Theory of spontaneous generation was rejected because:
| 1. | It explained origin of first life from non-living or inanimate matter and lacked experimental evidence. |
| 2. | It was based on the biogenesis concept. |
| 3. | It did not explain about the origin of first life on earth. |
| 4. | It proposed origin of life from outer space. |
Which of the following options is not correct regarding the early atmosphere?
(A) Oxygen was totally absent
(B) Atmosphere was reducing
(C) Temperature was high
(D) Presence of free molecules
(E) Presence of molecules like vapours
1. A, D and E
2. A and D
3. D and E
4. A only
A founder event favor microevolution in the founding population mainly because
| 1. | Mutations are more common in a head environment |
| 2. | A small founding population is subject to extensive sampling error in the composition of its gene pool |
| 3. | Gene flow increases |
| 4. | The low environment is likely to be patchy favouring diversifying selection |
If frequency of 'A' allele is 0.4 than, find out the frequency of 'B' allele and heterozygous genotype in a random mating population at equilibria.
1. 0.6 an 0.24
2. 0.6 and 0.96
3. 0.6 and 0.48
4. 0.6 and 0.50
The correct statement about the nature of evolution and natural selection is
| 1. | Evolution is a process while natural selection is the end result of a process which is unknown. |
| 2. | Natural selection is a process while evolution is the end result of a process which is unknown. |
| 3. | Both evolution and natural selection are the end results of a process which is unknown. |
| 4. | It is still unclear whether evolution and natural selection are process or end results of unknown processes. |
Select correct sequence about life cycle of Plasmodium:
| 1. | Sporozoites (human)RBCsLiver cellsGametocytes in bloodBlood meal (female mosquito)Multiply (female mosquito)Sporozoites (female mosquito) |
| 2. | Sporozoiters (human)Liver cellsRBCsGametocytes in bloodBlood meal (female mosquito)multiply (female mosquito)Sporozoites (female mosquito) |
| 3. | Gametocytes (female mosquito)Gametocytes (human)RBCsMultiplySporozoitesBlood meal (human)Sporozoites (female mosquito)Multiply (female mosquito)Gametocytes (female mosquito) |
| 4. | Sporozoites (human)Liver cellsGametocytes in bloodRBCsBlood meal(female mosquito)Multiply (female mosquito)Sporozoites (female mosquito) |
When a quick immune response is required, we can
1. Directly inject weakened pathogen at time of emergency.
2. Directly inject preformed antigens.
3. Directly inject preformed antibodies.
4. Directly inject immunosuppressants.
The excess use of herbicides and pesticides etc. has resulted in selection of resistant varieties in a much lesser time. Hence, resistant organisms/cells are appearing in a time span of months or years and not centuries. This statement is:
| 1. | In favour of evolution by anthropogenic action |
| 2. | Against the natural selection theory |
| 3. | In favour of sexual selection theory |
| 4. | In favour of genetic drift |
Industrial melanism cannot be explained by
| A. | Natural selection theory |
| B. | Mechanism of directional selection |
| C. | Mechanism of genetic drift |
| D. | Mechanism of founder's effect |
| E. | Mechanism of progressive selection |
1. A, C, D and E
2. A and D only
3. C and D only
4. D and E only
The relationship between Tetanus toxoid and Tetanus anti-toxin is that
(1) Tetanus toxoid injected in the body gives active immunity while Tetanus anti-toxin injected in the body gives passive immunity
(2) Tetanus toxoid injected in the body gives passive immunity while Tetanus anti-toxin injected in the body gives active immunity
(3) Tetanus toxoid injected in the body gives active immunity and Tetanus anti-toxin injected in the body also gives active immunity
(4) Tetanus toxoid injected in the body gives passive immunity and Tetanus anti-toxin injected in the body also gives passive immunity
Elephantiasis in man is caused by
1. Ancylostoma duodenale
2. Ascaris lumbricoides
3. Dracunculus medinensis
4. Wuchereria bancrofti
Salmonella typhi causes:
1. An acute infection of intestine that causes high fever and weakness
2. Enlargement of spleen and pain in stomach
3. Rose coloured rashes on the body
4. All of the above
Read the following statements about cancer and mark the incorrect ones
| (i) | Ionising radiations like x-rays and UV rays and non-ionizing radiations like gamma rays cause DNA damage leading to neoplastic transformation. |
| (ii) | CT scan uses non-ionizing radiations to accurately detect pathological and physiological changes in the living tissue. |
| (iii) | Antibodies against cancer-specific antigents are also used for detection of certain cancers. |
| (iv) | Some of the chemotherapeutic drugs are specific for certain cancers. |
| 1. | (i), (ii) and (iii) | 2. | (i) and (ii) |
| 3. | (iii) and (iv) | 4. | (ii) and (iv) |
Which one is true for humoral immunity?
| 1. | When a pathogen enters blood, B-lymphocytes get activated and differentiates into plasma cells |
| 2. | Undifferentiated B-lymphocytes remains as memory cells |
| 3. | Both (1) and (2) |
| 4. | B-lymphocytes produce an army of glycolipids against pathogen |
Antigens:
(1) Are always parts of microorganisms
(2) Can be pollens, egg-while, certain fruits, microorganisms and drugs
(3) Are always proteins
(4) Are always polysaccharides
Choose the correct option regarding the drug, its source, and its effect on the body
|
|
Drug |
Source |
Effect |
|
1. |
Coke |
Erythroxylum coca |
CNS depressant |
|
2. |
Heroin |
Papaver somniferum |
Stimulant |
|
3. |
Hashish |
Cannabis sativa |
Hallucinogen |
|
4. |
Datura |
Jimson's weed |
Stimulant |
Read the following statements:
(i) The main antibody produced as a result of allergic reaction is lgA.
