Match each item in Column I with one in Column II and select your answer from the codes given below:
| Column I | Column II | ||
| A. | Diatoms | a. | Protein-rich layer of pellicle |
| B. | Dinoflagellates | b. | Cell wall embedded with silica |
| C. | Euglenoids | c. | Spores with true walls |
| D. | Slime moulds | d. | Cellulose plates in the cell wall |
Codes:
| A | B | C | D | |
| 1. | b | a | c | d |
| 2. | b | d | a | c |
| 3. | b | c | a | d |
| 4. | b | a | d | c |
All the following statements regarding Basidiomycetes are correct except:
| 1. | The mycelium is branched and septate |
| 2. | Asexual spores and vegetative reproduction are generally not found |
| 3. | Sex organs are absent |
| 4. | Basidiospores are exogenously produced on the basidium |
Match the Scientists in Column I with their contribution in the field of virology given in Column II and select your answer from the codes given below:
| Column I | Column II | ||
| A. | Ivanowsky | a. | Recognized tobacco mosaic is caused by microbes smaller than bacteria |
| B. | Beijerinck | b. | Demonstrated extract of infected tobacco plants could cause infection in healthy plants |
| C. | Stanley | c. | Showed that viruses could be crystallized |
Codes:
| A | B | C | |
| 1. | a | b | c |
| 2. | a | c | b |
| 3. | b | a | c |
| 4. | b | a | d |
Sexual reproduction by non-flagellated but similar in size gametes is seen in:
1. Chlamydomonas
2. Volvox
3. Spirogyra
4. Fucus
In mosses, the sex organs are seen in:
1. Protonema stage
2. Leafy stage
3. Capsule of the sporophyte
4. Seta of the sporophyte
The features seen in the gymnosperm Cycas include:
| I. | Coralloid roots |
| II. | Unbranched stems |
| III. | Pinnate persistent leaves for a few years |
| IV. | Male cones and megasporophyllys borne on same plant |
1. I and II only
2. I and IV only
3. I, II and III only
4. I, II, III and IV
Both bryophytes and pteridophytes:
1. are vascular cryptogams
2. have a dominant sporophyte
3. reproduce only sexually
4. are haplo-diplontic
Identify the incorrectly matched pair:
| Animals | Feature present in both | |
| 1. | Balanoglossus and Pinctada | Open circulatory system |
| 2. | Branchiostoma and Ascidia | Persistent notochord |
| 3. | Aplysia and Pheretima | True coelom |
| 4. | Gorgonia and Pennatula | Cnidoblasts |
Pteropus is a/an:
| 1. | Limbless amphibian |
| 2. | Extinct reptile that evolved into mammals |
| 3. | Bony fish that migrates from sea water to fresh water for breeding |
| 4. | Mammal with flight |
The cells of which of the following regions of the root are responsible for growth of its length?
1. Root cap
2. Region of meristematic activity
3. Region of elongation
4. Region of maturation
Identify the correct statement regarding the stem morphology from the following:
| I: | Stems bear only axillary buds. |
| II: | Axillary buds may get modified into thorns as in Citrus |
| III: | Stem tendrils develop from axillary buds as in gourds. |
| 1. | Only I and II | 2. | Only I and III |
| 3. | Only II and III | 4. | I, II and III |
The type of placentation show in the given figure is seen in:
| 1. | Dianthus | 2. | Argemone |
| 3. | China rose | 4. | Marigold |
The given floral diagram can be of:
1. Asparagus
2. Atropa belladonna
3. Mustard
4. Indigofera
Parts of grasses removed by the grazing herbivores are regenerated due to the activity of:
1. Apical meristem
2. Intercalary meristem
3. Interfascicular cambium
4. Fascicular vascular cambium
In the dicot root, the vascular cambium:-
| 1. | is absent |
| 2. | is completely secondary in origin |
| 3. | does not form a continuous ring |
| 4. | originates from the tissue just above the phloem bundles |
The most abundant and widely distributed tissue in the bodies of complex animals is:
1. Epithelium
2. Connective
3. Muscular
4. Neural
The mouth part of the cockroach shown in the given diagram acts as a/an:
1. Upper lip
2. Tongue
3. Lower lip
4. Uvula
The tail of the phospholipid molecule:
| 1. | is hydrophilic and composed of phosphate |
| 2. | is hydrophilic and composed of saturated hydrocarbons |
| 3. | is hydrophobic and composed of saturated hydrocarbons |
| 4. | is hydrophobic and composed of unsaturated hydrocarbons |
In animal cells, lipid-like steroidal hormones are synthesized in the:
1. Nucleus
2. Rough endoplasmic reticulum
3. Smooth endoplasmic reticulum
4. Golgi apparatus
Unless specifically stained by a vital stain like Janus Green, which of the following will not be visible under a microscope?
