The coefficient of area expansion \(\beta\) of a rectangular sheet of a solid in terms of the coefficient of linear expansion \(\alpha\) is:
1. \(2\alpha\)
2. \(\alpha\)
3. \(3\alpha\)
4. \(\alpha^2\)
When \(0.15\) kg of ice at \(0^\circ \text{C}\) is mixed with \(0.30\) kg of water at \(50^\circ \text{C}\) in a container, the resulting temperature is \(6.7^\circ \text{C}.\)
The heat of fusion of ice is: (\(S_{\text{water}}=4186\) J kg–1 K–1)
1. \( 3.43 \times 10^4\) Jkg–1
2. \( 3.34 \times 10^4\) Jkg–1
3. \( 3.34 \times 10^5\) Jkg–1
4. \(4.34 \times 10^5\) Jkg–1
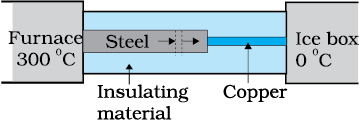
What is the temperature of the steel-copper junction in the steady-state of the system shown in the figure? The length of the steel rod = \(15.0\) cm, length of the copper rod = \(10.0\) cm, temperature of the furnace = \(300^{\circ}\text{C}\), temperature of the other end = \(0^{\circ}\text{C}\). The area of the cross section of the steel rod is twice that of the copper rod. (Thermal conductivity of steel = \(50.2 ~\text{J s}^{-1}\text{m}^{-1}\text{K}^{-1};\) and of copper \(= 385~\text{J s}^{-1}\text{m}^{-1}\text{K}^{-1})\).
A steel tape \(1\) m long is correctly calibrated for a temperature of \(27^\circ \text{C}\). The length of a steel rod measured by this tape is found to be \(63.0\) cm on a hot day when the temperature is \(45^\circ \text{C}\). What is the actual length of the steel rod on that day?
(Coefficient of linear expansion of steel = \(1.20\times 10^{-5}~\text{K}^{-1}\)).
1. \( 62.485 \) cm
2. \( 60.762 \) cm
3. \( 65.935 \) cm
4. \( 63.013\) cm
An iron bar
\(\left(L_{1}=0.1 \text{m} , A_{1}=0.02~\text{m}^{2} , K_{1}=79~\text{Wm}^{- 1} \text{K}^{-1}\right)\) and a brass bar
\(\left(L_{2}=0.1~\text{m} , A_{2}=0.02~\text{m}^{2} , K_{2}=109~\text{Wm}^{- 1} \text{K}^{- 1}\right)\) are soldered end to end as shown in the figure. The free ends of the iron bar and brass bar are maintained at \(373~ \text{K}\) and \(273~ \text{K}\) respectively. The temperature of the junction of the two bars is:

1. \(215~ \text{K}\)
2. \(315~ \text{K}\)
3. \(415~ \text{K}\)
4. \(115~ \text{K}\)
A large steel wheel is to be fitted onto a shaft of the same material. At 27 °C, the outer diameter of the shaft is 8.70 cm and the wheel's central hole has a diameter of 8.69 cm. The shaft is cooled using ‘dry ice’. At what temperature of the shaft does the wheel slip on the shaft?
(Assume the coefficient of linear expansion of the steel to be constant over the required temperature range and \(\alpha\) steel = \(1.20 \times 10^{-5} K^{-1}\))
2. -70°C
3. -69°C
4. -67°C
A hole is drilled in a copper sheet. The diameter of the hole is \(4.24~\text{cm}\) at \(27.0^\circ \text C.\) What is the change in the diameter of the hole when the sheet is heated to \(227^\circ \text C?\)
(the coefficient of linear expansion of copper \(\alpha=1.70\times 10^{-5}~\text K^{-1}\))
1. \(0.0144~\text{cm}\)
2. \(0.0234~\text{cm}\)
3. \(0.0123~\text{cm}\)
4. \(0.0111~\text{cm}\)
The coefficient of volume expansion of glycerine is \(49\times 10^{-5}~\text {K}^{-1}.\) What is the fractional change in its density for a \(30^\circ \text{C}\) rise in temperature?
1. \(1.44\times10^{-3}\)
2. \(1.57\times10^{-3}\)
3. \(1.57\times10^{-2}\)
4. \(1.44\times10^{-2}\)
A brass wire \(1.8~\text m\) long at \(27^\circ \text C\) is held taut with a little tension between two rigid supports. If the wire is cooled to a temperature of \(-39^\circ \text C,\) what is the tension created in the wire?
( Assume diameter of the wire to be \(2.0~\text{mm}\) , coefficient of linear expansion of brass \(=2.0 \times10^{-5}~\text{K}^{-1},\) Young's modulus of brass\(=0.91 \times10^{11}~\text{Pa}\) )
1. \(3.8 \times 10^3~\text N\)
2. \(3.8 \times 10^2~\text N\)
3. \(2.9 \times 10^{-2}~\text N\)
4. \(2.9 \times 10^{2}~\text N\)
A 10 kW drilling machine is used to drill a bore in a small aluminium block of mass 8.0 kg. How much is the rise in temperature of the block in 2.5 minutes?
(Assuming 50% of power is used up in heating the machine itself or lost to the surroundings? Specific heat of aluminium .)
1. 103°C
2. 109°C
3. 211°C
4. 197°C
In an experiment on the specific heat of a metal, a \(0.20~\text{kg}\) block of the metal at \(150^{\circ}\text{C}\) is dropped in a copper calorimeter (of water equivalent of \(0.025~\text{kg}\)) containing \(150~\text{cm}^{3}\) of water at \(27^{\circ}\text{C}.\) The final temperature is \(40^{\circ}\text{C}.\) The specific heat of the metal will be:
(the heat losses to the surroundings are negligible)
1. \(0 . 40 ~ \text{Jg}^{- 1} \text{K}^{- 1}\)
2. \(0 . 43 ~ \text{Jg}^{- 1} \text{K}^{- 1}\)
3. \(0 . 54 ~ \text{Jg}^{- 1} \text{K}^{- 1}\)
4. \(0 . 61 ~ \text{Jg}^{- 1} \text{K}^{- 1}\)
A body cools from \(80^\circ\text{C}\) to \(50^\circ\text{C}\) in \(5\) minutes. The time it takes to cool from \(60^\circ\text{C}\) to \(30^\circ\text{C}\) is:
(The temperature of the surroundings is \(20^\circ\text{C}\))
1. \(13~\text{min}\)
2. \(15~\text{min}\)
3. \(12~\text{min}\)
4. \(10~\text{min}\)

| (a) | The region \(AB\) represents ice and water in thermal equilibrium. |
| (b) | At \(B\) water starts boiling. |
| (c) | At \(C\) all the water gets converted into steam. |
| (d) | \(C\) to \(D\) represents water and steam in equilibrium at boiling point. |