In cymose inflorescence when shoot tip gets converted into flower, than flower is ?
1. It is always solitary
2. It is in cluster
3. It is always sympodial
4. It is always acropetal
In a flower staminode represents
1. Fertile stamen
2. Sterile stamen
3. Under develop stamen
4. Bunch of stamen
Match the following
A. Diadelphous (i) Malvaceae
B. Polyadelphous (ii) Fabaceae
C. Monodelphous (iii) Solanaceae
D. Epipetalous (iv) Rutaceae
1. A – ii, B – iv, C – i, D – iii
2. A – iv, B – i, C – ii, D – iii
3. A – iv, B – iii, C – i, D – ii
4. A – i, B – iv, C – iii, D – ii
These are the names of plants, maximum number of plants belong to which family
(i) Sem (ii) Sesbania (iii) Muliathi (iv) Petunia (v) Chilli (vi) belladonna (vii) Asparagus
(viii) Gloriosa (ix) Colchicum (x) Trifolium
1. Solanacea
2. Malvaceae
3. Fabaceae
4. Liliaceae
Which one of the following is not a medicinal plant
1. Lupin
2. Belladonna
3. Aloe
4. Ashwagandha
If false septum is present. Than what would be the placentation
1. Axile
2. Parietal
3. Marginal
4. Free central
Which of the following statement is not correct about Cajanus cajan
1. Seed is endospermic
2. Source of pulses
3. Vexillary aestivation
4. Ovary superior
Roots become modified to perform functions like
1. Additional support and absorption of water
2. Absorption and conduction of water and minerals
3. Storage of food and respiration
4. Absorption of minerals and storage of food
Select correct option with respect to Mangroves
(i) Grows in swampy area
(ii) Pneumatophores are present for respiration
(iii) Is halophyte
(iv) Shows in-situ germination of seed
1. All are correct
2. All are correct except (iv)
3. All are correct except (i) & (iv)
4. All are correct except (i), (iii), & (iv)
Fruit of coconut plant is developed from
1. Monocarpellary ovary
2. Superior ovary
3. Epigynous flower
4. Both a and b
The type of sub-aerial weak stem present in Pistia is also present in
1. Jasmine
2. Pineapple
3. Strawberry
4. Eichhornia
Read the following statement carefully
(i) In dicots, the leaf base expands into a sheath covering the stem partially or wholly
(ii) Veins provide rigidity to the leaf blade.
(iii) In monocots, leaf base is swollen and is known as pulvinus.
(iv) The petiole holds the blade to light
How many statement(s) is/are incorrect?
1. Two
2. Four
3. One
4. Three
Which of the following pairs of cells is related ontogenetically?
| 1. | Trachieds and tracheae |
| 2. | Sieve cells and sieve tube components |
| 3. | Sieve tube members and phloem |
| 4. | Sieve tube members and companion cells |
Conjunctive tissue is
1. parenchymatous tissue present between two vascular bundles in dicot stem
2. parenchymatous tissue present between xylem and phloem in root
3. sclerenchymatous tissue present in cortex
4. lateral meristem
Protoxylem and stomata in a dorsiventral leaf are present
1. on the abaxial and adaxial sides, respectively
2. on the adaxial and abaxial sides, respectively
3. on the adaxial side
4. on the abaxial side
The cells shown in the below diagram are of ______ tissue and can be seen in ______ layer of the dicot stem.
1. Collenchyma, Hypodermis
2. Sclerenchyma, Sclereids
3. Parenchyma, Pericycle
4. Parenchyma, Pith
Mechanical support to the growing parts of the plant such as young stem and petiole of leaf is provided by a living tissue called
| 1. | Collenchyma | 2. | Aerenchyma |
| 3. | Chlorenchyma | 4. | Sclerenchyma |
Which of the following is correct statement for intercalary meristems?
1. They lie at tip of root apex
2. They lie at base of stem always
3. They are short lived and consumed during primary growth
4. They are long lived and cause increase in length
Compare diagram ‘a’ with ‘b’ and find out which statement is incorrect about difference between these two diagrams.
| 1. | In ‘a’ mesophyll is of two type where as in case of ‘b’ mesophyll is of single type |
| 2. | In ‘a’ stomata is mainly at lower surface whereas in ‘b’ stomata is at both the surface |
| 3. | In ‘a’ vascular bundle is of different size whereas in ‘b’ vascular bundle is of more or less equal size |
| 4. | In ‘a’ xylem is towards adaxial surface whereas in ‘b’ xylem is towards abaxial surface |
A large number of proteins made by the ribosomes get modified in
| 1. | Lysosomes. |
| 2. | Trans face of Golgi apparatus. |
| 3. | Cisternae of Golgi apparatus. |
| 4. | Smooth endoplasmic reticulum. |
Three of the following statements regarding cell organelles are correct while one is wrong. Which one is wrong?
| 1. | Lysosomes are double membraned vesicles budded off from golgi bodies and contain digestive enzymes. |
| 2. | Endoplasmic reticulum consists of a network of membranous tubules and helps in transport, synthesis and secretion. |
| 3. | Leucoplasts are bound by two membranes, lack pigments but contain their own DNA and protein synthesizing machinery. |
| 4. | Sphaerosomes are single membrane bound and are associated with synthesis and storage of lipids. |
Consider the following cellular functions:
A. Transport of substances.
B. Synthesis of proteins.
C. Synthesis of steroidal hormones.
D. Glycogen metabolism.
E. Detoxification of drugs
F. Giving rise to sphaerosomes.
ER helps in how many of the above functions?