(ii) Modern-day lifestyle has resulted in lowering immunity and more sensitivity to allergens.
(iii) Recombinant DNA technology has allowed the production of antigenic polypeptides of pathogens in bacteria or yeasts.
(iv) Allergy is suppressed due to the release of histamine and serotonin.
(v) The drugs like anti-histamines, adrenaline, and steroids quickly reduce the symptoms of allergy.
Which of the above statements are incorrect?
1. (ii), (iii), (v)
2. (i), (iv)
3. (ii), (v)
4. (ii), (iii)
Allergy involves:
(1) Exaggerated response of immune system to certain antigens present in the environment
(2) Non-sensitivity of the immune system to the allergens
(3) Usually life-threatening hypersensitivity of immune system to the allergens
(4) Excess systhesis of anti-histamines by our body
Arrange sequentially the steps used in MOET (Multiple Ovulation Embryo Transfer Technology) for herd/cattle improvement
I. The fertilized egg at 8-32 celled stage are recovered non-surgically and transferred to surrogate mother
II. Female cattle (cow)is administered hormones with FSH-like activity
III. The animal is either mated with an elite bull or artificially inseminated
IV. Induction of follicular maturation and superovulation, i.e., 6-8 eggs are formed instead of one which these animals produce per cycle
1. IIVIIIII
2. IIIIVIII
3. IIIVIIII
4. IIIIIIIV
'Hisardale' is a breed of sheep developed by crossing
1. Bikaneri ewes and Marino rams
2. Merino ewes and Bikaneri rams
3. Deccani ewes and Bikaneri rams
4. Merino ewes and Apennine rams
Inbreeding
1. Refers to the mating of distantly related individuals
2. Refers to the mating of more closely related individuals
3. Reduces homozygosity
4. Reduces inbreeding depression
The restriction endonuclease breaks bonds between:
1. DNA-RNA hybrid
2. Introns
3. Nucleotides, i.e., breaks the phosphodiester bond
4. Pentose sugar and nitrogenous base, i.e., breaks N-glycosidic bond
Match the following:
a. Grey biotechnology I. Related to aquatic organisms
b. Blue biotechnology II. Related to agriculture
c. Green biotechnology III. Related to industrial application
d. Red biotechnology IV. Related to Medical application
(1) a-III, b-I, c-II, d-IV
(2) a-I, b-II, c-III, d-IV
(3) a-II, b-I, c-III, d-IV
(4) a-IV, b-II, c-III, d-I
Mark the restriction endonuclease which forms blunt or flush ends
1.
2.
3.
4.
During gel electrophoresis for separation of DNA fragment:
1. Smallest fragment will move to the farthest point towards cathode
2. Smallest fragment will move to the farthest point towards anode
3. Largest fragment will move to the farthest point towards cathode
4. Largest fragment will move to the farthesr point towards anode
Insulin cannot be synthesized by introducing natural human gene in bacteria because
1. Natural human genes are split genes they do not express their effects in bacteria
2. Prokaryotes are not able to remove intron
3. There is absence of enzyme responsible for removing C chain
4. All of these
Plasmids serve as useful cloning vectors due to all of the following traits, except
(1) Small size, making them easy to manipulate once they are isolated
(2) Easy to transfer from cell to cell by transformation
(3) Have inbuilt selectable markers which make them easy to detect and select
(4) By attaching a polylinker, it becomes easy to allow insertion of cloned DNA
When we ligate a foreign DNA at the sal I site of tetracycline resistance gene in a vector pBR 322, the ____a____ plasmid will lose tetracycline resistance due to insertion of foreign DNA but can still be selected out from ____b____ ones by plating the ___c____ on tetracycline containing medium
| a | b | c | |
| 1. | Non-recombinant | Recombinant | Transformant |
| 2. | Recombinant | Non-recombinant | Transformant |
| 3. | Transformant | Non-recombinant | Recombinant |
| 4. | Recombinant | Transformant | Non-recombinant |
If Pvu I restriction endonuclease is used for cleaving pBR322 cloning vector for inserting foreign DNA, which of the following can be expected in recombinants?
| 1. | They remain resistant to tetracycline but not to ampicillin |
| 2. | They would die in tetracycline containing medium but remain alive in ampicillin containing medium |
| 3. | Replication of recombinant plasmid get prevented |
| 4. | They do not show resistance to ampicillin as well as tetracycline |
After fertilization, the first phase of embryonic development in cleavage. Find the statement which is not appropriate for this phase
(1) It occurs in quick succession without any growth of the resultant blastomeres
(2) Ordinary they are rapid, rhythmic and repeated mitotic divisions
(3) In mammals including human beings cleavage divisions are among the slowest in animal kingdom
(4) Cleavage divisions are synchronous from the very beginning in mammals
When DNA is transcribed into mRNA, usually the mRNA remains single-stranded, but in some cases a RNA can be made that is complementary to the mRNA. This is called __________ and its main function is to _______:
1. Antisense RNA, block gene expression
2. Antisense RNA, amplify mRNA
3. Antisense RNA, enhance translation
4. Reverse transcription, enhance translation
Trace the path of physiologically mature sperm in the male reproductive tract
| 1. | Seminiferous tubulesRete testisVasa efferentiaEpididymisVas deferensEjaculatory ducturethra |
| 2. | Vas deferensEpididymisEjaculatory ductUrethra |
| 3. | Rete testisEpididymisVasa efferentiaVas deferensEjaculatory ducturethra |
| 4. | EpididymisEjaculatory ductVas deferenceUrethra |
Find the incorrect statement
1. scrotum helps in maintaining low temperature of the testes 2-2.5 less than normal, necessary for spermatogenesis
2. Each testis has about 250 testicular lobules
3. Transfer of sperms into female genital tract is insemination
4. Seminiferous tubules open into rete testis through vasa efferentia
The first movement of foetus observed by mother is called quickening. It is usually observed during
(1) End of 1st trimester
(2) Early 5th month
(3) End of third trimester
(4) Early 3rd month
If the menstrual cycle of female is 34 days. What will be time period of periodic abstinence as a natural method of contraception?
