What is true about cork cambium?
| 1. | It is extrastelar cambium in dicot stem. |
| 2. | In dicot stem it arises from the cells of cortex region. |
| 3. | It is also known as phellogen. |
| 4. | All the above |
Read the following statements with respect to pericycle and choose the suitable option
| (a) | It is the outermost portion of stele, that may be paranchymatous or sclerenchymatous. |
| (b) | It is absent in monocot stems. |
| (c) | It is always single layered. |
| 1. | Only (a) is correct |
| 2. | Only (b) is incorrect |
| 3. | Only (c) is incorrect |
| 4. | Both (a) & (b) are incorrect |
State True (T) or False (F) to the given statements and select the correct option
(A) Annual rings are distinct in plants growing in temperate regions.
(B) Lenticels occur in most woody trees and permit the exchange of gases.
(C) Due to stellar secondary growth, central cylinder of wood surrounded by secondary phloem is formed.
(D) The cells of endodermis opposite to protoxylem divide to give rise vascular cambium in dicot roots.
(A) (B) (C) (D)
1. T T F F
2. T T T F
3. T F T F
4. F F T T
Read the following statements about dicot stem and
choose the correct option.
| (a) | Vascular bundles are arranged in a ring. |
| (b) | Vascular bundles are conjoint, collateral and open type. |
| (c) | Endarch type of arrangement of secondary xylem. |
| 1. | Only (b) is correct |
| 2. | Only (c) is incorrect |
| 3. | Both (a) and (b) are incorrect |
| 4. | All three statements are correct |
What is incorrect for companion cell?
| 1. | It is specialized parenchymatous cell. |
| 2. | It helps in maintaining the pressure gradient in the sieve tubes. |
| 3. | It does not retain nucleus throughout the life. |
| 4. | It is absent in gymnosperms |
State True (T) or False (F) to the given statements and
select the correct option
| 1. | All tissue layers exterior to the vascular cambium constitute bark. |
| 2. | Root hypodermis is sclerenchymatous. |
| 3. | Ground tissue of leaves is called mesophyll. |
| 4. | Due to presence of casparian strips, endodermis is impervious to water. |
| (A) | (B) | (C) | (D) | |
| 1. | T | F | F | T |
| 2. | T | F | T | T |
| 3. | F | T | T | F |
| 4. | T | F | T | F |
Which of the following characters is/are related to isobilateral leaf?
| (a) | Stomata are present on both surfaces. |
| (b) | Mesophyll is differentiated into palisade and spongy parenchyma. |
| (c) | Sub-stomatal cavity is present below the stoma of the abaxial epidermis. |
| 1. | Only (a) |
| 2. | Only (c) |
| 3. | Only (a) and (c) |
| 4. | (a), (b) and (c) |
Which of the following floral features is not represented
by symbols in a floral formula of a plant family?
1. Relative positions of ovary with respect to other parts.
2. Adhesion of stamens.
3. Aestivation of calyx and corolla
4. Symmetry of flower.
Select the incorrect match
1. Green photosynthetic - Australian Acacia
petiole
2. Leaflets attached at - Silk cotton
tip of the petiole
3. Papilionaceous corolla - Bean
4. Epiphyllous stamens - Brinjal
Which of the given feature is not related to mustard?
1. Superior ovary.
2. Variable length of filaments of stamens.
3. Parietal placentation.
4. Opposite phyllotaxy.
Cymose inflorescence is dissimilar to racemose inflorescence in
1. Having limited growth of the main inflorenscence axis.
2. Having young flowers at top.
3. Showing centriperal manner of opening of flowers.
4. Having acropetal arrangement of flowers.
Read the following characters and mark the correct
ones for family Fabaceae.
(a) Flowers are arranged in acropetal manner on
floral axis.
(b) Flowers with radial symmetry.
(c) Hypogynous flower.
(d) Albuminous seeds.
(e) Monocarpellary ovary.
1. Only (a) and (c)
2. Only (a), (c), (d) and (e)
3. Only (a), (c) and (e)
4. Only (a) and (b)
Which of the following statements is not true for runner?
(1) Internodes are longer.
(2) Helps plants to spread to new niches.
(3) Roots are present at nodes.
(4) Found in plants like grasses, strawberry and jasmine.
State True (T) or False (F) to the given statements and select the correct option
| (A) | Abundance of lichens in any area indicates that the area is highly polluted with SO2. |
| (B) | Mycobiont partner of lichens is always heterotrophic. |
| (C) | Body of lichens is made up of phycobionts only. |
(A) (B) (C)
1. T T F
2. F T F
3. F F F
4. T F F
Which of the given statement is not true for viruses?
| 1. | They are nucleoproteins where protein is infectious in nature. |
| 2. | They can be crystallized and crystals consist largely of proteins. |
| 3. | Virus means venom or poisonous fluid. |
| 4. | A virus can never have both DNA and RNA as its genetic material. |
Mark the correct statement for Albugo.
(1) Causes white rust in members of Barassicaceae.
(2) Obligate saprophyte.
(3) Cell wall is made up of fungal cellulose.
(4) Mycelium is coenocytic and septate.
Organisms responsible for causing ‘red tide’ are also
characterized by
1. Presence of stiff cellulosic plates.
2. Obligate saprophyte.
3. Presence of two longitudinal flagella.
4. Filamentous body made up of trichomes.
How many of the following organisms possess
membrane bound cell organelles and autotrophic mode
of nutrition?
[Nitrobacter, Chromatium, Methanococcus, Nostoc,
Euglena, Gonyaulax, Paramoecium, Yeast,
Puccinia]
(1) 5 (2) 4
(3) 2 (4) 6
Group of organisms in which cell wall forms two thin overlapping shells are
1. Responsible for bioluminescence.
2. Chief producers of ocean.
3. Prokaryotes.
4. Heterotrophs.
According to the mass flow hypothesis:
(a) Sucrose is moved into the companion cells and then into the sieve tube by passive transport.
