A typical angiosperm anther is bilobed with each lobe having two theca i.e. dithecous. Theca is separated often by :
1. A longitudinal groove running lengthwise.
2. A longitudinal groove running breadth wise.
3. A transverse groove running breadth wise.
4. A transverse groove running lengthwise.
Four statements are given below. How many of them are correct?
(1) The anther is a tetragonal structure, with four microsporangia at corners, two in each lobe.
(2) Microsporangia develops further and becomes a pollen sacs.
(3) A typical microsporangia is surrounded by four layered wall, 3-protective and 1 nutritional.
(4) The nutritive layer is densely cytoplasmic with more than one nucleus
Options :
1. 3
2. 4
3. 2
4. 1
Which of the following structure is not associated with protection and dehiscence of anther?
1. Tapetum
2. Epidermis
3. Endothecium
4. Middle layer
For which of the following organisms there is no natural death ?
1. Bacteria reproducing by sporulation fission
2. Yeast reproducing by budding
3. Unicellular organisms reproducing by spores
4. Unicellular organisms reproducing by binary
Read the following statement carefully:
"Further development of zygote depends on the type of life cycle the organism has and the environment it is exposed to."
Identify the correctly matched pair with respect to the above statement
|
1. Thick walled zygote |
Haplontic life cycle |
|
2. Zygote forms new generation, by mitosis, represented by one cell |
Haplodiplontic life cycle |
|
3. Zygote undergoes meiosis to form haploid generation |
Diplontic life cycle |
|
4. Zygote forms multicellular diploid generation |
Haplontic life cycle |
In the given diagram the gas X can be:
1. Hydrogen sulfide
2. Carbon monoxide
3. Ammonia
4. Oxygen
In lactational amenorrhoea, there is no ovulation or menstruation during the period of intense lactation following parturition, due to high level of prolactin, which?
| 1. | Inhibits the release of gonadotropins |
| 2. | Inhibits the release of estrogen and progesterone |
| 3. | Stimulate the release of FSH and LH |
| 4. | Stimulates the release of estrogen and progesterone |
Triticale, the first man-made cereal crop has been obtained by crossing wheat with
(1) Rye
(2) Pearl millet
(3) Sugarcane
(4) Barley
To study whether a gene exhibits multiple allelism or not one must study:
1. An individual
2. A population
3. A species
4. With concentration
State True (T) or False (F) to the given statements and select the correct option
(A) Annual rings are distinct in plants growing in temperate regions.
(B) Lenticels occur in most woody trees and permit the exchange of gases.
(C) Due to stellar secondary growth, central cylinder of wood surrounded by secondary phloem is formed.
(D) The cells of endodermis opposite to protoxylem divide to give rise vascular cambium in dicot roots.
(A) (B) (C) (D)
1. T T F F
2. T T T F
3. T F T F
4. F F T T
Read the following statements about dicot stem and
choose the correct option.
| (a) | Vascular bundles are arranged in a ring. |
| (b) | Vascular bundles are conjoint, collateral and open type. |
| (c) | Endarch type of arrangement of secondary xylem. |
| 1. | Only (b) is correct |
| 2. | Only (c) is incorrect |
| 3. | Both (a) and (b) are incorrect |
| 4. | All three statements are correct |
Which of the following floral features is not represented
by symbols in a floral formula of a plant family?
1. Relative positions of ovary with respect to other parts.
2. Adhesion of stamens.
3. Aestivation of calyx and corolla
4. Symmetry of flower.
Select the incorrect match
1. Green photosynthetic - Australian Acacia
petiole
2. Leaflets attached at - Silk cotton
tip of the petiole
3. Papilionaceous corolla - Bean
4. Epiphyllous stamens - Brinjal
Which of the given feature is not related to mustard?
1. Superior ovary.
2. Variable length of filaments of stamens.
3. Parietal placentation.
4. Opposite phyllotaxy.
State True (T) or False (F) to the given statements and select the correct option
| (A) | Abundance of lichens in any area indicates that the area is highly polluted with SO2. |
| (B) | Mycobiont partner of lichens is always heterotrophic. |
| (C) | Body of lichens is made up of phycobionts only. |
(A) (B) (C)
1. T T F
2. F T F
3. F F F
4. T F F
Which of the given statement is not true for viruses?
| 1. | They are nucleoproteins where protein is infectious in nature. |
| 2. | They can be crystallized and crystals consist largely of proteins. |
| 3. | Virus means venom or poisonous fluid. |
| 4. | A virus can never have both DNA and RNA as its genetic material. |
Coralloid roots of gymnosperms are/have
1. Irregular and possess large number of roots hairs.
2. Symbiotic association with Rhizobium.
3. Symbiotic association with N2 - fixing cyanobacteria.
4. VAM
In pteridophytes, gametophyte that develops in the homosporous species is usually
1. Monoecious and has events, precursor to the seed habit.
2. Dioecious and does not lead to seed habit.
3. Monoecious and does not lead to seed habit.
4. Dioecious and has events, precursor to the seed habit.
Endoplasmic reticulum which is free of ribosomes is involved in all the given functions, except
| 1. | Synthesis of enzyme precursor for lysosomes. |
| 2. | Detoxification of drugs. |
| 3. | Uptake and release of Ca2+ ions during muscle contraction. |
| 4. | Glycogen synthesis. |
A membraneless cell organelle that is not found in higher plants is/has
| 1. | 9+2 arrangement of microtubules. |
| 2. | a role in formation of flagella in prokaryotes. |
| 3. | composed of rRNA and proteins. |
| 4. | involved in the formation of spindle fibers during cell division in animals. |
The "sodium-potassium pump" pumps ______.
