In vexillary aestivation:
1. The standard overlaps the wings
2. The standard overlaps the keel
3. The standard is overlapped by keel
4. The keel overlap the wings
Identify the incorrect statement:
| 1. | The presence of vessels is a characteristic of angiosperms |
| 2. | In roots the primary xylem is exarch |
| 3. | Gymnosperms lack albuminous cells and sieve cells |
| 4. | Bast fibers are generally absent in primary phloem |
Which of the following does not help in excretion in Periplaneta Americana?
1. Fat body
2. Urecose glands
3. Nephrocytes
4. None of these
There is a possibility of exposure of the maternal blood [of Rh -ve mother] to small amounts of Rh +ve blood from the fetus during the:
1. First trimester of any pregnancy
2. Third trimester of the second pregnancy only
3. Delivery of the child
4. There is no such possibility
Identify the incorrect statement :
| 1. | Heterotrophic bacteria are important decomposers |
| 2. | Diatoms are chief producers in the oceans |
| 3. | The pigments of euglenoids are identical to those present in higher plants |
| 4. | Very few protozoans are autotrophs |
Which of the following would not be a character of Asparagus?
1. Actinomorphic flower
2. Imbricate aestivation
3. Superior ovary
4. Endospermous seed
Dense irregular connective tissue is present in:
1. Tendons
2. Ligaments
3. Skin
4. Cartilage
All the following are the part of a eukaryotic cell endomembranous system except:
1. Vacuole
2. Lysosome
3. Golgi complex
4. Mitochondria
Which of the following is/are essential for imbibitions to take place?
I. Water potential gradient between the absorbent and the liquid imbibed
II. Affinity between the adsorbant and the liquid
1. Only II
2. Only I
3. Both I and II
4. None
How many ATP molecules are required to biologically fix one molecule of nitrogen into two molecules of ammonia?
1. 4
2. 8
3. 12
4. 16
Pneumotaxic centre which can moderate the functions of the respiratory rhythm centre is present at:
1. Pons region of brain
2. Thalamus
3. Spinal cord
4. Right cerebral hemisphere
Congestion of the lungs is one of the main symptoms in:
1. Hypotension
2. Coronary artery disease
3. Angina
4. Heart failure
If global warming continues, in general, how what would be the expected effect on the distributional range of some species ?
1. It will be an equatorial shift
2. It will be a pole ward shift
3. It will have no impact
4. It will affect only plants
What percent of energy in the sunlight availble to the primary producers is converted by them into net primary productivity ?
1. 1
2. 2
3. 5
4. 10
Both hydrach and xerarch successions:
1. Take same time
2. Have similar sere
3. Lead to mesic conditions
4. Have same pioneer species
The second trophic level in a lake is:
1. Phytoplankton
2. Zooplankton
3. Benthos
4. Fishes
The relation between species richness and area for a wide variety of taxa on a logarithmic scale is a:
1. rectangular hyperbola
2. straight line
3. sigmoid curve
4. sine curve
The intine of the pollen grain is a:
1. Thick and discontinuous layer made of sporopollenin
2. Thick and continuous layer made of sporopollenin
3. Thin and discontinuous layer made of cellulose and pectin
4. Thin and continuous layer made of cellulose and pectin
In the monosporic embryo sac of a flowering plant, the number of nuclei that get surrounded by cell walls and thus are organized into cells is:
1. 5
2. 6
3. 7
4. 8
Female flowers remain submerged in water and the pollen grains are released inside the water in:
1. Sea grasses
2. Vallisneria
3. Water lily
4. Water hyacinth
Scientists are trying to transfer apomictic genes into hybrid varieties of several of our food and vegetable crops so that:
1. They get resistance against common pathogens
2. The yield of these plants can be increased many folds
3. Hybrid seeds need not be produced every year
4. Their germplasm is easily stored in labs for further research
Which human male accessory reproductive duct receives a duct from the seminal vesicle?
1. Rete testis
2. Vas deferens
3. Epididymis
4. Urethra
The primary oocyte completes its first meiotic division within the:
1. Primary follicle
2. Secondary follicle
3. Tertiary follicle
4. Graafian follicle
Periodic abstinence is a natural method of contraception where the couples avoid or abstain from coitus during the period of the menstrual cycle when:
1. There is menstrual flow
2. The ovulation is most likely to occur
3. The corpus luteum is most active
4. The proliferative phase begins
Aneuploidy results from :
1. Point mutations
2. Gross structural changes in chromosomes
3. Failure of cytokinesis after telophase stage of cell division
4. Failure of segregation of chromatids during cell division
Identify the correct statement:
| 1. | Dryopithecus was more man like than Ramapithecus |
| 2. | Homo Habilis probably did not eat meat |
| 3. | Homo Erectus had a cranial capacity of around 800 cc |
| 4. | Agriculture came around 18000 years ago |
Which of the following Fungi is not capable of causing ringworm infection in a man?