(b) Inside the phloem, an osmotic pressure gradient is generated that facilitates the mass movement in the phloem.
(c) Water in the adjacent xylem moves into the phloem by an active process.
1. (a) and (c) are correct.
2. (b) and (c) are incorrect.
3. (a) and (c) are incorrect.
4. (a) and (b) are correct.
Select the incorrect statements
(a) Older dying leaves export most of their mineral content to younger leaves.
(b) Sulphur and calcium and frequently remobilized from senescing parts.
(c) Ions are absorbed from the soil by both passive and active transport.
(d) A C3 plant loses only half as much water as a C4 plant for the same amount of CO2 fixed
(1) (a), (b) and (c)
(2) Only (a) and (c)
(3) Only (b) and (d)
(4) All except (c)
Coralloid roots of gymnosperms are/have
1. Irregular and possess large number of roots hairs.
2. Symbiotic association with Rhizobium.
3. Symbiotic association with N2 - fixing cyanobacteria.
4. VAM
In pteridophytes, gametophyte that develops in the homosporous species is usually
1. Monoecious and has events, precursor to the seed habit.
2. Dioecious and does not lead to seed habit.
3. Monoecious and does not lead to seed habit.
4. Dioecious and has events, precursor to the seed habit.
Read the following statements and select the option with correct statements
| (a) | In Wolfia, the highly reduced female gametophyte present within ovule, is embryo sac. |
| (b) | The ploidy level of endosperm in Cycas and Eucalyptus is triploid. |
| (c) | Azolla is a water fern. |
| (d) | Majority of the red algae are marine with greater abundance in the warmer areas. |
1. (a) and (b) only
2. (b) and (c) only
3. (a), (b) and (c)
4. (a), (c) and (d)
Match the classes of pteridophyte given in column-I with their respective members given in column-II
| Column I | Column II | ||
| (a) | Psilopsida | (i) | Selaginella |
| (b) | Lycopsida | (ii) | Adiantum |
| (c) | Pteropsida | (iii) | Psilotum |
| (d) | Sphenopsida | (iv) | Equisetum |
Select the correct answer from the following
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (i) | (iv) | (ii) |
| 2. | (i) | (iii) | (ii) | (iv) |
| 3. | (iii) | (i) | (ii) | (iv) |
| 4. | (i) | (iv) | (iii) | (ii) |
Read the following statements and select the incorrect ones
| (a) | Mosses have an elaborate mechanism of spore dispersal. |
| (b) | In liverworts, the haploid free living sporophyte is formed by spore germination. |
| (c) | Vegetative reproduction in Polytrichum occurs by budding in the secondary protenema. |
| (d) | Marchantia is a heterosporous bryophyte. |
| (e) | Growth of bog moss ultimately fills ponds and lakes with soil |
1. (a), (b) and (c)
2. (d) and (e) only
3. (b) and (d) only
4. All except (a)
The members of phaeophyceae are characterized by all, except
| 1. | Presence of chlorophyll a, c and fucoxanthin pigments. |
| 2. | Production of pear-shaped and biflagellated asexual spores. |
| 3. | Production of pyriform gametes that bear laterally attached flagella. |
| 4. | Their occurrence, mostly in fresh water habitats. |
Which of the following characters, are defining features
of all living organisms?
(A) Growth from inside
(B) Sexual reproduction
(C) Metabolism
(D) Response to stimuli
(E) Cellular organisation
1. Only (C), (D) and (E)
2. Only (A) and (B)
3. Only (B), (C) and (D)
4. All except (B)
A taxonomic aid which gives actual account of habitat
and distribution of various plants of given area, is
1. Manual
2. Flora
3. Monograph
4. Catalogue
“The synaptonemal complex is formed during _A_
stage and dissolves during _B_ stage”.
Complete the above statement by choosing the correct
option for A and B
| A | B | |
| 1. | Diplotene | Diakinesis |
| 2. | Leptotene | Zygotene |
| 3. | Zygotene | Diplotene |
| 4. | Pachytene | Diplotene |
Chromosomes appear like a ‘ball of wool’ in
(1) Prophase (2) Telophase
(3) Anaphase (4) Metaphase
Which one of the following features differentiates G2
phase from G1 phase?
| 1. | Synthesis of proteins. |
| 2. | 4C content of DNA. |
| 3. | 2C content of DNA. |
| 4. | Synthesis of RNA. |
Endoplasmic reticulum which is free of ribosomes is involved in all the given functions, except
| 1. | Synthesis of enzyme precursor for lysosomes. |
| 2. | Detoxification of drugs. |
| 3. | Uptake and release of Ca2+ ions during muscle contraction. |
| 4. | Glycogen synthesis. |
A membraneless cell organelle that is not found in higher plants is/has
| 1. | 9+2 arrangement of microtubules. |
| 2. | a role in formation of flagella in prokaryotes. |
| 3. | composed of rRNA and proteins. |
| 4. | involved in the formation of spindle fibers during cell division in animals. |
Enzymes and electron carriers for the formation of
cellular energy are present in the mitochondria at
| 1. | Outer membrane only. |
| 2. | Inner membrane only. |
| 3. | Both outer and inner membrane. |
| 4. | Mitochondrial matrix only. |
Endoplasmic reticulum plays a role in origin of A which is lined by B and contains a fine tubule called C . Select the option that correctly fills the blanks:
A B C
1. Lysosome Plasma Membrane Golgi body
2. Cytoskeleton Vacuole Desmotubule
3. Plasmodesmata Plasma Membrane Desmotubule
4. Plasmodesmata Cell Wall Vacuole
Select the correct statement with respect to axoneme of eukaryotic flagella.
| 1. | It is composed of 9 peripheral triplet microfibrils of tubulin proteins. |
| 2. | It does not have covering of plasma membrane. |
| 3. | Central tubules are enclosed by a central sheath. |
| 4. | It gives rise to spindle apparatus during cell division. |
Nucleolus is
1. Bounded by a single membrane.
2. Always one per cell.
3. Present inside mitochondria.
4. The site for rRNA synthesis.
Select the features which are common for both ER and Golgi apparatus.
| (A) | Both are sites for synthesis of lipids and steroidal hormones. |
| (B) | Both are composed of cisternae, tubules and vesicles. |
| (C) | Both are parts of endomembrane system. |
| (D) | Both help in formation of plasma membrane during cytokinesis. |
1. Only (B)
2. Only (B) and (C)
3. Only (A) and (D)
4. All except (D)
What is the main arena of all cellular activities of a
cell?
