Passage cells are thin-walled cells found in:
| 1. | endodermis of roots facilitating rapid transport of water from cortex to pericycle |
| 2. | phloem elements that serve as entry points for substances for transport to other plant parts |
| 3. | testa of seeds to enable emergence of growing embryonic axis during seed germination |
| 4. | central region of style through which the pollen tube grows towards the ovary |
The opening of floral buds into flowers is a type of:
1. autonomic movement of locomotion
2. autonomic movement of variation
3. paratonic movement of growth
4. autonomic movement of growth
Telomere repetitive DNA sequences control the function of eukaryotic chromosomes because they:
1. act as replicons
2. are RNA transcription initiators
3. help chromosome pairing
4. prevent chromosome loss
Identify the odd combination of the habitat and the particular animal concerned.
| 1. | Dachigam National Park-Snow Leopard |
| 2. | Sunderbans-Bengal Tiger |
| 3. | Periyar-Snow Leopard |
| 4. | Rann of Kutch-Wild Ass |
In the leaves of C4 plants, malic acid formation during CO2 fixation occurs in the cells of:
1. mesophyll
2. bundle sheath
3. phloem
4. epidermis
A human male produces sperms with the genotypes AB, Ab, aB and ab pertaining to two diallelic characters in equal proportions. What is the corresponding genotype of this person?
1. AaBb
2. AaBB
3. AABb
4. AABB
Which of the following ecosystem types has the highest annual net primary productivity?
1. Tropical rainforest
2. Tropical deciduous forest
3. Temperate evergreen forest
4. Temperate deciduous forest
In human body, which one of the following is anatomically correct?
| 1. | Floating ribs-2 pairs | 2. | Collar bones-3 pairs |
| 3. | Salivary glands-1 pair | 4. | Cranial nerves-10 pairs |
Which of the following is a flowering plant with nodules containing filamentous nitrogen-fixing microorganism?
1. Casuarina equisetifolia
2. Crotolaria juncea
3. Cycas revoluta
4. Cicer arietinum
The population of an insect species shows an explosive increase in numbers during the rainy season followed by its disappearance at the end of the season. What does this show?
| 1. | S-shaped or sigmoid growth of this insect |
| 2. | the food plants mature and die at the end of the rainy season |
| 3. | Its population growth curve is of J-type |
| 4. | The population of its predators increases enormously |
One of the endangered species of Indian medicine plants is:
1. Podophyllum
2. Ocimum
3. Garlic
4. Nepenthes
In hexaploid wheat, the haploid (n) ar basic (x) numbers of chromosomes are:
1. n=7 and x=21
2. n=21 and x=21
3. n=21 and x=14
4. n=21 and x=7
In the cloning of cattle, a fertilized egg is taken out of the mother's womb and:
| 1. | the egg is divided into 4 pairs of cells which are implanted into the womb of other cows |
| 2. | in the eight-cell stage, cells are separated and cultured until small embryos are formed which are implanted into the womb of other cows |
| 3. | in the eight-cell stage, the individual cells are separated under an electrical field for further development in cultural media |
| 4. | from this, up to eight identical twins can be produced |
Which one of the following statements is correct?
| 1. | Extensive use of chemical fertilizers may lead to eutrophication of nearby water bodies |
| 2. | Both Azotobacter and Rhizobium in root nodules of plants |
| 3. | Cyanobacteria such as Anabaena and Nostoc are important mobilizers of phosphates and potassium for plant nutrition in the soil |
| 4. | At present it is not possible to grow maize without chemical fertilizers |
One of the important consequences of geographical isolation is:
1. no change in the isolated fauna
2. preventing speciation
3. speciation through reproductive isolation
4. random creation of new species
Flagellated male gametes are present in all three of which one of the following sets?
1. Anthoceros, Funaria, and Spirogyra
2. Zygnema, Saprolegnia, and Hydrilla
3. Fucus, Marsilea, and Calotropis
4. Riccia, Dryopteris, and Cycas
Molecular basis of organ differentiation depends on the modulation in transcription by:
1. RNA polymerase
2. ribosome
3. transcription factor
4. anticodon
Increased asthmatic attacks in certain seasons are related to:
1. hot and humid environment
2. eating fruits preserved in tin containers
3. inhalation of seasonal pollen
4. low-temperature
Which of the following is a slime mould?
1. Rhizopus
2. Physarum
3. Thiobacillus
4. Anabaena
Which one of the following is an example of a negative feedback loop in humans?
| 1. | Constriction of skin blood vessels and contraction of skeletal muscles when it is too cold |
| 2. | Secretion of tears after falling of sand particles into the eye |
| 3. | Salivation of the mouth at the sight of delicious food |
| 4. | secretion of sweat glands and constriction of the skin blood vessels when it is too hot |
Ergot of rye is caused by a species of:
1. Phytophthora
2. Uncinula
3. Ustilago
4. Claviceps
Geometric representation of age structure is a characteristic of:
1. Biotic community
2. Population
3. Landscape
4. Ecosystem
Which one of the following mammalian cells is not capable of metabolizing glucose to carbon dioxide aerobically?
1. White blood cells
2. Unsatiated muscle cells
3. Liver cells
4. Red blood cells
Feeling the tremors of an earthquake, a scared resident of the seventh floor of a multi-storey building starts climbing down the stairs rapidly. Which hormone initiated this act?
1. Thyroxine
2. Adrenaline
3. Glucagon
4. Gastrin
Among the human ancestors the brain size was more than 1000 CC in:
1. Homo neaderthalensis
2. Homo erectus
3. Ramapithecus
4. Homo habilis
The length of the DNA molecule greatly exceeds the dimensions of the nucleus in eukaryotic cells. How is this DNA accommodated?
1. Deletion of non-essential genes
2. Super-coiling in nucleosomes
3. DNAse digestion
4. Through the elimination of repetitive DNA
The wavelength of light absorbed by Pr form of phytochrome is:
1. 640 nm
2. 680 nm
3. 720 nm
4. 620 nm
Which part of the ovary in mammals acts as an endocrine gland after ovulation?
| 1. | Graafian follicle | 2. | Stroma |
| 3. | Germinal epithelium | 4. | Vitelline membrane |
Which one of the following is not a constituent of the cell membrane?
