Arrange the following process starting from the germ
cells of seminiferous tubule?
(i) Meiotic division in secondary spermatocytes.
(ii) Spermiation.
(iii) Spermiogenesis.
(iv) Capacitation.
1. (i), (ii), (iii), (iv)
2. (i), (iii), (ii), (iv)
3. (ii), (i), (iv), (iii)
4. (i), (ii), (iv), (iii)
Mark the incorrect match that is not related?
1. Sertoli cell - Spermiation
2. Spermatid - Spermiogenesis
3. Secondary spermatocyte - Mitotic division
4. Spermatozoa - Capacitation
Mark the incorrect match
| (i) | All spermatocytes have haploid structure. |
| (ii) | All of the spermatogonia changes into primary spermatocyte. |
| (iii) | FSH is required for the process of spermiogenesis. |
| (iv) | Seminal plasma is essential for maturation and motility of sperms. |
1. (i), (ii), (iii) and (iv)
2. (iii) and (iv) only
3. (i) and (ii) only
4. Only (iii)
Female accessory ducts related to reproductive system is
(i) Oviducts
(ii) Uterus
(iii) Vagina
1. (i), (ii), (iii)
2. (i) only
3. (i) and (ii) only
4. (iii) only
Which of the following duct is present outside the
testis?
(i) Rete testis
(ii) Vasa efferentia
(iii) Vas deferens
(iv) Epididymis
1. (i), (ii), (iii), (iv)
2. (iii) and (iv) only
3. (ii), (iii), (iv)
4. (iii) only
Mark the incorrect match
| 1. | Fimbriae - Collection of ovum after ovulation. |
| 2. | Endometrium - Undergoes cyclic changes during menstrual cycle. |
| 3. | Labia majora - Fleshy folds of tissues surrounds the vaginal opening. |
| 4. | Ovarian stroma - It is the inner medulla of ovary which contains all the stages of growing ovarian follicle. |
Which of the following is not a part of female external
genitalia?
1. Birth canal
2. Hymen
3. Clitoris
4. Labia minora
Which of the following structure can not be seen in ovary during post ovolutionary phase?
| (i) | Primary follicle. |
| (ii) | Tertiary follicle. |
| (iii) | Corpus luteum. |
| (iv) | Primary polar body. |
1. (i) and (ii) only
2. (ii) and (iv) only
3. (ii) only
4. (iii) and (iv) only
Which of the following takes maximum duration for its
completion?
1. Meiosis I in primary oocyte.
2. Formation of haploid spermatozoa.
3. Formation of ootid from secondary oocyte.
4. Physiological maturity of spermatozoa.
Which of the following structure can not be seen in
ovarian follicle if antrum is visible in that?
1. Zona pellucida.
2. Membrane granulosa.
3. Primary oocyte.
4. Polar body.
Ovulation can be delayed by
1. Declining the level of progesterone.
2. Preventing LH surge.
3. Increasing the level of estrogen.
4. Declining the level of FsH.
Which of the following is not an event of follicular phase
of menstrual cycle?
1. Primary follicle changes into grattian follicle.
2. Endometrium of uterus regenerates.
3. Secretion of LH increases gradually.
4. Secretion of progesterone rapidly increases
Which of the following is not correct about corpus
luteum?
1. It is formed by follicular cells and theca cells
of ovary.
2. It secretes large amounts of progesterone which is
essential for maintenance of the endometrium.
3. It degenerates after the implantation of blastocyst.
4. It secretes relatively less amount of estrogen.
Mark the wrong statement for the blastocyst?
1. It is madeup of trophoblast cells and inner cell
mass.
2. It is surrounded with endometrial cells after
implantation.
3. It is formed at ampullary isthmus junction of
fallopian tube.
4. Cells of blastocyst secrete HCG to maintain corpus
luteum.
Which of the following is termed as post pregnancy
hormone?
(i) HCG
(ii) Relaxin
(iii) Prolactin
(iv) Progesterone
1. (i), (ii), (iii), (iv)
2. (i) and (ii) only
3. (i), (ii), (iii)
4. (i) only
Placenta is formed by
1. Uterine endometrial tissue only.
2. Chorionic villi of foetus only.
3. Amnion and chorionic membrane.
4. Both (1) and (2)
Which of the following organ/structure appears in the
last during embryonic development?
1. Notochord
2. Genital organs
3. Limbs
4. Eye-lashes
Embryonic meoserm develops from
1. Syncytotrophoblast
2. Cytotrophoblast
3. Inner cell mass
4. All of these
During foetal development ‘stem cells’ can be obtained
from
1. Inner cell mass
2. Placenta
3. Morula
4. Trophoblast
Foetal ejection reflex is generated after
1. Increases in level of oxytocin level in maternal blood.
2. Mild uterine contraction.
3. Increase in P/E ration in maternal blood
(P-Progesterone) (E-estrogen)
4. Rupturing of placenta.
From this given diagram, of menstrual cycle a, b, c, d
represent the
1. Estrogen, Progesterone, FSH, LH
2. FSH, Estrogen, LH, Progesterone
3. FSH, LH, Estrogen, Progesterone.
4. FSH, LH, Progesterone, Estrogen
Which of the following is not related to first trimester of
embryonic development?
