| 1. | \(K_a \times (\wedge^\circ _M )^2\) | 2. | \(K_a \times {1 \over (\wedge^\circ _M )^3}\) |
| 3. | \(K_a \times {1 \over (\wedge^\circ _M )^2}\) | 4. | \(K_a\times (\wedge^\circ _M )^3 \) |
| 1. | \(1.2 \times 10^{-43}\) | 2. | \(2.4 \times 10^{-73}\) |
| 3. | \(6.3 \times 10^{-92}\) | 4. | \(1.5 \times 10^{-62}\) |
| Statement I | For \(\mathrm{Kl},\) molar conductivity increases steeply with dilution. |
| Statement II | For carbonic acid, molar conductivity increases slowly with dilution. |
| 1. | Statement I is incorrect and Statement II is correct. |
| 2. | Both Statement I and Statement II are correct. |
| 3. | Both Statement I and Statement II are incorrect. |
| 4. | Statement I is correct and Statement II is incorrect. |

| 1. | 0.03 V | 2. | 0.06 V |
| 3. | -0.06 V | 4. | -0.03 V |
| 1. | Cathode to the anode in solution. |
| 2. | Cathode to anode through external supply. |
| 3. | Cathode to anode through the internal supply. |
| 4. | Anode to the cathode through the internal supply. |
| 1. | The corrosion of iron can be minimised by forming contact with another metal with a higher reduction potential. |
| 2. | Iron corrodes in oxygen-free water |
| 3. | The corrosion of iron can be minimised by forming an impermeable barrier |
| 4. | Iron corrodes more rapidly in salt water because its electrochemical potential is higher. |
| List-I | List-II | ||
| (P) |  |
(i) | Conductivity decreases and then increases. |
| (Q) |  |
(ii) | Conductivity decreases and then does not change much. |
| (R) |  |
(iii) | Conductivity increases and then does not change much. |
| (S) |  |
(iv) | Conductivity does not change much and then increases. |
| P | Q | R | S | |
| 1. | (iii) | (iv) | (ii) | (i) |
| 2. | (iv) | (iii) | (ii) | (i) |
| 3. | (ii) | (iii) | (iv) | (i) |
| 4. | (i) | (iv) | (iii) | (ii) |
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. | \(\small{\mathrm{Cd}_{(\mathrm{s})}+\mathrm{NiO}_{2(\mathrm{~s})}+2 \mathrm{H}_2 \mathrm{O}_{(\mathrm{l})} \rightarrow \mathrm{Cd}(\mathrm{OH})_{2(\mathrm{~s})}+\mathrm{Ni}(\mathrm{OH})_{2(\mathrm{~s})}}\) |
| 2. | \(\small {Cd_{(s)} + NiO_{2{(s)}} + 2OH^-_{(aq)} \rightarrow Ni{(s)} + Cd(OH)_{2{(s)}}}\) |
| 3. | \(\small {Ni_{(s)}+ Cd(OH)_{2{(s)} }\rightarrow Cd_{(s)}+ Ni(OH)_{2{(s)}}}\) |
| 4. | \(\small {Ni(OH)_{2(s)}+Cd(OH)_{2(s)} \rightarrow Ni_{(s)} + Cd_{(s)}+2H_2O(l)}\) |
| 1. | \(I^-(aq) |I_2(s) || Br_2(l) |Br^-(aq) \) |
| 2. | \(Zn(s) | Zn^{2+} (aq) || Pb^{2+} (aq) | Pb(s) \) |
| 3. | \(Pt,H_2(g) |H_2O(l) | OH^-(aq) ||O_2(g) |OH^- (aq), Pt\) |
| 4. | \(Ag(s) | Ag^+ (aq) || Ag^+ (aq) |Ag(s) \) |
| (A) | During charging, PbSO4 on the anode is converted into PbO2. |
| (B) | During charging, PbSO4 on the cathode is converted into PbO2. |
| (C) | The lead storage battery consists of a grid of lead packed with PbO2 as the anode. |
| (D) | The lead storage battery uses a ~38% solution of sulfuric acid as an electrolyte. |
| Assertion (A): | Molar conductivity increases with a decrease in concentration. |
| Reason (R): | For strong electrolytes, \(Λ_m\) increases slowly with dilution and can be represented by the equation: \(\Lambda_m=\Lambda_m^0-A c^{1 / 2}\) |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
| Assertion(A): | \(\mathrm{E^0_{cell}}\) = 0 for a chloride ion concentration cell. |
| Reason(R): | For this concentration cell, the equation is given by: \(E_{\mathrm{cell}}=\frac{R T}{n F} \ln \left(\frac{\left[\mathrm{Cl}^{-}\right]_{\mathrm{LHS}}}{\left[\mathrm{Cl}^{-}\right]_{\mathrm{RHS}}}\right) \) |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
