Study the diagram given below and choose the correct information that can be deduced :
1. The core RNA polymerase is capable of catalyzing elongation only
2. Sigma factor and Rho factor are needed for termination of transcription
3. DNA helicase opens the strand of the DNA
4. Both strands of DNA are transcribed by the same RNA polymerase
Consider the transcription unit given in the following diagram and choose the correct statements :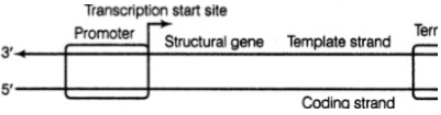
I. The promoter is situated upstream and 5' to the structural gene
II. It is the presence of the structural gene that defines the template strand
III. The coding strand does not code for anything and is displaced during transcription
1. I and II only
2. I and III only
3. II and III only
4. I,II and III
Consider the following statements:
I. Y chromosome determines maleness in humans and other mammals.
II. Sex-linked recessive characters are more commoniy seen in the males of the species.
III. In birds and butterflies, males are XX and females are XY.
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II and III
The feature lacking in bony fishes would be:
| 1. | Operculum | 2. | Swim bladder |
| 3. | Ammonotelism | 4. | Placoid scales |
A person can receive blood only from a donor of his/her blood group if the blood group of this person is:
1. O
2. AB
3. A or B
4. A, B, AB or O
Identify the correctly matched pair:
|
1. |
Asexual reproductive structure in Chlamydomonas |
Zygospores |
|
2. |
Vegetative propagule in Agave |
Offset |
|
3. |
Chromosome number in meiocyte of Ophioglossum |
630 |
|
4. |
Marchantia |
Dioecious plant |
Match each item in Column I with one in Column II and select the correct answer from the codes given:
|
A |
Codominance |
P |
Progeny resembles both parents |
|
B |
Incomplete dominance |
Q |
Progeny does not resemble either of the parents |
|
C |
Sex determination in Drosophila |
R |
Haplo-diploidy |
|
D |
Sex determination in Honey bee |
S |
Genic balance |
Codes :
| A | B | C | D | |
| 1. | P | Q | R | S |
| 2. | P | Q | S | R |
| 3. | Q | P | R | S |
| 4. | Q | P | S | R |
Regarding Mendelian disorders :
| 1. | Color blindness is more common in the females of the species. |
| 2. | Sickle cell anemia occurs due to the substitution of valine by glutamic acid in the globin chain of haemoglobin. |
| 3. | Individuals affected by phenylketonuria lack an enzyme that converts tyrosine to phenylalanine. |
| 4. | Thalassemia is inherited as an autosomal recessive trait. |
Given below in Column I are the criteria for a molecule that can act as a genetic material and the corresponding inferences drawn are given in Column II. Identify the criteria that is not correctly matched to the inference drawn:
|
|
Criteria |
Inference drawn |
|
1. |
It should be able to replicate |
Protein cannot be the genetic material |
|
2. |
It should be stable structurally and chemically |
DNA is a better genetic material than RNA |
|
3. |
It should provide the scope for slow changes |
RNA evolves faster than DNA |
|
4. |
It should be able to express in the form of Mendelian characters |
The protein synthesizing machinery has evolved around DNA |
Normally the process of DNA fingerprinting does not involve:
1. Southern blot
2. Elution
3. Radiolabelled VNTR probes
4. Autoradiography
Identify the biome that is not correctly matched with its mean annual temperature and mean annual precipitation:
|
|
Biome |
Mean annual temperature [0C] |
Mean annual precipitation [cm] |
|
1. |
Desert |
0 – 25 |
25 – 100 |
|
2. |
Temperate Forest |
10 – 22 |
50 – 220 |
|
3. |
Tropical forest |
20 – 25 |
150 – 425 |
|
4. |
Coniferous forest |
0 – 15 |
50 – 250 |
The relationship between cattle egret and grazing cattle can be best described as:
| 1. | Mutualism | 2. | Commensalism |
| 3. | Parasitism | 4. | Competition |
Which of the following will lead to decreased rate of decomposition of detritus?
1. Anaerobiosis
2. High temperature
3. Moist environment
4. High nitrogen content in detritus
Match the legislation in Column I with the corresponding year in Column II and select the correct answer from the codes given:
|
|
Legislation |
|
Year |
|
A |
Air [Prevention and Control of Pollution] Act |
P |
1974 |
|
B |
Environment [Protection] Act |
Q |
1981 |
|
C |
Water [Prevention and Control of Pollution] Act |
R |
1986 |
|
D |
National Forest Policy |
S |
1988 |
Codes
| Options: | (a) | (b) | (c) | (d) |
| 1. | R | Q | S | P |
| 2. | Q | R | P | S |
| 3. | Q | R | S | P |
| 4. | R | Q | P | - S |
Ozone hole develops over Antarctica each year between:
1. Early January and late April
2. Early March and late July
3. Late August and early October
4. Late September and early December
The steps of the cardiac cycle in sequence are:
1. isovolumic contraction, ejection, isovolumic relaxation, passive ventricular filling, active ventricular filling.
2. isovolumic relaxation, isovolumic contraction, ejection, passive ventricular filling, active ventricular filling.
3. isovolumic contraction, isovolumic relaxation, ejection, passive ventricular filling, active ventricular filling.
4. isovolumic contraction, ejection, isovolumic relaxation, active ventricular filling, passive ventricular filling.
