The pollen tube usually enters the embryo sac
1. Through one of the synergids
2. By directly penetrating the egg
3. Between one synergid and central cell
4. By knocking off antipodal cells.
Function of stem is :-
1. To bear leaves, flowers and branches.
2. Conduction of water and minerals to leaves.
3. Conduction of photosynthates
4. All of the above
How many enzymes in the list given below are protein digesting enzymes? Lactase, Trypsin, Pepsin, Rennin, Carboxypeptidase, Nuclease, Maltase, Ligase, Esterase, Ptylin, Lysozyme, Streptokinase
1. Six
2. Three
3. Four
4. Five
Read the given characteristics.
A - Cell wall form two thin overlapping shells.
B - Aseptate and coenocytic mycelium.
C - Walls are embedded with silica.
D - Smallest living cells which can survive without oxygen.
E - Pellicle makes body flexible.
F - Plasmogamy brought about by fusion of two vegetative cells.
G - Wall has stiff cellulose plates on outer surface.
H - Formation of zygospore by fusion of two gametes.
How many above mentioned characteristics are found in Phycomycetes , Chrysophytes, Euglenoids,
Dianoflagellates, Mycoplasma and Basidiomycetes respectively?
1. 2,2,1,1,1,1
2. 1,2, 1, 1,1,2
3. 1,1,1,1,2,2
4. 1,2,1,2,1,1
Photosynthesis in C4 plants is relatively less limited by atmospheric CO2 levels because:
1. The primary fixation of CO2 is mediated via PEP carboxylase
2. Effective pumping of CO2 into bundle sheath cells
3. Four carbon acids are the primary initial CO2 fixation products
4. RuBisCO in C4 plants has a higher affinity for CO2
cDNA probes are copied from the messenger RNA with help of
1. Restriction enzymes
2. Reverse transcriptase
3. DNA polymerase
4. Adenosine deaminase
Read the following four statements (1 – 4), find out certain mistakes in two of them.
| 1 | In our stomach lactic acid bacteria play a beneficial role by checking the growth of disease-causing microbes. |
| 2 | Lactic acid bacteria produce acid, that coagulates and completely digests the milk protein. |
| 3 | Lactic acid bacteria improves the nutritional quality by increasing the amount of Riboflavin |
| 4 | Lactic acid bacteria require a suitable temperature for their multiplication. |
Which of the two statements given above are not correct?
1. Statements (1) and (2)
2. Statement (1) and (3)
3. Statement (2) and (3)
4. Statements (2) and (4)
Which one of the following is not a function of cytokinin?
| 1. | Delay in senescence |
| 2. | Breaking of seed dormancy |
| 3. | Promoting bud dormancy |
| 4. | Promoting stomatal opening. |
The diagram below is a diagrammatic representation showing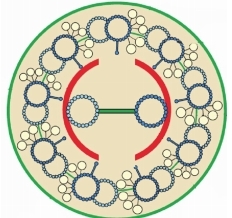
1. Section of a centriole
2. A cartwheel 9 + 0 structure of microtubular organisation
3. Internal structure of an eukaryotic flagella
4. Internal structure of a centrosome
What is incorrect statements-
1. All Organisms that can respond to stimuli are with a well developed nervous system..
2. Generally growth and reproduction are mutually exclusive events in higher organism.
3. Mules and sterile worker bees do not reproduce.
4. Metabolic reactions may take place in isolated cell-free systems
Mosses and ferns are found in moist and shady places because both
1. Require presence of water for fertilisation
2. Do not need sunlight for photosynthesis
3. Depend for their nutrition on microorganisms which survive only at low temperature
4. Cannot compete with sun-loving plants.
Which one of the following statements is correct?
| 1. | ‘Bt’ in Bt-cotton indicates that it is genetically modified organism produced through biotechnology. |
| 2. | Somatic hybridisation involves fusion of two complete plant cells carrying desired genes. |
| 3. | The anticoagulant hirudin is being produced from transgenic Brassica napus seeds. |
| 4. | ‘Flavr Savr’ variety of tomato has enhanced production of ethylene which improves its taste |
Fibrous thickenings of hygroscopic nature are found in this part of the anther
1. Epidermis
2. Endothecium
3. Middle layer
4. Tapetum.
If RQ is 0.6 in a respiratory metabolism, it would mean that
| 1. | Carbohydrates are used as respiratory material |
| 2. | Organic acids are used as respiratory substrate |
| 3. | The oxidation of the respiratory substrate consumed more oxygen than the amount of CO released |
| 4. | The oxidation of respiratory substrate consumed less oxygen than the amount of CO2 released |
The total number of progeny obtained through dihybrid cross of Mendel is 1280 in F2 generation. How
many are recombinants?
1. 240
2. 480
3. 360
4. 720
The amino acid substituted in sickle cell anaemia is
1. Glutamic acid for valine in alpha chain
2. Glutamic acid for valine in ß-chain
3. Valine for glutamic acid in the ɑ-chain
4. Valine for glutamic acid in the ß-chain
The process used in conversion of Pyruvate to Acetyl CoA is
1. Oxidative dehydration
2. Oxidative decarboxylation
3. Oxidative phosphorylation
4. Reductive phosphorylation.
Farmers sprayed litchi trees to supress populations of scale insects. This also killed populations of predatory lace wings that controlled the numbers of scale insects. Soon the spraying did no good and the damage to the litchi from the scale insects was greater than before spraying had occurred. Few predatory lace wings exist in the trees now because
1. They too are insects so they are r-selected.
2. Both insects were K-selected and resistance was a matter of chance.
3. The predator was r-selected and was therefore was more susceptible to the spray
4. The scale insect was r-selected and the high number of young included a resistant strain; the predator was k-selected and will take longer to produce a resistant variety.
Which of the following is not part of sexual life cycle of fungi?
1. Karyogamy
2. Plasmogamy
3. Zygotic meiosis
4. Gametic meiosis
Kingdom Protista
1. Are primarily terrestrial
2. Forms a link between archaebacteria and bacteria
3. Contain membrane bound organelles and have lack of zygote formation
4. Include Chrysophytes which are mostly microscopic and photosynthetic
Which of the following is false with respect to dinoflagellates
1. They are multi-coloured
2. Cell wall is covered by silica plates
3. The two flagella in which one lies laterally
4. Many of them release toxins which kill marine animals
Which of the following are parasites?
