Ethephon hastens the fruit ripening in tomatoes and apple and accelerates abscission in flowers and fruits. Above statement indicates that ethephon must release –
1. ABA
2. C2H4
3. IAA
4. GA7
Four potted plants (I, II, III, and IV) of cabbage (biennial plant) are subjected to different temperatures for several days as given in the table.
Plot Temperature
I 5°C
II 24°C
III 27°C
IV 32°C
Which potted plant will show flowering?
(1) I
(2) II
(3) III
(4) IV
Which statement concerning ATP synthesis is true?
| 1. | ATP can be synthesized through substrate level phosphorylation, photophosphorylation and oxidative phosphorylation. |
| 2. | The proton-motive force is the establishment of proton gradients and electrochemical potentials across the inner membrane of mitochondria. |
| 3. | Proton-motive force is essential for back flow of H+ from outer chamber of matrix of mitochondria through proton channel (F0) of F0 – F1 particle to produce ATP. |
| 4. | All |
Choose the correct names of A, B, C and D
|
|
A |
B |
C |
D |
|
1. |
1,3 di PGA |
3 PGAld |
Fr,1,6 di P |
Fr. 6P |
|
2. |
3 PGAld |
1,3 Di PGA |
Fr.1,6 di P |
Fr. 6P |
|
3. |
Fr,1, 6 Di P |
Fr. 6 P |
3 PGAld |
1,3 di PGA |
|
4. |
Fr,6P |
Fr.1,6 di P |
3 PGAld |
1,3 di PGA |
Study the pathway ATP synthesis through chemiosmosis given below –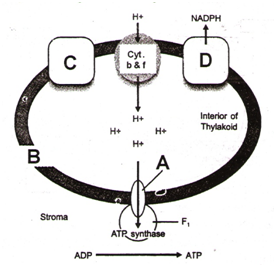
In which of the following options correct words for all the three blanks A, B, C and D are indicated –
| 1. | A – F1, B – Thylakoid membrane, C – Photosystem (I), D – Photosystem (II) |
| 2. | A – F0, B – Thylakoid membrane, C – Photosystem (I), D – Photosystem (II) |
| 3. | A – F1, B – Thylakoid membrane, C – Photosystem (II), D – Photosystem (I) |
| 4. | A – F0, B – Thylakoid membrane, C – Photosystem (II), D – Photosystem (I) |
Thomas Engelmann illuminated a filament of algae with light that passed through a prism, thus exposing different segments of the algal filament to different wavelengths of light. He added aerobic bacteria and found that these bacteria congregated in the areas illuminated by red and blue light. If you ran the same experiment without passing light through a prism, what would you predict?
| 1. | There would be no difference in results. |
| 2. | The number of bacteria would decrease along the entire length of the filament. |
| 3. | The bacteria would be relatively evenly distributed along the length of the filament. |
| 4. | The number of bacteria would increase along the entire length of the filament |
Study the cycle shown below and select the option which gives correct words for all the four blanks A, B, C and D –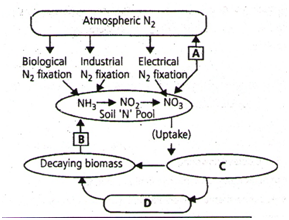
| A | B | C | D | |
| 1. | Nitri- fication |
Ammoni- fication |
Animals | Plants |
| 2. | Denitri- fication |
Ammon- ification |
Plants | Animals |
| 3. | Nitri- fication |
Denitri- fication |
Animals | Plants |
| 4. | Denitri- fication |
Nitrif- ication |
Plants | Animals |
I. Phloem sap can be transported in any required direction.
II. Phloem transports mainly water and sucrose but other sugars, hormones, and amino acids are also transported.
III. Cytoplasmic strands pass through the holes in the sieve plate forming continuous filaments.
IV. Ascent of sap is a pulling movement and the translocation of organic solutes is a pushing movement.
1. All are correct
2. All are incorrect
3. I, II, III are correct
4. III and IV are correct
Match the Column I with Column II –
|
|
Column-I |
|
Column-II |
|
1. |
Terminalization of chiasmata |
A. |
Zygotene |
|
2. |
Synapsis |
B. |
Diplotene |
|
3. |
Crossing over |
C. |
Metaphase I |
|
4. |
Dissolution of synaptonemal complex |
D. |
Diakinesis |
|
5. |
Best stage for the study of chiasmata |
E. |
Pachytene |
|
6. |
Nuclear membrane and nucleolus disappear |
|
|
|
7. |
Tetrads are arranged on equatorial line |
|
|
1. A – 2, B – 4,5, C – 7, D – 1,6, E – 3
2. A – 2, B – 3, C – 7, D – 1, 4, 6, E – 5
3. A – 2, B – 7, C – 3, D – 1, 4, 5, E – 6
4. A – 2, B – 1, C – 4, D – 5, 3, E – 6
The events shown below occur during different phases –
| A. | Centromere splits, chromatids separate and move to opposite poles. Chromatids are now called chromosome. |
| B. | Chromosomes cluster at opposite poles, decondensation of chromosome & reappearance of NM, GB, ER and nucleolus. |
| C. | Chromosomal replication. |
| D. | Kinetochores attach to spindle fibres and chromosome are arranged at equatorial plate. |
| E. | Spiration of chromosomes/condensation of chromosomal materials. |
Which of the following correctly identifies each of the phases described –
| Interphase | Prophase | Metaphase | Anaphase | Telophase | |
| 1. | C | E | D | A | B |
| 2. | C | D | E | A | B |
| 3. | C | E | D | B | A |
| 4. | C | A | D | E | B |
Choose the correct statements –
| I: | Mitochondria and Chloroplast transfer energy. |
| II: | Mitochondrion is a power-house of cell as it produces most of the cellular ATP. |
| III: | Mitochondria and chloroplast are found in all eukaryotic cells. |
| IV: | Mitochondria are the sites of anaerobic respiration. |
| V: | The matrix of mitochondria posses a single linear DNA, many RNA molecules, 80S ribosomes. |
1. IV and V
2. I, II
3. II, IV and V
4. III and V
Which of the following is the correct sequence / route of the secretory product?
| 1. | ER Vesicles Cis region of GB Trans region of GB Vesicle Plasma membrane. |
| 2. | RER GB Lysosome Nuclear membrane Plasma membrane. |
| 3. | ER Vesicles Trans region of GB Cis region of GB Vesicles Plasma membrane. |
| 4. | Lysosome ER GB Vesicles Cell membrane. |
| Column I | Column II |
| A. Spring wood or early wood B. Autumn wood or late wood |
I. Lighter in colour II. Density high III. Density low IV. Darker in colour V. Larger number of xylary elements VI. Vessels with wider cavity VII. Lesser number of xylary elements VIII. Vessels with small cavity |
Which of the following matching is correct?
