| (a) | Time required to consume 3 moles of X is \(2 \times 69.3 \) minutes |
| (b) | Value of \(k = { ln2 \over 2 \times 0.693 } min^{-1}\) |
| (c) | \(-\frac{d x}{d t}=2 \times 10^{-2} \frac{\mathrm{~m}}{\mathrm{~min}}~ at ~t=69.3 \mathrm{~min}\) |
| (d) | \(\frac{d y}{d t}=5 \times 10^{-3} \frac{\mathrm{~m}}{\mathrm{min}}~ at~t=138.6 \mathrm{~min}\) |
| Graph I: |  |
| Graph II: |  |
| 1. | Both graph I and graph II are not correct. |
| 2. | Graph I is not correct but graph II is correct. |
| 3. | Both graph I and graph II are correct. |
| 4. | Graph I is correct, but graph II is not correct. |
| (i) | A catalyst lowers the activation energy of a reaction. |
| (ii) | A catalyst allows the same rate of reaction to be achieved at a lower temperature. |
| (iii) | A catalyst mixes with the reactants and increases the overall concentration of reactants in the rate equation. |
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. | 1 | 2. | 5 |
| 3. | 2 | 4. | 8 |
| 1. | \(2.726 \times 10^{-5} \mathrm{~min}^{-1}\) | 2. | \(2.276 \times 10^{-5} \mathrm{~min}^{-1}\) |
| 3. | \(2.216 \times 10^{-5} \mathrm{~min}^{-1}\) | 4. | None of the above |
| 1. | \(\mathrm{s}^{-1}\) | 2. | \(\mathrm{L} \mathrm{mol}^{-1} \mathrm{~s}^{-1}\) |
| 3. | \(\mathrm{mol} \mathrm{L}^{-1} \mathrm{~s}^{-1}\) | 4. | \(\mathrm{L}^2 \mathrm{~mol}^{-2} \mathrm{~s}^{-1}\) |
| (a) | \(\ln\dfrac{A}{k}=\dfrac{E_a}{R T}\) |
| (b) | \(\dfrac{d \ln k}{d T}=\dfrac{E_a}{R T^2}\) |
| (c) | \(\log A=\log k+\dfrac{E_a}{2.303 R T}\) |
| (d) | \(\log \left(-\dfrac{E_a}{R T}\right)=\dfrac{k}{A}\) |
| 1. | A second-order reaction is always a multistep reaction. |
| 2. | A zero-order reaction is a multistep reaction. |
| 3. | A first-order reaction is always a single-step reaction. |
| 4. | A zero-order reaction is a single-step reaction. |
| 1. | The rate law includes all reactants in the balanced overall equation. |
| 2. | The overall order equals the sum of the reactant coefficients in the overall reaction. |
| 3. | The overall order equals the sum of the reactant coefficients in the slow step of the reaction. |
| 4. | The structure of the catalyst remains unchanged throughout the reaction progress. |
| Assertion (A): | A reaction can have zero activation energy. |
| Reason (R): | The minimum amount of energy required by reactant molecules so that their energy becomes equal to threshold value, is called activation energy. |
| 1. | (A) is False but (R) is True. |
| 2. | Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 3. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 4. | (A) is True but (R) is False. |
| 1. | 5.54 | 2. | 16.61 |
| 3. | 33.32 | 4. | 332.2 |

| (i) | The thermal decomposition of HI on a gold surface follows a zero-order reaction. |
| (ii) | Instantaneous rate = \(\operatorname{limit}_{t \rightarrow 0} \frac{\Delta C}{\Delta t}\) |
| (iii) | The rate of 1st order reaction is proportional to the first power of the concentration of the reactant. |
| (iv) | Radioactive reaction follows 1st order kinetics. |
| Assertion(A): | The active complex is an intermediate product. |
| Reason(R): | The active complex is unstable because of high energy. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
 (where K1 is the rate constant for the forward reaction and K2 is the rate constant for the backward reaction)
(where K1 is the rate constant for the forward reaction and K2 is the rate constant for the backward reaction)| Assertion (A): | Inversion of cane sugar is a first-order reaction. |
| Reason (R): | Inversion of cane sugar is a pseudo-unimolecular reaction. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True, but (R) is False. |
| 4. | Both (A) and (R) are False. |
Consider the reaction, 2A + B → Products.
