is the correct sequence of events of interphase:-
| a. | cells metabolically active |
| b. | Duplication of the DNA amount, but chromosome number remains constant |
| c. | M phased |
| d. | Synthesis of protein [Tubulin] |
| 1. | a → b → c → d |
| 2. | a → d → b → c |
| 3. | a → b → d → c |
| 4. | a → c → d → b |
Which one plant factor affects the photosynthesis process?
| 1. | CO2 Concentration |
| 2. | Light |
| 3. | Availability of water in the soil |
| 4. | Amount of chlorophyll |
'Founder effect' is related to -
1. Gene recombination and Natural selection
2. Genetic drift and origin of new species
3. isolation and Natural selection
4. Hybridization and origin of new species
Which contraceptive method is most widely accepted in India :
1. IUDs
2. Diaphragm
3. Vasectomy
4. Saheli
The fate of pyruvic acid during Aerobic respiration is
| 1. | Lactic acid fermentation |
| 2. | Alcoholic fermentation |
| 3. | Oxidative decarboxylation |
| 4. | Photophosphorylation |
Marsupial mole and kangaroo are examples of
1. Divergent evolution
2. Convergent evolution
3. Adaptive Radiation
4. Both 1 and 3
In nematode resistance by RNA interference, some specific genes were introduced which form dsRNA. These were introduced in
1. E.coli
2. Host plant
3. Agrobacterium
4. Nematode
Alien species invasion is one of the causes of Biodiversity loss. Introduction of Nile perch in victoria lake leads to the extinction of more than 200 species of-
1. Cichlid fish
2. Gambusia
3. Salmon fish
4. Catfish
In erythroblastosis, the fetal fetus dies due to:-
1. Elevated bilirubin level
2. Neonatal jaundice
3. Neonatal anemia
4. All of the above
Cerebroside present in the white matter of brain contain:-
1. Sphingosine + 1 fatty acid + choline.
2. Sphingosine +2 fatty acid + choline.
3. Sphingosine + 1 fatty acid + Galactose.
4. Sphingosine + Glycerol + choline
Vernalization can often be replaced by:-
1. Auxin
2. Cytokinins
3. Gibberellins
4. Ethylene
The first living organism was:-
1. Aerobic-heterotrophic
2. Aerobic-Autotrophic
3. Anaerobic-Heterotrophic
4. Anaerobic-Autrophic
The fruit developing from a single ovary is said to be:-
1. Composite fruit
2. Aggregate fruit
3. Simple fruit
4. Achenial fruit
The C-peptide is:-
1. Not present in proinsulin
2. Present in mature insulin
3. Remove during maturation of insulin
4. Also present in artificial insulin
Which of following stage of plasmodium is formed in mosquito:-
1. Sporozoite
2. Gametes
3. Both (1) and (2)
4. Gametocytes
Exploring molecular, genetic and species-level diversity for products of economic importance is known as -
1. Biomining
2. Biomedicining
3. Biofortification
4. Bioprospecting
Myosin filaments are:-
1. thin filament
2. found only in I band
3. absent from H zone
4. attached to M line
Which of the following statement are correct:-
| 1. | Lysosome contain a hydrolytic enzyme which acts in pH= 5 |
| 2. | Glycosylation complete in the Golgi body. |
| 3. | Mitochondria are stained with vital stain Janus green B. |
| 4. | All of the above. |
How many enzymes in the list given below found in pancreatic juice?
Enterokinase, Chymotrypsin, Aminopeptidase, Pepsin, Lactase, Rennin, Carboxypeptidase, Nuclease, Nucleotidase & Elastase:-
1. Six
2. Three
3. Five
4. Four
In Galapagos island, Darwin found different varieties of finches, originated from a common ancestor. The original variety of these finches was:-
1. Insectivorous
2. Vegetarian
3. Seed-eating
4. Cactus eating
'Sacred groves are also one of the important means of Biodiversity conservation. In respect of this find out the odd one-
| 1. | Khasi & Jaintia - Meghalaya |
| 2. | Aravalli hills - Rajasthan |
| 3. | Sarguja, Chanda, Bastar - Mizoram |
| 4. | Western Ghat - Maharashtra |
Match the following columns of groups and examples?
| Group | Example | ||
| (i) | Sphinopsida | (a) | Adiantum |
| (ii) | Bryopsida | (b) | Polysiphonia |
| (iii) | Pteropsida | (c) | Equisetum |
| (iv) | Liver Wort | (d) | Funaria |
| (v) | Rhodophyceae | (e) | Marchantia |
| 1. | i-a, ii-b, iii-c, iv-d, v-e |
| 2. | i-c, ii-a, iii-e, iv-b, v-d |
| 3. | i-c, ii-d, iii-a, iv-e, v-b |
| 4. | i-d, ii-a, iii-c, iv-e, v-b |
Grey seed colour(G) in pea is dominant to white seed colour(g). In the following crosses, the indicated parent with known phenotype but indicated parent with known phenotype, but unknown genotype, produced the progeny listed:
| Parent | Progeny | Female parent | ||
|
Female male |
Gray | White | Genotype | |
| (i) | Gray white | 81 | 82 | A |
| (ii) | Gray gray | 118 | 39 | B |
| (iii) | Gray white | 74 | 0 | C |
| (iv) | Gray gray | 90 | 0 | D |
Based on the segregation data, the possible genotype of each female parent -
1. A-Gg; B-Gg; C-GG; D-GG or Gg
2. A-GG; B-GG; C-GG; D-Gg
3. A-Gg; B-Gg; C-Gg; D-Gg
4. A-gg; B-gg; C-gg; D-Gg
Following structure represent:-
| 1. | Tertiary structure, of protein hollow woolen ball-like. |
| 2. | The sequence of amino acid, peptide bond N, C terminal. |
| 3. | Secondary structure, helical, structure |
| 4. | All of the above |
Which of the statement is not true for given structures?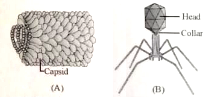
1. They both are made up of nucleoprotein
2. They both infect plants and bacteria
3. They are obligate intracellular parasites
4. A and B have RNA & DNA as genetics material respectively
Study the given table regarding vertebrates and identified A, B, C and D
| Taxon A |
Character Fish with circular mouth |
Example Hagfish |
| Osteichthyes | Gill is covered by operculum | B |
| Chondrichthyes | C | Scoliodon |
| D | 3 chambered heart | Lepidosiren |
| A | B | C | D |
| 1. Placodermi | Betta | Placoid Scales | Dipnoi |
| 2. Cyclostomata | Clarias | Placoid Scales | Dipnoi |
| 3. Cyclostomata | Pristis | Jawless | Bony Fish |
| 4. Osteichthyes | Pristis | Jawless | Bony Fish |
Bryophytes are:-
| 1. | Always homosporic and exhibit exospheric development of spores |
| 2. | Mostly sporophytic plants |
| 3. | Most successful terrestrial plants |
| 4. | Gametophytic with branched multicellular roots |
How many of the following statements are correct?