The amount of urine produced is generally increased when there is:
1. increased ADH secretion
2. increased atrial natriuretic hormone secretion
3. increased aldosterone secretion
4. sympathetic stimulation of the renal arteries
You are inserting your gene of interest into the Lac Z gene in a plasmid also containing a tetracycline-resistant gene. You plate your transformed bacteria on media containing tetracycline and X-gal. Which of the following results would indicate a clone with recombinant plasmids?
| 1. | A clone which did not grow on the tetracycline plates |
| 2. | A white colony on the tetracycline plates |
| 3. | A blue colony on the tetracycline plates |
| 4. | A red colony on the tetracycline plates |
Photochemical smog does not contain:
| 1. | O3 | 2. | PAN |
| 3. | NOx | 4. | CO2 |
Given below is a figure that indicates the external
features of cockroach
Choose the option which is correct for the features of
the labelled structure A,B,C and D
|
1. |
A – Tegmina |
- |
Transparent, membranous wings used in flight |
|
2. |
B– Anal styles |
- |
Jointed filamentous structures present in males but absent in females |
|
3. |
C-Compound eye |
- |
With the help of several ommatidia, a cockroach can receive several images of an object known as mosaic vision with more sensitivity and less resolution, being common during night |
|
4. |
D – Fore wing |
- |
Dark, opaque and leathery which are used in flight |
Peristalsis occurs
1. from the mouth to the small intestine
2. from the beginning of the esophagus to the anus
3. only in the stomach
4. only in the small and large intestines
Which group of plants show clear cut vegetative,
reproductive and senescent phases in their life cycle?
1. Mustard, Radish, Mango.
2. Peepal, Mango, Banyan.
3. Wheat, Rice, Cabbage.
4. Peepal, Bryophyllum, Rice.
Select the group of animals belonging to different
phyla?
1. Hirudinaria, Limulus, Laccifer.
2. Nereis, Dentalium, Aplysia.
3. Pheretima, Chaetopleura, Ophiura.
4. Cucumaria, Anteadon, Loligo.
Which of the following complex of mitochondrial ETS has two copper centers?
1. Cytochrome b c1 complex
2. NADH dehydrogenase complex
3. Succinate dehydrogenase complex
4. Cytochrome oxidase complex
Monascus purpureus is a
i. Bacteria
ii. Yeast
iii. Produces cyclosporin A
iv. Produces statins
1. i,iii
2. ii,iii
3. i,iv
4. ii,iv
Vertebrates and seastars may seen as different as two animal groups can be, yet they share
| 1. | The same type of body symmetry as adults. |
| 2. | A high degree of cephalization. |
| 3. | Certain developmental pattern, including the type of coelom formation. |
| 4. | The presence of endoskeleton that include cranium |
Which of the following statement is incorrect?
| 1. | Taxa can indicate categories at very different levels. |
| 2. | systematics takes into account evolutionary relationship between organisms. |
| 3. | Modern taxonomy do not take into account ecological information about organisms. |
| 4. | Each rank or taxon represents unit of classification. |
Most of the organelle duplication occurs during which
phase?
1. G1
2. G2
3. S
4. M
It begins with the simultaneous splitting of the
centromere of each chromosome, is true for which stage
of Cell division.
1. Anaphase-I
2. Anaphase-II
3. Anaphase
4. More than one
Which of the following have pigment similar to higher
plants?
1. Group which causes red tide.
2. Group which is chief producer of ocean.
3. Group when deprived of light behaves as heterotroph.
4. Group where spore have cellulosic cell wall
Which of the following fungus lacks dikaryophase?
1. Claviceps
2. Puccinia
3. Aspergillus
4. Trichoderma
Which of the following is not correctly matched?
| 1. | Inert Crystals - Virus | 2. | Free RNA - Viroids |
| 3. | Normal protein - Prions | 4. | Symbionts - Lichen |
Mark the incorrect match
| 1. | Radial conduction of water – Ray parenchyma. |
| 2. | Water filled cavities – Maize stem. |
| 3. | Phloem parenchyma – Mainly in monocots. |
| 4. | Trichomes – Prevents water loss |
How many of the following plants have stipulate leaf?
Tulip, Petunia, Lupin, Sweet pea, Indigofera,
Sunhemp, Aloe, Ashwagandha
1. 6
2. 4
3. 7
4. 3
In floral diagram a dot at the top of diagram represents
1. Position of ovary
2. Aestivation
3. Mother axis
4. Cohesion
In ribosome s-value provides indirect information about
1. Size and density.
2. Size & volume.
3. Volume and density.
4. Mass and size
Smooth endoplasmic reticulum performs all the
functions, except
| 1. | Production of glycoprotein. |
| 2. | Synthesis of lipid. |
| 3. | Synthesis of glycogen. |
| 4. | Synthesis of steroidal hormones |
Find out the correct match from the following table:-
| Column-I | Column-II | Column-III | |
| (i) | Radula | Rasping organ | Pila |
| (ii) | Hooks and suckers | Taenia | Annelida |
| (iii) | Tube feet | Asterias; Dentalium | Echinodermata |
| (iv) | Comb plates | Pleurobrachia; Aurelia | Ctenophora |
1. (i) only
2. (i) and (ii)
3. (iii) only
4. (ii) and (iii)
Which of the following epithelium their main function is to provide protection against chemical and mechanical stresses?