When concentration of B alone was doubled, the half-life did not change. When the concentration of A alone was doubled, the rate increased by two times. The unit of rate constant for this reaction is:
1. L mol–1 s–1
2. no unit
3. mol L–1s–1
4. s–1
The rate of a reaction is decreased by 3.555 times when the temperature was changed from 40°C to 30°C. The activation energy (in kJ ) of the reaction is:
(Take R=8.314 J and In 3.555=1.268)
1. 100 kJ/mol
2. 120 kJ/mol
3. 95 kJ/mol
4. 108 kJ/mol
92U235, nucleus absorb a neutron and disintegrate in 54Xe139, 38Sr94 and x . The product x is:
1. 3 - neutrons
2. 2 - neutrons
3. α - particle
4. β - particle
If the concentration of a solution is changed from 0.2 to 0.4, then what will be rate and rate constant. The reaction is of first order and rate constant is :
1.
2.
3.
4.
The graphs that represent a zero-order reaction are:
| (a) |  |
(b) |  |
| (c) |  |
(d) |  |
| 1. | a and b | 2. | b and c |
| 3. | c and d | 4. | a and d |
The correct statements among following about Maxwell, Boltzmann distribution of energy is -
| (a) | The fraction of molecules with the most probable kinetic energy decreases at higher temperatures |
| (b) | The fraction of molecules with the most probable kinetic energy increases at higher temperatures |
| (c) | Most probable kinetic energy increases at higher temperatures |
| (d) | Most probable kinetic energy decreases at higher temperatures |
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, c)
The value of the rate constant of a pseudo-first-order reaction:
| 1. | Depends on the concentration of reactants present in a small amount. |
| 2. | Depends on the concentration of reactants present in excess. |
| 3. | Is independent of the concentration of reactants. |
| 4. | Depends only on temperature. |
An incorrect statement about the collision theory of chemical reaction is:
| 1. | It considers reacting molecules or atoms to be hard spheres and ignores their structural features. |
| 2. | The number of effective collisions determines the rate of reaction. |
| 3. | The collision of atoms or molecules possessing sufficient threshold energy results in product formation. |
| 4. | Molecules should collide in the proper orientation for the collision to be effective with sufficient threshold energy and proper orientation. |
Consider the graph given in the figure. Which of the following options does not show an instantaneous rate of reaction in the 40s?

1. \({V_5 -V_2 \over 50-30}\)
2. \({V_4 -V_2 \over 50-30}\)
3. \({V_3 -V_2 \over 40-30}\)
4. \({V_3 -V_1 \over 40-20}\)
A graph of volume of hydrogen released vs time for the reaction between zinc and dil. HCl is given in the graph below.

The correct statement among the following based on the graph given above is:
For a reaction A → Product, with k = 2.0 × 10–2 s–1, if the initial concentration of A is 1.0 mol L–1, the concentration of A after 100 seconds would be :
| 1. | 0.23 mol L–1 | 2. | 0.18 mol L–1 |
| 3. | 0.11 mol L–1 | 4. | 0.13 mol L–1 |
The rate constant for the decomposition of hydrocarbons is 2.418 × 10–5 s–1 at 546 K. If the energy of activation is 179.9 kJ/mol, the value of the pre-exponential factor will be:
1.
2.
3.
4.