i. Axillary buds are present in axil of leaflets
ii. In salvia filaments of stamens are of the same length
iii. The apocarpous condition can be seen in lotus and rose
iv. Endospermous seeds can be seen in Solanaceae
v. Sterile stamens are called staminodes
1. Three
2. Four
3. Two
4. One
Select the incorrect statement regarding facilitated diffusion:-
1. It is a very specific process
2. It is a passive process
3. It helps the hydrophilic substances to be transported across the membrane
4. It is faster than active process
Which is not correctly matched?
1. Malpighian tubules - Cockroaches, Mosquito
2. Antennal glands - Planaria, Crab
3. Nephridia - Earthworm
4. Protonephridia - Amphioxus
The gametophytic phase and sporophytic phases are independent in:-
1. Pinus
2. Polytrichum
3. Dryopteris
4. Chara
Bacterial colonies that have a foreign DNA fragment inserted into the plasmid will appear white because-
1. X-gal can be cleaved by -galactosidase
2. Lac-Z gene shows insertional inactivation due to insertion of foreign DNA
3. Lac-Z gene is active
4. X-gal can be cleaved by permease
LSD is the most powerful psychological drug obtained from
1. Papaver somniferum
2. Claviceps purpurea
3. Cannabis sativa
4. Erythroxylum coca
Match column-I with column-II and select the correct option from the codes given below:-
| Column - I | Column - II | ||
| A | Parietal | i |
Dianthus |
| B | Axile | ii | Helianthus |
| C | Basal | iii | Argemone |
| D | Free central | iv | Hibiscus |
1. A-iii, B-i, C-ii, D-iv
2. A-iii, B-iv, C-ii, D-i
3. A-ii, B-iv, C-iii, D-i
4. A-iv, B-iii, C-i, D-ii
Which one is the supportive framework of epithelium?
1. Tendon
2. Ligament
3. Areolar tissue
4. Nervous tissue and blood
Which of the following is characteristic of prophase I of meiosis I:-
| 1. | Synapsis in zygotene, genetically different chromatid are seen in pachytene |
| 2. | Exchange of genes in leptotene, genetically similar chromatids are seen in zygotene. |
| 3. | Chiasmata are present in metaphase |
| 4. | Splitting of centromere in anaphase-I |
Phloem sap is mainly:-
1. Water and osmotically inactive disaccharides only
2. Water and PGRs only
3. Water and osmotically active disaccharides
4. Minerals and PGRs only
Which of the following is not the function of the large intestine?
1. Absorption of some water, minerals and certain drugs
2. Nutrient absorption
3. Secretion of mucus to lubricate feces
4. The temporary storage of feces in rectum
When both ovaries of human females removed then which hormone will decrease in blood?
1. Oxytocin
2. Estrogen
3. Prolactin
4. Gonadotropin hormone
Which of the following is wrongly matched in the given table?
| i | Golden rice | Transgenic rice | Vitamin A enriched |
| ii | Flavr savr | Transgenic tobacco | Delayed ripening |
| iii | Bt-cotton | Transgenic cotton | Bacterial and viral resistance |
| iv | Rosie | Transgenic cow | Milk contained human alpha lactalbumin |
1. i and ii
2. ii, iii, and iv
3. Only ii and iii
4. Only iii
In rocky intertidal communities removal of which of the following predator became the cause of the destruction of 10 species of invertebrates
| 1. | Monarch butterfly |
| 2. | Starfish pisaster |
| 3. | Paramecium Aurelia |
| 4. | Abingdon tortoise |
The holocrine gland is:-
1. Sweat gland
2. Sebaceous gland
3. Mammary gland
4. Mucus gland
Which of the following are the character of Liliaceae family?