1. Rough endoplasmic reticulum
2. Smooth endoplasmic reticulum
3. Golgi apparatus
4. Mitochondria
Fat soluble pigments like xanthophylls are present in:
1. Chromoplasts
2. Aleuroplasts
3. Elaioplasts
4. Amyloplasts
Phenyl mercuric acetate (PMA) results in
1. reduced photosynthesis
2. reduced transpiration
3. reduced respiration
4. killing of plants.
When the plants are grown in magnesium deficient but urea rich soil, the symptoms expressed are:
1. yellowish leaves
2. colourless petiole
3. dark green leaves
4. shoot apex die.
Choose the correct order for the steps of hemostasis:
1. Blood coagulation, platelet plug formation, blood vessel spasm
2. Platelet plug formation, blood coagulation, blood vessel spasm
3. Blood vessel spasm, platelet plug formation, blood coagulation
4. Blood vessel spasm, blood coagulation, platelet plug formation
The right atria of the human heart receive:
1. Oxygenated blood
2. Deoxygenated blood
3. Arterial blood
4. Venous blood
Which of the following statements about the kidneys is incorrect?
1. They consume over 20% of the oxygens used by the body at rest.
2. Approximately 1200ml of fluid is filtrated by the kidneys each day.
3. 120-125 ml of plasma is forced into the renal tubules each minute.
4. They compose less than 1% of the body weight.
The following statements refer to gas carriage by blood. Choose the correct statement:
| 1. | After leaving the lungs, each litre of blood contains about 20 ml of oxygen. |
| 2. | Haemoglobin is half saturated with oxygen when the PO2 is about 30 mm Hg |
| 3. | As the PCO2 rises, the affinity of haemoglobin for oxygen is increased. |
| 4. | Each litre of arterial blood contains about 50 mL of carbon dioxide. |
Regarding the control of water balance by the kidneys:
| 1. | The renal medulla has an osmotic gradient that decreases from the border with the cortex to the renal papilla. |
| 2. | ADH is secreted by anterior pituitary in response to a decrease in the osmolality of the blood. |
| 3. | A normal person cannot produce urine with an osmolality greater than 300 mOsmil.kg-1. |
| 4. | ADH acts on the cells of the collecting ducts to increase their permeability to water. |
Study the spirograph and identify the correctly matched volumes from the codes given: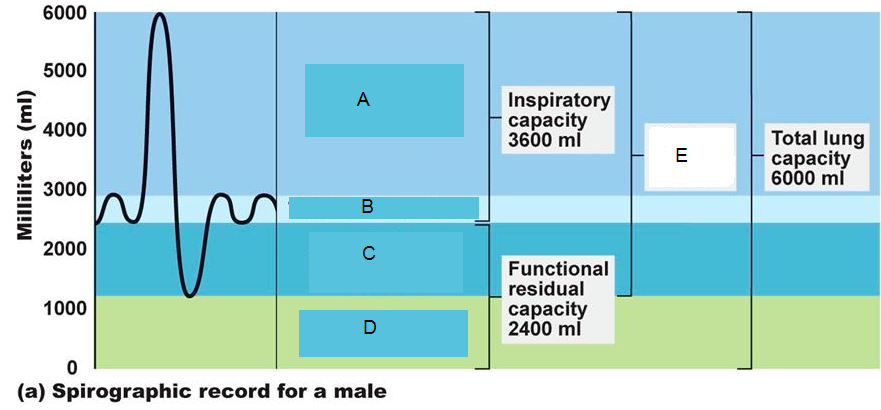
TV IRV ERV RV VITAL CAPACITY
1. A c B D E
2. A B C D E
3. B A C D E
4. C B A D E
What regulates the opening of oesophagus into the stomach?