1. Two
2. Four
3. Six
4. Five
Select the incorrect statement:
1. Leeuwenhoek first described a live cell
2. Robert Brown discovered the nucleus
3. Robert Hooke first saw a living cell
4. Schwann proposed that bodies of animals and plants are composed of cells and
products of cells
"One of the most important functions of the plasma membrane is the transport of molecules." Which of the following is correct with respect to this statement?
| 1. | Polar molecules can pass easily through the lipid bilayer |
| 2. | The membrane does not support passive transport |
| 3. | Neutral solutes require a carrier protein of membrane for transport |
| 4. | The memebrane is selectively permeable to some molecules present on either side of it |
Mitochondria and plastids are similar in all of the following aspects, except:
| 1. | Possess circular ds-DNA |
| 2. | Have endo-symbiotic origin |
| 3. | Are the sites of carbohydrate biosynthesis |
| 4. | Divide by fission |
Lysosomes are:
1. Double membrane bound vesicular structures
2. Rich in few hydrolases
3. Formed by packaging in Golgi apparatus
4. Formed by engulfing the food particles
Choose the correct statement with respect to the following diagram: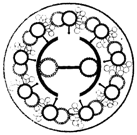
1. Section of flagella showing different parts seen in eukaryotes and prokaryotes
2. It shows the cartwheel organisation with 9 + 0 microtubular arrangement
3. They form structures which cause movement of either the cell or the surrounding fluid
4. It forms structure which is not covered by any membraneHow many of the following features
are associated', with the endomembrane system?
Which of the following is correct about cell cycle?
| 1. | All events occur in coordinated manner |
| 2. | All events are under genetic control |
| 3. | DNA synthesis occurs only during one specific stage in the cell cycle |
| 4. | All |
Identify A, B, C, D in below diagram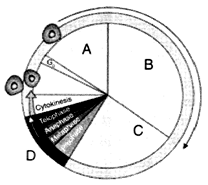
1. A-G1, B-S, C-G2, D-M Phase
2. A-G2, B-M Phase, C-G1, D-S
3. A-S, B-G2, C-G1, D-M Phase
4. A-M Phase, B-G1, C-G1, D-S
Events of telophase are
| 1. | chromosomes cluster at opposite spindle poles and their identity is lost as discrete elements |
| 2. | nuclear envelope assembles around the chromosome cluster |
| 3. | nucleolus, Golgi complex and ER reforms |
| 4. | all |
Select the total number of correct statements.
| I. | Cell-plate formation occurs in plant cell during cytokinesis. |
| II. | During cytokinesis, mitochondria and plastid get distributed between two daughter cells in mitosis. |
| III. | Liquid endosperm in coconut is syncytium |
| IV. | Furrow formation occur in animal cell during cytokinesis. |
1. 1 only
2. 2 only
3. 3 only
4. 4 only
Mitotic anaphase differs from metaphase in possessing
1. same number of chromosomes and half number of chromatids
2. half number of chromosomes and same number of chromatids
3. half number of chromosomes and half number of chromatids
4. same number of chromosomes and same number of chromatid
Mitosis differs from meiosis in
1. forming four haploid cells
2. pairing of homologous chromosomes and their subsequent separation
3. doubling of each chromosome and each pair showing four chromatids
4. duplication of chromosomes and subsequent separation of the duplicates
Meiosis involves
| 1. | two nuclear divisions and two chromosome divisions |
| 2. | two nuclear divisions and one chromosome division |
| 3. | one nuclear division and one chromosome division |
| 4. | one nuclear division and two chromosome divisions |
Synaptonemal complex has a role in
1. chromosome pairing
2. chromosome movement
3. chromosome segregation
4. chromosome organization
Cross-like configuration when nonsister chromatids of a bivalent come in contact during first meiotic division are
1. chiasmata
2. bivalents
3. chromomeres
4. centromeres
Significance of meiosis lies in the
1. reduction of chromosome number to one half
2. maintaining constancy of chromosome number during sexual reproduction
3. production of genetic variability
4. all of these
The type of reaction that would link monomers in the formation of a macromolecule is:
1. hydrophobic reaction
2. hydrolysis reaction
3. dehydration reaction
4. denaturation reaction
Cellulose is the most common organic compound on earth yet it is difficult for most animals to digest primarily because:
1. they don't have the proper enzyme to break the bonds between subunits
2. cellulose is made up of chitin, which is indigestible
3. the bonds holding cellulose subunits together are extremely strong, stronger than in any other macromolecule
4. there are many hydrogen bonds holding the subunits together
The molecule shown below can be: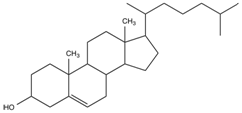
1. the major constituent of cell membranes
2. a major source of energy for animals
3. storage form of carbohydrate in animals
4. a precursor of steroid hormones in animals
The level of polypeptide folding in which the primary sequence coils around itself, stabilized by regularly spaced hydrogen bonds, is called:
1. primary structure
2. alpha helix
3. beta sheet
4. tertiary structure
The structure of ATP molecule most closely resembles that of:
| 1. | Ribonucleotide | 2. | Deoxyribonucleotide |
| 3. | Gamma amino acid | 4. | Saturated fatty acid |
Identify the incorrect statement regarding enzymes:
| 1. | Enzymes are proteins that function as catalysts. |
| 2. | Enzymes are specific. |
| 3. | Enzymes provide activation energy for reactions. |
| 4. | Enzyme activity can be regulated. |
In the graph given below which letter represents the reaction occurring in the presence of a competitive inhibitor of the enzyme?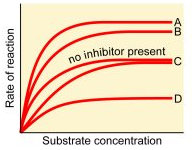
1. A
2. B
3. C
4. D
Which of the following cell organelles is present in the highest number in secretory cells?
| 1. | Mitochondria | 2. | Golgi complex |
| 3. | Endoplasmic reticulum | 4. | Lysosomes |
Non-membranous nucleoplasmic structures in the nucleus, are the sites for active synthesis of:
| 1. | protein | 2. | mRNA |
| 3. | rRNA | 4. | tRNA |
Which of the following nucleic acids is present in an organism having 70 S ribosomes only?