1. Cholesterol
2. Glycolipids
3. Proline
4. Phospholipids
The concept of chemical evolution is based on:
| 1. | crystallization of chemicals |
| 2. | interaction of water, air, and clay under intense heat |
| 3. | effect of solar radiation on chemicals |
| 4. | possible origin of life by a combination of chemicals under suitable environmental conditions |
In maize, hybrid vigour is exploited by:
1. bombarding the seeds with DNA
2. crossing of two inbred parental lines
3. harvesting seeds from the most productive plants
4. inducing mutations
In gymnosperms, the pollen chamber represents:
| 1. | a cell in the pollen grain in which the sperms are formed |
| 2. | a cavity in the ovule in which pollen grains are stored after pollination |
| 3. | an opening in the megagametophyte through which the pollen tube approaches the egg |
| 4. | the microsporangium in which pollen grains develop |
During transcription, RNA polymerase holoenzyme binds to a gene promoter and assumes a saddle-like structure. What is its DNA-binding sequence?
| 1. | TIAA |
| 2. | AATI |
| 3. | CACC |
| 4. | TATA |
In the prothallus of a vascular cryptogam, the antherozoids and eggs mature at different times. As a result:
| 1. | there is no change in the success rate of fertilization |
| 2. | there is a high degree of sterility |
| 3. | one can conclude that the plant is apomictic |
| 4. | self-fertilization is prevented |
Industrial melanism as observed in peppered moths proves that:
| 1. | The true black melanic forms arise from a recurring natural selection |
| 2. | The melanic form of the moth has no selective advantage over the lighter form in an industrial area |
| 3. | The lighter-form moth has no selective advantage either in polluted industrial areas or non-polluted areas |
| 4. | Melanism is a pollution-generated feature |
"Foolish seedling" disease of rice led to the discovery of:
1. GA
2. ABA
3. 2, 4-D
4. IAA
Differentiation of organs and tissues in a developing organism is associated with:
1. developmental mutations
2. differential expression of genes
3. lethal mutations
4. deletion of a gene
The first acceptor of electrons from an excited chlorophyll molecule of photosystem II is:
| 1. | cytochrome | 2. | iron-sulphur protein |
| 3. | ferredoxin | 4. | quinone |
Lysozyme that is present in perspiration, saliva, and tears destroys:
| 1. | certain fungi | 2. | certain types of bacteria |
| 3. | all viruses | 4. | most virus-infected cells |
During the transmission of nerve impulses through a nerve fibre, the potential on the inner side of the plasma membrane has which type of electric charge?
| 1. | First negative, then positive, and again back to negative |
| 2. | First positive, then negative, and continue to be negative |
| 3. | First negative, then positive, and then continues to be positive |
| 4. | First positive, then negative, and again back to positive |
The Okazaki fragments in DNA chain growth:
| 1. | result in transcription |
| 2. | polymerize in the 3'-to-5' direction and forms replication fork |
| 3. | prove semi-conservative nature of DNA replication |
| 4. | polymerize in the 5'-to-3' direction and explain 3'-to-5' DNA replication |
The two polynucleotide chains in DNA are:
1. parallel
2. discontinuous
3. antiparallel
4. semiconservative
Which one of the following is a viral disease of poultry?
1. Salmonellosis
2. Coryza
3. Newcastle disease
4. Pasteurellosis
Inheritance of skin colour in humans is an example of:
1. chromosomal aberration
2. point mutation
3. polygenic inheritance
4. codominance
One gene-one enzyme relationship was established for the first time in:
1. Neurosporacrassa
2. Salmonella typhimurium
3. Escherichia coli
4. Diplococcuspneumonia
A genetically engineered micro-organism used successfully in the bioremediation of oil spills is a species of:
1. Pseudomonas
2. Trichoderma
3. Xanthomonas
4. Bacillus
In which one of the following preparations are you likely to come across cell junctions most frequently?
1. Ciliated epithelium
2. Thrombocytes
3. Tendon
4. Hyaline cartilage
Which one of the following pairs is mismatched?
| 1. | Pila globosa - pearl oyster | 2. | Apis indica- honey bee |
| 3. | Laccifer lacca- lac insect | 4. | Bombyx mori- silkworm |
About 98 percent of the mass of every living organism is composed of just six elements including carbon, hydrogen, nitrogen, oxygen and:
1. phosphorus and sulphur
2. sulphur and magnesium
3. magnesium and sodium
4. calcium and phosphorus
Two genes R and Y are located very close on the chromosomal linkage map of the maize plant. When RRYY and rryy genotypes are hybridized, then F2 segregation will show:
1. higher number of the recombinant types
2. segregation in the expected 9:3:3:1 ratio
3. segregation in a 3:1 ratio
4. higher number of the parental types
What is common between parrot, platypus, and kangaroo?
1. Homeothermy
2. Toothless jaws
3. Functional post-anal tail
4. Oviparity
Which one of the following is being utilized as a source of bio-diesel in the Indian countryside?
1. Euphorbia
2. Beetroot
3. Sugarcane
4. Pongamia
In which one of the following the BOD (Biochemical Oxygen Demand) of sewage (S), distillery effluent (DE), paper mill effluent (PE) and sugar mill effluent (SE) have been arranged in ascending order?
1. SE < S < PE < DE
2. SE < PE < S <DE
3. PE < S < SE < DE
4. S < DE < PE <SE
In the human female, menstruation can be deferred by the administration of:
1. LH only
2. Combination of FSH and LH
3. Combination of estrogen and progesterone
4. FSH only
Select the correct statement from the following:
| 1. | Darwin variations are small and directionless |
| 2. | fitness is the end result of the ability to adapt and gets selected by nature |
| 3. | all mammals except whales and have seven cervical vertebrae |
| 4. | Mutations are random and directional |
Two plants can be conclusively said to belong to the same species if they:
| 1. | can reproduce freely with each other and form seeds |
| 2. | have more than 90 percent similar genes |
| 3. | look similar and possess identical secondary metabolites |
| 4. | have the same number of chromosomes |
A sequential expression of a set of human genes occurs when a steroid molecule binds to the:
1. transfer RNA
2. messenger RNA
3. DNA sequence
4. ribosome
In a coal fired power plant, electrostatic precipitators are installed to control the emission of:
1.
2.