1. sodium ions out and potassium ions in
2. sodium ions in and potassium ions out
3. sodium and potassium ions in
4. sodium and potassium ions out
Which of the following is not a characteristic of
the sympathetic system?
1. inhibits the digestive tract
2. dilates the bronchi
3. accelerates the heartbeat
4. constricts the pupil
The sliding filament theory of muscle contraction
involves
1. calcium ions releasing ATP for energy
2. calcium ions binding with troponin, which shifts
tropomyosin and allows the myosin-binding sites
on actin to be exposed
3. neuromuscular junctions directly producing the movement of actin and myosin
4. actin filaments moving the myosin filaments in
each sarcomere
Perhaps the biggest reason for the enormous success of Arthropods is?
1. Chitinous exoskeleton
2. Diverse appendages
3. Respiration by tracheoles
4. Internal fertilization
The filterable components of the blood that
move across from the glomerulus are______.
1. water, nitrogenous wastes, nutrients, and salts
2. blood cells and platelets and proteins
3. albumin, fats and other proteins
4. mainly blood cells
The maximum volume of air that can be moved
in and out during a single breath is called the
_________.
1. vital capacity
2. tidal volume
3. residual volume
4. dead space
The diaphragm and external intercostal muscles are ________ when expiration occurs.
1. contracted
2. relaxed
3. flexed
4. both relaxed (diaphragm) and flexed (intercostal
muscles)
When the heart beats, the familiar lub-dub
sound occurs as the valves of the heart _____.
1. open
2. close
3. expand
4. contract
The greater the number of blood vessels dilated, the ________.
1. higher the blood pressure
2. lower the blood pressure
3. faster the heartbeat
4. slower the heartbeat
The mucosa of the bladder is comprised of _____.
1. smooth muscle
2. squamous epithelium
3. transitional epithelium
4. simple columnar epithelium
Intercalated discs are associated with what type of tissues/muscles?
1. dense regular connective tissue
2. dense irregular connective tissue
3. cardiac muscle
4. skeletal muscle
Which cells are the first to phagocytize foreign
particles in the tissues such as bacteria in a
wound?
1. fibroblasts
2. white blood cells
3. macrophages
4. mast cells
One example of animals having a single opening
to the outside that serves both as mouth
and as anus is
1. Octopus
2. Asterias
3. Ascidia
4. Fasciola
These given statements are related with specific phase of meiosis, select the unrelated statement with the phase given below.
| Statement | Phase | ||
| 1. | The homologous chromosome separate, while sisterchromatids remain associated at their centromere |
– | Anaphase I |
| 2. | he bivalent chromosomes align on the equatorial plate | – | Metaphase I |
| 3. | Cytokinesis follows resulting in the formation of tetrad of cells | – | Telophase I |
| 4. | Terminalisation of chiasmata of prophase | – | Final stage |
Animals without excretory system, separate sexes, and calcareous endoskeleton are applicable for
1. Chaetopeura
2. Ophiura
3. Limulus
4. Spongilla
To make pancreatic enzyme in action, which of the following is required?
(i) Enterokinase (ii) Bile
(iii) Intrinsic factor (iv) HCl
1. (i), (ii), (iii) (iv)
2. (i), (ii) only
3. (i) only
4. (i), (iii) only
“This segment allows passage of small amounts of urea into the medullary interstitium to keep up the osmolarity”. Which segment does the statement suggest?
1. Descending limb of Henle’s loop.
2. Ascending limb of Henle’s loop.
3. Collecting duct.
4. Proximal convoluted tubule.
Eustachian tube connects:
1. External ear with middle ear
2. External ear with internal ear
3. Middle ear with pharynx
4. Middle ear with internal ear
Which one of the following is the correct description of a certain part of a normal human skeleton?
1. First vertebra is axis that articulates with the occipital condyles
2. The 9th and 10th pairs of ribs are called the floating ribs
3. Glenoid cavity is a depression to which the thigh bone articulates
4. Parietal bone and the temporal bone of the skull are joined by fibrous joint
Select the correct statement
| 1. | The base pairs in DNA are stacked 0.34 nm apart |
| 2. | The diameter of a right handed helical ds B-DNA molecules is 34 |
| 3. | Sugar-phosphate and hydrogen bonds are both present in a single nucleotide such as AMP |
| 4. | A and T of one strand compulsorily base pair with G and C respectively, of other strand is DNA |
Which is not a common mode of action between LNG20 and oral pill?
