Which of the following taxonomic categories contains organisms least similar to one another?
1. Class
2. Genus
3. Family
4. Species
The six most common atoms in organic molecules are:
| 1. | Carbon, Hydrogen, Oxygen, Helium, Calcium and Sulphur |
| 2. | Carbon, Hydrogen, Oxygen, Calcium, Magnesium and Sulphur |
| 3. | Carbon, Oxygen, Nitrogen, Sulphur, Phosphorous and Magnesium |
| 4. | Carbon, Hydrogen, Oxygen, Nitrogen, Phosphorous and Sulphur |
The unique properties of each amino acid are determined by its particular:
1. R group
2. Amino group
3. Kinds of peptide bonds
4. Number of bonds to other amino acids
Based on the graph of an enzyme-catalyzed reaction, which of the following could be used to increase the reaction rate beyond point C?

1. increase the amount of substrate
2. add more water
3. increase the temperature
4. decrease enzyme concentration
In feedback inhibition, a metabolic pathway is switched off by :
1. A rise in the temperature
2. Accumulation of the end product
3. Lack of a substrate
4. Competitive inhibition
Proteins that are to be used outside the cell are synthesized:
| 1. | in the mitochondria |
| 2. | on the rough endoplasmic reticulum |
| 3. | on the smooth endoplasmic reticulum |
| 4. | on free ribosomes |
The two types of cellular organelles that transform energy are:
| 1. | Chromoplasts and Leucoplasts |
| 2. | Mitochondria and Chloroplasts |
| 3. | Mitochondria and Endoplasmic reticulum |
| 4. | Chloroplasts and Golgi apparatus |
The arrangement of outer and central microtubules in a cilium is called the:
1. 9 + 1 pattern
2. 9 + 0 pattern
3. Flagellin pattern
4. 9 + 2 pattern
Pressure on a plant cell wall caused osmotic movement of water is called:
1. Osmotic pressure
2. Osmotic potential
3. Turgor pressure
4. Hypertonic pressure
Which of the following processes makes direct use of oxygen?
1. Glycolysis
2. Fermentation
3. Kreb's citric acid cycle
4. Electron transport
The main control over ATP production is allosteric inhibition, by ATP, of the enzyme that facilitates the formation of :
| 1. | Glucose from glycogen |
| 2. | Fructose 1,6 biphosphate from Fructose 6 phosphate |
| 3. | Acetyl coenzyme A from pyruvic acid |
| 4. | Citric acid from Acetyl coenzyme A and oxaloacetic acid. |
When a cell stops growing, say due to shortage of nutrients, this will occur in which phase of the cell cycle?
1.
2.
3. S
4.
Genetic information in a DNA molecule is coded in the:
1. Sequence of nucleotides
2. Base pairings
3. Proportion of each base present
4. The turning pattern of the helix
In DNA replication, the Okazaki fragments on the lagging strand are joined together by:
1. DNA ligase
2. DNA polymerase
3. Primase
4. Helicase
The metabolic activity of the different types of cells within an organism varies because of differences in the kinds of :
1. Genes in each cell
2. Ribosomes in each cell
3. Enzymes in each cell
4. Nucleoli in each cell
The enzyme that catalyzes the peptide bonding in prokaryotes is located in the:
1. Leader region of the mRNA
2. Central part of tRNA
3. Smaller subunit of the ribosome
4. Larger subunit of the ribosome
If a cell were treated with a chemical that blocked nucleic acid synthesis, which of the following processes would most likely be affected first?
1. DNA replication
2. tRNA synthesis
3. mRNA synthesis
4. Protein synthesis
In general, bacterial genes are regulated at the time of:
1. Transcription
2. Post transcription
3. Translation
4. Post translation
Restriction enzymes are synthesized by:
1. Bacteria only
2. Yeast and bacteria only
3. Eukaryotic cells only
4. All kinds of cells
The first human hormone produced by recombinant DNA technology was :
1. Estrogen
2. Growth hormone
3. Erythropoietin
4. Insulin
At what phase of meiosis are homologous chromosomes separated ?
1. Prophase l
2. Prophase ll
3. Anaphase l
4. Anaphase ll
One of the Mendel's pure strains of pea plants had green peas. How many different types of eggs could such a plant produce with regard to pea color?
1. 1
2. 2
3. 4
4. 8
A woman receives her X chromosomes from:
1. Her mother only
2. Her father only
3. Both her mother and father
4. Mitochondria of mother only
Most fossils are found in:
1. Granite
2. Sedimentary rocks
3. Lava flows
4. Black soil
Darwin believed that certain parts of the body get larger and more complex through the generations because they :
1. are used more extensively than other parts
2. contribute to greater reproductive success
3. are pre-determined to do so
4. are most similar to God's perfection
The wings of a bird and that of an insect are:
1. Analogous structures
2. Homologous structure
3. Vestigial structures
4. Useless structures
Genetic drift occurs when a few individuals of a species colonize an island. The particular phenomenon is known as
