Read the following four statements (A-D):
| A. | In transcription, adenosine pairs with uracil |
| B. | Regulation of lac operon by a repressor is referred to as positive regulation |
| C. | The human genome has approximately 50,000 genes |
| D. | Haemophilia is a sex-linked recessive disease |
How many of the above statements are right?
1. Three
2. Four
3. One
4. Two
How many organisms in the list given below are autotrophs?
| Lactobacillus, Nostoc, Chara, Nitrosomonas, Nitrobacter, Streptomyces, Saccharomyces, Trypanosoma, Porphyra, Wolfia |
1. Five
2. Six
3. Three
4. Four
How many plants in the list given below have marginal placentation?
Mustard, Gram, Tulip, Asparagus, Arhar, Sun hemp, Chilli, Colchicine, Onion, Moong, Pea, Tobacco, Lupin
| 1. | Five | 2. | Six |
| 3. | Three | 4. | Four |
Compared to a dicot root, a monocot root has:
1. Many xylem bundles
2. Inconspicuous annual rings
3. Relatively thicker periderm
4. More abundant secondary xylem
A test cross is carried out to:
| 1. | Predict whether two traits are linked |
| 2. | Assess the number of alleles of a gene |
| 3. | Determine whether two species or varieties will breed successfully |
| 4. | Determine the genotype of a plant at F2 |
Which one of the following categories of animals, is correctly described with no single exception in it?
| 1. | All bony fishes have four pairs of gills and an operculum on each side |
| 2. | All sponges are marine and have collared cells |
| 3. | All mammals are viviparous and possess a diaphragm for breathing |
| 4. | All reptiles possess scales, have a three-chambered heart, and are cold blood (poikilothermal) |
The rate of formation of new organic matter by rabbits in grassland is called:
1. Secondary productivity
2. Net primary productivity
3. Gross primary productivity
4. Net productivity
In genetic engineering, the antibiotic-resistance gene is used:
1. To select healthy vectors
2. As sequences from where replication starts
3. To keep the cultures free of infection
4. As selectable markers
The secretory phase in the human menstrual cycle is also called:
| 1. | Follicular phase lasting for about 6 days |
| 2. | Luteal phase and lasts for about 13 days |
| 3. | Follicular phase and lasts for about 13 days |
| 4. | Luteal phase and lasts for about 6 days |
In gobar gas, the maximum amount is that of:
1. Methane
2. Propane
3. Carbon dioxide
4. Butane
Through their effect on plant growth regulators, what do the temperature and light control in the plants?
1. Flowering
2. Closure of stomata
3. Fruit elongation
4. Apical dominance
Which one of the following human organs is often called the "graveyard" of RBCs?
1. Kidney
2. Spleen
3. Liver
4. Gall bladder
Which one of the following pairs of animals is similar to each other pertaining to the feature stated against them?
| 1. | Garden lizard and Crocodile - Three-chambered heart |
| 2. | Ascaris and Ancylostoma - Metameric segmentation |
| 3. | Sea horse and Flying fish - Cold-blooded (poikilothermal) |
| 4. | Pteropus and Ornithorhyncus - Viviparity |
The idea of mutations was brought forth by:
| 1. | Gregor Mendel, who worked on Pisum sativum |
| 2. | Hardy Weinberg, who worked on allele frequencies in a population |
| 3. | Charles Darwin, who observed a wide variety of organisms during sea voyages |
| 4. | Hugo do Vries, who worked on evening primrose |
Select the correct statement about biodiversity:
| 1. | Large-scale planting of Bt cotton has no adverse effect on biodiversity |
| 2. | Western Ghats have a very high degree of species richness and endemism |
| 3. | Conservation of biodiversity is just a fad pursued by developed countries |
| 4. | The desert areas of Rajasthan and Gujarat have a very high level of desert animal species as well as numerous rare animals |
Plants with ovaries having only one or a few ovules are generally pollinated by:
1. Butterflies
2. Birds
3. Wind
4. Bees
Consider the following four statements (I-IV) and select the option which includes all the correct ones only:
| I. | Single-cell Spirulina can produce large quantities of food rich in protein, minerals, vitamins, etc. |
| II. | Body weight-wise, the microorganism Methylophilus methylotrophus may be able to produce several times more proteins than that produced by a cow per day through its milk. |
| III. | Common button mushrooms are a very rich source of vitamin C. |
| IV. | A rice variety has been developed which is very rich in calcium. |
1. Statements (I), (III), and (IV)
2. Statements (II), (III), and (IV)
3. Statements (I), (II)
4. Statements (III), (IV)
Which one of the following biomolecules is correctly characterised?