(i) Functioning of heart.
(ii) Foetal movement.
(iii) Opening of Eye-lids.
(iv) Functioning of placenta
1. (iii) and (iv) only
2. (ii) and (iii) only
3. (ii), (iii), (iv)
4. (i) and (iii) only
Which of the following is correctly related to different
phases of menstrual cycle?
1. Ovulation always occurs at the mid of menstrual
cycle.
2. Meiosis I completion occurs in luteal phase of
cycle.
3. Thickening of endometrium is maximum in luteal
phase.
4. Level of progesterone is maximum in follicular phase
of menstrual cycle.
Gamete formation always occurs in
(i) Sexual reproduction.
(ii) Asexual reproduction.
(iii) Parthenogenesis.
1. (i) only
2. (i), (ii), (iii)
3. (i) and (iii) only
4. (i) and (ii) only
Common ejaculatory duct connects
1. Seminal vesicle to vas deferens.
2. Epididymis to vas deferens.
3. Seminal vesicle and vas deferens to the urethra.
4. Rete testis to vasa-efferentia.
Which of the following is not a part of seminal plasma?
1. Ca++
2. Fructose
3. Enzymes
4. Spermatozoa
Steroidal composition is present in
(i) Lippes loop
(ii) i-pills
(iii) Injectable and implants
(iv) Saheli
1. (i), (ii), (iii), (iv)
2. (ii) and (iii) only
3. (iv) only
4. (ii) only
Which of the following is the last step in the process of oogenesis?
1. Attachment of spermatozoa on the surface of the corona radiate.
2. Conversion of the primary oocyte into the secondary oocyte.
3. Sperm penetration through the zona pellucida.
4. Formation of ootid.
During fertilization spermatoza does not pass through
1. Birth canal.
2. Uterine tube.
3. Infundibulum of fallopian tube.
4. Isthmus of fallopian tube.
Mark the wrong statement for uterus?
1. The shape of it like an inverted pear.
2. It is supported by a ligament attached to the pelvic wall.
3. Its inner layer is the endometrium contains a thick layer of smooth muscle.
4. It is connected to the vagina through the cervical canal.
Odd one out in given barrier method of contraception
| 1. | Nirodh | 2. | Diaphragm |
| 3. | Vaults | 4. | Cervical cap |
Which of the following is not an application of IUDs?
| 1. | Create a sterile inflammatory response by producing a tissue injury of a minor degree. |
| 2. | Changing the intra-uterine environment and making it spermicidal. |
| 3. | Make the cervix hostile to sperms. |
| 4. | Hormonal changes prevent ovulation. |
As a contraception method, term sterilisation is used
for
1. Removal of testis
2. Vasectomy
3. Tubectomy
4. Both (2) and (3)
Amniocentesis can not detects problems like
1. Muscular dystrophy.
2. Down syndarome.
3. Spina bifida.
4. Cleft palate.
Intra uterine transfer of embryo is possible after
(i) In-vitro fertilization.
(ii) In-vivo fertilization.
(iii) ICSI
(iv) GIFT
1. (i), (ii), (iii) (iv)
2. (i), (ii), (iii)
3. (i) and (ii) only
4. (i) only
Marks the incorrect statement related to STDs?
| 1. | Most of this disease can be cured with antibiotics if treated early and can largely be prevented by the use of condoms. |
| 2. | All kinds of STDs can easily transfer from an infected mother to the foetus. |
| 3. | It can result in infertility and cancer of reproductive tracts. |
| 4. | Trichomoniasis is an STD that can be cured completely. |
Odd one out from the given IUDs
1. CuT
2. Cu7
3. Multiload 375
4. LNG-20
Which of the following mode of action of oral pills are common from the action of IUDs?
| (i) | Phagocytosis of spermatozoa. |
| (ii) | Prevention of ovulation. |
| (iii) | Preventing implantation. |
| (iv) | Retard entry of spermatozoa. |
1. (i), (ii), (iii), (iv)
2. (i), (ii), (iii)
3. (iii) and (iv) only
4. (iii) only
Regular oral contraceptive pills contain
| 1. | Progestogen only. |
| 2. | Progestogen - estrogen combination. |
| 3. | Estrogen only. |
| 4. | Both 1 and 2. |
A difference between the estrous cycle and the menstrual cycle
is that
1. The endometrial lining is shed in the menstrual cycle but reabsorbed in the estrous cycle.
2. Estrous cycles occur more frequently than menstrual cycles.
3. Ovulation occurs before the endometrium thickens in the estrous cycle.
4. Non-mammalian vertebrates have estrous cycles, whereas mammals have menstrual cycles.
Which the following male and female structures are
least alike in function?
