Grass leaves curl inwards during very dry weather. Select the most appropriate reason from the following :
1. Tyloses in vessels
2. Closure of stomata
3. Flaccidity of bulliform cells
4. Shrinkage of air spaces in spongy mesophyll
What triggers the activation of protoxin to active Bt toxin of Bacillus thuringiensis in bollworms?
1. Acidic pH of the stomach
2. Body temperature
3. Moist surface of midgut
4. Alkaline pH of the gut
Select the correctly written complete scientific name of Mango which was first described by Carolus Linnaeus:
1. Mangifera Indica
2. Mangifera indica Car. Linn.
3. Mangifera indica Linn.
4. Mangifera indica
Cells in phase:
1. terminate the cell cycle
2. exit the cell cycle
3. enter the cell cycle
4. suspend the cell cycle
Phloem in gymnosperms lacks:
1. Both sieve tubes and companion cells
2. Albuminous cells and sieve cells
3. Sieve tubes only
4. Companion cells only
Which of the following contraceptive methods involve the role of hormones?
| 1. | Pills, Emergency contraceptives, barrier methods |
| 2. | Lactational amenorrhea, Pills, Emergency contraceptives |
| 3. | Barrier method, Lactational amenorrhea, pills |
| 4. | CuT, Pills, barrier methods |
Which of the following statements is incorrect?
| 1. | Yeasts have filamentous bodies with long thread-like hyphae. |
| 2. | Morels and truffles are edible delicacies. |
| 3. | Claviceps is a source of many alkaloids and LSD. |
| 4. | Conidia are produced exogenously and ascospores endogenously. |
It takes a very long time for pineapple plants to produce flowers. Which combination of hormones can be applied to artificially induce flowering in pineapple plants throughout the year to increase yield?
1. Cytokinin and Abscisic acid
2. Auxin and Ethylene
3. Gibberellin and Cytokinin
4. Gibberellin and Abscisic acid
Conversion of glucose to glucose-6-phosphate, the first irreversible reaction of glycolysis, is catalyzed by:
| 1. | Phosphofructokinase | 2. | Aldolase |
| 3. | Hexokinase | 4. | Enolase |
Consider the following features:
| A: | Organ system level of organisation |
| B: | Bilateral symmetry |
| C: | True coelom with segmentation of body |
Select the correct option of animal groups that possess all the above characteristics:
| 1. | Annelida, Mollusca and Chordata |
| 2. | Annelida, Arthropoda and Chordata |
| 3. | Annelida, Arthropoda and Mollusca |
| 4. | Arthropoda, Mollusca and Chordata |
Which of the following muscular disorders is inherited?
1. Botulism
2. Tetany
3. Muscular dystrophy
4. Myasthenia gravis
The Earth Summit held in Rio de Janeiro in 1992 was called:
| 1. | for immediate steps to discontinue the use of CFCs that were damaging the ozone layer |
| 2. | to reduce CO2 emissions and global warming |
| 3. | for the conservation of biodiversity and sustainable utilization of its benefits |
| 4. | to assess the threat posed to native species by invasive weed species |
Which of the following can be used as biocontrol agent in the treatment of plant disease?
1. Lactobacillus
2. Trichoderma
3. Chlorella
4. Anabaena
Extrusion of the second polar body from the egg nucleus occurs:
| 1. | simultaneously with the first cleavage |
| 2. | after entry of sperm but before fertilization |
| 3. | after fertilization |
| 4. | before entry of sperm into the ovum |
Xylem translocates:
1. Water, minerals salts, some nitrogen and hormones
2. Water only
3. Water and mineral salts only
4. Water, mineral and some organic nitrogen only
The concept of "Omnis cellula-e-cellula" regarding cell division was first proposed by:
1. Aristotle
2. Rudolf Virchow
3. Theodor Schwann
4. Schleiden
Which of the following glucose transporters is insulin-dependent?
1. GLUT IV
2. GLUT I
3. GLUT II
4. GLUT III
Which of the following statements is correct?
| 1. | Cornea consists of dense matrix of collagen and is the most sensitive portion of the eye. |
| 2. | Cornea is an external, transparent and protective proteinaceous ring of the eyeball |
| 3. | Cornea consists of dense connective tissue of elastin and can repair itself |
| 4. | The Cornea is convex, transparent layer which is highly vascularised |
Match the following genes of the lac operon with their respective products:
| (a) | i gene | (i) | -galactosidase |
| (b) | Z gene | (ii) | Permease |
| (c) | A gene | (iii) | Repressor |
| (d) | Y gene | (iv) | transacetylase |
Select the correct option
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iii) | (iv) | (i) | (ii) |
| 2. | (i) | (iii) | (ii) | (iv) |
| 3. | (iii) | (i) | (ii) | (iv) |
| 4. | (iii) | (i) | (iv) | (ii) |
Respiratory Quotient (RQ) value of tripalmitin is:
| 1. | 0.09 | 2. | 0.9 |
| 3. | 0.7 | 4. | 0.007 |
Which of the following statements regarding mitochondria is incorrect?
| 1. | Mitochondrial matrix contains single circular DNA molecule and ribosomes. |
| 2. | Outer membrane is permeable to monomers of carbohydrates, fats, and proteins. |
| 3. | Enzymes of electron transport are embedded in the outer membrane. |
| 4. | Inner membrane is convoluted with infoldings. |
The shorter and longer arms of submetacentric chromosome are referred to as:
1. m-arm and n-arm respectively
2. s-arm and l-arm respectively
3. p-arm and q-arm respectively
4. q-arm and p-arm respectively
Purines found both in DNA and RNA are:
| 1. | Cytosine and thymine | 2. | Adenine and thymine |
| 3. | Adenine and guanine | 4. | Guanine and cytosine |
Which of the following methods is the most suitable for the disposal of nuclear waste?
| 1. | Bury the waste within rocks, deep below the earth's surface |
| 2. | Shoot the waste into the space |
| 3. | Bury the waste under the Antarctic ice-cover |
| 4. | Dump the waste within rocks, under deep ocean |
| 1. | Bronchioles and fallopian tubes |
| 2. | Bile duct and bronchioles |
| 3. | Fallopian tubes and pancreatic duct |
| 4. | Eustachian tube and salivary duct |
Variations caused by mutation, as proposed by Hugo de Vries, are:
1. small and directionless
2. random and directional
3. random and directionless
4. small and directional
How does steroid hormone influence the cellular activities?
| 1. | Using aquaporin channels as second messenger |
| 2. | Changing the permeability of the cell membrane |
| 3. | Binding the DNA and forming a gene-hormone complex |
| 4. | Activating cyclic AMP located on the cell membrane |
In Antirrhinum (Snapdragon), a red flower was crossed with a white flower and in F1 generation, pink flowers were obtained. When pink flowers were selfed, the F2 generation showed white, red, and pink flowers. Choose the incorrect statement from the following:
| 1. | Law of Segregation does not apply in this experiment. |
| 2. | This experiment does not follow the Principle of Dominance. |
| 3. | The pink colour in F1 is due to incomplete dominance. |
| 4. | Ratio of F2 is 1/4(red):2/4(pink):1/4(white). |
Placentation, in which ovules develop on the inner wall of the ovary or in peripheral part, is:
1. Free central
2. basal
3. Axile
4. Parietal
Select the correct group of biocontrol agents:
1. Nostoc, Azospirillum, Nucleopolyhedrovirus
2. Bacillus thuringiensis. Tobacco mosaic virus, Aphids
3. Trichoderma, Baculovirus, Bacillus thuringiensis
4. Oscilatoria, Rhizobium, Trichoderma
The correct sequence of phases in cell cycle is:
1.
