Intrinsic factor is secreted by which cells of the gastric glands?
1. mucous cells
2. chief cells
3. peptic cells
4. parietal cells
What hormone does the small intestine release in response to acidic chyme?
1. cholecystokinin
2. secretin
3. gastric inhibitory peptide
4. gastrin
What is the primary function of the large intestine?
1. to complete the absorption of most nutrients
2. to house gas-producing bacteria
3. to rid the body of toxins
4. to compact, store, and eliminate feces
What blocks pancreatic secretions from entering the duodenum?
1. pyloric valve
2. sphincter of Oddi
3. the common bile duct
4. cholecystokinin secretion
Which of the following triggers the formation of the others?
1. trypsin
2. enterokinase
3. carboxypeptidase
4. chymotrypsin
Which of the following acts to inhibit acid production?
1. secretin
2. somatostatin
3. cholecystokinin
4. gastrin
A person at rest has a respiratory rate of 12 breath per minute, a tidal volume of 500 mL and a dead space air volume of 150 mL. How much air is wasted (not involved in gas exchange) per minute?
1. 500 mL/min
2. 1800 mL/min
3. 4200 mL/min
4. 6000 mL/min
At lungs carbon dioxide diffuses out of the pulmonary capillaries into the alveoli. As a result hemoglobin’s ability to bind to oxygen would:
1. be unstable
2. stay the same
3. decrease
4. increase
Aging RBCs are phagocytized by macrophages in the mononuclear phagocyte system [sleen or liver] and their components are recycled, EXCEPT for the
1. iron atom from the heme group, which is excreted in bile
2. globin peptide chains, which are sent to the kidneys for excretion
3. globin peptide chains, which are converted into bilirubin
4. heme group (minus the iron atom), which is converted into a green pigment called biliverdin
The normal pacemaker of the human heart, sinoatrial node, is derived from the more primitive:
1. ventricle
2. bundle of His
3. conus arteriosus
4. sinus venosus
Amongst the following, a molecule that would lead to coagulation of the blood would be:
1. prostacyclin
2. tissue factor or thromboplastin
3. plasmin
4. tissue plasminogen activator
The efficiency of a respiratory system would be increased by all the following except:
1. increase the surface area available for diffusion of gases
2. decrease the distance over which the gases must diffuse
3. increase the concentration differences of gases inside and outside the system
4. dry the system out so the gases do not have to diffuse through water
How many of the statements given below are correct?
I. During inspiration the diaphragm moves upward while the thorax expands and moves outward.
II. The anatomical dead space represents the air that is trapped in the alveoli and is around 150mL.
III. Decreasing blood oxygen and increasing carbon dioxide cause the major increases seen in breathing rate during strenuous exercise.
IV. Oxygen dissociates faster from hemoglobin during exercise.
V. Increased pH shifts oxyHb-dissociation curve to the right.
1. 2
2. 3
3. 4
4. 5
Respiration is under neural and chemical regulation. The receptors for chemical regulation are located in the:
| 1. | walls of the carotid and aortic arteries and in the medulla oblongata. |
| 2. | walls of the carotid and aortic arteries only. |
| 3. | medulla oblongata and in the walls of the superior and inferior vena cavas. |
| 4. | walls of the carotid and aortic arteries and in the walls of the superior and inferior vena cavas. |
Choose the correct order for the steps of hemostasis:
1. blood coagulation, platelet plug formation, blood vessel spasm
2. platelet plug formation, blood coagulation, blood vessel spasm
3. blood vessel spasm, platelet plug formation, blood coagulation
4. blood vessel spasm, blood coagulation, platelet plug formation
During a cardiac cycle:
1. About 70 ml of blood is pumped by the two ventricles
2. The left atrium and the left ventricle contract simultaneously
3. More blood is pumped into the systemic than in pulmonary circulation
4. The atria contract prior to the ventricles
Kwashiorkor is characterized by:
1. Sufficient calorie intake but insufficient protein consumption
2. Insufficient calorie intake but sufficient protein consumption
3. Insufficient calorie intake and insufficient protein consumption
4. Sufficient calorie intake and sufficient protein consumption
Match each item in Column I with one in Column II and choose the correct option from the codes given below
|
|
Column I |
|
Column II |
|
A. |
2,3 DPG |
a. |
Potent stimulus for |
|
B. |
CO2 |
b. |
Primary stimulus for |
|
C. |
H+ |
c. |
Mainly acts on bodies carotid and aortic |
|
D. |
O2 |
d. |
Right shift of O2– Hb dissociation curve |
A B C D
1. a b c d
2. b d c a
3. d a b c
4. d b c c
What is the primary function of the Pneumotaxic Center in the regulation of respiration in human beings?
1. It increases the rate and depth of breathing
2. It decreases the rate of and depth of breathing
3. It controls the “switch off’ point of inspiration
4. It initiates the “Herring-Breur” reflex
Erythroblastosis fetalis can be prevented during a second pregnancy in a Rh negative mother who is likely to carry a Rh positive fetus by:
| 1. | Administering anti – Rh antibodies to the mother just before the delivery of the second child |
| 2. | Administering Rh antigen to the mother just after the delivery of the first child |
| 3. | Administering Rh antigen to the mother just before the delivery of the second child |
| 4. | Administering anti – Rh antibodies to the mother just after the delivery of the first child |
What is shown in the given figure: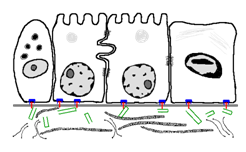
1. Type II pneumocytes lining the respiratory membrane
2. Lining of the small intestine with a brush border
3. Vascular endothelium showing gap junctions
4. Ciliated epithelial lining of the oviduct
Which of the following have no nuclei and originate as pinched-off cytoplasmic fragments of large cells in the bone marrow?