1. Vas deferens — oviduct.
2. Intestritial cells — Follicle cells.
3. Spermatogonia — oogonia.
4. Seminiferous tubule — vagina.
During human gestation organogensis occurs
1. In the first trimester.
2. In the second trimester.
3. In the third trimester.
4. While the embryo is in the oviduct.
Which of the following animal gestation is very close
to human beings?
1. Cows
2. Elephants
3. Giraffes
4. Dogs
Which of the following is not a function of endometrial
tissues?
1. It provides nutrition during the first 2 to 4 week of embryo.
2. Formation of placenta.
3. Formation of extra-embryonic membrane.
4. Site of implantation.
Menstruation and ovulation do not occur during
pregnancy because of
1. High level of progesterone in maternal blood.
2. Fallopian tube gets blocked after endometrial proliferation.
3. All the ovarian follicle degenerates.
4. Foetal development activates immunological response to kill any other secondary oocyte.
Select the most abundant green house gases that
A. NO
B. Methane
C. Carbon dioxide
D. CFCs
1. A and B
2. B and C
3. C and D
4. A and D
Which among the following is used to remove over 99
percent particulate matter present in the exhausts from
a thermal power plant?
1. Scrubber.
2. Incinerator.
3. Catalytic convertor.
4. Electrostatic precipitator
Mark the odd one with respect to shape of ecological pyramids.
1. Pyramid of energy.
2. Pyramid of number in grassland ecosystem.
3. Pyramid of biomass in aquatic ecosystem.
4. Pyramid of biomass in terrestrial ecosystem.
Consider the following statements and state True (T)
contributes to global warming or False (F)
a. The Eastern Ghats have higher amphibian species
diversity than the Western Ghats.
b. Habitat loss and fragmentation is the most
important cause driving animals and plants to
extinction.
c. Loss of biodiversity in a region may lead to lowered
resistance to environmental perturbations.
d. In onsite conservation approaches, biodiversity at
all levels is protected.
A B C D
1. F T T T
2. T F T T
3. F F T T
4. T T T F
Identify the incorrect statement with respect to predation.
1. Nature's way to transfer energy fixed by plants to
higher trophic levels.
2. Predators keep prey populations under control.
3. Predator increases the intensity of competition
among competing prey species.
4. Predators in nature are "prudent" .
Which among the following characteristics confers
stability to the DNA helical structure?
i. Presence of Uracil.
ii. Plane of one base pair stacking over the other in
DNA helix.
iii. Left handed fashion coiling of DNA helix.
iv. Presence of hydrogen bonds.
1. i and ii
2. ii and iii
3. iii and iv
4. ii and iv
All are the characteristics of an individual from trisomy
of chromosome 21, except
1. Short stature
2. Gynaecomastia
3. Palm crease
4. Furrowed tongue
Identify the correct sequence of steps in decomposition
A. Humification.
B. Fragmentation of detritus.
C. Leaching.
D. Mineralisation.
1. B A C D
2. A C B D
3. B C A D
4. D A B C
What is/are the role(s) of deoxyribonucleoside
triphosphates during DNA replication?
A. Act as substrate.
B. Catalyse the polymerisation of ribonucleic acid.
C. Provide energy for polymerisation reaction.
1. A only
2. B only
3. A and B
4. A and C
The F1 generation resembles both the parents
in case of
1. Complete Dominance.
2. Incomplete Dominance.
3. Codominance.
4. Both (2) and (3)
What is the ploidy of female gametophyte and PEN
respectively in typical angiosperms?
1. n, 3n
2. 2n, 3n
3. 2n, 2n
4. 3n, 3n
Identify the mismatched pair.
1. Anemophily — Maize.
2. Entomophily Vallisneria.
3. Hydrophily — Zostera.
4. Ornithophily—Bombax.
Identify the incorrect statement.
1. The two alleles of a gene pair are located on
homologous sites of homologous chromosomes.
2. Dominance is not an autonomous feature of a gene.
3. Recessive trait is seen due to functional enzyme
or enzyme produced in abundance .
4. Alleles are forms of a gene which code for a pair of
contrasting traits.
The large holes in 'Swiss cheese' are due to production
of CO2 by
1. Saccharomyces cerevisiae.
2. Lactobacillus acidophilus.
3. Propionibacterium sharmanii.
4. Penicillium roqueforti.
Pusa sadabahar variety is resistant to all, except
1. Tobacco mosaic virus.
2. Bacterial blight.
3. Chilly mosaic virus.
4. Leaf curl disease.
In mature pollen grain, vegetative cell differs from
generative cell as the former
1. Floats in the cytoplasm of generative cell.
2. Is smaller in size.
3. Is spindle shaped with dense cytoplasm.
4. Have abundant food reserve and large nucleus
Statement A - In seed plants, both male and female
gametes are non-motile.