1. Microsporum
2. Trichophyton
3. Epidermophyton
4. Trichoderma
Identify the incorrectly matched pair
|
1. |
Baculoviruses |
Species specific, broad spectrum insecticides |
|
2. |
Trichoderma |
Free living fungi common in root ecosystems |
|
3. |
Ladybird |
Biocontrol of aphids |
|
4. |
Organic farming |
Pests kept at manageable levels rather than completely eradicated |
An important biofertilizer in paddy fields is :
1. Azospirillum
2. Azotobacter
3. Anabaena
4. Rhizobium
Which of the following restriction sites is located within the gene for tetracycline resistance in the plasmid pBR322?
1. BamHI
2. Psti
3. Clal
4. Pvull
To protect tobacco plants against Meloidegyne incognita using RNAi, nematode specific genes were introduced in the plant using:
1. Transposons
2. Retrotransposons
3. Virus with RNA genome
4. Agrobacterium
The technique not used for transformation of plant cells in recombinant procedures is:
1. Biolistics
2. Agrobacterium mediation
3. Use of viruses
4. Micro-injection
In mosses, the sex organs are seen in:
1. Protonema stage
2. Leafy stage
3. Capsule of the sporophyte
4. Seta of the sporophyte
The features seen in the gymnosperm Cycas include:
| I. | Coralloid roots |
| II. | Unbranched stems |
| III. | Pinnate persistent leaves for a few years |
| IV. | Male cones and megasporophyllys borne on same plant |
1. I and II only
2. I and IV only
3. I, II and III only
4. I, II, III and IV
Fat soluble pigments like xanthophylls are present in:
1. Chromoplasts
2. Aleuroplasts
3. Elaioplasts
4. Amyloplasts
Muscularis layer in the wall of the alimentary canal is formed by smooth muscles usually arranged into:-
1. an inner circular and an outer longitudinal layer.
2. an inner longitudinal and an outer circular layer.
3. both circular layers
4. both longitudinal layers
During parturition, the fetal ejection reflex triggers the release of:
| 1. | An increasing amount of estrogen and a decreasing amount of progesterone from the placenta |
| 2. | A decreasing amount of estrogen and an increasing amount of progesterone from the placenta |
| 3. | Oxytocin from the maternal pituitary |
| 4. | GnRH from the fetal hypothalamus |
The skeletal structure shown in the given diagram shows the structure of :
| 1. | Morphine | 2. | Diacetyl morphine |
| 3. | Cannabinoid | 4. | Cocaine |
Identify the incorrectly matched pair:
|
|
Crop Variety |
Resistant to |
|
1. |
Himgiri |
Leaf and stripe rust |
|
2. |
Karan rai |
White rust |
|
3. |
Pusa sadabahar |
Bacterial blight |
|
4. |
Pusa shubhra |
Black rot |
In general, which of the following is not an adaptation seen in parasites in accordance with their life style?
1. Loss of unnecessary sense organs
2. Presence of adhesive organs
3. Low reproductive capacity
4. Loss of digestive system
Which of the following taxonomic categories contains organisms least similar to one another?
1. Class
2. Genus
3. Family
4. Species
The six most common atoms in organic molecules are:
| 1. | Carbon, Hydrogen, Oxygen, Helium, Calcium and Sulphur |
| 2. | Carbon, Hydrogen, Oxygen, Calcium, Magnesium and Sulphur |
| 3. | Carbon, Oxygen, Nitrogen, Sulphur, Phosphorous and Magnesium |
| 4. | Carbon, Hydrogen, Oxygen, Nitrogen, Phosphorous and Sulphur |
Proteins that are to be used outside the cell are synthesized:
| 1. | in the mitochondria |
| 2. | on the rough endoplasmic reticulum |
| 3. | on the smooth endoplasmic reticulum |
| 4. | on free ribosomes |
The main control over ATP production is allosteric inhibition, by ATP, of the enzyme that facilitates the formation of :
| 1. | Glucose from glycogen |
| 2. | Fructose 1,6 biphosphate from Fructose 6 phosphate |
| 3. | Acetyl coenzyme A from pyruvic acid |
| 4. | Citric acid from Acetyl coenzyme A and oxaloacetic acid. |
Genetic information in a DNA molecule is coded in the:
1. Sequence of nucleotides
2. Base pairings
3. Proportion of each base present
4. The turning pattern of the helix
In DNA replication, the Okazaki fragments on the lagging strand are joined together by:
1. DNA ligase
2. DNA polymerase
3. Primase
4. Helicase
The metabolic activity of the different types of cells within an organism varies because of differences in the kinds of :
1. Genes in each cell
2. Ribosomes in each cell
3. Enzymes in each cell
4. Nucleoli in each cell
The enzyme that catalyzes the peptide bonding in prokaryotes is located in the:
1. Leader region of the mRNA
2. Central part of tRNA
3. Smaller subunit of the ribosome
4. Larger subunit of the ribosome
If a cell were treated with a chemical that blocked nucleic acid synthesis, which of the following processes would most likely be affected first?