1. Mushrooms and toadstools
2. Funaria
3. Yeast and Aspergillus
4. Rust and smut fungi
Which of the following is not true for fermentation?
| 1. | Fermentation accounts for only a partial breakdown of glucose |
| 2. | In fermentation there is a net gain of only two molecules of ATP for each molecule of glucose |
| 3. | During fermentation NADH is oxidised to NAD+ very rapidly |
| 4. | Anaerobic respiration takes place in the cytoplasm |
Which element plays an important role in pollen germination and carbohydrate translocation?
1. Copper
2. Zinc
3. Boron
4. Molybdenum
The enzyme nitrogenase
1. Is present in the leaves of legumes
2. Is an iron copper protein
3. Catalyses conversion of atmospheric nitrogen to ammonia
4. Catalyses conversion of ammonia to amino acids
Study the given statements -
| i. | A dehydration reaction (or condensation reaction) is the process in which water molecules are produced as a polymer is formed from monomers. |
| ii. | The four main categories of macromolecules present in living systems are proteins, nucleic acids, carbohydrates, and lipids. |
| iii. | Glucose is the main monosaccharide used by human cells for energy. |
| iv. | The building blocks or monomers of nucleic acid molecules are called nucleosides. |
How many statements are correct?
1. i
2. ii
3. iii
4. iv
The given floral diagram helps us to deduce that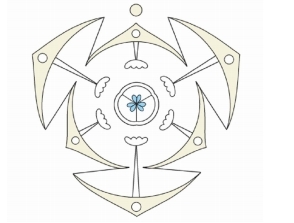
1. The flower is dicotyledonous
2. The flower shows parietal placentation
3. The flower is having six petals
4. The flower is epiphyllous
Which of the following statements is incorrect?
| 1. | The reaction centre of the photosystems is made up of a single chlorophyll a molecule |
| 2. | The reaction centre is same in both the photosystems |
| 3. | The reaction centre chlorophyll a of PS1 has an absorption peak at 700 nm |
| 4. | The various pigments are organised into light-harvesting complexes within the photosystems |
The heartwood in old trees is
1. Mostly made of dead elements with lignified walls
2. Conducts water and stores organic compounds like tannins, resins, oils etc.
3. Is light in colour and stores food.
4. Both a and b
Secondary growth in roots is different from stem in
1. Absence of cork cambium
2. Absence of medullary rays
3. Developing completely secondary vascular cambium
4. Absence of vascular cambium
Vivipary in plants means
1. Development of fruit after the fertilised seed is buried in the soil
2. Germination of seeds in crevices of the stem where some soil is deposited
3. Germination of seeds while inside the fruit when it is still on the parent tree
4. Organisation of several seeds
Which is not correct about methanogens?
1. they are archaebacteria
2. they live in marshy areas
3. Methane is their preferred carbon source
4. They are present in guts of several ruminant animals (cow, buffaloes) and they produce biogas (CH) from the dung of these animals.
Which of the following is not true for Pisum sativum.
1. Vexillary aestivation
2. Polycarpellary and unilocular ovary
3. Single style
4. Diadelphous androecium
According to the chemiosmotic hypothesis, there is a basic difference between respiration and photosynthesis. This is _____
| 1. | In chloroplast, H+ accumulation takes place in the lumen of thylakoid but in mitochondria, this takes place in perimitochondrial space. |
| 2. | Accumulation of H+ in chloroplast takes place in matrix i.e., stroma and in mitochondria, this takes place in cristae. |
| 3. | In both the mitochondria and chloroplast the accumulation of H+ takes place in inter-membranous space but with a high rate in mitochondria |
| 4. | Both 1 and 3 are correct. |
In rocky coasts of Scotland, studies show that the larger barnacle Balanus dominates the intertidal area and excludes another barnacle Chathamalus. This behaviour is explained by
1. Rivet popper hypothesis
2. Allen’s rule
3. Gause’s exclusion principle
4. Evil quartet
According to operon hypothesis, how does a regulator gene control the biochemical reactions in a cell?
1. By repression of transcription
2. By inactivating the enzyme
3. By inactivating the medium
4. By repression of migration of m-RNA
Pathogens of Arthropods effectively used as biological control agents include
1. Trichoderma
2. Baculoviruses
3. Agrobacterium tumefaciens
4. E. Col
In the above diagram showing a typical agaro gel
electrophoresis
1. Lane 1 shows undigested DNA fragment
2. Lane 4 shows largest DNA particles towards ‘y’
3. The procedure helps to form clones of DNA
4. The procedure takes place as the DNA fragments
are double stranded
By inserting an alien DNA at Bam H1 site of PBR322
1. The gene for ampicillin resistance gets inactivated
2. The gene for tetracycline resistance gets inactivated
3. Neither genes get inactivated
4. Both genes get inactivated
The below reaction depicts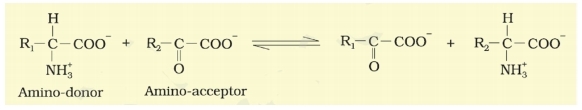
1. Reductive amination
2. Transamination
3. Action of enzyme nitrogenise
4. Formation of amide
Study the statements
i. The imbibitions or the uptake of water is the first step towards the germination of seed.
ii. The mobilization of food reserves is important for seed germination which involves the hydrolysis of stored polysaccharides, proteins, and lipids with the help of enzymes.
iii. The seed needs a suitable temperature between 25 to 35C for germination.
iv. Dormancy in seeds may be due to the presence of germination inhibitors such as abscisic acid, phenolic acid, short chain fatty acids and coumarin.
v. Scarification of seed involves scratching of seed coat to help in breaking of the dormancy caused by hard and impermeable seed coat.
vi. Stratification of seeds is subjecting the moist seeds to oxygen for variable periods of low or high temperatures.
Find out the correct statements-
1. All
2. iii
3. iv
4. none
Glycerol would enter the respiratory pathway after being converted to-
1. PGAL
2. DPGA
3. PGA
4. Acetyl CoA
Which of the following is incorrect for Bryophytes?
1. Zygotes do not undergo reduction division immediately.
2. Sporophyte is not free living but attached to photosynthetic gametophyte.
3. Spores germinate to produce sporophyte.
4. Sex organs are multicellular.
In C plants, for fixation of 6 CO how many ATP and NADPH are required respectively?
1. 12, 18
2. 18, 12
3. 10, 16
4. 16, 10
How many of the following belong to family Solanaceae ?
