Which of the following refers to correct example(s) of organisms that have evolved due to changes in environment brought about by anthropogenic action?
| (a) | Darwin's Finches of Galapagos islands |
| (b) | Herbicide-resistant weeds |
| (c) | Drug-resistant eukaryotes |
| (d) | Man-created breeds of domesticated animals like dogs |
1. (a) and (c)
2. (b), (c) and (d)
3. only (d)
4. only (a)
Meiotic division of the secondary oocyte is completed:
1. At the time of copulation
2. After zygote formation
3. At the time of fusion of a sperm with an ovum
4. Prior to ovulation
Which of the following is correct about viroids?
1. They have free RNA without protein coat.
2. They have DNA with protein coat.
3. They have free DNA without protein coat.
4. They have RNA with protein coat.
The plant parts which consist of two generations, one within the other, are:
| (a) | Pollen grains inside the anther |
| (b) | Germinated pollen grain with two male gametes |
| (c) | Seed inside the fruit |
| (d) | Embryo sac inside the ovule |
1. (a), (b) and (c)
2. (c) and (d)
3. (a) and (d)
4. (a) only
Experimental verification of the chromosomal theory of inheritance was done by:
| 1. | Sutton | 2. | Boveri |
| 3. | Morgan | 4. | Mendel |
Which of the following pairs is of unicellular algae?
1. Gelidium and Gracilaria
2. Anabaena and Volvox
3. Chlorella and Spirulina
4. Laminaria and Sargassum
Secondary metabolites such as nicotine, strychnine and caffeine are produced by plants for their:
1. Growth response
2. Defence action
3. Effect on reproduction
4. Nutritive value
By which method was a new breed 'Hisardale' of sheep found by using Bikaneri ewes and Marina rams?
1. Mutational breeding
2. Crossbreeding
3. Inbreeding
4. Outcrossing
The infectious stage of plasmodium that enters the human body is:
| 1. | Sporozoites
|
2. | Female gametocytes
|
| 3. | Male gametocytes
|
4. | Trophozoites |
The process responsible for facilitating loss of water in liquid form from the tip of grass blades at night and early morning is:
1. Root pressure
2. Imbibition
3. Plasmolysis
4. Transpiration
From his experiments, S.L. Miller produced amino acids by mixing which of the following in a closed flask:
1. and water vapour at
2. and water vapour at
3. and water vapour at
4. and water vapour at
In relation to Gross primary productivity and Net primary productivity of an ecosystem, which one of the following statements is correct?
| 1. | Gross primary productivity is always more than Net primary productivity. |
| 2. | Gross primary productivity and Net primary productivity are one and the same. |
| 3. | There is no relationship between Gross primary productivity and Net primary productivity. |
| 4. | Gross primary productivity is always less than net primary productivity. |
| 1. | Ori site | 2. | Palindromic sequence |
| 3. | Recognition site | 4. | Selectable marker |
Cuboidal epithelium with brush border of microvilli is found in :
1. Ducts of salivary glands
2. Proximal convoluted tubule of nephron
3. Eustachian tube
4. Lining of intestine
The body of the ovule is fused within the funicle at :
| 1. | Micropyle | 2. | Nucellus |
| 3. | Chalaza | 4. | Hilum |
In light reaction, plastoquinone facilitates the transfer of electrons from:
| 1. | Cytb6f complex to PS I | 2. | PS I to NADP+ |
| 3. | PS I to ATP synthase | 4. | PS II to Cytb6f complex |
Match the following diseases with the causative organism and select the correct option.
| Column-I | Column-II | ||
| (a) | Typhoid | (i) | Wuchereria |
| (b) | Pneumonia | (ii) | Plasmodium |
| (c) | Filariasis | (iii) | Salmonella |
| (d) | Malaria | (iv) | Haemophilus |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (iv) | (i) | (ii) |
| 2. | (ii) | (i) | (iii) | (iv) |
| 3. | (iv) | (i) | (ii) | (iii) |
| 4. | (i) | (iii) | (ii) | (iv) |
Match the following microorganism with product
| Column - I | Column - II | ||
| (a) | Clostridium butylicum | (i) | Cyclosporin A |
| (b) | Trichoderma polysporunt | (ii) | Butyric acid |
| (c) | Monascus purpureus | (iii) | Citric acid |
| (d) | Aspergillus niger | (iv) | Blood cholesterol lowering agent |
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (i) | (iv) | (iii) |
| 2. | (i) | (ii) | (iv) | (iii) |
| 3. | (iv) | (iii) | (ii) | (i) |
| 4. | (iii) | (iv) | (ii) | (i) |
Which of the following statements are true for the phylum-Chordata?
| (a) | In Urochordata, notochord extends from head to tail and is present throughout their life |
| (b) | In Vertebrata, notochord is present during the embryonic period only |
| (c) | Central nervous system is dorsal and hollow |
| (d) | Chordata is divided into 3 sub-phyla : Hemichordata, Tunicata and Cephalochordata |
Goblet cells of alimentary canal are modified from:
| 1. | Columnar epithelial cells | 2. | Chondrocytes |
| 3. | Compound epithelial cells | 4. | Squamous epithelial cells |
Which of the following is not an inhibitory substance governing seed dormancy?
1. Abscisic acid
2. Phenolic acid
3. Para-ascorbic acid
4. Gibberellic acid
Name the enzyme that facilitates opening of DNA helix during transcription.
| 1. | DNA helicase | 2. | DNA polymerase |
| 3. | RNA polymerase | 4. | DNA ligase |
Match the following:
| (a) | Inhibitor of catalytic activity of succinate dehydrogenase | (i) | Ricin |
| (b) | Possess peptide bonds | (ii) | Malonate |
| (c) | Cell wall material in fungi | (iii) | Chitin |
| (d) | Secondary metabolite | (iv) | Collagen |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (i) | (iv) | (ii) |
| 2. | (iii) | (iv) | (i) | (ii) |
| 3. | (ii) | (iii) | (i) | (iv) |
| 4. | (ii) | (iv) | (iii) | (i) |
Bilaterally symmetrical and acoelomate animals are exemplified by:
| 1. | Platyhelminthes | 2. | Aschelminthes |
| 3. | Annelida | 4. | Ctenophora |
Presence of which of the following conditions in urine is indicative of Diabetes mellitus:
1. Uremia and Renal Calculi
2. Ketonuria and Glycosuria
3. Renal Calculi and Hyperglycaemia
4. Uremia and Ketonuria
Ray florets have:
| 1. | Superior ovary | 2. | Hypogynous ovary |
| 3. | Half-inferior ovary | 4. | Inferior ovary |
Identify the substances having a glycosidic bond and peptide bond, respectively in their structure:
1. Glycerol, trypsin
2. Cellulose, lecithin
3. Inulin, insulin
4. Chitin, cholesterol
Which of the following statements is not correct?
| 1. | The proinsulin has an extra peptide called C-peptide |
| 2. | Functional insulin has A and B chains linked together by hydrogen bonds. |
| 3. | Genetically engineered insulin is produced in E. Coli. |
| 4. | In man, insulin is synthesized as proinsulin. |
Some dividing cells exit the cell cycle and enter the vegetative inactive stage. This is called the quiescent stage . This process occurs at the end of:
1.