1. Mature RBC
2. Reticulocytes
3. Thrombocytes
4. Macrophages
In the given diagram, the functional residual capacity will be depicted by: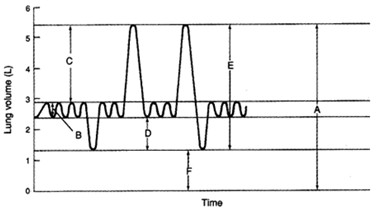
1. B + F
2. B + D + F
3. A – [B + C]
4. A – E
Some human infants, especially those born prematurely, suffer serious respiratory failure because of
1. The sudden change from the uterine environment to the air.
2. The incomplete development of the lung surface.
3. Lung collapse due to inadequate production of surfactant.
4. Mutations in the genes involved in lung formation.
An increase from pH 7.2 to pH 7.4 around hemoglobin causes:
1. Hemoglobin to release all bound oxygen molecules.
2. An increase in the affinity of hemoglobin to bind oxygen molecules.
3. Hemoglobin to denature.
4. An increase in the binding of H+ by hemoglobin.
“Binding of oxygen to hemoglobin makes hemoglobin a stronger acid” forms the physiological basis of:
1. Herring-Breur Reflex
2. Bohr effect
3. Haldane effect
4. Singultus
Which of the following statement is not correct about nitrogen?
1. Plants competes with microbes for limited nitrogen present in soil.
2. Nitrogen is a limiting nutrient for both natural and agricultural ecosystems.
3. During nitrification chemihetrotrophic bacteria play very important role.
4. Bacteria such as Pseudomonas and Thiobacillus can produce nitrogen gas from nitrate.
How many of the following statement is/are correct?
(1) Both Rhizobium and Frankia are free living in soil and can fix N2 only under symbiotic condition.
(2) Root nodules can be best observed before flowering.
(3) Formation of root nodule takes place as a result of division of pericycle cell.
(4) When Rhizobium is free living it is aerobic but when it is symbiont it becomes anaerobic to protect Mo-Fe protein.
Options:
1. 4
2. 3
3. 2
4. 1
How many of the following statement is correct about fate of ammonia in plants?
(1) At physiological pH NH3 is protonated to form NH4 which cannot be stored as it is toxic.
(2) Most plants can assimilate NH4+ as well as
(3) Assimilation of ammonia can take place by reductive amination as well as transamination.
(4) Tranport of fixed nitrogen in form of amides and ureides take place via xylem vessel.
Options :
1. 4 2. 3
3. 2 4. 1
Find odd one out with respect to Melvin Calvin
| 1. | He was professor of chemistry. |
| 2. | Calving along with J.A.Bassham studied firation of carbon in plants. |
| 3. | He proposed that plant can convert light energy into chemical energy by transferring electrons in an organized array of pigment molecules and other substance. |
| 4. | He discouraged use of radioactivity for human welfare. |
Half-leaf experiment, where a part of the leaf is enclosed in a test tube containing KOH is performed to show that:
1. H2O is required for photosynthesis
2. CO2 is required for photosynthesis
3. Light is required for photosynthesis
4. H2O is source of O2 released during photosynthesis
Match the following
|
Scientists |
Work |
||
|
(I) |
Julius van sachs |
(A) |
Only green part of plant could release oxygen. |
|
(II) |
T.W.Engelmann |
(B) |
Inferred that the O2 evolved during photosynthesis comes from H2O |
|
(III) |
Van Niel |
(C) |
First action spectrum in terms of O2 evolution. |
|
(IV) |
Ingenhousz |
(D) |
Provided evidence for production of glucose. |
Options :
| I | II | III | IV | |
| 1. | D | C | B | A |
| 2. | D | A | B | C |
| 3. | D | B | A | C |
| 4. | D | B | C | A |
Study the graph given below and find out the wrong statement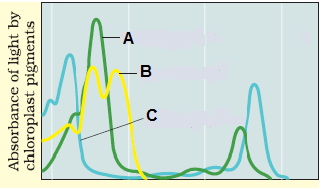
| 1. | A – chlorophyll b, B – Carotenoids, C – Chlorophyll a |
| 2 | A – Yellow-green, B – Yellow to yellow-orange, C – bright or blue-green |
| 3. | A – Major pigment, B – Accessory pigment, C – Protective pigment |
| 4. | A – Accessory pigment, B – Photoprotection, C - Major pigment |
Light reactions or the photochemical phase consists of all except :
| 1. | Absorption of light |
| 2. | Spliting of H2O and release of O2 |
| 3. | Formation of ATP and NADPH |
| 4. | Movement of Proton |
Which of the following statement is incorrect?
1. Diffusion is a slow process and not dependent upon a living system.
2. Diffusion across the membrane depends upon its solubility in lipids.
3. Porins are proteins that forms huge pores, in the inner membrane of the plastids, mitochondria and some bacteria.
4. Water channels are made up of eight different aquaporins.
Following are two statements, find the correct option
(I) In a flowering plant there is complex traffic of compounds moving in different directions.
(II) Each organ of a plant receives some substance and gives out some
Option :
1. Both (I) and (II) are correct
2. Only (I) is correct
3. Only (II) is correct
4. Neither (I) nor (II) is correct
Which of the following is not correct for imbibitions?
1. Imbibition plays a very important role in the emergence of the seedling.
2. In imbibition movement of the solvent is against the gradient.
3. Water potential gradient is essential for imbibition.
4. Affinity between liquid and solid is a pre-requisite for imbibitions.
How many of the following statements are correct?
(1) Vacuolar sap contributes to the solute potential of cells.
(2) The net direction and rate of osmosis depend upon the pressure gradient and concentration gradient.
(3) Osmotic pressure is a function of the solute concentration.
(4) Osmotic pressure is a positive pressure while the osmotic potential is negative.
Options :
1. 4
2. 3
3. 2
4. 1
How many of the following statements are correct?
(1) Less than one percent of water reaching the leaves is used in photosynthesis and plant growth.
(2) Turgidity of guard cells plays a very important role in stomatal opening and closing.