Chilli, Tulip, Petunia, Colchicine, Belladonna, Lupin, Tobacco, Indigifera,Tomato, Gloriosa,
Dolichos, Citrus.
1. 4
2. 6
3. 5
4. 3
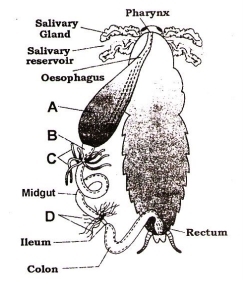
Identify structures A to D –
|
A |
B |
C |
D |
|
|
1. |
Gizzard |
Crop |
Hepatic caecae |
Malpighian tubules |
|
2. |
Crop |
Gizzard |
Hepatic caecae |
Malpighian tubules |
|
3. |
Crop |
Gizzard |
Malpighian tubules |
Hepatic caecae |
|
4. |
Gizzard |
Crop |
Malpighian tubules |
Hepatic caecae |
Identify A and B bonds in the following diagrammatic representation of a portion of glycogen –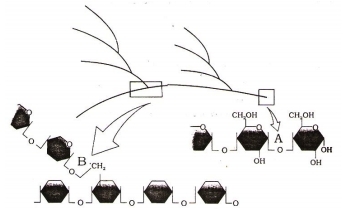
1. A = 1 – 4 -glycosidic bonds, B = 1 - 6 -glycosidic bonds
2. A = 1 - 6 -glycosidic bonds, B = 1 - 4 -glycosidic bonds
3. A = 1 - 1 -glycosidic bonds, B = 1 - 1 -glycosidic bonds
4. A = 1 - 4 -glycosidic bonds, B = 1 - 4 -glycosidic bonds
Match the column I with Column II correctly –
|
|
Column – I (Category) |
|
Column – II |
|
A. |
Pigments |
l. |
Concanavalin A |
|
B. |
Terpenoides |
ll. |
Monoterpenes, Diterpenes |
|
C. |
Alkaloids |
lll. |
Morphine, Codeine |
|
D. |
Lectins |
lV. |
Carotenoids, Anthocyanine |
1. A – IV, B – II, C – III, D – I
2. A – IV, B – III, C – II, D – I
3. A – I, B – IV, C – III, D – II
4. A – I, B – III, C – II, D – IV
The steps in catalytic cycle of an enzyme action are given in random order
I. The enzyme releases the products. Now enzyme is free to bind another substrate
II. The active sites, now in close proximity of substrate break the bond of substrate and E – P complex forms
III. Binding of substrate induces the enzyme to alter its shape fitting more tightly around the substrate
IV. The substrate binds to the active site of enzyme (i.e., fitting into the active site.
The correct order is
1. I, II, III, IV
2. IV, III, II, I
3. I, III, II, IV
4. I, II, IV, III
Which one of the following biomolecules is correctly characterized?
1. Lecithin – a phosphorylated glyceride found in cell membrane
2. Palmitic acid – an unsaturated fatty acid with 18 carbon atoms
3. Adenylic acid – adenosine with a glucose and phosphate molecule
4. Alanine amino acid – contains an amino group and an acidic group anywhere in the molecule
Find out the correct sequence of substrate, enzyme, and product.
1. Small intestine: Proteins Pepsin + Amino acids
2. Stomach: Fats Lipase + Micelles
3. Duodenum: Triglycerides Trypsin + Monoglycerides
4. Small intestine: Starch Amylase + Maltose
Which of the following statements is false?
1. The breakdown of most of the biomacromolecules occurs in the duodenum.
2. Simple substances (digested foods) are absorbed in the jejunum and ileum.
3. Very significant digestive activity occurs in the large intestine.
4. Undigested and unabsorbed substances are passed on to the large intestine.
The graph shows an oxygen dissociation curve for hemoglobin –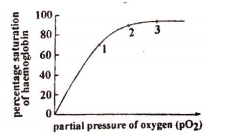
Where in the body will hemoglobin be saturated at the percentages shown at points 1, 2, and 3 on the graph?
|
Left ventricle |
Pulmonary vein |
Vena cava |
|
|
1. |
1 |
2 |
3 |
|
2. |
2 |
1 |
3 |
|
3. |
2 |
3 |
1 |
|
4. |
3 |
2 |
1 |
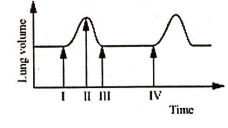
The given figure illustrates the changes in lung volume
during the process of breathing
The change from II to III indicates the
1. Movement of diaphragm away from the lungs
2. Expansion of the thoracic cavity
3. Movement of air out the lungs
4. Expansion of ribs
During ventricular systole –
1. Oxygenated blood is pumped into the aorta and deoxygenated blood is pumped into the pulmonary artery
2. Oxygenated blood is pumped into the pulmonary artery and deoxygenated blood is pumped into the aorta
3. Oxygenated blood is pumped into aorta and deoxygenated blood is pumped into pulmonary vein
4. Oxygenated blood is pumped into pulmonary vein and deoxygenated blood is pumped into pulmonary artery
A red blood cell, entering the right side of the heart passes by or through the following structures –
| (1) | Atrioventricular valves | (2) | Semi-lunar valves |
| (3) | Right atrium | (4) | Right ventricle |
| (5) | SAN |
1. 23145
2. 31524
3. 35124
4. 53142
Which one is correct?
| 1. | Notochord is mesodermal in origin and present in vertebrates only. |
| 2. | Notochord is a mesodermally derived rod like structure formed on the dorsal side in embryonic development in chordates. |
| 3. | All chordates are vertebrates. |
| 4. | All vertebrates possess bony vertebral column. |
Match column I with column II –
|
|
Column I |
|
Column – II |
|
A. |
PCT |
l. |
Concentrated urine formation |
|
B. |
DCT |
ll. |
Filtration of blood |
|
C. |
Loop of Henle |
lll. |
Reabsorption of 70-80% electrolytes |
|
D. |
Counter – current mechanism |
lV. |
Ionic Balance |
|
E. |
Renal corpuscle |
V. |
Maintenance of conc. gradient in medulla |
1. A – III, B – IV, C – I, D – V, E – II
2. A – III, B – V, C – IV, D – II, E – I
3. A – I, B – III, C – II, D – V, E – IV
4. A – III, B – I, C – IV, D – V, E – II
Which one of the following statements is correct with respect to kidney function regulation?