2.
3.
4.
Which part of the brain is responsible for thermoregulation?
1. Medulla oblongata
2. Cerebrum
3. Hypothalamus
4. Corpus callosum
Which one of the following is not a method of in situ conservation of biodiversity?
1. Sacred Grove
2. Biosphere Reserve
3. Wildlife Sanctuary
4. Botanical Garden
Which of the following pairs of gases is mainly responsible for green house effect?
1. Carbon dioxide and methane
2. Ozone and Ammonia
3. Oxygen and Nitrogen
4. Nitrogen and Sulphur dioxide
Persistent nucellus in the seed is known as:
| 1. | Tegmen | 2. | Chalaza |
| 3. | Perisperm | 4. | Hilum |
Match the Column I and Column II:
| Column I | Column II | ||
| (a) | P-waves | (i) | Depolarisation of ventricles |
| (b) | QRS complex | (ii) | Repolarisation of ventricles |
| (c) | T-wave | (iii) | Coronary ischemia |
| (d) | Reduction in the size of T-wave | (iv) | Depolarisation of atria |
| (v) | Repolarisation of atria |
| Options: | (a) | (b) | (c) | (d) |
| 1. | (ii) | (iii) | (v) | (iv) |
| 2. | (iv) | (i) | (ii) | (iii) |
| 3. | (iv) | (i) | (ii) | (v) |
| 4. | (ii) | (i) | (v) | (iii) |
Following statements describe the characteristics of the enzyme Restriction Endonuclease. Identify the incorrect statement:
| 1. | The enzyme recognizes a specific palindromic nucleotide sequence in the DNA |
| 2. | The enzyme cuts DNA molecules at an identified position within the DNA |
| 3. | The enzyme binds DNA at specific sites and cuts only one of the two strands |
| 4. | The enzyme cuts the sugar-phosphate backbone at specific sites on each strand |
Which of the following is true for Golden rice?
| 1. | It has yellow grains, because of gene introduced from a primitive variety of rice |
| 2. | It is Vitamin A enriched, with a gene from daffodil |
| 3. | It is pest resistant, with a gene from Bacillus thuringiensis |
| 4. | It is drought tolerant, developed using Agrobacterium vector |
Match the Column-I with Column-II
|
Column-I |
Column-II |
||
|
(a) |
Saprophyte |
(i) |
Symbiotic association of fungi with plant roots |
|
(b) |
Parasite |
(ii) |
Decomposition of dead organic materials |
|
(c) |
Lichens |
(iii) |
Living on living plants or animals |
|
(d) |
Mycorrhiza |
(iv) |
Symbiotic association of algae and fungi |
Choose the correct answer from the options given below:
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iii) | (iv) | (i) |
| 2. | (i) | (ii) | (iii) | (iv) |
| 3. | (iii) | (ii) | (i) | (iv) |
| 4. | (ii) | (i) | (iii) | (iv) |
What would be the heart rate of a person if the cardiac output is 5L, blood volume in the ventricles at the end of diastole is 100 mL and at the end of ventricular systole is 50 mL?
1. 125 beats per minute
2. 50 beats per minute
3. 75 beats per minute
4. 100 beats per minute
Which of the following statements is incorrect?
| 1. | Prions consist of abnormally folded proteins |
| 2. | Viroids lack a protein coat |
| 3. | Viruses are obligate parasites |
| 4. | Infective constituent in viruses is the protein coat |
Match the following structures with their respective location in organs:
|
(a) |
Crypts of Lieberkuhn |
(i) |
Pancreas |
|
(b) |
Glisson’s Capsule |
(ii) |
Duodenum |
|
(c) |
Islets of Langerhans |
(iii) |
Small intestine |
|
(d) |
Bruunner’s Glands |
(iv) |
Liver |
Select the correct option from the following:
| Options: | (a) | (b) | (c) | (d) |
| 1. | iii | ii | i | iv |
| 2. | iii | i | ii | iv |
| 3. | ii | iv | i | iii |
| 4. | iii | iv | i | ii |
Which of the following immune responses is responsible for the rejection of kidney graft?
| 1. | Cell-mediated immune response |
| 2. | Auto-immune response |
| 3. | Humoral immune response |
| 4. | Inflammatory immune response |
Identify the cells whose secretion protects the lining of gastro-intestinal tract from various enzymes:
1. Duodenal cells
2. Chief cells
3. Goblet cells
4. Oxyntic cells
Under which of the following conditions will there be no change in the reading frame of the following mRNA?
5'AACAGCGGUGCUAUU 3'
| 1. | Deletion of GGU from 7th, 8th and 9th positions |
| 2. | Insertion of G at the 5th position |
| 3. | Deletion of G from the 5th position |
| 4. | Insertion of A and G at the 4th and the 5th positions respectively |
Which of the following is a commercial blood cholesterol-lowering agent?
| 1. | Lipase | 2. | Cyclosporin A |
| 3. | Statin | 4. | Streptokinase |
Select the incorrect statement:
| 1. | Human males have one of their sex-chromosome much shorter than the other. |
| 2. | Male fruit fly is heterogametic. |
| 3. | In male grasshoppers, 50% of sperms have no sex chromosome. |
| 4. | In domesticated fowls, the sex of progeny depends on the type of sperm rather than the egg. |
Tidal Volume and Expiratory Reserve Volume of an athlete is 500 mL and 1000 mL respectively. What will be his Expiratory Capacity if the Residual Volume is 1200 mL?
| 1. | 2700 mL | 2. | 1500 mL |
| 3. | 1700 mL | 4. | 2200 mL |
Select the correct sequence for the transport of sperm cells in the male reproductive system:
| 1. | Testis → Epididymis → Vasa efferentia → Vas deferens → Ejaculatory duct → Inguinal canal → Urethra → Urethral meatus |
| 2. | Testis → Epididymis → Vasa efferentia → Rete testis → Inguinal canal → Urethra |
| 3. | Seminiferous tubules → Rete testis → Vasa efferentia → Epididymus → Vas deferens → Ejaculatory duct → Urethra → Urethral meatus |
| 4. | Seminiferous tubules → Vasa efferentia → Epididymis → Inguinal canal → Urethra |
Colostrum, the yellowish fluid, secreted by the mother during the initial days of lactation is very essential to impart immunity to newborn infants because it contains:
| 1. | Immunoglobulin A | 2. | Natural killer cells |
| 3. | Monocytes | 4. | Macrophages |
In some plants, the female gamete develops into an embryo without fertilization. This phenomenon is known as:
| 1. | Parthenogenesis | 2. | Autogamy |
| 3. | Parthenocarpy | 4. | Syngamy |
Identify the correct pair representing the causative agent of typhoid fever and the confirmatory test for typhoid.