| 1. | The bottleneck effect |
| 2. | The founder effect |
| 3. | Assortative mating |
| 4. | Random mating |
Directional selection favours:
| 1. | Both extremes of a trait |
| 2. | Intermediate form of a trait |
| 3. | Environmental differences |
| 4. | One extreme form over the other extreme form and over intermediate forms of the trait |
Most often the factor that initiates the speciation of two populations is:
| 1. | Geographic isolation |
| 2. | Inability of the gametes to fuse |
| 3. | Different courtship behaviours |
| 4. | Different copulatory organs |
Cells in some filamentous cyanobacteria that are specialized for nitrogen fixation are called:
1. Phycobilisomes
2. Chromatophores
3. Grana
4. Heterocysts
The protozoans that cause malaria in humans are :
1. Radiolarians
2. Dinoflagellates
3. Chrysophytes
4. Sporozoans
Red algae differ from the green algae and brown algae in having :
| 1. | No chlorophyll |
| 2. | No differentiated cells |
| 3. | Lacking sexual stages |
| 4. | No flagellated stages in their life cycle |
A moss sperm moves by means of :
1. Pseudopodia
2. Cilia
3. A flagellum
4. Two flagella
Which of the following is not an arthropod characteristic?
1. Jointed appendages
2. Non-segmented bodies
3. Periodic moulting
4. Articulated exoskeleton
Amniotes include all except:
(1) Amphibians
(2) Reptiles
(3) Birds
(4) Mammals
The oldest fossils resemble:
1. Amoeba
2. Red algae
3. Heterotrophic bacteria
4. Autotrophic bacteria
The first group of terrestrial plants to get rid of the swimming sperm was the:
| 1. | Club mosses | 2. | Ferns |
| 3. | Gymnosperms | 4. | Angiosperms |
Reptilian ancestors of the mammals were the :-
1. Therapsids
2. Thecodonts
3. Pterosaurs
4. Plesiosaurs
Lignin is a component of the secondary walls of:-
1. Epidermis
2. Collenchyma
3. Sclerenchyma
4. Parenchyma
The pericycle, which gives rise to lateral roots, consists of:
| 1. | Epithelial cells | 2. | Meristematic cells |
| 3. | Parenchymal cells | 4. | Endodermal cells |
The most abundant solute in the phloem sap is :
1. Potassium ions
2. Sucrose
3. Glucose
4. Starch
A plant requires potassium for:
(1) Holding its cells together
(2) Synthesizing chlorophyll
(3) DNA replication
(4) Opening and closing its stomata
The shoot apical meristem in a dicot embryo is located:
(1) Between the two cotyledons
(2) Within the root apex
(3) Around the suspensor
(4) In the endosperm
The asexual production of seeds is called as:
1. Adventition
2. Fragmentation
3. Self-fertilization
4. Apomixis
Primary and secondary growth are inhibited by:
1. Auxins
2. Gibberellins
3. Abscisic acid
4. Cytokinins
Which type of animal tissue changes the diameter of a blood vessel?
1. Epithelium
2. Connective
3. Muscle
4. Neural
Organisms with very high intrinsic growth rates have:
1. Long generation times
2. Short generation times
3. No courtship behaviour
4. No carrying capacities
Intraspecific competition is strongest when the:
1. Species overlap in their distribution
2. Populations overlap in their ranges
3. Population is at its carrying capacity
4. Reproductive rate is at its maximum
No predator can become proficient at acquiring prey because:
| 1. | Predators are not as intelligent as prey. |
| 2. | Predators are too large to be fast enough. |
| 3. | Prey populations evolve more rapidly than predator populations. |
| 4. | Prey populations evolve anti-predatory traits. |
Which of the following organisms will be the first to colonize a bare rock?
1. Annual plants
2. Lichens
3. Perennail plants
4. Herbs and shrubs
Most food chains are composed of
1. 1 or 2 species
2. 3 or 4 species
3. 9 or 10 species
4. More than 16 species
The phosphorus cycle is unusual in that it is entirely:
1. Within aquatic ecosystems
2. Within terrestrial ecosystems
3. Sedimentary
4. Gaseous
Most primary productivity in the surface water of lakes and oceans is by:
1. Floating plants
2. Red and brown algae
3. Zooplankton
4. Phytoplankton
The most effective means of conservation of biodiversity is:
1. Remove predators
2. Preserve habitats
3. Vaccinate against disease
4. Census the species during breeding season
What is eutrophication?
1. The nutrient enrichment of lakes and ponds
2. The decrease in the pH of a lake or pond
3. The increase of water temperature of a lake or pond
4. The depletion of nutrients inlakes and ponds
In what way does thermal pollution affect organisms?
1. Lowers the pH of water
2. Interrupts normal physiological pathways
3. Decreases oxygen supply in water
4. Increases oxygen supply in water
Describe urine in relationship to glomerular filtration tubular reabsorption and tubular secretion urine:
| 1. | glomerular filtration + tubular reabsorption + tubular secretion |
| 2. | glomerular filtration - tubular reabsorption -tubular secretion |