(3) In the dorsiventral leaf upper surface has a greater number of stomata.
(4) In guard cells cellulosic microfibrils are oriented radially.
Option :
1. 4
2. 3
3. 2
4. 1
The C4 plants are twice efficient with respect to C3 plants because
| 1. | They have ability to maximize availability of CO2 while minimizing H2O loss. |
| 2. | They have low CO2 requirement with minimum H2O loss. |
| 3. | There stomata remains always open and high water loss. |
| 4. | There stomata open during night, and minimum H2O loss. |
Match the following :
|
(1) |
Sink of nutrient |
(A) |
Fine vein endings |
|
(2) |
Unloading of mineral |
(B) |
Lateral meristeny |
|
(3) |
Mineral remobilisation |
(C) |
Storage organs |
|
(4) |
Unloading of sugar |
(D) |
Older parts |
Options :
1 2 3 4
1. A B C D
2. B A D C
3. A C B D
4. B C A D
Older dying leaves export much of their mineral content to younger leaves. Similarly, what is incorrect-?
1. Before leaf fall in deciduous plants, minerals are removed to other parts.
2. Elements most readily mobilized are phosphorus, nitrogen, and potassium.
3. Some elements that are structural components like calcium are not remobilized.
4. Deficiency symptoms of nitrogen are observed in younger parts.
Zinc is essential for
1. Biosynthesis of chlorophyll
2. Biosynthesis of auxin
3. Stomatal closing
4. Oxidation of carbohydrates
Blackman demonstrated that increasing illumination increased the photosynthetic rate up to a point when CO2 becomes limiting. If the light was not limiting, temperature becomes limiting. Emerson found that maximum CO2 fixation could be achieved with brief flashes of light. Mark the correct statement in the following
| 1. | Only one factor can be limited in photosynthesis |
| 2. | Photosynthesis consists of a light and dark reaction |
| 3. | The trapping of light by chloroplast is temperature-dependent |
| 4. | The trapping of light by chloroplast can occur only if CO2 is present |
The thylakoids are removed and kept in a culture medium containing CO2 and H2O. If the setup is exposed to light, hexose sugars are not formed as the end product. The most appropriate reason for this will be that
| 1. | Carbon assimilation cannot take place |
| 2. | The pigments (P–700 and P–680) are not linked |
| 3. | Enzymes are not available |
| 4. | The light-trapping device is not functional |
The main difference between active and passive transport across cell membrane is :
| 1. | passive transport is non-selective whereas active transport is selective |
| 2. | passive transport requires a concentration gradient across a biological membrane whereas active transport requires energy to move solutes. |
| 3. | passive transport is confined to anionic carrier proteins whereas active transport is confined to cationic channel. |
| 4. | active transport occurs more rapidly than passive |
Match the items given in Column - I with those in Column - II and choose the correct option:
| Column-I | Column-II | ||
| (a) | Rennin | (i) | Vitamin B12 |
| (b) | Enterokinase | (ii) | Facilitated transport |
| (c) | Oxyntic cells | (iii) | Milk proteins |
| (d) | Fructose | (iv) | Trypsinogen |
| Options: | (a) | (b) | (c) | (d) |
| 1. | iii | iv | ii | i |
| 2. | iv | iii | i | ii |
| 3. | iv | iii | ii | i |
| 4. | iii | iv | i | ii |
Kwashiorkor disease is due to:
| 1. | simultaneous deficiency of proteins and fats |
| 2. | simultaneous deficiency of proteins and calories |
| 3. | deficiency of carbohydrates |
| 4. | protein deficiency not accompanied by calorie deficiency |
Select the correct statement:
| 1. | Expiration occurs due to external intercostal muscles. |
| 2. | Intrapulmonary pressure is lower than atmospheric pressure during inspiration. |
| 3. | Inspiration occurs when atmospheric pressure is less than intrapulmonary pressure. |
| 4. | Expiration is initiated due to the contraction of the diaphragm. |
The maximum volume of air a person can breathe in after a forced expiration is known as:
1. Expiratory Capacity
2. Vital Capacity
3. Inspiratory Capacity
4. Total Lung Capacity
All the components of the nodal tissue are autoexcitable. Why does the SA node act as the normal pacemaker?
| 1. | SA node has the lowest rate of depolarisation |
| 2. | SA node is the only component to generate the threshold potential. |
| 3. | Only SA node can convey the action potential to the other |
| 4. | SA node has the highest rate of depolarisation. |
A specialised nodal tissue embedded in the lower corner of the right atrium, close to atrio-ventricular septum, delays the spreading of impulses to heart apex for about 0.1 sec.
This delay allows -
1. blood to enter aorta.
2. the ventricles to empty completely.
3. blood to enter pulmonary arteries.
4. the atria to empty completely.
Which of the following is not a feature of the active transport of solutes in plants?
1. Occurs against the concentration gradient
2. Non-selective
3. Occurs through membranes
4. Requires ATP
Which of the following bacteria reduce nitrate in soil into nitrogen?
1. Nitrobacter
2. Thiobacillus
3. Nitrococcus
4. Nitrosomonas
What will be the direction of flow of water when a plant cell is placed in a Hypotonic solution?