3. SPM
4. CO
Probiotics are:
1. safe antibiotics
2. cancer inducing microbes
3. new kind of food allergens
4. live microbial food supplement
Which pair of the following belongs to Basidiomycetes?
1. Bird's nest fungi and Pufballs
2. Puffballs and Claviceps
3. Peziza and Stinkhorns
4. Morchella and Mushrooms
Spore dissemination in some liverworts is aided by:
1. elaters
2. indusium
3. calypatra
4. peristome teeth
A person who is on a long hunger strike and is surviving only on water, will have:
1. more sodium in his urine
2. less amino acids in his urine
3. more glucose in his blood
4. less urea in his urine
Which one of the following pairs is not correctly matched?
| 1. | Abscisic acid - Stomatal closure |
| 2. | Gibberellic acid - Leaves fall |
| 3. | Cytokinin - Cell division |
| 4. | IAA - Cell wall elongation |
A common test to find the genotype of a hybrid is by:
1. crossing of one F2 progeny with a male parent
2. crossing of one F2 progeny with a female parent
3. studying the sexual behaviour of F1 progenies
4. crossing of one F1 progeny with a male parent
Which one of the following is a matching pair of a body feature and the animal possessing it?
1. Post-anal-tail - Octopus
2. Ventral Central nervous system - Leech
3. Pharyngeal gills slits absent in embryo - Chamaeleon
4. Ventral heart - Scorpion
Which of the following pairs are correctly matched?
| Animals | Morphological features | |
| A. | Crocodile | 4-chambered heart |
| B. | Sea urchin | Parapodia |
| C. | Obelia | Metagenesis |
| D. | Lemur | Thecodont |
1. A, C, and D
2, B, C, and D
3. Only A and D
4. Only A and B
If you suspect a major deficiency of antibodies in a person, to which of the following would you look for confirmatory evidence?
1. Serum albumins
2. Serum globulins
3. Fibrinogen in the plasma
4. Haemocytes
A person is having problems with calcium and phosphorus metabolism in his body. Which one of the following glands may not be functioning properly?
1. Parathyroid
2. Parotid
3. Pancreas
4. Thyroid
What is true about Nereis, Scorpion, Cockroach, and Silverfish?
| 1. | They all have jointed paired appendages |
| 2. | They all possess a dorsal heart |
| 3. | None of them is aquatic |
| 4. | They all belong to the same phylum |
The finches of the Galapagos islands provide evidence in favour of:
1. special creation
2. evolution due to mutation
3. retrogressive evolution
4. biogeographical evolution
Two cells A and B are contiguous. Cell A has osmotic pressure 10 atm, turgor pressure-7 atm, and diffusion pressure deficit 3 atm. Cell B has an osmotic pressure of 8 atm, turgor pressure of 3 atm, and diffusion pressure deficit of 5 atm. The result will be:
1. Movement of water from cell B to A
2. No movement of water
3. Equilibrium between the two
4. Movement of water from cell A to B
Which one of the following elements is not an essential micronutrient for plant growth?
1. Mn
2. Zn
3. Cu
4. Ca
Male gametes in angiosperms are formed by the division of:
1. microspore
2. generative cell
3. vegetative cell
4. microspore mother cell
Which one of the following is surrounded by a callose wall?
1. Microspore mother cell
2. Male gamete
3. Egg
4. Pollen grain
Which one of the following statements about mycoplasma is wrong?
1. They are also called PPLO
2. They are pleomorphic
3. They are sensitive to penicillin
4. They cause disease in plants
Which one of the following is not a bioindicator of water pollution?
1. Sludge-worms
2. Blood-worms
3. Stone files
4. Sewage fungus
Select the wrong statement from the following:
| 1. | Both chloroplast and mitochondria contain an inner and an outer membrane. |
| 2. | Both chloroplasts and mitochondria have an internal compartment, the thylakoid space bounded by the thylakoid membrane. |
| 3. | Both chloroplasts and mitochondria contain DNA. |
| 4. | The chloroplasts are generally much larger than mitochondria. |
Which one of the following pairs of organisms are exotic species introduced in India?
1. Ficus religiosa. Lantana camara
2. Lantana camara, Water hyacinth
3. Water hyacinth, Prosopis cineraria
4. Nile perch, Ficus religiosa
Which one of the following statements is correct?
| 1. | Stem cells are specialized |
| 2. | There is no evidence of the existence of gills during embryogenesis of mammals |
| 3. | All plant and animal cells are totipotent |
| 4. | Ontogeny repeats phylogeny |
Bowman's glands are located in the:
1. proximal end of uriniferous tubules
2. anterior pituitary
3. female reproductive system of cockroach
4. olfactory epithelium of our nose
Living organisms can be unexceptionally distinguished from non-living things on the basis of their ability for:
| 1. | responsiveness to touch |
| 2. | interaction with the environment and progressive evolution |
| 3. | reproduction |
| 4. | growth and movement |
Compared to a bull, a bullock is docile because of:
1. higher levels of thyroxin
2. higher levels of cortisone
3. lower levels of blood testosterone
4. lower levels of adrenalin/noradrenalin in its blood
What is common to whales, seals, and sharks?
1. Seasonal migration
2. Thick subcutaneous fat
3. Convergent evolution
4. Homeothermy
All enzymes of the TCA cycle are located in the mitochondrial matrix except one which is located in inner mitochondrial membranes in eukaryotes and in the cytosol in prokaryotes. This enzyme is:
| 1. | lactate dehydrogenase | 2. | isocitrate dehydrogenase |
| 3. | malate dehydrogenase | 4. | succinate dehydrogenase |
Which one of the following pairs of structures distinguishes a nerve cell from other types of cells?
1. Perikaryon and dendrites
2. Vacuoles and fibres
3. Flagellum and medullary sheath
4. Nucleus and mitochondria
Ultrasound of how much frequency is beamed into a human body for sonography?
1. 30-40 MHz
2. 15-30 MHz
3. 1-15 MHz
4. 45-70 MHz
The biological organisation starts with:
1. Submicroscopic molecular level
2. Cellular level
3. Organisms level
4. Atomic level
Which one of the following pairs is wrongly matched?
1. Methanogens - Gobar gas
2. Yeast - Ethanol
3. Streptomycetes - Antibiotic
4. Coliforms - Vinegar
In pea plants, yellow seeds are dominant to green. If a heterozygous yellow-seeded plant is crossed with a green-seeded plant, what ratio of yellow and green-seeded plants would you expect in F1generation?