i. Monocotyledonous plants
ii. Umbellate clusters
iii. Axile placentation
iv. 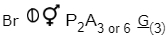
v. Ovary inferior
1. i, ii, & iii
2. i, ii, iii & iv
3. iv & v
4. iii & v
Match the items given in column-I with those given in column-II and select the correct option:-
| Column - I | Column - II | ||
| A | I |
Nitrate reductase |
|
| B | II | RuBisCO, PEPCase | |
| C | Mo | III | Alcohol dehydrogenase |
1. A-II, B-III, C-I
2. A-III, B-II, C-I
3. A-I, B-III, C-II
4. A-II, B-I, C-III
Mark the incorrectly matched pair regarding the structure with its location and functions:-
| 1. | Otolith organs | Located in the utriculus and sacculus, and is related to static equilibrium |
| 2. | Organ of Corti | Located on the basilar membrane and contains hair cells that act as auditory receptors |
| 3. | Visual cortex area | Located on the temporal lobe where sight impulses are analyzed and the retinal image is recognised. |
| 4. | Ear ossicles | Located in the middle ear cavity and increase the efficiency of transmission of sound waves to the inner ear. |
Find the correct sequence of the given phase of the menstrual cycle
a. Estrogenic phase
b. Secretory phase
c. Ovulation
d. Bleeding phase
1. d a c b
2. b a c d
3. d c a b
4. b a d c
Find an incorrect statement with respect to results of HGP:-
1. Dystrophin protein gene is the largest gene of the human genome
2. Chromosome Y has minimum of 231 genes
3. SNPs have been identified at about 1.4 million locations
4. The functions are unknown for over 90% of discovered genes.
Regarding competition, find out the wrong statement
1. Unrelated species could compete for the same resource
2. In competition, the fitness of one species is lowered in presence of other species
3. The Abingdon tortoise became extinct due to competitor starfish
4. Balanus led to exclusion of Chathamalus from rocky coasts of Scotland
Interspecific hybridization is the mating of
1. superior males and females of different breeds.
2. two different related species
3. animals within the same breed without having common ancestors
4. more closely related individuals within the same breed for 4-6 generations
Annelids do not represent:-
1. Circumpharyngeal nerve ring
2. Longitudinal muscles in the gut
3. Metameric segmentation
4. Mesodermal pouches in body cavity
Ground Tissue does not include:-
| (i) | Epidermis |
| (ii) | Vascular bundles |
| (iii) | Parenchyma |
| (iv) | Collenchyma |
| (v) | Sclerenchyma |
1. (i) & (ii)
2. (iv) & (v)
3. (i) & (iii)
4. (ii) & (v)
Morphology of chromosomes and shape of chromatid is best observed at:-
| 1. | Metaphase, Anaphase | 2. | Prophase, Metaphase |
| 3. | Anaphase, Telophase | 4. | Only telophase |
Select the correctly match pair:-
| 1. | Zinc - Helps to maintain the ribosome structure |
| 2. | Magnesium - Needed during the formation of mitotic spindles |
| 3. | Calcium - plays a role in opening and closing stomata |
| 4. | Manganese - Needed for splitting of water during photosynthesis |
The hormone that reduces the destruction of bones also enhances the deposition of in bones thus making them solid and strong. This hormone is:-
1. Collips hormone
2. Thyrocalcitonin
3. Thyroxine
4. Vasopressin
The part of the fallopian tube closest to the ovary is:-
| 1. | Ampulla | 2. | Infundibulum |
| 3. | Cervix | 4. | Isthmus |
Which of the following is not true for generation of dihybrid cross -
1. 9-phenotypic categories, 4 genotypic categories
2. 9-genotypic categories, 16 zygotic combination
3. 62.5% parental combination, 37.5% new combination
4. 4-types male gametes, 4 type female gemate
In meselson and Stahl's experiment, the E.coli cell with heavy DNA is continuously grown for three generations in containing medium. The ratio of containing DNA in the third generation would be-
1. 2 : 2 : 4
2. 0 : 1 : 3
3. 0 : 1 : 7
4. 7 : 1 : 0
The components of the ecosystem are seen to function as a unit when we consider which of the following aspect
1. Productivity and decomposition
2. Decomposition and energy flow
3. Productivity and energy flow
4. Productivity, decomposition, energy flow and nutrient cycling
How many of the following statements are not true for kingdom Monera ?
(i) Most of the members are prokaryotic
(ii) Some of the monerans can perform photosynthesis
(iii) All the members have cell wall of peptidoglycan
(iv) All the members of monera are devoid of true nucleus
(v) All the members are pathogenic
1. One
2. Two
3. Three
4. Four
Pseudocoelom is not found in:-
1. Enterobius
2. Ancylostoma
3. Echinococcus
4. Wuchereria
Identify the labelled parts in the given diagram and select the right option :-
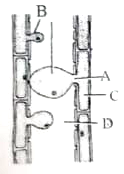
| A | B | C | D |
1. | Xylem parenchyma | Xylem vessel | Tyloses | Pit |
2. | Tyloses | Pit | Xylem parenchyma | Xylem vessel |
3. | Pit | Tyloses | Xylem parenchyma | Xylem vessel |
4. | Xylem vessel | Xylem parenchyma |
| Tyloses |
Which one option is correct for Dark reactions?
| 1. | Occurs in dark |
| 2. | Not depend on light |
| 3. | Dependent on products of light reaction |
| 4. | Occurs in lumen in thylakoid |
Which one of the following statements is incorrect with respect to the transport of carbon dioxide in blood?
1. About 20-25 percent of carbon dioxide is carried by haemoglobin in the form of carbaminohaemoglobin.
2. 70 percent of is carried in a dissolved state through plasma.
3. As the blood picks up carbon dioxide from tissue, accumulates inside RBCs of which some move out into the blood plasma and in exchange chloride ions move from plasma into RBCs.