1. Salmonella typhi/Widal test
2. Plasmodium vivax/UTI test
3. Streptococcus pneumoniae/Widal test
4. Salmonella typhi/Anthrone test
Expressed Sequence Tags (ESTs) refers to:
| 1. | Novel DNA sequence | 2. | Genes expressed as RNA |
| 3. | Polypeptide expression | 4. | DNA polymorphism |
Match the following hormones with the respective disease:
| (a) | Insulin | (i) | Addison's disease |
| (b) | Thyroxin | (ii) | Diabetes insipidus |
| (c) | Corticoids | (iii) | Acromegaly |
| (d) | Growth Hormone | (iv) | Goitre |
| (v) | Diabetes mellitus |
Select the correct option.
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iv) | (i) | (iii) |
| 2. | (v) | (i) | (ii) | (iii) |
| 3. | (ii) | (iv) | (iii) | (i) |
| 4. | (v) | (iv) | (i) | (iii) |
Which of the following factors is responsible for the formation of concentrated urine?
| 1. | Hydrostatic pressure during glomerular filtration. |
| 2. | Low levels of antidiuretic hormone. |
| 3. | Maintaining hyperosmolarity towards inner medullary interstitium in the kidneys. |
| 4. | Secretion of erythropoietin by Juxtaglomerular complex. |
Select the hormone-releasing Intra-Uterine Devices.
| 1. | Lippes Loop, Multitoad 375 |
| 2. | Vaults, LNG-20 |
| 3. | Multiload 375, Progestasert |
| 4. | Progestasert, LNG-20 |
Match the following organism with their respective characteristics:
| Column-I | Column-II | ||
| (a) | Pila | (i) | Flame cells |
| (b) | Bombyx | (ii) | Comb plates |
| (c) | Pleurobrachia | (iii) | Radula |
| (d) | Taenia | (iv) | Malpighian tubules |
Select the correct option from the following:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iii) | (ii) | (iv) | (i) |
| 2. | (iii) | (ii) | (i) | (iv) |
| 3. | (iii) | (iv) | (ii) | (i) |
| 4. | (ii) | (iv) | (iii) | (i) |
Which of the following sexually transmitted diseases is not completely curable?
| 1. | Chlamydiosis | 2. | Gonorrhoea |
| 3. | Genital warts | 4. | Genital herpes |
The drug called 'Heroin' is synthesized by:
| 1. | nitration of morphine | 2. | methylation of morphine |
| 3. | acetylation of morphine | 4. | glycosylation of morphine |
What is the site of perception of photoperiod necessary for induction of flowering in plants?
1. Leaves
2. Lateral buds
3. Pulvinus
4. Shoot apex
A gene locus has two alleles A, a. If the frequency of dominant allele A is 0.4, then what will be the frequency of homozygous dominant, heterozygous and homozygous recessive individuals in the population?
| 1. | 0.16 (AA); 0.36 (Aa); 0.48 (aa) |
| 2. | 0.36 (AA); 0.48 (Aa); 0.16 (aa) |
| 3. | 0.16 (AA); 0.24 (Aa); 0.36 (aa) |
| 4. | 0.16 (AA); 0.48 (Aa); 0.36 (aa) |
What map unit (Centimorgan) is adopted in the construction of genetic maps?
| 1. | A unit of distance between genes on chromosomes, representing 50% cross-over. |
| 2. | A unit of distance between two expressed genes, representing 10% cross-over. |
| 3. | A unit of distance between two expressed genes, representing 100% cross-over. |
| 4. | A unit of distance between genes on chromosomes. representing 1% cross-over. |
Concanavalin A is:
| 1. | a pigment | 2. | an alkaloid |
| 3. | an essential oil | 4. | a lectin |
Pinus seeds cannot germinate and establish without fungal association. This is because:
1. its seeds contain inhibitors that prevent germination.
2. its embryo is immature.
3. it has an obligate association with mycorrhizae.
4. it has a very hard seed coat.
The frequency of recombination between gene pairs on the same chromosome as a measure of the distance between genes was explained by:
| 1. | Sutton Boveri | 2. | T.H. Morgan |
| 3. | Gregor J. Mendel | 4. | Alfred Sturtevant |
In a species, the weight of a newborn ranges from 2 to 5 kg. 97% of the newborns with an average weight between 3 to 3.3 kg survive whereas 99% of the infants born with weights from 2 to 2.5 kg or 4.5 kg to 5 kg die. Which type of selection process is taking place?
1. Cyclical selection
2. Directional selection
3. Stabilizing selection
4. Disruptive selection
Match the hominids with their correct brain size:
| Column-I | Column-II | ||
| (a) | Homo habilis | (i) | 900 cc |
| (b) | Homo neanderthalensis | (ii) | 1350 cc |
| (c) | Homo erectus | (iii) | 650-800 cc |
| (d) | Homo sapiens | (iv) | 1400 cc |
Select the correct option.
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iv) | (iii) | (i) | (ii) |
| 2. | (iv) | (i) | (iv) | (ii) |
| 3. | (iii) | (ii) | (i) | (iv) |
| 4. | (iii) | (iv) | (i) | (ii) |
Select the correct option.
| 1. | There are seven pairs of vertebrosternal, three pairs of vertebrochondral and two pairs of vertebral ribs. |
| 2. | 8th, 9th and 10th pairs of ribs articulate directly with the sternum. |
| 3. | 11th and 12th pairs of ribs are connected to the sternum with the help of hyaline cartilage. |
| 4. | Each rib is a flat thin bone and all the ribs are connected dorsally to the thoracic vertebrae and ventrally to the sternum. |
What is the direction of movement of sugars in phloem?