1. 50:50
2. 9:1
3. 1:3
4. 3:1
Adaptive radiation refers to:
| 1. | adaptations due to geographical isolation |
| 2. | evolution of different species from a common ancestor |
| 3. | migration of members of a species to different geographical areas |
| 4. | power of adaptation in an individual to a variety of environments |
If the mean and the median pertaining to a certain character of a population are of the same value, the following is most likely to occur:
1. a normal distribution
2. a bi-modal distribution
3. a T-shaped curve
4. a skewed curve
When two species of different genealogy come to resemble each other as a result of adaptation, the phenomenon is termed:
1. divergent evolution
2. microevolution
3. co-evolution
4. convergent evolution
Which one of the following is a fat-soluble vitamin and its related deficiency disease?
| 1. | Ascorbic acid - Scurvy |
| 2. | Retinol - Xerophthalmia |
| 3. | Cobalamine - Beri-Beri |
| 4. | Calciferol - Pellagra |
ICBN stands for:
| 1. | Indian Congress of Biological Names |
| 2. | International Code of Botanical Nomenclature |
| 3. | Indian Code of Botanical Nomenclature |
| 4. | International Congress of Biological Names |
If you are asked to classify the various algae into distinct groups, which of the following characters you should choose?
| 1. | Types of pigments present in the cell |
| 2. | Nature of stored food materials in the cell |
| 3. | Structural organisation of thallus |
| 4. | Chemical compositions of the cell wall |
A plant requires magnesium for:
1. holding cells together
2. protein synthesis
3. chlorophyll synthesis
4. cell wall development
The overall goal of glycolysis, the Krebs cycle, and the electron transport system is the formation of:
1. ATP in small stepwise units.
2. ATP in one large oxidation reaction
3. Sugars
4. Nucleic acids
A high density of elephant population in an area can result in:
1. mutualism
2. Intraspecific competition
3. Interspecific completion
4. Predation on one another
A drop of each of the following is placed separately on four slides. Which of them will not coagulate?
| 1. | blood plasma |
| 2. | blood serum |
| 3. | Sample from the thoracic duct of the lymphatic system |
| 4. | Whole blood from the pulmonary vein |
For a critical study of secondary growth in plants, which one of the following pairs is suitable:
1. Sugarcane and sunflower
2. Teak and pine
3. Deodar and Fern
4. Wheat and maidenhair fern
The pOH of a solution at 25 °C that contains 1×10-10 M of hydronium ions is:
| 1. | 7.00 | 2. | 4.00 |
| 3. | 9.00 | 4. | 1.00 |
The compound that gives a pair of enantiomorphs is:
(en = NH2 CH2CH2 NH2)
1.
2.
3.
4.
The correct order of increasing thermal stability of K2CO3, MgCO3, CaCO3, and BeCO3 is:
1.
2.
3.
4.
A weak acid, HA, has a Ka of 1.00×10-5. If 0.100 mole of this acid is dissolved in one litre of water, the percentage of acid dissociated at equilibrium is closest to:
1. 99.0%
2. 1%
3. 99.9%
4. 0.100%
The number of moles of KMnO4 that will be needed to react with one mole of sulphite ion in an acidic solution is:
| 1. | \(3 \over 5\) | 2. | \(4 \over 5\) |
| 3. | \(2 \over 5\) | 4. | 1 |
Identify the incorrect statement among the following:
| 1. | There is a decrease in the radii of the atoms or ions as one proceeds from La to Lu. |
| 2. | Lanthanoid contraction is the accumulation of successive shrinkages |
| 3. | As a result of lanthanoid contraction, the properties of the 4d series of the transition elements have no similarities with the 5d series of elements |
| 4. | The shielding power of 4f electrons is quite weak. |
Among the following, the most characteristic oxidation states for lead and tin are, respectively:
1. +4, +2
2. +2, +4
3. +4, +4
4. +2, +2
The correct order of C-O bond length among CO, , CO2 is:
1.
2.
3.
4.
What is the correct sequence of acidity for the following compounds?
| 1. | CH3COOH > BrCH2COOH > ClCH2COOH > FCH2COOH |
| 2. | FCH2COOH > CH3COOH > BrCH2COOH > ClCH2COOH |
| 3. | BrCH2COOH > ClCH2COOH > FCH2COOH > CH3COOH |
| 4. | FCH2COOH > ClCH2COOH > BrCH2COOH > CH3COOH |
The following equilibrium constants are given:
N2 + 3H2 ⇌ 2NH3; K1
N2 + O2 ⇌ 2NO; K2
H2 + 1/2O2 ⇌ H2O; K3
The equilibrium constant for the oxidation of NH3 by oxygen to give NO is:
1.
2.
3.
4.
Which one of the following vitamins is water-soluble?
| 1. | Vitamin-B | 2. | Vitamin-E |
| 3. | Vitamin-K | 4. | Vitamin-A |
If there is no rotation of plane-polarized light by a compound in a specific solvent thought to be chiral, it may mean that:
1. The compound is certainly a chiral.
2. The compound is certainly meso.
3. There is no compound in the solvent.
4. The compound may be a racemic mixture.
Consider the following sets of quantum numbers:
| n | I | m | s | |
| (i) | 3 | 0 | 0 | +1/2 |
| (ii) | 2 | 2 | 1 | +1/2 |
| (iii) | 4 | 3 | -2 | -1/2 |
| (iv) | 1 | 0 | -1 | -1/2 |
| (v) | 3 | 2 | 3 | +1/2 |
Which of the following sets of quantum numbers is not possible?
1. ii, iii, and iv
2. i, ii, iii, and iv
3. ii, iv and v
4. i and iii
Which one of the following ions is the most stable in an aqueous solution?
(At. No. Ti = 22, V = 23, Cr = 24, Mn = 25)
1. \(\text{Cr}^{3 +}\)
2. \(\text V^{3 +}\)
3. \(\text{Ti}^{3 +}\)
4. \(\text{Mn}^{3 +}\)
Concentrated aqueous sulphuric acid is 98% H2SO4 by mass and has a density of 1.80 g mL-1. The volume of acid required to make one litre of 0.1 M H2SO4 solution is:
1. 11.10 mL
2. 16.65 mL
3. 22.20 mL
4. 5.55 mL
The ionic species that has the greatest proton affinity to form stable compound is:
1.