1. DNA replication
2. tRNA synthesis
3. mRNA synthesis
4. Protein synthesis
In general, bacterial genes are regulated at the time of:
1. Transcription
2. Post transcription
3. Translation
4. Post translation
Restriction enzymes are synthesized by:
1. Bacteria only
2. Yeast and bacteria only
3. Eukaryotic cells only
4. All kinds of cells
At what phase of meiosis are homologous chromosomes separated ?
1. Prophase l
2. Prophase ll
3. Anaphase l
4. Anaphase ll
One of the Mendel's pure strains of pea plants had green peas. How many different types of eggs could such a plant produce with regard to pea color?
1. 1
2. 2
3. 4
4. 8
A woman receives her X chromosomes from:
1. Her mother only
2. Her father only
3. Both her mother and father
4. Mitochondria of mother only
Most fossils are found in:
1. Granite
2. Sedimentary rocks
3. Lava flows
4. Black soil
Darwin believed that certain parts of the body get larger and more complex through the generations because they :
1. are used more extensively than other parts
2. contribute to greater reproductive success
3. are pre-determined to do so
4. are most similar to God's perfection
Cells in some filamentous cyanobacteria that are specialized for nitrogen fixation are called:
1. Phycobilisomes
2. Chromatophores
3. Grana
4. Heterocysts
The protozoans that cause malaria in humans are :
1. Radiolarians
2. Dinoflagellates
3. Chrysophytes
4. Sporozoans
Lignin is a component of the secondary walls of:-
1. Epidermis
2. Collenchyma
3. Sclerenchyma
4. Parenchyma
The asexual production of seeds is called as:
1. Adventition
2. Fragmentation
3. Self-fertilization
4. Apomixis
Most primary productivity in the surface water of lakes and oceans is by:
1. Floating plants
2. Red and brown algae
3. Zooplankton
4. Phytoplankton
Describe urine in relationship to glomerular filtration tubular reabsorption and tubular secretion urine:
| 1. | glomerular filtration + tubular reabsorption + tubular secretion |
| 2. | glomerular filtration - tubular reabsorption -tubular secretion |
| 3. | glomerular filtration + tubular reabsorption -tubular secretion |
| 4. | glomerular filtration -tubular reabsorption +tubular secretion |
Atrial natriuretic hormone (ANH) will promote a(n) ________ in the excretion of sodium and water; thus ________ blood volume and blood pressure.
1. decrease, decreasing
2. decrease; increasing
3. increase, decreasing
4. increase, increasing
Which of the following gastric secretory cells is correctly matched with its secretion(s)?
1. Chief cells - gastrin
2. parietal cells -- hydrochloric acid and intrinsic factor
3. enteroendocrine cells -- mucus and hormones
4. mucous cells -- pepsinogen and mucus
The neurotransmitter released by the nerve cell at the neuromuscular junction is:
1. Acetylcholine
2. Adrenaline
3. Serotonin
4. Dopamine
Match the columns A and B:
| Column I | Column II | ||
| (a) | Marginal placentation | (i) | Marigold |
| (b) | Axile placentation | (ii) | Dianthus |
| (c) | Parietal placentation | (iii) | Argemone |
| (d) | Free central placentation | (iv) | Chinarose |
| (e) | Basal placentation | (v) | Pea |
| (a) | (b) | (c) | (d) | (e) | |
| 1. | (v) | (iv) | (iii) | (ii) | (i) |
| 2. | (v) | (iii) | (iv) | (i) | (ii) |
| 3. | (iv) | (iii) | (v) | (ii) | (i) |
| 4. | (i) | (v) | (iv) | (iii) | (ii) |
ACTH controls the secretion of:
1. cortisol
2. aldosterone
3. epinephrine
4. testosterone
Which of the following is not likely to happen as a result of the rising levels of in our atmosphere?
1. Increased incidents of skin cancer
2. Rise in sea levels
3. Change in climatic patterns
4. Shifts in locations of deserts and fertile regions
An inferior ovary:
| 1. | is situated on the receptacle above the perianth and androecium. |
| 2. | is positioned below the sites of attachment for perianth and androecium. |
| 3. | forms a hypanthium. |
| 4. | is positioned below the receptacle. |
In the given diagrammatic representation of ATP synthesis in mitochondria, A, B and C are respectively:
1.
2.
3.
4.