| 3. | glomerular filtration + tubular reabsorption -tubular secretion |
| 4. | glomerular filtration -tubular reabsorption +tubular secretion |
Atrial natriuretic hormone (ANH) will promote a(n) ________ in the excretion of sodium and water; thus ________ blood volume and blood pressure.
1. decrease, decreasing
2. decrease; increasing
3. increase, decreasing
4. increase, increasing
Which of the following gastric secretory cells is correctly matched with its secretion(s)?
1. Chief cells - gastrin
2. parietal cells -- hydrochloric acid and intrinsic factor
3. enteroendocrine cells -- mucus and hormones
4. mucous cells -- pepsinogen and mucus
What structure closes off the nasopharynx during swallowing ?
1. tongue
2. epiglottis
3. uvula
4. hard palate
Fibrous connective tissue builds up in the lungs causing them not to inflate properly, and actually deflating them is called:
1. chronic bronchitis
2. emphysema
3. pulmonary fibrosis
4. asthma
The C-shaped cartilaginous rings around the trachea are open posteriorly to:
(1) allow for expansion of the esophagus during swallowing
(2) allow the vocal cords to relax
(3) prevent food from entering the nasal cavity during swallowing
(4) prevent food from entering the trachea
The trachea is lined with
1. ciliated simple squamous epithelium
2. stratified squamous epithelium
3. pseudostratified ciliated columnar epithelium
4. pseudostratified squamous epithelium
Primary lymphatic organs serve as the site for:
| 1. | antibody formation |
| 2. | lymph formation |
| 3. | lymphocytes to encounter and bind to antigens |
| 4. | lymphocyte formation and maturation |
Explain the reason there is a delay in processing the electrical signal from the AV node to the AV bundle. To:
1. allow the atria to complete their contraction prior to ventricular contraction
2. ensure the right and left atria contract at the same time
3. ensure the right and left ventricles contract at the same time
4 prevent an ectopic pacemaker
The vasomotor center that controls the diameter of blood vessels is located in the
1. medulla oblongata
2. pons
3. cerebrum
4. cerebellum
Which of the following is a hinge point?
1. At the base of the first metacarpal
2. Intervertebral disc
3. Interphalageal
4. Between carpal bone
The neurotransmitter released by the nerve cell at the neuromuscular junction is:
1. Acetylcholine
2. Adrenaline
3. Serotonin
4. Dopamine
AIDS is characterized by the presence of:
| 1. | Nosocomial infections |
| 2. | Iatrogenic infections |
| 3. | Opportunistic infections |
| 4. | Subclinical infections |
Hyperthyroidism [Graves' disease] is characterized by all the following except:
1. Toxic goiter
2. Exopthalmos
3. Bradycardia
4. Muscle weakness and termors
Match the columns A and B:
| Column I | Column II | ||
| (a) | Marginal placentation | (i) | Marigold |
| (b) | Axile placentation | (ii) | Dianthus |
| (c) | Parietal placentation | (iii) | Argemone |
| (d) | Free central placentation | (iv) | Chinarose |
| (e) | Basal placentation | (v) | Pea |
| (a) | (b) | (c) | (d) | (e) | |
| 1. | (v) | (iv) | (iii) | (ii) | (i) |
| 2. | (v) | (iii) | (iv) | (i) | (ii) |
| 3. | (iv) | (iii) | (v) | (ii) | (i) |
| 4. | (i) | (v) | (iv) | (iii) | (ii) |
If the goal were to create a plant resistant to an insecticide, which cell-based plant technology would be most effective ?
| 1. | clonal propagation |
| 2. | cybridization |
| 3. | protoplast fusion |
| 4. | mutant selection |
Which protein has been produced generating a transgenic sheep that is used for replacement therapy for individuals at risk from emphysema?
| 1. | Plasminogen activator (rPA) |
| 2. | α-antitrypsin (ATT) |
| 3. | Casein |
| 4. | Amyloid precursor proteins |
Gases found in biogas are:
1. Carbon dioxide is the only gas
2. Methane is the only gas
3. Carbon dioxide, hydrogen sulphide and Methane
4. Carbon dioxide, hydrogen cyanide and Methane
ACTH controls the secretion of:
1. cortisol
2. aldosterone
3. epinephrine
4. testosterone
Which of the following is not likely to happen as a result of the rising levels of in our atmosphere?
1. Increased incidents of skin cancer
2. Rise in sea levels
3. Change in climatic patterns
4. Shifts in locations of deserts and fertile regions
Phellogen in dicot roots originates from :
1. pericycle
2. endodermis
3. cortex
4. exodermis
Summer wood is:
(1) the same as heartwood.
(2) found to the outside of each annular ring of xylem.
(3) found to the inside of each annular ring of xylem.
(4) formed throughout the growing season.
An inferior ovary:
| 1. | is situated on the receptacle above the perianth and androecium. |
| 2. | is positioned below the sites of attachment for perianth and androecium. |
| 3. | forms a hypanthium. |
| 4. | is positioned below the receptacle. |
Aril is the edible part in :
1. Mango
2. Apple
3. Litchi