4. The greatest percentage of carbon dioxide is transported in blood plasma as bicarbonate ions.
The most important primary factor in determining the percent saturation of haemoglobin with oxygen is
1. Partial pressure of oxygen
2. Acidity
3. Partial pressure of carbon dioxide
4. Temperature
Analysis of urine helps in clinical diagnosis of many metabolic disorder as well as malfunctioning of kidney. Given below are some disorders
| a. | Glycosuria |
| b. | Ketonuria |
| c. | Haematuria |
| d. | Proteinuria |
Choose the correct option which indicates disorders of diabetes mellitus if not controlled?
1. a only
2. a & b only
3. a,b & c only
4. a,b,c & d
Select the correct statement with respect to gemmae
| a. | These are specialised reproductive structures in members of thallophytes and bryophytes |
| b. | These are green, unicellular & asexual buds |
| c. | These are formed in small receptacles located on thalli |
| d. | Single gemma forms two new individuals after germination |
| e. | These are chlorophyllous and multicellular structures |
| f. | They germinate and make monoecious thallus of Marchantia |
2. b,c,d and f
3. c,d and e
4. b,c and f
Number of chromosomes in gametes of maize, apple, rice and potato are respectively
1. 10,17,12,24
2. 20,34,24,48
3. 10,12,24,17
4. 16,17,12,24
Interflowering period in a perennial plant
1. Represents juvenile phase
2. Is used for building up resources for next flowering
3. Can be observed in both monocarpic and polycarpic plants
4. Can occur twice in one year in henbane
Read the following statements carefully and select the correct
a. Tapetum cells possess dense cytoplasm and generally have more than one nucleus
b. Sporopollenin is absent in the areas called germ pore
c. Generative cell is small, spindle shaped with a irregularly shaped nuclei
d. Spindle of first mitosis in pollen is asymmetric
1. a,b,c & d
2. a,b & c
3. a,c & d
4. a,b & d
Choose the correct sequence of greenhouse gases with respect to their relative contribution to global warming in increasing order
1.
2.
3.
4.
'Ozone depletion' means thinning of ozone layer during spring season and
1. This will allow UV-A to enter stratosphere which will cause snow blindness
2. This is caused by as major pollutant
3. Ozone is released during this as or ClO
4. First large hole was discovered over north pole
Match the following with respect to pollution
Column I Column II
(a) Incineration (i) Soil erosion
(b) Green Muffler (ii) Third poison
(c) Ground water pollution (iii) Solid waste
(d) Negative pollution (iv) Noise pollution
1. a(iii), b(iv), c(ii), d(i)
2. a(iii), b(iv), c(i), d(ii)
3. a(iv), b(iii), c(ii), d(i)
4. a(ii), b(iv), c(iii), d(i)
Match the following micro-organisms to the related products
| Column-I | Column-II | ||
| a. | Swiss cheese | (i) | Monascus |
| b. | Statin | (ii) | Propionibacterium |
| c. | Butyric acid | (iii) | Trichoderma |
| d. | Cyclosporin A | (iv) | Clostridium |
1. a(ii), b(i), c(iv), d(iii)
2. a(ii), b(i), c(iii), d(iv)
3. a(ii), b(iv), c(i), d(iii)
4. a(iii), b(i), c(ii), d(iv)
Statins produced by the yeast Monascus purpureus have have been commercialised as
1. Immunosuppressive agent
2. Blood-cholesterol lowering agent
3. Clot buster
4. Agents to increase immunity
Which of the following statement is correct ?
| 1. | Alcoholic fermentation by yeast and conversion of alcohol to acetic acid both are aerobic processes |
| 2. | Alcoholic fermentation is aerobic and conversion of alcohol to acetic acid is anaerobic one |
| 3. | Alcoholic fermentation by yeast is anaerobic and conversion of alcohol to acetic acid is aerobic one |
| 4. | Alcoholic fermentation by yeast and conversion of alcohol to acetic acid both are anaerobic processes |
Which of the following is correct option with respect to step labelled as C on the given graph ?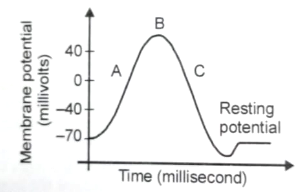
1. channels are opened and leak out to maintain a resting potential
2. channels open, and flows out causing rapid repolarisation
3. continues to flow out, producing a hyperpolarisation of membrane
4. Negatively charged protein molecule inside neuron responsible for this
During micturition , the contraction of detrusor muscles of urinary bladder is under the control of
1. Sympathetic nerve
2. Parasympathetic nerve
3. Pudendal nerve upon stimulation
4. Pudendal nerve upon inhibition
Given below is a diagrammatic representation of ribs and ribcage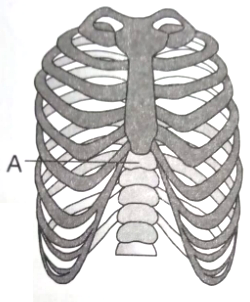
Identify the structure labelled as 'A' and choose the correct option with respect to its number in human body
1. 7 in number (cervical vertebrae)
2. 12 in number (lumbar vertebrae)
3. 5 in number (sacral vertebrae)
4. 12 in number (thoracic vertebrae)
The part of gut of the cockroach in which armarium and stomodeal valves are present in
1. Crop
2. Hepatic caeca
3. Gizzard
4. Ileum
Correct sequence of hormones increasing in their levels from the beginning of menstruation is
1. FSH, estrogen, LH, progesterone
2. Estrogen, LH, FSH, progesterone
3. FSH, progesterone, estrogen, LH
4. Estrogen, progesterone, FSH, LH
The latent heat of vaporisation of water is 540 cal at . Kb for water is
1.
2.
3.
4.
Which of the following complex species is not expected to exhibit optical isomerism ?
1.
2.
3.
4.
Which one of the following molecules is expected to exhibit diamagnetic behaviour
a.
b.
c.
d.