1. Inhibition of ovulation
2. Suppression of sperm motility and reduction of fertilizing ability of sperm
3. Alteration in quality of cervical mucus
4. Inhibition of implantation
Deficiency of which element causes the deficiency of nitrogen
1. Mo
2. K
3. Mn
4. S
The synthesis of one molecule of glucose during the Calvin cycle requires.
| 1. | 12 molecules of ATP and 18 molecules of NADPH2 |
| 2. | 6 molecules of ATP and 12 molecules of NADPH2 |
| 3. | 18 molecules of ATP and 12 molecules of NADPH2 |
| 4. | 12 molecules of each of ATP and NADPH2 |
Select the incorrect statement with respect to imbibition
1. It is diffusion process.
2. Affinity between the adsorbent and the liquid is not a pre-requisite.
3. It involves both capillary action and adsorption
4. Phycocolloids are best imbibants.
When a cell is placed in a solution whose osmotic concentration is equal to cell sap then:
1. Water moves inside the cell.
2. Water moves outside the cell.
3. No net movement of water occurs.
4. Cell will be plasmolysed.
Which if the following statement is not true regarding the plants?
| 1. | They show Kranz anatomy. |
| 2. | Decarboxylation process in bundle sheath cells. |
| 3. | Granal chloroplast is present in bundle sheath cells |
| 4. | PEPcase enzyme activity occurs in mesophyll cells. |
Vernalisation stimulates flowering in
1. Zamikand
2. Turmeric
3. Carrot
4. Ginger
Which of the following hormone is formed in the hypothalamus?
1. Adrenalin
2. Oxytocin
3. Insulin
4. Thyroxine
The enzymes of Krebs cycle where are produced are
| 1. | Isocitrate dehydrogenase succinate dehydrogenase and malic dehydrogenase. |
| 2. | Succinate thiokinase, succinate dehydrogenase and aconitase |
| 3. | Isocitrate dehydrogenase, |
| 4. | Isocitrate dehydrogenase, dehydrogenase and succinate dehydrogenase. |
Physical, psychomotor and mental development is retarded in an individual affected with
1. Down's syndrome
2. Sickle cell-anaemia
3. Turner's syndrome
4. Colour blindness
In protein synthesis process maximum unambiguously of genetic code is present when genetic code is:-
1. Singlet
2. Doublet
3. Triplet
4. Quadruplet
Adenohypophysis in humans consists of:
1. Pars distalis and Pars nervosa
2. Pars intermedia and Pars distalis
3. Pars nervosa and Pars intermedia
4. Anterior and posterior pituitary.
Which of the following enzymes is used in the case of fungus to cause the release of DNA along with other macromolecules?
1. Lysozyme
2. Cellulose
3. Chitinase
4. Amylase
The affected individuals are short saturated in disorders like
1. Turner's syndrome, phenylketonuria
2. Down's syndrome, Turner's syndrome
3. Klinefelter's syndrome, Down's syndrome
4. Turner's syndrome, Klinefelter's syndrome
Which of the following is not an invasive species?
1. Parthenium hysterophorus
2. Ocimum sanctum
3. Lantana Camara
4. Eicchornia crassipes
Which is not an effect of global warming
1. more extreme weather condition
2. melting of polar ice caps
3. Rise of sea level
4. Good fungal growth in soil
What are flocs?
1. Masses of anaerobic bacteria
2. Masses of aerobic fungi only
3. Masses of anaerobic bacteria and fungi
4. Masses of aerobic bacteria associated with fungal filaments
More conservative and scientifically sound estimate made by Robert May places the global species diversity at about
1. 1.5 million
2. 7 million
3. 1.7 million
4. 17 million
Consider the following statement and select correct set with respect to Global warming
a. El Nino effect
b. Cooling of stratosphere and earth surface
c. Shifting of climatic zones
d. Snow blindness
e. Impairment of hearing
f. Excessive deposition of ice at poles
g. Rise in sea level
h. Increases in eutrophication
1. a, c, e, g & h
2. a, c, d, e, f & g
3. a, b, c, d, f & h
4. a, c, g & h
Stanley Miller had put the Oparin-Haldane theory to test in 1953 by creating in the laboratory, the probable conditions on the primitive earth. In the experiment, simple amino acids were synthesized from which of the following mixtures, as observed after eighteen days?
1.
2.
3.
4.
Identify the gene and its protein that controls corn borer from the given options
1. cry I Ab and cry I Ab
2. cry I Ac and cry I Ab
3. cry II Ab and cry II Ab
4. cry I Ac and cry I Ac
The theory of spontaneous generation says that
1. Life originated from the decaying and rotting matter like straw, mud etc.
2. Life came on the earth from outer space
3. Life comes from pre-existing life only
4. Life started with the replication of self replicating metabolic capsules
If the initial amount of DNA is 8 C, then after S-phase the amount of DNA would be
1. 4 C
2. 8 C
3. 64 C
4. 16 C
Read the following statements:
| (a) | F 0 part of ATPase is associated with breakdown of proton gradient |
| (b) | A H-carrier contributes in creation of proton gradient |
| (c) | Movement of electrons in ETS is coupled to pumping of protons into the lumen |
| (d) | Formation of NADPH+ H+ is related with the creation of proton gradient |
How many of the above statements are correct?