1. A – II, IV, VII, VIII; B – I, III, V, VI
2. A – I, II, VIII, VIII; B – III, IV, V, VI
3. A – I, III, V, VI; B – II, IV, VII, VIII
4. A – I, III, VII, VIII; B – II, IV, V, VI
Given below is the diagram of a typical structure of dicotyledonous seeds. In which one of the options all the five parts A, B, C, D and E correct?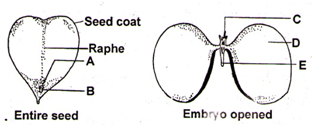
1. A – Hilum, B – Micropyle, C – Radicle, D – Cotyledon, E – Plumule
2. A – Hilum, B – Micropyle, C – Plumule, D – Cotyledon, E – Radicle
3. A – Micropyle, B – Hilum, C – Plumule, D – Cotyledon, E – Radicle
4. A – Hilum, B – Micropyle, C – Plumule, D – Radicle, E - Cotyledon
Which of the following combinations is false?
1. Apocarpous – Carpels free – Lotus, Rose.
2. Syncarpous – Carpels fused – Mustard, tomato.
3. Placentation – arrangement of ovules within ovary.
4. Arrangement of ovules within ovary – ovulation.
Which of the following is incorrect about A, B, C and D: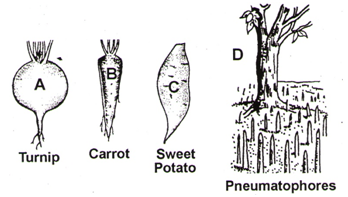
| 1. | Tap roots of carrot, turnip and adventitious root of sweet potato, get swollen and store food. |
| 2. | Pneumatophores help to get oxygen for respiration. |
| 3. | Pneumatophore is found in the plants that grow in sandy soil. |
| 4. | A, B and C are underground roots but D grows vertically upwards. |
Gemmae are the specialized structures produced in liverworts. These are
| 1. | Non-green, multi-cellular, asexual buds which develop in gemma cups. |
| 2. | Green, multi-cellular, asexual buds which develop in gemma cups. |
| 3. | Non-green, multi-cellular, diploid, sexual spores. |
| 4. | Green, unicellular, diploid, sexual spores. |
Which one is correct about heterosporous pteridophytes?
1. Microspore and megaspores develop into the male and the female gametophytes respectively.
2. The female gametophyte are retained on the parent sporophyte for a variable period.
3. The development of the zygote into the embryo takes place within the female gametophyte.
4. All
Which of the following statements does not characterize the red algae –
| 1. | Floridean starch (reserve food). |
| 2. | Both spores and gametes are non-motile. |
| 3. | Post fertilization development is like other algae. |
| 4. | Red algae can vary their ratio of photosynthetic pigments depending upon the light conditions. |
In 1971, T.O. Diener discovered a new infectious agent that was smaller than viruses –
| I. | It causes potato spindle tuber disease. |
| II. | It is free RNA. |
| III. | Molecular wt. of RNA is low. |
The above statements are assigned to –
1. Viruses
2. Viroids
3. Virulent
4. Mycoplasma
Which of the following is correct about class Deuteromycetes?
1. Some members are saprophytes or parasites.
2. A large number of members are decomposers of litter and help in mineral cycling.
3. Alternaria, Colletotrichum and Trichoderma are deuteromycetes.
4. All
Identify the blanks in the following figures –
| 1. | A – Cell wall, B – Cell membrane, C – Heterocyst, D – DNA, E – mucilaginous sheath. |
| 2. | A – Cell wall, B – Cell membrane, C – DNA, D – Heterocyst, E – Mucilaginous sheath. |
| 3. | A – Mucilaginous sheath, B – Cell membrane, C – DNA, D – Heterocyst, E – Cell wall. |
| 4. | A – Cell membrane, D – Cell wall, C – DNA, D – Heterocyst, E – Mucilaginous sheath. |
Which of the following statements about classification is not true?
| I. | Members of a family are less similar than members of an included genus. |
| II. | An order has more members than the number of members in an included genus. |
| III. | Families have more members than phyla. |
| IV. | The number of species in a taxon depends on their relative degree of similarity. |
1. Only III
2. Only IV
3. Only II
4. Only I
The following diagram refers to the process of transcription in Eukaryotes. Identify A, B, C and D –
| 1. | A – RNA polymerase II, B – Exon, C – Intron, D – Poly A tail |
| 2. | A – DNA polymerase II, B – Intron, C – Exon, D – Poly A tall |
| 3. | A – RNA polymerase II, B – Intron, C – Exon, D – Poly A tail |
| 4. | A – RNA polymerase II, B – Intron, C – Exon, D – Poly G tail |
Genetic code translates the language of
1. RNA into that of protein
2. RNA into that of DNA
3. amino acids into that of RNA
4. protein into that of DNA.
Wild type Escherichia coli growing on medium having glucose is transferred to lactose containing medium. Which one of the following change will occur?
1. The bacterium stops dividing
2. All operons are induced
3. Lac operon is suppressed
4. Lac operon is induced.
Choose the correct statements –
I. Binding of mRNA to the ribosome is loose and reversible.
II. The ribosome, which is composed of tRNA and proteins, consists of a small and large subunit.
III. Each of the 20 amino acids has a minimum of one specific tRNA molecule.
IV. tRNAMET binds to the first codon (AUG) – this is called the START codon.
V. The second tRNA binds to the second codon so that their attached monosaccharides are next to each other.
VI. The newly formed polypeptide may undergo post-transcriptional modification.
1. I, III and IV
2. II, IV, VI
3. II, V and VI
4. I, V, VI.
A test cross enables one to
1. Determine the viability of cross.
2. Distinguish between homozygous dominant and heterozygous dominant.
3. Determine whether two species can interbreed.
4. Determine the similarities in the DNA of two species.
The experiment shown in the figure has been carried out by Morgan to show the phenomenon of linkage and recombination. If in Cross I, genes are tightly linked and in Cross II, genes are loosely linked then what will be the percentage of recombinants produced in Cross I and Cross II respectively?
