As we go lower from kingdom to species, the number of common characters at each taxon:
1. Goes on decreasing
2. Goes on increasing
3. Remains same
4. There is no pattern to this
Members of Kingdom Protista:
1. are primarily aquatic
2. do not have membrane bound organelles
3. are all ciliated or flagellated
4. reproduce exclusively by asexual means
Asexaul reproduction by pear shaped biflagellate zoospores is seen in most:
1. Green algae
2. Brown algae
3. Red algae
4. Both (2) and (3)
Echinoderms are not characterized by:
| 1. | An absence of excretory system |
| 2. | Bisexual animals |
| 3. | Usually external fertilization |
| 4. | Indirect development with free swimming larva |
In vexillary aestivation:
1. The standard overlaps the wings
2. The standard overlaps the keel
3. The standard is overlapped by keel
4. The keel overlap the wings
Identify the incorrect statement:
| 1. | The presence of vessels is a characteristic of angiosperms |
| 2. | In roots the primary xylem is exarch |
| 3. | Gymnosperms lack albuminous cells and sieve cells |
| 4. | Bast fibers are generally absent in primary phloem |
Which of the following does not help in excretion in Periplaneta Americana?
1. Fat body
2. Urecose glands
3. Nephrocytes
4. None of these
There cannot be a living state without :
1. Metabolism
2. Capability of reproduction
3. Self-consciousness
4. Presence of DNA in a cell
There is a possibility of exposure of the maternal blood [of Rh -ve mother] to small amounts of Rh +ve blood from the fetus during the:
1. First trimester of any pregnancy
2. Third trimester of the second pregnancy only
3. Delivery of the child
4. There is no such possibility
Identify the incorrect statement :
| 1. | Heterotrophic bacteria are important decomposers |
| 2. | Diatoms are chief producers in the oceans |
| 3. | The pigments of euglenoids are identical to those present in higher plants |
| 4. | Very few protozoans are autotrophs |
All the following pteridophytes belong to the Class Pteropsida except:
1. Dryopteris
2. Pteris
3. Selaginella
4. Adiantum
Operculum and an air bladder are present in :
1. Clarias
2. Pristis
3. Trygon
4. Carcohrodon
Which of the following would not be a character of Asparagus?
1. Actinomorphic flower
2. Imbricate aestivation
3. Superior ovary
4. Endospermous seed
Dense irregular connective tissue is present in:
1. Tendons
2. Ligaments
3. Skin
4. Cartilage
All the following are the part of a eukaryotic cell endomembranous system except:
1. Vacuole
2. Lysosome
3. Golgi complex
4. Mitochondria
When the co-factor is removed from the enzyme, its catalytic activity:
1. remains same
2. is increased
3. is greatly reduced
4. is lost
The cell shown in the given diagram is in:
1. Early prophase
2. Late prophase
3. Metaphase
4. Telophase
Which of the following is/are essential for imbibitions to take place?
I. Water potential gradient between the absorbent and the liquid imbibed
II. Affinity between the adsorbant and the liquid
1. Only II
2. Only I
3. Both I and II
4. None
How many ATP molecules are required to biologically fix one molecule of nitrogen into two molecules of ammonia?