1. Simple epithelium.
2. Compound epithelium.
3. Columnar epithelium.
4. Cuboidal Epithelium
A list of different metabolites is given below in this list find out the number of primary & secondary metabolites, respectively & chooses the correct option:
Alkaloids, Abrin, Ricin, Carotenoids, Amino acids,
Glucose, Fructose, Fatty acids, Thymine, Uracil,
Lectins, Drugs.
1. Five and Six
2. Six and Six
3. Seven and five
4. Four and Eight
Read the following statement (A-D) :-
| (a) | Each protein is a polymer of amino acids. |
| (b) | A protein is a hetero polymer and not a homopolymer. |
| (c) | Dietary proteins are the source of essential amino acid. |
| (d) | Collagen is the most abundant protein in whole of the biosphere. |
How many of the above statement are right ?
1. Three
2. One
3. Two
4. Four
Theory of spontaneous generation was rejected because:
| 1. | It explained origin of first life from non-living or inanimate matter and lacked experimental evidence. |
| 2. | It was based on the biogenesis concept. |
| 3. | It did not explain about the origin of first life on earth. |
| 4. | It proposed origin of life from outer space. |
Which of the following options is not correct regarding the early atmosphere?
(A) Oxygen was totally absent
(B) Atmosphere was reducing
(C) Temperature was high
(D) Presence of free molecules
(E) Presence of molecules like vapours
1. A, D and E
2. A and D
3. D and E
4. A only
A founder event favor microevolution in the founding population mainly because
| 1. | Mutations are more common in a head environment |
| 2. | A small founding population is subject to extensive sampling error in the composition of its gene pool |
| 3. | Gene flow increases |
| 4. | The low environment is likely to be patchy favouring diversifying selection |
If frequency of 'A' allele is 0.4 than, find out the frequency of 'B' allele and heterozygous genotype in a random mating population at equilibria.
1. 0.6 an 0.24
2. 0.6 and 0.96
3. 0.6 and 0.48
4. 0.6 and 0.50
Trace the path of physiologically mature sperm in the male reproductive tract
| 1. | Seminiferous tubulesRete testisVasa efferentiaEpididymisVas deferensEjaculatory ducturethra |
| 2. | Vas deferensEpididymisEjaculatory ductUrethra |
| 3. | Rete testisEpididymisVasa efferentiaVas deferensEjaculatory ducturethra |
| 4. | EpididymisEjaculatory ductVas deferenceUrethra |
Find the incorrect statement
1. scrotum helps in maintaining low temperature of the testes 2-2.5 less than normal, necessary for spermatogenesis
2. Each testis has about 250 testicular lobules
3. Transfer of sperms into female genital tract is insemination
4. Seminiferous tubules open into rete testis through vasa efferentia
Decline in the level of which hormone of menstrual cycle is directly responsible for degeneration of corpus luteum?
1. Estrogen
2. Progesterone
3. LH
4. FSH
Oogenesis differs from spermatogenesis in a number of aspects. One of the following is however a similarity between the two
1. Growth phase is prolonged in both of them an starts before birth
2. Both give rise to non-motile gametes
3. Both produce equal number of gametes
4. Both occur inside primary sex organ
A contraceptive pill contains
| 1. | Progesterone and ostrogen |
| 2. | Spermicidal salts |
| 3. | Chemicals that cause automatic abortion |
| 4. | Chemicals that prevent fertilization of ovum |
Consider the following human disorders:
i. Haemophilia ii. Down’s Syndrome iii. Cystic fibrosis iv. Colour blindness
v. Night blindness.
Which of these disorders exhibit ‘Mendelian’ pattern of inheritance?
1. ii, iii and iv
2. i, ii and iii
3. i, iii and v
4. i, iii and iv
A plant with genotype AABbCcDD is self pollinated. Provided that the four genes are independently assorting, what proportion of the progeny will show the genotype AAbbccDD ?
1. 1/4
2. 1/16
3. 1/64
4. 1/256
In chemical synapse, postsynaptic membrane receptor
binds with
1. Excitatory neurotransmitter.
2. Inhibitory neurotransmitter.
3. Ca++ ions.
4. Both 1 and 2
Potato spindle tuber disease is caused by
1. A capsidated nucleic acid
2. ds, naked RNA
3. ss, naked RNA
4. Virusoid
A store house of collected plant specimens that are dried, pressed and preserved on sheets is known as
1. Flora
2. Manual
3. Herbarium
4. Botanical garden
Select an incorrect match
1. Psilopsida – Psilotum
2. Sphenopsida – Selaginella
3. Lycopsida – Lycopodium
4. Pteropsida – Dryopteris
Select incorrect statement(s):
(a) Both phloem loading and unloading are active processes.
(b) Loading of the phloem sets up root pressure that facilitates the mass movement in the phloem.
(c) As the hydrostatic pressure in the phloem sieve tube increases, pressure flow begins and the sap moves through
the phloem.
1. Both (b) & (c)
2. Both (a) & (c)
3. Only (b)
4. Only (c)
Match the following
Column-I Column-II
a. Mn (i) Nitrogenase
b. Fe (ii) Carbonic anhydrase
c. Mo (iii) Photolysis of water
d. Zn (iv) Catalase
1. a(iii), b(iv), c(i), d(ii)
2. a(ii), b(iii), c(i), d(iv)
3. a(ii), b(i), c(iv), d(iii)
4. a(i), b(iv), c(ii), d(iii)
RQ value for tripalmitin is
1. > 1
2. < 1
3. Zero
4. Infinite
Some tissue are listed below, find a correct set of tissues which are product of redifferentiation in a woody
dicotyledonous plant
| (a) | Phellogen |
| (b) | Phelloderm |
| (c) | Cork |
| (d) | Primary xylem |
| (e) | Secondary phloem |
| (f) | Interfascicular cambium |
1. (a), (b) & (c)
2. (a) & (f)
3. (b), (c), (e) & (f)
4. (b), (c) & (e)
Deltoid ridge is the characteristic feature of
1. Humerus
2. Radius
3. Femur
4. Tibia
Ability to abstract, reasoning, decision making, reality testing by judgement are regulated by
1. Frontal lobe
2. Parietal lobe
3. Temporal lobe
4. Occipital lobe
In which of the following organ immature lymphocytes differentiate into antigen sensitive lymphocytes?