In the diagrammatic representation of the Hatch and Slack pathway, A and B respectively represent:
| 1. | Mesophyll cells and Bundle sheath cells |
| 2. | Bundle sheath cells and Mesophyll cells |
| 3. | Bundle sheath cells and Palisade cells |
| 4. | Mesophyll cells and Guard cells |
The following cell undergoing mitosis, is at:
| 1. | Early prophase | 2. | Late prophase |
| 3. | Transition to metaphase | 4. | Early metaphase |
What is true for both (a) and (b)?

1. RNA is the genetic material
2. Capability to infect bacteria
3. Being obligate endoparasites
4. Can be killed by antibiotics
The following pie chart shows the relative contribution of radiatively active gases to greenhouse effect. The contribution of is shown by:
1. A
2. B
3. C
4. D
The following diagram shows the effect of sewage discharge on some important characteristics of a river. What can be A and B respectively?
1. BOD and DO
2. DO and BOD
3. BOD and COD
4. COD and BOD
Which of the following would not be a feature seen in a patient with the following karyotype ?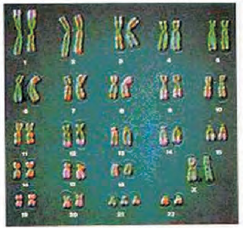
1. Many loops on finger tips
2. Congenital heart disease
3. Big and wrinkled tongue
4. Mucus clogging of airways
Given below is the initial amino acids of the beta chain of a hemoglobin molecule. What will be true about this hemoglobin molecule ?
| 1. | The RBCs carrying this molecule will undergo sickling at high oxygen tension |
| 2. | The RBCs carrying this molecules will undergo sickling at low oxygen tension |
| 3. | There will be a quantitative decrease in the synthesis of this molecule |
| 4. | This is normal beta chain of the hemoglobin molecule |
Consider the following statements:
| I. | Accessory pigments enable a wider range of light to be utilized for photosynthesis. |
| II. | The reaction center of PSI is P680 and that of PSII is P700. |
| III. | The stroma lamellae membranes lack both PSII and NADP reductase. |
Which of the above statements are true?
| 1. | l and ll only |
| 2. | l and lll only |
| 3. | ll and lll only |
| 4. | l, ll and lll |
Consider the following statements :
I. Many bony fishes, aquatic amphibians, and aquatic insects are ammonotelic.
II. Mammals, many terrestrial amphibians, and cartilaginous fishes are ureotelic.
III. Reptiles, birds, land snails, and insects are uricotelic.
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II and III
Consider the following statements:
| I. | Myasthenia gravis is an autoimmune disorder affecting the neuromuscular junction |
| II. | Muscular dystrophy is a progressive degeneration of skeletal muscles mostly due to a genetic disorder |
| III. | Tetany is rapid spasms in muscles due to high Ca++ in body fluid. |
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II and III
Consider the following statements:
I. Proximal radioulnar joint is a type of ellipsoidal joint.
II. The first carpo-metacarpal joint is a saddle joint.
III. Gleno-humeral joint is a ball and socket joint.
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II, and III
Consider the following statements:
I. Somatic neural system relays impulses from CNS to skeletal muscles.
II. Autonomic neural system transmits impulses from CNS to involuntary organs and smooth muscles.
III. Unmyelinated nerve fibres are commonly found in spinal and cranial nerves.
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II, and III
Consider the following statements:
| I: | The resting axonal membrane is nearly impermeable to sodium ions. |
| II: | Depolarization of the axonal membrane is due to the influx of sodium ions. |
| III: | The size of the action potential, if produced, does not depend on the strength of the stimulus. |
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II and III
Consider the following statements:
I. Acetylcholine is released at the electric synapse.
II. Electric synapses are faster.
III. It is more difficult to regulate an electric synapse with respect to a chemical synapse.
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II, and III
Consider the following statements:
I. Philadelphia chromosome is characteristically seen in Chronic Myeloid Leukemia.
II. Inversions are used to maintain balanced lethal systems.
III. It was actually a series of reciprocal translocations that was observed by Hugo de Vries in evening primrose
which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II and III
Match each item in Column I with one item in Column II and choose your answer from the codes given below:
| Column I | Column II | ||
| I. | Influx of sodium | 1. | Original establishment of RMP |
| II. | Efflux of potassium | 2. | Repolarization |
| III. | Na+/K+ pump | 3. | Depolarization |
| IV. | Ca++ | 4. | Plateau in cardiac muscles |
Codes :
| I | II | III | IV | |
| 1. | 3 | 2 | 4 | 1 |
| 2. | 2 | 1 | 4 | 3 |
| 3. | 2 | 1 | 3 | 4 |
| 4. | 3 | 2 | 1 | 4 |
Which one of the following phenomena supports Darwin's concept of natural selection in organic evolution -
1. production of 'Dolly', the sheep by cloning
2. Development of organs from 'stem' cells for organ transplantation
3. Development of transgenic animals
4. Prevalence of pesticide resistant insects
Mark the mis-matched pair
1. Polycarpic plants - Recovery phase
2. Offset - Water hyacinth
3. Marchantia - Monoecious plant
4. Bulbil - Agave
Low intelligence quotient abnormal skin and deafmutism are related to:
1. Low secretion of growth hormone
2. Hypothyroidism
3. Hyperparathyroidism
4. Hyposecretion of adrenal cortex hormone
Extracellular receptors must be required for the action of:
I. Oxytocin
II. Thyroxine
III. Epinephrine
IV. Glucagon
1. I, II, III & IV
2. I, IV only
3. I, III, IV
4. III & IV only
The correct order in which the O-O bond length increases in the following is
1. H2O2 < O2 < O3
2. O3 < H2O < O2
3. O2 < O3< H2O2
4. O2 < H2O2 < O3
Rate of diffusion of LPG (a mixture of n-butane and propane) is 1.25 times of SO3. Hence, mass fraction of n-butane in LPG is
1. 0.75
2. 0.25
3. 0.50
4. 0.67
One mole of methanol when burnt in O2 gives out 723 kJ mol-1 of heat. If one mole of O2 is used, what will be the amount of heat evolved?
1. 482 kJ
2. 241 kJ
3. 723 kJ
4. 924 kJ
Carbon suboxide (C3O2) has
1. Linear structure
2. Bent structure
3. Trigonal structure
4. Disorted tetrahedral structure
How many geometrical isomers are possible of the following?
CH3-CH=CH-CH=CH-CH3
1. 2 2. 3
3. 4 4. 6
The alkene among the following that has the smallest heat of hydrogenation is-
1.
2.
3.
4.
What is the current efficiency of an electrode deposition of Cu metal from CuSO4 solution in which 9.8 gm copper is deposited by the passage of 5 amperes current for 2 hours?
1. 41.4 %
2. 50%
3. 75%
4. 82.8 %
Mond's process is used for
1. Ni
2. Al
3. Fe
4. Cu
The number of S-S bonds in the trimer of sulphur trioxide is/are
1. zero
2. 1
3. 2
4. 3
Which nitro derivative of haloarene is most reactive towards nucleophilic substitution reaction?
1. 
2.
3.
4.
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Number of Chiral carbon atoms in cyclic b-D(+)-glucose is
1. 5
2. 4
3. 3
4. 2
Which among the following is most basic?
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
In the below-mentioned reaction, the major product (A) formed is:


| 1. |   |
2. |   |
| 3. |   |
4. | None of these |
To a 200 ml of 0.1 M weak acid HA solution 90 ml of 0.1 M solution of NaOH be added, Now, what volume of 0.1 M NaOH be added into above solution so that pH of resulting solution be 5.
[Ka(HA) = 10-5]
1. 2 ml
2. 20 ml
3. 10 ml
4. 15 ml
The configuration of given tartaric acid is

1. 2R, 3R 2. 2R, 3S
3. 2S, 3S 4. 2S, 3R
Consider the given reaction:
 Products formed
Products formed
| (A) |  |
(B) |  |
| (C) |  |
(D) |  |
1. (A) and (B) only 2. (A) and (C) only
3. (B) and (C) only 4. (A), (B), (C) and (D)
The dissociation of NH4OH can be suppressed by the addition of:
| 1. | NH4Cl | 2. | NH4NO3 |
| 3. | NaOH | 4. | All of the above |
What amount of CaCl2(i=2.47) is dissolved in 2L water so that its osmotic pressure is 0.5 atm at 27:-
1. 13.42 g
2. 19.24 g
3. 8.834 g
4. 1.820 g
On heating Li2CO3 and Na2CO3.10H2O, residue is left after heating. The weight loss is due to
1. Evolution of CO2 from Na2CO3.10H2O only
2. Evolution of CO2 from Li2CO3 only
3. Due to evolution of CO2 from Li2CO3 and H2O from Na2CO3.10H2O
4. H2O from Na2CO3.10H2O
Which among the following species has the highest ionization energy?
1. Ne
2. F
3. Li
4. B
If 'a' is the initial concentration and K is the rate constant of a zero-order reaction, the time for the reaction to go to completion will be
1.
2.
3.
4.