1. 98.7% and 62.8%
2. 1.3% and 37.2%
3. 37.2% and 1.3%
4. 62.8% and 98.7%
A. Genetic maps are extensively used as starting point in the sequencing of whole genome as was done in case of the Human genome sequencing project.
B. UV-rays can cause mutation in organisms.
C. Deletion and insertions of base pairs of DNA cause frame-shift mutations.
D. Mendelian disorders may be dominant or recessive.
1. All are correct
2. All are incorrect
3. a, b & d are correct
4. b, c & d are correct.
Which of the following is a false statement?
1. All organisms have evolved similar mechanism to multiply and produce offsprings.
2. Asexual reproduction is uniparental.
3. Sexual reproduction is biparental.
4. In asexual reproduction no fertilization occurs.
Which of the following is false about xenogamy?
1. It is the transfer of pollen grains from anther to stigma of another plant of the same species.
2. It produces genetic variation
3. It is genetically and ecologically (= functionally) cross pollination.
4. It occurs in cleistogamous flowers
Which of the following statements is false?
| I: | Vallisneria and Hydrilla are freshwater plants while sea-grasses (e.g., Zostera) are marine plants. |
| II: | Pollination in water lily / Lotus (Nymphea) and Eichhornia (water hyacinth) takes place by insects. |
| III: | In majority of aquatic plants, flowers emerge above the level of water and are pollinated by insects or wind. |
| IV: | In most of the water-pollinated species, pollen grains are protected from wetting due to presence of a mucilaginous covering. |
| V: | In hydrophilous plants, pollen grains are spherical. |
| 1. | All | 2. | None |
| 3. | V | 4. | IV |
Even in absence of pollinating agents seed setting is assured in?
1. Commellina
2. Zostera
3. Salvia
4. Fig
Identify structures A, B, C and phenomena – P1, P2, P3.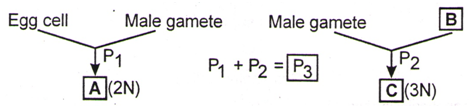
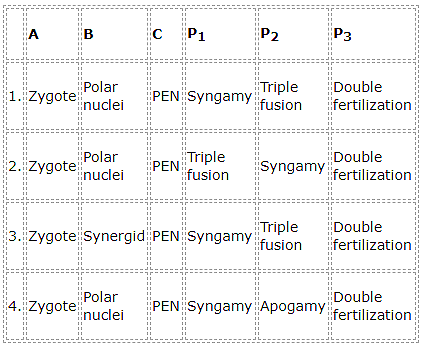
Some crop varieties produced by hybridization and selection, for disease resistance to fungi, bacteria, and viral diseases are released. Fill up the blanks-
|
Crop |
Variety |
Resistance to diseases |
|
Wheat |
A |
Leaf and stripe rust, hill bunt |
|
B |
Pusa swarnim (Karan rai) |
White rust |
|
C |
Pusa Shubhra, Pusa Snowball K–1 |
Black rot and Curl blight black rot |
|
Cowpea |
Pusa Komal |
D |
|
Chilli |
E |
Chilly mosaic virus, Tobacco mosaic virus & Leaf curl |
1. A – Himgiri, B – Brassica, C – Cauliflower, D – Bacterial blight, E – Pusa Gaurav
2. A – Himgiri, B – Brassica, C – Flat bean, D – Bacterial blight, E – Pusa Sadabahar
3. A – Brassica, B – Himgiri, C – Cauliflower, D – Bacterial blight, E – Pusa Sadabahar
4. A – Himgiri, B – Brassica, C – Cauliflower, D – Bacterial blight, E – Pusa Sadabahar.
Match the Column A with Column B –
|
|
Column A |
|
Column B |
|
I. |
Many people have deficiencies as they cannot buy fruits & vegetables |
A. |
Single cell proteins |
|
II. |
Crops with higher vitamins, proteins and fats are bred |
B. |
Micropropagation |
|
III. |
Growing microbes as the alternative source of proteins |
C. |
Somaclones |
|
IV. |
Capacity to generate plant from single cell or explant |
D. |
Hidden hunger |
|
V. |
Production of thousand plants through tissue culture |
E. |
Biofortification |
|
VI. |
Genetically identical plants |
F. |
Totipotency. |
1. I – D, II – E, III – F, IV – A, V – B, VI – C
2. I – D, II – E, III – F, IV – A, V – C, VI – B
3. I – D, II – E, III – A, IV – F, V – B, VI – C
4. I – F, II – E, III – A, IV – D, V – B, VI – C.
Name the blank spaces a, b, c and d given in the following table
|
Type of Microbe |
Scientific Name |
Commercial Product |
|
Bacterium |
A |
Clot buster enzyme |
|
B |
Aspergillus niger |
Citric acid |
|
Fungus |
Trichoderma polysporum |
C |
|
Bacterium |
D |
Butyric add |
1. A – streptococcus, B – Fungus, C – Cyclosporin–A, D – Clostridium butylicum.
2. A – Clostridium butylicum, B – Streptococcus, C – Fungus, D – Cyclosporin –A
3. A – Cyclosporin –A, B – Clostridium butylicum, C – Streptococcus, D – Fungus
4. A – Fungus, B – Cyclosporin –A, C – Clostridium butylicum, D – Streptococcus.
Consider the following statements (A – D) about organic farming:
(i) Utilizes genetically modified crops like Bt cotton.
(ii) Uses only naturally produced inputs like compost.
(iii) Does not use pesticides and urea.
(iv) Produces vegetables rich in vitamins and minerals.
Which of the above statements are correct?
1. (ii), (iii) and (iv)
2. (iii) and (iv) only
3. (ii) and (iii) only
4. (i) and (ii) only.
Study the population growth curves shown in the given diagram.