1. Thymus
2. Spleen
3. Peyer’s patches
4. Appendix
If a person shows the production of interferons in his body, he suffers from
1. Tetanus
2. Tuberculosis
3. Measles
4. Typhoid
The product has to be subjected through a series of processes before it is ready for marketing as a finished product called down stream processing. It includes
1. Separation
2. Purification
3. Quality control
4. Both 1 & 2
Insulin cannot be administered orally to diabetic patient because of its
1. Proteinaceous nature
2. Disulphide
3. Lipid nature
4. All of these
Which of the following was the first transgenic animal?
1. Transgenic salmon
2. Transgenic chicken
3. Transgenic mice
4. Transgenic rabbits
The respiratory centre in the brain is stimulated by
1. CO2 concentration in venous blood
2. O2 concentration in arterial blood
3. CO2 concentration in arterial blood
4. O2 concentration in venous blood
The patient of diabetes mellitus is characterized by
1. Ketonuria
2. Glycosuria
3. Haematuria
4. Both 1 & 2
Which of the following geographical region has maximum number of ‘Hot spots’ of biodiversity?
1. Asia-Pacific
2. Africa
3. South America
4. Europe and Central Asia
Which one of the following species is more susceptible to extinction due to narrow range of geographical distribution?
1. Blue whale
2. Woodland caribou
3. Rhinoceros
4. Bald eagle
Which of the following bacteria is generally an indicator of faecal pollution?
1. E. coli
2. Vibrio cholera
3. Daphnia
4. Cyclops
Antipodal cells are always present in all type of embryo sacs except in
1. Oenothera
2. Panaea
3. Allium
4. Adoxa
Which of the following PGR can cause tightening of epicotyl hook during seed germination?
1. GA
2. CK
3. C2H4
4. ABA
Male cones in Pinus
| 1. | Arise in place of long shoots |
| 2. | Develop singly |
| 3. | Are largest in plant kingdom |
| 4. | Are equivalent to male flowers of angiosperms |
Aggregate fruits
1. Are always true
2. Are actually aggregate of simple fruits
3. Arise from many ovaries of many flowers
4. Arise from inflorescence
Calvin cycle (C3 cycle)
| 1. | Is a feature of all chloroplast containing cells in plants |
| 2. | Is the main sugar-producing cycle in C3, C4, and CAM plants |
| 3. | Has both oxidative and reductive reactions |
| 4. | Occurs in darkness only |
Pollen grains in angiosperms
1. Show precocious germination
2. Are discharged at 2-3 celled stage
3. Have outer wall which is gametophytic in origin
4. More than one option is correct
Which of the following is both a green house gas as well as ozone depleting substance?
1. CO2
2. SO2
3. CFC
4. Water vapour
Choose wrong statement for ecological pyramids
1. Decomposers are not given place
2. Producers are always placed at base
3. Energy pyramid is always erect
4. Food web is taken in place of food chain
Medulla oblongata controls various involuntary movements of the body except
1. Vasomotor centre to regulate the diameter of blood vessels
2. It has reflex centre for swallowing, vomiting peristalsis, coughing and sneezing
3. It can modify or tone up the motor activities which originate from cerebrum
4. It has cardiac and respiratory centre to regulate heart beat and respiration
Neurohypophysis store and release hormones which are actually synthesised by the hypothalamus and are transported axonally here. Which of the following hormones belong to this category?
1. Glucocorticoid gonadotropins
2. FSH, LH
3. Oxytocin, Vasopressin
4. TSH, ACTH
Which part of the brain deals with a mixture of signals of smell and memories and is essential for learning?
1. Hypothalamus
2. Septum
3. Amygdala
4. Hippocampus
One of the following hormones do not enter inside the target cell but still leads to certain biochemical changes in the target tissue. Mark the same
1. Thyroxine
2. Cortisol
3. Glucagon
4. Progesterone
In case the wife is unable to bear the child due to defective uterus, a surrogate mother can be hired for bearing the child, developed through the technique of
1. GIFT
2. ZIFT
3. IUI
4. IVF-ET
When cotton bollworm attacks the cotton plant, bolls develop holes and opens prematurely and has poor lint. Loss of crop yield varies from 8-60%. Protective spray of insecticide is quite expensive. So which genes have been incorporated to protect the plant?
1. cry I and cry III
2. cry I Ac, cry II Ab
3. cry I A and cry IB
4. cry I Ab
Which of the following organ receives oxygenated blood only?
1. Liver
2. Lungs
3. Pancreas
4. Pituitary gland
Identify A, B and C in the diagrammatic representation of the mechanism of hormone action.