1. A membranous valve
2. A muscular sphincter
3. A mesodermal septum
4. A cartilaginous flap
Muscularis layer in the wall of the alimentary canal is formed by smooth muscles usually arranged into:-
1. an inner circular and an outer longitudinal layer.
2. an inner longitudinal and an outer circular layer.
3. both circular layers
4. both longitudinal layers
A person passes a lot of dilute urine and drinks a lot of water but does not have glycosuria. He is most likely suffering from :
1. Type 1 diabetes mellitus
2. Type 2 diabetes mellitus
3. Pituitary diabetes
4. Diabetes insipidus
The upper ends of the forearm bones articulate with each other in a:
1. pivot joint
2. ellipsoid joint
3. saddle joint
4. hinge joint
The substrate used for sperm metabolism is produced by the
1. prostate
2. seminal vesicle
3. Cowper's gland
4. testes
With respect to external fertilization, which of the following is not an advantage of internal fertilization?
1. A decreased risk of disease transmission
2. Minimal waste of gametes
3. Greater chances of individual egg fertilization
4. Relatively longer time of egg protection
Which of the following is the most common abiotic pollinating agent?
| 1. | Insects | 2. | Birds and bats |
| 3. | Wind | 4. | Water |
Seeds offer several advantages to angiosperms. Seed formation is more dependable because:
1. Seeds have better adaptive strategies for dispersal
2. Reproductive processes become independent of water
3. Hard seed coat provides protection to the young embryos
4. They generate new genetic combinations leading to variations
The given diagram exemplifies the fruit in:
1. Fig
2. Pineapple
3. Mulberry
4. Raspberry
Which of the following hormones acts on the Sertoli cells to stimulate secretion of some factors which help in the process of spermiogenesis?
1. FSH
2. LH
3. Testosterone
4. GnRH
The developing oocyte divides and releases the first polar body in :
1. Primary follicle
2. Secondary follicle
3. Tertiary follicle
4. Graffian follicle
During parturition, the fetal ejection reflex triggers the release of:
| 1. | An increasing amount of estrogen and a decreasing amount of progesterone from the placenta |
| 2. | A decreasing amount of estrogen and an increasing amount of progesterone from the placenta |
| 3. | Oxytocin from the maternal pituitary |
| 4. | GnRH from the fetal hypothalamus |
Progesterone alone or in combination with estrogen can also be used as contraceptives by females as injections or implants under their skin. When used in this manner, they:
| 1. | do not affect the release of the ovum |
| 2. | induce a foreign body reaction leading to rejection of the implanted blastocyst |
| 3. | can cause increased risk of ectopic pregnancy |
| 4. | have a much longer effective period |
A synthetic mRNA of repeating sequence 5'-CACACACACACACACAC... is used for a cell-free protein synthesizing system like the one used used by Nirenberg. If we assume that protein synthesis can begin without the need for an initiator codon, what product or products would you expect to occur after protein synthesis?
1. one protein, consisting of a single amino acid
2. three proteins, each consisting of a different, single amino acid
3. two proteins, each with an alternating sequence of two different amino acids
4. one protein, with an alternating sequence of two different amino acids
Fredrick Griffith accidentally discovered transformation when attempting to develop a vaccine for pneumonia. He injected mice with samples from S-strain (virulent) and/or R-strain (nonvirulent) pneumococci bacteria (Sterptococcus pneumoniae). Which of the following results is NOT consistent with Griffith's experiments?
| 1. | injected S-strain; mouse dies. |
| 2. | injected R-strain; mouse lives. |
| 3. | injected heat-killed S-strain; mouse lives. |
| 4. | injected mixture of heat-killed S-strain and live R-strain; mouse lives. |
Which scientists first gave experimental evidence that DNA is the genetic material?
| 1. | Avery, MacLeod , and McCarty who repeated the transformation experiments of Griffith, and chemically characterized the transforming principle. |
| 2. | Garrod, who postulated that Alcaptonuria, or black urine disease, was due to a defective enzyme. |
| 3. | Beadle and Tatum, who used a mutational and biochemical analysis of the bread mold Neurospora to extablish a direct link between genes and enzymes. |
| 4. | Meselson and Stahl who showed that DNA is replicated semiconservatively. |
Consider the cross AaBb x AaBb. If the alleles for both genes exhibit complete dominance, what genotypic ratio is expected in the resulting offspring?