2. S phase
3.
4. M phase
Identify the correct statement with regard to phase (Gap 1) of interphase:
| 1. | The reorganisation of all cell components takes place. |
| 2. | The cell is metabolically active and grows but does not replicate its DNA |
| 3. | Nuclear division takes place |
| 4. | DNA synthesis or replication takes place. |
The QRS complex in a standard ECG represents:
| 1. | Depolarisation of auricles | 2. | Depolarisation of ventricles |
| 3. | Repolarisation of ventricles | 4. | Repolarisation of auricles |
If the distance between two consecutive base pairs is 0.34 nm and the total number of base pairs of a DNA double helix in a typical mammalian cell is bp, then the length of the DNA is approximately:
| 1. | 2.5 meters | 2. | 2.2 meters |
| 3. | 2.7 meters | 4. | 2.0 meters |
Which of the following regions of the globe exhibits the highest species diversity?
| 1. | Madagascar | 2. | Himalayas |
| 3. | Amazon forests | 4. | Western Ghats of India |
Which of the following is put into anaerobic sludge digester for further sewage treatment?
| 1. | Floating debris | 2. | Effluents of primary treatment |
| 3. | Activated sludge | 4. | Primary sludge |
Dissolution of the synaptonemal complex occurs during :
| 1. | Zygote | 2. | Diplotene |
| 3. | Leptotene | 4. | Pachytene |
Select the option including all sexually transmitted diseases:
1. Gonorrhoea, Malaria, Genital herpes
2. AIDS, Malaria, Filaria
3. Cancer, AIDS, Syphilis
4. Gonorrhoea, Syphilis, Genital herpes
Select the correct statement.
1. Glucagon is associated with hypoglycemia.
2. Insulin acts on pancreatic cells and adipocytes.
3. Insulin is associated with hyperglycemia.
4. Glucocorticoids stimulate gluconeogenesis.
The product(s) of the reaction catalyzed by nitrogenase in root nodules of leguminous plants is/are.
1. Nitrate alone
2. Ammonia and oxygen
3. Ammonia and hydrogen
4. Ammonia alone
In-gel electrophoresis, separated DNA fragments can be visualized with the help of:
1. Ethidium bromide in UV radiation
2. Acetocarmine in UV radiation
3. Ethidium bromide in infrared radiation
4. Acetocarmine in bright blue light
In Which of the following techniques, the embryos are transferred to assist those females who cannot conceive?
| 1. | GIFT and ZIFT | 2. | ICSI and ZIFT |
| 3. | GIFT and ICSI | 4. | ZIFT and IUT |
Select the correct match:
| 1. | Phenylketonuria | Autosomal dominant trait |
| 2. | Sickle cell anemia | Autosomal recessive trait chromosome - 11 |
| 3. | Thalassemia | X linked |
| 4. | Haemophilia | Y linked |
Which of the following is not an attribute of a population?
1. Natality
2. Mortality
3. Species interaction
4. Sex ratio
The oxygenation activity of RuBisCo enzyme in photorespiration leads to the formation of:
| 1. | 1 molecule of the 3-C compound |
| 2. | 1 molecule of the 6-C compound |
| 3. | 1 molecule of a 4-C compound and 1 molecule of the 2-C compound |
| 4. | 2 molecules of the 3-C compound |
Match the following concerning essential elements and their functions in plants:
| Column I | Column II | ||
| (a) | Iron | (i) | Photolysis of water |
| (b) | Zinc | (ii) | Pollen germination |
| (c) | Boron | (iii) | Required for chlorophyll biosynthesis |
| (d) | Manganese | (iv) | IAA biosynthesis |
| (a) | (b) | (c) | (d) | |
| 1. | (iv) | (iii) | (ii) | (i) |
| 2. | (iii) | (iv) | (ii) | (i) |
| 3. | (iv) | (i) | (ii) | (iii) |
| 4. | (ii) | (i) | (iv) | (iii) |
Which is the important site of formation of glycoproteins and glycolipids in eukaryotic cells?
| 1. | Peroxisomes | 2. | Golgi bodies |
| 3. | Polysomes | 4. | Endoplasmic reticulum |
Select the correct events that occur during inspiration.
(a) Contraction of diaphragm
(b) Contraction of external inter-costal muscles
(c) Pulmonary volume decreases
(d) Intra pulmonary pressure increases
| 1. | (c) and (d) | 2. | (a), (b) and (d) |
| 3. | only (d) | 4. | (a) and (b) |
The roots that originate from the base of the stem are:
| 1. | Primary roots | 2. | Prop roots |
| 3. | Lateral roots | 4. | Fibrous roots |
The ovary is half inferior in:
1. Mustard
2. Sunflower
3. Plum
4. Brinjal
Match the following columns and select the correct option.
| Column I | Column-II | ||
| (a) | Floating Ribs | (i) | Located between second and seventh ribs |
| (b) | Acromion | (ii) | Head of the Humerus |
| (c) | Scapula | (iii) | Clavicle |
| (d) | Glenoid cavity | (iv) | Do not connect with the sternum |
| (a) | (b) | (c) | (d) | |
| 1. | (i) | (iii) | (ii) | (iv) |
| 2. | (iii) | (ii) | (iv) | (i) |
| 3. | (iv) | (iii) | (i) | (ii) |
| 4. | (iv) | (ii) | (i) | (iii) |
If the head of the cockroach is removed, it may live for a few days because:
| 1. | The cockroach does not have a nervous system |
| 2. | The head holds a small proportion of the nervous system while the rest is situated along the ventral part of its body |
| 3. | The head holds 1/3rd of the nervous system while the rest is situated along the dorsal part of its body |
| 4. | The supra-oesophageal ganglia of the cockroach are situated in ventral part of abdomen |
Identify the incorrect statement.