| Assertion (A): | 1 coulomb of electricity deposits 1 g-equivalent of substance. |
| Reason (R): | 1 Faraday is the charge on 1 mole of electricity. |
| 1. | Both (A) and (R) are True, and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True, but (R) is not the correct explanation of (A). |
| 3. | (A) is True, but (R) is False. |
| 4. | (A) is False, but (R) is True. |
Find the emf of the cell in which the following reaction takes place at 298 K:
\(\mathrm{Ni}(\mathrm{s})+2 \mathrm{Ag}^{+}(0.001 \mathrm{M}) \rightarrow \mathrm{Ni}^{2+}(0.001 \mathrm{M})+2 \mathrm{Ag}(\mathrm{s}) \)
\(\small{\text { (Given that } \mathrm{E}_{\text {cell }}^{\circ}=1.05 \mathrm{~V}; \dfrac{2.303 \mathrm{RT}}{\mathrm{F}}=0.059} )\)
| 1. | 1.05 V | 2. | 1.0385 V |
| 3. | 1.385 V | 4. | 0.9615 V |
| 1. | \(2.5 \times 10^{-3}\) | 2. | \(2 \times 10^{-3}\) |
| 3. | \(2.5 \times 10^{-4}\) | 4. | \(2 \times 10^{-4}\) |
| 1. | Mn | 2. | Zn |
| 3. | Cu | 4. | Fe |
For the following cell with hydrogen electrodes at two different pressures p1 and p2 , then the emf is given by:
Pt(H2) | H+(aq) |Pt (H2)
p1 1M p2
| 1. | \(\frac{R T}{F} \log _{e} \frac{P_{1}}{p_{2}}\) | 2. | \(\frac{R T}{2F} \log _{e} \frac{P_{1}}{p_{2}}\) |
| 3. | \(\frac{R T}{F} \log _{e} \frac{P_{2}}{p_{1}}\) | 4. | \(\frac{R T}{2F} \log _{e} \frac{P_{2}}{p_{1}}\) |
The decreasing order of electrical conductivity of the following aqueous solutions is :
0.1 M Formic acid (A),
0.1 M Acetic acid (B),
0.1 M Benzoic acid (C)
1. A > B > C
2. A > C > B
3. C > B > A
4. C > A > B
The value of Ecell in the reaction below will be:
\(\small{Pt(s)|Br^{-}(0.010 \ M)|Br_{2}(l) \ ||H^{+}(0.030 \ M)|H_{2}(g)(1 \ bar)|Pt(s)}\)
\(E_{Br^{-}/Br_{2}}^{o} \ = \ -1.09 \ V\)
1. +1.298 V
3. –1.298 V
4. –1.198 V
In electrolysis of NaCl when Pt electrode is taken then H2 is liberated at the cathode while with Hg cathode it forms sodium amalgam because:
| 1. | Hg is more inert than Pt |
| 2. | More voltage is required to reduce H+ at Hg than at Pt |
| 3. | Na is dissolved in Hg while it does not dissolve in Pt |
| 4. | The concentration of H+ ions is larger when the Pt electrode is taken |
For the disproportionation of copper:
2Cu+ → C u2+ + C u is:
(Given for Cu+2/Cu is 0.34 V & Eº for Cu+2/Cu+ is 0.15 V )
1. 0.49 V
2. – 0.19 V
3. 0.38 V
4. – 0.38 V
Given below are two statements:
| Assertion (A): | \(E^\circ\)Ag+/ Ag increases with an increase in the concentration of Ag+ ions. |
| Reason (R): | \(E^\circ\)Ag+/ Ag has a positive value. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
The incorrect statement about an inert electrode in a cell is:
| 1. | It does not participate in the cell reaction. |
| 2. | It provides a surface either for oxidation or for the reduction reaction. |
| 3. | It provides a surface for the conduction of electrons. |
| 4. | It provides a surface for redox reaction. |
One liter of 0.5 M KCl solution is electrolyzed for one minute in a current of 1.608 mA. Considering 100 % efficiency, the pH of the resulting solution will be :
| 1. | Seven (7) | 2. | Nine (9) |
| 3. | Eight (8) | 4. | Ten (10) |
Equivalent conductivity of at infinite dilution. If conductivity of saturated solution is x is:
(1)
(2)
(3)
(4)
A fuel cell develops an electrical potential from the combustion of butane at 1 bar and 298 K
C4H10(g) + 6.5O2(g) → 4 CO2(g) + 5H2O(l); Eo of a cell is:
(Given \(\Delta G^{o}\) \(= - 2746 k J / m o l e\) )
1. 4.74 V
2. 0.547 V
3. 4.37 V
4. 1.09 V
Consider the reaction given below:
The electrolysis of acetate solution produces ethane.