Select the correct option from the following:
| 1. | A = Steroid Hormone; B = Hormone receptor Complex; C = Protein |
| 2. | A = Protein Hormone; B = Receptor; C = Cyclic AMP |
| 3. | A = Steroid Hormone; B = Receptor; C = Second Messenger |
| 4. | A = Protein Hormone; B = Cyclic AMP; C = Hormone-receptor Complex |
Which is the most common type of embryo sac in angiosperms?
1. Tetrasporic with one mitotic stage of division
2. Monosporic with three sequential mitotic divisions
3. Monosporic with two sequential mitotic divisions
4. Bisporic with two sequential mitotic divisions
Given below are four statements pertaining to the separation of DNA fragments using gel electrophoresis. Identify the incorrect statements:
| (a) | DNA is a negatively charged molecule and so it is loaded on gel towards the anode terminal. |
| (b) | DNA fragments travel along the surface of the gel, whose concentration does not affect the movement of DNA. |
| (c) | The smaller the DNA fragment, the greater the distance it travels through it. |
| (d) | Pure DNA can be visualized directly by exposing it to UV radiation. |
Choose the correct answer from the options given below:
| 1. | (a), (c) and (d) | 2. | (a), (b) and (c) |
| 3. | (b), (c) and (d) | 4. | (a), (b) and (d) |
(CH3)3CCI + (CH3)3C K+ → Product
1. SN Product will be more
2. E2 Product will be more
3. Both will be the same
4. None of the above
The major end product of the following reaction is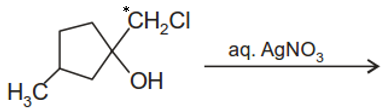
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Z
Common oxidizing agents used in organic chemistry are;
1. Fenton's reagent
2. Osmium tetroxide
3. Acidified KMnO4
4. All are correct
In E2 elimination, some compounds follow Hofmann's rule which means:
1. the double bond goes to the most substituted carbon
2. the compound is resistant to elimination
3. no double bond is formed
4. the double bond goes mainly towards the least substituted carbon
In the given reaction
The product will be:
1. 
2. 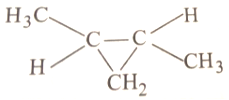
3. Mixture of 1 and 2
4.
Which among the following compounds is used for protection of carbonyl groups?
1. HCN
2.
3. ![]()
4.
Which has maximum dipole moment :-
1.
2.
3. 
4. ![]()
White fumes appear around the bottle of anhydrous AlCl3. This is due to -
1. Formation of Al(OH)3
2. Dimerisation of AlCl3
3. Hydrolysis of AlCl3 gives HCl gas
4. Adsorption of moisture
on heating (below 200 degrees celsius) liberates a gas. The same gas will be obtained below 600 degrees celsius by :
1. Heating
2. Heating
3. Heating
4. None of the above
Van-Arkel method of purification of metals involves converting the metal to a :
1. Volatile stable compound
2. Non-volatile stable compound
3. Non-volatile unstable compound
4. None of the above
The expression which gives th life 1st order reaction is :-
1.
2.
3.
4.
For a reaction A → B, the Arrhenius equation is given as \(log_{e}k \ = \ 4 \ - \ \frac{1000}{T}\) the activation energy in J/mol for the given reaction will be:
1. 8314
2. 2000
3. 2814
4. 3412
Which of the following compounds can act as both an oxidizing and a reducing agent?
| 1. | 2. | KMn | |
| 3. | 4. |
|
The charge of A sol is due to the absorbed
1.
2.
3.
4.
7.0 g of gas occupies 5.6 L of volume at STP. The gas is-
1. NO
2.
3. CO
4.
Which of the following alkenes will yield only ketones when treated with ozone?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
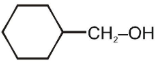
Amongst the following, the alcohol that reacts with conc. HCl /anhydrous ZnCl2 to form alkyl halide at room temperature, will be
1.
2.
3.
4. 
Consider the following reaction:-
A and B are isomeric compounds. Additionally, A is a monochloro derivative. These two can be differentiated easily by using:
1. AgN(aq)
2.
3. Conc.HN/
4. NaCl(aq)
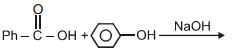
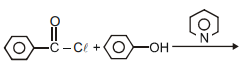
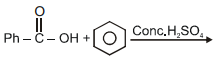
can be most suitably prepared by:-
1. 
2. 
3.
4. 
An acid that acts as a vitamin is
| 1. | Aspartic acid | 2. | Glumatic acid |
| 3. | Ascorbic acid | 4. | Saccharic acid |
The main product of the following reaction will be-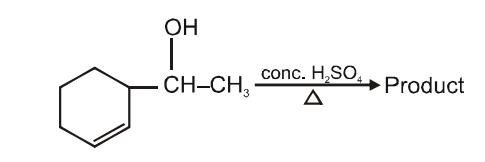
| 1. |  |
2. |  |
| 3. |  |
4. |  |
lUPAC name of complex [Cr(NO2)(NH3)5[ZnCl4] is:-
1. PentaamminenitritoNchromium(lll) terachloridozincate(ll)
2. PentaamminenitritoOchromium(lll) terachlorozincate(ll)
3. PentaamminenitroNchromium(lll) terachloridozincate(ll)
4. PentaamminenitroOchromium(lll) terachlorozincate(ll)
Among the following compounds, the paramagnetic compound with two unpaired electrons is:
1. K3[Fe(CN)6]
2. K2[NiCl4]
3. K2[CoCl4]
4. Na2[Ni(CN)4]
Out of Be, Mg, Ca, Sr and Ba,
Total number of metal whose nitrates are decomposed according to the following reaction are:
2M(NO3)2 Residue + 4NO2 + O2
1. 5
2. 4
3. 3
4. 2
The Brown ring test of is due to formation of complex :-
1.