| 1. | Sapwood is involved in conduction of water and minerals from roots to leaves. |
| 2. | Sapwood is the innermost secondary xylem and is lighter in colour |
| 3. | Due to deposition of tannins, resins, oils etc., heartwood is dark in colour. |
| 4. | Heart wood does not conduct water but gives mechanical support. |
Bt cotton variety that was developed by the introduction of toxin gene of Bacillus thuringiensis (BT) is resistant to:
| 1. | Fungal diseases | 2. | Plant nematodes |
| 3. | Insect predators | 4. | Insect pests |
The number of substrate level of phosphorylations in one turn of citric acid cycle is:
| 1. | One | 2. | Two |
| 3. | Three | 4. | Four |
Identify the wrong statement with regards to restriction enzyme:
| 1. | They cut the strand at palindromic sites. |
| 2. | They are useful in genetic engineering. |
| 3. | Sticky ends can be joined by using DNA ligases. |
| 4. | Each restriction enzyme functions by inspecting the length of a DNA sequence. |
Flippers of Penguins and Dolphins are examples of:
| 1. | Convergent evolution | 2. | Industrial melanism |
| 3. | Natural selection | 4. | Adaptive radiation |
Identify the wrong statement with reference to transport of oxygen:
| 1. | Partial pressure of CO2 can interfere with O2 binding with haemoglobin. |
| 2. | Higher H+ concentration in alveoli favours the formation of oxyhaemoglobin. |
| 3. | Low pCO2 in alveoli favours the formation of oxyhaemoglobin. |
| 4. | Binding of oxygen with haemoglobin is mainly related to partial pressure of O2. |
Identify the wrong statement with reference to the gene 'I' that controls ABO blood groups.
| 1. | A person will have only two of the 3 alleles. |
| 2. | When IA and IB are present together, they express same type of sugar. |
| 3. | Allele 'i' does not produce any sugar. |
| 4. | The gene (I) has three alleles. |
Identify the basic amino acid from the following.
| 1. | Glutamic Acid | 2. | Lysine |
| 3. | Valine | 4. | Tyrosine |
Name the plant growth regulator which upon spraying on sugarcane crop, increases the length of stem, thus increasing the yield of sugarcane crop.
1. Gibberellin
2. Ethylene
3. Abscisic acid
4. Cytokinin
Match the organism with its use in biotechnology.
| (a) | Bacillus thuringiensis | (i) | Cloning vector |
| (b) | Thermus aquaticus | (ii) | Construction of first rDNA molecule |
| (c) | Agrobacterium tumefaciens | (iii) | DNA polymerase |
| (d) | Salmonella typhimurium | (iv) | Cry proteins |
Select the correct option from the following:
| (a) | (b) | (c) | (d) | |
| 1. | (iv) | (iii) | (i) | (ii) |
| 2. | (iii) | (ii) | (iv) | (i) |
| 3. | (iii) | (iv) | (i) | (ii) |
| 4. | (ii) | (iv) | (iii) | (i) |
Which of the following statements is correct?
1. Adenine pairs with thymine through one H-bond.
2. Adenine pairs with thymine through three H-bonds.
3. Adenine does not pair with thymine.
4. Adenine pairs with thymine through two H-bonds.
Match the following columns and select the correct option:
| Column-I | Column-II | ||
| (a) | Gregarious, polyphagous pest | (i) | Asterias |
| (b) | Adult with radial symmetry and larva with bilateral symmetry | (ii) | Scorpion |
| (c) | Book lungs | (iii) | Ctenoplana |
| (d) | Bioluminescence | (iv) | Locusta |
| (a) | (b) | (c) | (d) | |
| 1. | (iv) | (i) | (ii) | (iii) |
| 2. | (iii) | (ii) | (i) | (iv) |
| 3. | (ii) | (i) | (iii) | (iv) |
| 4. | (i) | (iii) | (ii) | (iv) |
Which of the following would help in prevention of diuresis?
| 1. | Reabsorption of Na+ and water from renal tubules due to aldosterone |
| 2. | Atrial natriuretic factor causes vasoconstriction |
| 3. | Decrease in the secretion of renin by JG cells |
| 4. | More water reabsorption due to under secretion of ADH |
Choose the correct pair from the following :
| 1. | Polymerases | - | Break the DNA into fragments |
| 2. | Nucleases | - | Separate the two strands of DNA |
| 3. | Exonucleases | - | Make cuts at specific positions within DNA |
| 4. | Ligases | - | Join the two DNA molecules |
Identify the correct statement with reference to human digestive system
1. Serosa is the innermost layer of the alimentary canal.
2. Ileum is a highly coiled part.
3. Vermiform appendix arises from duodenum.
4. Ileum opens into small intestine.
Ernst Haeckel's embryological support for evolution was disapproved by:
| 1. | Alfred Wallace | 2. | Charles Darwin |
| 3. | Oparin | 4. | Karl Ernst von Baer |
Which of the following hormone levels will cause release of ovum (ovulation) from the Graafian follicle?
1. High concentration of Progesterone
2. Low concentration of LH
3. Low concentration of FSH
4. High concentration of estrogen
The specific palindromic sequence which is recognized by EcoRI is:
| 1. | 5' -GGAACC-3' 3'-CCTTGG-5' |
2. | 5'-CTTAAG-3' 3'-GAATTC-5' |
| 3. | 5'-GGATCC-3' 3'-CCTAGG-5' |
4. | 5'-GAATTC-3' 3'-CTTAAG-5' |
The first phase of translation is:
1. Recognition of DNA molecule
2. Aminoacylation of tRNA
3. Recognition of an anti-codon
4. Binding of mRNA to ribosome
Floridian starch has structure similar to :
1. Amylopectin and glycogen
2. Mannitol and algin
3. Laminarin and cellulose
4. Starch and cellulose
Strobili or cones are found in:
| 1. | Pteris | 2. | Marchantia |
| 3. | Equisetum | 4. | Salvinia |
How many true-breeding pea plant varieties did Mendel select as pairs, which were similar except in one character with contrasting traits?
| 1. | 2 | 2. | 14 |
| 3. | 6 | 4. | 4 |
Snow-blindness in Antarctic regions is due to:
| 1. | Inflammation of cornea due to high doses of UV-B radiation |
| 2. | High reflection of light from snow |
| 3. | Damage to the retina caused by infra-red rays |
| 4. | Freezing of fluids in the eye because of the low temperature |
The enzyme enterokinase helps in the conversion of
1. trypsinogen into trypsin
2. caseinogen into casein
3. pepsinogen into pepsin
4. protein into polypeptides
Match the following with respect to meiosis:
| Column-I | Column-II | ||
| (a) | Zygotene | (i) | Terminalization |
| (b) | Pachytene | (ii) | Chiasmata |
| (c) | Diplotene | (iii) | Crossing over |
| (d) | Diakinesis | (iv) | Synapsis |
Select the correct option from the following:
| Options: | (a) | (b) | (c) | (d) |
| 1. | iv | iii | ii | i |
| 2. | i | ii | iv | iii |
| 3. | ii | iv | iii | i |
| 4. | iii | iv | i | ii |
Which of the following statements about inclusion bodies is incorrect?