If for water are 1.86 and 0.52 K.kg.mol−1 respectively. The difference between boiling and freezing point of the aqueous solution be 102.38 then the solution is
1. 50 g of 1 molal KCI solution
2. 50 g of 2 molal urea solution
3. 1000 g of 1 molal glucose solution
4. All of these
Williamson's synthesis of ethers is an example of
1. Nucleophilic substitution reaction
2. Nucleophilic addition
3. Electrophilic addition
4. Electrophilic substitution
Calculate moles of ICI and ICI3 formed respectively if 25.4 g of and 14.2 g of are made to react completely to yield a mixture of ICI and ICI3:
[Given: Molecular mass of I2 -254, Cl2 - 71]
1. 0.5 and 0.2
2. 0.1 and 0.1
3. 0.1 and 0.3
4. 0.3 and 0.4
Which of the following exhibits a pπ-dπ bond?
| 1. | \(\mathrm{NO}_3^{-}\) | 2. | \( \mathrm{BO}_3^{3-} \) |
| 3. | \(\mathrm{CO}_3^{2-} \) | 4. | \(\mathrm{SO}_3^{2-} \) |
The wavenumber of the first line in the Balmer series of hydrogen is 15200 . The wavenumber of the first line in the Balmer series of is
1. 15200
2. 69800
3. 76000
4. 136,800
The hybridised state of Cu in is :
1.
2.
3.
4.
Packing fraction in bcc is
1. 0.52
2. 0.74
3. 0.60
4. 0.68
Which one of the following complexes is an outer orbital complex?
1.
2.
3.
4.
Which of the following gives a yellow precipitate with :
1.
2.
3.
4. All of the above
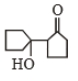
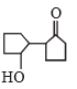
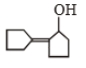
Self condensation of cyclopentanone is presence of dil. NaOH follwoed by heating gives
1. 
2. 
3. 
4. 
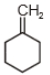
The major product in the acid catalysed dehydration of 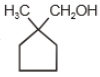 would be
would be
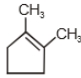
1. 
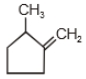
2. 
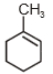
3. 
4. 
During electrolysis, 2A current is passed through an electrolytic solution for 965 s. The number of moles of electrons passed will be
1. 0.02
2. 0.01
3. 200
4. 0.037
Polyethene, PVC, teflon and neoprene are example of -
1. Homopolymer.
2. Addition hetropolymer.
3. Biodegradable polymer.
4. Copolymer.
The chemical added to soaps to impart antiseptic properties is :
1. Salvarsan
2. Bithional
3. Paracetamol
4. Iodine
Boric acid is an acid because its molecule
1. Contains replaceable ion
2. Gives up a proton
3. Accepts from water-releasing proton
4. Combines with proton from water molecule
The equivalent weight of in the reaction is
1. M/16
2. M/4
3. M/20
4. M/10
The structure of glycine (amino acid) (Zwitter ion) select the correct statement of the following.
1. Glycine, as well as other amino acids are amphoteric.
2. The acidic functional group in amino acids is
3. The basic functional group in amino acids is
4. All the statements are correct .
The kinetic energy for 14 grams of nitrogen gas at 127°C is nearly -
(mol. mass of nitrogen = 28 and gas constant = 8.31JK–1mol–1)
1. 1.0 J
2. 4.15 J
3. 2493 J
4. 3.3 J
is 60% ionised. Then the value of Van't Hoff factor is
1. 1.6
2. 2.4
3. 3
4. 3.4
The aqueous solution containing which one of the following ions will be colourless -
1. Fe2+
2 Mn2+
3. Ti3+
4. Sc3+
[Atomic number : Sc = 21, Fe = 26, Ti = 24, Mn = 25]
The number of moles of KMnO4 reduced by one mole of KI in alkaline medium is :-
1. One
2. Two
3. Five
4. One fifth
Which of the following pairs of a chemical reaction is certain to result in a spontaneous reaction -
1. Endothermic and decreasing disorder
2. Exothermic and increasing disorder
3. Endothermic and increasing disorder
4. Exothermic and decreasing disorder
In an adsorption experiment, a graph between log(x/m) versus log P was found to be linear with a slope of 45. The intercept on the y-axis was found to be 0.3.
What will be the value of x/m at a pressure of 3 atm for the Freundlich adsorption isotherm when 1/n remains constant?
[Antilog 0.3 = 2]
1. 2
2. 4
3. 6
4. 5
The incorrect statement among the following regarding reaction of is -
1. (acidic medium)
2.
3. (acidic medium)
4.
Two simple pendulums have time periods T and . They start vibrating at the same instant from the mean position in the same phase. The phase difference between them when bigger pendulum completes one oscillation will be:
1.
2.
3.
4.
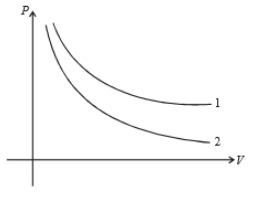

P-V graph of two gases during adiabatic processes are shown in figure. Plots 1 and 2 should correspond, respectively to :
1. He,
2. and He
3. He and Ar
4. and
| 1. | \(\frac{m_1}{m_2} \) | 2. | \(\frac{m_2}{m_1} \) |
| 3. | \(1 \) | 4. | \(\sqrt{\frac{\mathrm{m}_2}{\mathrm{~m}_1}}\) |
Heavier nuclei are unstable. The correct statement is :
1. binding energy per nucleon is small
2. get stability by nuclear fission
3. get stability by nuclear fusion
4. both (1) and (2)
The focal length of the plano-convex lens is 20 cm. If its plane surface is silvered, then
1. it behaves as a concave mirror of focal length 10 cm
2. it behaves as a convex mirror of focal length of 10 cm
3. it behaves as a concave mirror of focal length 20 cm
4. none of these
1.