1. Two
2. One
3. Four
4. Three
Choose the incorrect option for given below organism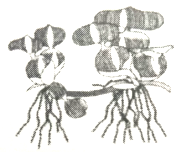
1. Scourge of the water bodies
2. Reproduction through offset
3. Found in running water
4. Drains oxygen from water
Sickle cell-anaemia disorder arises due to
1. Duplication of a segment of DNA
2. Substitution in a single base of DNA
3. Deletion of a segment of DNA
4. Duplication in a base pair of RNA
Mark the incorrect option with respect to lac operon
1. is under positive as well as negative control
2. controls catabolic pathway
3. shows feedback repression
4. Discovered by Jacob and Monod
In DNA fingerprinting, detection of hybridized DNA
1. Electrophoresis
2. Blotting
3. Autoradiography
4. Centrifugation
'Resource partitioning' is an important mechanism which promotes
1. Competitive release
2. Co-existence
3. Competitive exclusion
4. Antibiosis
Ecological succession is _________ and __________ change in species composition
1. Orderly and sequential
2. Unpredictable and orderly
3. Gradual and unsequential
4. Sequential and disorderly
Which one of the following shows maximum genetic diversity in India?
1. Mango
2. Groundnut
3. Rice
4. Maize
Recognizing the deleterious effects of ozone depletion an international treaty known as _______ was signed at ___________
1. Montreal Protocol, Canada
2. Kyoto Protocol, Brazil
3. Earth Summit, Montreal
4. World Summit, South Africa
Arrange CFC, in decreasing order according to their contribution in the greenhouse effect
1.
2.
3.
4.
Which of the following have been found to be very effective as emergency contraceptive as they could be used to avoid possible pregnancy due to rape if given within 72 hours?
A. Cervical caps
B. Progestogen-estrogen combination
C. Vaults
1. A only
2. A and B only
3. B and C only
4. B only
Natural selection in which more individuals acquire peripheral character value at both the ends of the distribution curve is
1. Stabilising natural selection
2. Disruptive natural selection
3. Directional natural selection
4. The curve never shows the formation of two peaks
Out of the following diseases which are caused due to bacterial infection?
| a. | Typhoid |
| b. | Elephantiasis |
| c. | Cholera |
| d. | Tuberculosis |
1. a & b only
2. b & c only
3. a, c & d only
4. a, b, c & d
Which of the following steps is/are catalysed by Taq polymerase in a PCR?
1. Denaturation of template DNA
2. Annealing of primers to template DNA
3. Extension of primer end on template DNA
4. All of these
Which carbohydrate splitting enzyme initiates the chemical process of digestion in the oral cavity?
1. Lysozyme
2. Salivary amylase
3. Pepsin
4. Rennin
Which one influences the activity of the kidney?
1. Vasopressin
2. Thyroxine
3. Vasopressin and aldosterone
4. Gonadotrophin
Which of the statement is incorrect with respect to the melatonin hormone?
1. Regulate diurnal variation
2. Influence metabolism
3. Secreted by pineal body
4. Non-influence pigmentation
Which hormone level reaches a peak during the luteal phase of the menstrual cycle?
1. Luteinizing hormone
2. Progesterone
3. Follicle-stimulating hormone
4. Estrogen
The mammary glands are paired structures (breasts) that contain glandular tissue and a variable amount of fat. The glandular tissue of the breast is divided into
1. 10-12 mammary lobes
2. 12-16 mammary tubules
3. 15-20 mammary alveoli
4. 15-20 mammary lobes
Which genotype will indicate colour blindness in male?
1.
2.
3.
4.
Female heterogamety found in :-
1. Birds
2. Drosophila
3. Human
4. Honey bee
In an operon, the RNA polymerase binds to
1. Regulator
2. Promoter gene
3. Operator gene
4. Constitutive gene
If an enzyme called beta-galactosidase is synthesized by E. coli, it is used to catalyze the hydrolysis of a disaccharide, lactose into:
1. Fructose
2. Galactose
3. Glucose
4. Both 2 and 3
Internal bleeding, muscular pain, fever, anaemia and blockage of the intestinal passage are the common symptoms of
1. Amoebiasis
2. Elephantiasis
3. Ascariasis
4. Typhoid
Which of the following is not a method of introducing alien DNA into host cells?
1. Microinjection
2. Heat shock method
3. Being placed along with the cell into a gene gun
4. Gel electrophoresis
Which of the following statement about Bacillus thuringiensis are correct?