1. 1:1:1:1
2. 9:3:3:1
3. 3:6:3:1:2:1
4. 1:2:1:2:4:2:1:2:1
The function of the rho protein is
1. to help terminate translation
2. to help RNA polymerase bind to the DNA
3. to help RNA polymerase find a promoter
4. to help terminate transcription
Two phenotypically normal individuals have an affected child. What can we conclude about the parents?
1. they both carried the diesease allele
2. they are not the parents of the child
3. they are affected
4. no conclusions can be drawn
What is the basis of pleiotropy?
1. A spontaneous mutation during the replication of DNA.
2. Interrelationship between various metabolic pathways in the body.
3. Chromosomal aberration as chromosomes are the vehicles of genes.
4. the behaviour of chromosomes during meiosis or gamete formation.
A female whose father was colorblind marries and normal male whose father was also colorblind. What is the probablility that their daughter will be colorblind?
1. 0 %
2. 25 %
3. 50 %
4. 75 %
DNA damage leading to neoplastic transformation can be brought about by ionizing radiations like :
1. UV rays
2. Gamma rays
3. Infrared waves
4. Radio waves
The skeletal structure shown in the given diagram shows the structure of :
| 1. | Morphine | 2. | Diacetyl morphine |
| 3. | Cannabinoid | 4. | Cocaine |
Inbreeding depression in animals can be overcome by:
1. A single outcross
2. Out cross for many generations
3. Cross-breeding
4. Inter-specific hybridization
Identify the incorrectly matched pair:
|
|
Crop Variety |
Resistant to |
|
1. |
Himgiri |
Leaf and stripe rust |
|
2. |
Karan rai |
White rust |
|
3. |
Pusa sadabahar |
Bacterial blight |
|
4. |
Pusa shubhra |
Black rot |
Meristem culture is especially important to obtain:
1. Drought resistant plants
2. Virus free plants
3. Biofortified plants
4. Androgenic haploids
Identify the incorrectly matched pair:
|
1 |
Streptococcus |
Clot buster |
|
2. |
Monascus purpureus |
Statins |
|
3. |
Trichoderma harziamum |
Immunosuppressant |
|
4. |
Aspergillus niger |
Citric acid |
Baculoviruses are:
1. Species specific, narrow spectrum insecticides
2. Species specific, broad spectrum insecticides
3. Non specific, narrow spectrum inseticides
4. Non specific, broad spectrum insecticides
Microinjection is suitable for :
1. Injecting an ovum into the sperm in IVF
2. Transforming animal cells
3. Injecting very small sized drug particles into neurons
4. Conferring antibiotic resistance to a certain strain of bacteria
Polymerase Chain Reaction is not used in :
| 1. | Confirming presence of a pathogen during early infection |
| 2. | Identifying the mutated genes in suspected cancer patients |
| 3. | Isolating the gene of interest from host DNA to be cloned by recombinant procedures |
| 4. | Detection of the presence of HIV in suspected AIDS patient |
If gene isolate from marrow cells producing ADA is introduced into cells at early embryonic stage, it could be a permanant cure of ADA deficiency because the early embryonic cells :
1. Are more differentiated than the later stage cells
2. Are constantly dividing and do not get differentiated
3. Are easy to manipulate than the later stage cells
4. Are virtually immortal
To test the safety of polio vaccine, the organisms used are transgenic :
1. Mice
2. Pigs
3. Monkey
4. Cattle
Very small animals are rarely found in polar region mainly because:
1. Smaller animals have a relatively slower heart rate
2. Smaller animals have a more surface area relative to their volume
3. Smaller animals are invariably herbivores
4. Smaller animals rely on diffusion for exchange of gases with the environment
When the population density reaches the carrying capacity, the logistic growth curve is said to be in:
1. Lag phase
2. A phase of acceleration
3. A phase of deceleration
4. Asymptote
In general, which of the following is not an adaptation seen in parasites in accordance with their life style?