| 1. | Palmitic acid - an unsaturated fatty acid with 18 carbon atoms |
| 2. | Adenylic acid - adenosine with a glucose phosphate molecule |
| 3. | Alanine amino acid - Contains an amino group and an acidic group anywhere in the molecule |
| 4. | Lecithin - a phosphorylated glyceride found in the cell membrane |
For its action, nitrogenase requires:
1. Light
2. Mn2+
3. Super oxygen radicals
4. High input of energy
Tobacco plants resistant to a nematode have been developed by the introduction of DNA that produced (in the host cells):
1. A particular hormone
2. An antifeedant
3. A toxic protein
4. Both sense and anti-sense RNA
Where do certain symbiotic microorganisms typically occur in the human body?
1. Oral lining and tongue surface
2. Vermiform appendix and rectum
3. Duodenum
4. Caecum
Identify the meiotic stage in which the homologous chromosomes separate while the sister chromatids remain associated at their centromeres.
1. Metaphase-II
2. Anaphase-I
3. Anaphase-II
4. Metaphase-I
Which one of the following cellular parts is correctly described?
| 1. | Centrioles - sites for active RNA synthesis |
| 2. | Ribosomes - those on chloroplasts are larger (80s) while those in the cytoplasm are smaller (70s) |
| 3. | Lysosomes - optimally active at a pH of about 8.5 |
| 4. | Thylakoids - flattened membranous sacs forming the grana of chloroplasts |
Cuscuta is an example of:
1. Brood parasitism
2. Predation
3. Endoparasitism
4. Ectoparasitism
| 1. | Areolar tissue | 2. | Bone |
| 3. | Cartilage | 4. | Ligament |
Given are the following five statements (I - V) for your consideration:
| I: | In Equisetum, the female gametophyte is retained on the parent sporophyte |
| II: | In Ginkgo, the male gametophyte is not independent |
| III: | The sporophyte in Marchantia is more developed than that in Polytrichum |
| IV: | Sexual reproduction in Volvox is isogamous |
| V: | The spores of slime moulds lack cell walls |
How many of the above statements is/are correct?
| 1. | Three | 2. | Four |
| 3. | One | 4. | Two |
Identify the molecules (a) and (b) shown below and select the right option giving their source and use.
| (I) |  |
| (II) |  |
|
Molecule |
Source |
Use |
|
|
1. |
(II) Heroin |
Cannabis sativa |
Depressant and slows down body functions |
|
2. |
(II) Cannabinoid |
Atropa belladonna |
Produces hallucination |
|
3. |
(I) Morphine |
Papaver somniferum |
Sedative and painkiller |
|
4. |
(I) Cocaine |
Erythroxylum coca |
Accelerates the transport of dopamine |
The figure below shows three steps (A, B, C) of Polymerase Chain Reaction (PCR). Select the option giving correct identification together with what it represents.
| 1. | A - Denaturation at a temperature of about 50ºC |
| 2. | C - Extension in the presence of heat-stable DNA polymerase |
| 3. | A - Annealing with two sets of primers |
| 4. | B - Denaturation at a temperature of about 98ºC separating the two DNA strands |
Identify the likely organisms (a), (b), (c), and (d) in the food web shown below:
|
(a) |
(b) |
(c) |
(d) |
|
|
1. |
Dog |
Squirrel |
Bat |
Deer |
|
2. |
Rat |
Dog |
Tortoise |
Crow |
|
3. |
Squirrel |
Cat |
Rat |
Pigeon |
|
4. |
Deer |
Rabbit |
Frog |
Rat |
Which one of the following pairs of chemical substances, is correctly categorised?
| 1. | Pepsin and prolactin - Two digestive enzymes secreted in the stomach |
| 2. | Troponin and myosin - Complex proteins in striated muscles |
| 3. | Secretin and rhodopsin - Polypeptide hormones |
| 4. | Calcitonin and thymosin - Thyroid hormones |
Vernalisation stimulates flowering in:
1. Turmeric
2. Carrot
3. Ginger
4. Zaminkand
The green revolution in India occurred during:
1. 1970s
2. 1980s
3. 1950s
4. 1960s
A fall in glomerular filtration rate (GFR) activates:
1. Adrenal cortex to release aldosterone
2. Adrenal medulla to release adrenaline
3. Posterior pituitary to release vasopressin
4. Juxta glomerular cells, to release renin
What is the function of germ pores?