4. Banana
The equivalent weight of an element is 15. It forms an acidic oxide with which potassium hydroxide forms a salt isomorphous with . The atomic weight of the element would be
1. 90
2. 13.16
3. 26.52
4. 52.64
A compound of vanadium has a magnetic moment of 1.73 BM. The electronic configuration o vanadium ion in the compound is
1.
2.
3.
4.
Which set of quantum theory is not consistent with quantum mechanical theory?
1. n = 2, l = 1, m = 1, s = +
2. n = 4, l = 3, m = 2, s = -
3. n = 3, l = 2, m = 3, s = +
4. n = 4, l = 3, m = 3, s = +
The correct order of increasing s-character of the orbital of B which overlaps with the orbital of F to form B-F bond in is
1.
2.
3.
4.
Which among the following species has the highest ionization energy?
1. Ne
2. F
3. Li
4. B
Gas is an open container that is heated from 27C to 127C. The fraction of the original amount of gas remaining in the container will be
1.
2.
3.
4.
If the entropy of vaporization of a liquid 100 and its enthalpy of vaporization is 50KJ/mol. The boiling point of the liquid is
1. 354.5 K
2. 454.5 K
3. 554.5 K
4. 445.5 K
How many unit cells are present in 1 g of Cu if it crystallizes in fcc?
1.
2.
3.
4.
If 'a' is the initial concentration and K is the rate constant of a zero-order reaction, the time for the reaction to go to completion will be
1.
2.
3.
4.
If for water are 1.86 and 0.52 K.kg.mol−1 respectively. The difference between boiling and freezing point of the aqueous solution be 102.38 then the solution is
1. 50 g of 1 molal KCI solution
2. 50 g of 2 molal urea solution
3. 1000 g of 1 molal glucose solution
4. All of these
Which of the following species cannot function as an electrophile?
1. HCl
2.
3.
4. CH2=CH2
An optically acti\.e carboxylic acid (X) the molecular formula and can be shown by Chemical and physical tests to contain the group — and at least one alkyl group. When (X) is reduced with hydrogen over platinum, the product formed is optically inactive.
1.
2.
3.
4.
Which compound will react readily in a ?
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Williamson's synthesis of ethers is an example of
1. Nucleophilic substitution reaction
2. Nucleophilic addition
3. Electrophilic addition
4. Electrophilic substitution
What will be the product (X) in the following reaction?
1.
2.
3. 
4. 
Consider the following reaction,
The compounds (B) and (C) are
1.
2.
3.
4.
Which compound is formed when iron reacts with carbon?
1.
2.
3.
4.
Which process of purification is represented by the following equation?
1. Cupellation
2. Poling
3. Van Arkel
4. Zone refining
What is the nature of hydrogen sulfide in the following reaction?
1. Reducing agent
2. Oxidizing agent
3. Acidic nature
4. Basic nature
The number of P - O - P bonds in cyclic metaphosphoric acid is
1. Zero
2. 2
3. 3
4. 4
Which of the following interhalogen compound is not possible?
1.
2.
3.
4.
Calculate moles of ICI and ICI3 formed respectively if 25.4 g of and 14.2 g of are made to react completely to yield a mixture of ICI and ICI3:
[Given: Molecular mass of I2 -254, Cl2 - 71]
1. 0.5 and 0.2
2. 0.1 and 0.1
3. 0.1 and 0.3
4. 0.3 and 0.4
What are the products formed when ammonia reacts with excess chlorine?
1.
2.
3.
4.
Consider the following reaction:
The rate of this reaction in terms of at T (K) is = 0.02 mol. . What is the value of (in units of mol. ) at the same temperature?
1. 0.02
2. 50
3. 0.06
4. 0.04
Which one of the following statements is not correct?
1. Physical adsorption decreases with increase in the temperature
2. Physical adsorption is multilayered
3. The activation energy of physical adsorption is very high
4. Enthalpy change of physical adsorption is about 20 kJ.
Which one of the following statements is correct?
1. Bronsted-Lowry concept could not explain the acidic nature of
2. The pH of 0.01M NaOH solution is 2
3. The ionic product of at 25C is
4. The pH of a solution can be calculated using the equation pH = log .
The products formed when heavy water is reacted when magnesium nitride, are ...
1.
2.
3.
4.
Select S-isomer
| 1. |  |
2. |  |
| 3. |  |
 |
Which of the following exhibits a pπ-dπ bond?
| 1. | \(\mathrm{NO}_3^{-}\) | 2. | \( \mathrm{BO}_3^{3-} \) |
| 3. | \(\mathrm{CO}_3^{2-} \) | 4. | \(\mathrm{SO}_3^{2-} \) |
The wavenumber of the first line in the Balmer series of hydrogen is 15200 . The wavenumber of the first line in the Balmer series of is
1. 15200
2. 69800
3. 76000
4. 136,800
On adding solution into KI solution, a negatively charged colloidal sol. is obtained When they are in
1. 100 ml of 0.1 + 100 ml of 0.1 MKl
2. 100 ml of 0.1 + 50 ml of 0.2 MKl
3. 100 ml of 0.2 + 100 ml of 0.1 MKl
4. 100 ml of 0.1 + 100 ml of 0.15 MKl
In the given reaction, the compound undergoes hydration with \(\text{HgSO}_4/\text{H}_2\text{SO}_4\). What is the major product A?