Statement B - In spermatophytes, pollen grains are
the carrier of male gametes.
1. Only statement A is correct.
2. Only statement B is correct.
3. Both statements A and B are incorrect.
4. Both statements A and B are correct
Which among the following traits of Pisum sativum is
only visible when both the alleles are identical?
1. Violet flower.
2. Inflated pod.
3. Axial flower.
4. Green seeds.
Examine the figures A, B, C and D. in which one of the
four options, all the labels for A, B, C and D are correct?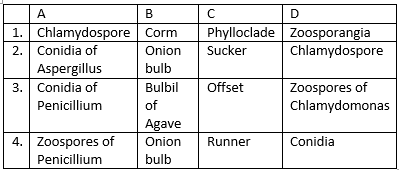
A plant breeder employ technique of emasculation
1. To cover the flower with a bag.
2. To prevent self-pollination.
3. To prevent stamen from being contaminated.
4. To produce female plant.
Look at the diagram given below and answer
appropriately
(i) Since ___A___ is associated with fruit, it is called
as ___(i)___ .
(ii) Since ___B___ is present, fruit cannot be called
as ___(ii)___ .
1. A - Pericarp, (i) - True fruits
2. B - Seed, (ii) - Ex-albuminous
3. A - Thalamus, (i) - True fruit
4. B - Seed, (ii) - Parthenocarpic
A typical anther shows
i. Two lobes with two theca each
ii. Each lobe as dithecous
iii. All cells in a given microsporangium as potential PMC
iv. Thousands of microspores per microsporangium
Out of these statements :
1. ii is incorrect.
2. i and ii are correct.
3. iii and iv are incorrect.
4. i, ii, iii and iv are correct.
Find correct match
|
|
Column-I |
|
Column-II |
|
a. |
Thick and swollen cotyledons |
(i) |
Castor. |
|
b. |
Unused endosperm in mature seed |
(ii) |
Legumes |
|
c. |
Unused nucellus in seed |
(iii) |
Cashew nut. |
|
d. |
Thalamus contributes in fruit formation |
(iv) |
Beet. |
1. a(iii), b(iv), c(ii), d(i)
2. a(i), b(iii), c(iv), d(ii)
3. a(ii), b(i), c(iii), d(iv)
4. a(ii), b(i), c(iv), d(iii)
Read the following statement and select the right choice
i. Semi-dwarf varieties, Jaya and Ratna were
developed in IRRI, Phillippines.
ii. Classical plant breeding involves crossing or
hybridization of pure lines.
iii. Saccharum barberi was originally grown in
South India.
iv. Genetic variability is the root of any breeding
programme.
1. Only i is correct.
2. ii and iv are correct.
3. i, ii and iii are correct.
4. ii, iii and iv are correct.
In nature, a given habitat has enough resources to
support a maximum possible number, beyond which
no further growth is possible. The limit is known as
1. Environmental resistance.
2. Intrinsic rate of natural increase.
3. Carrying capacity.
4. Exponentially growth.
Tropical environments, unlike temperate ones, are less
seasonal, relatively more constant and predictable.
Such constant environment promote
1. Deforestation.
2. Mass extinctions.
3. Over-exploitation.
4. Niche specialization.
Exploring molecular, genetic, and species-level
diversity for products of economic importance is
known as
1. Endemism
2. Bioinformatics
3. Tissue culture
4. Bioprospecting
It has been recommended that storage of nuclear waste,
after sufficient pre-treatment, should be done in suitably
shielded containers buried within rocks about _____
deep below the earth’s surface.
1. 100 m
2. 50 m
3. 500 m
4. 200 m
The rate of increase in biomass by heterotrophs or
consumers per unit time and area is known as
1. Gross primary productivity.
2. Net primary productivity.
3. Secondary productivity.
4. Community productivity.
The concept of Joint Forest Management was
introduced by the Government of India in
1. 1974
2. 1980
3. 1987
4. 1990
Which one of the following statement is false in respect
of flowering plants?
1. Parthenocarpy can be induced through the
application of growth hormones.
2. Integuments encircle the ovule except at the
tip where a small opening called the germ pore
is organized.
3. Endosperm development precedes embryo
development.
4. Apomicts have several advantages in horticulture
and agriculture
Which group of plants show clear cut vegetative,
reproductive and senescent phases in their life cycle?
1. Mustard, Radish, Mango.
2. Peepal, Mango, Banyan.
3. Wheat, Rice, Cabbage.
4. Peepal, Bryophyllum, Rice.
Choose unrelated for embryo development in flowering
plants :-
1. Develops at micropyle end of ovule.
2. Early stages of embryo development differs in
monocots and dicots.
3. In most cases zygotes divide first by transverse
division.