1. During summer when body loses lot of water by evaporation, the release of ADH is suppressed
2. When someone drinks lot of water, ADH release is suppressed
3. Exposure to cold temperature stimulates ADH release
4. An increase in glomerular blood flow stimulates formation of Angiotensim II
A motor unit is best described as –
1. All the nerve fibres and muscle fibres in a single muscle bundle
2. One muscle fibre and its single nerve fibre
3. A single motor neuron and all the muscle fibres that it innervates
4. It is the neuron which carries the message from muscle to CNS
Match the following and mark the correct option –
|
|
Column I |
|
Column – II |
|
A. |
Hinge joint |
l. |
Between humerus and pectoral girdle |
|
B. |
Pivot joint |
ll. |
Between carpals and metacarpals of thumb |
|
C. |
gliding joint |
lll. |
Between the carpals |
|
D. |
Saddle joint |
lV. |
Between atlas and axis |
|
E. |
Ball and Socket joint |
V. |
Knee joint |
1. A – V, B – IV, C – III, D – II, E – I
2. A – I, B – II, C – III, D – V, E – IV
3. A – I, B – III, C – II, D – V, E – IV
4. A – V, B – III, C – II, D – I, E – IV
Five events in the transmission of nerve impulse across the synapse are given:
| A. | Opening of specific ion channels allows the entry of ions, a new action potential is generated in the post synaptic neuron. |
| B. | Neurotransmitter binds to the receptor on post synaptic membrane. |
| C. | Synaptic vesicle fuses with pre-synaptic membrane, neurotransmitter releases into synaptic cleft. |
| D. | Depolarization of pre-synaptic membrane. |
| E. | Arrival of action potential at axon terminal. |
In which sequence do these events occur?
1. E → D → C → B → A
2. A → B → C → D → E
3. A → B → D → C → E
4. E → D → C → A → B
The swollen base of semicircular canals containing projecting ridge is called–
1. Macula
2. Jacobson’s organ
3. Crista ampullaris
4. Organs of Corti
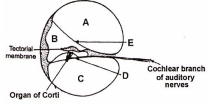
Go through the above sectional view of Cochlea. Identify A to E
| Options: | A | B | C | D | |
| 1. | Scala vestibule | Scala media | Scala tympani | Basilar membrane | Reissner’s membrane |
| 2. | Scala media | Scala vestibule | Scala tympani | Basilar membrane | Reissner’s membrane |
| 3. | Scala tympani | Scala media | Scala vestibule | Basilar membrane | Reissner’s membrane |
| 4. | Scala vestibule | Scala media | Scala tympani | Reissner’s membrane | Basilar membrane |
Match the Source gland with its respective hormone and function and select the correct option.
|
Source gland |
Hormone |
Function |
|
|
1. |
Anterior pituitary |
Oxytocin |
Contraction of uterus muscles during child birth |
|
2. |
Posterior pituitary |
Vasopressin |
Stimulates reabsorption of water in the distal tubules in the nephron |
|
3. |
Corpus luteum |
Estrogen |
Supports Pregnancy |
|
4. |
Thyroid |
Thyroxine |
Regulates blood calcium level |
The given table enlists various hormones and their chemical nature.
Hormone Chemical composition
(1) ___(i)____ Peptide
(2) Testosterone ___(ii)____
(3) Thyroxine ___(iii)____
(4) ___(iv)____ Amino-acid derivative
Which is the correct option that completes the given table?
1. i-Cortisol; ii-Steroids; iii-Polypeptide; iv-Estradiol
2. i-Insulin; ii-Proteins; iii-Polypeptide, iv-Epinephrine
3. i-Cortical; ii-Proteins; iii-Iodothyronines; iv-Estradiol
4. i-Insulin; ii-Steroids; iii-Iodothyronines; iv-Epinephrine
Which of the following is the common ancestor of Cycads and Dicotyledons?
1. Ginkgo’s
2. Seed ferns
3. Gnetales
4. Lycopods
In the following figures, there are three skulls. Which statement is correct about these skulls?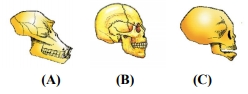
1. A – Skull of a baby chimpanzee
B – Skull of a man
C – Skull of adult human
2. A – Skull of an adult chimpanzee
B – Skull of a baby chimpanzee
C – Skull of adult human
3. A – Skull of an adult chimpanzee
B – Skull of adult human
C – Skull of a baby chimpanzee
4. A – Skull of a baby chimpanzee
B – Skull of adult human
C – Skull of an adult chimpanzee
Which of the following cannot be included under the secondary lymphoid organ?
1. Thymus
2. MALT
3. Spleen
4. Lingual tonsil
An example of an autoimmune disease is
1. Asthma
2. Cancer
3. Erythroblastosis foetalis
4. Myasthenia gravis
Which of the following is/are correct statement regarding contraception?
| (1) | Insertion of IUD could be effective contraceptive within 3 days of unprotected sex |
| (2) | Morning after pills are used as emergency contraceptives |
| (3) | IUDs prevent STDs. |
| (4) | Lactational amenorrhea is due to hyperprolactinemia |
1. (1) only
2. (1), (2) & (4) only
3. (1), (2) & (3) only
4. (1), (2), (3) & (4)
Mark the correct statement
(1) Pathogen specific response is acquired immunity
(2) Memory based response is acquired immunity
(3) Response against only intracellular pathogen is acquired immunity
(4) Response against only viral pathogen is acquired immunity
1. 1, 2, 3, 4
2. 1, 2 only
3. 2 only
4. 3 only
Anamnestic response is
1. Response against viral pathogens
2. Response against bacterial pathogens
3. Secondary response
4. Primary response
The primary and secondary immune response are carried out with the help of
1. B – lymphocyte only
2. T – lymphocyte only
3. Both B and T – lymphocyte
4. Neutrophils and monocytes
Sustained higher fever, weakness, stomach pain are related with
1. Cerebral malaria
2. Black fever
3. Typhoid
4. Black water fever
A person is injected with gamma-globulin against Snake venom. This is
1. Artificially acquired active immunity
2. Artificially acquired passive immunity
3. Naturally acquired active immunity
4. Naturally acquired passive immunity
Which of the following bacteria is known as natural genetic engineer of plants?