2. [Fe(H2O)6]+2
3.
4. [Fe(H2O)5(NO)]+2
lUPAC name of turnbull's blue is :-
1. lron(ll) hexacyanidoiron(lll)
2. lron(lll) hexacyanidoiron(lll)
3. lron(ll) hexacyanidoferrate(lll)
4. lron(lll) hexacyanidoferrate(ll)
Mish metal is the alloy of :-
1. Fe + Ce
2. La + Ce
3. Cu + Ni
4. Cu + C
Which of the following gas can be most readily liquefied?
(Given : Value of 'a' for
1. NH
2. Cl
3. SO
4. CO
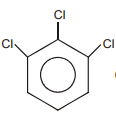
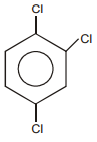
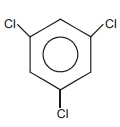
The following reaction is the best method to introduce F to benzene:
The structure of intermediate compound A is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
1 mol of Ar gas will have least density at :
1. STP
2. 0°C and 2 atm
3. 273°C and 2 atm
4. 273°C and 1 atm
Which of the following pairs of molecules have bond order three and are isoelectronic?
Which is the correct decreasing order of covalent nature?
1.
2.
3.
4.
Which of the following compounds is a planar and a polar compound?
1. TeCl4
2. SO2
3. SF6
4. XeF2
bond is present in:
| 1. | XeO3 | 2. | POCl3 |
| 3. | SO3 | 4. | All |
Which of the following order is incorrect :-
1. HF < HCl < HBr < HI (Acidic nature)
2. NaO > MgO > AlO (Basic nature)
3. Cu2> Zn2> Zn > Cu (I.P. order)
4. Ar< CI- < Cl < Ar (I.P. order)
The specific conductivity of a saturated solution of KI3 is 4.59 × 10-6 ohm-1 cm-1 and it's molar conductance is 1.53 ohm-1 cm2 mol-1. The Ksp Of KI3 will be :
1. 4 x 10-12
2. 27 x 27 x 10-9
3. 9 x 10-6
4. 4 x 10-6
A 100.0 mL dilute solution of Ag+ is electrolyzed for 15.0 minutes with a current of 1.25 mA and the silver is removed completely. The initial [Ag+] is :-
1. 2.32 x 10 M
2. 2.32 x 10 M
3. 2.32 x 10 M
4. 1.17 x 10 M
Which one of the following pair of solution can we expect to be isotonic at the same temperature?
1. 0.1 M urea and 0.1 M NaCl
2. 0.1 M urea and 0.2 M MgCl
3. 0.1 M NaCl and 0.1 M NaSO
4. 0.1 M Ca(NO) and 0.1 M NaSO
The heat of solution of BaCl2.2H2O(s) and anhydrous BaCl2(s) are 8.8 kJmol-1 and
–20.62 kJmol-1 respectively then the heat of hydration of BaCl2(s) will be:-
1. –12.25 kJ mol
2. –29.4 kJ mol
3. –11.37 kJ mol
4. +12.25 kJ mol
The maximum work done in expanding 16g isothermally at 300 K and occupying a volume of
5 dm3 until the volume becomes 25 dm3 is:
1. –2.01 J
2. 2.01× J
3. +2.81 J
4. +2.01J
If the molar concentration of is mol L–1, the concentration of chloride ions will be:
| 1. | 3.0 x 10-3 | 2. | 6.0 x 10-3 |
| 3. | 0.3 x 10-3 | 4. | 0.6 x 10-6 |
The percentage degree of hydrolysis of a salt of weak acid (HA) and weak base (BOH) in its 0.1 M solution is found to be 10%. If the molarity of the solution is 0.05 M, the percentage hydrolysis of the salt should be -
1. 5 %
2. 10 %
3. 20 %
4. None of the above
The ratio of energy of the electron in ground state of the Hydrogen to electron in first excited state of He is :-
1. 1 : 4
2. 1 : 1
2. 1 : 8
4. 1 : 16
The maximum number of electrons that can have principal quantum number n = 3 & spin quantum number = is :
1. 9
2. 18
3. 32
4. 6
The weight of a molecule of the compound is –
1. 1.4 × 10 g
2. 1.09 × 10 g
3. 5.025 × 10 g
4. 16.023 × 10 g
| 1. | \(2\) | 2. | \(1\) |
| 3. | \(3\) | 4. | \(4\) |
The root mean square velocity of the molecules of a gas is \(300 ~\text{m/s}.\) What will be the root mean square speed of the molecules if the atomic weight is doubled and the absolute temperature is halved?
| 1. | \(300 ~\text{m/s}\) | 2. | \(150 ~\text{m/s}\) |
| 3. | \(600 ~\text{m/s}\) | 4. | \(75 ~\text{m/s}\) |
Three rods made of the same material and having the same cross-section have been joined as shown in the figure. Each rod has the same length. The left and right ends are kept at \(0^{\circ}\text{C}~\text{and}~90^{\circ}\text{C},\) respectively. The temperature at the junction of the three rods will be:

1. \(45^{\circ}\text{C}\)
2. \(60^{\circ}\text{C}\)
3. \(30^{\circ}\text{C}\)
4. \(20^{\circ}\text{C}\)
A and B are two concentric metallic hollow spheres. If A is given a charge q while B is earthed as shown in figure, then
1. charge density of A and B are same
2. field inside and outside A is zero
3. field between A and B is not zero
4. field inside and outside B is zero
The potentiometer wire AB is 600 cm long. At what distance from A should the jockey J touch the wire to get zero deflection in the galvanometer?