The current efficiency of the process is 80%.
What volume of gases would be produced at 27 and 740 torr,
if the current of 0.5 amp is passed through the solution for 96.45 min?
1. 6.0 L
2. 0.60 L
3. 1.365 L
4. 0.91 L
The specific conductivity of a saturated solution of KI3 is 4.59 × 10-6 ohm-1 cm-1 and it's molar conductance is 1.53 ohm-1 cm2 mol-1. The Ksp Of KI3 will be :
1. 4 x 10-12
2. 27 x 27 x 10-9
3. 9 x 10-6
4. 4 x 10-6
In which electrolysis pH of solution decreases?
1. NaCl solution using Pt electrodes
2. solution using Pt electrodes
3. solution using Cu electrodes
4. All of these
Equivalent conductance of saturated \(\mathrm {BaSO}_4\) solution is \(400 \mathrm { ~ohm}^{-1}\) \(\mathrm {cm}^2\) \(\mathrm { ~equivalent}^{-1}\) and it's specific conductance is \(8 \times 10^{-5} \text { ohm}^{-1} \text {cm}^{-1}\) ; hence solubility product \(K_{sp}\) of \(\mathrm {BaSO}_4\) is :
1. \(4 \times 10^{-8} \text {M}^2\)
2. \(1 \times 10^{-8} \text {M}^2\)
3. \(2 \times 10^{-4} \text {M}^2\)
4. \(1 \times 10^{-4} \text {M}^2\)
Consider the following Galvanic cell.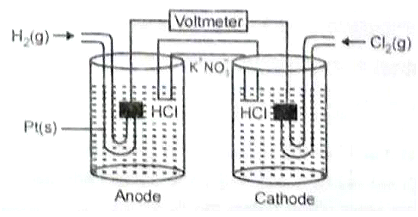
By what value the cell voltage change when concentration of ions in anodic and cathodic compartments both increased by a factor of 10 at 298
1. +0.0591
2. -0.0591
3. -0.1182
4. 0
For which of these electrodes will the reduction potential vary with pH?
l.
ll.
lll.
1. l only
2. ll only
3. l and ll only
4. l, ll and lll only
What is the standard Gibbs free energy change (ΔG° in kJ/mol) for a Daniell cell with a standard cell potential (E°cell) of 1.1 V, when 2 moles of Zn(s) are oxidized at 298 K?
| 1. | –212.3 | 2. | –106.2 |
| 3. | –424.6 | 4. | –53.1 |
Zinc gives H2 with H2SO4 and HCl but not with HNO3
because
1. Zinc acts as oxidising agent when reacts with
HNO3
2. HNO3 is a weaker acid then H2
SO4 and HCl
3. In electrochemical series zinc is above
hydrogen
4. is reduced is preference to hydronium ion
Given that
1. I¯ ions will be able to reduce bromine
2. Br¯ ion will be able to reduce iodine
3. Iodine will be able to reduce bromide ions
4. Bromine will be able to reduce iodide ions
The ionization constant of a weak acid is and the molar conductivity at infinite dilution is S. If the cell constant is 0.01 then conductance of 0.01 M acid solution is:
(1)
(2) 1.52 S
(3) 1.52 S
(4)
Given the cell: \(\mathrm{Cd}(s)\left|\mathrm{Cd}(\mathrm{OH})_2(s)\right| \mathrm{NaOH}(a q, 0.01 M) \mid H_2(g), 1 \text { bar } \mid P t(s)\)
with Ecell = 0.0 V. If E°Cd2+lCd = -0.39 V, then Ksp of Cd(OH)2 is:
1. 0.1
2.
3.
4. None of these
A fuel cell develops an electrical potential from the coombustion of butane at 1 bar and 298 K
What is of a cell?