1. Absorption of water for seed germination
2. Initiation of pollen tube
3. Release of male gametes
4. The emergence of radicle
Which one of the following options gives the correct categorization of six animals according to the type of nitrogenous wastes (A, B, C) they give out?
| A AMMONOTELIC |
B UREOTELIC |
C URICOTELIC |
|
| 1. | Frog, Lizards | Aquatic Amphibia, Humans | Cockroach, Pigeon |
| 2. | Aquatic Amphibia | Frog, humans | Pigeon, Lizards, Cockroach |
| 3. | Aquatic Amphibia | Cockroach, Humans | Frog, Pigeon, Lizards |
| 4. | Pigeon, Humans | Aquatic Amphibia, Lizards | Cockroach, Frog |
| Items | Category | Exception | |
| 1. | Kangaroo, Koala, wombat |
Australian marsupials | Wombat |
| 2. | Plasmodium, Cuscuta, Trypanosoma |
Protozoan parasites | Cuscuta |
| 3. | Typhoid, Pneumonia, Diphtheria |
Bacterial diseases | Diphtheria |
| 4. | UAA, UAG, UGA | Stop codons | UAG |
Which one of the following generally acts as an antagonist to Gibberellins?
1. Ethylene
2. ABA
3. IAA
4. Zeatin
Which one of the following organisms is scientifically correctly named, correctly printed according to the International Rules of Nomenclature, and correctly described?
| 1. | Plasmodium falciparum – a protozoan pathogen causing the most serious type of malaria |
| 2. | Felis tigris – The Indian tiger, well protected in Gir forests. |
| 3. | E.Coli – Full name Entamoeba coli, a commonly occurring bacterium in the human intestine |
| 4. | Musca domestica – The common house lizard, a reptile |
Read the following four statements (I-IV):
| I. | Colostrum is recommended for the newborn because it is rich in antigen |
| II. | Chikungunya is caused by a Gram-negative bacterium |
| III. | Tissue culture has proved useful in obtaining virus-free plants |
| IV. | Beer is manufactured by distillation of fermented grape juice |
How many of the above statements are wrong?
1. Three
2. Four
3. One
4. Two
Which one of the following organisms is correctly matched with its three characteristics?
| 1. | Tomato: Twisted aestivation, Axile placentation, Berry |
| 2. | Onion: Bulb, Imbricate aestivation, Axile placentation |
| 3. | Maize: C 3 pathway, Closed vascular bundles, Scutellum |
| 4. | Pea: C 3 pathway, Endospermic seed, Vaxillary aestivation |
The second stage of hydrosere is occupied by plants like:
1. Typha
2. Salix
3. Vallisneria
4. Azolla
Which one of the following statements is correct with respect to immunity?
| 1. | The antibodies against smallpox pathogens are produced by T-lymphocytes |
| 2. | Antibodies are protein molecules each of which has four light chains |
| 3. | Rejection of a kidney graft is the function of B-lymphocytes |
| 4. | Preformed antibodies need to be injected to treat the bite by a viper snake |
Which one of the following represents a palindromic sequence in DNA?
| 1. | 5'-CCAATG-3' 3'-GAATCC-5' |
2. | 5'-CATTAG-3' 3'-GATAAC-5' |
| 3. | 5'-GATACC-3' 3'-CCTAAG-5' |
4. | 5'-GAATTC-3' 3'-CTTAAG-5' |
For its activity, carboxypeptidase requires:
1. Iron
2. Niacin
3. Copper
4. Zinc
Given below is the diagrammatic sketch of a certain type of connective tissue. Identify the parts labeled A, B, C, and D and select the right option for them:

| Part-A | Part-B | Part-C | Part-D | |
| 1. | Mast cell | Macrophage | Fibroblast | Collagen fibres |
| 2. | Macrophage | Collagen fibres | Fibroblast | Mast cell |
| 3. | Mast cell | Collagen fibres | Fibroblast | Macrophage |
| 4. | Macrophage | Fibroblast | Collagen fibres | Mast cell |
In the five-kingdom classification, Chlamydomonas and Chlorella have been included in:
| 1. | Algae | 2. | Plantae |
| 3. | Monera | 4. | Protista |
Read the following four statements (I-IV):
| I. | Both, photophosphorylation and oxidative phosphorylation involve uphill transport of protons across the membrane |
| II. | In dicot stems, a new cambium originates from cells of pericycle at the time of secondary growth |
| III. | Stamens in flowers of Gloriosa and Petunia are polyandrous |
| IV. | Symbiotic nitrogen-fixers occur in a free-living state and also in soil |
How many of the above statements are right?