1. Loss of unnecessary sense organs
2. Presence of adhesive organs
3. Low reproductive capacity
4. Loss of digestive system
The rate of formation of new organic matter by consumers is called as:
1. Secondary productivity
2. Standing crop
3. Standing state
4. Net primary productivity
The limitations of ecological pyramids include all the following except:
1. The do not take into account the same species belonging to two or more trophic levels
2. They do not represent relationships between organisms at different trophic levels
3. They assume a simple food chain and do not consider food webs
4. Saprotrophs are not given any place in the ecological pyramids
About what percent of the global carbon is contained in the atmosphere?
1. 71
2. 49
3. 17
4. about 1
Which of the following has maximum global biodiversity?
1. Angiosperms
2. Algae
3. Lichens
4. Mosses
What is applicable to both Lates niloticus and Lantana camara ?
1. They are on the verge of extinction due to over-exploitation by humans.
2. They are alien species that became invasive in certain environments causing threat to indigenous biodiversity.
3. They are mutualists and likely to undergo co-extinction in recent future.
4. They are keystone species and are vital to the stability of tropical ecosystems.
Eventually, the only solution for the treatment of e-wastes, provided it is carried out in an environment friendly manner, is:
1. Incineration
2. Burying in landfill sites
3. Dumping in developing countries
4. Recycling
Match each item in Column I with one item in Column II and choose the correct answer from the code given below:
| A. | Air [Prevention and Control of Pollution] Act | a. | 1981 |
| B. | Water [Prevention and Control of Pollution] Act | b. | 1974 |
| C. | Environment [Protection] Act | c. | 1986 |
| D. | Montreal Protocol | d. | 1987 |
Codes:
| A | B | C | D | |
| 1. | a | b | c | d |
| 2. | b | a | d | c |
| 3. | d | c | b | a |
| 4. | c | d | a | b |
The reaction of 

1.
2.
3.
4. 
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Which of the following alkanes will give the maximum number of isomers when a monochloro-substituted product is obtained?
| 1. |  |
2. |  |
| 3. | \(\mathrm{CH_4}\) |
4. |  |
An open flask containing air is heated from 300 K to 500 K. The percentage of air that will escape into the atmosphere if pressure is kept constant is-
1. 80
2. 40
3. 60
4. 20
Most stable radical is
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Which of the following is the most stable carbonium ion?
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The stability of the given compounds, in descending order, is:
| A. |  |
B. |  |
| C. |  |
D. |  |
1. A > B > C > D
2. D > B > C > A
3. D > C > B > A
4. A > B > D > C
The decreasing order of cases of leaving group?
-OAC -OMe -OSO2Me -OSO2CF3
(I) (II) (III) (IV)
1. I>II>III>IV
2. IV>III>I>II
3. II>I>III>IV
4. IV>III>II>I
Which among the following is most basic?
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Compound PdCl4.6H2O is a hydrated complex, 1 molal aqueous solution of it has freezing point 269.28 K. Assuming 100 % ionization of complex, the molecular formula of the complex is-
(Kf for water = 1.86 K kg mole-1)
1. [Pd(H2O)6]Cl4
2. [Pd(H2O)4Cl2]Cl2.2H2O
3. [Pd(H2O)3Cl3]Cl.3H2O
4. [Pd(H2O)Cl4]4H2O
White phosphorous reacts with calcium to form a certain compound which on hydrolysis produces
1. PH3
2. H3PO4
3. P4O6
4. P4O10
The correct order of ease of hydrolysis is :
1. CCl4 < SiCl4 < PCl5 < AlCl3
2. AlCl3 < CCl4 < PCl5 < SiCl4
3. CCl4 < AlCl3 < PCl5 < SiCl4
4. CCl4 < AlCl3 < SiCl4 < PCl5
Select the incorrect statement among the following
1. Aspirin is both analgesic and antipyretics
2. Ampicillin is a natural antibiotic
3. Sulphadiazine is a synthetic antibacterial
4. Some disinfectants can be used as antiseptics in lower concentrations
Calculate the molar solublity of Fe(OH)2 at a pH of 8
[Ksp of Fe(OH)2 = 1.6 X 10-14]
1. 0.06
2. 0.016
3. 0.01
4. 0.16
Ionic radii are :