1. Bi-directional
2. Non-multidirectional
3. Upward
4. Downward
Polyblend, a fine powder of recycled modified plastic, has proved to be a good material for:
1. making tubes and pipes
2. making plastic sacks
3. use as a fertilizer
4. construction of roads
Which of the following ecological pyramids is generally inverted?
| 1. | Pyramid of biomass in a sea |
| 2. | Pyramid of numbers in grassland |
| 3. | Pyramid of energy |
| 4. | Pyramid of biomass in a forest |
Use of an artificial kidney during hemodialysis may result in:
| (a) | Nitrogenous waste build-up in the body |
| (b) | Non-elimination of excess potassium ions |
| (c) | Reduced absorption of calcium ions from gastro-intestinal tract |
| (d) | Reduced RBC production |
Which of the following options is the most appropriate?
1. (a) and (d) are correct
2. (a) and (b) are correct
3. (b) and (c) are correct
4. (c) and (d) are correct
Which of the following pair of organelles does not contain DNA?
| 1. | Nuclear envelope and Mitochondria |
| 2. | Mitochondria and Lysosomes |
| 3. | Chloroplast and Vacuoles |
| 4. | Lysosomes and vacuoles |
Which of the following is the most important cause for animals and plants being driven to extinction?
1. Alien species invasion
2. Habitat loss and fragmentation
3. Drought and floods
4. Economic exploitation
What is the fate of the male gametes discharged in the synergid?
| 1. | One fuses with the egg and the other fuses with central cell nuclei |
| 2. | One fuses with the egg and the other(s) degenerate(s) in the synergid |
| 3. | All fuse with the egg |
| 4. | One fuses with the egg and the other(s) fuse(s) with a synergid nucleus |
Which of the following protocols did aim for reducing the emission of chlorofluorocarbons into the atmosphere?
1. Geneva protocol
2. Montreal protocol
3. Kyoto protocol
4. Gothenburg protocol
Due to increasing air-borne allergens and pollutants, many people in urban areas are suffering from respiratory disorder causing wheezing due to:
| 1. | reduction in the secretion of surfactants by pneumocytes |
| 2. | benign growth on mucous lining of nasal cavity |
| 3. | inflammation of bronchi and bronchioles |
| 4. | proliferation of fibrous tissues and damage of the alveolar walls |
From an evolutionary point of view, retention of the female gametophyte with developing young embryo on the parent sporophyte for some time, is first observed in:
| 1. | Gymnosperms | 2. | Liverworts |
| 3. | Mosses | 4. | Pteridophytes |
What is the genetic disorder in which an individual has an overall masculine development gynaecomastia, and is sterile?
1. Down's syndrome
2. Turner's syndrome
3. Klinefelter's syndrome
4. Edward syndrome
Which of the following features of genetic code does allow bacteria to produce human insulin by recombinant DNA technology?
1. Genetic code is specific
2. Genetic code is not ambiguous
3. Genetic code is redundant
4. Genetic code is nearly universal
Match the following :
|
(a) |
Lactobacillus |
(i) |
Cheese |
|
(b) |
Saccharomyces cerevisiae |
(ii) |
Curd |
|
(c) |
Aspergillus niger |
(iii) |
Citric Acid |
|
(d) |
Acetobacter aceti |
(iv) |
Bread |
|
(v) |
Acetic acid |
| Options: | (a) | (b) | (c) | (d) |
| 1. | (ii) | (i) | (iii) | (v) |
| 2. | (ii) | (iv) | (v) | (iii) |
| 3. | (ii) | (iv) | (iii) | (v) |
| 4. | (iii) | (iv) | (v) | (i) |
DNA precipitation, out of a mixture of biomolecules, can be achieved by treatment with:
1. Chilled chloroform
2. Isopropanol
3. Chilled ethanol
4. Methanol at room temperature
Thiobacillus is a group of bacteria helpful in carrying out:
1. Denitrification
2. Nitrogen fixation
3. Chemoautotrophic fixation
4. Nitrification
Which of the following statements is not correct?
| 1. | Lysosomes are formed by the process of packaging in the endoplasmic reticulum. |
| 2. | Lysosomes have numerous hydrolytic enzymes |
| 3. | The hydrolytic enzyme of lysosomes are active under acidic pH |
| 4. | Lysosomes are membrane bound structure. |
Select the incorrect statement:
| 1. | Inbreeding helps in the accumulation of superior genes and the elimination of undesirable genes |
| 2. | Inbreeding increases homozygosity |
| 3. | Inbreeding is essential to evolve pure lines in any animal |
| 4. | Inbreeding selects harmful recessive genes that reduce fertility and productivity |
Select the correct sequence of organs in the alimentary canal of cockroach starting from mouth:
| 1. | Pharynx → Oesophagus → Ileum → Crop → Gizzard → Colon → Rectum |
| 2. | Pharynx → Oesophagus → Crop → Gizzard → Ileum → Colon → Rectum |
| 3. | Pharynx → Oesophagus → Gizzard → Crop → Ileum → Colon → Rectum |
| 4. | Pharynx → Oesophagus → Gizzard → Ileum → Crop → Colon → Rectum |
Consider the following statements:
| A: | A coenzyme or metal ion that is tightly bound to enzyme protein is called prosthetic group |
| B: | A complete catalytic enzyme with its bound prosthetic group is called apoenzyme |
Select the correct option
1. (A) is false but (B) is true
2. Both (A) and (B) are true
3. (A) is true but (B) is false
4. Both (A) and (B) are false
Which of the statements given below is not true about the formation of Annual Rings in trees?
| 1. | Annual rings are not prominent in trees of temperate regions. |
| 2. | Annual rings are a combination of spring wood and autumn wood produced in a year. |
| 3. | Differential activity of cambium causes light and dark bands of tissue - early and late wood respectively. |
| 4. | Activity of cambium depends upon variation in climate. |
Which one of the following statements regarding post-fertilization development in flowering plants is incorrect?
| 1. | Ovules develop into an embryo sac |
| 2. | Ovary develops into fruit |
| 3. | Zygote develops into an embryo |
| 4. | Central cell develops into endosperm |
Which of the following equipment is essentially required for growing microbes on a large scale for the industrial production of enzymes?