| 1. |  |
2. |  |
| 3. |  |
4. |  |
Carbon monoxide reacts with sodium hydroxide at 200C under pressure to give
1. Sodium formate
2. Sodium oxalate
3. formic acid
4. Oxalic acid
A gas with formula diffuses through the porous plug at a rate of one-sixth of the rate of diffusion of hydrogen gas under similar conditions. The formula of gas is
1.
2.
3.
4.
A monobasic acid is obtained in
1.
2.
3.
4. None of these
The most powerful oxidant among the following is
1. HOCl
2.
3.
4.
What is A in the following reaction?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
A compound is formed by elements A and B and is cubic. A atoms is at the comers and B atoms are at the face center. The formula of the compound is
1. AB
2.
3.
4.
One mole of a gas is heated at constant pressure to raise its temperature by 1c. The work done in joules is:
1. -4.3
2. -8.314
3. -16.62
4. unpredictable
Borax is used as a buffer since
1. its aqueous solution contains an equal amount of weak acid and
2. its salt it is easily available
3. its aqueous solution contains an equal amount of strong acid and its salt
4. its aqueous solution contains salt only
The hybridised state of Cu in is :
1.
2.
3.
4.
An acid solution of pH 6 is diluted hundred times. The pH of the solution becomes
1. 6.85
2. 6
3. 4
4. 8
The heat of hydration is maximum for
1.
2.
3.
4.
Among unpaired electron is present in
1.
2.
3.
4.
The vapour density of undecomposed is 46. When heated, vapour density decreases to 24.5 due to its dissociation to . The % dissociation of is :
1. 40
2. 57
3. 67
4. 87
A physical quantity x depends on quantities y and z as follows: x = Ay + Btan Cz, where A, B, and C are constants. Which of the following does not have the same dimension?
1. x and B
2. C and
3. y and B/A
4. x and A
A physical quantity determined by measuring a, b, c, d, and e separately with the percentage error of 2%, 2%, 2%, 2%, and 2% respectively. The minimum amount of error is contributed to the measurement of
1. b
2. a
3. d
4. e
Which of the following cannot be speed-time graph?
| (A) |  |
| (B) |  |
| (C) |  |
| (D) |  |
| (E) |  |
(A)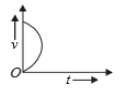
(B) 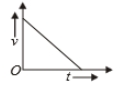
(C)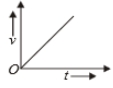
(D) 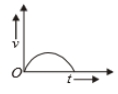
(E) 
1. (B) and (D)
2. (C) and (E)
3. (D) only
4. (A) only
Following diagram performs logic function of