Which option is the best for curve (i) and (ii)
| Type of curve (i) | Type of curve (ii) | Status of food & space for curve (i) | Status of food & space for curve (ii) | |
| 1. | Logistic curve | Exponential curve | Unlimited | Limited |
| 2. | Exponential curve | Logistic curve | Unlimited | Limited |
| 3. | Logistic curve | Exponential curve | Limited | Unlimited |
| 4. | Exponential curve | Logistic curve | Limited | Unlimited |
Which of the following is most appropriately defined?
1. Host is an organism which provides food to another organism.
2. Amensalism is a relationship in which one species is benefited whereas the other is unaffected.
3. Predator is an organism that catches and kills other organism for food.
4. Parasite is an organism which always lives inside the body of the organism and may kill it.
The secondary productivity means
1. Rate of increase in the biomass of autotrophs.
2. Rate of increase in the biomass of heterotrophs.
3. The rate at which the organic molecules are used up by an autotroph.
4. The rate at which inorganic molecules are used up by an autotroph.
Find out the correct order of succession levels in xerarch.
1. Lichen, moss stage, annual herb stage, perennial herb stage, shrub stage, forest.
2. Annual herb stage, perennial herb stage, lichen, moss stage, shrub stage, forest
3. Perennial herb stage, annual herb stage, lichen, moss stage, shrub stage, forest
4. Shrub stage, forest, annual herb stage, perennial herb stage, lichen, moss stage.
Measuring Biochemical Oxygen Demand (BOD) is a method used for:
1. Estimating the amount of organic matter in sewage water.
2. Working out the efficiency of oil driven automobile engines.
3. Measuring the activity of Saccharomyces cerevisae in producing curd on a commercial scale.
4. Working out the efficiency of R.B.Cs. about their capacity to carry oxygen.
A. DNA polymorphism plays important role in evolution and speciation
B. 99.9% nucleotides bases are exactly same in all people
C. Chromosome 1 has the fewest genes (231) and Y-chromosomes has most genes (2968)
D. BAC and YAC have been used in Human Genome Project
1. All correct
2. All incorrect
3. A, B, D are correct
4. B, C, D are correct
Select the group of animals belonging to different
phyla?
1. Hirudinaria, Limulus, Laccifer.
2. Nereis, Dentalium, Aplysia.
3. Pheretima, Chaetopleura, Ophiura.
4. Cucumaria, Anteadon, Loligo.
Similarity between protochordata and hemichordata is
1. Presence of notochord.
2. Exclusively marine.
3. Dorsal hollow and single central nervous system.
4. Exclusively parasitic.
According to the discovery made in 1980’s, RNA can
act like enzyme to assemble new RNA molecules on
RNA template. Which of the following statements is
not proved by this theory?
1. Coacervates may not have been the first step in
the evolution of life.
2. Perhaps the first macromolecule was RNA.
3. Coacervates were the basis for the first cell.
4. After the formation of RNA, the stability of molecule
improved by surrounding RNA within a coacervate.
Identify the incorrect statement regarding the blood vascular system of cockroach:
| 1. | Blood vessels are poorly developed and open into hemocoel |
| 2. | The hemolymph is composed of colourless plasma and hemocytes |
| 3. | Heart lies along the mid-ventral line of thorax and abdomen |
| 4. | Blood from sinuses enter heart through ostia |
Nucleotide is made up of
1. Heterocyclic compound, Monosaccharide,
Phosphoric acid.
2. Nitrogenous base, hexose sugar, phosphate.
3. Hetrocylic compound and pentose sugar only.
4. Hetrocyclic compound and nucleoside.
NAD and NADP are consider as
| 1. | Apoenzyme and cofactor respectively. |
| 2. | Coenzyme and co-factor respectively. |
| 3. | Both as co-enzyme. |
| 4. | Apoenzyme and holoenzyme respectively |
Adipose tissue and areolar tissue are considered as:
1. Loose connective tissue and dense connective tissue respectively.
2. Dense regular and dense irregular tissue respectively.
3. Both are specialised connective tissue.
4. Both are loose connective tissue.
Statement (I) : Compound epithelium is always made
up of more than one layer of cells.
Satement (II) : Its main function is to provide protection
against chemical and mechanical stresses.
1. Both statement is correct.
2. (I) is correct and (II) is incorrect.
3. (I) is incorrect and (II) is correct.
4. Both are incorrect statement.
Common bile duct opens into:
1. Gall bladder
2. Jejunum
3. Hepato-pancreatic duct
4. Stomach
Mucus neck cell is a part of
1. Gastric gland.
2. Intestinal gland.
3. Salivary gland.
4. Both 1 and 2
According to Bohr’s effect
1. Hemoglobin oxygen binding affinity is inverely
related to acidity.
2. Hemoglobin oxygen binding affinity is inverely
related to concentration of CO2
3. Hemoglobin oxygen binding affinity is directly related
to acidity.
4. Both (a) and (b)
Chloride shift occurs when
1. Blood gives up oxygen and receives CO2
from tissues.
2. Blood receive oxygen from lungs.
3. When the hemoglobin saturation is minimum in the
blood.
4. When the blood pH is maximum.
The evolutionary process giving rise to new species
adapted to new habitats and ways of life is called
adaptive radiation. For example,
1. Darwin’s finches in Galapagos Islands.
2. Australian marsupials that radiated to form new
species.
3. Wolf and Tasmanian wolf.
4. Both (a) and (b)
‘Erythroblastosis Foetalis’ occurs when
I. Mother is Rh+
and foetus is Rh–
II. Mother is Rh–
and foetus is Rh+
III. Mother and foetus both are Rh+.
IV. Mother and foetus both are Rh–
1. I & II
2. Only I
3. Only II
4. II, III & IV
In ECG, ventricular contraction occurs
1. Just after P wave and before Q wave
2. Just after Q wave but before T wave
3. In between S-T segment
4. After the end of T wave
Which of the following cannot be expected in the filtrate when it arrives to descending limb of Henle’s loop?
| I. | Albumin | II. | Glucose |
| III. | Urea | IV. | Amino acid |
1. I, II, III & IV
2. I only
3. II, IV only
4. I, II & IV
Gill clefts and notochord appear in the embryonic
development of all vertebrates from fishes to mammals.