1. a,b
2. a,c
3.b,c,d
4.a,b,d
A solution of ( - ) - 1 - chloro - 1 - phenylethane in toluene racemizes slowly in the presence of a small amount of , due to the formation of:
1. carbanion
2. carbene
3. carbocation
4. free radical
A piston filled with 0.04 mol of an ideal gas expands reversibly from 50.0 mL to 375 mL at a constant temperature of 37.0ºC. As it does so, it absorbs 208 J of heat. The values of q and w for the process will be-
(R = 8.314 J/mol K) (ln 7.5 = 2.01)
| 1. | q = +208 J, w = -208 J | 2. | q = -208 J, w = -208 J |
| 3. | q = -208 J, w = + 208 J | 4. | q = +208 J, w = + 208 J |
The molarity of a solution obtained by mixing 750 mL of 0.5(M) HCl with 250 mL of 2(M)HCl will be :
1. 0.875 M
2. 1.00 M
3. 1.75 M
4. 0.975 M
Arrange the following compounds in order of decreasing acidity :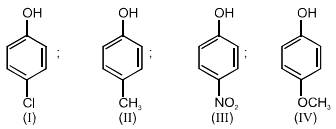
1. II > IV > I > III
2. I > II > III > IV
3. III > I > II > IV
4. IV > III > I > II
The amount (in mol) of bromoform (CHBr3) produced when 1.0 mol of acetone reacts completely with 1.0 mol of bromine in the presence of aqueous NaOH is
1.
2.
3. 1
4. 2
For gaseous state, if most probable speed is denoted by C*, average speed by and mean square speed by C, then for a large number of molecules the ratios of these speeds are :
1. C* : : C = 1.225 : 1.128 : 1
2. C* : : C = 1.128 : 1.225 : 1
3. C* : : C = 1 : 1.128 : 1.225
4. C* : : C = 1 : 1.225 : 1.128
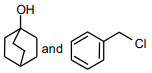
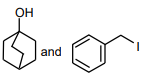
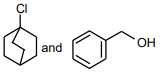
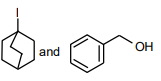
The following compound 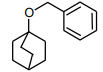
can readily be prepared by Williamson ether synthesis by reaction between
1. 
2. 
3. 
4. 
When the temperature of a reaction increases from 300 K to 310 K, the rate of the reaction doubles. What is the activation energy for this reaction?
\((R=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1} \text { and } \log 2=0.301)\)
| 1. | \(53.6 \mathrm{~kJ} \mathrm{~mol}^{-1} \) | 2. | \(68.6 \mathrm{~kJ} \mathrm{~mol}^{-1} \) |
| 3. | \(59.5 \mathrm{~kJ} \mathrm{~mol}^{-1} \) | 4. | \(70.5 \mathrm{~kJ} \mathrm{~mol}^{-1}\) |
A compound with molecular mass 180 is acylated with to get a compound with molecular mass 390. The number of amino groups presents per molecule of the former compound is :
1. 2
2. 5
3. 4
4. 6
The higher stabilities of tert-butyl cation over isopropyl cation, and trans-2-butene over propene, respectively, are due to orbital interactions involving.
1.
2.
3.
4.
Benzaldehyde can be converted to benzyl alcohol in concentrated aqueous NaOH solution using
1. acetone
2. acetaldehyde
3. formic acid
4. formaldehyde
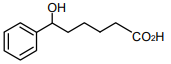
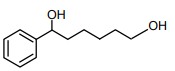
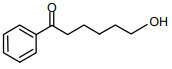
The major product of the following reaction is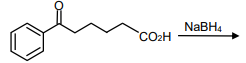
1. 
2. 
3. 
4. 
Which of the following statements is false?
| 1. | V2+ < Cr2+ < Mn2+ < Fe2+ : Paramagnetic behaviour |
| 2. | Ni2+ < Co2+ < Fe2+ < Mn2+ : Ionic size |
| 3. | Co3+ < Fe3+ < Cr3+ < Sc3+ : Stability in aqueous solution |
| 4. | Sc < Ti < Cr < Mn : Number of oxidation states |
Among the following species, the H–X–H angle (X=B,N or P) follows the order
1.
2.
3.
4.
The order of stability of the following carbocations :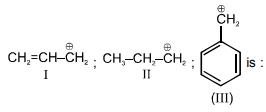
1. III > II > I
2. II > III > I
3. I > II > III
4. III > I>II
The ionic radii of Na+, F–, O2–, N3– follow the order
1. O2– > F– > Na+ > N3–
2. N3– > Na+ > F– > O2–
3. N3– > O2– > F– > Na+
4. Na+ > F– > O2– > N3–
The oxoacid of phosphorus having the strongest reducing property is
1. H3PO3
2. H3PO2
3. H3PO4
4. H4P2O7
Consider the following reaction,
\(\small{\mathrm {xMnO_4^−+yC_2O_4^{2−}+zH^+→xMn^{2+}+2yCO_2+{z\over2} H_2O}}\)
The values of x, y, and z in the reaction are, respectively :
1. 5, 2 and 16
2. 2, 5 and 8
3. 2, 5 and 16
4. 5, 2 and 8
Among C, S and P, the element(s) that produce (s) SO2 on reaction with hot conc. H2SO4 is/are
1. only S
2. only C and S
3. only S and P
4. C, S and P
Which of the following statements is false?