1. Agrobacterium tumefaciens
2. Bacillus thuringiensis
3. Rhizobium meliloti
4. Pseudomonas putida
Small volume cultures cannot yield appreciable quantity of products. So to produce the large quantities, the instrument used is
1. Biolistics
2. Biofermenter
3. Bioreactors
4. Biomagnifier
In which cells of the patient, the functional ADA gene is introduced as a part of the treatment of SCID?
1. Erythrocytes
2. Lymphocytes
3. Platelets
4. Liver cells
Mark the incorrect statement:
| 1. | By using Agrobacterium vectors nematode specific genes were introduced into the host plant. |
| 2. | Bacillus thuringiensis forms protein crystals during a particular phase of growth. |
| 3. | C-peptide is not present in mature insulin and is removed during maturation into insulin. |
| 4. | All vectors naturally contain antibiotic-resistance gene. |
Choose the correct match with respect to cranial capacity
1. Neanderthal man – 1800 cc
2. Homo habilis – 650 cc
3. Homo erectus – 200 cc
4. Australopithecus – 900 cc
Mode of action of IUD’s excludes
1. Blocking the entry of sperms through cervix
2. Phagocytosis of sperms within uterus
3. Suppressing sperm motility
4. Making the uterus unsuitable for implantation
The figure given below illustrates the changes taking place during the human menstruation cycle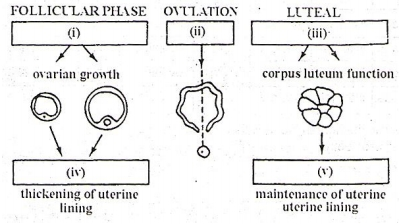
In each of the boxes shown in the figure write the name of the hormone, or hormones controlling the stage in the human menstrual cycle.
| 1. | (i) FSH, (ii) LH, (iii) LH, (iv) Estrogen, (v) Progesterone |
| 2. | (i) LH, (ii) FSH, (iii) LH, (iv) Estrogen, (v) Progesterone |
| 3. | (i) FSH, (ii) LH, (iii) FSH, (iv) Estrogen, (v) Progesterone |
| 4. | (i) FSH, (ii) LH, (iii) LH, (iv) Progesterone, (v) Estrogen |
Identify A and B, and their respective functions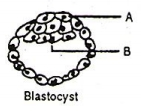
I. IUDs increase phagocytosis of sperms
II. Release Cu ions that suppress sperm motility and fertilizing capacity of sperms
III. Make the uterus unsuitable for implantation
IV. Make the cervix hostile to sperms
V. Prevent semen from entering the female reproductive tract.
1. I, II, III, IV, V
2. I, II, III, IV
3. I, II
4. II, III, IV
Mark the correct statement regarding natural selection and related speciation?
(1) Environmental changes lead to rapid speciation
(2) Anthropogenic action resulted in selection of resistant varieties in a much lesser time scale
(3) Natural selection results in availability of only dominant species in a population
(4) Natural selection maintains consistent differences in the contribution of various genotypes to the
next generation.
1. 1, 2, 3, 4
2. 2, 4 only
3. 4 only
4. 3, 4 only
Those that are better fit in an environment; leave more progeny than other’s, this statement explain -
1. Lamarck use and disuse theory
2. Hugo de Vries mutation theory
3. Founder effect
4. Natural selection theory of Charles Darwin
Mark the incorrect statement?
| 1. | Force of natural selection started with cellular form of life |
| 2. | According to natural selection rate of appearance of new forms is linked to life cycle or the life span |
| 3. | According to natural selection theory of fitness is based on characteristic which are inherited |
| 4. | Natural selection produces new genetic variability in a population. |
Column – I Column – II
A. Lippes loop I. Non-medicated IUDs
B. Multiload 375 II. Hormone releasing IUDs
C. CuT III. Copper releasing IUDs
D. Cu7
E. LNG – 20
F. Progestasert
The correct match is –
1. I – A; II – B, F; III – C, D, E
2. I – A; II – E, F; III – B, C, D
3. I – B; II – E, F; III – A, C, D
4. I – B; II – A, F; III – C, D, E
Identify A, B, C, D in the given diagram of E. Coli cloning vectors pBR 322.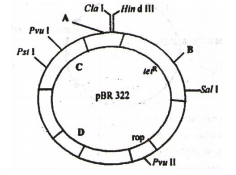
|
|
A |
B |
C |
D |
|
1. |
Hind I |
Eco RI |
ampR |
ori |
|
2. |
Hind I |
Bam HI |
kanR |
ampR |
|
3. |
Bam HI |
Pst I |
ori |
ampR |
|
4. |
Eco RI |
Bam HI |
ampR |
ori |

1.
2. Cl-
3. Cl
4. Cl2+

The product (B) in the above-mentioned reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Aluminium is extracted from alumina (Al2O3) by electrolysis of a molten mixture of :
1. Al2O3 + HF + NaAlF4
2. Al2O3 + CaF2 + NaAlF4
3. Al2O3 + Na3AlF6 + CaF2
4. Al2O3 + KF +Na3AlF6
Pb and Sn are extracted from their chief ore by:
1. carbon reduction and self-reduction respectively.
2. self-reduction and carbon reduction respectively.
3. electrolysis and self-reduction respectively.
4. self-reduction and self-reduction respectively.
Sulphur burns according to the reaction
What volume of air, at 1 atm and 273 K, containing 21% oxygen by volume is required to completely burn sulphur present in 200 g of the sample?
(This sample contains 20% inert material which does not burn)
1. 23.52 litre
2. 320 litre
3. 112 litre
4. 533.33 litre
The equivalent weight of in the given reaction is:
1. 16.25
2. 36.5
3. 73
4. 85.1
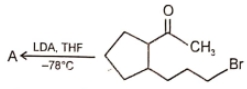
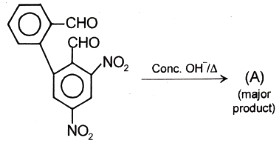
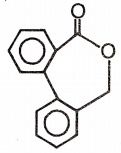
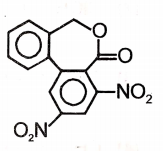
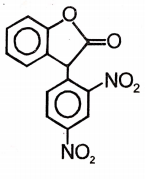
Major product 'A' and 'B' are respectively
1. 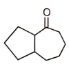
2. 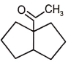
3. 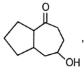
4. 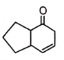
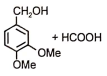
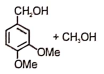
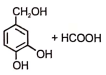
The following reaction gives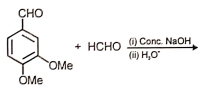
1.  2.