This supports the theory of
1. Recapitulation
2. Vetamorphosis
3. Biogenesis
4. Abiogenesis
Blood present in afferent arteriole and efferent arteriole will differ in
I. Amount of protein.
II. Number of blood cells.
III. Amount of glucose.
IV. Amount of urea.
1. I, II, III & IV
2. III & IV only
3. IV only
4. III only
Cardiac muscles can be best defined as
1. Many cardiac muscle cells assemble in a branching
pattern.
2. Single cardiac muscle fibre enclosed by connective
tissue layer.
3. Group of multinucleated muscle fibres enclosed by
connective tissue layer.
4. Group of unstriated muslce fibres enclosed by
endocardium.
Which of the following is required for the breaking of
cross bridge during muscle contraction?
1. ATP and Ca++
2. ADP and Ca++
3. Only Ca++
4. ATP only
Zygomatic and sphenoid bone is an example of
1. Cranial bones.
2. Carnial bone and facial bone respectively.
3. Facial bone and cranial bone respectively.
4. Facial bones
Mark the incorrect match:
|
1. |
Cerebral aqueduct |
→ |
A canal passes through the midbrain, connecting 3rd ventricle to 4th ventricle of brain. |
|
2. |
Corpus callosum |
→ |
Thick band of nerve fibres that divides the cerebral cortex lobes into left and right hemispheres. |
|
3. |
Limbic system |
→ |
The inner part of forebrain involved in the regulation of sexual behaviour. |
|
4. |
Pons |
→ |
Consist of fibre tracts that interconnect different regions of the brain. |
Vestibular apparatus is composed of:
I. Semi-circular canals
II. Otolith organs
III. Organ of Corti
IV. Crista and macula
1. I, II, III & IV
2. I, II only
3. IV only
4. II only
‘Retinal’ is present in
1. Rods.
2. Cones.
3. Both 1 and 2.
4. All the retinal layer
Low intelligence quotient abnormal skin and deaf-mutism is related to
1. Low secretion of growth hormone.
2. Hypothyroidism.
3. Hyperparathyroidism.
4. Hypo secretion of adrenal cortex hormone
Extra cellular receptors must be required for the action of
I. Oxytocin.
II. Thyroxine.
III. Epinephrine.
IV. Glucagon.
1. I, II, III & IV
2. I, IV only
3. I, III, IV
4. III & IV only
Mark the correct statement for pBR322 as a plasmid
vector
I. Contains relaxed origin of replication.
II. Two genes coding for resistance to antibiotics have
been introduced.
III. There are single recognition sites for a number of
restriction enzymes at various points.
IV. Insertional inactivation is a useful selection method
for identifying recombinant vectors with insert.
1. I, II, III & IV
2. I, II only
3. II, III only
4. II only
If two enzymes A and B are used for restriction mapping
of 9 kb molecule, cleavage with A gives fragments of 2
and 7 kb and B gives fragments and 3 and 6 kb. If A
and B cut at opposite end of molecule what will be the
length of fragment after double digestion?
1. 2 kb, 3 kb, 4 kb.
2. 1 kb, 2 kb, 6 kb.
3. 3 kb, 3 kb, 3 kb.
4. 2 kb, 2 kb, 5 kb
Which of the following is very effective sedative and
pain killer?
1. Cocaine.
2. Heroin.
3. Morphine.
4. Cannabis extract
Cellular oncogenes can be best explained as
| 1. | Mutated gene. |
| 2. | Present in normal cells. |
| 3. | Present only in the cells of benign tumour. |
| 4. | Genes due to which cells loose the property of contact inhibition. |
___A___ and ___B___ provide micro-environments for
the development and maturation of T-lymphocytes. Here
A and B is :-
1. Spleen and thymus gland.
2. Thymus gland and MALT.
3. Bone marrow and thymus gland.
4. Spleen and MALT.
Recombinant DNA technology has allowed the
production of __A__ of pathogen in bacteria or yeast.
Here A is:-
| 1. | Pro-toxin. |
| 2. | Anti-bodies. |
| 3. | Antigenic polypeptide. |
| 4. | Antigenic polysaccharide. |
Vaccines are available for the disease like
I. Polio.
II. Diptheria.
III. Pneumonia.
IV. Tetanus.
1. I, II, III & IV
2. I and IV only
3. I only
4. I, II & IV only
Odd one out ?
1. PMNL - neutrophils
2. Monocytes.
3. Natural killer cells.
4. Interferons.
Gene pool of a population tends to remain stable if the population is large, without large-scale mutations, without migration and with
| 1. | Random mating. |
| 2. | Moderate environmental changes. |
| 3. | Natural selection. |
| 4. | Reduction in predators. |
There are two opposing views about the origin of modern
man. According to one view, Homo erectus in Asia
were the ancestors of modern man. A study of variation
in DNA, however, suggested African origin of modern
man. What kind of observation of DNA variation could
suggest this?
1. Greater variation in Asia than in Africa.
2. Greater variation in Africa than in Asia.
3. Similar variation in Africa and Asia.
4. Variation only in Asia and no variation in Africa
Which of the following cells play major roles in rejection of a tissue graft (such as a kidney transplant from an unrelated donor)?
1. Cytotoxic and Helper T- cells
2. Plasma cells
3. Macrophages
4. B-cells
At which stage of HIV infection does one usually show
symptoms of AIDS?
1. When the infecting retrovirus enters host cells.
2. When viral DNA is produced by reverse
transcriptase.
3. When HIV replicates rapidly in helper
T-lymphocytes and damages large number of these.