2.
3.
4.
The energy needed in breaking of a drop of liquid of radius R into n drops of radius r is given by (T is surface tension and P is atmospheric pressure) :
1.
2.
3.
4.
An incompressible liquid travels as shown in the figure. The speed of the fluid in the lower branch will :

1. 1 m/s
2. 1.5 m/s
3. 2.25 m/s
4. 3 m/s
The current through the \(5~\Omega\) resistor is:
| 1. | \(3.2~\text A\) | 2. | \(2.8~\text A\) |
| 3. | \(0.8~\text A\) | 4. | \(0.2~\text A\) |
In the figure, a carbon resistor has bands of different colours on its body as shown. The value of the resistance is:
1. 2.2 k
2. 3.3 k
3. 5.6 k
4. 9.1 k
A circular coil of wire of radius 'r' has 'n' turns and carries a current 'I'. The magnetic induction (B) at a point on the axis of the coil at a distance from its center is :
1.
2.
3.
4.
Magnetic permeability is maximum for
1. Diamagnetic susbtance
2. Paramagnetic substance
3. Ferromagnetic substance
4. All of these
| 1. | \(\left[MLT^{-2}\right]\) | 2. | \(\left[ML^{-1}T^{2}\right]\) |
| 3. | \(\left[ML^{-1}T^{-2}\right]\) | 4. | \(\left[MLT^{2}\right]\) |
Interference fringes are produced using white light in a double-slit arrangement. When a mica sheet of uniform thickness of the refractive index 1.6 (relative to air) is placed in the path of light from one of the slits, the central fringe moves by a distance.
This distance is equal to the width of 30 interference bands. If the light of wavelength 4800 is used, the thickness (in m) of mica is —
1. 90
2. 12
3. 14
4. 24
Which of the following cannot be speed-time graph?
| (A) |  |
| (B) |  |
| (C) |  |
| (D) |  |
| (E) |  |
(A)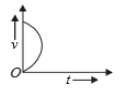
(B) 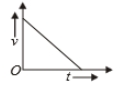
(C)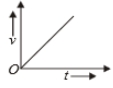
(D) 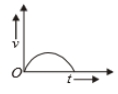
(E) 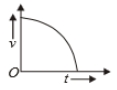
1. (B) and (D)
2. (C) and (E)
3. (D) only
4. (A) only
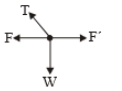
A point of mass m is suspended from a light thread of length l, fixed at O,
is whirled in a horizontal circle at constant speed as shown. From a
frame stationary with respect to the mass, the forces acting on the mass are-
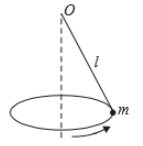

| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
1. 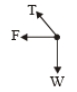
2. 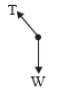
3. 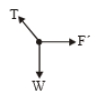
4. 
The centre of mass of a body
1. depends on the choice of co-ordinate system
2. is independent of the choice of co-ordiante system
3. may or may not depend on the choice of co-ordinate system
4. none of these
If vector be a force acting on a particle having the position vector and be the torque of this force about the origin, then
1.
2.
3.
4.
The moment of inertia of a uniform rod about a perpendicular axis passing through one end is . The same rod is bent into a ring and its moment of inertia about a diameter is . Then is
1.
2.
3.
4.
A planet revolves in an elliptical orbit around the sun. The linear speed of the planet will be maximum at :

1. A
2. B
3. C
4. D
If a spring of spring constant k is stretched by a length x under tension T, the energy stored is
1.
2.
3.
4.
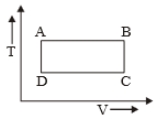
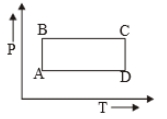
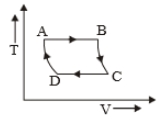
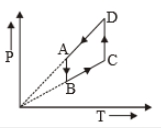
The P-V diagram of a certain process (Carnot cycle) is shown in the figure. The process is represented as: 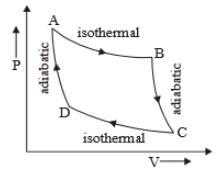

1. 
2. 
3. 
4. 
