| 1. | Relative lowering of vapor pressure is independent of temperature. |
| 2. | Osmotic pressure always depends on the nature of the solute. |
| 3. | Elevation of boiling point is independent of the nature of the solvent. |
| 4. | Lowering of freezing point is proportional to the molar concentration of solute. |
A 0.03 M aqueous solution of a weak acid HX has an acid dissociation constant \(K_a = 1.2 \times 10^{-5}\), for the dissociation\(\mathrm{HX}_{(\mathrm{aq})} \rightleftharpoons \mathrm{H}_{(\mathrm{aq})}^{+}+\mathrm{X}_{(\mathrm{aq})}^{-} \). Calculate the osmotic pressure (in atm) of the solution at 300 K.
| 1. | 4 atm | 2. | 2 atm |
| 3. | 1 atm | 4. | 6 atm |
| 1. | The freezing point is raised. |
| 2. | The freezing point is lowered. |
| 3. | The freezing point does not change. |
| 4. | The boiling point does not change. |
| 1. | Leaves the vapour increases |
| 2. | Leaves the solution increases |
| 3. | Leaves the solution decreases |
| 4. | Leaves the vapour decreases |
| Statement I: | The proportionality constant, Kf, depends on the nature of the solvent as well as the solute. |
| Statement II: | When blood cells are placed in water containing less than 0.9 % (mass/volume) salt, blood cells shrink due to osmosis. |
| 1. | Statement I is correct; Statement II is incorrect |
| 2. | Statement I is incorrect; Statement II is incorrect |
| 3. | Statement I is incorrect; Statement II is correct |
| 4. | Statement I is correct; Statement II is correct |
| 1. | The gas in contact with the liquid should behave as an ideal gas. |
| 2. | There should not be any chemical interaction between the gas and the liquid. |
| 3. | The pressure applied should be high. |
| 4. | All of the above. |
| 1. | 2.463 atm | 2. | 1.231 atm |
| 3. | 4.926 atm | 4. | 369 atm |
| 1. | Nitric acid (68%) and water (32%) mixture is an example of the minimum boiling azeotrope. |
| 2. | Osmosis can be reversed if pressure lower than the osmotic pressure is applied to the solution. |
| 3. | As compared to other colligative properties, the magnitude of osmotic pressure is large even for very dilute solutions. |
| 4. | The condition of anoxia in mountain climbers is an application of Raoult's law. |
| 1. | Sodium chloride in liquid cyclohexane |
| 2. | Bromine in liquid hexane |
| 3. | Hydrogen in liquid water |
| 4. | Potassium bromide in liquid tetrachloromethane |
| Assertion (A): | The preservation of meat by salting and fruits by adding sugar is done to protect them from bacterial action. |
| Reason (R): | Due to reverse osmosis. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
| Assertion(A): | For 0.1 M aq. NaOH and for 0.1 M aq. CH3COOH solution, the ebulioscopic constant is different. |
| Reason(R): | The ebulioscopic constant depends on the nature of the solute. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True and (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
| Assertion (A): | 0.1 M solution of glucose has the same decrement in the freezing point as 0.1 M solution of urea. |
| Reason (R): | Kf for both has the same value. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
| Assertion (A): | The solubility of all solutes in water generally increases with higher temperatures. |
| Reason (R): | The solubility of the solutes is dependent on temperature. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
| 1. | 103.57 oC | 2. | 101.428 oC |
| 3. | 374.428 K | 4. | 372.196 K |
| 1. | \(P-P_s \over P \) = i (\(n \over n+N\)) | 2. | \(P-P_s \over P_s\) = i (\(n \over n+N\)) |
| 3. | \(P-P_s \over P \) = \(i n \over in+N\) | 4. | \(P-P_s \over P_s\) = \(in \over in+N\) |
The vapour pressure of pure liquids A and B are 450 and 700 mm Hg respectively, at 350 K. If the total vapour pressure is 600 mm Hg then the composition of the vapour phase are:
1. XA=0.30; XB=0.70
2. XA=0.40; XB=0.60
3. XA=0.20; XB=0.90
4. XA=0.50; XB=0.50
| 1. | 1.24 g | 2. | 1.45 g |
| 3. | 1.65 g | 4. | 1.85 g |
Solution A contains 7 g/L of
1. Solution A is greater than B
2. Both have the same osmotic pressure
3. Solution B is greater than A
4. Cannot be determined
The boiling point of 1M Urea solution is 101o C. The boiling point of 1M NaCl solution will be (Assume that NaCl is 100% dissociated):
1. 101oC
2. 275 K
3. 375 K
4. 202oC
The boiling point of an aqueous solution is . The freezing point of this solution will be:
(Given: Kb = 0.52 k/m, Kf = 1.86 k/m)
1.