A. One of the toxin produced by the bacteria is thurioside, which is active against different groups in insect larve.
B. The toxin accumulates inside the bacteria during sporulation
C. Upon ingestion by susceptible insects they are converted into active form and kill them by inhibition of ion transport in the midgut
D. The proteins encoded by the gene cry II Ab control corn borer
1. B only
2. A and B
3. A, B and C
4. A,B,C and D
Major event that occurs during anaphase of mitosis which brings about equal distribution of chromosomes is:
| 1. | Condensation of chromatin |
| 2. | Replication of genetic material |
| 3. | Splitting of centromere |
| 4. | Pairing of homologous chromosomes |
An absorption line of the lowest frequency in the transition of hydrogen spectra is:
1. n=1 to n=2
2. n=3 to n=8
3. n=2 to n=1
4. n=8 to n=3
Overlap of which of the following atomic orbitals would be maximum to form the strongest covalent bond.
1. 1s-2s (σ)
2. 1s-2p (σ)
3. 2p-2p (π)
4. 2p-2p (σ)
Which of the alkali metal chloride is having the highest m.p. ?
1. LiCl
2. NaCl
3. KCl
4. RbCl
A solution of CuCl in NH4OH is used to measure the amount of gas by simply measuring change in volume. The gas is
1. CO2
2. H2
3. CO
4. All of the above.
When allylic alcohol (CH₂=CH-CH₂OH) is oxidized in the presence of bromine, which of the following products will be formed?
1. Oxalic acid
2. Formic acid
3. Succinic acid
4. Acrylic acid
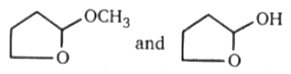
Above compounds can be differentiated by following reagent:
1. 2-4 DNP (Brady reagent)
2. Tollen's reagent
3. Bromine water reagent
4. NaHSO3
The concentration of the Ag+ ions in a saturated solution of Ag2C2O4 is 2.2 x 10-4 M. The solubility product of Ag2C2O4 is
1. 2.42 x 10-8
2. 2.66 x 10-12
3. 4.5 x 10-11
4. 5.3 x 10-12
30 ml of 0.06 M solution of the protonated form of an anion acid methionine (H2A+) is treated with 0.09 M NaOH. Calculate pH after addition of 20 ml of base.[ pKa1, = 2.28 and pKa2 = 9.2.]
1. 5.5
2. 5.74
3. 9.5
4. None of the above
The calomel electrode is reversible with respect to-
1. Mercury
2.
3.
4.
The number of isomeric sodium salt that will be required to obtain neopentane is:
1. 3
2. 1
3. 4
4. 6
Which of the following statement is not true about the effect of an increase in temperature on the distribution of molecular speeds of an ideal gas?
1. The area under the curve remains same even at the higher temperature.
2. The distribution pattern becomes more uniform.
3. Fraction of molecules with speed greater than a particular high speed will increase.
4. The fraction of molecules having most probable speed increases.
gas, molecular mass of gas A is:
1. 2
2. 32
3. 44
4. 28
Major product.
1.
2.
3.
4.
An aqueous solution of a protein has an osmotic pressure of 3.8 mm of Hg at temperature of 300 K. If concentration of protein is (w/v )then calculate molecular mass of protein.
1. 2000
2. 20000
3. 200
4. 1000
Which of the following reactions is a pseudo first order reaction?
1.
2.
3.
4.
The cation which is expected to act as ligand is:
1.
2.
3.
4.
The pair that has both members from the same period of the Periodic Table is
| 1. | Cl, Br | 2. | Ca, Cl |
| 3. | Na, Ca | 4. | Na, Cl |
An example of an oxide is
1. Zinc blende
2. Bauxite
3. Feldspar
4. Malachite
The electronic configuration that represents the maximum magnetic moment is:
1.
2.
3.
4. (high spin)
The major product in the above-mentioned reaction is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Ether is more volatile
than alcohol despite having the same molecular formula. This is due to -
1. Dipolar character of ethers
2. Alcohols having resonance structures
3. Intermolecular hydrogen bonding in ethers
4. Intermolecular hydrogen bonding in alcohols
Which of the test is used for the detection of secondary amines?
1.Libermann's nitroso test
2. Lucas test
3. Tollen's test
4. Carbylamine reaction
Mole percentage of in a non-stoichiometric oxide of iron. O, will be
1. 88%
2. 12%
3. 91.67%
4. 84.48%
Plastic household crockery is prepared by using :
1. Melamine and tetrafluoroethane.
2. Malonic acid and hexamethylene imine.
3. Melamine and vinyl acetate.
4. Melamine and formaldehyde.
The equivalent weight of for acid solution is
1. 79
2. 52.16
3. 158
4. 31.6
Which of the following statements regarding ozone is incorrect?
1. Powerful oxidising agent.
2. Bleaching agent
3. Paramagnetic
4. Bent shape
If volume containing gas is compressed to half, how many moles of gas remained in the closed vessel?
1. Just double
2. Just half
3. Same
4. More than double
Which is an extensive property?