1. 320 cm
2. 120 cm
3. 20 cm
4. 450 cm
A circular coil A has a radius R and the current flowing through it is I. Another circular coil B has radius 2R and if 2I is the current flowing through it, then the magnetic field at the centre of the circular coil are in the ratio of
1. 4:1
2. 2:1
3. 3:1
4. 1:1
In an AC circuit, the voltage applied is E=. The resulting current in the circuit . The power consumption in the circuit is given by
1.
2.
3. P = 0
4.
In an electromagnetic wave, energy density associated with a magnetic field will be:
| 1. | 2. | ||
| 3. | 4. |
|
A liquid of refractive index \(1.33\) is placed between two identical plano-convex lenses, with refractive index \(1.50\). Two possible arrangements, \(P\) and \(Q\), are shown. The system is:

| 1. | divergent in \(P\), convergent in \(Q\) | 2. | convergent in \(P\), divergent in \(Q\) |
| 3. | convergent in both | 4. | divergent in both |
In a photoelectric effect experiment
1. on increasing intensity and keeping frequency fixed, the saturation current decreases.
2. on increasing intensity and keeping frequency fixed, the saturation current remains constant.
3. on increasing intensity, saturation current may increase.
4. on increasing frequency, saturation current may increase.
A particle is projected with a velocity \(v\) such that its range on the horizontal plane is twice the greatest height attained by it. The range of the projectile is: (where \(g\) is acceleration due to gravity)
| 1. | \(\frac{4 v^2}{5 g} \) | 2. | \(\frac{4 g}{5 v^2} \) |
| 3. | \(\frac{v^2}{g} \) | 4. | \( \frac{4 v^2}{\sqrt{5} g}\) |
The acceleration 'a' of the box shown in the figure to descend so that the block of mass M exerts a force Mg/4 on the floor of the box, should be: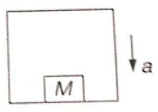
1. g/4
2. g/2
3. 3g/4
4. 4g
A block A of mass m is placed over a plank B of mass 2m. Plank B is placed over a smooth horizontal surface. The co-efficient of friction between A and B is . Block A is given a velocity towards right. Acceleration of B relative to A is
1.
2. g
3.
4. zero
A body constrained to move in y-direction is subjected to a force given by . The work done by this force in moving the body a distance of 10 m along the y-axis is
1. 20 J
2. 150 J
3. 60 J
4. 190 J
A man '\(A\)', mass \(60\) kg, and another man '\(B\)', mass \(70\) kg, are sitting at the two extremes of a \(2\) m long boat, of mass \(70\) kg, standing still in the water as shown. They come to the middle of the boat. (Neglect friction). How far does the boat move on the water during the process?
| 1. | \(5\) cm leftward | 2. | \(5\) cm rightward |
| 3. | \(7\) cm leftward | 4. | \(7\) cm rightward |
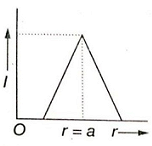
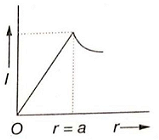
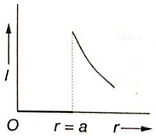
Which of the following graphs represents correctly the variation of intensity of gravitational field l with the distance r from the centre of a spherical shell of mass M and radius a?
1. 
2. 
3. 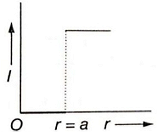
4. 
An elongation of 0.1% in a wire of cross-sectional area causes a tension of 100 N. The young's modulus is
1.
2.
3.
4.
A cylinder of height 20 m is completely filled with water. The velocity of efflux of water (in ) through a small hole on the side wall of the cylinder near its bottom is
1. 10
2. 20
3. 25.5
4. 5
A train moves towards a stationary observer at a speed of 34 m/s. The train sounds a whistle and its frequency registered is . If the train's speed is reduced to 17 m/s, the frequency registered is . If the speed of sound is 340 m/s , then the ratio is
1.
2.
3. 2
4.
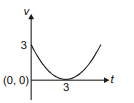
Position of a particle moving along the y-axis is given by , then which of the following represent the correct variation of speed (v) with respect to time (t)?
1. 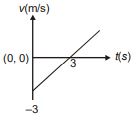 2.
2. 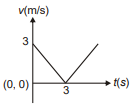
3. 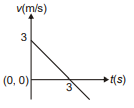 4.
4. 
A man moving in the west direction observes wind is blowing towards the south. If the man doubles his speed in the same direction, then the direction of wind with respect to man will be:
1. North-West
2. South-West
3. North-East
4. East-South
An ideal gas undergoes a cyclic process ABCA as shown.