| 1. | Single-stranded DNA with a protein coat |
| 2. | Double-stranded circular naked DNA |
| 3. | Double-stranded DNA enclosed in nuclear membrane |
| 4. | Double-stranded circular DNA with histone proteins |
After meiosis I, the resultant daughter cells have
| 1. | same amount of DNA as in the parent cell in S |
| 2. | twice the amount of DNA in comparison to haploid |
| 3. | same amount of DNA in comparison to haploid gamete |
| 4. | four times the amount of DNA in comparison to haploid gamete |
Which of the following organic compounds is the main constituent of Lecithin?
| 1. | Arachidonic acid | 2. | Phospholipid |
| 3. | Cholesterol | 4. | Phosphoprotein |
In the dicot root, the vascular cambium originates from:
| 1. | Tissue located below the phloem bundles and a portion of pericycle tissue above protoxylem |
| 2. | Cortical region |
| 3. | Parenchyma between endodermis and pericycle |
| 4. | Intrafascicular and interfascicular tissue in a ring |
Which of the following shows whorled phyllotaxy?
| 1. | Mustard | 2. | China rose |
| 3. | Alstonia | 4. | Calotropis |
Regeneration of damaged growing grass following grazing is largely due to:
| 1. | Lateral meristem | 2. | Apical meristem |
| 3. | Intercalary meristem | 4. | Secondary meristem |
Bicarpellary ovary with obliquely placed septum is seen in:
1. Brassica
2. Aloe
3. Solanum
4. Sesbania
Match Column-I with Column-II.
| Column-I | Column-II | ||
| (a) | Golgi apparatus | (i) | Synthesis of protein |
| (b) | Lysosomes | (ii) | Trap waste and excretory products |
| (c) | Vacuoles | (iii) | Formation of glycoproteins and glycolipids |
| (d) | Ribosomes | (iv) | Digesting biomolecules |
Choose the right match from the options given below:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iii) | (iv) | (ii) | (i) |
| 2. | (iv) | (iii) | (i) | (ii) |
| 3. | (iii) | (ii) | (iv) | (i) |
| 4. | (i) | (ii) | (iv) | (iii) |
Prosthetic groups differ from co-enzymes, in which:
| 1. | they require metal ions for their activity. |
| 2. | they (prosthetic groups) are tightly bound to apoenzymes. |
| 3. | their association with apoenzymes is transient. |
| 4. | they can serve as co-factors in a number of enzyme-catalyzed reactions. |
Crossing over takes place between which chromatids and in which stage of the cell cycle?
| 1. | Non-sister chromatids of non-homologous chromosomes at Zygotene stage of prophase I. |
| 2. | Non-sister chromatids of homologous chromosomes at Pachytene stage of prophase I. |
| 3. | Non-sister chromatids of homologous chromosomes at Zygotene stage of prophase I. |
| 4. | Non-sister chromatids of non-homologous chromosomes at Pachytene stage of prophase I. |
"Ramachandran plot" is used to confirm the structure of:
1. RNA
2. Proteins
3. Triacylglycerides
4. DNA
Match the placental types Column-I with their examples Column-II.
| Column-I | Column-II | ||
| (a) | Basal | (i) | Mustard |
| (b) | Axile | (ii) | China rose |
| (c) | Parietal | (iii) | Dianthus |
| (d) | Free central | (iv) | Sunflower |
Choose the correct answer from the following options:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (ii) | (iii) | (iv) | (i) |
| 2. | (i) | (ii) | (iii) | (iv) |
| 3. | (iv) | (ii) | (i) | (iii) |
| 4. | (iii) | (iv) | (i) | (ii) |
Due to discovery of which of the following in 1980 the evolution was termed as RNA world?
| 1. | mRNA, tRNA, rRNA synthesize proteins |
| 2. | In some virus RNA is genetic material |
| 3. | RNA have enzymatic property |
| 4. | RNAis not found in all cells 1980 |
Telomerase is an enzyme which is a
1. repetitive DNA
2. RNA
3. simple protein
4. ribonucleoprotein
In curve of enzyme catalyzed reaction, the value of velocity of enzyme reaction at point A will be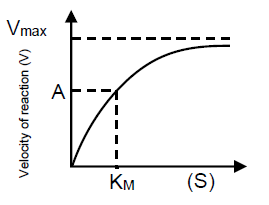
1. and velocity may increase by increasing temperature
2. and velocity may increase by increasing substrate
3. and velocity may increase by increasing by catalyst
4. and velocity remain constant even changing any factor in to the medium.
Which of the following is the least likely to be involved in stabilizing the three-dimensional folding of most proteins?
1. Ester bonds
2. Hydrogen bonds
3. Electrostatic interaction
4. Hydrophobic interaction
Cellulose, the most important constituent of plant cell wall is made of
1. Unbranched chain of glucose molecules linked by α 1, 4 glycosidic bond
2. Branched chain of glucose molecules linked by β 1, 4 glycosidic bond in straight chain and β 1, 6 glycosidic bond at the site of branching
3. Unbranched chain of glucose molecules linked by β 1, 4 glycosidic bond
4. Branched chain of glucose molecules linked by α 1, 6 glycosidic bond at the site of branching
P-protein is found in
1. Parenchyma
2. Collenchyma
3. Sieve tube
4. Xylem
Tracheids are
| 1. | Elongated cells with tapering ends and unperforated end walls |
| 2. | Elongated cells with tapering ends and perforated end wall |
| 3. | Rounded cells with lignified walls |
| 4. | Both 1 and 2 |
Interxylary phloem formed in Salvadora due to abnormal secondary growth is also known as
1. Included phloem
2. Internal phloem
3. External phloem
4. Vestigial phloem
Cambium found in vascular bundles of dicot stem is
1. Intercalary meristem
2. Fasicular cambium
3. Secondary meristem
4. All of the above
Vessels are found in
| 1. | All angiosperms |
| 2. | Most angiosperms, few gymnosperms and few pteridophytes |
| 3. | All angiosperms and few gymnosperms |
| 4. | Most angiosperms and few gymnosperms |
The cells of the quiescent center are characterized by
1. having dense cytoplasm and prominent nuclei
2. having light cytoplasm and small nuclei
3.dividing regularly to add to the corpus
4. dividing regularly to add to tunica
Water containing cavities in vascular bundles are found in
1. Sunflower
2. Maize
3. Cycas
4. Pinus
Prokaryotic and eukaryotic flagella differ in
| 1. | Type of movement and placement |
| 2. | Location and mode of functioning |
| 3. | Microtubular structure and function |
| 4. | Microtubular organisation and type of movement |
In eubacteria, a cellular component that resembles eukaryotic cells is
1. Plasma membrane
2. Nucleus
3. Ribosomes
4. Cell wall
The solid linear cytoskeletal elements having a diameter of 6 nm and made up of a single type of monomer are known as :
1. Microtubules
2. Microfilaments
3. Intermediate filaments
4. Lamins
During which phase(s) of cell cycle, amount of DNA in a cell remains at 4 C level if the initial amount is denoted as 2C ?