1. Temperature
2. Chemical potential
3. Gibbs free energy
4. Molar volume
Which of the following elements has the highest second ionization enthalpy ?
1. Cr
2. Mn
3. Fe
4. Ti
During the adsorption of Krypton on activated charcoal at low temperature
1.
2.
3.
4.
The molecule among the following
that is an antidepressant is:
1. Chloroxylenol
2. Bithional
3. Cocaine
4. Penicillin-G
An ideal gas is expanded irreversibly against 10 bar pressure from 20 litres to 30 litres. Calculate 'w' if the process is isoenthalpic.
| 1. | 0 | 2. | +100J |
| 3. | -100 J | 4. | -10 kJ |

The IUPAC name of the above-mentioned compound is:
1. Pent-1-en-3-yne
2. Pent-1-ene-4-yne
3. Pent-4-yn-1-ene
4. Pent-1-en-4-yne
The IUPAC name of [\(\mathrm {C r \left(en\right)_{3}}\)]\(\mathrm{C l}_{3}\) is:
| 1. | Triethylenediaminechromium (III) chloride |
| 2. | Tris(ethylenediammine)chromium (II) chloride |
| 3. | Tris(ethylenediamine)chromium (III) chloride |
| 4. | Tris(ethylenediammine)chromium (I) chloride |
Alkali metals act as a :
1. Good dehydrating agent.
2. Good reducing agent.
3. Good oxidising agent.
4. None of the above.
Formula for agate is
1.
2.
3.
4.
Which of the following sulphur compounds can be used as a refrigerant?
1.
2.
3.
4.
In the equation,
the coefficients of are respectively:
| 1. | 6, 7 | 2. | 6, 14 |
| 3. | 5, 7 | 4. | 5, 14 |
How many EDTA (ethylenediaminetetraacetic acid) molecules are required to make an octahedral complex with a ion?
1. six
2. three
3. one
4. two
Which of the following does not react with periodic acid?
1.
2.
3.
4.
In a set of reactions, propanoic acid yielded a compound (D).
\(CH_3CH_2COOH\ \text{(A)} \xrightarrow[]{SOCl_2}\ \text{(B)} \)
\(\ \xrightarrow[]{NH_3}\ \text{(C)}\ \xrightarrow[Br_2]{KOH}\ \text{(D)}\)
The formula for (D) is:
1.
2.
3.
4.
Which of the following compounds, when heated at 483 K turns to caramel
1. Glucose
2. Sucrose
3. Fructose
4. Lactose
How many moles of magnesium phosphate, \(Mg_ 3 ( PO_ 4 )_ 2\) will contain 0.25 mole of oxygen atoms?
1. 0.02
2.
3.
4.
Which one of the following has a coordinate bond?
1.
2.
3.
4.
The reaction 2A + B + C D + E is found to be a first-order reaction with respect to A, second-order reaction with respect to B, and zero-order reaction with respect to C. If the concentrations of A, B, and C are doubled, the rate of the reaction will be:
1. 72 times
2. 8 times
3. 24 times
4. 36 times
Due to a small magnet , intensity at a distance x in the end on position is 9 Gauss. What will be the intensity at same distance in equatorial position ?
1. 9 Gauss
2. 4 Gauss
3. 36 Gauss
4. 4.5 Gauss
The displacement time graph of a moving particle is shown in the figure below. The instantaneous velocity of the particle is negative at the point:
| 1. | D | 2. | F |
| 3. | C | 4. | E |
A \(5\) m long uniformly thick string rests on a horizontal frictionless surface. It is pulled by a horizontal force of \(5\) N from one end. The tension in the string at \(1\) m from the end where the force is applied is:
| 1. | zero | 2. | \(5\) N |
| 3. | \(4\) N | 4. | \(1\) N |

Fluid flows through a pipe in a horizontal plane. The fluid has a relative density of \(0.9\) and its velocity at \(P\) (where cross-section area is \(A\)) is \(4\) m/s. What is its pressure at \(Q\)? (where cross-section area is \(\frac{A}{4},\) The pressure at \(P\) is \(2.8\times 10^{5}~\text{Pa})\)
1. \(2.36\times 10^{5}~\text{Pa}\)
2. \(3.88\times 10^{5}~\text{Pa}\)
3. \(2.08\times 10^{5}~\text{Pa}\)
4. \(1.72\times 10^{5}~\text{Pa}\)
A charged ball B hangs from a silk thread S which makes an angle with a large charged conducting sheet P, as shown in the figure. The surface charge density of the sheet is proportional to
1. cos
2. cot
3. sin
4. tan
In the figure shown, the value of so that electric field at centre ‘C’ is along ‘y’-axis only, where and are linear charge densities on semicircular and straight wire respectively, is -
1.
2. 2
3.
4.