1. ONCl and are isoelectronic.
2. molecule is bent
3. Ozone is violet-black in solid-state
4. Ozone is diamagnetic gas
The complex that can exhibit linkage isomerism is:
| 1. | [Co(NH3)5H2O]Cl3 | 2. | [Co(NH3)5(NO2)]Cl2 |
| 3. | [Co(NH3)5NO3](NO3)2 | 4. | [CO(NH3)5Cl]SO4 |
The tendency of X in BX3 (X = F,Cl, OMe, NMe) to form a bond with boron follows the order
1. BCl3 < BF3 < B(OMe)3 < B(NMe2)3
2. BF3 < BCl3 < B(OMe)3 < B(NMe2)3
3. BCl3 < B(NMe2)3 < B(OMe)3 < BF3
4. BCl3 < BF3 < B(NMe2)3 < B(OMe)3
On complete hydrogenation, natural rubber produces
1. polyethylene
2. ethylene-propylene copolymer
3. polyvinyl chloride
4. polypropylene
1 gram of a carbonate on treatment with excess HCl produces 0.01186 mole of . The molar mass of in g mol–1 is:
1. 84.3
2. 118.6
3. 11.86
4. 1186
Given :
Based on the above thermochemical equations, the value of at 298 K for the reaction
1. 85.2 kJ/mol
2. 15.2 kJ/mol
3. -74.8 kJ/mol
4. 74.8 kJ/mol
The freezing point of benzene decreases by when 0.2 g of acetic acid is added to 20 g ofbenzene. If acetic acid associates to form a dimer in benzene, percentage association of acetic acid inbenzene will be : ( for benzene = 5.12 K kg )
1. 80.4%
2. 74.6%
3. 94.6%
4. 64.6%
The most abundant elements by mass in the body of a healthy human adult are : Oxygen (61.4%); Carbon (22.9%), Hydrogen (10.0%) ; and Nitrogen (2.6%). The weight which a 75 kg person would gain if all atoms are replaced by atoms is :
1. 37.5 Kg
2. 7.5 Kg
3. 10 Kg
4. 15 Kg
The formation of which of the following polymers involves hydrolysis reaction ?
1. Bakelite
2. Nylon 6,6
3. Terylene
4. Nylon 6
Given :
Among the following, the strongest reducing agent is :
In the following reactions, ZnO is respectively acting as a/an :
a.
b.
1. base and base
2. acid and acid
3. acid and base
4. base and acid
A gaseous hydrocarbon gives upon combustion 0.72 g of water and 3.08 g. of .The empirical formula of the hydrocarbon is :
1.
2.
3.
4.
A pair in which both species are not likely to exist is:
| 1. | \(H^+_2,He^{2-}_2\) | 2. | \(H^-_2,He^{2+}_2\) |
| 3. | \(H^{2+}_2,He_2\) | 4. | \(H^+_2,He^{2+}_2\) |
Which of the following exists as covalent crystals in the solid-state ?
1. Iodine
2. Silicon
3. Sulphur
4. Phosphorus
The coagulating power of electrolytes with ions Na+, Al3+, and Ba2+for arsenic sulphide sol increases in the order :
1.
2.
3.
4.
Which of the following represents the correct order of increasing first ionization enthalpy for Ca, Ba, S, Se, and Ar?
1. Ca < S < Ba < Se < Ar
2. S < Se < Ca < Ba < Ar
3. Ba < Ca < Se < S < Ar
4. Ca < Ba < S < Se < Ar
Energy of an electron is given by E = . Wavelength of light required to excite an electron in an hydrogen atom from level n = 1 to n = 2 will be :
1.
2.
3.
4.
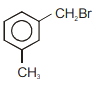
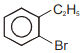
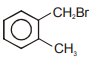
Compound (A), , gives a white precipitate when warmed with alcoholic . Oxidation of (A) gives an acid (B), . (B) easily forms anhydride on heating. Identify the compound (A).
1. 
2. 
3. 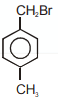
4. 
On treatment of 100 mL of 0.1 M solution of with excess ions are precipitated. The complex is :
1.
2.
3.
4.
How many litres of water must be added to 1 litre an aqueous solution of HCl with a pH of 1 to create an aqueous solution with pH of 2 ?
1. 0.1 L
2. 0.9 L
3. 2.0 L
4. 9.0 L
The first ionisation potential of Na is 5.1 eV. The value of electron gain enthalpy of will be :
1. -2.55 eV
2. -5.1 eV
3. -10.2 eV
4. +2.55 eV
The increasing order of the reactivity of the following halides for the reaction is:
(I)
(II)
(III)
1. (II) < (I) < (III)
2. (I) < (III) < (II)
3. (II) < (III) < (I)
4. (III) < (II) < (I)
Which of the following reactions will not result in the formation of carbon-carbon bonds ?
1. Reimer-Tiemann reaction
2. Cannizaro reaction
3. Wurtz reaction
4. Friedel-Crafts acylation
The potential energy increases when-
1. a conservative force does positive work.
2. a non-conservative force does positive work.
3. a conservative force does negative work.
4. a non-conservative force does negative work.
A constant force of 20 N is applied in horizontal direction on system of blocks kept on frictionless surface as shown in the figure. The contact force between 3 kg & 2 kg blocks is and between 2 kg and 5 kg blocks is , then-
1.
2.
3.
4. can't say
Three blocks are connected by a string as shown in figure. Find the maximum value of m if both the blocks on the horizontal plane move together.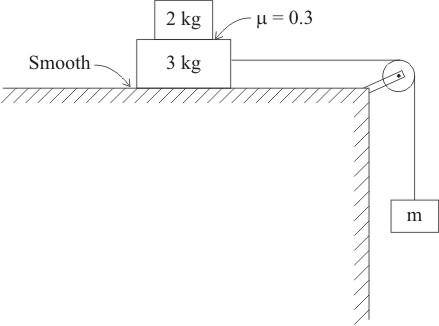
1. 1 kg
2. 2.1 kg
3. 1.5 kg
4. 0.5 kg
Two particles A and B are moving in uniform circular motion in concentric circles of radii with speeds respectively. If their time period of motion is same, then the ratio of their radii is-
1.