2.
3.
4.
CH3— CHCl—CH2—CH3 has a chiral center. Which one of the following represents its R configuration?
1. \(H_{3}C - \underset{H}{\overset{C_{2}H_{5}}{\underset{|}{\overset{|}{C}}}} - {Cl}\)
2. \(H - \underset{Cl}{\overset{C_{2}H_{5}}{\underset{|}{\overset{|}{C}}}} - {CH}_{3}\)
3. \({Cl} - \underset{H}{\overset{C_{2}H_{5}}{\underset{|}{\overset{|}{C}}}} - {CH}_{3}\)
4. \(H - \underset{C_{2}H_{5}}{\overset{{CH}_{3}}{\underset{|}{\overset{|}{C}}}} - {Cl}\)
0.5 molal aqueous solution of a weak acid (HX) is 20 % ionised. The lowering in the freezing point of the solution will be:
[Kf for water = 1.86 K kg mol-1]
1. -1.12 K
2. 0.56 K
3. 1.12 K
4. -0.56 K
Which one of the following polymers is prepared by condensation polymerization?
1. Nylon-6, 6
2. Teflon
3. Rubber
4. Styrene
The Langmuir adsorption isotherm is deduced using the assumption:
| 1. | The adsorption takes place in multilayers |
| 2. | The adsorption sites are equivalent in their ability to adsorb the particles |
| 3. | The heat of adsorption varies with coverage |
| 4. | The adsorbed molecules interact with each other |
The reaction of hydrogen and iodine monochloride is given as:
H2(g) + 2ICl(g) → 2HCl(g) + I2(g)
This reaction is of first order with respect to H2(g) and ICl(g), for which of the following proposed mechanisms:
Mechanism A:
H2(g) + 2ICl(g) → 2HCl(g) + I2(g)
Mechanism B:
H2(g) + ICl(g) →HCl(g) + HI(g); slow
HI(g) + ICl(g) →HCl(g) + I2(g); fast
1. B Only
2. A and B both
3. Neither A nor B
4. A only
RNA and DNA are chiral molecules due to:
1. L- sugar component
2. Chiral bases
3. Chiral phosphate exter unit
4. D- sugar component
In which of the following the hydration energy is higher than the lattice energy?
1.
2.
3.
4.
Which compound, upon reduction with lithium aluminium hydride, yields a secondary amine?
1. Nitroethane
2. Methylisocyanide
3. Acetamide
4. Methyl cyanide
Consider the following reactions:
| (i) | H+(aq) + OH-(aq) → H2O(l) ΔH = -x1 kJmol-1 |
| (ii) | H2(g) + 1/2O2(g) → H2O(l) ΔH = -x2 kJmol-1 |
| (iii) | CO2(g) + H2(g) → CO (g) + H2O(l) ΔH = -x3 kJmol-1 |
| (iv) | C2H2(g) + 5/2O2(g) → 2CO2 + H2O(l) ΔH = -x4 kJmol-1 |
Enthalpy of formation of H2O(l) is :
1.
2.
3.
4.
The bond energy of H—H and Cl-Cl is 430 kJ
mol-1 and 240 kJ mol-1 respectively and ΔHf for HCl is -90 kJ mol-1. The bond enthalpy of HCl is:
1. 290
2. 380
3. 425
4. 245
Reduction of aldehydes and ketones into hydrocarbons using amalgam and conc. HCl is called:
1. Clemmensen reduction
2. Cope reduction
3. Dow reduction
4. Wolff-Kishner reduction
Which one of the following anions is present in the chain structure silicates?
1.
2.
3.
4.
The fraction of total volume occupied by the atoms present in a simple cube is:
1.
2.
3.
4.
For the following:
(i) I-
(ii) Cl-
(iii) Br-
The increasing order of nucleophilicity would be:
1. I-<Br-<Cl-
2. Cl-<Br-<I-
3. I- <Cl-<Br-
4. Br-<Cl-<I-
Which one of the following orders correctly represents the increasing acid strengths of the given acids?
1.
2.
3.
4.
The product formed in the below-mentioned reaction is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
A configuration with the lowest ionization enthalpy among the following is:
1. \(1 s^2 2 s^2 2 p^5\)
2. \(1 s^2 2 s^2 2 p^3\)
3. \(1 s^2 2 s^2 2 p^6 3 s^1\)
4. \(1 s^2 2 s^2 2 p^6\)
Predict the product C obtained in the following reaction of 1-butyne:
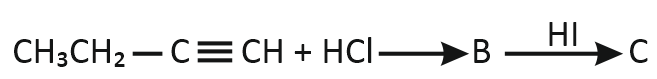
| 1. | 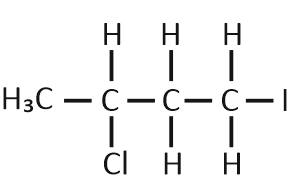 |
| 2. | 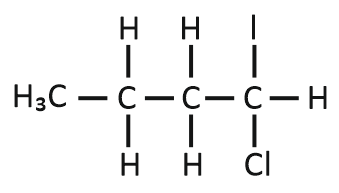 |
| 3. | 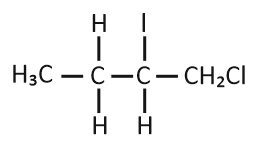 |
| 4. | 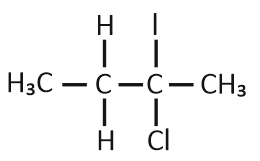 |
An element X has the following isotopic composition:
200X: 90%
199X: 8.0%
202X: 2.0%
The weighted average atomic mass of the naturally-occurring element X is closest to:
1. 200 amu
2. 201 amu
3. 202 amu
4. 199 amu
In a first order reaction A \(\overset{ }{\rightarrow}\) B, if k is rate constant and initial concentration of the reactant A is 0.5 M then the half-life is :
(1) \(\frac{0 . 693}{0 . 5 k}\)
(2) \(\frac{log 2}{k}\)
(3) \(\frac{log 2}{k \sqrt{0 . 5}}\)
(4) \(\frac{ln 2}{k}\)
| 1. | Carbon and hydrogen act as suitable reducing agents for metal sulphides |
| 2. | The \(\Delta_f G^0\) of the sulphide is greater than those for \(C S_2\) and \(\mathrm{H}_2 \mathrm{~S}\) |
| 3. | The \(\Delta_f G^0\) is negative for roasting of sulphur ore to oxide. |
| 4. | Roasting of the sulphide to the oxide is thermodynamically feasible |
If 60% of a first-order reaction was completed in 60 min, 50% of the same reaction would be completed in approximately:
(log 4 = 0.60, log 5 = 0.69)
| 1. | 50 min | 2. | 45 min |
| 3. | 60 min | 4. | 40 min |
For a given reaction,
Cu(s) + 2Ag+ (aq) → Cu2+(aq)+2Ag(s);
E0=0.46 V at 298 K . The equilibrium constant will be :
| 1. | 2. | ||
| 3. | 4. |
A compound with the molecular formula C5H10 that yields acetone on ozonolysis is:
1. 2-Methyl-2-butene
2. 2-Methyl-1-butene
3. Cyclopentane
4. 3-Methyl-1-butene
Sulphide ores of metals are usually concentrated by the froth flotation process. Which one of the following sulphide ores offers an exception and is concentrated by chemical leaching?