1. Water will flow in both directions.
2. Water will flow out of the cell.
3. Water will flow into the cell.
4. No flow of water in any direction.
In Hatch and Slack pathway, the primary CO2 acceptor is -
1. Oxaloacetic acid
2. Phosphoglyceric acid
3. Phosphoenol pyruvate
4. Rubisco
One scientist cultured Cladophora in a suspension of Azotobacter and illuminated the culture by splitting light through a prism. He observed that bacteria accumulated mainly in the region of:
| 1. | Violet and green light | 2. | Indigo and green light |
| 3. | Orange and yellow light | 4. | Blue and red light |
The enzyme that is not present in succus entericus is
1. maltase
2. nucleases
3. nucleosidase
4. lipase
The vitamin nicotinamide can be synthesized in our body from
1. tyrosine
2. tryptophan
3. valine
4. phenylalanine
Select the correct match of the digested products in humans given in
column I with their absorption site and mechanism in column II.
|
Column I |
Column II |
|
1. Glycines and Glucose |
Small intestine and active absorption |
|
2. Fructose and Na+ |
Small intestine and passive absorption |
|
3. Glycerol and Fatty acids |
Duodenum and move as chylomicrons |
|
4. Cholesterol and Maltose |
Large intestine and active absorption |
Cattle fed with spoilt hay of sweet clover which contains dicumarol
1. are healthier due to a good diet
2. catch infections easily
3. may suffer vitamin K deficiency and prolonged bleeding
4. may suffer from beri beri due to deficiency of B vitamins
In acute constipation, purgatives that are used to stimulate intestinal peristalsis and evacuation of fluid faeces contain salts of
1. Sodium
2. Magnesium
3. Potassium
4. Calcium
One of the constituents of the pancreatic juice while poured into the duodenum in humans is.
1. Enterokinase
2. Trypsinogen
3. Chymotrypsin
4. Trypsin
Oxygen binding to haemoglobin in blood is:
| 1. | directly proportional to the concentration of in the medium. |
| 2. | inversely proportional to the concentration of in the medium. |
| 3. | directly proportional to the concentration of CO in the medium. |
| 4. | independent of the concentration of CO in the medium. |
People who have migrated from the planes to an area adjoining Rohtang Pass about six months back
1. have more RBCs and their haemoglobin has a lower binding affinity to
2. are not physically fit to play games like football
3. suffer from altitude sickness with symptoms like nausea, fatigue, etc.
4. have the usual RBC count but their haemoglobin has very high binding affinity to
Lungs do not collapse between breaths and some air always remains in the lungs which can never be expelled because
1. there is a negative pressure in the lungs
2. there is a negative intrapleural pressure pulling at the lung walls
3. there is a positive intrapleural pressure
4. pressure in the lungs in higher than the atmospheric pressure
Which of the following match is correct?
1. Emphysema : reduction of surface area of alveoli and bronchi
2. Pneumonia : occupational disease with asbestos
3. Silicosis : inflammation of alveoli
4. Asthma : excessive secretion of bronchial mucus
Which one of the following statements is incorrect?
1. The residual air in lungs slightly decreases the efficiency of respiration in mammals
2. The presence of non-respiratory air sacs, increases the efficiency of respiration in birds
3. In insects, circulating body fluids serve to distribute oxygen to tissues
4. The principle of countercurrent flow facilitates efficient respiration in gills of fishes
A large proportion of oxygen is left unused in the human blood even after its uptake by the body tissues. This
1. raises the of blood to 75 mm of Hg
2. is enough to keep oxyhaemoglobin
3. helps in releasing more to the epithelial tissues
4. acts as a reserve during muscular exercise
Which one of the following is a matching pair of a certain body feature and its value/count in a normal human adult?
1. Urea -5-10 mg/100 ml of blood
2. Blood sugar (fasting) - 80-100 mg/100 ml
3. Total blood volume - 3 - 4 litres
4. ESR in Wintrobe method -9-15 mm in males and 20-34 mm in females
You are required to draw blood from a patient and to keep it in a test tube for analysis of blood corpuscles and plasma. You are also provided with the following four types of test tubes. Which of these will you not use for the purpose?
1. Test tube containing calcium bicarbonate
2. Chilled test tube
3. Test tube containing heparin
4. Test tube containing sodium oxalate
Systemic heart refers to
1. The heart that contracts under stimulation from the nervous system.
2. Left auricle and left ventricle in higher vertebrates.
3. Entire heart in lower vertebrates.
4. The two ventricles together in humans.
Which of the following substances, if introduced into the bloodstream, would cause coagulation of blood at the site of its introduction?
1. Thromboplastin
2. Fibrinogen
3. Heparin
4. Prothrombin
In a standard ECG which one of the following alphabets is the correct representation of the respective activity of the human heart?
1. P-depolarization of ventricles
2. R-repolarization of ventricles
3. S-start of systole
4. T-end of diastole
Erythropoiesis starts in
1. Kidney
2. Liver
3. Spleen
4. Red bone marrow
A few drops of sap were collected by cutting across a plant stem by a suitable method. The sap was tested chemically. Which one of the following test results indicates that it is phloem sap?
1. Acidic
2. Alkaline
3. Low refractive index
4. The absence of sugar
Root pressure develops due to
1. active absorption
2. low osmotic potential in soil
3. passive absorption
4. increase in transpiration
The translocation of organic solutes in sieve tube members is supported by:
1. root pressure and transpiration pull
2. P-proteins
3. mass flow involving a carrier and ATP
4. cytoplasmic streaming
Potometer works on the principle of
1. Potential difference between the tip of the tube and that of the plant
2. Amount of water absorbed equals the amount transpired
3. Osmotic pressure
4. Root pressure
The rupture and fractionation do not usually occur in the water column in vessel/tracheids during the ascent of sap because of
1. lignified thick walls
2. cohesion and adhesion
3. weak gravitational pull
4. transpiration pull
The most abundant intracellular cation is:
1.
2.
3.
4.
Nitrifying bacteria
1. convert free nitrogen to nitrogen compounds
2. convert proteins into ammonia
3. reduce nitrates to free nitrogen
4. oxidize ammonia to nitrates
Sulphur is an important nutrient for optimum growth and productivity in:
1. pulse crops
2. cereals
3. fibre crops
4. oilseed crops
If by radiation all nitrogenase enzymes are inactivated, then there will be no
1. Fixation of atmospheric nitrogen
2. Conversion from nitrate to nitrite in legumes
3. Conversion from ammonium to nitrate in soil
4. Fixation of nitrogen in legumes
In photosystem-I, the first electron acceptor is :
1. ferredoxin
2. cytochrome
3. plastocyanin
4. an iron-sulphur protein
As compared to a plant, how many additional molecules of ATP are needed for net production of one molecule of hexose sugar by plants ?