2.
3. 1:1
4. None of the above
A particle moves along a path \(ABCD\) as shown in the figure. The magnitude of the displacement of the particle from \(A\) to \(D\) is:
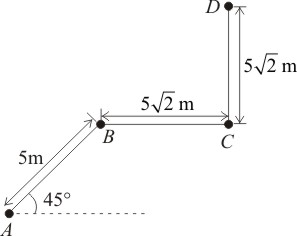
1. m
2. \(10\) m
3. m
4. \(15\) m
A force F is given in terms of time t as F = AsinCt. The dimensions of are given by:
1.
2.
3.
4.
A ball is projected at a certain angle with initial velocity \(u\). It covers horizontal range \(R\). With what initial velocity it should be projected keeping the angle of projection the same so that its horizontal range becomes \(2.25R\)?
| 1. | \(2.5u\) | 2. | \(1.5u\) |
| 3. | \(2.25u\) | 4. | \(0.25u\) |
A wire of negligible mass and length \(2\) m is stretched by hanging a \(20\) kg load to its lower end keeping its upper end fixed. If work done in stretching the wire is \(50\) J, then the strain produced in the wire will be:
1. \(0.5\)
2. \(0.1\)
3. \(0.4\)
4. \(0.25\)
A stone of mass 5kg tied to a string of length m is brought to a position where the string makes angle with the vertical as shown in the figure, and released. The angular acceleration of the stone just after the release is (string is massless)-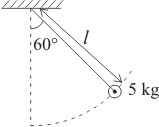
1. 1 rad/
2. 0.5 rad/
3. 1.5 rad/
4. 2.5 rad/
A tank is filled with water up to a height h and the pressure at the bottom of the tank is 3P where P is the atmospheric pressure. The height of a point from the bottom of the tank where pressure is is:
1.
2.
3.
4. None of these
A particle of mass M is at distance 'a' from the surface of a thin spherical shell of mass M and radius a. If are the potentials at the centre and the surface of the shell respectively, then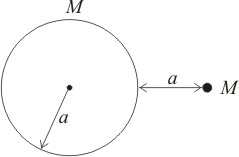
1.
2.
3.
4. Can't say
Moment of inertia of a solid sector about axis passing through center and perpendicular to plane of sector as shown in figure, if the mass of the sector is M and radius is R, is-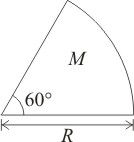
1.
2.
3.
4.
A circle of radius \(a\) is cut out from a square of uniform density and side \(4a\). If initial mass of the square is \(M\), then the centre of mass of the remaining part is: (where \(O\) is center of square)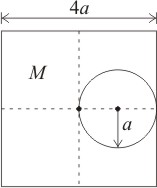
1. \(\frac{a}{16-\pi}\) to the left of \(O\).
2. \(\frac{a}{16-\pi}\) to the right of \(O\).
3. \(\frac{\pi a}{16-\pi}\) to the right of \(O\).
4. \(\frac{\pi a}{16-\pi}\) to the left of \(O\).
A pencil is used to draw part of a circle of radius R using a normal force N. If the coefficient of kinetic friction between the pencil and the surface is , then the work done by the friction force is-
1.
2.
3.
4.
A sinusoidal wave is given in the figure. The points moving upward are: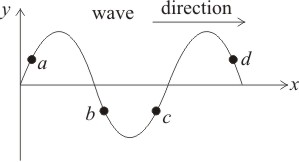
1. a and b
2. b and d
3. c and d
4. b and c
The distance moved by a particle in simple harmonic motion in time where T is the time period of the particle-
1.
2.
3.
4. 3 A
A stationary wave between two fixed points A and B is shown in the figure. Then: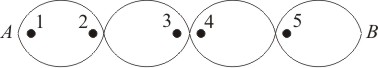
1. Points 1 and 2 are in phase but points 3 and 4 are not in phase.
2. Points 1 and 3 are in phase but points 3 and 5 are not in phase.
3. Points 2 and 3 are in phase but points 2 and 4 are not in phase.
4. None of these.
A spring of certain length and having spring constant k is cut into two pieces of lengths in ratio 1:2. The spring constants of the two pieces are in a ratio-
1. 1 : 1
2. 1 : 4
3. 1 : 2
4. None of these
The rms speed of oxygen atoms is v. If the temperature is halved and the oxygen atoms combine to form oxygen molecules, then the rms speed will be:
1.
2.
3. 2v
4.
The efficiency of a Carnot engine working between 800 K and 500 K is -
1. 0.4
2. 0.625
3. 0.375
4. 0.5
An ideal gas mixture filled inside a balloon expands according to the relation =constant. The pressure inside the balloon is-
1. increasing
2. decreasing
3. constant
4. can't say
A body of density \(0.7\) g/cm3 floats on a lake of water. The fraction of the body that is outside water is:
1. \(30 \text{%}\)
2. \(70 \text{%}\)
3. \(25 \text{%}\)
4. \(50 \text{%}\)
Hot coffee in a mug cools from \(90^{\circ}\text{C}\) to \(70^{\circ}\text{C}\) in \(4.8\) minutes. The room temperature is \(20^{\circ}\text{C}.\) Applying Newton's law of cooling, the time needed to cool it further by \(10^{\circ}\text{C}\) should be nearly:
| 1. | \(4.2\) minute | 2. | \(3.8\) minute |
| 3. | \(3.2\) minute | 4. | \(2.4\) minute |
A current-carrying wire \(LN\) is bent in the form shown below. If the wire carries a current of \(10 ~\text A\) and it is placed in a magnetic field of \(5 ~\text T\) which acts perpendicular to the paper outward, then it will experience a force of:

1. \(0~\text N\)
2. \(5~\text N\)
3. \(30~\text N\)
4. \(20~\text N\)
The most suitable curve for a paramagnetic substance between magnetization M and magnetising field H is:
1. A
2. B
3. C
4. D
A magnetic rod is inside a coil of wire which is connected to an ammeter. If the rod is stationary, which of the following statements is true?