1. Three
2. Four
3. One
4. Two
Domestic sewage in large cities:
| 1. | is processed by aerobic and then anaerobic bacteria in the secondary treatment in Sewage Treatment Plant (STPs) |
| 2. | when treated in STPs, does not require the aeration step as the sewage contains adequate oxygen |
| 3. | has very high amounts of suspended solids and dissolved salts |
| 4. | has a high BOD as it contains both aerobic and anaerobic bacteria |
| 1. | Salvinia - Prothallus | 2. | Viroids – RNA |
| 3. | Mustard-Synergids | 4. | Ginkgo-Archegonia |
What is it that forms the basis of DNA Fingerprinting?
| 1. | The relative difference in the DNA occurrence in blood, skin and saliva |
| 2. | The relative amount of DNA in the ridges and grooves of the fingerprints |
| 3. | Satellite DNA occurs as highly repeated short DNA segments |
| 4. | The relative proportions of purines and pyrimidines in DNA |
Which one of the following characteristics is common in both humans and adult frogs?
1. Internal fertilization
2. Nucleated RBCs
3. Ureotelic mode of excretion
4. Four-chambered heart
Represented below is the inheritance pattern of a certain type of traits in humans. Which one of the following conditions could be an example of this pattern?
| 1. | Sickle cell anemia | 2. | Haemophilia |  |
| 3. | Thalassemia | 4. | Phenylketonuria |
The four sketches (A, B, C, and D) given below, represent four different types of animal tissues. Which one of these is correctly identified in the options given, along with its correct location and function?
| Tissue | Location | Function | ||
| 1. | (C) | Collagen fibres | Cartilage | Attach skeletal muscles to bones |
| 2. | (D) | Smooth muscle tissue | Heart | Heart contraction |
| 3. | (A) | Columnar epithelium | Nephron | Secretion and absorption |
| 4. | (B) | Glandular epithelium | Intestine | Secretion |
The first clinical gene therapy was given for treating:
1. Chickenpox
2. Rheumatoid arthritis
3. Adenosine deaminase deficiency
4. Diabetes mellitus
Sacred groves are especially useful in:
1. preventing soil erosion
2. year-round flow of water in rivers
3. conserving rare and threatened species
4. generating environmental awareness
Which one of the following is a wrong statement regarding mutations?
| 1. | Cancer cells commonly show chromosomal aberrations |
| 2. | UV and Gamma rays are mutagens |
| 3. | Change in a single base pair of DNA does not cause mutation |
| 4. | Deletion and insertion of base pairs cause frame-shift mutations |
Biolistics (gene-gun) is suitable for:
| 1. | Transformation of plant cells |
| 2. | Constructing recombinant DNA by joining with vectors |
| 3. | DNA fingerprinting |
| 4. | Disarming pathogen vectors |
Which one of the following statements is wrong?
| 1. | Vegetative cell is larger than generative cell |
| 2. | Pollen grains in some plants remain viable for months |
| 3. | Intine is made up of cellulose and pectin |
| 4. | When pollen is shed at a two-celled stage, double fertilization does not take place |
Identify the human development stage shown below as well as the related right place of its occurrence in a normal pregnant woman, and select the right option for the two together:

| Developmental stage | Site of occurrence | |
| 1. | Blastula | End part of fallopian tube |
| 2. | Blastocyst | Uterine wall |
| 3. | 8-celled morula | Starting point of fallopian tube |
| 4. | Late morula | Middle part of fallopian tube |
A red precipitate is obtained when an ethanol solution of dimethylglyoxime is added to ammoniacal Ni(II).
Which of the following statements is not true?
1. Complex has symmetrical H-bonding.
2. Red complex has a tetrahedral geometry.
3. Dimethylglyoxime functions as a bidentate ligand.
4. Red complex has a square planar geometry.
Which orbital does the additional electron occupy when O₂ is converted to the O₂⁻ ion?
| 1. | 2. | ||
| 3. | 4. |
Consider the reaction:
What sort of reaction is it?
1. Free radical addition – elimination reaction
2. Electrophilic substitution-elimination reaction
3. Nucleophilic addition – elimination reaction
4. Electrophilic addition – elimination reaction
The given sequence among the following arrangements that is not strictly according to the property indicated against it.
| 1. | H2O < H2S < H2Se < H2Te : \(\small{\text{increasing} \ p K_{a}\ \text{values}}\) |
| 2. | \(\small{\mathrm{N H_{3} < P H_{3} < A s H_{3} < S b H_{3}} : \text{increasing acidic character}}\) |
| 3. | \(\small{\mathrm{C O_{} < S i O_{} < S n O_{} < P b O_{2}} : \text{increasing oxidising power}} \) |
| 4. | \(\small{\mathrm{H F < H C l < H B r < H I} : \text{increasing acidic strength}} \) |
The Gibb's energy for the decomposition of \(\mathrm{A l_{2} O_{3}}\) at \(\mathrm{500~ ^\circ C}\) is as follows:
2/3Al2O3 → 4/3Al + O2 ; ∆rG = + 960 k J mol–1
The potential difference needed for the electrolytic reduction of aluminium oxide (Al2O3) at \(\mathrm{500~ ^\circ C}\) is at least,
1. 3.0 V
2. 2.5 V
3. 5.0 V
4. 4.5 V
Given that the equilibrium constant for the reaction
has a value of 278 at a particular temperature, the value of the equilibrium constant for the following reaction at the same temperature will be:
1.