The current in a coil varies with time \(t\) as \(I= 3 t^{2} +2t\). If the inductance of coil be \(10\) mH, the value of induced emf at \(t=2~\text{s}\) will be:
1. \(0.14~\text{V}\)
2. \(0.12~\text{V}\)
3. \(0.11~\text{V}\)
4. \(0.13~\text{V}\)
A student measures the value of g with the help of a simple pendulum using the formula g = . The errors in the measurements of L and T are L and T respectively. In which of the following cases is the error
in the value of g the minimum?
1. L = 0.5cm, T = 0.5s
2. L = 0.2cm, T = 0.2s
3. L = 0.1cm, T = 1.0s
4. L = 0.1cm, T = 0.1s
The ratio of rotational and translational kinetic energies of a solid sphere is
1.
2.
3.
4.
If a 50 kg mass is swinging in a vertical plane on a string at rest then the power delivered by gravity when the mass is moving with a velocity of 2 m/sec upwards in a direction, making an angle of with the vertical will be: (g = 9.8 m/)
| 1. | \(980 \mathrm{~W} \) | 2. | \(490 \mathrm{~W} \) |
| 3. | \(490 \sqrt{3}~ W \) | 4. | \(245 \mathrm{~W}\) |
A gas is found to obey the law = constant. The initial temperature and volume are . If the gas expands to a volume , its final temperature becomes:
1.
2.
3.
4.
Five equal capacitors connected in series have a resultant capacitance of \(4~\mu\text{F}\). The total energy stored in these when these are connected in parallel and charged to \(400\) V is:
1. \(1~\text{J}\)
2. \(8~\text{J}\)
3. \(16~\text{J}\)
4. \(4~\text{J}\)
In the following circuit, the battery \(E_1\) has an emf of \(12\) volts and zero internal resistance while the battery \(E\) has an emf of \(2\) volts. If the galvanometer \(G\) reads zero, then the value of the resistance \(X\) in ohms is:
| 1. | \(10\) | 2. | \(100\) |
| 3. | \(500\) | 4. | \(200\) |
A plane electromagnetic wave travels in free space along \(x\text-\)axis. At a particular point in space, the electric field along \(y\text-\)axis is \(9.3~\text{Vm}^{-1}.\) The magnetic induction is:
| 1. | \(3.1\times 10^{-8}~\text{T}\) | 2. | \(3\times 10^{-5}~\text{T}\) |
| 3. | \(3\times 10^{-6}~\text{T}\) | 4. | \(9.3\times 10^{-6}~\text{T}\) |
| 1. | \(8~\text{cm}\) inside the sphere | 2. | \(12~\text{cm}\) inside the sphere |
| 3. | \(4~\text{cm}\) inside the sphere | 4. | \(3~\text{cm}\) inside the sphere |
Boxes A and B are sliding to the right across a frictionless table.The hand H is slowing them down. The mass of A is larger than the mass of B. Rank in order, from largest to smallest, the horizontal forces on A, B and H
1.
2.
3.
4.
A force of 5 N making an angle with the horizontal acting on an object displaces it by 0.4 m along the horizontal direction. If the object gains kinetic energy of 1 J then the component of the force is:
| 1. | 1.5 N | 2. | 2.5 N |
| 3. | 3.5 N | 4. | 4.5 N |
The centre of mass of the triangle shown in figure has coordinates-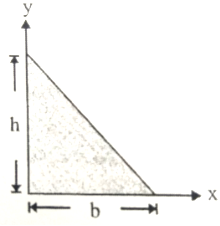
1.
2.
3.
4.
A car is moving on a circular level road of curvature 300 m. If the coefficient of friction is 0.3 and acceleration due to gravity is 10 , the maximum speed that car can have is-
1.
2.
3.
4.
Two rods, one made of aluminium and the other made of steel, having initial lengths \(l_1\) and \(l_2\) are connected together to form a single rod of length . The coefficient of linear expansion for aluminium and steel are and respectively. If the length of each rod increases by the same amount when their temperature is raised by \(t^\circ \mathrm{C},\) then the ratio \(\frac{l_1}{l_1+l_2}\) is:
1.
2.
3.
4.
The moment of inertia of a thin uniform circular disc about one of its diameter is I. Its moment of inertia about an axis perpendicular to the circular surface and passing through its center will be:
1.
2. 2 l
3.
4.
The network shown in figure is a part of a complete circuit. If at a certain instant, the current \(i\) is \(10\) A and is increasing at the rate of \(4\times 10^{3}\) A/sec, then \(V_A-V_B\) is:

| 1. | \(6\) V | 2. | \(-6\) V |
| 3. | \(10\) V | 4. | \(-10\) V |
An inductor \((L)\) and resistance \((R)\) are connected in series with an AC source. The phase difference between voltage \((V)\) and current \((i)\) is \(45^{\circ}.\)
If the phase difference between \(V\) and \(i\) remains the same, then the capacitive reactance and impedance of the \(LCR\) circuit will be:
1. \(2R, R\sqrt{2}\)
2. \(R, R\sqrt{2}\)
3. \(R, R\)
4. \(2R, R\sqrt{3}\)
In a Young's double slit experiment the intensity at a point where the path difference is ( being the wavelength of the light used) is I. If denotes the maximum intensity, is equal to :-
1.