The half-life for radioactive decay of 14C is 5730 years. A wood sample contains only 80% of the 14C. The age of the wood sample would be-
1. 1898 years
2. 1765 years
3. 1931 years
4. 1860 years
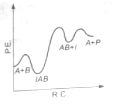
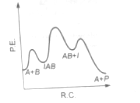
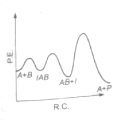
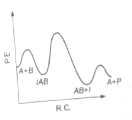
The following mechanism has been proposed for the exothermic catalyzed complex reaction
If is much smaller than , the most suitable qualitative plot of potential energy (P.E.) versus reaction co-ordinate (R.C.) for the above reaction
(1) 
(2) 
(3) 
(4) 
Consider the reaction.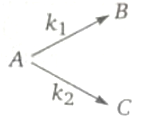
The rate constant for two parallel reactions were found to be and . If the corresponding energy of activation of the parallel reaction are 100 and 120 kJ/mol respectively, The net energy of activation of A is:
(1) 150 kJ/mol
(2) 320 kJ/mol
(3) 116 kJ/mol
(4) 220 kJ/mol
Select the correct option based on the statements below:
| Statement 1: | The overall order of a reaction is the sum of the powers of all the reactants in the rate expression. |
| Statement 2: |
Examples of higher-order reactions (order > 2) are abundant. |
| 1. |
Both Statement 1 and Statement 2 are true. |
| 2. |
Both Statement 1 and Statement 2 are false |
| 3. |
Statement 1 is true, but Statement 2 is false. |
| 4. |
Statement 1 is false, but Statement 2 is true. |
For study in chemical kinetics, the following relationship is observed
In K (sec-1) = 12.4 -
the activation energy(in cal) of the reaction will be:
1. 2.303 x 8.314 x 800
2. 2.303 x 800
3. 8.314 x 800
4. 2 x 800
The rate constant of the given reaction-
R(g) \(\xrightarrow[]{\Delta }\) 2P(g) is 2.48 × 10–4 s–1.
A 1 : 1 molar ratio of R to P in the reaction mixture is attained after:
1. 32 min
2. 27.3 min
3. 20 min
4. 0 min
The hydrolysis of ester is alkaline medium is a
1. 1st order reaction with molecularity 1
2. 1st order reaction with molecularity 2
3. 2nd order reaction with molecularity 1
4. 2nd order reaction with molecularity 2
1. 0.0125 M
2. 0.025 M
3. 0.05 M
4. None of these
1. 1 : 0.301
2. 0.125 : 0.602
3. 1 : 602
4. None of these
|
Ex. No. |
[A/M] |
[B/M] |
Initial rate |
|
1. |
0.01 |
0.01 |
|
|
2. |
0.02 |
0.01 |
|
|
3. |
0.02 |
0.01 |
1. M/sec
2. M/sec
3. M/sec
4. M/sec
Plots showing the variation of the rate constant (k) with temperature (T) are given below. The plot that follows Arrhenius equation is –
1.
2.
3.
4.
For a first order reaction A P, the temperature (T) dependent rate constant(k) was found to follow the equation logk = – (2000) + 6.0. The pre-exponential factor A and the activation energy Ea,respectively, are-
1. 1.0 × 106 s–1 and 9.2 kJ mol–1
2. 6.0 s–1 and 16.6 kJ mol–1
3. 1.0 × 106 s–1 and 16.6 kJ mol–1
4. 1.0 × 106 s–1 and 38.3 kJ mol–1
The decomposition of a gaseous substance (A) to yield gaseous products (B) and (C)
follows first order kinetics. If initially only (A) is present and 10 minutes after the
start of the reaction the pressure of (A) is 200 mm Hg and that of over all
mixture is 300 mm Hg, then the rate constant for 2A B + 3 C is –
1. (1/600) ln 1.25 sec–1
2. (2.303/10) log 1.5 min–1
3. (1/10) ln 1.25 sec–1
4. None of the above
The rate of the simple reaction
2NO+O2 2NO2, when the volume of the reaction vessel is doubled –
1. Will grow eight times of its initial rate
2. Reduce to one-eighth of its initial rate
3. Will grow four times of its initial rate
4. Reduce to one-fourth of its initial rate
For the reaction, , the rate law for the disappearance of will be
1.
2.
3.
4.
The following data were obtained during the first-order thermal decomposition of at a constant volume.
SO2Cl2(g) → SO2(g) + Cl2(g)
| Experiment | Time/s | Total pressure/atm |
| 1 | 0 | 0.5 |
| 2 | 100 | 0.6 |
The rate of the reaction when total pressure is 0.65 atm will be:
1.
2.
3.
4.
1 mole of a gas changes linearly from its initial state (2 atm, 10 lt) to its final state (8 atm, 4 lt). The maximum rate constant is equal to 20 and the value of activation energy is 40 kJ, assuming that the activation energy does not change in this temperature range. The value of the rate constant, at the maximum temperature that the gas can attain, is:
1.
2.
3.
4.
What is the percentage of the reactant molecules crossing over the energy barrier at 325 K?