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
An organ pipe open at one end is vibrating in first overtone and is in resonance with another pipe open at both ends and vibrating in third harmonic. The ratio of the length of two pipes is
1. 1:2
2. 4:1
3. 8:3
4. 3:1
In the circuit shown in the figure, the potential difference across the capacitor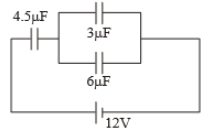

1. 8 volts
2. 6 volts
3. 4 volts
4. volts
Identify the correct statement from the following
1. Cyclotron frequency is dependent on speed of the charged particle
2. Kinetic energy of chared particle in cyclotron does not depend on its mass
3. Cyclotron frequency does not depend on speed of charged particle
4. Kinetic energy of charged particle in cyclotron is independent of its charge
A small square loop of wire of side 'l' is placed inside a large square loop of side 'L' (L>>l). If the loops are coplanar and their centres coincide, the mutual induction of the system is directly proportional to
1. L/l
2. l/L
3.
4.
| 1. | real |
| 2. | virtual |
| 3. | depends on the nature of the object |
| 4. | none of these |
A bridge rectifier is constructed shown in figure. Alternating input is given across A and C. If output is taken across BD, then it is

1. zero
2. same as input
3. half wave rectified
4. full wave rectified
A cannon ball has the same range R on a horizontal plane for two different angles of projection. If are the greatest height in the two paths for which this is possible, then:
1.
2.
3.
4.
A particle of mass 5 gm executing SHM has amplitude of 8 cm. If it makes 16 vibrations per second. Its energy at mean position is
1.
2.
3.
4.
If is finite then
1. is parallel to
2. =
3. ||=||
4. and are mutually perpendicular
A wire of length L and radius r is fixed at one end and a force F applied to the other end produces an extension l. The extension produced in another wire of the same material of length 2L and radius 2r by a force 2F is
1. l
2. 2l
3. l/2
4. 4l
A gas is formed of molecules, each molecules possessing n degree of freedom. The value of for the gas is
1.
2. 1+
3.
4.
Two waves having intensities in the ratio of 9:1 produce interference. The ratio of maximum to minimum intensity is equal to
1. 10:8
2. 9:1
3. 4:1
4. 2:1
A point Q lies on the perpendicular bisector of an electrical dipole of dipole moment p. If the distance of Q from the dipole is r ( much larger than the size of the dipole), then electric field at Q is proportional to?
1. and
2. p and
3.
4. p and
The magnetic field perpendicular to the plane of a conducting ring of radius r changes at the rate dB/dt. Then
1. the potential difference between diametrically opposite points on the ring is half of the induced emf
2. the emf induced in the ring is
3. all the points on the ring are at the same potential
4. none of the above
The Rydberg constant R for hydrogen is
1.
2.
3.
4.
Variation in electric potential is maximum if one goes :
1. along the line of force
2. perpendicular to the line of force
3. in any direction
4. no variation in any direction
The photoelectric work function for a metal surface is 4.125 eV. The cut off wavelength for this surface is
1. 4125
2. 2062.5
3. 3000
4. 6000
Carbon, Silicon, and Germanium atoms have four valence electrons each. Their valence and conduction bands are separated by energy gaps represented by \(\left(E_g\right)_C,(E_g)_{Si}~\text{and}~(E_g)_{Ge}\) respectively. Which one of the following relationships is true in their case?
1. \(\left(E_g\right)_C<\left(E_g\right)_{G e} \)
2. \(\left(E_g\right)_C>\left(E_g\right)_{S i} \)
3. \(\left(E_g\right)_C=\left(E_g\right)_{S i} \)
4. \(\left(E_g\right)_C<\left(E_g\right)_{S i}\)
For the circuit shown in the figure, the equation of current is:

1. \({ i}=(1~\text A) \sin \left(\omega {t}-\frac{\pi}{4}\right) \)
2. \({i}=(100~\text A) \sin \left(\omega{t}+\frac{\pi}{4}\right) \)
3. \({i}=(1 ~\text A) \sin \omega t \)
4. \({i}=(1~\text A) \sin \left(\omega {t}+\frac{\pi}{4}\right)\)
The current gain of the common-base n-p-n transistor is 0.96. What is the current gain if it is used as common-emitter amplifier ?
1. 16
2. 24
3. 20
4. 32
If and represent the wavelengths of visible light, x-rays and microwaves respectively, then-
1.
2.
3.
4.
A force F acting on an object varies with distance x as shown here.
The force is in N and x in m. The work done by the force in moving the object from x = 0 to x = 6 m is
1. 18.0 J
2. 13.5 J
3. 4.5 J
4. 9.0 J
The linear momentum p of a body of mass 5 kg varies with time t as,
p = 5t2+ t + 5
It follows that the body is moving with :-
1. Constant acceleration.
2. Constant speed.
3. Variable acceleration which is increasing with time.
4. Variable retardation which is decreasing with time.