| 1. | Bioreactor | 2. | BOD incubator |
| 3. | Sludge digester | 4. | Industrial oven |
Given the following cell reaction:
\(\mathrm{2Fe^{3+}(aq) \ + \ 2I^{-}(aq)\rightarrow 2Fe^{2+}(aq) \ + \ I_{2}(aq)}\)
[Given: \(F = 96500\) \(C\) \(mol^{- 1}\)]
1. \(23 . 16\) \(kJ\) \(mol^{- 1}\)
2. \(- 46 . 32\) \(kJ\) \(mol^{- 1}\)
3. \(- 23 . 16\) \(kJ\) \(mol^{- 1}\)
4. \(46 . 32\) \(kJ\) \(mol^{- 1}\)
The compound that is most difficult to protonate is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The manganate and permanganate ions are tetrahedral, due to:
| 1. | The π-bonding involves the overlap of d-orbitals of oxygen with d-orbitals of manganese. |
| 2. | The π-bonding involves the overlap of p-orbitals of oxygen with d-orbitals of manganese. |
| 3. | Absence of π-bonding. |
| 4. | The π-bonding involves the overlap of p-orbitals of oxygen with p-orbitals of manganese. |
An alkene "A" on reaction with and gives propanone and ethanal in equimolar ratio. The addition of HCl to alkene "A" gives "B" as the major product. The structure of product "B" is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
For the second-period elements, the correct increasing order of first ionisation enthalpy is:
| 1. | Li < Be < B < C < O < N < F < Ne |
| 2. | Li < Be < B < C < N < O < F < Ne |
| 3. | Li < B < Be < C < O < N < F < Ne |
| 4. | Li < B < Be < C < N < O < F < Ne |
A gas at 350 K and 15 bar has a molar volume 20 percent smaller than that for an ideal gas under the same conditions. The correct option about the gas and its compressibility factor (Z) is:
| 1. | Z<1 and repulsive forces are dominant. |
| 2. | Z>1 and attractive forces are dominant. |
| 3. | Z>1 and repulsive forces are dominant. |
| 4. | Z<1 and attractive forces are dominant. |
For a cell involving one electron \(E_{cell}^{\ominus} = 0 . 59 V\) at 298 K. The equilibrium constant for the cell reaction is :
\(\mathrm{[Given~ that~ \frac {2.303 ~RT}{F} = 0.059 ~V~ at~ T = 298 K]}\)
| 1. | \(1 . 0 \times \left(10\right)^{30}\) | 2. | \(1 . 0 \times \left(10\right)^{2}\) |
| 3. | \(1 . 0 \times \left(10\right)^{5}\) | 4. | \(1 . 0 \times \left(10\right)^{10}\)
|
Which composition will make the basic buffer?
| 1. | 100 mL of 0.1 M HCl+100 mL of 0.1 M NaOH |
| 2. | 50 mL of 0.1 M NaOH+25 mL of 0.1 M CH3COOH |
| 3. | 100 mL of 0.1 M CH3COOH+100 mL of 0.1 M NaOH |
| 4. | 100 mL of 0.1 M HCl+200 mL of 0.1 M NH4OH |
The correct thermal stability order for \(\mathrm{{H}_2 {E}},\) \(\mathrm{{ E}= {O}, {S}, {Se}, {Te}} \text { and } \mathrm{Po}\) among the following is:
1. \(\mathrm{{H}_2 {Se}<{H}_2 {Te}<{H}_2 {Po}<{H}_2 {O}<{H}_2 S}\)
2. \(\mathrm{{H}_2 {~S}<{H}_2 {O}<{H}_2 {Se}<{H}_2 {Te}<{H}_2 {Po}}\)
3. \(\mathrm{{H}_2 {O}<{H}_2 {~S}<{H}_2 {Se}<{H}_2 {Te}<{H}_2 {PO}}\)
4. \(\mathrm{{H}_2 {Po}<{H}_2 {Te}<{H}_2 {Se}<{H}_2 {~S}<{H}_2 {O}}\)
For an ideal solution, the correct option is:
1. at constant T and P
2. at constant T and P
3. at constant T and P
4. at constant T and P
The biodegradable polymer is:
1. Buna-S
2. Nylon-6, 6
3. Nylon 2-nylon 6
4. Nylon-6
Enzymes that utilize ATP in phosphate transfer require an alkaline earth metal (M) as the cofactor. M is:
| 1. | Sr | 2. | Be |
| 3. | Mg | 4. | Ca |
If the rate constant for a first order reaction is k, the time (t) required for the completion of 99% of the reaction is given by:
1. t = 2.303/k
2. t = 0.693/k
3. t = 6.909/k
4. t = 4.606/k
A diatomic molecule, among the following species, that only has bonds according to the Molecular Orbital Theory (MOT) is:
| 1. | Be2 | 2. | O2 |
| 3. | N2 | 4. | C2 |
| 1. | \(0 . 5 \times \left(10\right)^{- 10}\) | 2. | \(0 . 5 \times \left(10\right)^{- 15}\) |
| 3. | \(0 . 25 \times \left(10\right)^{- 10}\) | 4. | \(0 . 125 \times \left(10\right)^{- 15}\) |
Which of the following mixtures forms the maximum boiling azeotrope?
| 1. | Heptane + Octane | 2. | Water + Nitric acid |
| 3. | Ethanol + Water | 4. | Acetone + Carbon disulphide |
4d, 5p, 5f and 6p orbitals are arranged in the order of decreasing energy. The correct option is:
| 1. | 5f > 6p > 4d > 5p | 2. | 5f > 6p > 5p > 4d |
| 3. | 6p > 5f > 5p > 4d | 4. | 6p > 5f > 4d > 5p |
Which of the following is an amphoteric hydroxide?
1.
2.
3.
4.
The incorrect statement among the following is:
1. SnF4 is ionic in nature.
2. PbF4 is covalent in nature.
3. SiCl4 is easily hydrolysed.
4. GeX4 (X = F, Cl, Br, I) is more stable than GeX2
Under the isothermal condition, a gas at \(300 \mathrm{~K}\) expands from \(0.1 \mathrm{~L}\) to \(0.25 \mathrm{~L}\) against a constant external pressure of 2 bar. The work done by the gas is:
| 1. | \(30 ~\mathrm {J} \) | 2. | \(-30 ~\mathrm{J} \) |
| 3. | \(5~ \mathrm{kJ}\) | 4. | \(25~ \mathrm{J}\) |
The number of sigma () and pi () bonds in pent-2-en-4-yne are:
1. 13 bonds and no bond
2. 10 bonds and 3 bonds
3. 8 bonds and 5 bonds
4. 11 bonds and 2 bonds
Match the Xenon compounds in Column-I with their structure in Column-II and assign the correct code:
| Column-I | Column-II | ||
| (a) | XeF4 | (i) | Pyramidal |
| (b) | XeF6 | (ii) | Square planar |
| (c) | XeOF4 | (iii) | Distorted octahedral |
| (d) | XeO3 | (iv) | Square pyramidal |
Code:
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (iv) | (i) | (ii) |
| 2. | (i) | (ii) | (iii) | (iv) |
| 3. | (ii) | (iii) | (iv) | (i) |
| 4. | (ii) | (iii) | (i) | (iv) |
A process among the following shows decrease in entropy is :
1. \(2 \text H \left(g\right)\rightarrow\text H_{2} \left(g\right)\)
2. Evaporation of water
3. Expansion of a gas at a constant temperature
4. Sublimation of solid to gas
The most suitable reagent for the following conversion is-
| 1. | Hg2+/ H+, H2O | 2. | Na/liquid NH3 |
| 3. | H2, Pd/C, quinoline | 4. | Zn/HCl |
The product of the below-mentioned reaction is:
| 1. | 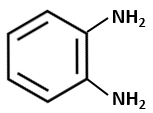 |
2. |  |
| 3. |  |
4. |  |
Match the name of process given in Column-I with the name of compound given in Column-II and mark the correct option.