1. Cell wall
2. Cell membrane
3. Nucleus
4. Cytoplasm
Which of the following is correct for the function of calcium?
1. Helps in the synthesis of middle lamella.
2. It is required during the formation of spindle fibres.
3. It is required by meristematic as well as
differentiating tissues.
4. All of the above.
The symptoms due to toxicity of elements are difficult to identify as
1. Toxicity of micronutrients lead to the deficiency of macronutrients express the symptoms.
2. Deficiency of macronutrient leads to the toxicity micronutrients.
3. Toxicity of macronutrient leads to the deficiency of micronutrient.
4. More than one option is correct.
In plant cells, synthesis of ATP by the chemiosmotic
mechanism occurs during
| 1. | Photosynthesis only. |
| 2. | Respiration only. |
| 3. | Both photosynthesis and respiration. |
| 4. | Photorespiration only. |
In a plant system, the rate of photosynthesis may
become equal to the rate of respiration at
(1) CO2 compensation point.
(2) Light coompensation point.
(3) Light saturation point.
(4) More than one option is correct.
Which of the following plant hormones would most likely
be found in high concentrations in a mature, slightly
over-ripe fruit
1. Cytokinin and ethylene.
2. ABA and ethylene.
3. GA and ABA.
4. Auxin and cytokinin
The "sodium-potassium pump" pumps ______.
1. sodium ions out and potassium ions in
2. sodium ions in and potassium ions out
3. sodium and potassium ions in
4. sodium and potassium ions out
Which of the following is not a characteristic of
the sympathetic system?
1. inhibits the digestive tract
2. dilates the bronchi
3. accelerates the heartbeat
4. constricts the pupil
The iris __________.
1. regulates light entrance through the pupil
2. refracts light rays
3. absorbs stray light rays
4. contains receptors for sight
The most versatile joints that permit the most
movement are the _____________.
1. hinge joints
2. ball-and-socket joints
3. fibrous and cartilaginous joints
4. vertebral joints
Creatine phosphate ____________.
1. can be used in the reaction: actin + myosin =
actomyosin
2. is used to regenerate ADP to ATP
3. is the molecule that triggers contraction
4. forms the core strand of myosin
The sliding filament theory of muscle contraction
involves
1. calcium ions releasing ATP for energy
2. calcium ions binding with troponin, which shifts
tropomyosin and allows the myosin-binding sites
on actin to be exposed
3. neuromuscular junctions directly producing the movement of actin and myosin
4. actin filaments moving the myosin filaments in
each sarcomere
The receptors for non-steroid peptide hormones
are on the ______.
1. plasma membrane
2. nuclear envelope
3. DNA receptor complex
4. peptide chain
Perhaps the biggest reason for the enormous success of Arthropods is?
1. Chitinous exoskeleton
2. Diverse appendages
3. Respiration by tracheoles
4. Internal fertilization
If insufficient PTH is produced, the blood calcium level drops, resulting in ______.
1. reduced growth in childhood or parathyroid dwarfism
2. tetany, where the body shakes from continuous muscle contraction
3. osteoporosis
4. blood clotting
A person with Addison disease ___________.
| 1. | is unable to replenish blood glucose levels under stressful conditions |
| 2. | develops dramatically more male features |
| 3. | develops a rounded face and edema |
| 4. | has overgrowth of hands and face |
The filterable components of the blood that
move across from the glomerulus are______.
1. water, nitrogenous wastes, nutrients, and salts
2. blood cells and platelets and proteins
3. albumin, fats and other proteins
4. mainly blood cells
Drinking alcohol causes diuresis because it
inhibits the secretion of ________.
1. ANH
2. ADH
3. angiotensin
4. aldosterone
The maximum volume of air that can be moved in and out during a single breath is called the_________.
| 1. | vital capacity | 2. | tidal volume |
| 3. | residual volume | 4. | dead space |
The diaphragm and external intercostal muscles are ________ when expiration occurs.
| 1. | contracted |
| 2. | relaxed |
| 3. | flexed |
| 4. | both relaxed (diaphragm) and flexed (intercostal muscles) |
When blood levels of carbon dioxide rise, the rate and depth of breathing _________.
| 1. | decreases | 2. | increases |
| 3. | stays the same | 4. | stops |
Which of the following is not a character of deuterostomes?
1. Radial cleavage
2. Schizocoelous coelom
3. Indeterminate cleavage
4. Blastopore forms anus
Which of the following statements is NOT correct?
1. Microvilli increase the surface area of the small intestine for the absorption of nutrients.
2. Enzymes located on the brush border finish the digestion of chyme.
3. Absorption is an active process in the small intestine.
4. Sugars and amino acids cross columnar epithelial cells to enter the lacteal.
Which of the following comparisons is NOT
correct?
1. jaundice--abnormally large amount of bilirubin
in the skin and the whites of the eyes
2. ulcer--open sore in the wall of the stomach
3. peritonitis--inflammation of the cecum
4. cirrhosis--liver first becomes fatty and then replaced
by fibrous scar tissue
When the heart beats, the familiar lub-dub
sound occurs as the valves of the heart _____.
1. open
2. close
3. expand
4. contract
The greater the number of blood vessels dilated, the ________.
1. higher the blood pressure
2. lower the blood pressure
3. faster the heartbeat
4. slower the heartbeat
If blood is transferred to a test tube and is prevented
from clotting, it separates into ___.