| 1. | These are involved in the ingestion of food particles. |
| 2. | They lie freely in the cytoplasm. |
| 3. | These represent reserve material in cytoplasm. |
| 4. | They are not bound by any membrane. |
Match the following columns and select the correct option:
| Column-I | Column-I | ||
| (a) | Eosinophils | (i) | Immune response |
| (b) | Basophils | (ii) | Phagocytosis |
| (c) | Neutrophils | (iii) | Acidophils |
| (d) | Lymphocytes | (iv) | Release granules containing histamine |
| (a) | (b) | (c) | (d) | |
| 1. | (iv) | (i) | (ii) | (iii) |
| 2. | (i) | (ii) | (iv) | (iii) |
| 3. | (ii) | (i) | (iii) | (iv) |
| 4. | (iii) | (iv) | (ii) | (i) |
The transverse section of a plant shows following anatomical features:
| a. | Large number of scattered vascular bundles surrounded by bundle sheath. |
| b. | Large conspicuous parenchymatous ground tissue. |
| c. | Vascular bundles conjoint and closed. |
| d. | Phloem parenchyma absent. |
Identify the category of plant and its part :
1. Monocotyledonous root
2. Dicotyledonous stem
3. Dicotyledonous root
4. Monocotyledonous stem
Match the following columns and select the correct option:
| Column-I | Column-II | ||
| (a) | Pituitary gland | (i) | Grave's disease |
| (b) | Thyroid gland | (ii) | Diabetes mellitus |
| (c) | Adrenal gland | (iii) | Diabetes insipidus |
| (d) | Pancreas | (iv) | Addison's disease |
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iii) | (ii) | (i) | (iv) |
| 2. | (iii) | (i) | (iv) | (ii) |
| 3. | (ii) | (i) | (iv) | (iii) |
| 4. | (iv) | (iii) | (i) | (ii) |
Match the following columns and select the correct option.
| Column-I | Column-II | ||
| a. | Placenta | (i) | Androgens |
| b. | Zona pellucida | (ii) | Human chronic Gonadotropin |
| c. | Bulbo-urethral glands | (iii) | Layer of the ovum |
| d. | Leydig cells | (iv) | Lubrication of the Penis |
| a | b | c | d | |
| 1. | (i) | (iv) | (ii) | (iii) |
| 2. | (iii) | (ii) | (iv) | (i) |
| 3. | (ii) | (iii) | (iv) | (i) |
| 4. | (ii) | (iv) | (i) | (ii) |
In water hyacinth and water lily, pollination takes place by:
1. Water currents only
2. Wind and water
3. Insects and water
4. Insects or wind
According to Robert May, global species diversity is about:
1. 20 Million
2. 50 Million
3. 7 Million
4. 1.5 Million
Match the following columns and select the correct option.
| Column I | Column II | ||
| (a) | 6 - 15 pairs of gill slits | (i) | Trygon |
| (b) | Heterocercal caudal fin | (ii) | Cyclostomes |
| (c) | Air Bladder | (iii) | Chondrichthyes |
| (d) | Poison sting | (iv) | Osteichthyes |
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iii) | (iv) | (i) | (ii) |
| 2. | (iv) | (ii) | (iii) | (i) |
| 3. | (i) | (iv) | (iii) | (ii) |
| 4. | (ii) | (iii) | (iv) | (i) |
The process of growth is maximum during:
1. Lag phase
2. Senescence
3. Dormancy
4. Log phase
Match the following columns and select the correct option.
| Column-I | Column-II | ||
| a. | Bt cotton | (i) | Gene therapy |
| b. | Adenosine deaminase deficiency | (ii) | Cellular defense |
| c. | RNAi | (iii) | Detection of HIV infection |
| d. | PCR | (iv) | Bacillus thuringiensis |
| a | b | c | d | |
| 1. | (iii) | (ii) | (i) | (iv) |
| 2. | (ii) | (iii) | (iv) | (i) |
| 3. | (i) | (ii) | (iii) | (iv) |
| 4. | (iv) | (i) | (ii) | (iii) |
Match the following columns and select the correct option
| Column-I | Column-II | ||
| a. | The organ of Corti | (i) | Connects the middle ear and pharynx |
| b. | Cochlea | (ii) | Coiled part of the labyrinth |
| c. | Eustachian tube | (iii) | Attached to the oval window |
| d. | Stapes | (iv) | Located on the basilar membrane |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (i) | (iv) | (ii) |
| 2. | (iv) | (ii) | (i) | (iii) |
| 3. | (i) | (ii) | (iv) | (iii) |
| 4. | (ii) | (i) | (i) | (iv) |
Which one of the following is the most abundant protein in animals?
| 1. | Collagen
|
2. | Lectin
|
| 3. | Insulin
|
4. | Hemoglobin |
Identify the wrong statement with a reference to immunity.
| 1. | When ready-made antibodies are directly given, it is called “Passive immunity". |
| 2. | Active immunity is quick and gives a full response. |
| 3. | Foetus receives some antibodies from the mother, it is an example of passive immunity |
| 4. | When exposed to antigen(living or dead) antibodies are produced in the host's body. It is called "Active immunity |
Montreal Protocol was signed in 1987 for the control of:
| 1. | Emission of ozone-depleting substances |
| 2. | Release of Green House gases |
| 3. | Disposal of e-waste |
| 4. | Transport of genetically modified organisms from one country to another |
Match the trophic levels with their correct species examples in the green land ecosystem
| Column-I | Column-II | ||
| (a) | Fourth trophic level | (i) | Cow |
| (b) | Second trophic level | (ii) | Vulture |
| (c) | First trophic level | (iii) | Rabbit |
| (d) | Third trophic level | (iv) | Grass |
Select the correct option:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iii) | (ii) | (i) | (iv) |
| 2. | (iv) | (iii) | (ii) | (i) |
| 3. | (i) | (ii) | (iii) | (iv) |
| 4. | (ii) | (iii) | (iv) | (i) |
1. Benzene + Toluene
2. Acetone + Chloroform
3. Chloroethane + Bromoethane
4. Ethanol + Acetone
The incorrect statement about carbon monoxide is-
| 1. | It reduces the oxygen-carrying ability of blood. |
| 2. | The carboxyhaemoglobin (haemoglobin bound to CO) is less stable than oxyhaemoglobin. |
| 3. | It is produced due to incomplete combustion. |
| 4. | It forms carboxyhaemoglobin. |
The number of Faradays (F) required to produce 20 g of calcium from molten CaCl2 (Atomic mass of Ca = 40 g mol–1) is:
1. 2
2. 3
3. 4
4. 1
Hydrolysis of sucrose is given by the following reaction
Sucrose + H2O Glucose + Fructose
If the equilibrium constant (Kc) is 21013 at 300 K, the value of at the same temperature will be:
1. 8.314 J mol–1 K–1300 Kln (21013)
2. 8.314 J mol–1 K–1300 Kln (31013)
3. –8.314 J mol–1 K–1300 Kln (41013)
4. –8.314 J mol–1 K–1300 Kln (21013)
| 1. | \(\Delta_{\mathrm{r}} \mathrm{H}>0\) and \(\Delta_{\mathrm{r}} \mathrm{S}<0 \) | 2. | \(\Delta_{\mathrm{r}} \mathrm{H}<0\) and \( \Delta_{\mathrm{r}} \mathrm{S}>0 \) |
| 3. | \(\Delta_{\mathrm{r}} \mathrm{H}<0 \) and \(\Delta_{\mathrm{r}} \mathrm{S}<0 \) | 4. | \(\Delta_{\mathrm{r}} \mathrm{H}>0\) and \( \Delta_{\mathrm{r}} \mathrm{S}>0\) |
Paper chromatography is an example of:
1. Partition chromatography
2. Thin layer chromatography
3. Column chromatography
4. Adsorption chromatography
The rate constant for a first order reaction is . The time required to reduce 2.0 g of the reactant to 0.2 g is:
| 1. | 200 s | 2. | 500 s |
| 3. | 1000 s | 4. | 100 s |
Compound of sulphur that has linkage among the following is:
1.