4. Within 15 days of sexual contact with an infected
person
How does a bacterial cell protect its own DNA from restriction enzymes?
| 1. | By adding methyl groups to adenine and cytosines. |
| 2. | By reinforcing bacterial DNA structure with covalent phosphodiester bonds. |
| 3. | By adding histones to protect the double-stranded DNA. |
| 4. | By forming “sticky ends” of bacterial DNA to prevent the enzyme from attaching |
DNA fragments separated by gel electrophoresis are shown. Mark the correct statement :
| 1. | Band 3 contains more positively charged DNA molecules than band 1. |
| 2. | Band 3 indicates more charge density than bands 1 and 2. |
| 3. | Band 1 has longer DNA fragment than bands 2 and 3. |
| 4. | All bands have equal lengths and charges but differ in base composition. |
Level of estrogen and progesterone are minimum at
the time of
1. Follicular phase.
2. Ovulation.
3. Secretory phase.
4. Onset of menstrual phase.
Fertilization membrane is formed to
1. Faciliate entry of sperm into egg.
2. Provide stability to egg.
3. Prevent ectopic pregnancy.
4. Prevent polyspermy
Seminal plasma of humans is rich in
1. Fructose and certain enzymes, poor in Ca2+.
2. Fructose, Ca2+ and certain enzymes.
3. Fructose, Ca2+ but no enzymes.
4. Glucose, certain enzymes but no Ca2+
Which one of the following is not the function of
placenta?
1. Faciliates removal of carbon dioxide and waste
material from embryo.
2. Secrete oxytocin during parturition.
3. Faciliates supply of oxygen and nutrients to embryo.
4. Secretes estrogen.
Diaphragms, cervical caps and vaults prevent conception by
| 1. | Increasing phagocytosis of sperms within uterus. |
| 2. | Suppressing sperm motility. |
| 3. | Inhibiting ovulation and implantation. |
| 4. | Blocking the entry of sperms through the cervix. |
An absorption line of the lowest frequency in the transition of hydrogen spectra is:
1. n=1 to n=2
2. n=3 to n=8
3. n=2 to n=1
4. n=8 to n=3
Which of the alkali metal chloride is having the highest m.p. ?
1. LiCl
2. NaCl
3. KCl
4. RbCl
A solution of CuCl in NH4OH is used to measure the amount of gas by simply measuring change in volume. The gas is
1. CO2
2. H2
3. CO
4. All of the above.
When allylic alcohol (CH₂=CH-CH₂OH) is oxidized in the presence of bromine, which of the following products will be formed?
1. Oxalic acid
2. Formic acid
3. Succinic acid
4. Acrylic acid
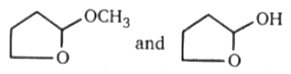
Above compounds can be differentiated by following reagent:
1. 2-4 DNP (Brady reagent)
2. Tollen's reagent
3. Bromine water reagent
4. NaHSO3
The concentration of the Ag+ ions in a saturated solution of Ag2C2O4 is 2.2 x 10-4 M. The solubility product of Ag2C2O4 is
1. 2.42 x 10-8
2. 2.66 x 10-12
3. 4.5 x 10-11
4. 5.3 x 10-12
The calomel electrode is reversible with respect to-
1. Mercury
2.
3.
4.
The volume of gases liberated at STP when a charge of 2F is passed
through aqueous solution of sodium phosphate, is :
1. 11.2L
2. 44.8L
3. 33.6L
4. 22.4L
Which of the following statement is not true about the effect of an increase in temperature on the distribution of molecular speeds of an ideal gas?
1. The area under the curve remains same even at the higher temperature.
2. The distribution pattern becomes more uniform.
3. Fraction of molecules with speed greater than a particular high speed will increase.
4. The fraction of molecules having most probable speed increases.
gas, molecular mass of gas A is:
1. 2
2. 32
3. 44
4. 28
Major product.
1.
2.
3.
4.
The pair that has both members from the same period of the Periodic Table is
| 1. | Cl, Br | 2. | Ca, Cl |
| 3. | Na, Ca | 4. | Na, Cl |
The electronic configuration that represents the maximum magnetic moment is:
1.
2.
3.
4. (high spin)
Which of the aldehyde is most reactive toward nucleophilic addition reaction?
1.
2.
3. HCHO
4. All the equally reactive
The number of structural and configurational isomers of a bromo compound, formed by the addition of HBr to 2-pentyne respectively are
(1) 1 & 2
(2) 2 & 4
(3) 4 & 2
(4) 2 & 1
Ether is more volatile
than alcohol despite having the same molecular formula. This is due to -
1. Dipolar character of ethers
2. Alcohols having resonance structures
3. Intermolecular hydrogen bonding in ethers
4. Intermolecular hydrogen bonding in alcohols
Which of the test is used for the detection of secondary amines?
1.Libermann's nitroso test
2. Lucas test
3. Tollen's test
4. Carbylamine reaction
Plastic household crockery is prepared by using :
1. Melamine and tetrafluoroethane.
2. Malonic acid and hexamethylene imine.
3. Melamine and vinyl acetate.
4. Melamine and formaldehyde.
The equivalent weight of for acid solution is
1. 79
2. 52.16
3. 158
4. 31.6
Which of the following statements regarding ozone is incorrect?
1. Powerful oxidising agent.
2. Bleaching agent
3. Paramagnetic
4. Bent shape
If volume containing gas is compressed to half, how many moles of gas remained in the closed vessel?
1. Just double
2. Just half
3. Same
4. More than double
Which is an extensive property?
1. Temperature
2. Chemical potential
3. Gibbs free energy
4. Molar volume
Which of the following elements has the highest second ionization enthalpy ?
1. Cr
2. Mn
3. Fe
4. Ti
The molecule among the following
that is an antidepressant is:
1. Chloroxylenol
2. Bithional
3. Cocaine
4. Penicillin-G
Identify the option which is correct with respect to Vanderwaal constant-a & b for gases.
1.
2. a CO2 > a H2 b H2
3.
4.

The IUPAC name of the above-mentioned compound is:
1. Pent-1-en-3-yne
2. Pent-1-ene-4-yne
3. Pent-4-yn-1-ene
4. Pent-1-en-4-yne
The only non-metal which is liquid at ordinary temperature is
1. Hg
2.
3.
4. None of these
Give the major product of the following reaction:
(1)
(2)
(3)
(4)
Formula for agate is
1.
2.
3.
4.
Which of the following sulphur compounds can be used as a refrigerant?
1.
2.
3.
4.
In the equation,
the coefficients of are respectively:
| 1. | 6, 7 | 2. | 6, 14 |
| 3. | 5, 7 | 4. | 5, 14 |
How many EDTA (ethylenediaminetetraacetic acid) molecules are required to make an octahedral complex with a ion?
1. six
2. three
3. one
4. two
The Lanthanide contraction is responsible for the fact that:
(1) Zr and S have almost the same radius.