1. formed elements and plasma
2. granular leukocytes and agranular leukocytes
3. red blood cells and white blood cells
4. inorganic and organic substances
The male and female cockroaches can be distinguished by their:
1. size
2. wings
3. anal styles
4. color
Without the presence of enzymes, the reactions
necessary to sustain life would require
___________________ in order to occur.
1. larger cells
2. higher temperatures
3. larger proteins
4. smaller atoms
Cofactors
1. break hydrogen bonds in proteins
2. help facilitate enzyme activity
3. increase activation energy
4. are very rare in living organisms
In the formation of a macromolecule, what type of reaction would join two subunits together?
| 1. | hydrophobic reaction | 2. | hydrolysis reaction |
| 3. | dehydration reaction | 4. | denaturation reaction |
How much of the cardiac output passes through the kidneys?
(1) 10%
(2) 20%
(3) 50%
(4) 65%
Which of the following statements is not correct with respect to the epithelial tissue?
| 1. | It covers the external surface of the body and the internal surface of many organs. |
| 2. | The neighbouring cells are held together by cell junctions and there is very little intercellular material. |
| 3. | The epithelial cells rest on a cellular basement membrane that separates it from underlying connective tissue. |
| 4. | There is no blood vessel supplying the nutrients to the epithelial cells. |
When secretory granules leave cell by exocytosis with no loss of other cellular material, the glands are called
1.Holocrine 2.Merocrine
3. Apocrine 4. Autocrine
The mucosa of the bladder is comprised of _____.
1. smooth muscle
2. squamous epithelium
3. transitional epithelium
4. simple columnar epithelium
Which of these could appear in the urine from dieting or the utilization of excess lipids?
(1) urea
(2) uric acid
(3) glycine
(4) Ketone
Which area actually secretes renin into the blood?
| 1. | macula densa |
| 2. | juxtaglomerular apparatus |
| 3. | juxtaglomerular cells |
| 4. | cortical nephron |
Intercalated discs are associated with what type of tissues/muscles?
1. dense regular connective tissue
2. dense irregular connective tissue
3. cardiac muscle
4. skeletal muscle
Which of the following is mismatched?
1. matrix - extracellular material in connective tissue
2. cardiac muscle cells - uninucleate.
3. chondrocytes - cartilage cells
4. ligaments - bind muscles to bone
Endocrine and exocrine glands are formed from
what type of tissue?
1. connective
2. blood
3. muscle
4. epithelial
Goblet cells are specialized cells of the
1. stratified squamous epithelium
2. cuboidal epithelium
3. columnar epithelium
4. simple squamous epithelium
Which cells are the first to phagocytize foreign
particles in the tissues such as bacteria in a
wound?
1. fibroblasts
2. white blood cells
3. macrophages
4. mast cells
The air sacs in the lungs are comprised of
_____ cells.
1. simple squamous
2. simple columnar
3. simple cuboidal
4. transitional
The sebaceous glands of the skin produce secretions
by the _____ method of secretion.
1. merocrine
2. eccrine
3. holocrine
4. apocrine
Neuroglial cells help neurons in each of these
ways, with the exception of _______________.
1. supporting and binding nervous tissue
2. carrying on phagocytosis
3. playing a role in cell-to-cell communications
4. transmitting nervous impulses
Mark the correct statement:
1. Electrical synapses are more common in our neural system than chemical synapses.
2. The new potential in the post-synaptic neuron may be either excitatory or inhibitory.
3. Hypothalamus is the major coordination center for sensory and motor signaling.
4. The tracts of nerve fibers that connect two cerebral hemispheres are called corpora quadrigemina.
Go through the following characterization of Sympathetic and. Parasympathetic Nervous System.
I. Dilates pupil,
II. Heart rate, BP (Vasoconstriction) Glycogenolysis, dilates bronchi.
III. Constricts pupil.
IV. Induces ejaculation.
V. Gastric secretion, salivary secretion, peristalsis.
VI. Erection and urination.
1. Sympathetic - III, V, VI; Parasympathetic - I, II, IV
2. Sympathetic - I, II, IV; Parasympathetic - III, V, VI
3. Sympathetic M II, IV, VI; Parasympathetic - I, III, V
4. Sympathetic - I, III, V; Parasympathetic - II, IV, VI
One example of animals having a single opening
to the outside that serves both as mouth
and as anus is
1. Octopus
2. Asterias
3. Ascidia
4. Fasciola
Which of the following groups of animals is
correctly matched with its one characterstic
feature without even a single exception?
| 1. | Mammalia: | give birth to young ones. |
| 2. | Reptilia: | possess 3-chambered heart with one incompletely divided ventricle |
| 3. | Chordata: | possess a mouth provided with an upper and a lower jaw |
| 4. | Chondrichthyes: | possess cartilaginous endoskeleton |
Planaria possess high capacity of
1. metamorphosis
2. regeneration
3. alternation of generation
4. bioluminescence
Some hormones can act on their target cells through second messengers. Identify the one that does not:
1. cortisol
2. adrenaline
3. FSH
4. calcitonin
The quantum number not obtained from Schrodinger’s wave equation is:
| 1. | \(n\) | 2. | \(l\) |
| 3. | \(m\) | 4. | \(s\) |
The incorrect statement about the nodal plane among the following is:
| 1. | A plane on which there is a zero probability of finding an electron. |
| 2. | A plane on which there is maximum probability that the electron will be found. |
| 3. | ψ2 is zero at nodal plane. |
| 4. | None of the above. |
The incorrect order of electronegativity is :
| 1. | Cl > S > P > Si | 2. | Si > Al > Mg > Na |
| 3. | F > Cl > Br > I | 4. | None of the above. |
Lattice energy of (I) , (II) and (III) are in the order -
1. I > II > III
2. I < II < III
3. I < III < II
4. II < I < III
combine readily because of the formation of -
1. a covalent bond
2. a hydrogen bond
3. a co-ordinate bond
4. an ionic bond
Which can not be explained by VBT -
1. Overlapping
2. Bond formation
3. Paramagnetic nature of oxygen
4. Shapes of molecules
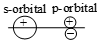
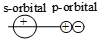
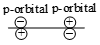
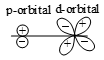
Which of the following leads to bonding ?