1. G0 and G1
2. G1 and S
3. Only G2
4. G2 and M
Which of the following plants are used as green manure in crop fields and in sandy soils
1.Crotolariajuncea and Alhagicamelorum
2. Calotropisprocera and Phyllanthusnintri
3. Saccharummunja and Lantana camara
4. Dichanthiumannulatum and Azollanilotica
A racemose type of inflorescence with its main axis almost flat is called
1. Corymb
2. Umbel
3. Spike
4. Capitulum
An ovule bent to come at right angles to funicle is
1. Anatropous
2. Orthotropous
3. Campylotropous
4. Hemitropous.
Anthesis is
1. Opening of floral bud
2. Development of anthers
3. Maturation of anthers
4. Reception of pollen by stigma
Which one of the following organisms is correctly matched with its three characteristics?
1. Pea: C3 pathway, Endospermic seed, Vexillary aestivation
2. Tomato: Twisted aestivation, Axile placentation, Berry
3. Onion: Bulb, Imbricate aestivation, Axile placentation
4. Maize : C3 pathway, Closed vascular bundles, Scutellum
Juicy hair like structures observed in lemon fruit develop from
1. Exocarp
2. Mesocarp
3. Endocarp
4. Mesocarp and endocarp.
Dry indehiscent single-seeded fruits formed from bicarpellarysyncarpous inferior ovary is
1. Berry
2. Cremocarp
3. Cypsela
4. Caryopsis.
How many plants in the list given below have composite fruits that develop from an inflorescence walnut, poppy, radish, fig, pineapple, apple, tomato, mulberry
1. Four
2. Five
3. Two
4. Three
An aggregate fruit is one which developed from
1. Multicarpellarysyncarpous gynoecium
2. Multicarpellaryapocarpous gynoecium
3. Complete inflorescence
4. Multicarpellary superior ovary
How many plants among Indigofera, Sesbania, Salvia, Allium, Aloe, mustard, groundnut, radish, gram and turnip have stamens with different lengths in their flowers?
1. Six
2. Three
3. Four
4. Five
A bacterium divides every 35 minutes. If a culture containing 105 cells per mL is grown for 175 minutes. What will be the cell concentration per mL after 175 minutes ?
1. 175 × 105 cells
2. 85 × 105 cells
3. 35 × 105 cells
4. 32 × 105 cells
At what stage of the cell cycle are histone proteins synthesized in a eukaryotic cell
1. During telophase
2. During S-phase
3. During G2 stage of prophase
4. During entire prophase
Four healthy people in their 20s got involved in injuries resulting in damage and death of a few cells of the following. Which of the cells are least likely to be replaced by new cells ?
| 1. | Osteocytes |
| 2. | Liver cells |
| 3. | Neurons |
| 4. | Malpighian layer of the skin |
During mitosis, endoplasmic reticulum and nucleolus begin to disappear at:
| 1. | Late metaphase | 2. | Early prophase |
| 3. | Late prophase | 4. | Early metaphase |
During which phase(s) of cell cycle, amount of DNA in a cell remains at 4C level if the initial amount is denoted as 2C ?
1. G0 and G1
2. G1 and S
3. Only G2
4. G2 and M
A somatic cell that has just completed the S phase of its cell cycle, as compared to gamete of the same species, has
1. Twice the number of chromosomes and twice the amount of DNA
2. Same number of chromosomes but twice the amount of DNA
3. Twice the number of chromosomes and four times the amount of DNA
4. Four times the number of chromosomes and twice the amount of DNA
Salting out action of the soap is based on:
1. Complex ion formation
2. Common ion effect
3. Solubility product
4. Acid-base neutralization
The most hydrolyzed salt among the following is-
(Assume that Kb of all weak bases is the same)
1. NH4Cl
2. CuSO4
3. AlCl3
4. All are equally hydrolyzed.
The equivalent weight of a metal is 4.5 and the molecular weight of its chloride is 80. The atomic weight of the metal is:-
1. 18
2. 9
3. 4.5
4. 36
For N2 + 3H2 2NH3, one mole of N2 and three moles of H2 are at pressure of 4 atm. Equilibrium pressure is found to be 3 atm. Hence, Kp is
1. 1/(0.5)x(0.15)3 2. 1/(0.5)x(1.5)3
3. 3x3/(0.5)x(1.5)3 4. none of the above
If the value of an equilibrium constant for a particular reaction is 1.6 x 1012, then at equilibrium the system will contain
1. 100% reactants
2. mostly reactants
3. mostly products
4. reactants and products in ratio of 1:1
Buffer solutions have constant acidity and alkalinity because
1. these give unionised acid or base or base on reaction with added acid or alkali
2. acids and alkalies in these solutions are shielded from attack by other ions
3. they have large excess of H+ or OH- ions
4. they have fixed value of pH
The enthalpy and entropy change for the reaction :
Br2 (l) + Cl2 (g) 2BrCl (g)
are 30 kJ mol-1 and 105 J K-1 mol-1 respectively.