(1) 10th to 7th day of menstrual cycle
(2) 13th to 24th day of menstrual cycle
(3) 18th to 23rd day of menstrual cycle
(4) 20th to 27th day of menstrual cycle
Decline in the level of which hormone of menstrual cycle is directly responsible for degeneration of corpus luteum?
1. Estrogen
2. Progesterone
3. LH
4. FSH
Oogenesis differs from spermatogenesis in a number of aspects. One of the following is however a similarity between the two
1. Growth phase is prolonged in both of them an starts before birth
2. Both give rise to non-motile gametes
3. Both produce equal number of gametes
4. Both occur inside primary sex organ
The hormones released by placenta
a. Resue degeneration of corpus luteum
b. Maintain high levels of progesterone
c. Prepare mammary glands for lactation
d. Cause morning sickness
(1) a & b only
(2) a, b, c & d
(3) a, b & d only
(4) a & d only
Which of the following occur during pre ovulatory phase?
a. Formation of corpus heamorrhagicum
b. Luteal phase
c. Secretory phase
d. Follicular phase
e. Proliferative phase
1. d & e only
2. a, b & c only
3. a, b & e only
4. c & d only
Chorion is made up of
(1) Trophoblast outside and somatopleuric extra embyonic mesoderm inside
(2) Trophoblast inside and somatopleuric extraembryonic mesoderm outside
(3) Endoderm inside and splanchnopleuric extraembryonic mesoderm outside
(4) Endoderm inside and somatopleuric extraembryonic mesoderm outside
Which hormone level in female suppresses release of GnRH, providing negative feedback control exactly parallel to the control of testosterone in males?
(1) Progesterone
(2) Estrogen
(3) LH
(4) FSH
Pills are very effective with lesser side effects and are well accepted by the females. They prevent conception by following means
| 1. | Inhibit ovulation |
| 2. | Inhibit implantation |
| 3. | After quality of cervix mucus to prevent/retard entry of sperms |
| 4. | All of these |
A contraceptive pill contains
| 1. | Progesterone and ostrogen |
| 2. | Spermicidal salts |
| 3. | Chemicals that cause automatic abortion |
| 4. | Chemicals that prevent fertilization of ovum |
Which one of the following is tested by the technique of amniocentesis?
1. Biochemical abnormalities in the foetus
2. Errors of metabolism in the foetus
3. Chromosomal abnormalities in the foetus
4. All of the above
Which of the following STDs cannot be treated with antibiotics?
(1) Genital herpes
(2) Chancroid
(3) Syphilis
(4) Gonorhoea
The following graph of relative concentrations of the four hormones present in the blood plasma of a woman during menstrual cycle. Identify the hormones:
A B C D
1. FSH Progesterone LH Oestrogen
2. LH Progesterone FSH Oestrogen
3. FSH Oestrogen LH Progesterone
4. LH Oestrogen FSH Progesterone
In a population, homozygous & heterozygous individuals are in a certain proportion. This proportion can't be changed by
1. Natural Selection
2. Mutation
3. Recombination
4. Use and Disuse of organs
To form four fully developed Polygonum type of embryosacs in angiosperms total number of meiosis and mitosis divisions generations required are :-
1. 4, 12
2. 1, 3
3. 4, 4
4. 1, 12
A : All organisms have to reach a certain stage of growth and maturity in their life, before they can reproduce sexually.
B : Male and female gametes must be physically brought together to facilitate syngamy :-
1. A - correct B - incorrect
2. Both A and B are correct
3. Both A and B are incorrect
4. A - incorrect B – correct
Genes which are tightly linked on chromosome show :
1. Very low recombination
2. High recombination
3. Very low parental combination
4. Clear cut independent assortment
In large number of insects the mechanism of sex determination is of XO type, then which of the
following statements is true for XO type determination :
1. All eggs bear an additional X-chromosome besides the autosome
2. All sperms bear only autosome
3. All eggs bear only autosomes
4. All sperms bear only X-chromosomes
In Morgan's experiment, the F2 ratio deviated very significantly from 9:3:3:1, This is due to :
1. Independent assortment
2. Segregation
3. Linkage
4. Dominance
Three region's in DNA - a promoter, structural gene and terminator are collectively known as :
1. Replication unit
2. Transcription unit
3. Translation unit
4. Operon
Which of the following is not correct with respect to salient features of the double helix structure of DNA?
1. The plane of one base pair stacks over the other
2. The backbone is constituted by sugar-phosphate and the bases project outside
3. The two chains have anti-parallel polarity
4. The pitch of the helix is 3.4 nm
In pea plant, the intermediate size of starch grains is due to
1. Dominant epistasis
2. Codominance
3. Incomplete dominance
4. Recessive epitasis
Read the following four statements (A-D)
A. The characters never blend in heterozygous condition
B. Change in a single base pair of DNA does not cause mutation
C. Cancer cells commonly show chromosomal aberrations
D. In chicken, sex chromosomes in male are ZW and in females are ZZ
How many of the above statement is/are right?