1. Zero
2. Six
3. Two
4. Twelve
Photosynthesis in \(C_4\) plants is relatively less limited by atmospheric \(CO_2\) levels because
1. The primary fixation of \(CO_2\) is mediated via PEP carboxylase
2. Effective pumping of \(CO_2\) into bundle sheath cells
3. Four carbon acids are the primary initial \(CO_2\) fixation products.
4. Rubisco in \(C_4\) plants has a higher affinity for \(CO_2\)
The -plants are photosynthetically more efficient than -plants because
| 1. | the CO2 compensation point is more |
| 2. | CO2 generated during photorespiration is trapped and recycled through PEP carboxylase |
| 3. | the CO2 efflux is not prevented |
| 4. | they have lower CO2 compensation point. |
The first acceptor of electrons from an excited chlorophyll molecule of photosystem II is :
1. cytochrome
2. iron-sulphur protein
3. ferredoxin
4. quinone
During photorespiration, the oxygen consuming reaction(s) occur in :
1. stroma of chloroplasts and mitochondria
2. stroma of chloroplasts and peroxisomes
3. grana of chloroplasts and peroxisomes
4. stroma of chloroplasts
A molten ionic hydride on electrolysis gives:
1. H+ ion moving towards the cathode.
2. H+ ion moving towards the anode.
3. H2 is liberated at anode.
4. H2 is liberated at cathode.
A 1000 g aqueous solution of CaCO3 contains 10 g of calcium carbonate. The hardness of the solution is:
1. 10 ppm
2. 100 pm
3. 1000 ppm
4. 10000 ppm
Which of the following statement about the sulphate of alkali metal is correct?
1. Expect all sulphate of other alkali metals are soluble in hot water
2. All sulphate salts of alkali metals except lithium sulphate forms alum.
3. All sulphate salts of alkali metals except lithium sulphate do not decompose at high temperature
4. All of the above
Which of the following statements is not true about the dilute solutions of alkali metals in liquid ammonia?
1. They are deep blue coloured solutions.
2. They are highly conducting in nature.
3. They are diamagnetic in nature.
4. Ammoniated cations and ammoniated anions are formed in a solution.
In which of the following compounds, hydrogen exists in the atomic state –
1. Metallic hydrides
2. Ionic hydrides
3. Molecular hydrides
4. Water
Find the volume strength of solution.
1. 8.96
2. 2.16
3. 9.16
4. 5.14
Calcium can be obtained by the electrolysis of-
1. and
2.
3.
4.
A hydrated water soluble salt, A in solution gives a turbidity with dilute hydrochloric acid evolving a gas with a suffocation odour. The solution of A decolourises lodine solution. A is likely to be
1. KI
2.
3.
4.
The boiling point of calcium is abnormally high as compared to magnesium because-
1. In calcium d-orbital forms a weak metallic bond
2. In calcium p-orbital forms a weak metallic bond
3. In calcium d-orbital forms a strong metallic bond
4. In calcium s-orbital forms a strong metallic bond
Alkali metal ions are-
1. Diamagnetic and coloured.
2. Diamagnetic and colourless.
3. Paramagnetic and coloured.
4. Paramagnetic and colourless.
Tin cry refers to-
1. Conversion of white to grey tin
2. Tin plating
3. Conversion of white tetrahedral tin to white rhombohedral tin
4. Emission of sound while bending a tin rod
If a person is injured by the shot of a gun and all the pellets could not be removed, it may cause poisoning by:
1. Hg
2. Pb
3. Fe
4. As
The correct statement regarding polymeric silicon dioxide is -
| 1. | Each silicon atom is surrounded by four oxygen atoms and each oxygen atom is bonded to two silicon atoms. |
| 2. | Each silicon atom is surrounded by two oxygen atoms and each oxygen atom is bonded to two silicon atoms. |
| 3. | Silicon atom is bonded to two oxygen atoms. |
| 4. | These are double bonds between silicon and oxygen atoms. |
Ionic mobility of which of the following alkali metal ions is lowest when aqueous solution of their salts are put under an electric field?
1. K
2. Rb
3. Li
4. Na
Boric acid is an acid because its molecule:
| 1. | contains replaceable H+ ion. |
| 2. | gives up a proton. |
| 3. | accepts OH– from water releasing proton into the solution. |
| 4. | combines with protons from water molecules. |
The correct statement among the following is :
| 1. | Sn2+ is an oxidizing agent while Pb4+ is a reducing agent. |
| 2. | Sn2+ and Pb2+ both are oxidizing agents. |
| 3. | Sn4+ is a reducing agent while Pb4+ is an oxidizing agent. |
| 4. | Sn2+ is a reducing agent while Pb4+ is an oxidizing agent. |
The correct order of atomic radii of the group 13 elements is-
1. B < Al < In < Ga < Tl
2. B < Al < Ga < In < Tl
3. B < Ga < Al < Tl < In
4. B < Ga < Al < In < Tl
An incorrect statement among the following regarding hydrogen is:
| 1. | Hydrogen never acts as a cation in ionic salts. |
| 2. | Hydronium ion, H3O+ exists freely in a solution. |
| 3. | Dihydrogen does not act as a reducing agent. |
| 4. | Hydrogen has three isotopes of which protium is the most common. |
Which of the following biologically important ions is also a constituent of the sodium pump?
1. Ca2+
2. Mg2+
3. K+
4. Fe2+
A compound among the following having a similar structure to graphite is:
1. B
2. B4C
3. B2H6
4. BN
The number of carbon atoms per unit cell of a diamond is:
1. 8
2. 6
3. 1
4. 4
| 1. | Sheet silicate | 2. | Pyrosilicate |
| 3. | Three dimensional silicate | 4. | Linear chain silicate |
An active ingredient in bleaching powder that aids in the bleaching action is:
1.