The temperature at which the reaction will be in equilibrium is :
| 1. | 285.7 K | 2. | 273.4 K |
| 3. | 450.9 K | 4. | 300.1 K |
For the equilibrium of the reaction,\(\mathrm{HgO}(\mathrm{~s}) \rightleftharpoons \mathrm{Hg}(\mathrm{~g})+\frac{1}{2} \mathrm{O}_ 2(\mathrm{~g}), \mathrm{K_P}\) for the reaction at a total pressure of "P" will be:
| 1. | \(\mathrm{K}_P={2 \over 3^{3/2}}\mathrm P^{3/2}\) | 2. | \(\mathrm{K}_P={2 \over 3^{1/2}}\mathrm P^{1/2}\) |
| 3. | \(\mathrm{K}_P={1 \over 3^{2/3}}\mathrm P^{3/2}\) | 4. | \(\mathrm{K}_P={1 \over 3^{2/3}}\mathrm P\) |
Ionisation constant of HA (weak acid) and BOH (weak base) are 3.0 x 10–7 each at 298K. The percent degree of hydrolysis of BA at the dilution of 10L is :
1. 25
2. 50
3. 75
4. 40
When a gas undergoes adiabatic expansion, it gets cooled due to -
1. Loss of energy
2. Fall in pressure
3. Decrease in velocity
4. Increase in energy with work done
A Beckmann thermometer is used to measure -
1. High temperature
2. Low temperature
3. Normal temperature
4. All temperatures
The enthalpy of neutralization of HCN by NaOH is –12.13 kJ mol–1. The enthalpy of ionisation of HCN will be -
1. 45.07 kJ
2. 4.310 kJ
3. 451.9 kJ
4. 33.77 kJ
A brown ring complex compound is formulated as The oxidation state of iron is
1. 1
2. 2
3. 3
4. 0
The critical volume of a gas is . The radius of the molecule will be , in cm
1.
2.
3.
4.
Enthalpy change when 1.00 g water is frozen at is :
1. 0.0797 kcal
2. -0.0797 kcal
3. 1.435 kcal
4. -1.435 kcal
If of a reaction is 100 kJ mol-1,
then the activation energy for the forward reaction must be
1. Greater than 100 kJ mol-1
2. Less than 100 kJ mol-1
3. Equal to 100 kJ mol-1
4. None of the above.
A mixture of 2.3 g formic acid and 4.5 g oxalic acid is treated with conc. . The evolved gaseous mixture is passed through KOH pellets.
Weight (in g) of the remaining product at STP will be:
1. 1.4
2. 3.0
3. 2.8
4. 4.4
The characteristics associated with adsorption are:
1. ∆G, ∆H, and ∆S are all negative.
2. ∆G and ∆H are negative, but ∆S is positive.
3. ∆G and ∆S are negative, but ∆H is positive.
4. ∆G is negative, but ∆H and ∆S are positive.
At room temperature, MY and NY3, two nearly insoluble salts, have the same Ksp values of 6.2 × 10-13. The true statement regarding MY and NY3 is:
| 1. | The molar solubility of MY in water is less than that of NY3. |
| 2. | The salts MY and NY3 are more soluble in 0.5 M KY than in pure water. |
| 3. | The addition of the salt of KY to a solution of MY and NY3 will have no effect on their solubilities. |
| 4. | The molar solubilities of MY and NY3 in water are identical. |
When equal volumes of 0.1 M NaOH and 0.01 M HCl are mixed, the pH of the resulting solution will be:
| 1. | 12.65 | 2. | 2.0 |
| 3. | 7.0 | 4. | 1.04 |
The heat of combustion of carbon to CO2 is –393.5 KJ/mol. The heat released upon the formation of 35.2 g of CO2 from carbon and oxygen gas is:
| 1. | –315 KJ | 2. | +315 KJ |
| 3. | –630 KJ | 4. | +630 KJ |
The Ksp of Ag2CrO4, AgCl, AgBr, and Agl are respectively, 1.1 × 10–12, 1.8 × 10–10, 5.0 × 10–13, 8.3 × 10–17. Which one of the following salts will precipitate last if solution is added to the solution containing equal moles of NaCl, NaBr, Nal, and Na2CrO4?
1. Agl
2. AgCl
3. AgBr
4. Ag2CrO4
The correct statement for a reversible process in a state of equilibrium is:
1. G = – 2.30RT log K
2. G = 2.30RT log K
3. Go = – 2.30RT log K
4. Go = 2.30RT log K
(a) H2O2 + O3 → H2O + 2O2
(b) H2O2 + Ag2O → 2Ag + H2O + O2
The role of hydrogen peroxide in the above reactions is respectively:
| 1. | Oxidizing in (a) and reducing in (b) |
| 2. | Reducing in (a) and oxidizing in (b) |
| 3. | Reducing in (a) and (b) |
| 4. | Oxidizing in (a) and (b) |
The change in oxidation number of chlorine when Cl2 gas reacts with hot and concentrated sodium hydroxide solution is:
1. Zero to +1 and Zero to –5
2. Zero to –1 and Zero to +5
3. Zero to –1 and Zero to +3
4. Zero to +1 and Zero to –3
The highest oxidation number of nitrogen among the following compounds is:
| 1. | 2. | ||
| 3. | 4. |
The standard enthalpy of vaporization for water at 100 oC is 40.66 kJ mol–1. The internal energy of vaporization of water at 100 oC (in kJ mol–1) is:
(Assume water vapour behaves like an ideal gas.)
| 1. | +37.56 | 2. | –43.76 |
| 3. | +43.76 | 4. | +40.66 |
The bond energy of H—H and Cl-Cl is 430 kJ
mol-1 and 240 kJ mol-1 respectively and ΔHf for HCl is -90 kJ mol-1. The bond enthalpy of HCl is:
1. 290
2. 380
3. 425
4. 245
A certain hydrate has the formula . A quantity of 54.2 g of the compound is heated in an oven to drive off the water. If the water vapour generated exerts a pressure of 24.8 atm in a 2.0 L container at 120, the value of x is-
1. 2
2. 5
3. 6
4. 7
The correct order of temperature of a real gas is-
I. Boyle's temperature
II. Critical temperature
III. Inversion temperature
1. III > I > II
2. I > II > III
3. II > I > III
4. I > III > II
Which of the following is a redox reaction?