1. 
2. 
3. 
4. 
Which of the following pairs of species have similar shapes?
| 1. | PCl3, NH3 | 2. | CF4, SF4 |
| 3. | PbCl2, CO2 | 4. | PF5 , IF5 |
CHCH product
Product is -
1. Cu–CC–Cu
2. CH2=CH–CCH
3. CH=C–Cu
4. Cu–CC–NH4
B CH3–CCH A
A and B are -
1. ![]()
2. ![]()
3. CH3CH2CHO (both)
4. ![]()
For the reaction
B2H6 (g) + 3O2 (g) B2O3 (s) + 3H2O(v)
E = – 2143.2 kJ.
Calculate H for the reaction at 25ºC–
1. – 2145 kJ mole–1
2. – 2138.6 kJ mole–1
3. – 2133.2 kJ mole–1
4. – 2143.2 kJ mole–1
Using bond enthalpies (symbolized by e) an estimated value of DHº for the reaction
H2C=CH2 (g) + H2 (g) CH3–CH3 (g)
would be–
1.
2.
3.
4.
The standard heat of combustion of propane is –2220.1 kJ mol–1. The standard heat of vaporisation of liquid water is 44.0 kJ mol–1. The enthalpy change for the reaction is–
C3H8 (g) + 5O2 (g) 3CO2 (g) + 4H2O(g)
| 1. | –2220.1 kJ | 2. | –2044.1 kJ |
| 3. | –2396.1 kJ | 4. | –2176.1 kJ |
The standard heat of combustion of solid boron is equal to–
1. Hºf (B2O3)
2. 1/2 Hºf (B2O3)
3. 2Hºf (B2O3)
4. –1/2Hºf (B2O3)
Pure ammonia is placed in a vessel at a temperature where its dissociation constant (a) is appreciable. At equilibrium :
1. does not change significantly with pressure
2. does not change with pressure
3. concentration of does not change with pressure
4. concentration of hydrogen is less than that of nitrogen
One mole of at 300 K is kept in a closed container under one atmosphere. It is heated to 600 K when 20% by mass of decomposes to . The resultant pressure
is
1. 1.2 atm
2. 2.4 atm
3. 2.0 atm
4. 1.0 atm
The correct order of acidic strength is -
1. Cl2O7 >SO2>P4O10
2. CO2 >N2O5 <SO3
3. Na2O >MgO >Al2O3
4. K2O >CaO >MgO
Match the items under list (1) with items under list (2) select the correct answers from the sets (A), (B), (C) and (D) -
List (1) molecule
(a) PCl5 (b) F2O
(c) BCl3 (d) NH3
List (2) shape
(i) V-shaped
(ii) Triangular planar
(iii) Trigonal bipyramidal
(iv) Trigonal pyramidal
(v) Tetrahedral
1. a - i, b - v, c - iv, d - iii
2. a -ii, b -iii, c - i , d - ii
3. a - iv , b - iii, c - ii, d - v
4. a - iii, b - i, c - ii, d - iv
The equilibrium constant of the reaction A2(g) + B2(g) 2AB (g) at 100ºC is 50. If a one litre flask containing one mole of A2 is connected to a two litre flask containing two moles of B2, how many moles of AB will be formed at 373 K ?
1. 2.8
2. 1.9
3. 2.1
4. 3.6
In the preparation of quick lime from lime stone, the reaction CaCO3(s) CaO(s) + CO2(g) led to a set of Kp values fitting an empirical equation:
log Kp = 7.282 –
If the reaction is carried out in open air, what temperature would be predicted from this equation for complete decomposition of limestone?
1. T = 1167.26 K
2. T = 1670.55 K
3. T = 1650.80 K
4. None of these
For the equilibrium
LiCl.3NH3(s) LiCl.NH3(s) + 2NH3
KP = 9 atm2, at 40ºC. A 5 litre contains 0.1 mole of LiCl.NH3. How many moles of NH3 should be added to the flask at this
temperature to drive the backward reaction for completion
1. 0.78
2. 0.80
3. 0.90
4. None of these
A sample of CaCO3(s) is introduced into a sealed container of volume 0.821 litre and heated to 1000 K until equilibrium is reached. The equilibrium constant for the reaction CaCO3(s) CaO(s) + CO2(g) is 4 × 10–2 atm at this temperature. The mass of CaO is present at equilibrium is-
1. 22.4 mg
2. 25 mg
3. 27.2 mg
4. None of these
At 100 ºC and 1 atm, if the density of liquid water is 1.0 g cm–3 and that of water vapor is 0.0006 g cm-3, then the volume occupied by water molecules in 1 litre of steam at that temperature will be:
| 1. | 6 cm3 | 2. | 60 cm3 |
| 3. | 0.6 cm3 | 4. | 0.06 cm3 |
A 2L solution (X) contain 0.02 mole of [Co(NH3)5SO4]Br and 0.02 mol [Co(NH3)5 Br] SO4. 2 L of this solution is -
taken :
X + AgNO3 (excess) Y mol AgBr
Y + BaCl2 (excess) mol BaSO4 + Z
Values of Y and Z are -
1. 0.01, 0.02 2. 0.02, 0.02
3. 0.02, 0.01 4. 0.01, 0.01
What volume (in ml) at STP of SO2 gas is oxidized by 100 ml of 0.1 (M) H2Cr2O7 in acid solution ?
1. 672 ml 2. 224 ml
3. 448 ml 4. 112 ml
25 ml of a 0.1 (M) solution of a stable cation of transition metal z reacts exactly with 25 ml of 0.04 (M) acidified KMnO4 solution. Which of the following is most likely to represent the change in oxidation state of z correctly ?
1.