4. Most zygote divide only after certain amount
of endosperm is formed.
A: All organisms have to reach a certain stage of growth
and maturity in their life, before they can reproduce
sexually.
B: Male and female gametes must be physically
brought together to facilitate syngamy :
1. A - correct B – incorrect.
2. Both A and B are correct.
3. Both A and B are incorrect.
4. A - incorrect B - correct.
Which one of the following plant does not show
dioecious condition:-
1. Marchantia
2. Papaya
3. Date palm
4. Coconut
Read the following statements :-
(A) Desert lizard lack physiological ability that
mammals have to deal with high
temperature of their habitat.
(B) Kangaroo rat has the ability to concentrate its urine.
1. Only (A) is correct.
2. Both statements are incorrect.
3. Only (B) is correct.
4. Both statements are correct.
How many statements are correct about decomposition?
| (A) | Oxygen independent process. |
| (B) | Faster if detritus is rich in lignin and chitin. |
| (C) | Light is the most important climatic factor for decomposition. |
| (D) | Warm and moist environment favour decomposition. |
1. One
2. Two
3. Three
4. Four
Which one of the following belongs to more than one
trophic level ?
1. Grasshopper
2. Insectivorous plant
3. Cow
4. Lion
Which one of the following is wrong about primary
succession ?
1. Lichen is pioneer community in lithosere.
2. Formation of soil does not take place.
3. The establishment of new biotic community is
generally slow.
4. With time water body is converted into land.
According to Robert Constanza, the most expensive
ecological service is :-
1. Purification of air.
2. Soil formation.
3. Nutrient cycling.
4. Climate regulation.
Find the odd one out about levels of biological
organization:-
1. Biome
2. Community
3. Species
4. Population
In mycorrhiza association the fungal symbiont
helpful in :
1. Phosphorus nutrition.
2. Resistance to root borne pathogen.
3. Tolerance to salinity and drought.
4. All the above.
Virus free plants can be obtained by :-
1. Only apical meristem.
2. Only axillary meristem.
3. Apical and axillary meristem.
4. Embryo culture.
Atlas 66 is a variety of :-
1. Wheat
2. Bhindi
3. Rice
4. Sugar cane
Practical, hygienic, efficient, and cost-effective solution
to human waste disposal is:-
1. Natural toilets.
2. Eco-friendly toilets.
3. Eco-san toilets.
4. Biological sanitation.
For a zero order reaction if the time taken for 75%
completion of the reaction is 60 min than what is
its t1/2?
1. 30 minutes
2. 40 minutes
3. 50 minutes
4. 45 minutes
For a reaction, PCl5 → PCl3+ Cl2
, the rate can be expressed as
Relation between k1, k2 and k3 is
1.
2.
3.
4.
Which of the following is a correct statement?
1. Crystals having Frenkel defect have high
coordination number
2. Ferromagnetic substances becomes
paramagnetic above curie’s temperature
3. Percentage by volume of empty space in
ABAB....... type of packing is 74%
4. Increasing pressure, increases coordination
number due to increase in radius ratio
Which of the following is incorrect statement
regarding solubility of gas in liquid?
1. Gases which can react with solvent are more
soluble in solvent than the one which can not
react
2. Dissolution of gas is exothermic process
3. Solubility increases with increase in pressure
of the gas above the liquid
4. Gases can dissolve only in water
Which of the following is not correct for reactions
following 1st order kinetics?
1.
2.
3.
4.
Which of the following is true about any catalysed
process?
1. ΔHreaction for a catalysed process is different
from uncatalyzed process
2. Catalysed process also undergo the same
path as the uncatalyzed process
3. Activation energy for catalyzed path is different
from uncatalyzed process
4. All of these
Which of the following statement is correct?
1. Tyndall effect is shown by emulsion but not by
gel
2. Tyndall effect is shown by true solution
3. Tyndall effect is shown by emulsion and gel
but not by true solution
4. Tyndall effect will be shown by emulsion, gel
and true solution all
Gold number is measurement of
1. Metallic nature of gold
2. Electroplated gold
3. Protective power of lyophilic colloid
4. Protective power of lyophobic colloid
Softening of hard water using ‘Zeolite’ involves
1. Adsorption of Ca2+ and Mg2+ of hard water
replacing Na+ from zeolite
2. Adsorption of Ca2+ and Mg2+ of hard water
replacing Al3+ from zeolite
3. Adsorption of Ca2+ and Mg2+ of hard water
replacing both Na+ and Al3+ from zeolite
4. Adsorption of Cl–, and of hard
water replacing silicate ions from zeolite
AgI sol is prepared by adding KI into AgNO3
solution. The electrolyte having maximum
coagulation value of this AgI sol is
1. CaCl2
2. Na2SO4
3. AlPO4
4. K3[Fe(CN)6]
Mixing of two liquids A and B gives a solution
which show negative deviation from Raoult’s law,
then
1. ΔHmix < 0, ΔVmix < 0 and ΔSmix < 0
2. ΔHmix < 0, ΔVmix > 0 and ΔSmix < 0
3. ΔHmix < 0, ΔVmix < 0 and ΔSmix > 0
4. ΔHmix < 0, ΔVmix > 0 and ΔSmix > 0
The amount of ice that will separate out on cooling
a solution containing 50 g of ethylene glycol in 200
g of water to –9.3ºC will be -
[Kf for water = 1.86 K kg mol–1]
1. 18.9 g
2. 30.3 g
3. 21.9 g
4. 38.7 g
Which one of the following statements about unit is correct?