1. Directly proportional to square of effective nuclear charge.
2. Inversely proportional to effective nuclear charge .
3. Inversely proportional to square of effective nuclear charge .
4. Directly proportional to effective nuclear charge.
To a 200 ml of 0.1 M weak acid HA solution 90 ml of 0.1 M solution of NaOH be added, Now, what volume of 0.1 M NaOH be added into above solution so that pH of resulting solution be 5.
[Ka(HA) = 10-5]
1. 2 ml
2. 20 ml
3. 10 ml
4. 15 ml
If carbon dioxide is 2% dissociated at equilibrium 2CO2 (g)2CO(g) + O2(g). The mole fraction of CO2 at equuilibrium is
1. 1.01/0.98
2. 0.98/1.01
3. 0.01/0.98
4. 0.098/1.01
Oxidation number of carbon in CH2Cl2 is
1. -4
2. +4
3. -2
4. zero
A mixture contains Na2CO3 and NaHCO3 and wt. of mixture is 10gm. Mixture on heating liberates 56ml of CO2 at S.T.P, wt of Na2CO3 in mixture is
1. 9.58 gm
2. 5 gm
3. 7.5 gm
4. 8 gm
The correct order of relative rates of hydrogenation of alkenes is:
1. Ethylene > propene > 2-butene > 2-methyl-2-butene
2. 2-methyl-2-butene > 2-butene > Propene > Ethylene
3. 2-butene > propene > ethylene > 2-methyl-2-butene
4. Propene > 2-butene > ethylene > 2-methyl-2-butene
For N2 + 3H2 2NH3, one mole of N2 and three moles of H2 are at pressure of 4 atm. Equilibrium pressure is found to be 3 atm. Hence, Kp is
1. 1/(0.5)x(0.15)3 2. 1/(0.5)x(1.5)3
3. 3x3/(0.5)x(1.5)3 4. none of the above
V versus T curves at constant pressure P1 and P2 for an ideal gas are as shown in the figure. The correct statement is-
1. P1>P2
2. P1<P2
3. P1=P2
4. All of the above
Consider the given reaction:
 Products formed
Products formed
| (A) |  |
(B) |  |
| (C) |  |
(D) |  |
1. (A) and (B) only 2. (A) and (C) only
3. (B) and (C) only 4. (A), (B), (C) and (D)
The dissociation of NH4OH can be suppressed by the addition of:
| 1. | NH4Cl | 2. | NH4NO3 |
| 3. | NaOH | 4. | All of the above |
What amount of CaCl2(i=2.47) is dissolved in 2L water so that its osmotic pressure is 0.5 atm at 27:-
1. 13.42 g
2. 19.24 g
3. 8.834 g
4. 1.820 g
On heating Li2CO3 and Na2CO3.10H2O, residue is left after heating. The weight loss is due to
1. Evolution of CO2 from Na2CO3.10H2O only
2. Evolution of CO2 from Li2CO3 only
3. Due to evolution of CO2 from Li2CO3 and H2O from Na2CO3.10H2O
4. H2O from Na2CO3.10H2O
In the reaction,
2-butene ; (B) is
1. Butane
2. 1-Butene
3. Butyraldehyde
4. Butanone
In the Cannizzaro reaction given below,
the slowest step is
1. The attack of OH- ion to the carbonyl group
2. The transfer of hydride ion to the carbonyl group
3. The abstration of proton from the carboxylic acid
4. The deprotonation of Ph-CH2OH
Which of the following gives the fastest Lucas test?
| 1. |  |
2. | |
| 3. |  |
4. |  |
1.
2.
3.
4.