1. 4
2. 8
3. 12
4. 16
Consider the following two statements:
| I: | Except for plants in shade or in dense forests, light is rarely a limiting factor in nature for photosynthesis. |
| II: | Light saturation occurs at 50% of the full sunlight. |
| 1. | Both statements are correct and II explains I |
| 2. | Both statements are incorrect |
| 3. | I is correct and II is incorrect |
| 4. | I is incorrect and II is correct |
The sites of perception of light/dark duration in the plants are the:-
1. Shoot apices
2. Leaves
3. Roots
4. Branches
The useful purpose served by lactate fermentation is:
| 1. | Make lactose available for gluconeogenesis |
| 2. | Production of additional ATP in anaerobic conditions |
| 3. | Regeneration of NAD+ |
| 4. | Increased availability of oxygen for the skeletal muscle |
The mucosal layer in the stomach form irregular folds known as:-
1. villi
2. lumen
3. rugae
4. crypts of Lieberkuhn
Pneumotaxic centre which can moderate the functions of the respiratory rhythm centre is present at:
1. Pons region of brain
2. Thalamus
3. Spinal cord
4. Right cerebral hemisphere
Congestion of the lungs is one of the main symptoms in:
1. Hypotension
2. Coronary artery disease
3. Angina
4. Heart failure
The condition where urea accumulates in blood is:
1. Glycosuria
2. Uremia
3. Ketonuria
4. Acidosis
Scapula is a large triangular flat bone situated in the dorsal part of the thorax between:
1. the second and fifth ribs
2. the second and seventh ribs
3. the third and sixth ribs
4. the third and eighth ribs
The amino acid derivative among the following hormone is:-
1. Insulin
2. Epinephrine
3. Estradiol
4. Testosterone
To safeguard our water resources, the Water (Prevention and Control of Pollution) Act, was passed in:
1. 1951
2. 1974
3. 1985
4. 1987
If global warming continues, in general, how what would be the expected effect on the distributional range of some species ?
1. It will be an equatorial shift
2. It will be a pole ward shift
3. It will have no impact
4. It will affect only plants
The logistic growth curve of animal population growth is more realistic than J shaped curve because:
1. Asexual mode of reproduction is rare in higher animals
2. Resources are finite and become limiting sooner or later.
3. Most animals are conformers rather than regulators
4. Abiotic factors affect animals more than plants
Which of the following is not a function of predators?
| 1. | They act as conduits for energy transfer across trophic levels |
| 2. | They keep prey populations under control |
| 3. | They help in the stabilization of the ecosystems |
| 4. | They decrease the species diversity in a community |
What percent of energy in the sunlight availble to the primary producers is converted by them into net primary productivity ?
1. 1
2. 2
3. 5
4. 10
Both hydrach and xerarch successions:
1. Take same time
2. Have similar sere
3. Lead to mesic conditions
4. Have same pioneer species
The second trophic level in a lake is:
1. Phytoplankton
2. Zooplankton
3. Benthos
4. Fishes
The relation between species richness and area for a wide variety of taxa on a logarithmic scale is a:
1. rectangular hyperbola
2. straight line
3. sigmoid curve
4. sine curve
The intine of the pollen grain is a:
1. Thick and discontinuous layer made of sporopollenin
2. Thick and continuous layer made of sporopollenin
3. Thin and discontinuous layer made of cellulose and pectin
4. Thin and continuous layer made of cellulose and pectin
In the monosporic embryo sac of a flowering plant, the number of nuclei that get surrounded by cell walls and thus are organized into cells is:
1. 5
2. 6
3. 7
4. 8
Female flowers remain submerged in water and the pollen grains are released inside the water in:
1. Sea grasses
2. Vallisneria
3. Water lily
4. Water hyacinth
Scientists are trying to transfer apomictic genes into hybrid varieties of several of our food and vegetable crops so that:
1. They get resistance against common pathogens
2. The yield of these plants can be increased many folds
3. Hybrid seeds need not be produced every year
4. Their germplasm is easily stored in labs for further research
Which human male accessory reproductive duct receives a duct from the seminal vesicle?
1. Rete testis
2. Vas deferens
3. Epididymis
4. Urethra
The primary oocyte completes its first meiotic division within the:
1. Primary follicle
2. Secondary follicle
3. Tertiary follicle
4. Graafian follicle
The human fetus develops limbs and digits:
1. After one month of pregnancy
2. By the end of second month of pregnancy
3. During the fifth month of pregnancy
4. By the end of second trimester of pregnancy
Periodic abstinence is a natural method of contraception where the couples avoid or abstain from coitus during the period of the menstrual cycle when:
1. There is menstrual flow
2. The ovulation is most likely to occur
3. The corpus luteum is most active
4. The proliferative phase begins
In a monohybrid cross F1 progeny resemble neither of the parents. What would be true in this case?