| Column-I | Column-II | ||
| (a) | Pure nitrogen | (i) | Chlorine |
| (b) | Haber process | (ii) | Sulphuric acid |
| (c) | Contact process | (iii) | Ammonia |
| (d) | Deacon's process | (iv) | Sodium azide or Barium azide |
| (a) | (b) | (c) | (d) | |
| 1. | (iv) | (iii) | (ii) | (i) |
| 2. | (i) | (ii) | (iii) | (iv) |
| 3. | (ii) | (iv) | (i) | (iii) |
| 4. | (iii) | (iv) | (ii) | (i) |
Which of the following series of transitions in the spectrum of hydrogen atoms falls in the visible region?
1. Brackett series
2. Lyman series
3. Balmer series
4. Paschen series
Among the following, the narrow-spectrum antibiotic is:
1. chloramphenicol
2. penicillin G
3. ampicillin
4. amoxicillin
Which mixture of the solutions will lead to the formation of a negatively charged colloidal [AgI]I- sol?
1. 50 mL of 0.1 M AgNO3 + 50 mL of 0.1 M KI
2. 50 mL of 1 M AgNO3 + 50 mL of 0.5 M KI
3. 50 mL of 1 M AgNO3 + 50 mL of 2 M KI
4. 50 mL of 2 M AgNO3 + 50 mL of 1.5 M KI
The reaction among the following that proceeds through an electrophilic substitution reaction is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The structure of intermediate A in the following reaction is:

| 1. |  |
2. |  |
| 3. |  |
4. |  |
What is the correct electronic configuration of the central atom in \(K_{4}FeCN_{6}\) based on crystal field theory:
| 1. | \(e^{4} t_{2 g}^{ 2}\) | 2. | \(t_{2 g}^{ 4} e_{g}^{2}\) |
| 3. | \(t_{2 g}^{ 6} e_{g}^{0}\) | 4. | \(e^{3} t_{2 g}^{ 3}\) |
Among the following, the one that is not a greenhouse gas is:
1. Sulphur dioxide
2. Nitrous oxide
3. Methane
4. Ozone
False statement regarding PCl5 is:
| 1. | PCl5 molecule is non-reactive. |
| 2. | Three equatorial P-Cl bonds make an angle of 120° with each other. |
| 3. | Two axial P-Cl bonds make an angle of 180° with each other. |
| 4. | Axial P-Cl bonds are longer than equatorial P-Cl bonds. |
Which one is a malachite from the following?
1. CuCO3.Cu(OH)2
2. CuFeS2
3. Cu(OH)2
4. Fe3O4
Which of the following species is not stable?
| 1. | 2. | ||
| 3. | 4. |
|
A compound is formed by cation C and anion A. The anions form hexagonal close packed (hcp) lattice and the cations occupy 75 % of octahedral voids. The formula of the compound is:
1.
2.
3.
4.
The correct structure of tribromo octoxide is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The method used to remove the temporary hardness of water is-
1. Synthetic resins method
2. Calgon's method
3. Clark's method
4. Ion-exchange method
The non-essential amino acid among the following is:
| 1. | Lysine | 2. | Valine |
| 3. | Leucine | 4. | Alanine |
| 1. | 40 mol | 2. | 10 mol |
| 3. | 20 mol | 4. | 30 mol |
Disproportionation reactions are:
| (a) | |
| (b) | |
| (c) | |
| (d) |
| 1. | (a) and (d) only | 2. | (a) and (b) only |
| 3. | (a), (b) and (c) | 4. | (a), (c) and (d) |
For the chemical reaction ) the correct option is:
| 1. | \(3\frac {d[H_2]} {dt} = 2 \frac {d[NH_3]} {dt} \) | 2. | \(-\frac {1} {3}\frac {d[H_2]} {dt} = -\frac {1} {2} \frac {d[NH_3]} {dt} \) |
| 3. | \(- \frac{d \left[N_{2}\right]}{dt} = 2 \frac{d [NH_{3}]}{dt}\) | 4. | \(- \frac{d \left[N_{2}\right]}{dt} = \frac{1}{2} \frac{d [NH_{3}]}{dt}\) |
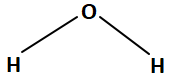
1. H3O+ and H2F+, respectively.
2. OH– and H2F+, respectively.
3. H3O+ and F–, respectively.
4. OH– and F–, respectively.
In which of the following devices, the eddy current effect is not used?
1. Electric heater
2. Induction furnace
3. Magnetic braking in train
4. Electromagnet
The average velocity of a particle executing SHM in one complete vibration is:
1. zero
2. \(\dfrac{A \omega}{2}\)
3. \(A \omega\)
4. \(\dfrac{A \left(\omega\right)^{2}}{2}\)
The speed of a swimmer in still water is \(20~\text{m/s}.\) The speed of river water is \(10~\text{m/s}\) and is flowing due east. If he is standing on the south bank and wishes to cross the river along the shortest path, the angle at which he should make his strokes with respect to the north is given by:
| 1. | \(45^{\circ}\) west of north | 2. | \(30^{\circ}\) west of north |
| 3. | \(0^{\circ}\) west of north | 4. | \(60^{\circ}\) west of north |
In the circuits shown below, the readings of the voltmeters and the ammeters will be:

| 1. | \(V_2>V_1~\text{and}~i_1= i_2\) | 2. | \(V_2=V_1~\text{and}~i_1> i_2\) |
| 3. | \(V_2=V_1~\text{and}~i_1= i_2\) | 4. | \(V_2>V_1~\text{and}~i_1> i_2\) |
A copper rod of \(88\) cm and an aluminium rod of an unknown length have an equal increase in their lengths independent of an increase in temperature. The length of the aluminium rod is:
\(\left(\alpha_{Cu}= 1.7\times10^{-5}~\text{K}^{-1}~\text{and}~\alpha_{Al}= 2.2\times10^{-5}~\text{K}^{-1}\right)\)
| 1. | \(68~\text{cm}\) | 2. | \(6.8~\text{cm}\) |
| 3. | \(113.9~\text{cm}\) | 4. | \(88~\text{cm}\) |
| 1. | W m–1 K–1 | 2. | J m K–1 |
| 3. | J m–1 K–1 | 4. | W m K–1 |
For a \(\mathrm{p}\text{-}\)type semiconductor, which of the following statements is true?