1. Two
2. Three
3. Four
4. One
In capping of RNA
1. Unusual nucleotide (methyl guanosine triphosphate) is added to the 5'-end of hnRNA
2. Adenylate residues (200-300) are added at 3'-end
3. Adenylate residues is added to the 5'-end of hnRNA
4. Unusual nucleotide (methyl guanosine triphosphate) is added at 3'-end
Consider the statements regarding Mendel’s work. Find out the incorrect option.
1. Mendel selected 14 true-breeding pea plant varieties, as pairs which were similar except for one character with contrasting traits.
2. To determine the genotype of a tall plant at F2, Mendel crossed the tall plant from F2 with a dwarf plant. This he called a test cross.
3. Mendel proposed that something was being stably passed down, unchanged, from parent to offspring through the gametes, over successive generations. He called these things as ‘factors’.
4. Based on his observations on monohybrid crosses Mendel proposed three general rules to consolidate his understanding of inheritance in monohybrid crosses.
Drosophila was used by Morgan for his experiments as
1. It completes its life cycle in two months.
2. Large size male can be easily grown on sucrose medium.
3. Hereditary variations can be seen with low power microscope.
4. It is a saprophyte.
Consider the following statements:
i. The most interesting molecule with autocalytic and heterocatalytic functions in the living system.
ii. Most abundant genetic material.
iii. Functions as adaptor or structural molecule.
iv. A long polymer of deoxyribonucleotides.
Which of these are correct for DNA?
1. (i), (ii) and (iv)
2. (i), (iii) and (iv)
3. (ii), (iii) and (iv)
4. (i), (ii), (iii) and (iv)
Law of independent assortment does not hold true because:
1. Distantly located genes assorted independently
2. Closely located genes assorted together
3. Genes of same chromosome always assort independently
4. Genes are DNA fragments
Which one is incorrect (with respect to mutation)?
1. It results in alteration of DNA sequence
2. Usually mutation change gene in next generation
3. Point mutation arise due to change in single base pair
4. Mutation is also a phenomenon that lead to variation in DNA
Test cross is:
| 1. | Cross between two homozygous genotypes for different traits of the same character |
| 2. | Cross between two heterozygous genotypes of different character |
| 3. | Cross between one homozygous and one heterozygous genotype for different traits of the same character |
| 4. | Cross between one heterozygous and homozygous of similar traits of the same character |
Adventitive embryo in Citrus by apomixis develops from:
1. Integuments only
2. Nucellus and synergids
3. Integuments and nucellus
4. Antipodals, synergids and integuments
| A: | Pollen grains carry the male gamete from microsporangium to micropyle of ovule in angiosperms. |
| B: | Pollen grain itself a male gamete further divides and form another one on stigma only. |
| 1. | Both A and B are correct | 2. | A is correct only |
| 3. | B is correct only | 4. | Both A and B are incorrect |
Seed-set is assured even in absence of pollinators, when:
1. Both flowers ( and
) of a plant mature at the same time
2. The numbers of flowers clustered into an inflorescence to make them conspicuous, colorless and rich in nectar
3. The flowers are cleistogamous
4. The flowers are functionally cross-pollinating but genetically self-pollinating
Find incorrect statement:
1. A polycarpellary, apocarpous gynoecium is found in Michelia
2. Many ovules are present in an ovary of papaya and orchids
3. Yellowish, powdery pollen grains found in Hibiscus
4. The more pistils may be fused together to form monocarpellary syncarpous gynoecium
A female is haemophilic because mother of female is
1. Carrier and father should be normal
2. Carrier and father should be haemophilic
3. Haemophilic and father should be normal
4. Normal and father should be normal
If father is colourblind and mother is carrier for
colourblindness then find out percentage of
colourblind progeny :-
1. 25%
2. 50%
3. 75%
4. 100%
In 5 types of nucleotide and 100 types of amino
acid participate in protein synthesis than genetic
code will be
1. Doublet
2. Triplet
3. Tetraplet
4. Can't determine
When a man with hairy ear marries a normal
woman, what percentage of their daugthers would
be expected to have hairy ear ?
1. 100%
2. 50%
3. 75%
4. 0%
Read the following statements (A-D):-
(1) DNA is a acidic substance
(2) The two chains of DNA have antiparallel
polarity
(3) 5-Methyl uracil is another chemical name of
thymine
(4) The two chains of B-DNA are coiled in a right
handed fashion
How many of the above statements are correct
1. 4
2. 3
3. 2
4. 1
These are two statements related to sexual reproduction :
(I) In both plants and animals, hormones are responsible for the transitions between the three phases.
(II) Interaction between hormones and certain environmental factors regulate the reproductive processes and the associated behavioural expressions of organisms.
Options :
1. Both (I) and (II) is correct
2. Only (I) is correct
3. Only (II) is correct
4. Neither (I) nor (II) is correct
Consider the following human disorders:
i. Haemophilia ii. Down’s Syndrome iii. Cystic fibrosis iv. Colour blindness
v. Night blindness.
Which of these disorders exhibit ‘Mendelian’ pattern of inheritance?
1. ii, iii and iv
2. i, ii and iii
3. i, iii and v
4. i, iii and iv
A plant with genotype AABbCcDD is self pollinated. Provided that the four genes are independently assorting, what proportion of the progeny will show the genotype AAbbccDD ?
1. 1/4
2. 1/16
3. 1/64
4. 1/256
To protect and improve the quality of our environment, government of India has passed Environment
(Protection) Act in the year ______
1. 1974
2. 1981
3. 1986
4. 1987
Read the following statements and choose the correct option
Statement A : Electrostatic precipitator can remove over 99% particulate matter present in exhaust from a thermal power plant.
Statement B : According to CPCB particulate size 2.5 micrometers or less in diameter (PM 2.5) are responsible for causing the greatest harm to human health.