2.
3.
4.
In which of the following the hydration energy is higher than the lattice energy?
1.
2.
3.
4.
Al2O3 can be converted to anhydrous AlCl3 by heating:
1.
2. with NaCl in solid-state
3. a mixture of and carbon in dry gas
4. with gas
The incorrect statement among the following is:
1. SnF4 is ionic in nature.
2. PbF4 is covalent in nature.
3. SiCl4 is easily hydrolysed.
4. GeX4 (X = F, Cl, Br, I) is more stable than GeX2
The method used to remove the temporary hardness of water is-
1. Synthetic resins method
2. Calgon's method
3. Clark's method
4. Ion-exchange method
Heavy water is ......% heavier than ordinary water
1. 11
2. 20
3. 25
4. 15
Benzene is oxidised by in the presence of to:
1. phenol
2. cyclohexane
3. benzaldehyde
4. benzoic acid
For the bleaching of hair, the substance used is:
1.
2. Bleaching powder
3.
4.
Para- hydrogen at room temp is:
1. less stable than ortho- hydrogen
2. more stable than ortho- hydrogen
3. as stable as ortho- hydrogen
4. none of these
Reaction among the following where acts as a reducing agent is-
a.
b.
c.
d.
1. a, b
2. c, d
3. a, c
4. b, d
Boric acid is polymeric because of:
1. Its acidic nature
2. Presence of hydrogen bonds
3. Its monobasic nature
4. Its geometry
Boric acid on heating at gives:
1.
2.
3.
4.
When reacts with , the product formed is-
1.
2.
3.
4. None of the above
The soldiers of the Napolean army while at the Alps during freezing winter suffered a serious problem with regards to the tin buttons of their uniforms. White Metallic tin buttons get converted to grey powder. This transformation is related to-
1. An interaction with water vapour contained in humid air
2. A change in the crystalline structure of tin
3. A change in the partial pressure of in the air
4. An interaction with of air at low temperature
The amphoteric oxide among the following is -
| 1. | \(\mathrm{CO}_2 \) | 2. | \(\mathrm{PbO}_2 \) |
| 3. | \(\mathrm{SiO}_2 \) | 4. | \(\mathrm{SeO}_2\) |
Aluminium chloride in acidified aqueous solution forms a complex 'A'.
The formula of A and hybridisation state of Al in 'A' is respectively:
1. [Al(H2O)6]3+, sp3d2
2. [Al(H2O)4]3+, sp3
3. [Al(H2O)4]3+, dsp2
4. [Al(H2O)]3+, d2sp3
The compound among the following that used in cosmetic surgery is:
1. Silica
2. Silicates
3. Silicones
4. Zeolites
A compound "X", upon reaction with H2O, produces a colourless gas "Y" with a rotten fish smell. Gas 'Y' is absorbed in a solution of CuSO4 to give Cu3P2 as one of the products. The compound 'X' will be:
1. Ca3P2
2.
3.
4. Ca3(PO4)2
In the Solvay process of manufacture of sodium carbonate, the raw materials used are :
1. Aqueous NaOH, and
2. Molten NaOH, and CO
3. Brine NaCl, and CO
4. Brine NaCl, and
Litharge is not commonly used in:
1. Manufacture of special glasses
2. Glazing pottery
3. Preparing paints
4. Lead storage battery
The products formed on heating Pb(NO3)2
1.
2. Pb(NO2
3.
4.
Insoluble compound in acetic acid is-
1. Calcium oxide
2. Calcium carbonate
3. Calcium oxalate
4. Calcium hydroxide
1. 15 g
2. 30 g
3. 50 g
4 60 g
In the figure below is shown the flow of liquid through a horizontal pipe. Three tubes A, B, and C are connected to the pipe. The radius of the pipe at A, B, and C is respectively 2 cm, 1 cm, and 2 cm. It can be said that the :

1. Height of the liquid in the tube A is maximum
2. Height of the liquid in the tubes A and B is the same
3. Height of the liquid in all the three tubes is the same
4. Height of the liquid in the tubes A and C is the same
The P-V diagram for a thermodynamic system is shown in the figure. The work done by the system during the cyclic process ABCA is

1. Zero
2. 90 J
3. 30 J
4. 60 J
An ideal gas changes from state 'a' to state 'b' as shown in figure. What is the work done by the gas in the process?

1. zero
2. positive
3. negative
4. infinite
A beaker containing a liquid is kept inside a big closed jar. If the air inside the jar is continuously pumped out, the pressure in the liquid near the bottom of the beaker will
1. Increase
2. Decrease
3. Remain constant
4. First decrease and then increase
In making an alloy, a substance of specific gravity and mass is mixed with another substance of specific gravity and mass ; then the specific gravity of the alloy is
1.
2.
3.
4.
Two solids A and B float in water. It is observed that A floats with of its body immersed in water and B floats with of its volume above the water level. The ratio of the density of A to that of B is
1. 4 : 3
2. 2 : 3
3. 3 : 4
4. 1 : 2
\({P \over \rho g} + h+ {v^2 \over 2 g} = \text {constant}\\ \small{(A)}~~~~\small {(B)}~~~\small {(C)}\)
The terms \(A\), \(B\) and \(C\) are generally called respectively:
1. Gravitational head, pressure head and velocity head
2. Gravity, gravitational head and velocity head
3. Pressure head, gravitational head and velocity head
4. Gravity, pressure and velocity head
A manometer connected to a closed tap reads pascal. When the tap is opened the reading of the manometer falls to pascal. Then the velocity of flow of water is
1. 7
2. 8
3. 9
4. 10
Two small drops of mercury, each of radius r, coalesce to form a single large drop. The ratio of the total surface energies before and after the change is
1.
2.