1. The parental traits would not appear in any of the F2 -progenies
2. The F2 phenotypic ratio will be different from the F2 genotypic ratio
3. It could be a case of incomplete dominance
4. The F2 phenotypic ratio will be similar to any Mendelian monohybrid cross
The two alleles of a gene pair are located on:
1. Homologous sites on homologous chromosomes
2. Heterologous sites on homologous chromosomes
3. Homologous sites on heterologous chromosomes
4. Heterologous sites on heretologous chromosomes
Male heterogamety is not seen in:
1. Humans
2. Melandrium album
3. Birds
4. Fruit fly
The trait shown in the given pedigree chart is most likely a/an:
1. Autosomal recessive trait
2. Autosomal dominant trait
3. Sex linked recessive trait
4. Sex linked dominant trait
Aneuploidy results from :
1. Point mutations
2. Gross structural changes in chromosomes
3. Failure of cytokinesis after telophase stage of cell division
4. Failure of segregation of chromatids during cell division
DNA chemically is less reactive and structurally more stable than RNA. Therefore,
1. DNA has evolved from the RNA
2. RNA can directly code for proteins
3. DNA is the better genetic material than RNA
4. The protein synthesizing machinery has evolved around RNA
A transcription unit does not contain:
1. A promoter
2. The structural gene
3. A terminator
4. An operator
Regulation of lac operon by repressor is referred to as:
1. Inducible regulation
2. Repressible regulation
3. Negative regulation
4. Positive regulation
The similarities between the eyes of an octopus and of a mammal are a result of:
| 1. | Convergent evolution | 2. | Divergent evolution |
| 3. | Saltation | 4. | Retrograde evolution |
Which of the following would lead to deviation from Hardy-Weinberg equilibrium?
1. Random mating
2. Lack of mutations
3. No gene flow or gene migration
4. Natural selection
The now extinct reptile group that evolved into mammals was:
1. Therapsids
2. Dinosaurs
3. Thecodants
4. Sauropsids
Identify the correct statement:
| 1. | Dryopithecus was more man like than Ramapithecus |
| 2. | Homo Habilis probably did not eat meat |
| 3. | Homo Erectus had a cranial capacity of around 800 cc |
| 4. | Agriculture came around 18000 years ago |
Which of the following Fungi is not capable of causing ringworm infection in a man?
1. Microsporum
2. Trichophyton
3. Epidermophyton
4. Trichoderma
Opportunistic infections appear in AIDS patients when the HIV has destroyed :
1. Macrophages
2. T cytotoxic cells
3. T helper cells
4. B cells
Proto-oncogenes are seen in :
1. Normal cells
2. Malignant cells
3. Virus infected cells
4. Cells exposed to X-rays
What is incorrect regarding opioids?
| 1. | Their receptors are in CNS and CVS |
| 2. | They are generally taken by snorting and injection |
| 3. | They are CNS depressants |
| 4. | They can be powerful sedatives and painkillers |
Pusa Shubhra, an improved variety of cauliflower, is resistant to:
1. White rust
2. Bacterial blight
3. Black rot
4. Leaf curl
To obtain a virus free plant, what can be used as an explant in micropropogation ?
| 1. | Apical meristem |
| 2. | Axillary meristem |
| 3. | Either apical or axillary meristem |
| 4. | Embryo |
Identify the incorrectly matched pair
|
1. |
Baculoviruses |
Species specific, broad spectrum insecticides |
|
2. |
Trichoderma |
Free living fungi common in root ecosystems |
|
3. |
Ladybird |
Biocontrol of aphids |
|
4. |
Organic farming |
Pests kept at manageable levels rather than completely eradicated |
An important biofertilizer in paddy fields is :
1. Azospirillum
2. Azotobacter
3. Anabaena
4. Rhizobium
What limitation of traditional hybridization used for plant and animal breeding has been overcome by
the use of rDNA techniques?