1. Argentite
2. Galena
3. Copper pyrite
4. Sphalerite
The efficiency of a fuel cell is given by:
1.
2.
3.
4.
The compound on hydrolysis of 50% aqueous sodium hydroxide that produces the corresponding alcohol and acid is:
1.
2.
3.
4.
Identify the correct order of the size of the following:
1.
2.
3.
4.
Consider the following compounds:
| (i) | C6H5COCl | (ii) |  |
| (iii) |  |
(iv) |  |
What is the correct decreasing order of their reactivity towards hydrolysis?
1. (ii) > (iv) > (iii) > (i)
2. (i) > (ii) > (iii) > (iv)
3. (iv) > (ii) > (i) > (iii)
4. (ii) > (iv) > (i) > (iii)
The product formed in aldol condensation is:
| 1. | A \(\beta\)-hydroxy acid |
| 2. | A \(\beta\)-hydroxy aldehyde or a \(\beta\)-hydroxy ketone |
| 3. | An \(\alpha\)-hydroxy aldehyde or ketone |
| 4. | An \(\alpha\)-\(\beta\) unsaturated ester |
If NaCl is doped with 10-4mol% of SrCl2, the concentration of cation vacancies will be (NA= 6.02×1023mol-1):
1.
2.
3.
4.
The d-electron configurations of Cr2+, Mn2+, Fe2+ and Ni2+ and 3d4, 3d5, 3d6 and 3d8 respectively.
Which one of the following aqua complexes will exhibit the minimum paramagnetic behaviour?
(At. No. Cr = 24, Mn = 25, Fe = 26, Ni = 28)
1. \(\left[\mathrm{Mn}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}\)
2. \(\left[\mathrm{Fe}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}\)
3. \(\left[\mathrm{Ni}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}\)
4. \(\left[\mathrm{Cr}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}\)
The order of decreasing reactivity towards an electrophilic reagent, for the following:
(i) Benzene (ii) Toluene
(iii) Chlorobenzene and
(iv) Phenol
would be:
1. (i) > (ii) > (iii) > (iv)
2. (ii) > (iv) > (i) > (iii)
3. (iv) > (iii) > (ii) > (i)
4. (iv) > (ii) > (i) > (iii)
In which of the following pairs, the two species are iso-structural?
1.
2.
3.
4.
Nickel shows the ferromagnetic property at room temperature. If the temperature is increased beyond Curie's temperature, then it will show:
1. paramagnetism
2. anti-ferromagnetism
3. no magnetic property
4. diamagnetism
In the radioactive decay process, the negatively charged emitted β-particles are:
| 1. | the electrons present inside the nucleus |
| 2. | the electrons produced as a result of the decay of neutrons inside the nucleus |
| 3. | the electrons produced as a result of collisions between atoms |
| 4. | the electrons orbiting around the nucleus |

| 1. | \(1.8 \times 10^8 ~\text{m/s}\) | 2. | \(2.4 \times 10^8~\text{m/s}\) |
| 3. | \(3.0 \times 10^8~\text{m/s}\) | 4. | \(1.2 \times 10^8~\text{m/s}\) |
What is the value of inductance L for which the current is maximum in a series LCR circuit with C = 10 μF and ω=1000 s-1?
1. 100 mH
2. 1 mH
3. cannot be calculated unless R is known
4. 10 mH
Three-point charges \(+q\), \(-2q\) and \(+q\) are placed at points \((x=0,y=a,z=0)\), \((x=0, y=0,z=0)\) and \((x=a, y=0, z=0)\), respectively. The magnitude and direction of the electric dipole moment vector of this charge assembly are:
| 1. | \(\sqrt{2}qa\) along \(+y\) direction |
| 2. | \(\sqrt{2}qa\) along the line joining points \((x=0,y=0,z=0)\) and \((x=a,y=a,z=0)\) |
| 3. | \(qa\) along the line joining points \((x=0,y=0,z=0)\) and \((x=a,y=a,z=0)\) |
| 4. | \(\sqrt{2}qa\) along \(+x\) direction |
A nucleus has a mass represented by \(M(A, Z).\) If \(M_P\) and \(M_n\) denote the mass of proton and neutron respectively and BE the binding energy, then:
1.
2.
3.
4.