1. with NaOH
2. Formation of in the atmosphere
3. Evaporation of
4. Oxides of nitrogen forming nitrogen & oxygen from lightening
In the following redox reaction,
The value of x,y,z are :
1. x = 6, y = 1, z = 7
2. x = 3, y = 6, z = 8
3. x = 6, y = 2, z = 7
4. x = 4, y = 6, z = 7
| 1. | 7.01 | 2. | 2 |
| 3. | 12 | 4. | 9 |
Which of the following cannot act both as a Bronsted acid and as a Bronsted base?
| 1. | \(\mathrm{H C O_{3}^{-}}\) | 2. | \(\mathrm{NH_3}\) |
| 3. | \(\mathrm{HCl}\) | 4. | \(\mathrm{H S O_{4}^{-}}\) |
The molar solubility of in 0.1 M solution of NaF will be:
| 1. | 2. | ||
| 3. | 4. |
The oxidation state of Cr in is:
| 1. | –6 | 2. | +12 |
| 3. | +6 | 4. | +4 |
In water-saturated air, the mole fraction of water vapor is 0.02. If the total pressure of the saturated air is 1.2 atm, the partial pressure of dry air is-
1. 1.17 atm
2. 1.76 atm
3. 1.27 atm
4. 0.98 atm
The volume occupied by 1.8 g of water vapour at 374 and 1 bar pressure will be -
[Use R=0.083 bar ]
1. 96.66 L
2. 55.87 L
3. 3.10 L
4. 5.31 L
What is the amount of work done by an ideal gas, if the gas expands isothermally from \(10^{-3}~m^3\) to \(10^{-2}~m^3\) at \(300~K\)against a constant pressure of \(10^{5}~Nm^{-2}\)?
| 1. | \(+270 ~kJ\) | 2. | \(–900 ~J\) |
| 3. | \(+900 ~kJ\) | 4. | \(–900~ kJ\) |
Reversible expansion of an ideal gas under isothermal and adiabatic conditions are shown in the figure:
ABIsothermal expansion
ACAdiabatic expansion
Which of the following options is not correct?
| 1. | \(\Delta S_{\text {isothermal }}>\Delta S_{\text {adiabatic }} \) | 2. | \(T_A=T_B \) |
| 3. | \(W_{\text {isothermal }}>W_{\text {adiabatic }} \) | 4. | \(T_C>T_A\) |
Oxygen gas generated by the decomposition of potassium chlorate is collected. The volume of oxygen collected at 24C and an atmospheric pressure of 760 mm Hg is 128 ml. Calculate the mass (in grams) of oxygen gas obtained. The pressure of water vapour at 24C is 22.4 mm Hg
1. 1.36 g
2. 1.52 g
3. 0.163 g
4. 1.63 g
Concentration of the Ag+ ions in a saturated solution of Ag2C2O4 is 2.2 x 10-4 mol-1 solubility product of Ag2C2O4 is
1. 2.42 x 10-8
2. 2.66 x 10-12
3. 4.5 x 10-11
4. 5.3 x 10-12
Which of the following pairs constitutes a buffer?
1. HNO2 and NaNO2
2. NaOH and NaCl
3. HNO3 and NH4NO3
4. HCl and KCl
Which of the following thermodynamic quantities is an outcome of the second law of thermodynamics ?
1. Work
2. Enthalpy
3. Internal energy
4. Entropy
A solid sphere, a hollow sphere, and a disc, all having the same mass and radius, are placed at the top of a smooth incline and released. Least time will be taken in reaching the bottom by
| 1. | the solid sphere |
| 2. | the hollow sphere |
| 3. | the disc |
| 4. | all will take the same time |
A loaded truck has to take a sharp turn to the left. The center of gravity of the truck can be altered by shifting the concentrated load. To avoid toppling, the load must be shifted
1. up and left
2. up and right
3. down and left
4. down and right
What minimum friction coefficient is required for hollow sphere rolling without slipping on the inclined plane with an angle of inclination 37 ?
1.
2.
3.
4. None of these
A particle of mass m is moving at speed v perpendicular to a rod of length d and mass M = 6m which pivots around a frictionless axle running through its centre. It strikes and sticks to the end of the rod. The moment of inertia of the rod about its centre is . Then the angular speed of the system just after the collision is:

1. 2v/3d
2. 2v/d
3. v/d
4. v
A particle of mass 'm' is moving in a horizontal circle of radius 'r' under a centripetal force equal to –K/r2, where K is a constant. The total energy of the particle will be:
1.
2.
3.
4.
The graph between and is (E = kinetic energy and p = momentum)
1.
2.
3.
4.
The distance between the centres of moon and earth is D. The mass of earth is 81 times the mass of the moon. At what distance from the centre of the earth the gravitational force will be zero?
1.
2.
3.
4.
A geostationary satellite is revolving around the earth. To make it escape from gravitational field of earth, its velocity must be increased by
1. 100%
2. 41.4%
3. 50%
4. 59.6%
Two similar springs P and Q have spring constants KP and KQ, such that KP > KQ. They are stretched, first by the same amount (case a) and then by the same force (case b). The work done by external force, WP and WQ on the springs P and Q in case (a) and case (b) respectively are related as,
1. WP=WQ ; WP>WQ
2. WP=WQ ; WP=WQ
3. WP>WQ ; WQ>WP
4. WP<WQ ; WQ<WP
Three identical spherical shells, each of mass m and radius r are placed as shown in figure. Consider an axis XX’, which is touching to two shells and passing through diameter of third shell. Moment of inertia of the system consisting of these three spherical shells about XX' axis is
1.mr2
2. 3mr2
3. mr2
4. 4mr2
A solid cylinder of mass 3kg is rolling on a horizontal surface with velocity 4 It collides with a horizontal spring of force constant 200 . The maximum compression produced in the spring will be
1. 0.5 m
2. 0.6 m
3. 0.7 m
4. 0.2 m
magnitude \(P_0\). The instantaneous velocity of this car is proportional to:
1. \(t^2 P_0\)
2. \(t^{\frac{1}{2}}\)
3. \(t^{\frac{-1}{2}}\)
4. \(\frac{t}{\sqrt{m}}\)
Which one of the following plots represent the variation of gravitational field on a particle at distance r, due to a thin spherical shell of radius R? (r is measured from the centre of the spherical shell).