| 1. | unit has one peroxy linkage. |
| 2. | has two tetrahedral units joined through Cr—O— Cr. |
| 3. | The Cr — O — Cr bond angle is equal to 109°28′. |
| 4. | All of the above. |
In the complex [Fe(H2O)5NO]2⊕ the oxidation state
of iron is
1. +4
2. +3
3. +2
4. +1
Which one of the following is a diamagnetic ion?
| 1. | La3+ | 2. | Lu3+ |
| 3. | Ce4+ | 4. | All of the above. |
Which one of the following complexes can show
geometrical isomerism?
1. [PtCl2(NH3)2]
2. [Cr(ox)3]3(–)
3. [Co(NH3)6]3+
4. All of these
Which one of the following is the strongest ligand?
| 1. | F- | 2. | NH3 |
| 3. | CO | 4. | H2O |
The complex showing spin-only magnetic moment
1.732 B.M is
1. [NiCl4]2(–)
2. [Ti(H2O)6]3+
3. [Ni(CN)4]2(–)
4. [Co(NH3)6]3+
The mass of silver(eq. mass = 108) displaced by that quantity of current which displaced 5600 mL of hydrogen at STP is:
1. 54 g
2. 108 g
3. 5.4 g
4. none of these
Compound A is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Without losing its concentration ZnCl2 solution cannot be kept in contact with.
1. Au
2. Al
3. Pb
4. Ag
Compound A can be
1.
2.
3.
4. All the above compounds will form
CH3CH2CO-NH2 can be converted into CH3CH2NH2
in the presence of:
1. LiAlH4
2. H2/Pt
3. Sn/HCl
4. NaOBr/OH–
When ‘Z’ is treated with Sn/HCl it gives
1. Acetamide
2. Aniline
3. Benzamide
4. Benzylamine
What additional product is formed, besides the tertiary amine, when the given compound undergoes a reaction with moist silver oxide and heat?
| 1. | Toluene | 2. | Styrene |
| 3. | Ethyl benzene | 4. | Benzene |
The polymer that is addition as well as copolymer is:
1. PHBV
2. PVC
3. Buna-S
4. Neoprene
The molecule among the following that acts as an antiseptic is -
1. Iodine + C2H5OH
2. Boric acid
3. H2O2
4. All of the above
Cetyltrimethyl ammonium bromide is an example of :
1. Artificial sweetener
2. Cationic detergent
3. Soap
4. Anionic detergent
The Grignard reagent is added to methanal and hydrolyzed to produce:
| 1. | Primary alcohol | 2. | Tertiary alcohol |
| 3. | Methane | 4. | Methanoic acid |
Pb and Sn are extracted from their chief ore by:
1. carbon reduction and self-reduction respectively.
2. self-reduction and carbon reduction respectively.
3. electrolysis and self-reduction respectively.
4. self-reduction and self-reduction respectively.
HVZ reaction cannot be given by:
| 1. | Butyric acid | 2. | Acetic acid |
| 3. | Benzoic acid | 4. | Phenylacetic acid |
Which of the following acids is the strongest?
1. CH3COOH
2. C6H5COOH
3. C6H5OH
4. C2H5COOH
| 1. | Phenol | 2. | Benzene |
| 3. | Salicylic acid | 4. | Soda ash |
Which of the following compounds produces a pink colour when combined with Schiff's reagent?
1. CH3CHO
2. CH3CH2COOH
3. C3H5COOH
4. C6H5COC6H5
The most acidic amongst the following is
1. HClO
2. HBrO
3. HClO3
4. HClO4
In S8 unit shown below, the oxidation number and
valency of S-atom respectively are
1. 0, 0
2. 2, 2
3. 0, 2
4. 2, 0
The number of lone pairs in XeF2, XeO3 and XeOF4
at the Xe atom respectively are
1. 3, 1, 1
2. 3, 2, 1
3. 1, 2, 3
4. 3, 2, 2
The correct order of bond angle of group 16
hydrides is
1. H2O > H2S > H2Se > H2Te
2. H2Te > H2Se > H2S > H2O
3. H2S > H2Se> H2O> H2Te
4. H2S > H2O> H2Se>H2Te
Iodide ion on oxidation in the basic medium by KMnO4
will give:
1.