2. 
3.  4.
4. 
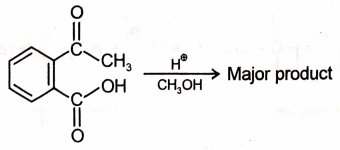
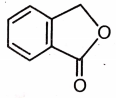
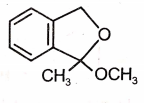
Which of the following is the most suitable major product?
1. 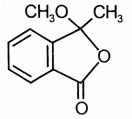 2.
2. 
3. 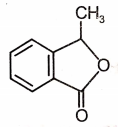 4.
4. 
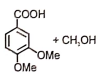
1.  2.
2. 
3.  4.
4. 
Compound (A) in the given reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
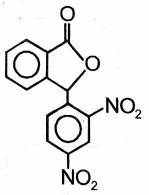
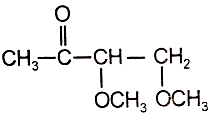
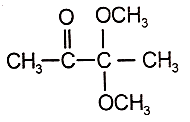
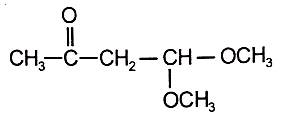
An organic compound (A) of the molecular formula gives a positive Iodoform test but does not reduce Tollen's reagent. When (A) is subjected to acid-catalyzed hydrolysis, another compound B is formed which gives positive Tollen's test. Identify the unknown compound (A).
1. 
2. 
3. 
4. 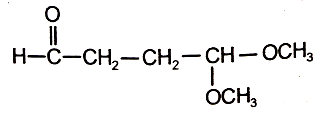
1.
2.
3.
4.
Given the following reaction:
The structure of the major product is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
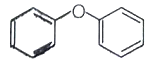
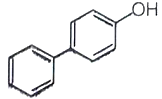
The major product of the following reaction is -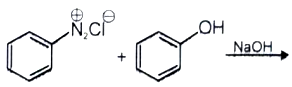
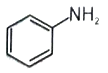
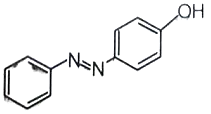
-
1. 
2. 
3. 
4. 
KOH react with O3. The product formed is/are
1. KO2, O2 and H2O
2. KO3, O2 and H2O
3. K2O2, O2 and H2O
4. K2O, O2 and H2O
Sodium metal dissolves in liquid ammonia and forms a deep blue solution. The colour is due to the absorption of light by:
1. Sodium ions
2. Ammoniated electrons
3. Free electrons
4. Ammoniated sodium ions
Which of the following compound can be used as a purgative?
1. HgCl2
2. ZnCl2
3. Hg2Cl2
4. ZnSO4.7H2O
The unpaired electron in Eu3+ will be the same in:
1. Yb2+
2. Ho3+
3. Sm2+
4. Gd3+
When excess of KCN is added to a solution of CuSO4, the final product obtained is,
1. K2[Cu(CN)4]
2. K3[Cu(CN)4]
3. CuCN
4. K[Cu(CN)2]
The most abundant transition metal in human body is
1. Copper
2. Iron
3. Zinc
4. Manganese
The total number of the optically active isomer of B is
1. 2
2. 3
3. 4
4. 6
1,2-dibromopropane on treatment with X moles of followed by treatment with ethyl bromine gave a pent-2-yne. The value of X is:
1. 1
2. 2
3. 3
4. 4
How many chiral centers are present in the given compound?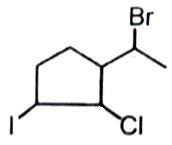
1. 1
2. 2
3. 3
4. 4
An amphoteric oxide among the following is -
1. CaO
2. CO2
3. SiO2
4. SnO2
If α is the degree of dissociation of Na2SO4, the vant Hoff’s factor (i) used for
calculating the molecular mass is
1. 1 + α
2. 1 - α
3. 1 + 2α
4. 1 – 2 α
An ionic compound has a unit cell consisting of A ions at the corners of a cube and B ions on the centers of the faces of the cube. The empirical formula for this compound would be :
1. AB
2. A2B
3. AB3
4. A3B
Polyamide is represented by:
1. Teflon
2. Nylon – 6,6
3. Terylene
4. Bakelite
Aluminium metal can be produced by the electrolysis of molten aluminium oxide at about 1000 °C.
The cathode reaction is: \(Al^{3 +} + 3 e^{-} \rightarrow Al\)
Given that the atomic mass of aluminium is 27 amu and 1 Faraday = 96,500 C, calculate the quantity of electricity (in coulombs) required to produce 5.12 kg of aluminium by this method:
1. \(5 . 49 \times 10^{1 } C\) of electricity
2. \(5 . 49 \times 10^{4 } C\) of electricity
3. \(1 . 83 \times 10^{7 } C\) of electricity
4. \(5 . 49 \times 10^{7 } C\) of electricity
Consider the reaction: carried out at constant temperature and
pressure. If ∆H and ∆U are the enthalpy and internal energy changes for the
reaction. The correct expressions among the following is -
1. ∆H = 0
2. ∆H = ∆U
3. ∆H <∆U
4. ∆H > ∆U
The solubility product of a salt having general formula MX2, in water is: . The concentration of M2+ ions in the aqueous solution of the salt is
1.
2.
3.
4.
Benzene and toluene form nearly ideal solutions. At 20C, the vapour pressure of
benzene is 75 torr and that of toluene is 22 torr. The partial vapour pressure of
benzene at 20 C for a solution containing 78 g of benzene and 46 g of toluene in
torr is
1. 50
2. 25
3. 37.5
4. 53.5
Two solutions of a substance (non-electrolyte) are mixed in the following manner.
480 ml of 1.5 M first solution +520 mL of 1.2 M second solution. What is the molarity of the final mixture?
1. 1.20 M
2. 1.50 M
3. 1.344 M
4. 2.70 M
Hydrogen ion concentration in mol / L in a solution of pH = 5.4 will be:
1.
2.
3.
4.