1. High cost
2. Insertion of undesirable genes
3. Complexity of the procedure
4. Antigenicity of the product
Which of the following restriction sites is located within the gene for tetracycline resistance in the plasmid pBR322?
1. BamHI
2. Psti
3. Clal
4. Pvull
To protect tobacco plants against Meloidegyne incognita using RNAi, nematode specific genes were introduced in the plant using:
1. Transposons
2. Retrotransposons
3. Virus with RNA genome
4. Agrobacterium
A permanent cure for ADA deficiency could be:
| 1. | Enzyme replacement therapy |
| 2. | Bone marrow transplant |
| 3. | Introduction of a functional ADA cDNA into lymphocytes |
| 4. | Introduction of gene isolated from marrow cells producing ADA into early embryonic cells |
The technique not used for transformation of plant cells in recombinant procedures is:
1. Biolistics
2. Agrobacterium mediation
3. Use of viruses
4. Micro-injection
A positive carbylamine test is shown by which one of the following?
1. N, N-diethyl aniline
2. N-methyl propyl amine
3. N, N-diethyl butyl amine
4. 2, 4-dimethyl aniline
In the reaction, N2O4(g) 2NO2(g), is that part of N2O4 which dissociates. The number of moles at equilibrium will be:
1.
2.
3.
4.
XY
Y is:
1. 
3. 4.
A metallic oxide contains 40% oxygen then equivalent weight of metal is
1. 48
2. 36
3. 24
4. 12
Carbon atom in C2(CN)4 are
1. sp hybridised
2. sp2 hybridised
3. sp and sp2 hybridised
4. sp, sp2 and sp3 hybridised
The correct order in which the O-O bond length increases in the following is
1. H2O2 < O2 < O3
2. O3 < H2O < O2
3. O2 < O3< H2O2
4. O2 < H2O2 < O3
Rate of diffusion of LPG (a mixture of n-butane and propane) is 1.25 times of SO3. Hence, mass fraction of n-butane in LPG is
1. 0.75
2. 0.25
3. 0.50
4. 0.67
One mole of methanol when burnt in O2 gives out 723 kJ mol-1 of heat. If one mole of O2 is used, what will be the amount of heat evolved?
1. 482 kJ
2. 241 kJ
3. 723 kJ
4. 924 kJ
In which solution, AgCl has minimum solubility ?
1. 0.05 M AgNO3
2. 0.01 M NaCl
3. Pure Water
4. 0.01 M NH4OH
50 ml of HCl (pH=1) is mixed with another 100 ml of HCl (pH=2) then pH of resulting solution will be approximately
1. 1.2
2. 1.4
3. 1.6
4. 1.8
3Cl2 + 6NaOH 5NaCl + NaClO3 + 3H2O, the equivalent weight of NaOH will be
1. 40
2. 48
3. 60
4. 80
Carbon suboxide (C3O2) has
1. Linear structure
2. Bent structure
3. Trigonal structure
4. Disorted tetrahedral structure
How many geometrical isomers are possible of the following?
CH3-CH=CH-CH=CH-CH3
1. 2 2. 3
3. 4 4. 6
The alkene among the following that has the smallest heat of hydrogenation is-
1.
2.
3.
4.
+ A(Major), the compound 'A' is
(1 mole)
1.
2.