2.
3.
4.
1 mole NaCl is mixed with 6 mole of H2O. As a result, relative lowering in vapour pressure is observed to be 0.2. In these conditions, percentage ionisation of NaCl will be
1. 25
2. 50
3. 75
4. 100
0.6 mL of acetic acid (CH3COOH), having density 1.06 g mL–1, is dissolved in 1 litre of water. The depression in freezing point observed for this strength of acid was 0.0205°C. The value of van’t Hoff factor and the dissociation constant of acid are respectively:
1. 1.041; 1.86 × 10–5
2. 2.073; 1.86 × 10–5
3. 0.091; 1.76 × 10–5
4. 1.041; 1.76 × 10–5
The solubility of \(H_2S\) in water at STP is 0.195 m. The value of Henry's constant is:
| 1. | 274 atm | 2. | 285 atm |
| 3. | 295 atm | 4. | 278 atm |
Match the following type of intermolecular interactions present in the pair of compounds given:
| (a) | Methanol and Acetone | (i) | Van der Waal’s forces of attraction |
| (b) | Acetonitrile and Acetone | (ii) | Ion-dipole interaction |
| (iii) | Dipole-dipole interaction |
| (a) | (b) | |
| 1. | (iii) | (ii) |
| 2. | (ii) | (ii) |
| 3. | (iii) | (iii) |
| 4. | (iii) | (i) |
When 19.5 g of fluoroacetic acid (CH2FCOOH) is dissolved in 500 g of water, the observed depression in the freezing point was found to be 1.00 K. The Van’t Hoff factor and the dissociation constant of the fluoroacetic acid, respectively are: (Given: \(K_f\) for water = 1.86 K kg mol⁻¹, Molar mass of CH2FCOOH = 78 g mol⁻¹)
| 1. | 20.75 , 4.77 × 10–3 | 2. | 1.075 , 4.77 × 10–2 |
| 3. | 2.073 , 3.07 × 10–4 | 4. | 1.075 , 3.07 × 10–3 |
A solution containing 30 g of non-volatile solute in 90 g of water has a vapour pressure of 2.8 kPa at 298 K. Further, 18 g of water is then added to the solution and the new vapour pressure becomes 2.9 kPa at 298 K.
The molar mass of the solute will be :
1. 23 g mol−1
2. 34 g mol−1
3. 15 g mol−1
4. 46 g mol−1
Camphor is often used in molecular mass determination because:
1. It has a very high cryoscopic constant
2. It is volatile
3. It is a solvent for organic substances
4. It is readily available
The vapour pressure of a solvent decreased by 10 mm of Hg when a non-volatile solute was added to the solvent. The mole fraction of the solute in solution is 0.2. What would be the mole fraction of the solvent if the decrease in vapour pressure is 20 mm of Hg:
1. 0.2
2. 0.4
3. 0.6
4 0.8
The addition of water vapour does not change the density of:
1. CCl4
2. CS2
3. Ether
4. Coke
Select the correct option based on statements below:
| Assertion (A): | When methyl alcohol is added to water, boiling point of water increases. |
| Reason (R): | When a volatile solute is added to a volatile solvent, elevation in boiling point is observed. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Which of the following binary mixtures will have the same composition in the liquid and the vapour phase?
| (a) | Benzene-toluene | (b) | Water-nitric acid |
| (c) | Water-ethanol | (d) | n-Hexane-n-heptane |
| 1. | a and b | 2. | b and c |
| 3. | c and d | 4. | a and d |
Three aqueous solutions of NaCl are labelled as ‘A', ‘B’ and ‘C with concentrations 0.1 M, 0.01 M and 0.001 M, respectively. The value of van’t Hoff factor for these solutions will be in the order of :
1. iA < iB < iC
2. iA > iB > iC
3. iA = iB = iC
4. iA < iB > iC
The boiling point of water at 750 mm Hg is 99.63°C. How much sucrose is to be added to 400 g of water such that it boils at 100°C?