1.
2.
3. 
4.
Two particles which are initially at rest, move towards each other under the action of their mutual attraction.If their speeds are v and 2v at any instant, then the speed of centre of mass of the system will be
1. 2v 2. 0
3. 1.5v 4. v
A particle of mass M is situated at the centre of a spherical shell of mass M and radius a.The gravitational potential at a point situated at a/2 distance from the centre will be
1. 2.
3. 4.
A thin circular ring of mass M and radius r is rotating about its axis with constant angular velocity Two objects each of mass m are attached gently to the opposite ends of a diameter of the ring. The ring now rotates with angular velocity given by
1. 2.
3. 4.
The ratio of the radii of gyration of a circular disc to that of a circular ring, each of the same mass and radius, around their respective axes is:
1. \(\sqrt{3}:\sqrt{2}\)
2. \(1:\sqrt{2}\)
3. \(\sqrt{2}:\sqrt{1}\)
4. \(\sqrt{2}:\sqrt{3}\)
Water falls from a height of 60 m at the rate of 15 kg/s to operate a turbine. The losses due to frictional forces are 10% of energy. How much power is generated by the turbine?
(g = 10 m/s2)
1. 8.1 kW
2. 10.2 kW
3. 12.3 kW
4. 7.0 kW
For silver, Young's modulus is and Bulk modulus is . Its Poisson's ratio will be
1. -1
2. 0.5
3. 0.39
4. 0.25
One end of a uniform wire of length \(L\) and of weight \(W\) is attached rigidly to a point in the roof and a weight \(W_1\) is suspended from its lower end. If \(S\) is the area of cross-section of the wire, the stress in the wire at a height \(\frac{3L}{4}\) from its lower end is:
1. \(\frac{W_1}{S}\)
2. \(\frac{W_1+\left(\frac{W}{4}\right)}{S}\)
3. \(\frac{W_1+\left(\frac{3W}{4}\right)}{S}\)
4. \(\frac{W_1+W}{S}\)
The potential energy U between two molecules as a function of the distance X between them has been shown in the figure. The two molecules are -
1. Attracted when x lies between A and B and are repelled when X lies between B and C
2. Attracted when x lies between B and C and are repelled when X lies between A and B
3. Attracted when they reach B
4. Repelled when they reach B
When shearing force is applied to a body, then the elastic potential energy is stored in it. On removing the force, this energy:
1. converts into kinetic energy.
2. converts into heat energy.
3. remains as potential energy.
4. None of the above
When a force is applied on a wire of uniform cross-sectional area and length 4m, the increase in length is 1 mm. Energy stored in it will be
1. 6250 J
2. 0.177 J
3. 0.075 J
4. 0.150 J
Consider a drop of rainwater having a mass of \(1~\text{gm}\) falling from a height of \(1~\text{km}.\) It hits the ground with a speed of \(50~\text{m/s}.\) Take \(g\) as constant with a value \(10~\text{m/s}^2.\) The work done by the
(i) gravitational force and the (ii) resistive force of air is:
| 1. | \((\text{i})~1.25~\text{J};\) \((\text{ii})~-8.25~\text{J}\) |
| 2. | \((\text{i})~100~\text{J};\) \((\text{ii})~8.75~\text{J}\) |
| 3. | \((\text{i})~10~\text{J};\) \((\text{ii})~-8.75~\text{J}\) |
| 4. | \((\text{i})~-10~\text{J};\) \((\text{ii})~-8.75~\text{J}\) |
Two wires are made of the same material and have the same volume. The first wire has a cross-sectional area \(A\) and the second wire has a cross-sectional area \(3A\). If the length of the first wire is increased by \(\Delta l\) on applying a force \(F\), how much force is needed to stretch the second wire by the same amount?
| 1. | \(9F\) | 2. | \(6F\) |
| 3. | \(4F\) | 4. | \(F\) |
A moving block having mass \(m\) collides with another stationary block having a mass of \(4m.\) The lighter block comes to rest after the collision. When the initial velocity of the lighter block is \(v,\) then the value of the coefficient of restitution \((e)\) will be:
| 1. | \(0.5\) | 2. | \(0.25\) |
| 3. | \(0.8\) | 4. | \(0.4\) |
A body initially at rest and sliding along a frictionless track from a height \(h\) (as shown in the figure) just completes a vertical circle of diameter \(\mathrm{AB}= D.\) The height \({h}\) is equal to:

| 1. | \({3\over2}D\) | 2. | \(D\) |
| 3. | \({7\over4}D\) | 4. | \({5\over4}D\) |
Three objects, \(A:\) (a solid sphere), \(B:\) (a thin circular disk) and \(C:\) (a circular ring), each have the same mass \({M}\) and radius \({R}.\) They all spin with the same angular speed about their own symmetry axes. The amount of work \(({W})\)required to bring them to rest, would satisfy the relation:
| 1. | \({W_C}>{W_B}>{W_A} ~~~~~~~~\) |
| 2. | \({W_A}>{W_B}>{W_C}\) |
| 3. | \({W_B}>{W_A}>{W_C}\) |
| 4. | \({W_A}>{W_C}>{W_B}\) |
The bulk modulus of a spherical object is \(B.\) If it is subjected to uniform pressure \(P,\) the fractional decrease in radius is:
| 1. | \(\frac{B}{3P}\) | 2. | \(\frac{3P}{B}\) |
| 3. | \(\frac{P}{3B}\) | 4. | \(\frac{P}{B}\) |
| 1. | \(0.25 ~\text{rad/s}^2 \) | 2. | \(25 ~\text{rad/s}^2 \) |
| 3. | \(5 ~\text{m/s}^2 \) | 4. | \(25 ~\text{m/s}^2 \) |
Two discs of the same moment of inertia are rotating about their regular axis passing through centre and perpendicular to the plane of the disc with angular velocities \(\omega_1\) and \(\omega_2\). They are brought into contact face to face with their axis of rotation coinciding. The expression for loss of energy during this process is:
1. \(\frac{1}{4}I\left(\omega_1-\omega_2\right)^2\)
2. \(I\left(\omega_1-\omega_2\right)^2\)
3. \(\frac{1}{8}I\left(\omega_1-\omega_2\right)^2\)
4. \(\frac{1}{2}I\left(\omega_1-\omega_2\right)^2\)
Which of the following statements are correct?