1. Only satatement A is correct.
2. Only statement B is correct.
3. Both statements A and B are correct.
4. Both statements A and B are incorrect.
Triangular age pyramid represents
1. Expanding population.
2. Declining population.
3. Mature population.
4. Both 1 and 3
What do you mean by bioprospecting?
1. Biological analysis of living things to classify them.
2. Exploring molecular, genetic and species level diversity for product of economic importance.
3. Exploring forests to identify diversity present their.
4. It is branch of biology which deals with prospect of conservation.
Read the following statements and choose the correct option
Statement 1 : The success of mammals is largely due to their ability to maintain a constant body temperature.
Statement 2 : Majority of the animals and nearly all plants cannot maintain a constant internal environment.
1. Only statement 1 is correct.
2. Only statement 2 is correct.
3. Both statements are correct.
4. Both statements are incorrect.
In aquatic ecosystem, major channel for energy flow is
1. Grazing food chain.
2. Detritus food chain.
3. Auxillary food chain.
4. Parasitic food chain.
State True (T) or False (F) for the following statements and select the correct option
a. Mammals from colder climates generally have smaller ears and limbs to minimize the heat loss.
b. Experience of altitude sickness is due to high atmospheric pressure at high altitudes.
c. The size of a population for any species is not a static parameter.
d. Resources for growth for most of the animal population are infinite and become limiting sooner or later.
a b c d
1. T T T T
2. F T T F
3. F F F F
4. T F T F
Which of the following statements is/are correct?
(1) The entire sequence of communities that successively change in a given area is called sere.
(2) The natural reservoir of phosphorus is rock.
(3) Ecological pyramids do not accommodate food web.
1. Only statement (1) is correct.
2. Only statement (2) is correct.
3. All (1), (2) and (3) are correct.
4. All (1), (2) and (3) are incorrect
What is/are the correct explanation(s) for the species richness of the tropics?
1. Tropics had less evolutionary time for species diversification.
2. Tropics provide a relatively constant environment.
3. Tropics receive more solar energy which contributes to greater productivity.
4. Both 2 and 3 are correct.
Which one is not an effect of increase in the level of greenhouse gases?
1. CO2 fertilization effect.
2. EI Nino effect.
3. Snow blindness.
4. Warming of troposphere and cooling of stratosphere and thermosphere
Biomagnification of DDT in aquatic food chain leads to high concentration of DDT in fish eating birds. It results in
(1) Disturbance in calcium metabolism in birds.
(2) Thinning of egg shell and their premature breaking.
(3) Decline in birds population
1. (1) and (2) only
2. (2) and (3) only
3. (1) and (3) only
4. All (1), (2) and (3)
Which one is not a cause of biodiversity loss?
1. Alien species invasion.
2. Co-extinction.
3. Endemism.
4. Overexploitation
Different varieties of cheese are known by their characteristic texture, flavour and taste that is due to?
1. Flavouring agent used
2. Living agent used
3. It is inherent quality
4. Arise due to molecular interaction between chemicals
During biological treatment of sewage water the term activated sludge refers to?
1. Activated charcoal which is used for purification
2. Masses of aerobic bacteria and fungal filament which is sedimented
3. Different biochemical’s that comes with polluted water
4. It is digested solid waste which can be used as active fertilizer
Find out which one of the following statement is not correct with respect to gobar-gas plant?
1. It has a floating cover which keeps on rising as gas is produced
2. It is developed by IARI and KVIC
3. Main gas produced is butane, isobutene & propane
4. Spent slurry may be used as fertilizer
Element ‘B’ forms ccp structure and ‘A’ occupies half of the octahedral voids, while
oxygen atoms occupy all the tetrahedral voids. The structure of bimetallic oxide is
1.
2.
3.
4.
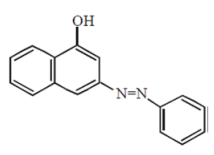
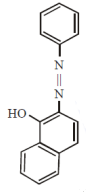
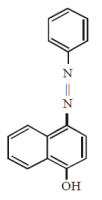
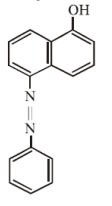
Coupling of benzene diazonium chloride with 1 – naphthol in alkaline medium will give:
1. 
2. 
3. 
4. 
The size of the iso-electronic species Ar and is affected by
1. Principal quantum number of valence shell
2. Azimuthal quantum number of valence shell
3. electron – electron interaction in the outer orbitals
4. nuclear charge
In the following compounds, the decreasing order of basic strength will be:
1.
2.
3.
4.
The correct order of the spin only magnetic moment of metal ions in the following low
spin complexes,
1.
2.
3.
4.
An organic compound ‘X’ showing the following solubility profile is: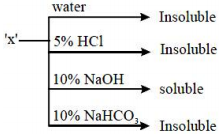
1. Oleic acid
2. o – Toluidine
3. Benzamide
4. m – Cresol
Diborane reacts independently with to produce, respectively:
1.
2.
3.
4.
The lanthanoide that would show colour is:
1.
2.
3.
4.
The major product of the following reaction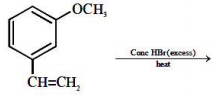
1. 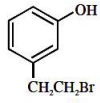
2. 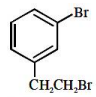
3. 
4. 
100 mL of a water sample contains 0.81 g of calcium bicarbonate and 0.73 g of
magnesium bicarbonate. The hardness of this water sample expressed in terms of
equivalents of is:
(molar mass of calcium bicarbonate is and magnesium bicarbonate is 146 g )
1. 10,000 ppm
2. 1,000 ppm
3. 5,000 ppm
4. 100 ppm
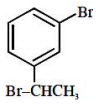
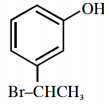
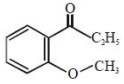
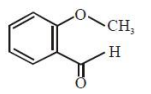
An organic compound neither reacts with neutral ferric chloride solution nor with Fehling solution. It however, reacts with Grignard reagent and gives positive iodoform test. The compound is:
1. 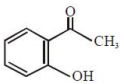
2. 