1. AND gate
2. NAND gate
3. XOR gate
4. OR gate
A plane is flying horizontally at 98 m/s and an object which reaches the releases ground in 10 sec. The angle its velocity makes with horizontal while hitting the ground is (g = 9.8 m/)
1. 55
2. 45
3. 60
4. 75
A point of mass m is suspended from a light thread of length l, fixed at O,
is whirled in a horizontal circle at constant speed as shown. From a
frame stationary with respect to the mass, the forces acting on the mass are-
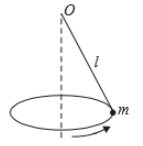

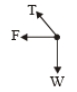
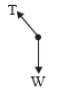
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
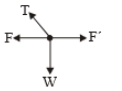
1. 
2. 
3. 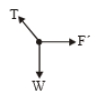
4. 
A balloon of mass M is descending at a constant acceleration . When a mass m is released from the balloon it starts rising with the same acceleration . Assuming that its volume does not change, what is the value of m?
1.
2.
3.
4.
The figure shows the position-time (x-t) graph of one-dimensional motion of a body of mass 0.4 kg. The magnitude of each impulse is-
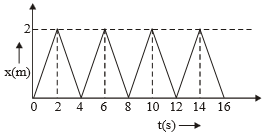

1. 0.4 N-s
2. 0.8 N-s
3. 1.6 N-s
4. 0.2 N-s
The centre of mass of a body
1. depends on the choice of co-ordinate system
2. is independent of the choice of co-ordiante system
3. may or may not depend on the choice of co-ordinate system
4. none of these
If vector be a force acting on a particle having the position vector and be the torque of this force about the origin, then
1.
2.
3.
4.
The moment of inertia of a uniform rod about a perpendicular axis passing through one end is . The same rod is bent into a ring and its moment of inertia about a diameter is . Then is
1.
2.
3.
4.
A planet revolves in an elliptical orbit around the sun. The linear speed of the planet will be maximum at :