3. 2:1
4. 1:2
Two bubbles A and B are joined through a narrow tube where bubble A is bigger. Then
1. The size of A will increase
2. The size of B will increase
3. The size of B will increase until the pressure equals
4. None of these
A capillary tube made of glass is dipped into mercury. Then
1. Mercury rises in the capillary tube
2. Mercury rises and flows out of the capillary tube
3. Mercury descends in the capillary tube
4. Mercury neither rises nor descends in the capillary tube
In rainy season, on a clear night the black seat of a bicycle becomes wet because -
1. It absorbs water vapour
2. Black seat is good absorber of heat
3. Black seat is good radiator of heat energy
4. None of the above
A piece of blue glass heated to a high temperature and a piece of red glass at room temperature, are taken inside a dimly lit room. Then -
1. The blue piece will look blue and red will look as usual
2. Red look brighter red and blue look ordinary blue
3. Blue shines like brighter red compared to the red piece
4. Both the pieces will look equally red.
We consider a thermodynamic system. If ΔU represents the increase in its internal energy and W the work done by the system, which of the following statements is true ?
1. ΔU = –W in an adiabatic process
2. ΔU = W in an isothermal process
3. ΔU = –W in an isothermal process
4. ΔU = W in an adiabatic process
In a reversible isochoric change -
1. ΔW = 0
2. ΔQ = 0
3. ΔT = 0
4. ΔU = 0
If the door of a refrigerator is kept open, then which of the following is true ?
1. Room is cooled
2. Room is heated
3. Room is either cooled or heated
4. Room is neither cooled nor heated
The total energy radiated from a black body source is collected for one minute and is used to heat a quantity of water. The temperature of water is found to increase from 20 to 20.5 . If the absolute temperature of the black body is doubled and the experiment is repeated with the same quantity of water at 20, the temperature of water will be
1. 21 2. 22
3. 24 4. 28
Thermodynamic processes are indicated in the following diagram:
Match the following:
| Column-I | Column-II | ||
| (P) | Process I | (a) | Adiabatic |
| (Q) | Process II | (b) | Isobaric |
| (R) | Process III | (c) | Isochoric |
| (S) | Process IV | (d) | Isothermal |
| 1. | P → c, Q → a, R → d, S→ b |
| 2. | P→ c, Q → d, R → b, S → a |
| 3. | P → d, Q → b, R → b, S → c |
| 4. | P → a, Q → c, R → d, S → b |
A rectangular film of liquid is extended from \((4~\text{cm} \times 2~\text{cm})\) to \((5~\text{cm} \times 4~\text{cm}).\) If the work done is \(3\times 10^{-4}~\text J,\) then the value of the surface tension of the liquid is:
| 1. | \(0.250~\text{Nm}^{-1}\) | 2. | \(0.125~\text{Nm}^{-1}\) |
| 3. | \(0.2~\text{Nm}^{-1}\) | 4. | \(8.0~\text{Nm}^{-1}\) |
| 1. | \(\dfrac{3}{2}R\) | 2. | \(\dfrac{5}{2}R\) |
| 3. | \(2R\) | 4. | \(R\) |
The temperature inside a refrigerator (reversible process) is t2oC and the room temperature is t1oC. The amount of heat delivered to the room for each joule of
electrical energy consumed, ideally, will be:
1.
2.
3.
4.
| 1. | \([2+(n+1)r ]\rho\) | 2. | \([2+(n-1)r] \rho\) |
| 3. | \([1+(n-1)r] \rho\) | 4. | \([1+(n+1)r ]\rho\) |
A gas is compressed isothermally to half its initial volume. The same gas is compressed separately through an adiabatic process until its volume is again reduced to half. Then,
| 1. | compressing the gas through an adiabatic process will require more work to be done. |
| 2. | compressing the gas isothermally or adiabatically will require the same amount of work. |
| 3. | which of the case (whether compression through isothermal or through the adiabatic process) requires more work will depend upon the atomicity of the gas. |
| 4. | compressing the gas isothermally will require more work to be done. |
Two metal wires of identical dimensions are connected in series. If \(\sigma_1~\text{and}~\sigma_2\)
1. \(\dfrac{2\sigma_1 \sigma_2}{\sigma_1+\sigma_2}\)
2. \(\dfrac{\sigma_1 +\sigma_2}{2\sigma_1\sigma_2}\)
3. \(\dfrac{\sigma_1 +\sigma_2}{\sigma_1\sigma_2}\)
4. \(\dfrac{\sigma_1 \sigma_2}{\sigma_1+\sigma_2}\)
The value of the coefficient of volume expansion of glycerine is \(5\times10^{-4}~\text{K}^{-1}.\) The fractional change in the density of glycerine for a rise of \(40^\circ \text{C}\) in its temperature is:
| 1. | \(0.015\) | 2. | \(0.020\) |
| 3. | \(0.025\) | 4. | \(0.010\) |
The heart of a man pumps \(5~\text{L}\) of blood through the arteries per minute at a pressure of \(150~\text{mm}\) of mercury. If the density of mercury is \(13.6\times10^{3}~\text{kg/m}^{3}\) \(g = 10~\text{m/s}^2,\) then the power of the heart in watts is:
| 1. | \(1.70\) | 2. | \(2.35\) |
| 3. | \(3.0\) | 4. | \(1.50\) |
Water rises to a height ‘h’ in the capillary tube. If the length of the capillary tube is made less than ‘h’, then,