2.
3.
4.
The compound that is used as antifreeze in automobile radiators is:
| 1. | Glycol | 2. | Nitrophenol |
| 3. | Ethyl alcohol | 4. | Methyl alcohol |
Molar conductivities at infinite dilution of
NaCl, HCl, and are 126.4, 425.9, and 91.0 S cm2 mol–1 respectively.
for will be:
| 1. | \(180.5~S~cm^2~mol^{-1}\) | 2. | \(290.8~S~cm^2~mol^{-1}\) |
| 3. | \(390.5~S~cm^2~mol^{-1}\) | 4. | \(425.5~S~cm^2~mol^{-1}\) |
Vapour pressure of chloroform \(\mathrm{(CHCl_3)}\) and dichloromethane \(\mathrm{(CH_2Cl_2)}\) at are 200 mmHg and 41.5 mmHg respectively. Vapour pressure of the solution was obtained by mixing 25.5 g of \(\mathrm{(CHCl_3)}\) and 40 g of \(\mathrm{(CH_2Cl_2)}\) at the same temperature will be: (Molecular mass of \(\mathrm{(CHCl_3)}\) = 119.5 u and molecular mass of \(\mathrm{(CH_2Cl_2)}\) = 85 u)
| 1. | 90.40 mm Hg | 2. | 119.5 mm Hg |
| 3. | 75 mm Hg | 4. | 173.9 mm Hg |
A certain gas takes three times as long to effuse out as helium. Its molecular mass will be:
1. 36 u
2. 64 u
3. 9 u
4. 27 u
The set of monomers that forms a biodegradable polymer is:
1. H2N-CH2-COOH and H2N-(CH2)5-COOH
2. HO-CH2-CH2-OH and HOOC-C6H4-COOH
3. C6H5-CH=CH2 and CH2=CH-CH=CH2
4. CH2=CH-CN and CH2=CH-CH=CH2
The catalytic activity of transition metals and their compounds is ascribed mainly to:
1. their unfilled d-orbitals
2. their ability to adopt variable oxidation states
3. their chemical reactivity
4. their magnetic behaviour
Consider the following reaction:
A2(g) + B2(g) ⇋ 2AB(g)
At equilibrium, the concentrations of A2 = 3.0×10–3 M; B2 = 4.2×10–3 M and AB = 2.8×10–3M.
The value \(K_C\) for the above-given reaction in a sealed container at 527°C is:
| 1. | 3.9 | 2. | 0.6 |
| 3. | 4.5 | 4. | 2.0 |
Standard reduction potentials of the half-reactions are given below:
The strongest oxidizing and reducing agents, respectively, are:
1. and
2. and
3. and
4. and
Four diatomic species are listed below. Identify the correct order in which the bond order is increasing in them.
1. O2– < NO < C22– < He2+
2. C22– < He2+ < O2– < NO
3. He2+ < O2– < NO < C22–
4. NO < O2– < C22– < He2+
A low spin complex of -cation in an octahedral field will have the following energy:
1.
2.
3.
4.
( = Crystal field splitting energy in an octahedral field, P = Electron pairing energy)
A yellow precipitate with iodine and alkali is given by:
(i) Methyl acetate
(ii) Acetamide
(iii) 2-Hydroxypropane
(iv) Acetophenone
| 1. | (i) and (ii) | 2. | (ii) and (iii) |
| 3. | (iii) and (iv) | 4. | (iv) and (i) |
The orbital angular momentum of a p-electron is given as:
| 1. | \(\sqrt{3}~ \frac{h}{2 \pi} \) | 2. | \(\sqrt{\frac{3}{2}} \frac{h}{\pi} \) |
| 3. | \(\sqrt{6}~ \sqrt{\frac{h}{2 \pi}} \) | 4. | \(\frac{h}{\sqrt{2 }\pi}\) |
Which one of the following does not correctly represent the correct order of the property indicated against it?
| 1. | Ti3+< V3+< Cr3+< Mn3+: increasing magnetic moment |
| 2. | Ti < V < Cr < Mn : increasing melting point |
| 3. | Ti < V < Mn < Cr : increasing 2nd ionization enthalpy |
| 4. | Ti < V < Cr < Mn : increasing number of oxidation states |
Chloramphenicol is an:
1. antihistaminic
2. antiseptic and disinfectant
3. antibiotic-broad spectrum
4. antifertility drug
Consider the following reaction:
The product 'A' is:
1.