| 1. | Electrons are the majority carriers and pentavalent atoms are the dopants. |
| 2. | Electrons are the majority carriers and trivalent atoms are the dopants. |
| 3. | Holes are the majority carriers and trivalent atoms are the dopants. |
| 4. | Holes are the majority carriers and pentavalent atoms are the dopants. |
A cylindrical conductor of radius \(R\) is carrying a constant current. The plot of the magnitude of the magnetic field \(B\) with the distance \(d\) from the centre of the conductor is correctly represented by the figure:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Body \(\mathrm{A}\) of mass \(4m\) moving with speed \(u\) collides with another body \(\mathrm{B}\) of mass \(2m\) at rest. The collision is head-on and elastic in nature. After the collision, the fraction of energy lost by the colliding body \(\mathrm{A}\) is:
| 1. | \(\dfrac{5}{9}\) | 2. | \(\dfrac{1}{9}\) |
| 3. | \(\dfrac{8}{9}\) | 4. | \(\dfrac{4}{9}\) |
The correct Boolean operation represented by the circuit diagram given above is:
1. \(\mathrm{NOR}\)
2. \(\mathrm{AND}\)
3. \(\mathrm{OR}\)
4. \(\mathrm{NAND}\)
When an object is shot from the bottom of a long, smooth inclined plane kept at an angle of \(60^\circ\) with horizontal, it can travel a distance \(x_1\) along the plane. But when the inclination is decreased to \(30^\circ\) and the same object is shot with the same velocity, it can travel \(x_2\) distance. Then \(x_1:x_2\) will be:
| 1. | \(1:2\sqrt{3}\) | 2. | \(1:\sqrt{2}\) |
| 3. | \(\sqrt{2}:1\) | 4. | \(1:\sqrt{3}\) |
| 1. | \(\dfrac{3}{2}mgR\) | 2. | \(mgR\) |
| 3. | \(2mgR\) | 4. | \(\dfrac{1}{2}mgR\) |
| 1. | \(3.4~\text{eV},~3.4~\text{eV}\) |
| 2. | \(-3.4~\text{eV},~-3.4~\text{eV}\) |
| 3. | \(-3.4~\text{eV},~-6.8~\text{eV}\) |
| 4. | \(3.4~\text{eV},~-6.8~\text{eV}\) |
| 1. | isochoric | 2. | isothermal |
| 3. | adiabatic | 4. | isobaric |
A hollow metal sphere of radius \(R\) is uniformly charged. The electric field due to the sphere at a distance \(r\) from the centre:
| 1. | decreases as \(r\) increases for \(r<R\) and for \(r>R\). |
| 2. | increases as \(r\) increases for \(r<R\) and for \(r>R\). |
| 3. | is zero as \(r\) increases for \(r<R\), decreases as \(r\) increases for \(r>R\). |
| 4. | is zero as \(r\) increases for \(r<R\), increases as \(r\) increases for \(r>R\). |
Pick the wrong statement in the context with a rainbow.
| 1. | Rainbow is a combined effect of dispersion, refraction, and reflection of sunlight. |
| 2. | When the light rays undergo two internal reflections in a water drop, a secondary rainbow is formed. |
| 3. | The order of colors is reversed in the secondary rainbow. |
| 4. | An observer can see a rainbow when his front is towards the sun. |
| 1. | \(6.4\times10^{-6}~\text{m}^{3}/\text{s}\) | 2. | \(12.6\times10^{-6}~\text{m}^{3}/\text{s}\) |
| 3. | \(8.9\times10^{-6}~\text{m}^{3}/\text{s}\) | 4. | \(2.23\times10^{-6}~\text{m}^{3}/\text{s}\) |
Which of the following acts as a circuit protection device?
| 1. | Fuse | 2. | Conductor |
| 3. | Inductor | 4. | Switch |
| 1. | \(\frac{4F}{3}\) | 2. | \(F\) |
| 3. | \(\frac{9F}{16}\) | 4. | \(\frac{16F}{9}\) |
Which colour of the light has the longest wavelength?
1. violet
2. red
3. blue
4. green
The radius of the circle, the period of revolution, initial position and direction of revolution are indicated in the figure.
The \(y\)-projection of the radius vector of rotating particle \(P\) will be:
| 1. | \(y(t)=3 \cos \left(\dfrac{\pi \mathrm{t}}{2}\right)\), where \(y\) in m |
| 2. | \(y(t)=-3 \cos 2 \pi t\) , where \(y\) in m |
| 3. | \(y(t)=4 \sin \left(\dfrac{\pi t}{2}\right)\), where \(y\) in m |
| 4. | \(y(t)=3 \cos \left(\dfrac{3 \pi \mathrm{t}}{2}\right) \), where \(y\) in m |
| 1. | \(2\) protons only. |
| 2. | \(2\) protons and \(2\) neutrons only. |
| 3. | \(2\) electrons, \(2\) protons, and \(2\) neutrons. |
| 4. | \(2\) electrons and \(4\) protons only. |
A solid cylinder of mass \(2~\text{kg}\) and radius \(4~\text{cm}\) is rotating about its axis at the rate of \(3~\text{rpm}.\) The torque required to stop after \(2\pi\) revolutions is:
1. \(2\times 10^6~\text{N-m}\)
2. \(2\times 10^{-6}~\text{N-m}\)
3. \(2\times 10^{-3}~\text{N-m}\)
4. \(12\times 10^{-4}~\text{N-m}\)
In a double-slit experiment, when the light of wavelength \(400~\text{nm}\) was used, the angular width of the first minima formed on a screen placed \(1~\text{m}\) away, was found to be \(0.2^{\circ}.\) What will be the angular width of the first minima, if the entire experimental apparatus is immersed in water? \(\left(\mu_{\text{water}} = \dfrac{4}{3}\right)\)
1. \(0.1^{\circ}\)
2. \(0.266^{\circ}\)
3. \(0.15^{\circ}\)
4. \(0.05^{\circ}\)
At point A on the earth's surface, the angle of dip is, . At a point B on the earth's surface, the angle of dip is, . We can interpret that:
| 1. | A and B are both located in the southern hemisphere. |
| 2. | A and B are both located in the northern hemisphere. |
| 3. | A is located in the southern hemisphere and B is located in the northern hemisphere. |
| 4. | A is located in the northern hemisphere and B is located in the southern hemisphere. |
| 1. | \(\dfrac{1}{2}MgL\) | 2. | \(Mgl\) |