3. Equimolar mixture of (1) and (2)
4. No reaction
In a nitrating mixture (HNO3 + H2SO4), HNO3 acts as
1. Acid
2. Base
3. Acid as well as Base
4. Neither acid nor base
K4[Fe(CN)6] is 60% ionised. Then the value of Van't Hoff factor is
1. 1.6
2. 2.4
3. 3
4. 3.4
The concentration of reactant X decreases from 0.1 M to 0.005 M in 40 minutes. If the reaction follows first order kinetics, the rate of reaction when concentration of X is 0.01 M will be
1. 1.73 x 10-4 M min-1
2. 3.47 x 10-4 M min-1
3. 3.47 x 10-5 M min-1
4. 7.50 x 10-4 M min-1
Mond's process is used for
1. Ni
2. Al
3. Fe
4. Cu
Ammonia on reaction with excess of chlorine gives
1. NCl3 and HCl
2. N2 and NH4Cl
3. NCl3 and NH4Cl
4. N2 and HCl
The number of S-S bonds in the trimer of sulphur trioxide is/are
1. zero
2. 1
3. 2
4. 3
The crystal field stabilisation energy for the high spin d4 octahedral complex is:
1. -1.8 o
2. -1.6 o
3. -1.2 o
4. -0.6 o
Which nitro derivative of haloarene is most reactive towards nucleophilic substitution reaction?
1. 
2.
3.
4.
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Victor Meyer Test is used for the distinguished of 1, 2 and 3
1. Alcohols
2. Alkyl halides
3. Nitro Compounds
4. All of these
(CH3)3C-O-CH3 + HI Products, the product are
1. (CH3)3C-OH + CH3l
2. (CH3)3C-l + CH3OH
3. (CH3)3C-l + CH3l
4. (CH3)3C-OH + CH3OH
CH2=CH-CHO+HCNA(Major),
the compound 'A' in the above mentioned reaction is :
1. CH2=CH-CH-CH(OH)(CN)
2. CH3-CH(CN)-CHO
3. CH2(CN)-CH2-CHO
4. CH3-C(OH)(CN)-CH3
Which compound does not liberate gas with ?
1.
2. 2,4,6- trinitro phenol
3. HCl
4. Phenol
An organic compound X on treatment with acidified K2Cr2O7, gives compound Y which reacts with I2, and NaOH to form CHI3. The compound X can be-
1. CH3OH
2. CH3CHO
3. CH3COCH3
4. CH3CH(OH)CH3
Number of Chiral carbon atoms in cyclic b-D(+)-glucose is
1. 5
2. 4
3. 3
4. 2
Two forces A and B have a resultant . If B is doubled, the new resultant is perpendicular to A. Then
1.
2.
3.
4.
The quantity has dimensional formula same as [This question includes concepts from 12th syllabus]
1.
2.
3.
4. Power
A body of mass (m) elastically collides with another stationary body of mass 3m. The fractional loss of K.E. of the body is:
1.
2.
3.
4.
A body of mass (m) is projected with velocity v0 at an angle of projection . The magnitude of angular momentum about origin after time t from projection is :
1. Increase with time
2. decrease with time
3. independent of time
4. none of these
Bernoulli's theorem is based on :
1. conservation of energy
2. conservation of mass
3. conservation of momentum
4. conservation of angular momentum
Two spherical soap bubbles of radii r1 and r2 in vaccum collapse under isothermal condition. The resulting bubble has radius equal to :
1.
2.
3.
4.
When a block of iron floats in Hg at , a fraction of its volumen= is submerged, while at temperature of a fraction is seen to be submersed. If the coefficient of volume expansion of iron is and that of mercury is , then the ratio can be expressed as:
1.
2.
3.
4.
The mass of 1 liter of He under a pressure 2 atmosphere and temperature is:
1. 0.16 g
2. 0.32 g
3. 0.48 g
5. 0.64 g
mole of monoatomic gas is mixed with mole of diatomic gas such that , then:
1.
2.
3.
4.
Hot water cools from 60 to 50 in first 10 minutes and from 50 to 42 in next 10 minutes. The temperature of surrounding is :
1.
2.
3.
4.
Two simple pendulums have time periods T and . They start vibrating at the same instant from the mean position in the same phase. The phase difference between them when bigger pendulum completes one oscillation will be:
1.
2.
3.
4.