2.
3.
4.
The reagent that can be used to distinguish between 1-butyne and 2-butyne is:
1. \(\mathrm{HCl}\)
2. \(\mathrm{O_{2}}\)
3. \(\mathrm{B r_{2}}\)
4. \(\mathrm{N a N H_{2}}\)
For real gases van der Waals equation is written as
where 'a' and 'b' are van der Waals constants.
Two sets of gases are:
(I) (II)
The gases given in set-I are in increasing order of 'b' and gases given in set-II are in decreasing order of 'a'. Select the correct order from the following:
1.
2.
3.
4.
Activation energy and rate constant (k1 and k2) of a chemical reaction at two different temperatures (T1 and T2) are related by:
| 1. | \(\ln \frac{k_2}{k_1}=-\frac{E_a}{R}\left(\frac{1}{T_2}-\frac{1}{T_1}\right)\) |
| 2. | \(\ln \frac{k_2}{k_1}=-\frac{E_a}{R}\left(\frac{1}{T_2}+\frac{1}{T_1}\right)\) |
| 3. | \(\ln \frac{k_2}{k_1}=\frac{E_a}{R}\left(\frac{1}{T_2}-\frac{1}{T_1}\right)\) |
| 4. | \(\ln \frac{k_2}{k_1}=-\frac{E_a}{R}\left(\frac{1}{T_1}-\frac{1}{T_2}\right)\) |
Which of the following exhibits only a +3 oxidation state?
| 1. | Th | 2. | Ac |
| 3. | Pa | 4. | U |
Equal volumes of two monoatomic gases, A and B, at same temperature and pressure are mixed. The ratio of specific heats (Cp/Cv) of the mixture will be:
1. 1.50
2. 3.3
3. 1.67
4. 0.83
Structure of a mixed oxide is cubic close-packed (CCP). The cubic unit cell of mixed oxide is composed of oxide ions. One-fourth of the tetrahedral voids are occupied by divalent metal A and the octahedral voids are occupied by a monovalent metal B. The formula of the oxide is:
1. A2BO2
2. A2B3O4
3. AB2O2
4. ABO2
Four successive members of the first series of the transition metals are listed below. For which one of them does the standard potential value have a positive sign?
1. Ni (Z = 28)
2. Cu (Z = 29)
3. Fe (Z = 26)
4. Co (Z = 27)
In replacement reaction
The reaction will be most favorable if M happens to be:
1. K
2. Rb
3. Li
4. Na
An organic compound () (A), when treated with nitrous acid, gave an alcohol, and N2 gas was evolved. (A) on warming with CHCl3 and caustic potash gave (C) which on reduction gave isopropylmethylamine. The structure of (A) is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. | \( \frac{\sqrt{3}}{4} \) | 2. | \( \frac{1}{2} \) |
| 3. | \( \frac{1}{8} \) | 4. | \( \frac{1}{4}\) |
The power dissipated in the circuit shown in the figure is \(30~\text{Watts}\). The value of \(R\) is:
1. \(15~\Omega\)
2. \(10~\Omega\)
3. \(30~\Omega\)
4. \(20~\Omega\)
The dimensions of are:
1.
2.
3.
4.
An ideal gas goes from state \(A\) to state \(B\) via three different processes as indicated in the \((P\text-V)\) diagram.
If \(Q_1,Q_2,Q_3\) indicate the heat absorbed by the gas along the three processes and \(\Delta U_1, \Delta U_2, \Delta U_3\) indicate the change in internal energy along the three processes respectively, then:
| 1. | \(Q_3>Q_2>Q_1\) and \(\Delta U_1= \Delta U_2= \Delta U_3\) |
| 2. | \(Q_1=Q_2=Q_3\) and \(\Delta U_1> \Delta U_2> \Delta U_3\) |
| 3. | \(Q_3>Q_2>Q_1\) and \(\Delta U_1> \Delta U_2> \Delta U_3\) |
| 4. | \(Q_1>Q_2>Q_3\) and \(\Delta U_1= \Delta U_2= \Delta U_3\) |
To get output \(Y = 1\) in the given circuit which of the following input will be correct?