| 3. | \(MgL\) | 4. | \(\dfrac{1}{2}Mgl\) |
A parallel plate capacitor of capacitance \(20~\mu\text{F}\) is being charged by a voltage source whose potential is changing at the rate of \(3~\text{V/s}.\) The conduction current through the connecting wires, and the displacement current through the plates of the capacitor would be, respectively:
| 1. | zero, zero | 2. | zero, \(60~\mu\text{A}\) |
| 3. | \(60~\mu\text{A},\) \(60~\mu\text{A}\) | 4. | \(60~\mu\text{A},\) zero |
A mass \(m\) is attached to a thin wire and whirled in a vertical circle. The wire is most likely to break when:
| 1. | inclined at an angle of \(60^{\circ}\) from vertical. |
| 2. | the mass is at the highest point. |
| 3. | the wire is horizontal. |
| 4. | the mass is at the lowest point. |
| 1. | \(2:1\) | 2. | \(4:9\) |
| 3. | \(9:4\) | 4. | \(1:2\) |
| 1. | \(90^{\circ}\) |
| 2. | \(180^{\circ}\) |
| 3. | \(0^{\circ}\) |
| 4. | equal to the angle of incidence |
Two similar thin equi-convex lenses, of focal length \(f\) each, are kept coaxially in contact with each other such that the focal length of the combination is \(F_1\). When the space between the two lenses is filled with glycerin which has the same refractive index as that of glass \((\mu = 1.5),\) then the equivalent focal length is \(F_2\). The ratio \(F_1:F_2\) will be:
| 1. | \(3:4\) | 2. | \(2:1\) |
| 3. | \(1:2\) | 4. | \(2:3\) |
| 1. | \(1:4\) | 2. | \(2:1\) |
| 3. | \(1:2\) | 4. | \(4:1\) |
In an experiment, the percentage errors that occurred in the measurement of physical quantities \(A,\) \(B,\) \(C,\) and \(D\) are \(1\%\), \(2\%\), \(3\%\), and \(4\%\) respectively. Then, the maximum percentage of error in the measurement of \(X,\) where \(X=\frac{A^2 B^{\frac{1}{2}}}{C^{\frac{1}{3}} D^3}\), will be:
| 1. | \(10\%\) | 2. | \(\dfrac{3}{13}\%\) |
| 3. | \(16\%\) | 4. | \(-10\%\) |
A block of mass \(10~\text{kg}\) is in contact with the inner wall of a hollow cylindrical drum of radius \(1~\text{m}.\) The coefficient of friction between the block and the inner wall of the cylinder is \(0.1.\) The minimum angular velocity needed for the cylinder, which is vertical and rotating about its axis, will be:
\(\left(g= 10~\text{m/s}^2\right )\)
| 1. | \(10~\pi~\text{rad/s}\) | 2. | \(\sqrt{10}~\pi~\text{rad/s}\) |
| 3. | \(\dfrac{10}{2\pi}~\text{rad/s}\) | 4. | \(10~\text{rad/s}\) |
A \(800\) turn coil of effective area \(0.05~\text{m}^2\) is kept perpendicular to a magnetic field \(5\times 10^{-5}~\text{T}\). When the plane of the coil is rotated by \(90^{\circ}\)around any of its coplanar axis in \(0.1~\text{s}\), the emf induced in the coil will be:
| 1. | \(0.02~\text{V}\) | 2. | \(2~\text{V}\) |
| 3. | \(0.2~\text{V}\) | 4. | \(2\times 10^{-3}~\text{V}\) |
Two particles \(A\) and \(B\) are moving in a uniform circular motion in concentric circles of radii \(r_A\) and \(r_B\) with speeds \(v_A\) and \(v_B\) respectively. Their time periods of rotation are the same. The ratio of the angular speed of \(A\) to that of \(B\) will be:
| 1. | \( 1: 1 \) | 2. | \(r_A: r_B \) |
| 3. | \(v_A: v_B \) | 4. | \(r_B: r_A\) |
A soap bubble, having a radius of \(1~\text{mm}\), is blown from a detergent solution having a surface tension of \(2.5\times 10^{-2}~\text{N/m}\). The pressure inside the bubble equals at a point \(Z_0\) below the free surface of the water in a container. Taking \(g = 10~\text{m/s}^{2}\), the density of water \(= 10^{3}~\text{kg/m}^3\), the value of \(Z_0\) is:
1. \(0.5~\text{cm}\)
2. \(100~\text{cm}\)
3. \(10~\text{cm}\)
4. \(1~\text{cm}\)
A body weighs \(200\) N on the surface of the earth. How much will it weigh halfway down the centre of the earth?
| 1. | \(100\) N | 2. | \(150\) N |
| 3. | \(200\) N | 4. | \(250\) N |
An electron is accelerated through a potential difference of \(10,000~\text{V}\). Its de-Broglie wavelength is, (nearly):
\(\left(m_e = 9\times 10^{-31}~\text{kg}\right )\)
1. \(12.2~\text{nm}\)
2. \(12.2\times 10^{-13}~\text{m}\)
3. \(12.2\times 10^{-12}~\text{m}\)
4. \(12.2\times 10^{-14}~\text{m}\)
Two parallel infinite line charges with linear charge densities \(+\lambda~\text{C/m}\) and \(+\lambda~\text{C/m}\) are placed at a distance \({R}.\) The electric field mid-way between the two line charges is:
| 1. | \(\frac{\lambda}{2 \pi \varepsilon_0 {R}}~\text{N/C}\) | 2. | zero |
| 3. | \(\frac{2\lambda}{ \pi \varepsilon_0 {R}} ~\text{N/C}\) | 4. | \(\frac{\lambda}{ \pi \varepsilon_0 {R}}~\text{N/C}\) |
An increase in the temperature of a gas-filled container would lead to:
| 1. | decrease in intermolecular distance. |
| 2. | increase in its mass. |
| 3. | increase in its kinetic energy. |
| 4. | decrease in its pressure. |
A particle moving with velocity \(\vec{v}\) is acted by three forces shown by the vector triangle \({PQR}.\) The velocity of the particle will:

| 1. | change according to the smallest force \({\overrightarrow{Q R}}\) |
| 2. | increase |
| 3. | decrease |
| 4. | remain constant |
A disc of radius \(2~\text{m}\) and mass \(100~\text{kg}\) rolls on a horizontal floor. Its centre of mass has a speed of \(20~\text{cm/s}\). How much work is needed to stop it?
| 1. | \(1~\text{J}\) | 2. | \(3~\text{J}\) |
| 3. | \(30~\text{J}\) | 4. | \(2~\text{J}\) |