3. 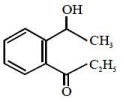
4. 
For the reaction the values of initial rate at different reactant concentrations are given in the table below: The rate law for the reaction is:
|
|
|
Initial Rate |
|
0.05 |
0.05 |
0.045 |
|
0.10 |
0.05 |
0.090 |
|
0.20 |
0.10 |
0.72 |
1.
2.
3.
4.
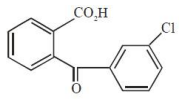
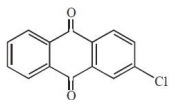
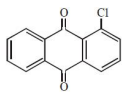
The major product of the following reaction is: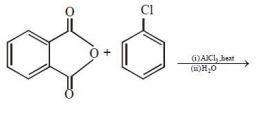
1. 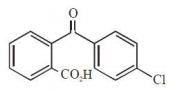
2. 
3. 
4. 
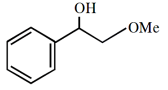
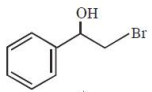
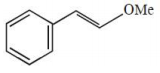
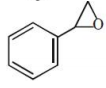
What is the major product formed in the following reaction?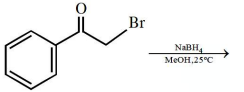
1. 
2. 
3. 
4. 
For silver . If the temperature (T) of 3 moles of silver is
raised from 300 K to 1000 K at 1 atm pressure, the value of will be close to:
1. 13 kJ
2. 62 kJ
3. 16 kJ
4. 21 kJ
The correct order of hydration enthalpies of alkali metal ions is:
1.
2.
3.
4.
Adsorption of a gas follows Freundlich adsorption isotherm. x is the mass of the gas
adsorbed on mass m of the adsorbent. The plot of log versus log p is shown in the
given graph, is proportional to: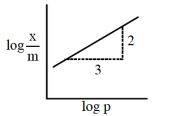
1.
2.
3.
4.
The vapour pressures of pure liquids A and B are 400 and 600 mm Hg, respectively at
298 K. On mixing the two liquids, the sum of their initial volumes is equal to the volume of the final mixture. The mole fraction of liquid B is 0.5 in the mixture. The vapour pressure of the final solution, the mole fractions of components A and B in vapour phase, respectively are:
1. 500 mmHg, 0.4, 0.6
2. 500 mmHg, 0.5, 0.5
3. 450 mmHg, 0.5, 0.5
4. 450 mmHg, 0.4, 0.6
Given that
. Then strongest oxidizing agent is:
1.
2.
3.
4.
The quantum number of four electrons are given below:
I.
II.
III.
IV.
The correct order of their increasing energies will be:
1. I < III < II < IV
2. I < II < III < IV
3. IV < II < III < I
4. IV < III < II < I
If solubility product of
1.
2.
3.
4.
Which one of the following equations does not correctly represent the first law of
thermodynamics for the given processes involving an ideal gas? (Assume non –
expansion work is zero)
1. Adiabatic process:
2. Isochoric process:
3. Cyclic process: q w
4. Isothermal process: q w
The IUPAC name of the following compound is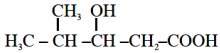
1. 3-Hydroxy – 4 – methylpentaonic acid
2. 4 – Methyl – 3 – hydroxypentanoic acid
3. 2 – Methyl – 3 – hydroxylpentan-5-oic acid
4. 4, 4 – Dimethyl – 3 – hydroxybutanoic acid
In order to oxidize a mixture of one mole of each of
1. 1
2. 1.5
3. 2
4. 3
Maltose on treatment with dilute HCI gives:
1. D – Fructose
2. D – Galactose
3. D – Glucose and D – Fructose
4. D – Glucose
With respect to an ore, Ellingham diagram helps to predict the feasibility of its
1. Electrolysis
2. Thermal reduction
3. Vapour phase refining
4. Zone refining
Magnesium powder burns in air to give:
1. MgO and
2. MgO and
3. MgO only
4.
The number of water molecule(s) not coordinated to copper ion directly in CuSO45H2O,
is:
1. 1
2. 3
3. 2
4. 4
Which of the following coordination complexes will exhibit optical activity?
(Note: en=ethane-1,2-diamine; A and B are monodentate ligands)
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Among the following, the molecule expected to be stabilized by anion formation is :
1.
2.
3.
4. NO
The degenerate orbitals of are :
1. and
2.
3.
4.
The given plots represent the variation of the concentration of a reactant R with time for
two different reactions (i) and (ii). The respective orders of the reactions are:
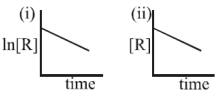
1. 1, 1
2. 0, 2
3. 0, 1
4. 1, 0
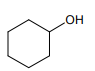
The organic compound that gives the following qualitative analysis is:
Test Inference
(a) Dil. HCl Insoluble
(b) NaOH solution Soluble
(c) /water Decolourization
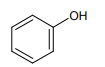
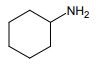
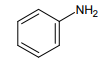
1. 
2. 
3. 
4. 
Excessive release of into the atmosphere results in:
1. formation of smog
2. depletion of the ozone
3. global warming
4. polar vortex
The ore that contains the metal in the form of fluoride is:
1. sphalerite
2. malachite
3. magnetite
4. cryolite
What is the correct IUPAC name for the following compound?