(2) Zr and Nb have similar oxidation state.
(3) Zr and Hf have almost the same radius.
(4) Zr and Zn have the same oxidation state.
Which of the following does not react with periodic acid?
1.
2.
3.
4.
Which of the following is an example of additional polymerization?
(1) Proteins
(2) Teflon
(3) Nylon-66
(4) Glyptal (polyester)
In a set of reactions, propanoic acid yielded a compound (D).
\(CH_3CH_2COOH\ \text{(A)} \xrightarrow[]{SOCl_2}\ \text{(B)} \)
\(\ \xrightarrow[]{NH_3}\ \text{(C)}\ \xrightarrow[Br_2]{KOH}\ \text{(D)}\)
The formula for (D) is:
1.
2.
3.
4.
A liquid was mixed with ethanol and a drop of concentrated was added. A compound with a fruity smell was formed. The liquid was
1.
2.
3.
4.
Which of the following compounds, when heated at 483 K turns to caramel
1. Glucose
2. Sucrose
3. Fructose
4. Lactose
How many moles of magnesium phosphate, \(Mg_ 3 ( PO_ 4 )_ 2\) will contain 0.25 mole of oxygen atoms?
1. 0.02
2.
3.
4.
Which one of the following ions is not isoelectronic with ?
1.
2.
3.
4.
Which one of the following has a coordinate bond?
1.
2.
3.
4.
Which of the following is a path function?
1. Internal energy
2. Enthalpy
3. Work
4. Entropy
The molality of pure water is:
1. 1 m
2. 18 m
3. 55.5 m
4. None of the above
What will be the electrode potential of that hydrogen electrode is filled with HC1 solution of pH value 1.0?
1. -59.15V
2. +59.15
3. +59.15 mV
4. -59.15 mV
The reaction 2A + B + C D + E is found to be a first-order reaction with respect to A, second-order reaction with respect to B, and zero-order reaction with respect to C. If the concentrations of A, B, and C are doubled, the rate of the reaction will be:
1. 72 times
2. 8 times
3. 24 times
4. 36 times
The displacement time graph of a moving particle is shown in the figure below. The instantaneous velocity of the particle is negative at the point:
| 1. | D | 2. | F |
| 3. | C | 4. | E |
A \(5\) m long uniformly thick string rests on a horizontal frictionless surface. It is pulled by a horizontal force of \(5\) N from one end. The tension in the string at \(1\) m from the end where the force is applied is:
| 1. | zero | 2. | \(5\) N |
| 3. | \(4\) N | 4. | \(1\) N |
Three rings each of mass M and radius R are arranged
as shown in the figure. The moment of inertia of the
system about YY’ will be :
1.
2.
3.
4.
Fluid flows through a pipe in a horizontal plane. The fluid has a relative density of \(0.9\) and its velocity at \(P\) (where cross-section area is \(A\)) is \(4\) m/s. What is its pressure at \(Q\)? (where cross-section area is \(\frac{A}{4},\) The pressure at \(P\) is \(2.8\times 10^{5}~\text{Pa})\)
1. \(2.36\times 10^{5}~\text{Pa}\)
2. \(3.88\times 10^{5}~\text{Pa}\)
3. \(2.08\times 10^{5}~\text{Pa}\)
4. \(1.72\times 10^{5}~\text{Pa}\)
A charged ball B hangs from a silk thread S which makes an angle with a large charged conducting sheet P, as shown in the figure. The surface charge density of the sheet is proportional to
1. cos
2. cot
3. sin
4. tan
If the activity of a radioactive sample drops to 1/32 of its initial value after 7.5 Hours, its half-life will be:
1. 3 Hours
2. 4.5 Hours
3. 7.5 Hours
4. 1.5 Hours
The ratio of rotational and translational kinetic energies of a solid sphere is
1.
2.
3.
4.
A wave is represented by
y = a sin(At - Bx + C)
where A, B, C are constants. The dimensions of A, B, C are
(1)
(2)
(3) T, L, M
(4)
If a 50 kg mass is swinging in a vertical plane on a string at rest then the power delivered by gravity when the mass is moving with a velocity of 2 m/sec upwards in a direction, making an angle of with the vertical will be: (g = 9.8 m/)
| 1. | \(980 \mathrm{~W} \) | 2. | \(490 \mathrm{~W} \) |
| 3. | \(490 \sqrt{3}~ W \) | 4. | \(245 \mathrm{~W}\) |
When \(2\) point charges \(+q\) and \(+3q\), held at a distance r from each other are released, they have an acceleration of a and \(2a\) respectively. When we distribute the total charge equally between them, keep them at the same distance as the original and release them, their accelerations now would be:
1. \(2a\) and \(4a\)
2. \(\frac{4a}{3}\) and \(\frac{8a}{3}\)
3. \(\frac{9a}{4}\) and \(\frac{9a}{2}\)
4. \(\frac{2a}{3}\) and \(\frac{4a}{3}\)
Five equal capacitors connected in series have a resultant capacitance of \(4~\mu\text{F}\). The total energy stored in these when these are connected in parallel and charged to \(400\) V is:
1. \(1~\text{J}\)
2. \(8~\text{J}\)
3. \(16~\text{J}\)
4. \(4~\text{J}\)
In the following circuit, the battery \(E_1\) has an emf of \(12\) volts and zero internal resistance while the battery \(E\) has an emf of \(2\) volts. If the galvanometer \(G\) reads zero, then the value of the resistance \(X\) in ohms is:
| 1. | \(10\) | 2. | \(100\) |
| 3. | \(500\) | 4. | \(200\) |
A long straight wire carrying a current of 30 A is placed in an external uniform magnetic field of induction . The magnetic field is acting parallel to the direction of current. The magnitude of the resultant magnetic induction in testa at a point perpendicular distance 2.0 cm away from the wire is
()
(1)
(2)
(3)
(4)
In an LR circuit, the inductive reactance is equal to resistance R of the cirucit. An emf E = is applied to the circuit. The power consumed in the circuit is
(1)
(2)
(3)
(4)
| 1. | \(8~\text{cm}\) inside the sphere | 2. | \(12~\text{cm}\) inside the sphere |
| 3. | \(4~\text{cm}\) inside the sphere | 4. | \(3~\text{cm}\) inside the sphere |
Which of the following postulates of the Bohr model led to the quantization of energy of the hydrogen atom?