If we consider that 1/6, in place of 1/12;
mass of carbon atom is taken to be the relative
atomic mass unit, the mass of one mole of a substance will
1. Decrease twice
2. Increase two fold
3. Remain unchanged
4. Be a function of the molecular mass of the substance
|
Electrolyte |
KCl |
KNO3 |
HCl |
NaOAc |
NaCl |
|
(Scm2mol-1) |
149.9 |
145.0 |
426.2 |
91.0 |
126.5 |
Calculate Using appropriate molar conductances of the electrolytes listed
above at infinite dilution in H2O at 25°C
1. 517.2
2. 552.7
3. 390.7
4. 217.5
A schematic plot of In Keq versus inverse of temperature for a reaction is shown
below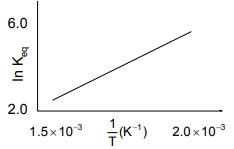
The reaction must be
1. exothermic
2. endothermic
3. one with negligible enthalpy change
4. highly spontaneous at ordinary
temperature
The number and type of bonds between two carbon atoms in calcium carbide are
1. One sigma, one pi
2. One sigma, two pi
3. Two sigma, one pi
4. Two sigma, two pi
Heating an aqueous solution of aluminium chloride to dryness will give
1. AlCl3
2. Al2Cl6
3. Al2O3
4. Al(OH)Cl2
The decreasing order of nucleophilicity among the nucleophiles
(i)
(ii)
(iii)
(iv)
1. (i), (ii), (iii), (iv)
2. (iv), (iii), (ii), (i)
3. (ii), (iii), (i), (iv)
4. (iii), (ii), (i), (iv)
Alkyl halides react with dialkyl copper reagents to give
1. alkenes
2. alkyl copper halides
3. alkanes
4. alkenyl halides
Reaction of cyclohexanone with dimethylamine in the presence of catalytic amount of an acid forms a compound if water during the reaction is continuously removed. The
compound formed is generally known as
1. a Schiff’s base
2. an enamine
3. an imine
4. an amine
An amount of solid NH4HS is placed in a flask already containing ammonia gas at a certain temperature and 0.50 atm. Pressure. Ammonium hydrogen sulphide decomposes to yield NH3 and H2S gases in the flask. When the decomposition reaction reaches equilibrium, the total pressure in the flask rises to 0.84 atm. The equilibrium constant for NH4HS decomposition at this temperature is
1. 0.30
2. 0.18
3. 0.17
4. 0.11
1. E1
2. E2
3.
4. elimination
The major product of the reaction is
1. I
2. II
3. III
4. IV
The displacement (\(Y\)) as a function of position (\(x\)) and time (\(t\)) is given as \(Y= Ae^{(bx+Ct)}\). Which of the following expressions has dimensions different from others?
| 1. | \(YC\) | 2. | \(AC\) |
| 3. | \(\frac{C}{b}\) | 4. | \(bC\) |
A car A is going north-east at 80 km/h and another car B is going south-east at 60 km/h. Then the direction of the velocity of A relative to B makes with the north an angle such that tan is:
1. 1/7
2. 3/4
3. 4/3
4. 3/5
A man is crossing a river flowing with a velocity of \(5\) m/s. He reaches a point directly across the river at a distance of \(60\) m in \(5\) s. His velocity in still water should be:
1. \(12\) m/s
2. \(13\) m/s
3. \(5\) m/s
4. \(10\) m/s
A monkey of mass 20 kg is holding a vertical rope. The rope can break when a mass of 25 kg is suspended from it. What is the maximum acceleration with which the monkey can climb up along the rope?
1.
2.
3.
4.
A block A kept on an inclined surface just begins to slide if the inclination is . The block is replaced by another block B and it is found that it just begins to slide if the inclination is .
1. Mass of A > mass of B
2. Mass of A < mass of B
3. Mass of A = mass of B
4. All the three are possible
The work done is pushing a block of mass 10 kg from bottom to the top of a frictionless inclined plane 5 m long and 3 m high is (considering g = 9.8 m/)
1. 392 J
2. 294 J
3. 98 J
4. 0.98 J
A particle is released from rest at origin. It moves under the influence of the potential field . The kinetic energy at x = 2 m is-
1. 2 J
2. 1 J
3. 1.5 J
4. 0 J
A particle of mass 3m is projected from the ground at some angle with horizontal. The horizontal range is R. At the highest point of its path, it breaks into two pieces of mass m and 2m. The smaller mass comes to rest and larger mass finally falls at a distance x from the point of projection where x is equal to-
1.
2.
3.
4. 3R
Let and be moments of inertia of a body about two axes, A and B, respectively. The axis A passes through the centre of mass of the body, but B does not. Which of the following is correct?
1. <
2. If <, the axes are parallel.
3. If the axes are parallel, <
4. If the axes are not parallel,
A homogeneous cubical brick lies motionless on a rough inclined surface. The half of the brick which applies greater pressure on the plane is
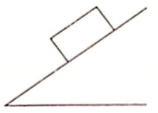
1. left half
2. right half
3. both apply equal pressure
4. the answer depends on coefficient of friction
The linear speed of a uniform spherical shell after rolling down an inclined plane of vertical height h from rest is
1.
2.
3.
4.
A satellite of the earth is revolving in circular orbit with a uniform velocity V. If the gravitational force suddenly disappears, the satellite will
1. continue to move with the same velocity in the same orbit
2. move tangentially to the original orbit with velocity V
3. fall down with increasing velocity
4. come to a stop somewhere in its original orbit
A man grows into a giant such that his linear dimensions increase by a factor of \(9.\) Assuming that his density remains the same, the stress in the leg will change by a factor of:
1. \(\dfrac{1}{81}\)
2. \(9\)
3. \(\dfrac{1}{9}\)
4. \(81\)
A tank has an orifice near its bottom. The volume of the liquid flowing per second out of the orifice does not depend upon
1. Area of the orifice
2. Height of the liquid level above the orifice
3. Density of liquid
4. Acceleration due to gravity
The density of a material A is 1500 kg/ and that of another material B is 2000 kg/. It is found that the heat capacity of 8 volumes of A is equal to heat the capacity of 12 volumes of B. The ratio of specific heats of A and B will be
1. 1:2
2. 3:1
3. 3:2
4. 2:1
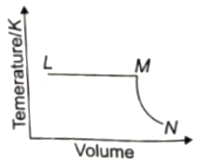
A fixed mass of ideal gas undergoes changes of pressure and volume starting at L, as shown in figure.
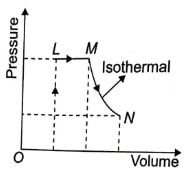
Which of the following is correct?
1.