2.
3.
4.
In context with beryllium, which one of the following statements is incorrect?
1. It is rendered passive by nitric acid
2. lt forms Be2C
3. Its salts rarely hydrolyse
4. Its hydride is electron-deficient and polymeric
The function of "Sodium pump" is a biological process operating in each and every cell of all animals. Which of the following biologically important ions is also a constituent of this pump?
1. Ca2+ 2. Mg2+ 3. K+ 4. Fe2+
Alumina is insoluble in water because -
1. It is a covalent compound
2. It has high lattice energy and low heat of hydration
3. It has low lattice energy and high heat of hydration
4. Al3+ and O2– ions are not excessively hydrated.
The structure of aluminium bromide is best represented as -
The compound which has one isopropyl group is -
1. 2,2,3,3-tetramethyl pentane
2. 2,2-dimethyl pentane
3. 2,2,3-trimethyl pentane
4. 2-methyl pentane
The IUPAC name of the compound is:
1. 1-amino-1-phenyl-2-methyl propane
2. 2-methyl-1-phenyl propanamine
3. 2-methyl-1-amino-1-phenyl propane
4. 1-isopropyl-1-phenyl methyl amine
The IUPAC name of is ?
1. 2-chloro-4-N-ethylpentanoic acid
2. 2-chloro-3-(N, N-diethyl amino)-propanoic acid
3. 2-chloro-2-oxo diethylamine
4. 2-chloro-2-carboxy-N-ethyl ethane
When temperature is increased -
1. % of eclipsed form increses
2. % of skew form increases
3. % of staggered form increases
4. No effect on any form
Which of the following compounds can not exist as enantiomers -
1. CH3–CH(OH) COOH
2.
3. C6H5CH2–CH3
4. C6H5CHClCH3
Pair of groups exerting (–I) effect is -
1. – NO2 & – CH3
2. – NO2 & – Cl
3. – Cl & – CH3
4. – CH3 & – C2H5
'M' effect is the resonance of -
1. electrons only
2. electrons only
3. and both
4. (+) ve and (–) charge.
In which of the following, delocalization of positive charge is possible?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Which of the following is/are not correctly ordered for resonance stability -
A gas at a pressure of 5.0 atm is heated from 0º to 546 ºC and simultaneously compressed to one-third of its original volume. Hence final pressure is -
1. 10.0 atm
2. 30.0 atm
3. 45.0 atm
4. 5.0 atm
An open vessel containing air is heated from 300 K to 400 K. The fraction of air originally present which goes out of it is at 400 K -
1. 3/4
2. 1/3
3. 2/3
4. 1/8
N2 + 3H2 2NH3. 1 mol N2 and 4 mol H2 are taken in 15 L flask at 27 ºC. After complete conversion of N2 into , 5 L of H2O is added. Pressure set up in the flask is -
1.
2.
3.
4.
A sample of air contains only N2, O2 and H2O. It is saturated with water vapours and total pressure is 640 torr. The vapour pressure of water is 40 torr and the molar ratio of N2 : O2 is 3 : 1. The partial pressure of N2 in the sample is -
1. 540 torr
2. 900 torr
3. 1080 torr
4. 450 torr
The phenomenon in which atmospheric gases trap the heat radiations from the sun near the earth’s surface and keeps it warm is known as-
1. Natural greenhouse effect
2. Tyndall effect
3. Heating effect
4. Joule’s effect
The gas that damages the ozone layer is -
1. CFCs
2. CO2
3. CH4
4. SO2
A physical quantity X is expressed as
v (velocity) =
The dimensions of X are
1. ML–1T–2
2. MLT–2
3. MLT–1
4. ML–1T–3
The ratio of maximum and minimum magnitudes of the resultant of two vectors and is 3:2. The relation between A and B is
1. A = 5B
2. 5A = B
3. A = 3B
4. A = 4B
In head on collision of two point particles, loss
in kinetic energy is given by
With usual notations (except k), the
dimensional formula of quantity k is
1. [M0L0T–1]
2. [M0L0T0]
3. [M0LT–1]
4. [M0L2T–2]
A particle starting from the point \((1,2)\) moves in a straight line in the \(XY\)-plane. Its coordinates at a later time are \((2,3).\) The path of the particle makes what angle with the \(x\)-axis?
1. \(30^\circ\)
2. \(45^\circ\)
3. \(60^\circ\)
4. data is insufficient
A ball is thrown vertically upwards with a velocity \(u\) with respect to ground from a balloon descending with velocity \(v\) with respect to ground. The ball will pass the balloon after time:
1. \(\frac{u-v}{2g}\)
2. \(\frac{u+v}{2g}\)
3. \(\frac{2(u-v)}{g}\)
4. \(\frac{2(u+v)}{g}\)
A projectle is fired horizontally from an
inclined plane (of inclination 45° with
horizontal) with speed =50 m/s. If g=10m/s2,
the range measured along the incline is
1. 500 m
2. 500 m
3. 200m
4. none of these
Two particles are projected from a point at the same instant with velocities whose horizontal and vertical components are u1, v1 and u2, v2 respectively. The interval between their passing through the other common point of their path is
1.
2.
3.
4.
A ball is thrown vertically upwards from the ground. It crosses a point at the height of 25 m twice at an interval of 4 secs. The ball was thrown with the velocity of
1. 20m/sec.
2. 25 m/sec.
3. 30m/sec.
4. 35 m/sec
In the shown system, each of the blocks is at rest. The value of \(\theta\) is:

1. \(\tan^{- 1} \left(1\right)\)
2. \(\tan^{- 1} \left(\frac{3}{4}\right)\)
3. \(\tan^{- 1} \left(\frac{4}{3}\right)\)
4. \(\tan^{- 1} \left(\frac{3}{5}\right)\)
A force F acting on a body depends on its
displacement x as F ∝ xn. The power delivered
by F will be independent of x if n is–
1.
2.
3.
4.