The position of a particle with respect to time \(t\) along the \({x}\)-axis is given by \(x=9t^{2}-t^{3}\) where \(x\) is in metres and \(t\) in seconds. What will be the position of this particle when it achieves maximum speed along the \(+{x} \text-\text{direction}?\)
1. \(32~\text m\)
2. \(54~\text m\)
3. \(81~\text m\)
4. \(24~\text m\)
The total power dissipated in watts in the circuit shown below is:


| 1. | \(16\) W | 2. | \(40\) W |
| 3. | \(54\) W | 4. | \(4\) W |
In the following circuit, the output \(Y\) for all possible inputs \(A\) and \(B\) is expressed by the truth table:

| 1. | A | B | Y | 2. | A | B | Y |
| 0 | 0 | 0 | 0 | 0 | 1 | ||
| 0 | 1 | 0 | 0 | 1 | 1 | ||
| 1 | 0 | 0 | 1 | 0 | 1 | ||
| 1 | 1 | 1 | 1 | 1 | 0 | ||
| 3. | 0 | 0 | 1 | 4. | 0 | 0 | 0 |
| 0 | 1 | 0 | 0 | 1 | 1 | ||
| 1 | 0 | 0 | 1 | 0 | 1 | ||
| 1 | 1 | 1 | 1 | 1 | 1 |
Assuming the sun to have a spherical outer surface of radius \(r,\) radiating like a black body at temperature \(t^\circ \text{C},\) the power received by a unit surface of the earth (normal to the incident rays) at a distance \(R\) from the centre of the sun will be:
(where \(\sigma\) is Stefan's constant)
| 1. | \(\dfrac{4\pi r^2\sigma t^4}{R^2}\) | 2. | \(\dfrac{r^2\sigma(t+273)^4}{4\pi R^2}\) |
| 3. | \(\dfrac{16\pi^2r^2\sigma t^4}{R^2}\) | 4. | \(\dfrac{r^2\sigma(t+273)^4}{R^2}\) |
A particle starting from the origin \((0,0)\) moves in a straight line in the \((x,y)\) plane. Its coordinates at a later time are (, \(3).\) The path of the particle makes an angle of __________ with the \(x\)-axis:
1. \(30^\circ\)
2. \(45^\circ\)
3. \(60^\circ\)
4. \(0\)
A wheel has an angular acceleration of \(3.0\) rad/s2 and an initial angular speed of \(2.0\) rad/s. In a time of \(2\) s, it has rotated through an angle (in radians) of:
| 1. | \(6\) | 2. | \(10\) |
| 3. | \(12\) | 4. | \(4\) |
The resistance of an ammeter is 13 Ω and its scale is graduated for a current up to 100 A. After an additional shunt has been connected to this ammeter, it becomes possible to measure currents up to 750 A by this ammeter. The value of shunt resistance is:
1. 20
2. 2
3. 0.2
4. 2 k
Under the influence of a uniform magnetic field a charged particle is moving in a circle of radius R with constant speed v. The time period of the motion:
1. depends on v and not on R.
2. depends on both R and v.
3. is independent of both R and v.
4. Depends on R and not on v.
Two condensers, one of capacity \(C\) and the other of capacity \(\frac{C}2\) are connected to a \(V\) volt battery, as shown in the figure.

The energy stored in the capacitors when both condensers are fully charged will be:
1. \(2CV^2\)
2. \({1 \over4}CV^2\)
3. \({3 \over4}CV^2\)
4. \({1 \over2}CV^2\)
A uniform rod \(AB\) of length \(l\) and mass \(m\) is free to rotate about point \(A\). The rod is released from rest in the horizontal position. Given that the moment of inertia of the rod about \(A\) is \(\dfrac{ml^2}{3}\) the initial angular acceleration of the rod will be:

1. \(\dfrac{2g}{3l}\)
2. \(\dfrac{mgl}{2}\)
3. \(\dfrac{3}{2}gl\)
4. \(\dfrac{3g}{2l}\)
The frequency of a light wave in a material is 2×1014 Hz and the wavelength is 5000 Å. The refractive index of the material will be:
1. 1.40
2. 1.50
3. 3.00
4. 1.33
In the energy band diagram of a material shown below, the open circles and filled circles denote holes and electrons respectively. The material is a/an: 
1. p-type semiconductor
2. insulator
3. metal
4. n-type semiconductor
A car moves from \(X\) to \(Y\) with a uniform speed \(v_u\) and returns to \(X\) with a uniform speed \(v_d.\) The average speed for this round trip is:
| 1. | \(\dfrac{2 v_{d} v_{u}}{v_{d} + v_{u}}\) | 2. | \(\sqrt{v_{u} v_{d}}\) |
| 3. | \(\dfrac{v_{d} v_{u}}{v_{d} + v_{u}}\) | 4. | \(\dfrac{v_{u} + v_{d}}{2}\) |
A particle executes simple harmonic oscillation with an amplitude a. The period of oscillation is T. The minimum time taken by the particle to travel half of the amplitude from the equilibrium position is:
1.
2.
3.
4.
A \(5~\text W\) emits monochromatic light of wavelength \(5000~\mathring{A}.\) When placed \(0.5~\text m\) away, it liberates photoelectrons from a photosensitive metallic surface. When the source is moved \(1.0~\text m\) away, the number of photoelectrons liberated is reduced by a factor of:
1. \(4\)
2. \(8\)
3. \(16\)
4. \(2\)
A block \(B\) is pushed momentarily along a horizontal surface with an initial velocity \(v.\) If \(\mu\) is the coefficient of sliding friction between \(B\) and the surface, the block \(B\) will come to rest after a time:
1. \(v \over g \mu\)
2. \(g \mu \over v\)
3. \(g \over v\)
4. \(v \over g\)
Two radioactive substances A and B have decay constants 5λ and λ respectively. At t = 0, they have the same number of nuclei. The ratio of the number of nuclei of A to those of B will be after a time interval:
1.
2.
3.
4.