2. 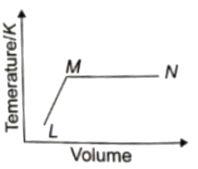
3. 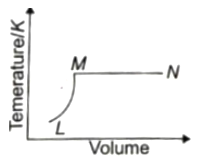
4. 
1 kg of gas does 20 kJ of work and receives 16 kJ of heat when it is expanded between two states. The second kind of expansion can be found between the same initial and final states, which requires a heat input of 9 kJ. The work done by the gas in the second expansion will be:
| 1. | 32 kJ | 2. | 5 kJ |
| 3. | -4 kJ | 4. | 13 kJ |
A closed container is fully insulated from outside. One half of it is filled with an ideal gas X separated by a plate P from the other half Y which contains a vacuum as shown in the figure. When P is removed, X moves into Y. Which of the following statement is correct?
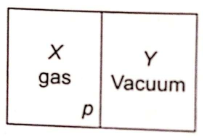
1. No work is done by X
2. X decreases in temperature
3. X increases in internal energy
4. X doubles in pressure.
The maximum displacement of a particle executing SHM is 1 cm and the maximum acceleration is . Then the time period is
1. 0.25 s
2. 4.00 s
3. 1.57 s
4. s
Two trains move towards each other at the same speed. The speed of sound is 340 . If the pitch of the whistle of one when heard on the other becomes 9/8 times, then the speed of each train is -

1. 2
2. 40
3. 20
4. 100
A tuning fork of frequency 280 Hz produces 10 beats/s when sounded with a vibrating sonometer string. When the tension in the string increases slightly, it produces 11 beats/s. The original frequency of the vibrating sonometer string is
1. 269 Hz
2. 291 Hz
3. 270 Hz
4. 290 Hz
When a negative charge is released and moves in the electric field, it moves towards a position of:
| 1. | lower electric potential and lower potential energy. |
| 2. | lower electric potential and higher potential energy. |
| 3. | higher electric potential and lower potential energy. |
| 4. | higher electric potential and higher potential energy. |
A charged particle q is placed at the centre O of the cube(ABCDEFGH) of length L . Another same charge q is placed at a distance L from O. Then the electric flux through BCFG is-
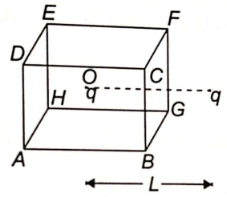
1.
2. zero
3.
4. None
A point charge q = 50C is located in the x-y plane at the point whose position vector is. What is the electric field at the point whose position vector is ?
1.
2.
3.
4.
The terminal voltage across a battery of emf E can be
1. 0
2. >E
3. <E
4. All of above
If the reading of ammeter in figure is 0.75 A, the reading of ammeter will be: [Assume ideal ammeters]
1. 1.5 A
2. 3 A
3. 4.5 A
4. 6 A
What is the equivalent capacitance of the system of capacitors between A and B?
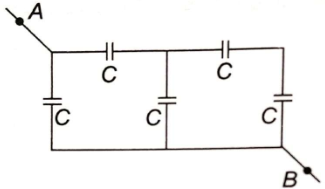
1.
2. 1.6 C
3. C
4. none
A current of i Ampere is flowing through each of the bent wires as shown. The magnitude of the magnetic field at O is-
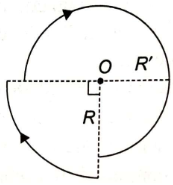
1.
2.
3.
4.
The direction of the magnetic force on the electron as shown in the diagram is along-
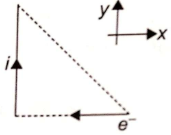
1. y-axis
2. z-axis
3. -y-axis
4. -z-axis
The variation of the intensity of magnetization (I) with respect to the magnetizing field (H) in a diamagnetic substance is described by which graph in the figure?
1. OD
2. OC
3. OB
4. OA
The number of turns in a coil of wire of fixed radius & length is \(600\) and its self-inductance is \(108\) mH. The self-inductance of a coil of \(500\) turns will be:
1. \(74\) mH
2. \(75\) mH
3. \(76\) mH
4. \(77\) mH
A resistance of 50 , an inductance of 20/ H and a capacitor of 5/ F are connected in series with an AC source of 230 V and 50 Hz. The impedance of circuit is-
1. 5
2. 50
3. 5 K
4. 500
| 1. | wavelength is \(2\) times and frequency becomes half. |
| 2. | wavelength is half and frequency remains unchanged. |
| 3. | wavelength and frequency both remain unchanged. |
| 4. | None of the above. |
A plano-convex lens has a curved surface of a radius of 100 cm. If = 1.5, then the focal length of the lens is:
1. 50 cm
2. 100 cm
3. 200 cm
4. 500 cm
A prism is made up of a material of refractive index . The angle of the prism is A. If the angle of minimum deviation is equal to the angle of the prism, then the value of A is:
1. 30
2. 45
3. 60
4. 75
A light of wavelength \(\lambda\) in air enters a medium of refractive index \(\mu.\) Two points in this medium lying along the path of this light are at a distance \(x\) apart. The phase difference between these points is:
1. \(\dfrac{2 \pi \mu x}{\lambda}\)
2. \(\dfrac{2 \pi x}{\mu \lambda}\)
3. \(\dfrac{2 \pi \left(\mu - 1\right) x}{\lambda}\)
4. \(\dfrac{2 \pi x}{\left(\lambda - 1\right) \lambda}\)
Two polaroids are kept crossed to each other. Now one of them is rotated through an angle of \(45^{\circ}\)
1. \(15\%\)
2. \(25\%\)
3. \(50\%\)
4. \(60\%\)
Two radiations containing photons of energies twice and five times the work function of metal are incident successively on the metal surface. The ratio of the maximum velocity of photoelectrons emitted in the two cases will be-
1. 1:2
2. 2:1
3. 1:4
4. 4:1
The following nuclear reaction is an example of-
1, fission
2. fusion
3. alpha decay
4. beta decay
The activity of a radioactive substance is at time and at time . Then the ratio is:
1.
2.
3.
4.
Which of the following statements is correct?
1. The depletion region of P-N junction diode increases with forward biasing
2. The depletion region of P-N junction diode decreases with reverse biasing
3. The depletion region of P-N junction diode does not change with biasing
4. The depletion region of P-N junction diode decreases with forward biasing
In the given transistor circuit, the base current is 35 A. The value of is
1. 100 k
2. 200 k
3. 300 k
4. 400 k