| 1. | The rod induces a small current. |
| 2. | The rod loses its magnetic field. |
| 3. | There is no induced current. |
| 4. | None of these. |
A resistor is connected to a coil with 30 turns and area of . A magnet is moving down as shown in the figure. The magnetic field increases from 0.01 T to 0.06 T in 20 ms. Find the magnitude of the current going through the resistor.
1. 2.5 A
2. 0.1 A
3. 0.25 A
4. 0.01 A
The ammeter A reads 3 A and a non-ideal voltmeter V reads 30 V. The value of resistance R is-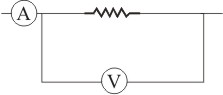
1. Exactly 10 ohm
2. Less than 10 ohm
3. more than 10 ohm
4. can't say
Three bulbs with individual power ratings of \(12~\text{W}, ~2~\text{W}~\text{and}~6~\text{W}\) respectively are connected as per the circuit diagram below. Find the amount of heat dissipated by each in \(10\) seconds.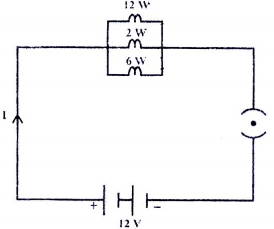
1. \(8~\text{J}, ~1.33~\text{J}, ~4~\text{J}\)
2. \(120~\text{J}, ~20~\text{J}, ~60~\text{J}\)
3. \(10~\text{J}, ~0.277~\text{J}, ~2.5~\text{J}\)
4. \(12~\text{J}, ~1.66~\text{J}, ~5~\text{J}\)
Relation between magnetic field at the center of cube and magnetic field at the center of circular loop is-
1.
2.
3.
4. data are insufficient
In the figure, where along the line connecting the two charges is the electric potential (V) due to the two charges zero?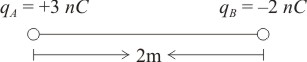
1. 1.2 m to the left of
2. 1.2 m to the left of
3. 1.2 m to the right of
4. 1.2 m to the right of
The direction of the Electric field at point C due to charge q and -2q at points A and B such that C is a point on the perpendicular bisector of AB above line AB and charge q is negative-
In the given figure if \(V = 4~\text{volt}\) each plate of the capacitor has a surface area of\(10^{-2}~\text{m}^2\) and the plates are \(0.1\times10^{-3}~\text{m}\)apart, then the number of excess electrons on the negative plate is:

In a semiconductor diode, P-side is earthed and N-side has a potential of -2 V. Then, the diode shall-
1. Conduct
2. Non conduct
3. Conduct partially
4. Break down
If input frequency is 50Hz, then ripple frequency of a half-wave rectifier will be equal to-
1. 100 Hz
2. 50 Hz
3. 25 Hz
4. Data is insufficient
Size of image formed by a convex lens is one fourth of size of real object. If focal length of the lens is 30 cm, then distance of the object from the lens is-
1. 150 cm
2. 120 cm
3. 90 cm
4. 60 cm
Two waves of equal amplitude interfere destructively resulting in a wave with zero amplitude. Then phase difference between the two waves is-
1.
2.
3.
4. 0
The correct order of frequencies of different radiations in a photoelectric experiment is-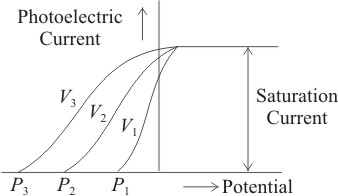
1.
2.
3.
4.
The kinetic energy of a photoelectron emitted on shining light of wavelength m on a metal surface of work function 0.1 eV is-
1. 0.01 eV
2. 0.02 eV
3. 1 eV
4. 0.1 eV
An electron is in an excited state in a hydrogen like atom. It has a total energy of -3.4 eV. The kinetic energy is E and its de-Broglie wavelength is . Then-
1. E=6.8 eV, =6.6
2. E=3.4 eV, =6.6
3. E=3.4 eV, =6.6
4. E=6.8 eV, =6.6
Compound X is found to radioactively decay with a rate constant equal to . If a time of 20 min passes by, what percentage of compound X has decayed within this period?
1. 63%
2. 50%
3. 37%
4. 25%
As the doping of a pure semiconductor increases, the resistance of the semiconductor-
1. Remains the same
2. Decreases
3. increases
4. None of these
In an AC circuit, alternating voltage \(e=200 \sqrt{2} \sin 100 t\) Volt is connected to a capacitor of capacity \(1~\mu \text{F}\). The RMS value of the current in the circuit is:
1. \(100\) mA
2. \(200\) mA
3. \(20\) mA
4. \(10\) mA
Energy density associated with an electromagnetic wave is-
1.
2.
3.
4. None
Select the incorrect option-
1. A plane mirror always forms a virtual image
2. The virtual image formed by a concave mirror is always enlarged
3. The critical angle is always defined in the denser medium
4. A glass slab cannot deviate the light