2.
3.
4.
A block is connected to a relaxed spring and kept on a smooth floor. The block is given a velocity towards the right. Just after this:
| 1. | the speed of block starts decreasing but acceleration starts increasing. |
| 2. | the speed of the block as well as its acceleration starts decreasing. |
| 3. | the speed of the block starts increasing but its acceleration starts decreasing. |
| 4. | the speed of the block as well as acceleration start increasing. |
1. \(\dfrac{17g}{15}\)
2. \(\dfrac{19g}{10}\)
3. \(\dfrac{23g}{10}\)
4. \(\dfrac{11g}{10}\)
Three equal masses \(m\) are placed at the three vertices of an equilateral triangle of sides \(r.\) The work required to double the separation between masses will be:

| 1. | \(Gm^2\over r\) | 2. | \(3Gm^2\over r\) |
| 3. | \({3 \over 2}{Gm^2\over r}\) | 4. | None of the above |
In a photoelectric phenomenon, the number of photo-electrons emitted depends on
1. The intensity of incident radiation
2. The frequency of incident radiation
3. The velocity of incident radiation
4. The work function of the photo cathode
| 1. | total energy of electron in \(1\text{st}\) orbit of \(\mathrm{He}^{+}\) |
| 2. | total energy of electron in \(3\text{rd}\) orbit of \(\mathrm{He}^{+}\) |
| 3. | total energy of electron in \(2\text{nd}\) orbit of \(\mathrm{Li}^{++}\) |
| 4. | total energy of electron in \(3\text{rd}\) orbit of \(\mathrm{Li}^{++}\) |
C and Si both have same lattice structure, having 4 bonding electrons in each. However, C is insulator where as Si is intrinsic semiconductor. This is because
1. In case of C the valence band is not completely filled at absolute zero temperature
2. In case of C the conduction band is partially filled even at absolute zero temperature
3. The four bonding electrons in the case of C lie in the second orbit, whereas in the case of Si they lie in the fourth
4. The four bonding electrons in the case of C lie in the second orbit, whereas in the case of Si they lie in the third orbit
An ideal gas is taken through the cycle \(A\rightarrow B\rightarrow C\rightarrow A\) as shown in the figure below. If the net heat supplied to the gas is \(10~\text{J}\), then the work done by the gas in the process \(B\rightarrow C\) is:

| 1. | \(-10~\text{J}\) | 2. | \(-30~\text{J}\) |
| 3. | \(-15~\text{J}\) | 4. | \(-20~\text{J}\) |
A capacitor is connected to a battery. The force of attraction between the plates when the separation between them is halved
1. remains the same
2. becomes eight times
3. becomes four times
4. becomes double
In the circuit shown, the potential difference between points C and B will be
1. (8/9) V
2. (4/3) V
3. (2/3) V
4. 4 V
A microscope has an objective of focal length 1.5 cm and an eye-piece of focal length 2.5 cm. If the distance between objective and eye-piece is 25 cm, what is the approximate value of magnification produced for relaxed eye?
1. 75
2. 110
3. 140
4. 25
| 1. | Neutrons | 2. | Alpha particles |
| 3. | Beta particles | 4. | Gamma photons |
The circuit has two oppositely connected ideal diodes in parallel. What is the current flowing in the circuit?
1. 1.33 A
2. 1.71 A
3. 2.00 A
4. 2.31 A
Air is pushed into a soap bubble of radius r to double its radius. If the surface tension of the soap solution is S, the work done in the process is
1.
2.
3.
4.
A mass m is suspended from two springs of spring constant as shown in the figure below. The time period of vertical oscillations of the mass will be
1.
2.
3.
4.
A man grows into a giant such that his linear dimensions increase by a factor of \(9.\) Assuming that his density remains the same, the stress in the leg will change by a factor of:
1. \(\dfrac{1}{81}\)
2. \(9\)
3. \(\dfrac{1}{9}\)
4. \(81\)
A current of i Ampere is flowing through each of the bent wires as shown. The magnitude of the magnetic field at O is-
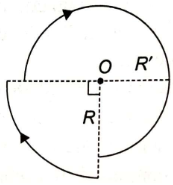
1.
2.
3.
4.
The direction of the magnetic force on the electron as shown in the diagram is along-
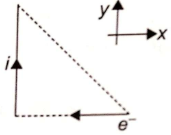
1. y-axis
2. z-axis
3. -y-axis
4. -z-axis
A light of wavelength \(\lambda\) in air enters a medium of refractive index \(\mu.\) Two points in this medium lying along the path of this light are at a distance \(x\) apart. The phase difference between these points is:
1. \(\dfrac{2 \pi \mu x}{\lambda}\)
2. \(\dfrac{2 \pi x}{\mu \lambda}\)
3. \(\dfrac{2 \pi \left(\mu - 1\right) x}{\lambda}\)
4. \(\dfrac{2 \pi x}{\left(\lambda - 1\right) \lambda}\)