2.
3.
4.
Liquation can be used for
1. Fe
2. Pb
3. Mn
4. Ag
Total electron transfer during reaction of KMnO4 in
acidic, basic and neutral medium per molecule
respectively is
1. 5, 3, 1
2. 5, 1, 3
3. 3, 1, 5
4. 1, 3, 5
Which of the following molecules can readily undergo the SN2 mechanism?
| 1. | \(\left(\mathrm{CH}_3\right)_3 \mathrm{C}-\mathrm{Cl} \) | 2. | \(\mathrm{CH}_2=\mathrm{CH}-\mathrm{Cl} \) |
| 3. | \(\mathrm{CH}_2=\mathrm{CH}-\mathrm{CH}_2-\mathrm{CH}_2 \mathrm{Cl}\) | 4. |  |
|
Which set of reactants among the following produces butane nitrile? |
1. C3H7Br + KCN
2. C4H9Br + KCN
3. C3H7OH + KCN
4. C4H9OH + KCN
Sandmeyer's reaction described by among the following is:
| 1. | |
| 2. | |
| 3. | |
| 4. |  |
The product 'B' in the below mentioned reaction is-
1. Phenol
2. Benzol
3. Sodium benzoate
4. Benzal
| 1. | Ultraviolet light has a wavelength shorter than \(4000~\mathring{A}\). |
| 2. | Infrared light has a wavelength longer than \(7000~\mathring{A}\). |
| 3. | Red light has a wavelength near about \(7000~\mathring{A}\). |
| 4. | Violet light has a wavelength near about \(7000~\mathring{A}\). |
Hydrogen (H), Deuterium (D), singly ionized helium and doubly ionized lithium () all have one electron around the nucleus. Consider n = 2 to n = 1 transition. The wavelengths of emitted radiations are respectively. Then approximately
1.
2.
3.
4.
The wavelength of radiation emitted is when an electron jumps from the third to the second orbit of hydrogen atom. For the electron jump from the fourth to the second orbit of the hydrogen atom, the wavelength of radiation emitted will be
(a) (b)
(c) (d)
The energy of lowest state of hydrogen is -13.6 eV. The ionisation energy of first excited state will be
(A) 13.6 eV (B) 4.2 eV
(C) 6.8 eV (D) 3.4 eV
The half life of is 3 minutes. What fraction of a 10 gram sample of , will remain after 15 minutes?
(1) (2) (3) (4)
A nuclear reaction along with the masses of the particle taking part in it is as follows;
The energy Q liberated in the reaction is
(1) 1.234 MeV (2) 0.931 MeV
(3) 0.465 MeV (4) 1.862 MeV
A concave lens of focal length \(25~\text{cm}\) can produce an image \(\frac1{10}\text{th}\) the size of the object. The distance of the object from the lens is:
1. \(225~\text{cm}\)
2. \(250~\text{cm}\)
3. \(150~\text{cm}\)
4. \(175~\text{cm}\)
A battery of internal resistance r, when connected across 2Ω resistor supplies a current of 4A. When the same battery is connected across a 5Ω resistor, it supplies a current of 2A. The value of internal resistance r is:
1. 2Ω
2. 1Ω
3. 0.5Ω
4. zero Ω
If E be the electric field inside a parallel plate capacitor due to Q and -Q charges on the two plates, then electrostatic force on plate having charge -Q due to the plate having charges +Q will be
1. -QE
2.
3. QE
4.

In the circuit shown in the figure, the current supplied by the battery is:
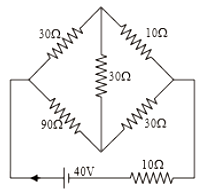
1. 2 A
2. 1 A
3. 0.5 A
4. 0.4 A
An electric Kettle draws 4.2A at 250V. How much time it would take to boil 1kg of water at C completely to 100 ?
1. 320 seconds
2. 64 seconds
3. 3840 seconds
4. 960 seconds
The electric Potential at a point in space due to charge \( Q \) is \( Q \times 10^{12} \, \text{V} \). The value of electric field at that point will be
1. \( \frac{Q}{9} \times 10^{12} \, \frac{N}{C} \)
2. \( \frac{Q}{9} \times 10^{15} \, \frac{N}{C} \)
3. \( \frac{Q}{9} \times 10^{9} \, \frac{N}{C} \)
4. \( Q \times 10^{15} \, \frac{N}{C} \)
A metallic sphere is given some charge. Electric energy is stored:
1. only inside the sphere.
2. only outside the sphere.
3. both inside and outside.
4. neither inside nor outside.