( Kb H2O = 0.52 K kg mol−1)
1. 97.33g
2. 121.67g
3. 145.14g
4. 124.79g
Benzene and toluene form an ideal solution over the entire range of composition. The vapour pressure of pure benzene and toluene at 300 K are 50.71 mm Hg and 32.06 mm Hg respectively.
The mole fraction of benzene in vapour phase, if 80 g of benzene is mixed with 100 g of toluene, would be:
1. 0.41
2. 0.68
3. 0.72
4. 0.59
The mass of a non-volatile solute (molar mass 40 g mol–1) that should be dissolved in 114 g octane to reduce its vapour pressure to 80 % would be:
1. 6 g
2. 7 g
3. 8 g
4. 10 g
Which of the following represents a metastable system?
1. an unsaturated solution
2. a dilute solution
3. a saturated solution
4. a supersaturated solution
A solution of acetone in ethanol :
1. Shows a negative deviation from Raoult's law.
2. Shows a positive deviation from Raoult's law.
3. Behaves like a near-ideal solution.
4. Obeys Raoult's law.
During osmosis.flow of water through a semipermeable membrane is
(1) from both sides of semi-permeable membrane with unequal flow rates
(2) from solution having lower concentration only
(3) from solution having higher concentration only
(4) from both sides of semi-permeable membrane with equal flow rates
Which of the following graph is correct for an ideal binary liquid solution containing non-volatile solute?
| 1. |  |
2. |  |
| 3. |  |
4. | All are correct |
The latent heat of vaporization of water is 540 cal g-1 at 100 oC. Kb for water is:
1. \(0 . 562 K m^{- 1}\)
2. \(1 . 86 K m^{- 1}\)
3. \(0 . 515 K m^{- 1}\)
4. \(5 . 12 K m^{- 1}\)
The partial pressure of ethane over a saturated solution containing 3.56 X 10-2 g of ethane is 3 bar. If the solution contains 9.00 X 10-2 g of ethane, then what will be the partial pressure of the gas?
| 1. | 0.76 bar | 2. | 0.16 bar |
| 3. | 7.82 bar | 4. | 3.12 bar |
Sodium chloride (relative molecular mass=58.5) has a solubility of 36.0 g per 100 g of water. The density of the solution is 1.13 g/ml. Which of the following calculations would give the solubility in moles per litre?
1. 36.0 × 10/58.5
2. 36.0 × 1000/(58.5 × 1.13)
3. 36.0 × 10 × 1.13/58.5
4. 36.0 × 1000 × 1.13/(58.5 × 136)
Mole of K2SO4 to be dissolved in 12 moles water
to lower its vapour pressure by 10 mm of Hg at a
temperature at which vapour pressure of pure
water is 50 mm of Hg is
1. 3 mol
2. 0.5 mol
3. 1 mol
4. 2 mol
A dilute sulphuric acid (H₂SO₄) solution has pH = 0.
Assuming complete dissociation, what is the molarity of the solution?