1. A
2. B
3. C
4. D
If a spring of spring constant k is stretched by a length x under tension T, the energy stored is
1.
2.
3.
4.
The pressure at the bottom of a tank of water is 3P, where P is atmospheric pressure. If the water is drawn out until the level of water is lowered by one fifth, then the pressure at the bottom of the tank is :
1. 2P
2. 13P/5
3. 8P/5
4. 4P/5
During severe winter in the low-temperature zones of the world, the superficial parts of the lakes are frozen, leaving water below. The freezing at the bottom is prevented mainly because of
1. The conductivity of ice is low
2. The water has a large specific heat
3. The water has a large latent heat of fusion
4. The temperature of the earth at the bottom of the lake is high
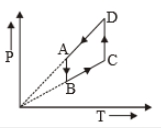
The P-V diagram of a certain process (Carnot cycle) is shown in the figure. The process is represented as: 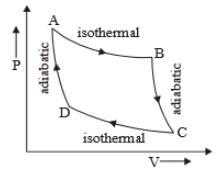

1. 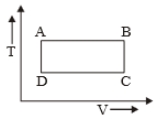
2. 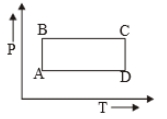
3. 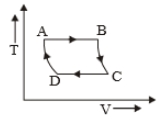
4. 
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The molar heat capacity at constant volume of oxygen gas at STP is nearly and it approaches as the temperature is increased. This because at the higher temperature
1. oxygen becomes triatomic
2. oxygen does not behave as an ideal gas
3. oxygen molecules rotate more vigorously
4. oxygen atoms start vibrating
Two identical springs are connected to mass m as shown (k = spring constant). If the time period of the configuration in (i) is 2s, the time period of the configuration (ii) is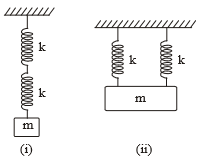

1.
2. 1s
3.
4.
The displacement of a wave disturbance propagating in the positive X-direction is given by : at t =0 and at t= 2 sec, where x and y are in m. If the shape of the wave disturbance does not change during the propaation , what is the velocity of the wave ?
1. 1 m/sec
2. 1.5 m/sec
3. 0.5 m/sec
4. 2 m/sec
An organ pipe open at one end is vibrating in first overtone and is in resonance with another pipe open at both ends and vibrating in third harmonic. The ratio of the length of two pipes is
1. 1:2
2. 4:1
3. 8:3
4. 3:1
In moving from A to B along an electric field line, the electric tied dos 6.4 x J of work on an electron. If are equipotential surface, then the potential difference is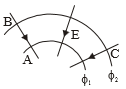

1. -4 V
2. 4 V
3. 0
4. 64 V
The electric potential at a point (x, y) in the x y-plane is given by, V = - Kxy. The
electric field intensity at a distance r from the origin varies as
1.
2. r
3. 2r
4. The electric potential at a ponit (x, y) in
the x y -plane is given by, V = —Kv . The
electric field intensity at a distance r from
the origin varies as
In the circuit shown in the figure, the potential difference across the capacitor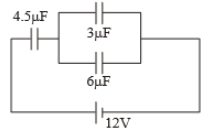

1. 8 volts
2. 6 volts
3. 4 volts
4. volts
A uniform wire of resistance 20 ohms having resistance 1 /m is bent in the adjoining form of a circle as shown in the figure. If the equivalent resistance between M and N is 1.8 ohm, then the length of the shorter section is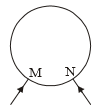