Given that
| 1. | 80.62 % | 2. | 85.23 % |
| 3. | 89.27 % | 4. | None of the above |
The gas phase decomposition 2N2O5 → 4NO2 + O2 follows the first order rate law, K = 7.5 × 10-3 sec-1. The initial pressure of N2O5is 0.1 atm. The time of decomposition of N2O5 so that the total pressure becomes 0.15 atm will be -
| 1. | 54 sec | 2. | 5.4 sec |
| 3. | 3.45 sec | 4. | 34.55 sec |
The relationship between temperature and the variance in reaction rate is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
In a reaction, the rate = k[A]1[B]-2/3 the order of the reaction is-
1. 1/3
2. 2
3. -1/3
4. Zero
A graph between log and log a (abscissa), a being the initial
concentration of A in the reaction. For reaction AProduct, the rate law is :
| 1. | \(\frac{-d A}{d t}=\mathrm{K}\) | 2. | \(\frac{-d A}{d t}=K A\) |
| 3. | \(\frac{-d A}{d t}=K A^2\) | 4. | \(\frac{-d A}{d t}=K A^3\) |
Consider a reaction \(A(g) \xrightarrow { k=0.1 M m^{-1}}2B(g)\). If initial concentration of A is 0.5 M then select graph.
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Listed in the table are forward and reverse rate constants for the reaction
2N0(g) (g) + (g)
|
Temperature |
||||
| 1400 | 0.29 | 1.1 | ||
| 1500 | 1.3 | 1.4 |
Select the correct statement :
1. Reaction is exothermic and value of equilibrium constant () at 1400 K is 3.79x
2. Reaction is endothermic and value of at 1400 K is 2.63x
3. Reaction is exothermic and value of at 1400 K is 2.63 X
4. Reaction is endothermic and value of at 1500 K is 9.28 x
A first-order reaction is 15 % completed in 20 minutes. The amount of time required to complete 60 % of the reaction is:
| 1. | 112.8 min | 2. | 120.7 min |
| 3. | 100.4 min | 4. | 140.7 min |
The plot of log k vs helps to calculate :
1. Energy of activation.
2. Rate constant of reaction.
3. Order of the reaction.
4. Energy of activation as well as the frequency.
In the following reaction: XA YB where the -ve sign indicates the rate of disappearance of the reactant. Then X: Y is
1. 1:2
2. 2:1
3. 3:1
4. 3:10
A gaseous reaction A2(g) → B(g) + (g) shows increase in pressure from 100 mm to 120 mm in 5 minutes. The rate of disappearance of A2 will be :
| 1. | 4 mm min-1 | 2. | 8 mm min-1 |
| 3. | 16 mm min-1 | 4. | 2 mm min-1 |

Half-life is independent of the concentration of a reactant. After 10 minutes, the volume of N2 gas is 10 L and after complete reaction, it is 50 L. Hence, the rate constant is:
1. log 5 min-1
2. log 1.25 min-1
3. log 2 min-1
4. log 4 min-1
For a first-order reaction A Products, the rate of reaction at [A] = 0.2 M is 1.0 x 10-2 mol litre-1 min-1. The half-life period for the reaction will be:
| 1. | 832 sec | 2. | 440 sec |
| 3. | 416 sec | 4. | 14 sec |
Rate constant of reaction can be expressed by Arrhenius equation as,
In this equation, represents:
1. the energy above which all the colliding molecules will react
2. the energy below which colliding molecules will not react
3. the total energy of the reacting molecules at a temperature, T
4. the fraction of molecules with energy greater than the activation energy of the reaction
For a given reaction, the presence of a catalyst reduces the energy of activation by 2 kcal at 27 oC. The rate of reaction will be increased by:
1. 20 times
2. 14 times
3. 28 times
4. 2 times
How much faster would a reaction proceed at 25°C than at 0°C if the activation energy is 65 kJ?
1. 2 times
2. 16 times
3. 11 times
4. 6 times
In the Arrhenius equation K = Ae-Ea/RT, the quantity e-Ea/kT is referred as:
1. Boltzmann factor.
2. Frequency factor.
3. Activation factor.
4. None of the above.
For an exothermic chemical process occurring in two steps as;
(i) A+BX(Slow)
(ii) XAB (Fast)
The progress of the reaction can be best described by:
| 1. |  |
2. |  |
| 3. |  |
4. | All of the above. |