1. Water rises up to the tip of the capillary tube and then starts overflowing like a fountain.
2. Water rises up to the top of the capillary tube and stays there without overflowing.
3. Water rises up to a point little below the top and stays there.
4. Water does not rise at all.
A certain number of spherical drops of a liquid of radius \({r}\) coalesce to form a single drop of radius \({R}\) and volume \({V}.\) If \({T}\) is the surface tension of the liquid, then:
| 1. | the energy \(= 4{VT}\left( \frac{1}{{r}} - \frac{1}{{R}}\right)\) is released. |
| 2. | the energy \(={ 3{VT}\left( \frac{1}{{r}} + \frac{1}{{R}}\right)}\) is released. |
| 3. | the energy \(={ 3{VT}\left( \frac{1}{{r}} - \frac{1}{{R}}\right)}\) is released. |
| 4. | the energy is neither released nor absorbed. |
| 1. | surface tension. |
| 2. | density. |
| 3. | angle of contact between the surface and the liquid. |
| 4. | viscosity. |
| 1. | \(275~\text{K}\) | 2. | \(325~\text{K}\) |
| 3. | \(250~\text{K}\) | 4. | \(380~\text{K}\) |
A tube of length \( L\) is filled completely with an incompressible liquid of mass \(M\) and closed at both ends. The tube is then rotated in a horizontal plane about one of its ends with a uniform angular velocity \(\omega\). The force exerted by the liquid at the other end is:
| 1. | \(ML \omega^2 \over 2\) | 2. | \(ML^2 \omega \over 2\) |
| 3. | \(ML \omega^2 \) | 4. | \(ML^2 \omega^2 \over 2\) |
The plots of intensity versus wavelength for three black bodies at temperatures , and respectively are as shown. Their temperatures are such that :
1.
2.
3.
4.
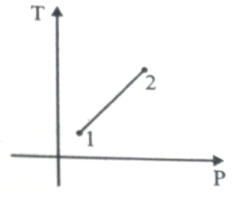
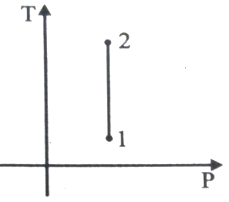
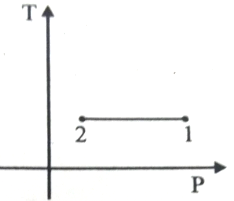
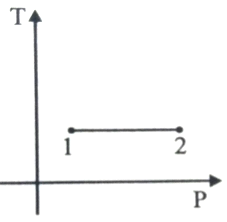
For the P-V diagram given for an ideal gas,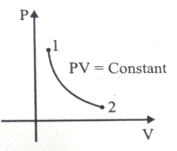
Out of the following which one correctly represents the T-P diagram?
1. 
2. 
3.
4.
A gas is compressed from a volume of to a volume of at constant pressure of 100 N/. Then it is heated at constant volume by supplying 150 J of energy. As a result, the internal energy of the gas :
1. increases by 250 J
2. decreases by 250 J
3. increases by 50 J
4. decreases by 50 J
Two moles of an ideal monoatomic gas occupies a volume V at 27. The gas expands adiabatically to a volume of 2V. Calculate final temperature of the gas .
1. 295 K
2. 189 K
3. 395 K
4. 298 K
The ratio of work done by an ideal monoatomic gas to the heat supplied to it in an isobaric process is:
1. 3/5
2. 2/3
3. 3/2
4. 2/5
An object kept in a large room having an air temperature of \(25^\circ \text{C}\) takes \(12 ~\text{min}\) to cool from \(80^\circ \text{C}\) to \(70^\circ \text{C}.\) The time taken to cool for the same object from \(70^\circ \text{C}\) to \(60^\circ \text{C}\) would be nearly:
| 1. | \(10 ~\text{min}\) | 2. | \(12 ~\text{min}\) |
| 3. | \(20 ~\text{min}\) | 4. | \(15 ~\text{min}\) |
Two small spherical metal balls, having equal masses, are made from materials of densities \(\rho_1\) and \(\rho_2\) such that \(\rho_1=8\rho_2\)
| 1. | \(\dfrac{79}{72}\) | 2. | \(\dfrac{19}{36}\) |
| 3. | \(\dfrac{39}{72}\) | 4. | \(\dfrac{79}{36}\) |
In a U-tube, as shown in the figure, the water and oil are in the left side and right side of the tube respectively. The height of the water and oil columns are \(15~\text{cm}\) and \(20~\text{cm}\) respectively. The density of the oil is:
\(\left[\text{take}~\rho_{\text{water}}= 1000~\text{kg/m}^{3}\right]\)
| 1. | \(1200~\text{kg/m}^{3}\) | 2. | \(750~\text{kg/m}^{3}\) |
| 3. | \(1000~\text{kg/m}^{3}\) | 4. | \(1333~\text{kg/m}^{3}\) |
A deep rectangular pond of surface area \(A\), containing water (density = \(\rho,\) specific heat capacity = \(s\)), is located in a region where the outside air temperature is at a steady value of \(-26^{\circ}\text{C}\). The thickness of the ice layer in this pond at a certain instant is \(x\). Taking the thermal conductivity of ice as \(k\), and its specific latent heat of fusion as \(L\), the rate of increase of the thickness of the ice layer, at this instant, would be given by:
| 1. | \(\dfrac{26k}{x\rho L-4s}\) | 2. | \(\dfrac{26k}{x^2\rho L}\) |
| 3. | \(\dfrac{26k}{x\rho L}\) | 4. | \(\dfrac{26k}{x\rho L+4s}\) |
\(1~\text g\) of water of volume \(1~\text{cm}^3\) at \(100^\circ \text{C}\) is converted into steam at the same temperature under normal atmospheric pressure \(\approx 1\times10^{5}~\text{Pa}.\) The volume of steam formed equals \(1671~\text{cm}^3.\) If the specific latent heat of vaporization of water is \(2256~\text{J/g},\) the change in internal energy is:
| 1. | \(2423~\text J\) | 2. | \(2089~\text J\) |
| 3. | \(167~\text J\) | 4. | \(2256~\text J\) |
A fixed mass of an ideal gas undergoes a change in which it is supplied with \(3500~\text J\) of thermal energy. At the same time, this gas does \(3500~\text J\) of work on its surroundings. Which type of change does the gas undergo during this time?
1. adiabatic
2. isothermal
3. isochoric
4. isomeric