The heat exchange between the system and the surrounding during the process will be:
| 1. | 10 J | 2. | 5 J |
| 3. | 15 J | 4. | 20 J |
The equivalent capacitance between the points \(A\) and \(B\) in the given network is:

1. \(25~\mu\text{F}\)
2. \(16~\mu\text{F}\)
3. \(21~\mu\text{F}\)
4. \(12~\mu\text{F}\)
A battery of 10 volt and internal resistance 30 is connected to a variable resistance R. The power dissipated in R is maximum when R is equal to -
1. 30
2. 15
3. 60
4. 20
A rod \(AB\) of length \(l\) is moving with constant speed \(v\) in a uniform magnetic field on a conducting \(U\)-shaped wire as shown. If the rate of loss of heat energy across resistance \(R\) is \(Q,\) then the force needed parallel to velocity to keep rod moving with constant speed \(v\) is:

1. \(Qv\)
2. \(\dfrac{Q}{v}\)
3. \(\dfrac{Q^2}{v}\)
4. \(Q^2v\)
Select the incorrect statement regarding magnetic material.
| 1. | The magnetic dipole moment of each atom of para and ferromagnetic substance is zero and that of diamagnetic material is non-zero in the absence of external magnetizing force |
| 2. | When the diamagnetic substance is brought near the north or south pole of a bar magnet, it is repelled |
| 3. | Magnetic susceptibility of diamagnetic substances is temperature independent and that of para and ferromagnetic substances is temperature-dependent |
| 4. | All of these |
A beam of light falls normally on a plane surface. This surface reflects 40% of incident light and the rest is absorbed. If the power of the incident beam is 24 W, then the force exerted by the beam on the surface is
1. 1.12 × N
2. 1.12 × N
3. 1.12 × N
4. 1.12 × N
When the frequency of incident radiation on a metal surface is v, the maximum KE of the photoelectron is . If the frequency of incident radiation is doubled, then the maximum KE becomes K. Select correct statement(s).
1. K = 2
2. K < 2
3. K =
4. K > 2
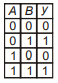
Which of the following truth table is correct for function y in the given combination of logic gates?
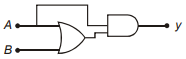
1. 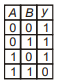 2.
2. 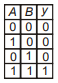
3. 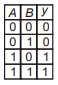 4.
4. 
A solid sphere with a velocity (of the centre of mass) v and angular velocity is gently placed on a rough horizontal surface. The frictional force on the sphere:
1. must be forward (in direction of v)
2. must be backward (opposite to v)
3. cannot be zero
4. None of these
A ring and a disc with the same moment of inertia roll along a plane surface at the same speed. If be the rotational kinetic energy of the ring and be that of the disc, then:
1. >
2. <
3. =
4. The relation depends upon the radii of the ring and disc
A truck of mass \(30,000~\text{kg}\) moves up an inclined plane of slope \(1\) in \(100\) \(\left(\tan\theta = \frac{1}{100}\right)\) at a speed of \(30~\text{km/h}\). The power of the truck is: (given \(g=10~\text{ms}^{-2}\)):
| 1. | \(25~\text{kW}\) | 2. | \(10~\text{kW}\) |
| 3. | \(5~\text{kW}\) | 4. | \(2.5~\text{kW}\) |
An ideal gas is taken through the process as shown in the figure. Then:
| 1. | In the process AB, the work done by the system is positive. |
| 2. | In process AB, heat is rejected out of the system. |
| 3. | In the process AB, internal energy increases. |
| 4. | In the process AB, internal energy decreases, and in the process BC, internal energy increases. |
A particle is executing SHM with an amplitude of 4 cm. At the mean position, velocity of the particle is 10 cm/s. The distance of the particle from the mean position when its speed becomes 5 cm/s is
1. cm
2. cm
3. 2 cm
4. 2 cm
The magnetic moment of a circular orbit of radius ‛\(r\)‛ carrying a charge '\(q\)' and rotating with velocity \(v\) is given by:
1. \(\frac{qvr}{2\pi}\)
2. \(\frac{qvr}{2}\)
3. \(qv\pi r\)
4. \(qv\pi r^2\)
If the Young's double slit experiment is performed with white light, then which of the following is not true?
1. the central maximum will be white
2. there will not be a completely dark fringe
3. the fringe next to the central will be red
4. the fringe next to the central will be violet
| 1. | \(1.0 \times 10^6 ~\text{V/m}\) | 2. | \(1.0 \times 10^5 ~\text{V/m}\) |
| 3. | \(2.0 \times 10^5 ~\text{V/m}\) | 4. | \(2.0 \times 10^6 ~\text{V/m}\) |
Which logic gate is represented by the following combination of logic gates?
1. OR
2. NAND
3. AND
4. NOR
A force F is given in terms of time t as F = AsinCt. The dimensions of are given by:
1.
2.
3.
4.
A body of density \(0.7\) g/cm3 floats on a lake of water. The fraction of the body that is outside water is:
1. \(30 \text{%}\)
2. \(70 \text{%}\)
3. \(25 \text{%}\)
4. \(50 \text{%}\)
An ideal fluid is flowing in a steady-state from section \(A\) to \(B\) through a pipe in a vertical plane as shown. Select the incorrect statement.
| 1. | Total energy per unit volume is the same at both sections A and B. |
| 2. | The incoming flow rate at A is equal to the outgoing flow rate at B. |
| 3. | Loss in kinetic energy of the fluid is equal to gain in potential energy from section A to section B. |
| 4. | The flow of fluid from A to B is laminar flow. |
The conducting shells A and B are arranged as shown below. If the charge on the shell B is q, then electric flux linked with the spherical Gaussian surface S is-
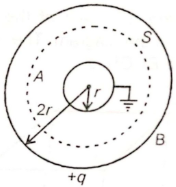
1.
2.
3.
4.
The time period of a simple pendulum in a stationary lift is T. If the lift moves upwards with an acceleration g, then the new time period will be:
1. Infinite
2.
3.
4. 0.707 T