A pendulum clock runs faster by \(5\) s per day at \(20^{\circ}\mathrm {C}\) and goes slow by \(10\) s per day at \(35^{\circ}\mathrm {C}\). It shows the correct time at a temperature of:
1. \(27.5^{\circ}\mathrm {C}\)
2. \(25^{\circ}\mathrm {C}\)
3. \(30^{\circ}\mathrm {C}\)
4. \(33^{\circ}\mathrm {C}\)
For an ideal gas V-T curves at constant pressure and are shown in figure. From the figure

1.
2.
3.
4.
A thin rod of length L is bent to form a semicircle. The mass of the rod is M. The gravitational potential at the centre of the circle is :
1.
2.
3.
4.
A point \(P\) lies on the axis of a ring of mass \(M\) and radius \(a\) at a distance \(a\) from its centre \(C\). A small particle starts from \(P\) and reaches \(C\) under gravitational attraction. Its speed at \(C\) will be:
1. \(\sqrt{\frac{2 GM}{a}}\)
2. \(\sqrt{\frac{2 GM}{a} \left(1 - \frac{1}{\sqrt{2}}\right)}\)
3. \(\sqrt{\frac{2 GM}{a} \left(\sqrt{2} - 1\right)}\)
4. zero
The energy needed in breaking of a drop of liquid of radius R into n drops of radius r is given by (T is surface tension and P is atmospheric pressure) :
1.
2.
3.
4.
A tank is filled with water up to a height H. A hole is made at a depth h below the free surface of the water in the tank. The water coming out of the orifice strikes at a distance S from the walls of the tank. Then S is given by :
1. \(\sqrt{2 g(H-h)}\)
2. \(\sqrt{g(H-h)}\)
3. \(\sqrt{2 g h}\)
4. \(\sqrt{4 h(H-h)}\)
An incompressible liquid travels as shown in the figure. The speed of the fluid in the lower branch will :

1. 1 m/s
2. 1.5 m/s
3. 2.25 m/s
4. 3 m/s

1. \(\frac{3g}{2L}\)
2. \(\frac{3g}{\sqrt{2}L}\)
3. \(\frac{3g}{2\sqrt{2}L}\)
4. \(\frac{6g}{\sqrt{2}L}\)
From a disc of mass M and radius '2a', a concentric disc of radius 'a' is cut out. The moment of inertia of remaining disc is
1.
2.
3.
4.
A meter bridge is set up to determine unknown resistance \(x\) using a standard \(10~\Omega\) resistor. The galvanometer shows the null point when the tapping key is at a \(52\) cm mark. End corrections are \(1\) cm and \(2\) cm respectively for end \(A\) and \(B\). Then the value of \(x\) is:

1. \(10.2~\Omega\)
2. \(10.6~\Omega\)
3. \(10.8~\Omega\)
4. \(11.1~\Omega\)
The current through the \(5~\Omega\) resistor is:
| 1. | \(3.2~\text A\) | 2. | \(2.8~\text A\) |
| 3. | \(0.8~\text A\) | 4. | \(0.2~\text A\) |
| 1. | \(28\) C | 2. | \(30.5\) C |
| 3. | \(8\) C | 4. | \(82\) C |
In the figure, a carbon resistor has bands of different colours on its body as shown. The value of the resistance is:
1. 2.2 k
2. 3.3 k
3. 5.6 k
4. 9.1 k

A circular coil of wire of radius 'r' has 'n' turns and carries a current 'I'. The magnetic induction (B) at a point on the axis of the coil at a distance from its center is :
1.
2.
3.
4.
The dots in the figure depict a magnetic field that is perpendicular to the plane of the paper and emanates from it. The trajectory of a particle in the plane of the paper is depicted by the curve \(ABC\). What exactly is the particle?

| 1. | Proton. | 2. | Electron. |
| 3. | Neutron. | 4. | It cannot be predicted. |
| 1. | the speed will change. |
| 2. | the direction will change. |
| 3. | both (1) and (2) |
| 4. | none of the above |
The grid (each square of 1 m 1m), represents a region in space containing a uniform electric field, if potentials at points O,A,B,C,D,E,F & G,H are respectively 0,-1,-2,1,2,0,-1,1 and 0 volts, find the electric field intensity

1.
2.
3.
4.