2.
3.
4.
The reaction between benzaldehyde and acetophenone in the presence of dilute NaOH is known as:
1. Cannizzaro's reaction
2. Cross Cannizzaro's reaction
3. Cross aldol condensation
4. Aldol condensation
An element has a body-centered cubic (BCC) structure with a cell edge of 288 pm. The atomic radius is:
1. pm
2. pm
3. pm
4. pm
Which of the following is a cationic detergent?
1. Sodium stearate
2. Cetyl trimethyl ammonium bromide
3. Sodium dodecylbenzene sulphonate
4. Sodium lauryl sulfate
The calculated spin only magnetic moment of Cr2+ ion is:
1. 4.90 BM
2. 5.92 BM
3. 2.84 BM
4. 3.87 BM
HCl was passed through a solution of CaCl2, MgCl2, and NaCl. Compound(s) that get crystallize is/are-
1. Only NaCl
2. Only MgCl2
3. NaCl, MgCl2, and CaCl2
4. Both MgCl2 and CaCl2
Match the following and identify the correct option
| (a) CO(g)+H2(g) | (i) Mg(HCO3)2+Ca(HCO3)2 |
| (b) Temporary hardness of water | (ii) An electron deficient hydride |
| (c) B2H6 | (iii) Synthesis gas |
| (d) H2O2 | (iv) Non-planar structure |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (ii) | (i) | (iv) |
| 2. | (iii) | (iv) | (ii) | (i) |
| 3. | (i) | (iii) | (ii) | (iv) |
| 4. | (iii) | (i) | (ii) | (iv) |
Which of the following statement(s) is/are correct about the elimination reaction of 2-Bromopentane to form pent-2-ene:
(a) -Elimination reaction
(b) Follows Zaitsev rule
(c) Dehydrohalogenation reaction
(d) Dehydration reaction
| 1. | (a), (c), (d) | 2. | (b), (c), (d) |
| 3. | (a), (b), (d) | 4. | (a), (b), (c) |
The correct order of increasing field strength of ligands is:
1.
2.
3.
4.
The correct statement among the following is:
| 1. | Blister copper has a blistered appearance due to the evolution of CO2. |
| 2. | Vapour phase refining is carried out for nickel by the Van Arkel method. |
| 3. | Pig iron can be moulded into a variety of shapes. |
| 4. | Wrought iron is impure iron with 4 % carbon. |
Sucrose on hydrolysis gives:
1. -D-Glucose + -D-Glucose
2. -D-Glucose + -D-Fructose
3. -D-Fructose + -D-Fructose
4. -D-Glucose + -D-Fructose
What is the alteration in the oxidation state of carbon in the given reaction?
\(\mathrm{{CH_4}_{(g)} + 4{Cl_2}_{(g)} \rightarrow {CCl_4}_{(l)} + 4 HCl_{(g)}}\)
| 1. | 0 to +4 | 2. | –4 to +4 |
| 3. | 0 to –4 | 4. | +4 to +4 |
Which of the following metal ions activates many enzymes participating in the oxidation of glucose to produce ATP and with Na it is also responsible for the transmission of nerve signals:
1. Copper
2. Calcium
3. Potassium
4. Iron
Which of the following alkane cannot be made in good yield by Wurtz reaction?
| 1. | 2,3-Dimethylbutane | 2. | n-Heptane |
| 3. | n-Butane | 4. | n-Hexane |
Measuring Zeta potential is useful in determining which of the following property of a colloidal solution:
1. Solubility.
2. Stability of the colloidal particles.
3. Size of the colloidal particles.
4. Viscosity.
The freezing point of depression constant (Kf ) of benzene is 5.12 K kg mol–1. The freezing point depression for the solution of molality 0.078 m containing a non-electrolyte solute in benzene is:
1. 0.80 K
2. 0.40 K
3. 0.60 K
4. 0.20 K
Which amine gives the carbylamine test?
| 1. | 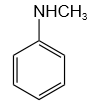 |
2. |  |
| 3. |  |
4. |  |
The natural polymer among the following options is:
1. Poly(Butadiene-styrene)
2. Polybutadiene
3. Poly(Butadiene-acrylonitrile)
4. Cis-1,4-polyisoprene
Identify the incorrect statement.
| 1. | The transition metals and their compounds are known for their catalytic activity due to their ability to adopt multiple oxidation states and to form complexes. |
| 2. | Interstitial compounds are those that are formed when small atoms like H, C or N are trapped inside the crystal lattices of metals. |
| 3. | The oxidation states of chromium in CrO42– and Cr2O72– are not the same. |
| 4. | Cr2+(d4) is a stronger reducing agent than Fe2+(d6) in water. |
Which of the following set of molecules will have zero dipole moment?
| 1. | Boron trifluoride, hydrogen fluoride, carbon dioxide, 1 3-dichlorobenzene |
| 2. | Nitrogen trifluoride, beryllium difluoride, water, 1 3 -dichlorobenzene |
| 3. | Boron trifluoride, beryllium difluoride, carbon dioxide, 1 4-dichlorobenzene |
| 4. | Ammonia, beryllium difluoride, water, 1, 4-dichlorobenzene |
On electrolysis of dilute sulphuric acid using Platinum (Pt) electrode, the product obtained at the anode will be:
1. Oxygen gas
2. gas
3. gas
4. Hydrogen gas
Which of the following is produced when anisole reacts with HI?