A block of mass \(1\) kg lying on the floor is subjected to a horizontal force given by, \(F=2\sin\omega t\) newtons. The coefficient of friction between the block and the floor is \(0.25\). The acceleration of the block will be:
1. positive and uniform
2. positive and non–uniform
3. zero
4. depending on the value of \(\omega\).
A person used force (F), shown in figure to
move a load with constant velocity on given
surface.Identify the correct surface profile :-
1.
2.
3.
4.
A body of mass m dropped from a height h reaches the ground with a speed of 1.4. The work done by air drag is:
1. –0.2mgh
2. –0.02mgh
3. –0.04mgh
4. mgh
A weightless string passes through a slit over
a pulley. The slit offers frictional force f to
the string. The string carries two weights
having masses m1 and m2 where m2 > m1, then
acceleration of the weights is
1.
2.
3.
4.

| 1. | \( \sqrt{\dfrac{2 g \sin \theta}{L}\left(L^2-b^2\right)} \) | 2. | \( \sqrt{\dfrac{g \sin \theta}{2 L}\left(L^2-b^2\right)} \) |
| 3. | \( \sqrt{\dfrac{g \sin \theta}{L}\left(L^2-b^2\right)}\) | 4. | None of these |
A metal block is resting on a rough wooden
surface. A horizontal force applied to the block
is increased uniformly. Which of the following
curves correctly represents velocity of the
block ?
1.
2.
3.
4.
Choose the correct option-
1. If only conservative forces act on a particle,
the kinetic energy remains constant.
2. If the net force acting on an object is zero,
then the object is at rest.
3. If net mechanical work is done on a body,
the body must accelerate.
4. If net mechanical work is done on a body,
the speed of body remains unchanged.
A particle A of mass 1kg moves along the line
3x – 4y = 0 with a speed of 10 ms–1 and another
particle B of mass 2kg moves along the
line 12y + 5x = 0 with a speed of v ms–1. Both
of them start simultaneously from the origin,
with A and B moving in first and fourth quadrants
respectively, such that their centre of
mass is always on x-axis. The value of v is :-
1.
2.
3.
4. None
A disc of the radius R is confined to roll without slipping at A and B. If the plates have the velocities v and 2v as shown, the angular velocity of the disc is
1. Anticlockwise
2. Clockwise
3. Anticlockwise
4. Clockwise
A uniform rod of mass 2M is bent into four adjacent semicircles, each of radius r, all lying in the same plane. The moment of inertia of the bent rod about an axis through one end A and perpendicular to the plane of the rod is:
1. 22
2. 88
3. 44
4. 66
A hollow cylinder with an inner radius of R, an outer radius of 2R, and a mass M is rolling without slipping at the speed of its centre v. Its kinetic energy will be:
1.
2.
3.
4. None of these
A particle of mass M is at a distance 'a' from
center of a thin spherical shell of equal mass
and having radius 'a'. Select correct alternative.
1. Gravitational field and potential both are
zero at centre of the shell.
2. Gravitational field is zero not only inside
the shell but at a point outside the shell
also.
3. Inside the shell, gravitational field alone
is zero.
4. Neither gravitational field nor gravitational
potential is zero inside the shell.
A particle is projected vertically upwards the
surface of the earth (radius Re) with a speed equal
to one fourth of escape velocity. What is the
maximum height attained by it from the surface
of the earth ?
1.
2.
3.
4. None
The following figure shows two air-filled
bulbs connected by a U-tube partly filled with
alcohol. What happens to the levels of alcohol
in the limbs X and Y when an electric bulb
placed midway between the bulbs is lighted?
1. The level of alcohol in limb X falls while
that in limb Y rises
2. The level of alcohol in limb X rises while
that in limb Y falls
3. The level of alcohol falls in both limbs
4. There is no change in the levels of alcohol
in the two limbs
A rubber cord has a cross-sectional area \(1~\text{mm}^2\) and the total unstretched length \(10~\text{cm}.\) It is stretched to \(12~\text{cm}\) and then released to project a mass of \(80~\text g.\) The Young's modulus for rubber is \(5\times10^{8}~\text{N-m}^2.\) What would be the velocity of mass when the rubber cord is unstretched?
1. \(5~\text{m/s}\)
2. \(3~\text{m/s}\)
3. \(7~\text{m/s}\)
4. \(2~\text{m/s}\)
A liquid drop at temperature T, isolated from its surroundings, breaks into a number of droplets. The temperature of the droplets will be–
| 1. | equal to T |
| 2. | greater than T |
| 3. | less than T |
| 4. | either (1), (2) or (3) depending on the surface tension of the liquid. |
Find the rate of flow of glycerin of density 1.25 × 103 kg/m3 through the conical section of a pipe, if the radii of its ends are 0.1 m and 0.04 m and the pressure drop across its length is 10 N/m2.
1. 3.14 × 10–4 m3/s
2. 6.28 × 10–4 m3/s
3. 12.56 × 10–4 m3/s
4. 1.57 × 10–4 m3/s
Water flows through a frictionless duct with a cross-section varying as shown in the figure. Pressure \(P\) at points along the axis is represented by:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Statement 1 : When a large soap bubble and
a small soap bubble are connected by a
capillary tube, the large bubble expands while
the small bubble shrinks.
and
Statement 2 : The excess pressure inside a
bubble is inversely proportional to radius and
air flows from small bubble to large bubble.