1. 1M
2. M/2
3. M/10
4. Cannot be determined
A mixture of CO & CO2 has a density of 1.5 gL–1 at
20°C and 740 mm Hg. The mole fraction of CO in the
mixture is around
1. 0.65
2. 0.56
3. 0.43
4. 1
A metal oxide contains 40% oxygen by weight. Its
equivalent weight is
1. 12
2. 20
3. 40
4. 60
If the relative decrease in vapour pressure is 0.4 for a solution containing 1 mol NaCl in 3 mol of , then percentage ionisation of NaCl will be -
1. 60%
2. 80%
3. 40%
4. 100%
60 g m of Urea is dissolved in 1100 gm solution. To keep as 1 mol/g, water separated in the form of ice is
1. 40 gm
2. 60 gm
3. 100 gm
4. 200 gm
When mercuric iodide is added to the aqueous solution of potassium iodide is
(1) Freezing point is raised
(2) Freezing point is lowered
(3) Freezing point does not change
(4) Boiling point does not change
Mole of to be dissolved in 12 moles water to lower its vapour pressure by 10 mm of Hg at a temperature at which vapour pressure of pure water is 50 mm of Hg is
(1) 3 mol
(2) 0.5 mol
(3) 1 mol
(4) 2 mol
Equal volumes of 0.1 M and 0.2 M NaCl are mixed. The concentration of ions in the mixture will be:
| 1. | 0.1 M | 2. | 0.05 M |
| 3. | 0.2 M | 4. | 0.15 M |
The Henry's law constant for the solubility of N2 gas in water at 298 K is 1.0 × 105 atm. The mole fraction of N2 in air is 0.8. The number of moles of N2 formed from air dissolved in 10 moles of water at 298 K and 5 atm pressure is-
| 1. | 4.0 × 10–4 mol | 2. | 4.0 × 10–5 mol |
| 3. | 5.0 × 10–4 mol | 4. | 4.0 × 10–6 mol |
The solubility of a specific non-volatile salt is 4 g in 100 g of water at 25C. If 2.0 g , 4.0 g and 6.0 g of the salt added of 100 g of water at 25C, in system X,Y and Z. The vapour pressure would be in the oreder :
1. X < Y < Z
2. X > Y > Z
3. Z > X = Y
4. X > Y = Z
Dimer of acetic acid in liquid benzene is in equilibrium with acetic acid monomer at certain temperature and pressure. If 25% of the dimer molecules are separated out then
1. Freezing point of the solution reduces
2. Average molar mass of solute increases
3. Boiling point of solution increases
4. Molar mass of solute decreases
How much ethyl alcohol must be added to 1.0 L of water so that solution will not freeze at -4F ? (Kf = 1.86C/m)
1. < 20 g
2. < 10.75 g
3. < 494.5 g
4. > 494.5 g
Water and chlorobenzene are immiscible liquids. Their mixture boils at 89oC under a reduced pressure of . The vapour pressure of pure water at 89oC is . Weight percent of chlorobenzene in the distillate is :
1. 50
2. 60
3. 78.3
4. 38.46
If two liquids A and B are completely immiscible with each other ( each one will behave independtly of the other ) are present in a closed vessel. The total vapour pressure of the system will be :
1. less than 100 torr
2. greater than 200 torr
3. between 100 to 200 torr
4. 300 torr
An aqueous solution of 2% non volatile solute
exerts a pressure of 1.004bar at the normal
boiling point of the solvent.What is the
molecular mass of the solute?
1. 4.135g / mol
2. 22.1g / mol
3. 90.1g / mol
4. 41.35g / mol
During osmosis, the flow of water through a semi-permeable membrane is:
| 1. | From a solution having higher concentration only. |
| 2. | From both sides of the semi-permeable membrane with equal flow rates. |
| 3. | From both sides of the semi-permeable membrane with unequal flow rates. |
| 4. | From a solution having lower concentration only. |
100 mL of liquid A was mixed with 25 mL of liquid B to give a non-ideal solution of A-B. The volume of this mixture will be:
1. 75 mL
2. Exact 125 mL
3. Between 75 mL and 125 mL
4. Close to 125 mL but will not exceed 125 mL
| 1. | \(\dfrac{dlnp}{dT}=\frac{-\Delta H_v}{RT}\) | 2. | \(\dfrac{dlnp}{dT^2}=\frac{-\Delta H_v}{T^2}\) |
| 3. | \(\dfrac{dlnp}{dT}=\frac{\Delta H_v}{RT^2}\) | 4. | \(\dfrac{dlnG}{dT^2}=\frac{-\Delta H_v}{RT^2}\) |
The density of dry air containing only and is 1.15 g/L at 740 mm and 300 K. What is % composition of by weight in the air:
1. 78%
2. 75.5%
3. 70.08%
4. 72.25%
0.1 M KI and 0.2 M are mixe in a 3:1 volume ratio. The depression of the freezing point of the resulting solution will be [ = 1.86 K kg ]
1. 3.72 K
2. 1.86 K
3. 0.93 K
4. 0.279 K.
A saturated solution of has a vapor pressure of 17.20 mm Hg at 20°C, while pure water vapor pressure is 17.25 mm Hg. Solubility product () of at 20°C
1. 9.8x
2.
3. 2.56x
4. 7x
At 300 K, 40 mL of (g) dissolved in 100 g of water at 1.0 atm. What mass of ozone dissolved in 400 g of water at a pressure of 4.0 atm at 300 K?