| 1. | 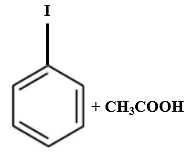 |
2. | 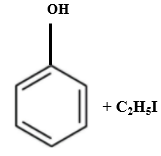 |
| 3. | 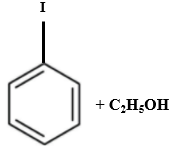 |
4. | 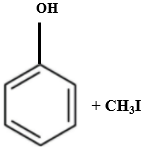 |
The number of protons, neutrons and electrons in \( _{71}^{175}Lu\) respectively, are:
1. 104, 71 and 71
2. 71, 71 and 104
3. 175, 104 and 71
4. 71, 104 and 71
Match the following:
| Oxide | Nature | ||
| (a) | CO | (i) | Basic |
| (b) | BaO | (ii) | Neutral |
| (c) | Al2O3 | (iii) | Acidic |
| (d) | Cl2O7 | (iv) | Amphoteric |
Which of the following is the correct option?
| a | b | c | d | |
| 1. | (ii) | (i) | (iv) | (iii) |
| 2. | (iii) | (iv) | (i) | (ii) |
| 3. | (iv) | (iii) | (ii) | (i) |
| 4. | (i) | (ii) | (iii) | (iv) |
A tertiary butyl carbocation is more stable than a secondary butyl carbocation because-
1. + R effect of groups
2. R effect of CH3 groups
3. Hyperconjugation
4. l effect of CH3 groups
Which of the following contains the maximum number of atoms ?
| 1. | 1 g of Mg(s) | 2. | 1 g of O2 (g) |
| 3. | 1 g of Li(s) | 4. | 1 g of Ag(s) |
Which of the following is a basic amino acid?
| 1. | Alanine | 2. | Tyrosine |
| 3. | Lysine | 4. | Serine |
The correct option for free expansion of an ideal gas under adiabatic condition is:
1. and
2. and
3. and
4. and
Identify the INCORRECT match.
| Name | IUPAC Official Name | ||
| a. | Unnilunium | (i) | Mendelevium |
| b. | Unniltrium | (ii) | Lawrecium |
| c. | Unnilhexium | (iii) | Seaborgium |
| d. | Unununnium | (iv) | Darmstadtium |
| 1. | (b)- (ii) | 2. | (c)- (iii) |
| 3. | (d) -(iv) | 4. | (a)- (i) |
A molecule that cannot exist is:
| 1. | 2. | ||
| 3. | 4. |
Identify the correct statements from the following:
| (a) | \(\mathrm{CO}_2(\mathrm{~g})\) is used as a refrigerant ice cream and frozen food. |
| (b) | The structure of \(\mathrm{C}_{60} \) customs12 six carbon rings and 25 five carbon rings |
| (c) | ZSM-5, a type of zeolite, is used to convert alcohol into gasoline. |
| (d) | CO is a colorless and odorless gas. |
| 1. | (a) and (c) only | 2. | (b) and (c) only |
| 3. | (c) and (d) only | 4. | (a), (c) and (d) only |
An alkene on ozonolysis gives methanal as one of the products. Its structure is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. | sec-Butyl alcohol | 2. | tert-Butyl alcohol |
| 3. | iso-Butyl alcohol | 4. | iso-Propyl alcohol |
A mixture of and Ar gases in a cylinder contains 7 g of and 8 g of Ar. If the total pressure of the mixture of the gases in the cylinder is 27 bar, the partial pressure of is:
[Use atomic masses (in g ): N=14, Ar=40]
1. 12 bar
2. 15 bar
3. 18 bar
4. 9 bar
An increase in the concentration of the reactants of a reaction leads to a change in:
| 1. | Heat of reaction | 2. | Threshold energy |
| 3. | Collision frequency | 4. | Activation energy |
Given that the ionic product of is 2 × . The solubility of in 0.1 M NaOH is ;
1. 2 × M
3. 1 × M
4. 2 × M
The product 'X' in the below mentioned reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Urea reacts with water to form A which will decompose to form B. B when passed through \(\left(Cu\right)^{2 +}\) (aq), deep blue colour solution C is formed. What is the formula of C from the following?
| 1. | \(\left[Cu \left(NH_{3}\right)_{4}\right]^{2 +}\) | 2. | \(Cu \left(\right. OH \left.\right)_{2}\) |
| 3. | \(CuCO_{3} . Cu (OH)_{2}\) | 4. | \(CuSO_{4}\) |
A screw gauge has the least count of \(0.01~\text{mm}\) and there are \(50\) divisions in its circular scale. The pitch of the screw gauge is:
| 1. | \(0.25~\text{mm}\) | 2. | \(0.5~\text{mm}\) |
| 3. | \(1.0~\text{mm}\) | 4. | \(0.01~\text{mm}\) |
The mean free path for a gas, with molecular diameter \(d\) and number density \(n,\) can be expressed as:
| 1. | \( \dfrac{1}{\sqrt{2} n \pi {d}^2} \) | 2. | \( \dfrac{1}{\sqrt{2} n^2 \pi {d}^2} \) |
| 3. | \(\dfrac{1}{\sqrt{2} n^2 \pi^2 d^2} \) | 4. | \( \dfrac{1}{\sqrt{2} n \pi {d}}\) |
| 1. | four times | 2. | one-fourth |
| 3. | zero | 4. | doubled |
In a certain region of space with volume \(0.2~\text m^3,\) the electric potential is found to be \(5~\text V\) throughout. The magnitude of the electric field in this region is:
| 1. | \(0.5~\text {N/C}\) | 2. | \(1~\text {N/C}\) |
| 3. | \(5~\text {N/C}\) | 4. | zero |
Which of the following graph represents the variation of resistivity () with temperature (\(T\)) for copper?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
A wire of length \(L,\) area of cross section \(A\) is hanging from a fixed support. The length of the wire changes to \({L}_1\) when mass \(M\) is suspended from its free end. The expression for Young's modulus is:
| 1. | \(\dfrac{{Mg(L}_1-{L)}}{{AL}}\) | 2. | \(\dfrac{{MgL}}{{AL}_1}\) |
| 3. | \(\dfrac{{MgL}}{{A(L}_1-{L})}\) | 4. | \(\dfrac{{MgL}_1}{{AL}}\) |
In a guitar, two strings \(A\) and \(B\) made of same material are slightly out of tune and produce beats of frequency \(6~\text{Hz}\). When tension in \(B\) is slightly decreased, the beat frequency increases to \(7~\text{Hz}\). If the frequency of \(A\) is \(530~\text{Hz}\), the original frequency of \(B\) will be:
| 1. | \(524~\text{Hz}\) | 2. | \(536~\text{Hz}\) |
| 3. | \(537~\text{Hz}\) | 4. | \(523~\text{Hz}\) |
| 1. | \(2.05~\text A\) | 2. | \(2.5~\text A\) |
| 3. | \(25.1~\text A\) | 4. | \(1.7~\text A\) |
A ball is thrown vertically downwards with a velocity of \(20\) m/s from the top of a tower. It hits the ground after some time with the velocity of \(80\) m/s . The height of the tower is: (assuming \(g = 10~\text{m/s}^2)\)
| 1. | \(340\) m | 2. | \(320\) m |
| 3. | \(300\) m | 4. | \(360\) m |
An electron is accelerated from rest through a potential difference of \(V\) volt. If the de Broglie wavelength of an electron is \(1.227\times10^{-2}~\text{nm}.\) What will be its potential difference?