| \(A\) | \(B\) | \(C\) | |
| 1. | 1 | 0 | 1 |
| 2. | 1 | 1 | 0 |
| 3. | 0 | 1 | 0 |
| 4. | 1 | 0 | 0 |
Two metallic spheres of radii \(1~\text{cm}\) and \(3~\text{cm}\) are given charges of \(-1\times 10^{-2}~\text{C}\) and \(5\times 10^{-2} ~\text{C}\), respectively. If these are connected by a conducting wire, then the final charge on the bigger sphere is:
| 1. | \(3\times 10^{-2}~ \text{C}\) | 2. | \(4\times 10^{-2}~\text{C}\) |
| 3. | \(1\times 10^{-2}~\text{C}\) | 4. | \(2\times 10^{-2}~\text{C}\) |
Two radiations of photons energies \(1\) eV and \(2.5\) eV, successively illuminate a photosensitive metallic surface of work function \(0.5\) eV. The ratio of the maximum speeds of the emitted electrons is:
1. \(1:2\)
2. \(1:1\)
3. \(1:5\)
4. \(1:4\)
The moment of inertia of a uniform circular disc is maximum about an axis perpendicular to the disc and passing through:
1. \(C\)
2. \(D\)
3. \(A\)
4. \(B\)
A train moving at a speed of towards a stationary object, emits a sound of frequency 1000 Hz. Some of the sound reaching the object gets reflected back to the train as an echo. The frequency of the echo as detected by the driver of the train is:(speed of sound in air is )
1. 4000 Hz
2. 5000 Hz
3. 3000 Hz
4. 3500 Hz
The half-life of a radioactive nucleus is 50 days. The time interval between the time when of it has decayed and the time when of it had decayed is:
1. 50 days
2. 60 days
3. 15 days
4. 30 days
A car of mass \(m\) is moving on a level circular track of radius \(R\). If \(\mu_s\) represents the static friction between the road and tyres of the car, the maximum speed of the car in circular motion is given by:
| 1. | \(\sqrt{\dfrac{Rg}{\mu_s} }\) | 2. | \(\sqrt{\dfrac{mRg}{\mu_s}}\) |
| 3. | \(\sqrt{\mu_s Rg}\) | 4. | \(\sqrt{\mu_s m Rg}\) |
A circular platform is mounted on a frictionless vertical axle. Its radius \(R = 2~\text{m}\) and its moment of inertia about the axle is \(200~\text{kg m}^2\). It is initially at rest. A \(50~\text{kg}\) man stands on the edge of the platform and begins to walk along the edge at the speed of \(1~\text{ms}^{-1}\) relative to the ground. The time taken by man to complete one revolution is:
| 1. | \(\dfrac{3\pi}{2}\text{s}\) | 2. | \(2\pi~\text{s}\) |
| 3. | \(\dfrac{\pi}{2}\text{s}\) | 4. | \(\pi~\text{s}\) |
If the momentum of an electron is changed by \(p,\) then the de-Broglie wavelength associated with it changes by \(0.5\%.\) The initial momentum of an electron will be:
| 1. | \(400p\) | 2. | \(\frac{p}{100}\) |
| 3. | \(100p\) | 4. | \(200p\) |
| 1. | \(v_o=v_e\) | 2. | \(v_e=\sqrt{2v_o}\) |
| 3. | \(v_e=\sqrt{2}~v_o\) | 4. | \(v_o=\sqrt{2}~v_e\) |
The equation of a simple harmonic wave is given by \(y=3\sin \frac{\pi}{2}(50t-x)\) where \(x \) and \(y\) are in meters and \(t\) is in seconds. The ratio of maximum particle velocity to the wave velocity is:
| 1. | \(\frac{3\pi}{2}\) | 2. | \(3\pi\) |
| 3. | \(\frac{2\pi}{3}\) | 4. | \(2\pi\) |
A proton carrying \(1~\text{MeV}\) kinetic energy is moving in a circular path of radius \(R\) in a uniform magnetic field. What should be the energy of an \(\alpha \text- \)particle to describe a circle of the same radius in the same field?