1. 0.1 g
2. 1.2 g
3. 0.48 g
4. 4.8 g
A sample of liquid of mass 18.0 g is injected into an evacuated 7.6 L flask maintained at 27.0C. If vapour pressure of at 27C is 24.63 mm Hg. What weight percentage of the water will be vapourised when the system comes to equilibrium? Assume water vapours behave as an ideal gas. The volume occupied by the liquid water is negligible compared to the volume of the container :
1. 1%
2. 10%
3. 18%
4. 20%
Which has highest freezing point?
1. 0.01 M NaCl
2. 0.05 M Urea
3. 0.01 M MgCl2
4. 0.02 M NaCl
The boiling point of an aqueous solution of a non-volatile solute is 100.15C. What is the freezing point of an aqueous solution obtained by diluting the above solution with an equal volume of water? The value of Kb and Kf for water are 0.512C and 1.86CK molality
:
(1)
(2)
(3)
(4)
At Abu mountains, water boils at 96. What amount of NaCl be added in 1 kg water so that it boils at 100. .
1. 225 g
2. 450 g
3. 200 g
4. 125 g
Two solutions () containing and () containing are separated by semipermeable membrane as shown below. If on reaction with , produces blue colour of , the blue colour will be noticed in: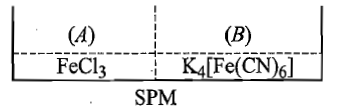
(1) (A)
(2) (B)
(3) in both (A) and (B)
(4) neither in (A) nor in (B)
Elevation in boiling point for 1 molal is three times of one molal aqueous solution of urea. Calculate the value of x, assuming 100% ionization of complex salt. solvent.
1. 5
2. 4
3. 1
4. 6
During osmosis, the flow of water through a semi-permeable membrane is :
1. From solution having higher concentration only
2. From both sides of the semi-permeable membrane with equal flow rates
3. From both sides of the semi-permeable membrane with unequal flow rates
4. From solution having lower concentration only
When mercuric iodide is added to the aqueous solution of potassium iodide is
1. Freezing point is raised
2. Freezing point is lowered
3. Freezing point does not change
4. The boiling point does not change
Which has the highest freezing point?
1. 0.01 M NaCl
2. 0.05M Urea
3. 0.01 M MgCl2
4. 0.02 M NaCl
What is the required amount of K₂SO₄ in moles to dissolve in 12 moles of water to decrease
the vapour pressure by 10 mmHg, if the vapour pressure of pure water is 50 mmHg?
1. 3 mol
2. 0.5 mol
3. 1 mol
4. 2 mol
If partial pressure of oxygen is 0.5 atm and KH = 1.4 × 10-3 M/atm, then the amount of oxygen dissolved in 100 ml water at 298K is-
| 1. | 22.4 mg | 2. | 22.4 g |
| 3. | 2.24 g | 4. | 2.24 mg |
Which solution has highest freezing point?
1. 1% glucose solution
2. 1% urea solution
3. 1% sucrose solution
4. All have same freezing
The
salt that possesses the highest freezing point
1. 0.01 M NaCl
2. 0.05 M Urea
3. 0.01 M MgCl2
4. 0.02 M NaCl
The mole(s) of K2SO4 to be dissolved in 12 moles of water to lower its vapor pressure by 10 mm of Hg at a temperature at which vapor pressure of pure water is 50 mm of Hg is :
| 1. | 3 mol | 2. | 0.5 mol |
| 3. | 1 mol | 4. | 2 mol |
A solution is prepared by dissolving 18 g of glucose in 1000 g of water. When this solution is cooled to –0.2 °C,
some ice separates. Calculate the amount of ice that separates from the solution.
[Freezing point depression constant of water, Kf(H2O) = 1.86 K molal-1]
| 1. | 70 g | 2. | 140 g |
| 3. | 90 g | 4. | 210 g |
Given that the pH of a 0.1 M monobasic acid is 2, what is the osmotic pressure at a given temperature T?
1. 0.1RT
2. 0.11RT
3. 1.1RT
4. 0.01RT
The latent heat of vapourisation of water is 540 cal g-1 at 100. Kb for water is
1. 0.56 K.mole-1
2. 1.86 K.mole-1
3. 0.51 K.mole-1
4. 5.12 K.mole-1