| 1. | \(10^{2}~\text{V}\) | 2. | \(10^{3}~\text{V}\) |
| 3. | \(10^{4}~\text{V}\) | 4. | \(10^{5}~\text{V}\) |
For the logic circuit shown, the truth table is:
| 1. |
|
|||||||||||||||
| 2. |
|
|||||||||||||||
| 3. |
|
|||||||||||||||
| 4. |
|
A short electric dipole has a dipole moment of \(16 \times 10^{-9} ~\text{C-}\text{m}. \) The electric potential due to the dipole at a point at a distance of \(0.6~\text{m}\) from the centre of the dipole situated on a line making an angle of \(60^{\circ}\) with the dipole axis is:
\(\left( \dfrac{1}{4\pi \varepsilon_0}= 9\times 10^{9}~\text{N-m}^2/\text{C}^2 \right) \)
| 1. | \(200~\text{V}\) | 2. | \(400~\text{V}\) |
| 3. | zero | 4. | \(50~\text{V}\) |
An iron rod of susceptibility \(599\) is subjected to a magnetizing field of \(1200~\text{A m}^{-1}.\) The permeability of the material of the rod is:
\((\mu_0 = 4 \pi\times 10^{-7}~\text{T mA}^{-1})\)
1. \(8.0\times 10^{-5}~\text{T mA}^{-1}\)
2. \(2.4\pi\times 10^{-5}~\text{T mA}^{-1}\)
3. \(2.4\pi\times 10^{-7}~\text{T mA}^{-1}\)
4. \(2.4\pi\times 10^{-4}~\text{T mA}^{-1}\)
The increase in the width of the depletion region in a \(\mathrm{p\text{-}n}\) junction diode is due to:
1. reverse bias only
2. both forward bias and reverse bias
3. increase in forwarding current
4. forward bias only
A capillary tube of radius \(r\) is immersed in water and water rises in it to a height \(h.\) The mass of the water in the capillary is \(5\) g. Another capillary tube of radius \(2r\) is immersed in water. The mass of water that will rise in this tube is:
| 1. | \(5.0\) g | 2. | \(10.0\) g |
| 3. | \(20.0\) g | 4. | \(2.5\) g |
| 1. | \(4.5\times10^{13}~\text J\) | 2. | \(1.5\times10^{13}~\text J\) |
| 3. | \(0.5\times10^{13}~\text J\) | 4. | \(4.5\times10^{16}~\text J\) |
The solids which have the negative temperature coefficient of resistance are:
| 1. | insulators only |
| 2. | semiconductors only |
| 3. | insulators and semiconductors |
| 4. | metals |
A ray is incident at an angle of incidence \(i\) on one surface of a small angle prism (with the angle of the prism \(A\)) and emerges normally from the opposite surface. If the refractive index of the material of the prism is \(\mu,\) then the angle of incidence is nearly equal to:
| 1. | \(\dfrac{2A}{\mu}\) | 2. | \(\mu A\) |
| 3. | \(\dfrac{\mu A}{2}\) | 4. | \(\dfrac{A}{2\mu}\) |
For which one of the following Bohr models is not valid?
| 1. | Singly ionised helium atom \(\big(\mathrm{He}^{+}\big).\) |
| 2. | Deuteron atom. |
| 3. | Singly ionised neon atom \(\big(\mathrm{Ne}^{+}\big).\) |
| 4. | Hydrogen atom. |
Assume that light of wavelength 600 nm is coming from a star. The limit of resolution of telescope whose objective has a diameter of 2 m is:
1.
2.
3.
4.
A body weighs \(72~\text{N}\) on the surface of the earth. What is the gravitational force on it at a height equal to half the radius of the earth?
| 1. | \(32~\text{N}\) | 2. | \(30~\text{N}\) |
| 3. | \(24~\text{N}\) | 4. | \(48~\text{N}\) |
A charged particle having drift velocity of \(7.5\times10^{-4}~\text{ms}^{-1}\) in an electric field of \(3\times10^{-10}~\text{Vm}^{-1},\) has mobility of:
1. \(2.5\times 10^{6}~\text{m}^2\text{V}^{-1}\text{s}^{-1}\)
2. \(2.5\times 10^{-6}~\text{m}^2\text{V}^{-1}\text{s}^{-1}\)
3. \(2.25\times 10^{-15}~\text{m}^2\text{V}^{-1}\text{s}^{-1}\)
4. \(2.25\times 10^{15}~\text{m}^2\text{V}^{-1}\text{s}^{-1}\)
For transistor action, which of the following statements is correct?