| 1. | \(1~\text{MeV}\) | 2. | \(0.5~\text{MeV}\) |
| 3. | \(4~\text{MeV}\) | 4. | \(2~\text{MeV}\) |
Three masses are placed on the \(x\)-axis: \(300~\text{g}\) at origin, \(500~\text{g}\) at \(x= 40~\text{cm}\) and \(400~\text{g}\) at \(x= 70~\text{cm}.\) The distance of the centre of mass from the origin is:
1. \(45~\text{cm}\)
2. \(50~\text{cm}\)
3. \(30~\text{cm}\)
4. \(40~\text{cm}\)
In a coil of resistance \(10\) \(\Omega\), the induced current developed by changing magnetic flux through it is shown in the figure as a function of time. The magnitude of change in flux through the coil in Weber is:
| 1. | \(2\) | 2. | \(6\) |
| 3. | \(4\) | 4. | \(8\) |
A parallel plate capacitor has a uniform electric field \(E\) in the space between the plates. If the distance between the plates is \(d\) and the area of each plate is \(A,\) the energy stored in the capacitor is:
| 1. | \(\dfrac{E^2 Ad}{\varepsilon_0}\) | 2. | \(\dfrac{1}{2}\varepsilon_0E^2 Ad\) |
| 3. | \(\varepsilon_0EAd\) | 4. | \(\dfrac{1}{2}\varepsilon_0E^2 \) |
A car of mass m starts from rest and accelerates so that the instantaneous power delivered to the car has a constant magnitude \(P_0\). The instantaneous velocity of this car is proportional to:
| 1. | \(t^{\frac{1}{2}}\) | 2. | \(t^{\frac{-1}{2}}\) |
| 3. | \(\frac{t}{\sqrt{m}}\) | 4. | \(t^2 P_0\) |
Which one of the following plots represents the variation of a gravitational field on a particle with distance \(r\) due to a thin spherical shell of radius \(R?\)
(\(r\) is measured from the centre of the spherical shell)
| 1. | 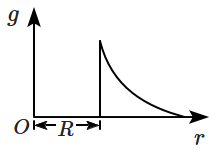 |
2. |  |
| 3. |  |
4. |  |
The input resistance of a silicon transistor is 100 Ω. If the base current is changed by 40 µA, it results in a change in collector current by 2 mA. This transistor is used as a common emitter amplifier with a load resistance of 4 kΩ. The voltage gain of the amplifier is:
1. 3000
2. 4000
3. 1000
4. 2000
For the angle of minimum deviation of a prism to be equal to its refracting angle, the prism must be made of a material whose refractive index:
| 1. | lies between \(2\) and \(\sqrt{2}\) |
| 2. | is less than \(1\) |
| 3. | is greater than \(2\) |
| 4. | lies between \(\sqrt{2}\) and \(1\) |
The transition from the state \(n=3\) to \(n=1\) in hydrogen-like atoms results in ultraviolet radiation. Infrared radiation will be obtained in the transition from:
1. \(3\rightarrow 2\)
2. \(4\rightarrow 2\)
3. \(4\rightarrow 3\)
4. \(2\rightarrow 1\)
A rod of length \(10~\text{cm}\) lies along the principal axis of a concave mirror of focal length \(10~\text{cm}\) in such a way that its end closer to the pole is \(20~\text{cm}\) away from the mirror. The length of the image is:
1. \(15~\text{cm}\)
2. \(2.5~\text{cm}\)
3. \(5~\text{cm}\)
4. \(10~\text{cm}\)
A slab of stone with an area \(0.36~\text{m}^{2}\) and thickness of \(0.1~\text{m}\) is exposed on the lower surface to steam at \(100^\circ\text{C}.\) A block of ice at \(0^{\circ}\text{C}\) rests on the upper surface of the slab. In one hour \(4.8~\text{kg}\) of ice is melted. The thermal conductivity of the slab will be:
(Given latent heat of fusion of ice \(= 3.36\times10^{5}~\text{JKg}^{-1}\))
1. \(1.29~\text{J/m/s/}^{\circ}\text{C}\)
2. \(2.05~\text{J/m/s/}^{\circ}\text{C}\)
3. \(1.02~\text{J/m/s/}^{\circ}\text{C}\)
4. \(1.24~\text{J/m/s/}^{\circ}\text{C}\)
A stone is dropped from a height \(h.\) It hits the ground with a certain momentum \(p.\) If the same stone is dropped from a height \(100\%\) more than the previous height, the momentum when it hits the ground will change by:
1. \(41\%\)
2. \(200\%\)
3. \(100\%\)
4. \(68\%\)
A cell having an emf \(\varepsilon\) and internal resistance \(r\) is connected across a variable external resistance \(R\). As the resistance \(R\) is increased, the plot of potential difference \(V\) across \(R\) is given by:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
A magnetic needle suspended parallel to a magnetic field requires \(\sqrt{3}~\text{J}\) of work to turn it through \(60^\circ\)
1. \(3\) N-m
2. \(\sqrt{3} \) N-m
3. \(\frac32\) N-m
4. \(2\sqrt{3}\) N-m
The ratio of the amplitude of a magnetic field to the amplitude of an electric field for an electromagnetic wave propagating in a vacuum is equal to:
| 1. | reciprocal of speed of light in vacuum. |
| 2. | the ratio of magnetic permeability to the electric susceptibility of vacuum. |
| 3. | unity. |
| 4. | the speed of light in a vacuum. |