The electrostatic field due to a charged conductor just outside the conductor is:
| 1. | zero and parallel to the surface at every point inside the conductor. |
| 2. | zero and is normal to the surface at every point inside the conductor. |
| 3. | parallel to the surface at every point and zero inside the conductor. |
| 4. | normal to the surface at every point and zero inside the conductor. |
X and Y are large, parallel conducting plates close to each other. Each face has an area A. X is given a charge Q. Y is without any charge. Point A, B, and C are as shown in the figure. Find the incorrect option.


1. the field at B is
2. the field at B is
3. the fields at A, B and C are of the same magnitude
4. the fields at A and C are of the same magnitude but in opposite directions
A 4 capacitor is charged by a 200 V supply. It is then disconnected from the supply and is connected to another uncharged 2 capacitor. How much electrostatic energy of the first capacitor is lost in the form of heat and electromagnetic radiation?
1.
2.
3.
4.
| 1. | \(\left[MLT^{-2}\right]\) | 2. | \(\left[ML^{-1}T^{2}\right]\) |
| 3. | \(\left[ML^{-1}T^{-2}\right]\) | 4. | \(\left[MLT^{2}\right]\) |
A particle has initial velocity and has acceleration . Its speed after 10 s:
1. 7 units
2. units
3. 8.5 units
4. 10 units
The gravity in space is given by . Two particles are simultaneously projected with velocity and . Then, the ratio of their times of flight
1. 1:1
2. 1:2
3. 2:1
4. none
A mass of 0.5kg moving with a speed of 1.5 m/s on a horizontal smooth surface collides with a nearly weightless spring of force constant k = 50 N/m. The maximum compression of the spring would be
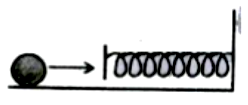

1. 0.5 m
2. 0.15 m
3. 0.12 m
4. 1.5 m
Interference fringes are produced using white light in a double-slit arrangement. When a mica sheet of uniform thickness of the refractive index 1.6 (relative to air) is placed in the path of light from one of the slits, the central fringe moves by a distance.
This distance is equal to the width of 30 interference bands. If the light of wavelength 4800 is used, the thickness (in m) of mica is —
1. 90
2. 12
3. 14
4. 24
A position dependent force \(F=7-2x+3x^2\) N acts on a small body of mass \(2\) kg and displaces it from \(x = 0\) to \(x = 5\) m. The work done in joule is:
| 1. | \(70\) | 2. | \(270\) |
| 3. | \(35\) | 4. | \(135\) |
The initial activity of a certain radioactive isotope was measured as 16000 counts per minute. That the only activity measured was due to this and that its activity after 12 hours was 2000 counts/minute. Its half-life, in hours, is nearest to-
1. 9.0
2. 6.0
3. 4.0
4. 3.0
Magnetic moment due to the motion of the electron in energy state of a hydrogen atom is proportional to
1. n
2.
3.
4.
How much energy is contained in a particle that has a mass of
A radioactivity nuclide can decay simultaneously by two different processes that have decay constants and . The effective decay constant of the nuclide is Then-
1.
2.
3.
4.
A semiconductor is known to have an electron concentration of \(8\times 10^{13}~\text{cm}^{-3},\) and a hole concentration of \(5\times 10^{2}~\text{cm}^{-3}.\) The semiconductor is:
| 1. | \(\mathrm{n}\text-\)type | 2. | \(\mathrm{p}\text-\)type |
| 3. | intrinsic | 4. | insulator |
In the below-mentioned reaction, the major product (A) formed is:


| 1. |   |
2. |   |
| 3. |   |
4. | None of these |
Magnetic permeability is maximum for
1. Diamagnetic susbtance
2. Paramagnetic substance
3. Ferromagnetic substance
4. All of these
A small telescope has an objective lens of focal length 144 cm and an eye-piece of focal length 6.0 cm. The magnifying power of the telescope is ( when the final image is at infinity)
1. 24
2. 25
3. 30
4. 150
The configuration of given tartaric acid is

1. 2R, 3R 2. 2R, 3S
3. 2S, 3S 4. 2S, 3R