| 1. | Base, emitter, and collector regions should have the same size. |
| 2. | Both emitter junction, as well as the collector junction, are forward biased. |
| 3. | The base region must be very thin and lightly doped. |
| 4. | Base, emitter, and collector regions should have the same doping concentrations. |
The capacitance of a parallel plate capacitor with air as a medium is \(6~\mu\text{F}.\) With the introduction of a dielectric medium, the capacitance becomes \(30~\mu\text{F}.\) The permittivity of the medium is:
\(\left(\varepsilon_0=8.85 \times 10^{-12} ~\text{C}^2 \text{N}^{-1} \text{m}^{-2}\right )\)
1. \(1.77 \times 10^{-12}~ \text{C}^2 \text{N}^{-1} \text{m}^{-2}\)
2. \(0.44 \times 10^{-10} ~\text{C}^2 \text{N}^{-1} \text{m}^{-2}\)
3. \(5.00 ~\text{C}^2 \text{N}^{-1} \text{m}^{-2}\)
4. \(0.44 \times 10^{-13} ~\text{C}^2 \text{N}^{-1} \text{m}^{-2}\)
| 1. | \(9.98~\text{m}\) | 2. | \(9.980~\text{m}\) |
| 3. | \(9.9~\text{m}\) | 4. | \(9.9801~\text{m}\) |
Two bodies of mass, \(4~\text{kg}\) and \(6~\text{kg}\), are tied to the ends of a massless string. The string passes over a pulley, which is frictionless (see figure). The acceleration of the system in terms of acceleration due to gravity (\(g\)) is:
| 1. | \(\dfrac{g}{2}\) | 2. | \(\dfrac{g}{5}\) |
| 3. | \(\dfrac{g}{10}\) | 4. | \(g\) |
A cylinder contains hydrogen gas at a pressure of \(249~\text{kPa}\) and temperature \(27^\circ\text{C}.\) Its density is:
(\(R=8.3~\text{J mol}^{-1} \text {K}^{-1}\))
1. \(0.2~\text{kg/m}^{3}\)
2. \(0.1~\text{kg/m}^{3}\)
3. \(0.02~\text{kg/m}^{3}\)
4. \(0.5~\text{kg/m}^{3}\)
| 1. | \(1:1\) | 2. | \(1:c\) |
| 3. | \(1:c^2\) | 4. | \(c:1\) |
A long solenoid of \(50~\text{cm}\) length having \(100\) turns carries a current of \(2.5~\text{A}\). The magnetic field at the centre of the solenoid is:
\(\big(\mu_0 = 4\pi\times 10^{-7}~\text{TmA}^{-1} \big)\)
1. \(3.4\times 10^{-4}~\text{T}\)
2. \(6.28\times 10^{-5}~\text{T}\)
3. \(3.14\times 10^{-5}~\text{T}\)
4. \(6.28\times 10^{-4}~\text{T}\)
A resistance wire connected in the left gap of a meter bridge balances a \(10~\Omega\) resistance in the right gap at a point which divides the bridge wire in the ratio \(3:2\). lf the length of the resistance wire is \(1.5~\text{m}\), then the length of \(1~\Omega\) of the resistance wire will be:
| 1. | \(1.0\times 10^{-1}~\text{m}\) | 2. | \(1.5\times 10^{-1}~\text{m}\) |
| 3. | \(1.5\times 10^{-2}~\text{m}\) | 4. | \(1.0\times 10^{-2}~\text{m}\) |
The energy required to break one bond in DNA is \(10^{-20}~\text{J}\). This value in eV is nearly:
1. \(0.6\)
2. \(0.06\)
3. \(0.006\)
4. \(6\)
| 1. | \(_{40}^{91}\mathrm{Zr}\) | 2. | \(_{36}^{101}\mathrm{Kr}\) |
| 3. | \(_{36}^{103}\mathrm{Kr}\) | 4. | \(_{56}^{144}\mathrm{Ba}\) |
Two cylinders \(A\) and \(B\) of equal capacity are connected to each other via a stop cock. A contains an ideal gas at standard temperature and pressure. \(B\) is completely evacuated. The entire system is thermally insulated. The stop cock is suddenly opened. The process is:
| 1. | adiabatic | 2. | isochoric |
| 3. | isobaric | 4. | isothermal |
Light with an average flux of \(20~\text{W/cm}^2\) falls on a non-reflecting surface at normal incidence having a surface area \(20~\text{cm}^2\). The energy received by the surface during time span of \(1\) minute is:
1. \(12\times 10^{3}~\text{J}\)
2. \(24\times 10^{3}~\text{J}\)
3. \(48\times 10^{3}~\text{J}\)
4. \(10\times 10^{3}~\text{J}\)
The quantities of heat required to raise the temperature of two solid copper spheres of radii \(r_1\) and \(r_2\) \((r_1=1.5~r_2)\) through \(1~\text{K}\) are in the ratio:
| 1. | \(\dfrac{9}{4}\) | 2. | \(\dfrac{3}{2}\) |
| 3. | \(\dfrac{5}{3}\) | 4. | \(\dfrac{27}{8}\) |
| 1. | \(\dfrac{3}{2}k_BT\) | 2. | \(\dfrac{5}{2}k_BT\) |
| 3. | \(\dfrac{7}{2}k_BT\) | 4. | \(\dfrac{1}{2}k_BT\) |
An AC voltage source is connected to a series \(LCR\) circuit. When \(L\) is removed from the circuit, the phase difference between current and voltage is \(\dfrac{\pi}{3}\). If \(C\) is instead removed from the circuit, the phase difference is again \(\dfrac{\pi}{3}\) between current and voltage. The power factor of the circuit is:
1. \(0.5\)
2. \(1.0\)
3. \(-1.0\)
4. zero
Two particles of mass \(5~\text{kg}\) and \(10~\text{kg}\) respectively are attached to the two ends of a rigid rod of length \(1~\text{m}\) with negligible mass. The centre of mass of the system from the \(5~\text{kg}\) particle is nearly at a distance of:
1. \(50~\text{cm}\)
2. \(67~\text{cm}\)
3. \(80~\text{cm}\)
4. \(33~\text{cm}\)
The phase difference between displacement and acceleration of a particle in a simple harmonic motion is:
1. \(\dfrac{3\pi}{2}\text{rad}\)
2. \(\dfrac{\pi}{2}\text{rad}\)
3. zero
4. \(\pi ~\text{rad}\)
The Brewster's angle for an interface should be:
1. \(30^{\circ}<i_b<45^{\circ}\)
2. \(45^{\circ}<i_b<90^{\circ}\)
3. \(i_b=90^{\circ}\)
4. \(0^{\circ}<i_b<30^{\circ}\)
Dimensions of stress are:
| 1. | \( {\left[{ML}^2 {T}^{-2}\right]} \) | 2. | \( {\left[{ML}^0 {T}^{-2}\right]} \) |
| 3. | \( {\left[{ML}^{-1} {T}^{-2}\right]} \) | 4. | \( {\left[{MLT}^{-2}\right]}\) |
The colour code of resistance is given below:
The values of resistance and tolerance, respectively are:
1. 47 k, 10%
2. 4.7 k, 5%
3. 470 , 5%
4. 470 k, 5%
A spherical conductor of radius \(10~\text{cm}\) has a charge of \(3.2 \times 10^{-7}~\text{C}\) distributed uniformly. What is the magnitude of the electric field at a point \(15~\text{cm}\) from the centre of the sphere?
\(\left(\frac{1}{4\pi \varepsilon _0} = 9\times 10^9~\text{N-m}^2/\text{C}^2\right)\)
1. \(1.28\times 10^{5}~\text{N/C}\)
2. \(1.28\times 10^{6}~\text{N/C}\)
3. \(1.28\times 10^{7}~\text{N/C}\)
4. \(1.28\times 10^{4}~\text{N/C}\)
What would be the torque about the origin when a force \(3\hat{j}~\text N\) acts on a particle whose position vector is \(2\hat{k}~\text m?\)
| 1. | \(6\hat{j}~\text{N-m}\) | 2. | \(-6\hat{i}~\text{N-m}\) |
| 3. | \(6\hat{k}~\text{N-m}\) | 4. | \(6\hat{i}~\text{N-m}\) |