1. 2m
2. 5m
3. 1.8 m
4. 18 m
The current flowing in the given circuit is 0. IA. The potential difference between the point Y and X is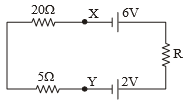

1. 4.0 V
2. 3.0 V
3. 2.5 V
4. 2.0 V
Two electric bulbs rated watt-V volts and watt-V volts are connected in parallel and V volt are applied to it. The total power will be
1.
2.
3.
4.
A wire carrying current 1is shaped as shown in the figure. The section AB is a quarter circle of radius r. The magnetic field at C is directed
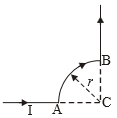

1. at an angle /4 to the plane of the paper
2. along the bisector of the angle ACB towards AB
3. along the bisector of the angle, ACB away from AB
4. perpendicular to the plane of the paper and directed into the paper
Identify the correct statement from the following
1. Cyclotron frequency is dependent on speed of the charged particle
2. Kinetic energy of chared particle in cyclotron does not depend on its mass
3. Cyclotron frequency does not depend on speed of charged particle
4. Kinetic energy of charged particle in cyclotron is independent of its charge
The half-life of a radioactive substance is 20 minutes. The approximate time interval () between the time when of it has decayed and time when of it had decayed is
1. 7 min
2. 14 min
3. 20 min
4 28 min
A bridge rectifier is constructed shown in figure. Alternating input is given across A and C. If output is taken across BD, then it is

1. zero
2. same as input
3. half wave rectified
4. full wave rectified
The current gain of the common-base n-p-n transistor is 0.96. What is the current gain if it is used as a common-emitter amplifier?
1. 16
2. 24
3. 20
4. 32
A black body has maximum radiation intensity at the wavelength at 2000 K. Its corresponding wavelength at 3000 K will be
1.
2.
3.
4.
The relation connecting magnetic susceptibility , and relative permeability is
1.
2.
3.
4.
When a metallic plate swings between the poles of a magnet
1. no effect of the plate
2. eddy current are set up inside the plate and the direction of the current is along the motion of the plate
3. eddy current are set up inside the plate and then direction of the current oppose the motion of the plate
4. eddy currents are set up inside the plate
A small square loop of wire of side 'l' is placed inside a large square loop of side 'L' (L>>l). If the loops are coplanar and their centres coincide, the mutual induction of the system is directly proportional to
1. L/l
2. l/L
3.
4.
An AC source is rated at 220 V, 50Hz, the time taken for the voltage to change from the peak value to zero is
1. 50 sec
2. 0.02 sec
3. 5 sec
4. 5 x sec
| 1. | real |
| 2. | virtual |
| 3. | depends on the nature of the object |
| 4. | none of these |
An endoscope is employed by a physician to view the internal parts of a body organ. It is based on the principle of
1. refraction
2. reflection
3. total internal reflection
4. dispersion
A point object is placed beyond the focus of a convex lens cut into two halves, each of which are separated by a small distance. Then the number of images fomed will be

1. zero
2. one
3. two
4. four
nth bright fringe of red light coincides with (n+1)th bright fringe of green light . The value of n =?
1. 4
2. 5
3. 3
4. 2
Mature sperms in humans are stored in:
| 1. | Seminal vesicles | 2. | Epididymis |
| 3. | Vas deferens | 4. | Rete testis |
Menstruation is triggered by an abrupt decline in the amount of:
1. Estrogen
2. Progesterone
3. LH
4. FSH

1. \(60~\text{N}\)
2. \(\frac{60}{\sqrt{2}}~\text{N}\)
3. \(30~\text{N}\)
4. \(\frac{30}{\sqrt{2}}~\text{N}\)
A particle of mass m collides with another stationary particle of mass M such that the second particle starts moving and the first particle stops just after collision. Then which of the following conditions must always be valid ?
1.
2.
3.
4.
The potential energy U between two atoms in a diatomic molecule as a function of the distance x between atoms has been shown in the adjoining figure. The atoms are 

1. attracted when x lies between A and B and repelled when x lies between B and C
2. attracted when x lies between B and C and repelled when x lies between A and BA
3. attracted when they reach B
4. repelled when they reach B