1. 5-Chloro-4-methyl-1-nitrobenzene
2. 2-Methyl-5-nitro-1-chlorobenzene
3. 3-Chloro-4-methyl-1-nitrobenzene
4. 2-Chloro-1-methyl-4-nitrobenzene
The element having the greatest difference between its first and second ionization energies, is
1. Ca
2. K
3. Ba
4. Sc
For a reaction,
; identify dihydrogen () as a limiting reagent in the following reaction mixtures.
1.
2.
3.
4.
The major product of the following reaction is :![]()
1.
2.
3.
4.
The osmotic pressure of a dilute solution of an ionic compound XY in water is four times that of a solution of 0.01 M BaCl2 in water. Assuming complete dissociation of the given ionic compounds in water, the concentration of XY (in mol ) in solution is
1.
2.
3.
4.
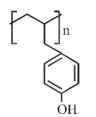
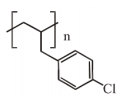
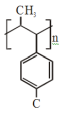
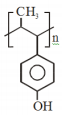
The major product of the following reaction is: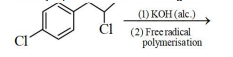
1. 
2. 
3. 
4. 
The increasing order of reactivity of the following compounds towards aromatic
electrophilic substitution reaction is
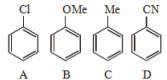
1. D < B < A < C
2. A < B < C < D
3. D < A < C < B
4. B < C < A < D
The correct order of the oxidation states of nitrogen in is :
1.
2.
3.
4.
The standard Gibbs energy for the given cell reaction in kJ at 298 K is :
Eo = 2 V at 298 K
[Faraday’s constant F = 96500 C ]
1. -192
2. 384
3. -384
4. 192
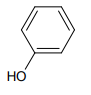
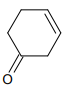
The major product of the following reaction is:
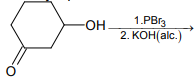
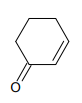
1. 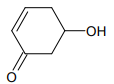
2. 
3. 
4. 
Three concentric metallic spherical shells of radii R, 2R, 3R, are given charges respectively. It is found that the surface charge densities on the outer surfaces of the shells are equal. Then, the ratio of the charges given to the shells, , is
(1) 1:2:3
(2) 1: 3:5
(3) 1:4:9
(4) 1:8:18
A charge Q is placed at each of the opposite corners of a diagonal of square. A charge q is placed at each of the other two corners. If the net electrical force on Q is zero, then Q/q equals
(1) -2
(2) -1
(3) 1
(4)
An electron is moving around the nucleus of a hydrogen atom in a circular orbit of radius r. The coulomb force on the electron is (Where )
(1)
(2)
(3)
(4)
Three identical point charges, as shown are placed at the vertices of an isoceles right angled triangle. Which of the numbered vectors coincides in direction with the electric field at the mid point M of the hypotenuse ?
(1) 1
(2) 2
(3) 3
(4) 4
Two metallic spheres of radii 1 cm and 3 cm are given charges of , respectively. If these are connected by a conducting wire, the final charge on the bigger sphere is
(1)
(2)
(3)
(4)
In the rectangle, shown below, the two corners have charges . The work done in moving a charge +3.0 from B to A is
(1) 2.8 J
(2) 3.5 J
(3) 4.5 J
(4) 5.5 J
Two parallel plates of area A are separated by two different dielectrics as shown in the figure. The net capacitance is:

1.
2.
3.
4.
In the given circuit, a charge of is given to the upper plate of the capacitor. Then in the steady state, the charge on the upper plate of the capacitor is
(1) +32
(2) +40
(3) +48
(4) +80
In the figure , a carbon resistor has bands of different colours on its body as mentioned in the figure. The value of the resistance is
(1) 2.2
(2) 3.3
(3) 5.6
(4) 9.1
A metallic wire of resistance of 12 is bent to form a square. The resistance between two diagonal points would be
(1) 12
(2) 24
(3) 6
(4) 3
A circuit consists of five identical conductors as shown in figure. The two similar conductors are added as indicated by the dotted lines. The ratio of resistances before and after addition will be
(1) 7 / 5
(2) 3 / 5
(3) 5 / 3
(4) 6 / 5
A galvanometer having a coil resistance of 60 shows full scale deflection when a current of 1.0 amp passes through it. It can be converted into an ammeter to read currents upto 5.0 amp by
(1) Putting in parallel a resistance of 240
(2) Putting in series a resistance of 15
(3) Putting in series a resistance of 240
(4) Putting in parallel a resistance of 15
A rectangular coil of length 0.12 m and width 0.1 m having 50 turns of wire is suspended vertically in a uniform magnetic field of strength 0.2 Weber/. The coil carries a current of 2A. If the plane of the coil is inclined at an angle of with the direction of the field, the torque required to keep the coil in stable equilibrium will be
(1) 0.20 Nm
(2) 0.24 Nm
(3) 0.12 Nm
(4) 0.15 Nm
Magnetic field induction at the centre O of a square loop of side 'a' carrying current I as shown in figure is
(1)
(2)
(3)
(4) zero
A magnet of magnetic moment is placed along the x-aixs in a magnetic field . The torque acting on the magnet is
(1)
(2)
(3)
(4)
Angle of dip is 90° at:
(1) Geomagnetic Equator (2) Geographic Equator
(3) Geographic Pole (4) Magnetic pole
A rectangular, a square, a circular and an elliptical loop, all in the (x-y) plane, are moving out of a uniform magnetic field with a constant velocity . The magnetic field is directed along the negative z-axis direction. The induced emf, during the passage of these loops, out of the field region, will not remain constant for
(1) The rectangular, circular and elliptical loops
(2) The circular and the elliptical loops
(3) Only the elliptical loop
(4) Any of the four loops
A rectangular loop has a sliding connector PQ of length l and resistance R and it is moving with a speed as shown. The set-up is placed in a uniform magnetic field going into the plane of the paper. The three currents are
(1)
(2)
(3)
(4)
A lamp consumes only 50% of peak power in an a.c. circuit. What is the phase difference between the applied voltage and the circuit current ?