1. Statement-1 is ture, statement-2 true;
statement-2 is the correct explanation of
statement-1
2. Statement-1 is false, statement-2 is true
3. Statement-1 is ture, statement-2 is false
4. Statement-1 is true, statement-2 is true;
statement -2 is not correct explanation of
statement-1
The graph \(AB\) shown in the figure is a plot of the temperature of a body in degrees Celsius and degrees Fahrenheit. Then:

| 1. | the slope of the line \(AB\) is \(9/5\) |
| 2. | the slope of the line \(AB\) is \(5/9\) |
| 3. | the slope of the line \(AB\) is \(1/9\) |
| 4. | the slope of the line \(AB\) is \(3/9\) |
At ordinary temperatures, the molecules of a diatomic gas have only translational and rotational kinetic energies. At high temperatures, they may also have vibrational energy. As a result of this compared to lower temperatures, a diatomic gas at higher temperatures will have:
| 1. | lower molar heat capacity. |
| 2. | higher molar heat capacity. |
| 3. | lower isothermal compressibility. |
| 4. | higher isothermal compressibility. |
At a pressure of \(24\times 10^{5}~\text{dyne/cm}^2\), the volume of \(O_2\) is \(10\) litre and mass is \(20\text{g}\). The rms velocity will be:
| 1. | \(800~\text{m/s}\) | 2. | \(400~\text{m/s}\) |
| 3. | \(600~\text{m/s}\) | 4. | Data is incomplete. |
A horizontal cylinder has two sections of unequal cross-sections in which two pistons, A and B, can move freely. The pistons are joined by a string. Some gas is trapped between the pistons. If this gas is heated, the pistons will:
| 1. | move to the left. |
| 2. | move to the right. |
| 3. | remain stationary. |
| 4. | move either to the left or to the right depending on the initial pressure of the gas. |
In the \((P\text-V)\) diagram shown, the gas does \(5~\text J\) of work in the isothermal process \(ab\) and \(4~\text J\) in the adiabatic process \(bc.\) What will be the change in internal energy of the gas in the straight path from \(c\) to \(a?\)

1. \(9~\text J\)
2. \(1~\text J\)
3. \(4~\text J\)
4. \(5~\text J\)
\(ABCA\) is a cyclic process. Its \(P\text-V\) graph would be:

| 1. |  |
2. |  |
| 3. |  |
4. |  |
When \(x\) amount of heat is given to a gas at constant pressure, it performs \(\frac{x}{3}\) amount of work. The average number of degrees of freedom per molecule of the gas is:
1. \(3\)
2. \(4\)
3. \(5\)
4. \(6\)
A block is given a initial push along rough horizontal fixed table surface and after some time it stops due to friction. For the system of block and table,
1. temperature increase
2. heat supplied is positive
3. heat supplied is negative
4. total energy decreases
The shape of a wave propagating in the positive \(x\) or negative \(x\) direction is given by; \(y=\frac{1}{\sqrt{1+x^2}}\) at \(t = 0 \) and \(y=\frac{1}{\sqrt{2-2x+x^2}}\) at \(t = 1~\text s\) where \( x\) and \(y\) are in meters. The shape of the wave disturbance does not change during propagation, then the velocity of the wave is:
1. \(1~\text{m/s}\) in the positive \( x\) direction
2. \(1~\text{m/s}\) in the negative \( x\) direction
3. \(\dfrac{1}{2}~\text{m/s}\) in the positive \( x\) direction
4. \(\dfrac{1}{2}~\text{m/s}\) in the negative \( x\) direction
Two pendulums (with the same angular amplitudes) of time periods \(3~\text{s}\) and \(7~\text{s}\) respectively start oscillating simultaneously from two opposite extreme positions with the same angular amplitudes. After how much time they will be in phase?
1. \(\dfrac{21}{8}~\text{s}\)
2. \(\dfrac{21}{4}~\text{s}\)
3. \(\dfrac{21}{2}~\text{s}\)
4. \(\dfrac{21}{10}~\text{s}\)
An observer moving at a velocity of 20 m/s is moving away from a source moving at a speed of 10 m/s towards the observer.
If the frequency of the source is 'f', then find the frequency observed by the observer is: ( )
1.
2.
3.
4. f
Two particles A and B are performing SHM along x and y-axis respectively with equal amplitude and frequency of 2 cm & 1Hz respectively. Equilibrium positions of the particles A and B are at the
coordinates (3 cm, 0) and (0, 4 cm) respectively. At t = 0, B is at its equilibrium position and moving towards the origin, while A is nearest to the origin and moving away from the origin.
Equation of motion of particle A can be written as-
1. x = (2 cm) cos 2t
2. x = (3 cm) – (2 cm) cos 2t
3. x = (2 cm) sin 2t
4. x = (3 cm) – (2 cm) sin 2t
Two particles A and B are performing SHM along x and y-axis respectively with equal amplitude and frequency of 2 cm & 1Hz respectively. Equilibrium positions of the particles A and B are at the coordinates (3 cm, 0) and (0, 4 cm) respectively. At t = 0, B is at its equilibrium position and moving towards the origin, while A is nearest to the origin and moving away from the origin.
Minimum and maximum distance between A and B during the motion is-
1. cm and cm
2. 3 cm and 7 cm
3. 1 cm and 5 cm
4. 9 cm and 16 cm
In each of the following questions, Match column I and column II and select the correct match out of the four given choices-
| Column I | Column II | ||
| \(\mathrm{(A)}\) |  |
\(\mathrm{(I)}\) | \(T=2\pi\sqrt{\dfrac{m(k_1+k_2)}{k_1k_2}}\) |
| \(\mathrm{(B)}\) |  |
\(\mathrm{(II)}\) | \(T=2\pi\sqrt{\dfrac{m}{(k_1+k_2)}}\) |
| \(\mathrm{(C)}\) |  |
\(\mathrm{(III)}\) | \(T=2\pi\sqrt{\dfrac{m}{k}}\) |
| \(\mathrm{(D)}\) |  |
\(\mathrm{(IV)}\) | \(T=2\pi\sqrt{\dfrac{m}{2k}}\) |
| 1. | \(\mathrm{A\text-I,B\text- II,C\text-III,D\text-IV}\) |
| 2. | \(\mathrm{A\text-II,B\text-I ,C\text- III,D\text-IV}\) |
| 3. | \(\mathrm{A\text- II,B\text-I ,C\text- IV,D\text- III}\) |
| 4. | \(\mathrm{A\text- I,B\text- II,C\text-IV,D\text-III}\) |