A tuning fork and sonometer give 5 beats per second when the length of the wire is 1 m and 1.05 m respectively. The frequency of fork is -
1. 210 Hz
2. 205 Hz
3. 410 Hz
4. 420 Hz
Two identical metal plates are given charges respectively. If they are now brought close to form a parallel plate capacitor with capacitance , the potential difference between them is :
1.
2.
3.
4.
Three copper wires have their lengths in the ratio 5:3:1 and their masses are in the ratio 1:3:5. Their electrical resistance will be in the ratio of :
1. 5:3:1
2. 1:3:5
3. 125:15:1
4. 1:15:125
The equivalent resistance between \(A\) and \(B\) is:

1. \(3~\Omega\)
2. \(6~\Omega\)
3. \(9~\Omega\)
4. \(12~\Omega\)
| 1. | \(B\) acts along the \(x\text-\)axis |
| 2. | \(B\) acts along the \(y\text-\)axis |
| 3. | \(B\) acts along the \(z\text-\)axis |
| 4. | \(B\) can act along any of the above direction for the net force to be zero |
1.
2.
3.
4. None of these
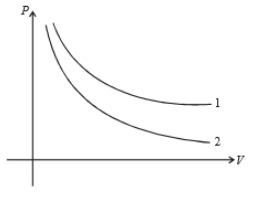

P-V graph of two gases during adiabatic processes are shown in figure. Plots 1 and 2 should correspond, respectively to :
1. He,
2. and He
3. He and Ar
4. and
Density (D) of nucleus related to mass no. (A) as
1.
2.
3.
4.
In a common-emitter transistor, the phase difference between input and output is:
1. Zero
2. /2
3. /3
4.

1. \(V=V_L-V_C\)
2. \(V=\sqrt{V_R^2+V_L^2}\)
3. \(V=\sqrt{V_R^2+V_L^2-V_C^2}\)
4. zero
Heavier nuclei are unstable. The correct statement is :
1. binding energy per nucleon is small
2. get stability by nuclear fission
3. get stability by nuclear fusion
4. both (1) and (2)
The minimum distance between two tetrahedral voids is (a = cell edge length)
1. a
2
2. a
√2
3. a
√3
4. √3 a
2
What is the current efficiency of an electrode deposition of Cu metal from CuSO4 solution in which 9.8 gm copper is deposited by the passage of 5 amperes current for 2 hours?
1. 41.4 %
2. 50%
3. 75%
4. 82.8 %
A small square loop of wire of side l is placed inside a large square of wire of side L (L>>l). The loops are co-planar and their centres coincide. The mutual inductance of the system is proportional to :
1. l/L
2.
3. L/l
4.
The sun (diameter D) subtends an angle radian at the pole of a concave mirror of focal length f. The diameter of the image of the sun formed by the mirror is:
1.
2.
3.
4.
When neutron moving with Kinetic Energy 2eV collides with
stationary in the ground state, the collision will be:
1. Must be elastic
2. may be inelastic
3. Both
4. Must be perfectly inelastic
The potential difference applied to an X-ray tube is 5 kV and current through it is 3.2 mA. The number of electrons striking the target per sec is:
1.
2.
3.
4.
| 1. | \(\frac{m_1}{m_2} \) | 2. | \(\frac{m_2}{m_1} \) |
| 3. | \(1 \) | 4. | \(\sqrt{\frac{\mathrm{m}_2}{\mathrm{~m}_1}}\) |
In a E.M.W, phase difference between and is :
1. zero
2.
3.
4.
The focal length of the plano-convex lens is 20 cm. If its plane surface is silvered, then
1. it behaves as a concave mirror of focal length 10 cm
2. it behaves as a convex mirror of focal length of 10 cm
3. it behaves as a concave mirror of focal length 20 cm
4. none of these
The forces required to just move a body up an inclined plane is double the force required to just prevent it from sliding down. If is angle of friction and is the angle which the plane makes with horizontal, then
1.
2.
3.
4.