(1)
(2)
(3)
(4)
The instantaneous value of current in an AC circuit is \(I = 2\sin\left(100\pi t +\dfrac{\pi}{3}\right)~\text{A}\). The current will be maximum for the first time at:
| 1. | \(t = \dfrac{1}{100}s\) | 2. | \(t = \dfrac{1}{200}s\) |
| 3. | \(t = \dfrac{1}{400}s\) | 4. | \(t = \dfrac{1}{600}s\) |
A LCR series AC circuit is tuned to resonance. The impedence of the circuit is now
(1) R
(2)
(3)
(4)
A coil of inductive reactance 31 has a resistance of 8. It is placed in series with a condenser of capacitative reactance 25. The combination is connected to an a.c. source of 110 volt. The power factor of the circuit is
(1) 0.80
(2) 0.33
(3) 0.56
(4) 0.64
A beam of cathode rays accelerated through a potential difference V is subjected to crossed Electric (E) and Magnetic fields (B). The fields are adjusted such that the beam is not deflected. The specific charge of the cathode rays is given by
(1)
(2)
(3)
(4)
A particle of mass 1 mg has the same wavelength as an electron moving with a velocity of . The velocity of the particle is
(1)
(2)
(3)
(4)
(Mass of electron=9.1)
The threshold frequency for a metallic surface corresponds to an energy of 6.2eV and the stopping potential for a radiation incident on this surface is 5V. The incident radiation lies in
(1) Infra-red region
(2) Visible region
(3) X-ray region
(4) Ultra-violet region
Two identical photo-cathodes receive light of frequencies . If the velocities of the photo electrons (of mass m) coming out are respectively , then
(1)
(2)
(3)
(4)
In the orbit, the energy of an electron is for hydrogen atom. The energy required to take the electron from first orbit to second orbit will be
(1) 10.2 eV
(2) 12.1 eV
(3) 13.6 eV
(4) 3.4 eV
If the wavelength of the first line of the Balmer series of hydrogen is 6561 , the wavelength of the second line of the series should be
(1) 13122
(2) 3280
(3) 4860
(4) 2187
Suppose an electron is attracted towards the origin by a force where 'k' is a constant and 'r' is the distance of the electron from the origin. By applying Bohr model to this system, the radius of the orbital of the electron is found to be and the kinetic energy of the electron to be . Then which of the following is true?
(1)
(2)
(3)
(4)
Which of the relation is correct between time period and number of orbit in which electron is revolving ?
(1)
(2)
(3)
(4)
Hydrogen atom from excited state comes to the ground state by emitting a photon of wavelength . If R is the Rydberg constant, the principal quantum number n of the excited state is
(1)
(2)
(3)
(4)
A nucleus with mass number 220 initially at rest emits an -particle. If the Q-value of the reaction is 5.5 MeV, calculate the kinetic energy of the -particle-
1. 4.4 MeV
2. 5.4 MeV
3. 5.6 MeV
4. 6.5 MeV
The power obtained in a reactor using disintegration is 1000 kW. The mass decay of per hour is
(1) 1 microgram
(2) 10 microgram
(3) 20 microgram
(4) 40 microgram
In the given reaction
radioactive radiations are emitted in the sequence
(1)
(2)
(3)
(4)
A piece of semiconductor is connected in series in an electric circuit. On increasing the temperature, the current in the circuit will
(1) Decrease
(2) Remain unchanged
(3) Increase
(4) Stop flowing
A junction diode has a resistance of 25 when forward biased and 2500 when reverse biased. The current in the diode, for the arrangement shown will be
(1)
(2)
(3)
(4)
Zener breakdown takes place if
(1) Doped impurity is low
(2) Doped impurity is high
(3) Less impurity in N-part
(4) Less impurity in P-part
A small object is placed 10 cm infront of a plane mirror. If you stand behind the object 30 cm from the mirror and look as its image, the distance focused for your eye will be
(1) 60 cm
(2) 20 cm
(3) 40 cm
(4) 80 cm
Convergence of concave mirror can be decreased by dipping in
(1) Water
(2) Oil
(3) Both
(4) None of these
The reason of seeing the Sun a little before the sunrise is
(1) Reflection of the light
(2) Refraction of the light
(3) Scattering of the light
(4) Dispersion of the light
A boy is trying to start a fire by focusing Sunlight on a piece of paper using an equiconvex lens of focal length 10 cm. The diameter of the Sun is m and its mean distance from the Earth is m. What is the diameter of the Sun's image on the paper?
(1)
(2)
(3)
(4)
Deviation of is observed from a prism whose angle is small and whose refractive index is 1.5. The angle of prism is
(1) 7.5
(2) 10
(3) 5
(4) 3.3
In single slit diffraction pattern
(1) Central fringe has negligible width than others
(2) All fringes are of same width
(3) Central fringes do not exist
(4) None of the above
Two light sources are said to be coherent if they are obtained from:
(1) two independent point sources emitting light of the same wavelength.
(2) a single point source.
(3) a wide source.
(4) two ordinary bulbs emitting light of different wavelengths.
Monochromatic green light of wavelength 5 m illuminates a pair of slits 1 mm apart. The separation of bright lines on the interference pattern formed on a screen 2 m away is
(1) 0.25 mm
(2) 0.1 mm
(3) 1.0 mm
(4) 0.01 mm