(1) 4.74 V
(2) 0.547 V
(3) 4.37 V
(4) 1.09 V
108 g fairly concentrate solution of is electrolyzed using 0.1 F of electricity. The weight of resulting solution is:
(1) 94 g
(2) 11.6 g
(3) 96.4 g
(4) None of these
Pt | Cl2 (P1) | HCl (0.1 M) | Pt | Cl2 (P2) ; cell reaction will be spontaneous if :
1. P1 = P2 2. P1 > P2
3. P2 > P1 4. P1 = P2 = 1 atm
Molar conductance of 0.1 M acetic acid is 7 ohm–1 cm2 mol–1 .
If the molar conductance of acetic acid at infinite dilution is 380.8 ohm–1 cm2 mol–1,
the value of dissociation constant will be -
1. 226 × 10–5 mol dm–3
2. 1.66 × 10–3 mol dm–1
3. 1.66 × 10–2 mol dm–3
4. 3.442 × 10–5 mol dm–3
An increase in equivalent conductance of a strong electrolyte with dilution is mainly due to:
| 1. | increase in ionic mobility of ions |
| 2. | 100% ionization of electrolyte at normal dilution |
| 3. | increase in both, i.e, number of ions and ionic mobility of ions |
| 4. | increase in the number of ions |
| 1. | 0.34 V | 2. | -0.34 V |
| 3. | 0.22 V | 4. | -0.22 V |
The volume of gases liberated at STP when a charge of 2F is passed
through aqueous solution of sodium phosphate, is :
1. 11.2L
2. 44.8L
3. 33.6L
4. 22.4L
Value of \(\land_{m}^{0}\) for \(\) \(SrCl_{2}\) (strong electrolyte) in water at 25°C from the data below is:
| Conc. (mol/litre) | 0.25 | 1 |
| \(\land_{m} \Omega^{- 1 } c m^{2 } m o l^{- 1}\) | 260 | 250 |
1. 270 Ω-1 cm2 mol-1
2. 260 Ω-1 cm2 mol-1
3. 250 Ω-1 cm2 mol-1
4. 255 Ω-1 cm2 mol-1
The calomel electrode is reversible with respect to-
1. Mercury
2.
3.
4.
If is 1.69 V and is 1.40 V, then will be :
1. 0.19 V
2. 2.945 V
3. 1.255 V
4. None of the above
Standard electrode potential data are useful for understanding the suitability of an oxidant in aredox titration. Some half cell reactions and their standard potential are given below
Identify the only incorrect statement regarding the quantitative estimation of aqueous
(1) can be used in aqueous
(2) can be used in aqueous
(3) can be used in aqueous
(4) can be used in aqueous
Zinc gives H2 with H2SO4 and HCI but not with HNO3 because
(1) Zinc acts as oxidizing agent when react with HNO3(2) HNO 3 is a weaker acid then H2SO4 and HCI
(3) In electrochemical series, the zine is above hydrogen
(4) NO is reduced is a preference to hydronium ion
During electrolysis of conc. H2SO4, perdisulphuric acid (H2S2O8), and O2
form in equimolar amount. The amount of H2 that will form simultaneously will be :
1. Thrice that of O2 in moles.
2. Twice that of O2 in moles.
3. Equal to that of O2 in moles.
4. Half of that of O2 in moles.
Al2O3 is reduced by electrolysis at low potentials and high currents. If 4.5x104 A of
current is passed through molten Al2O3 for 6 h,what mass of aluminium is produced ?
(Assume 100% current efficiency, at. mass of Al = 27 g mol-1)
1. 9.0 x 103 g
2. 9.1 x 104 g
3. 2.4 x 105 g
4. 1.3 x 104g
The quantity of electricity required to reduce 12.3 g of nitrobenzene to aniline with 50 % current efficiency is:
1. 1 F
2. 0.6 F
3. 0.5 F
4. 1.2 F
Three Faradays of electrical charge are passed consecutively through four separate electrolytic cells containing aqueous solutions of AgNO3, CuSO4, Al(NO3)3, and NaCl. What will be the molar ratio of the respective metals deposited at each cathode?
| 1. | 1 : 2 : 3 : 1 | 2. | 6 : 3 : 2 : 6 |
| 3. | 6 : 3 : 0 : 0 | 4. | 3 : 2 : 1 : 0 |
If an iron rod is dipped in CuSO4 solution, then:
1. Blue colour of the solution turns red.
2. Brown layer is deposited on iron rod.
3. No change occurs in the colour of the solution.
4. None of the above.