(1) the electron goes around the nucleus in circular orbits
(2) the angular momentum of the electron can only be an integral multiple of h / 2
(3) The magnitude of the linear momentum of the electron is quantized
(4) Quantization of energy is itself a postulate of the Bohr model
Which of the following is a wrong description of binding energy of a nucleus?
(1) It is the energy required to break a nucleus into its constituent nucleons
(2) It is the energy released when free nucleons combine to form a nucleus
(3) It is the sum of the rest mass energies of its nucleons minus the rest mass energy of the nucleus
(4) It is the sum of the kinetic energy of all the nucleons in the nucleus
A gas bubble oscillates with a time period T proportional to where P is pressure, d is the density and E is the energy. The values of a, b & c are
(1)
(2)
(3)
(4)
Two balls are thrown simultaneously at two different angles with the same speed from the same position so that both have equal range. If be the maximum heights attained in two cases, then the summation is equal to
(1)
(2)
(3)
(4)
The fundamental interval, that is the number of division between LFP & UFP on the two scales X and Y are 50 and 150 respectively. The ice point on both the scales is . If the temperature on the X-scale is , then what is the temperature on the Y-scale ?
A force of 5 N making an angle with the horizontal acting on an object displaces it by 0.4 m along the horizontal direction. If the object gains kinetic energy of 1 J then the component of the force is:
| 1. | 1.5 N | 2. | 2.5 N |
| 3. | 3.5 N | 4. | 4.5 N |
A standing wave pattern is established in a string as shown. The wavelength of one of the component travelling waves is
(1) 0.25 m
(2) 0.5 m
(3) 1 m
(4) 4 m
An object of mass 10 kg falls from rest through a vertical distance of 10 m and acquires a velocity of 10 m/s. The work done by the push of air on the object is
1. 500J
2. -500J
3. 250J
4. -250J
A small block of mass m is kept on a rough inclined plane of angle fixed in elevator. The elevator goes up with a uniform velocity v and the block does not slide down. The work done by the frictional force on the block in time t will be:
1.
2.
3.
4. 0
The given figure shows the electric lines of force emerging from a charged body. If the electric fields at A and B are respectively and if the distance between A and B is r, then:
(1)
(2)
(3)
(4)
The moment of inertia of a thin uniform circular disc about one of its diameter is I. Its moment of inertia about an axis perpendicular to the circular surface and passing through its center will be:
1.
2. 2 l
3.
4.
A particle falls towards the earth from infinity. Its velocity on reaching the earth would be-
1. Infinite
2.
3.
4. gR
What is the equivalent resistance between \(A\) and \(B\) in the figure below if \(R= 3~\Omega?\)

1. \(9~\Omega\)
2. \(12~\Omega\)
3. \(15~\Omega\)
4. None of these
The ratio of de-Broglie wavelengths of a proton and an alpha particle moving with the same velocity is:
1. \(1\)
2. \(2\)
3. \(4\)
4. \(0.25\)
The network shown in figure is a part of a complete circuit. If at a certain instant, the current \(i\) is \(10\) A and is increasing at the rate of \(4\times 10^{3}\) A/sec, then \(V_A-V_B\) is:

| 1. | \(6\) V | 2. | \(-6\) V |
| 3. | \(10\) V | 4. | \(-10\) V |
A gaseous mixture consists of 32 g of helium and 16 g of oxygen. The ratio of of the mixture is:
1. 1.4
2. 1.54
3. 1.59
4. 1.62
A condenser is charged to 500V and then the plates are joined through a resistance. The heat produced in the resistance (in joule) is
1. 0.10J
2. 0.15J
3. 0.25J
4. 0.3J
Two coherent monochromatic light beams of intensities I and 4I are superposed. The maximum and minimum possible intensities in the resulting beam are
(1) 5I and I
(2) 5I and 3I
(3) 9I and I
(4) 9I and 3I
In the nuclear reaction given below
The value of 'a' is given by
(1) 16
(2) 12
(3) 10
(4) 14
A current of 2 ampere is flowing through a coil of radius 0.1 m and having 10 turns. The magnetic moment of the coil will be:
1.
2.
3.
4.
If instantaneous current is given by amperes, then the r.m.s value of current is
1. 4 amperes
2. amperes
3. amperes
4. zero amperes
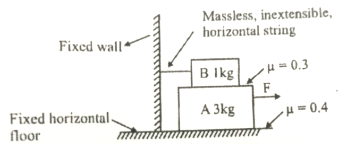
1. \(\dfrac{17g}{15}\)
2. \(\dfrac{19g}{10}\)
3. \(\dfrac{23g}{10}\)
4. \(\dfrac{11g}{10}\)
A double convex lens of f=6 cm is made of glass of refractive index 1.5. The radius of curvature of the surface which is half that of other surface will be
1. 1.5cm
2. 2.5cm
3. 3.5 cm
4. 4.5cm
If for water is and the velocity of light in vaccum is m/s, the time taken for light to travel a distance of 450m in water will be
1.
2.
3.
4.
| 1. | total energy of electron in \(1\text{st}\) orbit of \(\mathrm{He}^{+}\) |
| 2. | total energy of electron in \(3\text{rd}\) orbit of \(\mathrm{He}^{+}\) |
| 3. | total energy of electron in \(2\text{nd}\) orbit of \(\mathrm{Li}^{++}\) |
| 4. | total energy of electron in \(3\text{rd}\) orbit of \(\mathrm{Li}^{++}\) |
C and Si both have same lattice structure, having 4 bonding electrons in each. However, C is insulator where as Si is intrinsic semiconductor. This is because
1. In case of C the valence band is not completely filled at absolute zero temperature
2. In case of C the conduction band is partially filled even at absolute zero temperature
3. The four bonding electrons in the case of C lie in the second orbit, whereas in the case of Si they lie in the fourth
4. The four bonding electrons in the case of C lie in the second orbit, whereas in the case of Si they lie in the third orbit
The resultant force of two equal forces of F each is F . The angle between them will be :-
(1)
(2)
(3)
(4)