If a soap bubble is given some charge, then its radius:
| 1. | increases. |
| 2. | decreases. |
| 3. | remains unchanged. |
| 4. | may increase or decrease depending upon whether the given charge is positive or negative. |
A positively charged particle enters a magnetic field of \(B \hat {i}\) with a velocity \(V \hat {j.}\)
The particle will move
| 1. | In a circle |
| 2. | In y-z plane |
| 3. | With constant speed |
| 4. | All of these are correct |
A current wire is hidden in a wall. Its position can be located with the help of a:
| 1. | Moving coil galvanometer |
| 2. | Voltmeter |
| 3. | Hotwire ammeter |
| 4. | Magnetic needle |
1. R
2. 2R
3. 3R
4.
The vertical component of the earth's magnetic field is zero at:
1. The magnetic poles.
2. The magnetic equator.
3. The geographic poles.
4. The geographic equator.

Use of transistor as an amplifier works in the region -
1. Cut off region
2. Saturation region
3. Passive region
4. Active region
For a transistor amplifier, the power gain and voltage gain are 150 and 10 respectively. The current gain is
1.
2.
3. 15
4. 1500

| 1. | \(5\) | 2. | \(10\) |
| 3. | \(15\) | 4. | \(20\) |
When the temperature of the silicon sample is increased from to , the conductivity of silicon will be
1. Increased
2. Decreased
3. Remain same
4. Zero
A light beam containing red, yellow and blue color, falls on the interface of glass air as shown. If the yellow light is just totally internally reflected , then emergent ray in air contains-
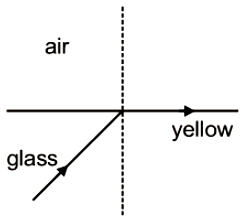
1. Both red and blue color
2. Only red color
3. Only blue color
4. No color

A transistor is used in common emitter mode in an amplifier circuit. When a signal of 10 mV is added to the base-emitter voltage, the base current changes by 15 A and the collector current changes by 1.5 mA. The current gain b will be
1. 50
2. 48
3. 100
4. 200
The current through an ideal p-n junction diode shown in the circuit will be -
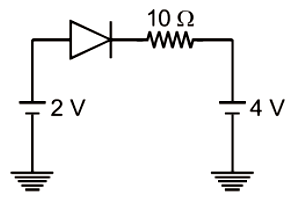
1. 5 A
2. 0.2 A
3. 0.6 A
4. Zero
A closed-loop (Of any shape) carrying current is lying in the x-y plane. A uniform magnetic field B is present in the region such that the loop experiences zero force :
1. B acts along the x-axis
2. B acts along the y-axis
3. B acts along the z-axis
4. B can any of the above direction for the net force to be zero
Below figures (1) and (2) represent lines of force. Which is the correct statement?
1. Figure (1) represents magnetic lines of force
2. Figure (2) represents magnetic lines of force
3. Figure (1) represents electric lines of force
4. Both figure (1) and figure (2) represent magnetic lines of force
Points A and B are situated perpendicular to the axis of a 2cm long bar magnet at large distances X and 3X from its centre on opposite sides. The ratio of the magnetic fields at A and B will be approximately equal to
(a) 1 : 9 (b) 2 : 9
(c) 27 : 1 (d) 9 : 1
In an electromagnetic wave in free space, the root mean square value of the electric field is V/m. The peak value of the magnetic field is
(a) 1.41
(b)
(c)
(d)
The interference pattern is obtained with two coherent light sources of intensity ratio n. In the interference pattern, the ratio will be
1.
2.
3.
4.
When a metallic surface is illuminated with radition of wavelength , the stopping potential is V. If the same surface is liiuminated with radiation of wavelength 2, the stopping potential is .The threshold wavelength for metallic surface is -
(a) 5 (b)
(c) 3 (d) 4
Which of the following figures represent the variation of particle momentum and the associated de-Broglie wavelength?
(a)


At resonance, the value of the power factor of the circuit is -
1. 1
2. Zero
3. Between zero and 1
4. More than 1
1. 25V
2. 75V
3. 35V
4. Zero
Energy stored in an inductor can be made four times when
(1) Inductance is doubled
(2) Current is made times
(3) Current is doubled
(4) Can never to be made

When the light of wavelength is made to fall on metal in a photoelectric experiment the maximum kinetic energy of emitted electrons is found to be 2.5eV. The work function of the metal is
1. 2.5eV
2. 3.1eV
3. 0.9eV
4. 1.5eV
Phenomen of polarisation justify which nature of light ?
1. longitudinal
2. transverse
3. both
4. geometrical

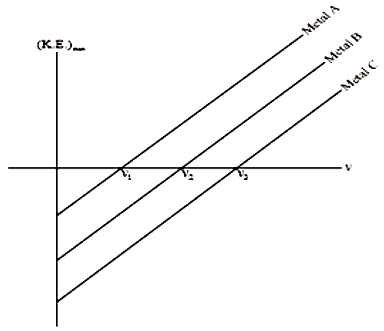
1.
2.
3.
4. None of these