| (a) | centre of mass of a body always coincides with the centre of gravity of the body . |
| (b) | centre of gravity of a body is the point about which the total gravitational torque on the body is zero. |
| (c) | a couple on a body produce both translational and rotation motion in a body. |
| (d) | mechanical advantage greater than one means that small effort can be used to lift a large load. |
| 1. | (a) and (b) | 2. | (b) and (c) |
| 3. | (c) and (d) | 4. | (b) and (d) |
The moment of the force, \(\overset{\rightarrow}{F} = 4 \hat{i} + 5 \hat{j} - 6 \hat{k}\) at point (\(2,\) \(0,\) \(-3\)) about the point (\(2,\) \(-2,\) \(-2\)) is given by:
| 1. | \(- 8 \hat{i} - 4 \hat{j} - 7 \hat{k}\) | 2. | \(- 4 \hat{i} - \hat{j} - 8 \hat{k}\) |
| 3. | \(- 7 \hat{i} - 8 \hat{j} - 4 \hat{k}\) | 4. | \(- 7 \hat{i} - 4 \hat{j} - 8 \hat{k}\) |
A solid sphere is in rolling motion. In rolling motion, a body possesses translational kinetic energy (Kt) as well as rotational kinetic energy (Kr) simultaneously. The ratio Kt : (Kt + Kr) for the sphere will be:
1. 7:10
2. 5:7
3. 10:7
4. 2:5
A solid sphere is rotating freely about its axis of symmetry in free space. The radius of the sphere is increased keeping its mass the same. Which of the following physical quantities would remain constant for the sphere?
| 1. | angular velocity |
| 2. | moment of inertia |
| 3. | rotational kinetic energy |
| 4. | angular momentum |
| 1. | \(wx \over d\) | 2. | \(wd \over x\) |
| 3. | \(w(d-x) \over x\) | 4. | \(w(d-x) \over d\) |
A mass falls from a height \(h\) and its time of fall \(t\) is recorded in terms of time period \(T\) of a simple pendulum. On the surface of the earth, it is found that \(t=2T\). The entire setup is taken on the surface of another planet whose mass is half of that of the Earth and whose radius is the same. The same experiment is repeated and corresponding times are noted as \(t'\) and \(T'\). Then we can say:
| 1. | \(t' = \sqrt{2}T\) | 2. | \(t'>2T'\) |
| 3. | \(t'<2T'\) | 4. | \(t' = 2T'\) |
An object flying in the air with velocity \((20 \hat{i}+25 \hat{j}-12 \hat{k})\) suddenly breaks into two pieces whose masses are in the ratio of \(1:5.\) The smaller mass flies off with a velocity \((100 \hat{i}+35 \hat{j}+8 \hat{k})\). The velocity of the larger piece will be:
1. \( 4 \hat{i}+23 \hat{j}-16 \hat{k}\)
2. \( -100 \hat{i}-35 \hat{j}-8 \hat{k} \)
3. \( 20 \hat{i}+15 \hat{j}-80 \hat{k} \)
4. \( -20 \hat{i}-15 \hat{j}-80 \hat{k}\)
The stress-strain curves are drawn for two different materials \(X\) and \(Y.\) It is observed that the ultimate strength point and the fracture point are close to each other for material \(X\) but are far apart for material \(Y.\) We can say that the materials \(X\) and \(Y\) are likely to be (respectively):
| 1. | ductile and brittle |
| 2. | brittle and ductile |
| 3. | brittle and plastic |
| 4. | plastic and ductile |
An object of mass \(500~\text g\) initially at rest is acted upon by a variable force whose \(x\)-component varies with \(x\) in the manner shown. The velocities of the object at the points \(x=8~\text m\) and \(x=12~\text m\) would have the respective values of nearly:

| 1. | \(18~\text {m/s}\) and \(22.4~\text {m/s}\) | 2. | \(23~\text {m/s}\) and \(22.4~\text {m/s}\) |
| 3. | \(23~\text {m/s}\) and \(20.6~\text {m/s}\) | 4. | \(18~\text {m/s}\) and \(20.6~\text {m/s}\) |
A solid cylinder of mass 2 kg and radius 50 cm rolls up an inclined plane of angle of inclination . The centre of mass of the cylinder has a speed of 4 m/s. The distance travelled by the cylinder on the inclined surface will be
| 1. | 2.2 m | 2. | 1.6 m |
| 3. | 1.2 m | 4. | 2.4 m |
| 1. | \(6\sqrt{2}~\text{hr}\) | 2. | \(12\sqrt{2}~\text{hr}\) |
| 3. | \(\frac{24}{2.5}~\text{hr}\) | 4. | \(\frac{12}{2.5}~\text{hr}\) |
Assuming that the gravitational potential energy of an object at infinity is zero, the change in potential energy (final - initial) of an object of mass \(m\) when taken to a height \(h\) from the surface of the earth (of radius \(R\) and mass \(M\)), is given by:
| 1. | \(-\frac{GMm}{R+h}\) | 2. | \(\frac{GMmh}{R(R+h)}\) |
| 3. | \(mgh\) | 4. | \(\frac{GMm}{R+h}\) |