A hollow cylinder has a charge \(q\) coulomb within it (at the geometrical centre). If \(\phi\) is the electric flux in units of Volt-meter associated with the curved surface \(B,\) the flux linked with the plane surface \(A\) in units of volt-meter will be:

1. \(\frac{1}{2}\left(\frac{q}{\varepsilon_0}-\phi\right)\)
2. \(\frac{q}{2\varepsilon_0}\)
3. \(\frac{\phi}{3}\)
4. \(\frac{q}{\varepsilon_0}-\phi\)
A transformer is used to light a \(100~\text{W}\) and \(110~\text{V}\) lamp from a \(220~\text{V}\) main. If the main current is \(0.5~\text{A},\) the efficiency of the transformer is approximately:
1. \(30\%\)
2. \(50\%\)
3. \(90\%\)
4. \(10\%\)
A particle of mass \(m\) moves in the XY plane with a velocity \(v\) along the straight line AB. If the angular momentum of the particle with respect to the origin \(O\) is \(L_A\) when it is at \(A\) and \(L_B\) when it is at \(B,\) then:
| 1. | \(L_A>L_B\) |
| 2. | \(L_A=L_B\) |
| 3. | the relationship between \(L_A\) and \(L_B\) depends upon the slope of the line \(AB.\) |
| 4. | \(L_A<L_B\) |
The total energy of an electron in the ground state of a hydrogen atom is -13.6 eV. The kinetic energy of an electron in the first excited state is:
1. 3.4 eV
2. 6.8 eV
3. 13.6 eV
4. 1.7 eV
A steady current of 1.5 A flows through a copper voltmeter for 10 min. If the electrochemical equivalent of copper is 30 × 10-5 g C-1, the mass of copper deposited on the electrode will be:
1. 0.40 g
2. 0.50 g
3. 0.67 g
4. 0.27 g
In a mass spectrometer used for measuring the masses of ions, the ions are initially accelerated by an electric potential \(V\) and then made to describe semi-circular paths of radius \(R\) using a magnetic field \(B\). If \(V\) and \(B\) are kept constant, the ratio of \(\left(\frac{\text{Charge on the ion}}{\text{Mass of the ion}} \right)\) will be proportional to:
1. \(\frac{1}{R}\)
2. \(\frac{1}{R^2}\)
3. \(R^2\)
4. \(R\)
Three resistances \(\mathrm P\), \(\mathrm Q\), and \(\mathrm R\), each of \(2~\Omega\) and an unknown resistance \(\mathrm{S}\) form the four arms of a Wheatstone bridge circuit. When the resistance of \(6~\Omega\) is connected in parallel to \(\mathrm{S}\), the bridge gets balanced. What is the value of \(\mathrm{S}\)?
| 1. | \(2~\Omega\) | 2. | \(3~\Omega\) |
| 3. | \(6~\Omega\) | 4. | \(1~\Omega\) |
The electric and magnetic fields of an electromagnetic wave are:
| 1. | In phase and parallel to each other |
| 2. | In opposite phases and perpendicular to each other |
| 3. | In opposite phases and parallel to each other |
| 4. | In phase and perpendicular to each other |
A mass of \(2.0\) kg is put on a flat pan attached to a vertical spring fixed on the ground as shown in the figure. The mass of the spring and the pan is negligible. When pressed slightly and released, the mass executes a simple harmonic motion. The spring constant is \(200\) N/m. What should be the minimum amplitude of the motion, so that the mass gets detached from the pan?
(Take \(g=10\) m/s2)
| 1. | \(8.0\) cm |
| 2. | \(10.0\) cm |
| 3. | any value less than \(12.0\) cm |
| 4. | \(4.0\) cm |
Two satellites of Earth, \(S_1\), and \(S_2\), are moving in the same orbit. The mass of \(S_1\) is four times the mass of \(S_2\). Which one of the following statements is true?
| 1. | The time period of \(S_1\) is four times that of \(S_2\). |
| 2. | The potential energies of the earth and satellite in the two cases are equal. |
| 3. | \(S_1\) and \(S_2\) are moving at the same speed. |
| 4. | The kinetic energies of the two satellites are equal. |
Charges +q and –q are placed at points A and B, respectively; which are at a distance 2L apart. C is the midpoint between A and B. The work done in moving a charge +Q along the semicircle CRD is:
1.
2.
3.
4.
A beam of electrons passes un-deflected through mutually perpendicular electric and magnetic fields. Where do the electrons move if the electric field is switched off and the same magnetic field is maintained?
| 1. | in an elliptical orbit. |
| 2. | in a circular orbit. |
| 3. | along a parabolic path. |
| 4. | along a straight line. |
A black body is at \(727^\circ\text{C}.\) The rate at which it emits energy is proportional to:
| 1. | \((727)^2\) | 2. | \((1000)^4\) |
| 3. | \((1000)^2\) | 4. | \((727)^4\) |
An engine has an efficiency of . When the temperature of the sink is reduced by , its efficiency is doubled. the temperature of the source is:
1. 124oC
2. 37oC
3. 62oC
4. 99oC
Monochromatic light of frequency 6.0×1014 Hz is produced by a laser. The power emitted is 2×10-3 W. The number of photons emitted, on average, by the source per second is:
1.
2.
3.
4.
For a cubic crystal structure, which one of the following relations indicating the cell characteristic is correct?
1.
2.
3.
4.
A common emitter amplifier has a voltage gain of 50, an input impedance of 100Ω, and an output impedance of 200Ω. The power gain of the amplifier is:
1. 500
2. 1000
3. 1250
4. 100
The phase difference between the instantaneous velocity and acceleration of a particle executing simple harmonic motion is:
1. 0.5
2.
3. 0.707
4. zero
A vertical spring with a force constant \(k\) is fixed on a table. A ball of mass \(m\) at a height \(h\) above the free upper end of the spring falls vertically on the spring so that the spring is compressed by a distance \(d\). The net work done in the process is:
1. \(mg(h+d)+\frac{1}{2}kd^2\)
2. \(mg(h+d)-\frac{1}{2}kd^2\)
3. \(mg(h-d)-\frac{1}{2}kd^2\)
4. \(mg(h-d)+\frac{1}{2}kd^2\)
\(\overrightarrow{A}\) and \(\overrightarrow B\) are two vectors and \(\theta\) is the angle between them. If \(\left|\overrightarrow A\times \overrightarrow B\right|= \sqrt{3}\left(\overrightarrow A\cdot \overrightarrow B\right),\) then the value of \(\theta\) will be:
| 1. | \(60^{\circ}\) | 2. | \(45^{\circ}\) |
| 3. | \(30^{\circ}\) | 4. | \(90^{\circ}\) |
Dimensions of resistance in an electrical circuit, in terms of dimension of mass M, length L, time T, and current I, would be:
1.
2.
3.
4.
A particle moving along the x-axis has acceleration \(f,\) at time \(t,\) given by, \(f=f_0\left ( 1-\frac{t}{T} \right ),\) where \(f_0\) and \(T\) are constants. The particle at \(t=0\) has zero velocity. In the time interval between \(t=0\) and the instant when \(f=0,\) the particle’s velocity \( \left ( v_x \right )\) is:
1. \(f_0T\)
2. \(\frac{1}{2}f_0T^{2}\)
3. \(f_0T^2\)
4. \(\frac{1}{2}f_0T\)