A calorie is defined as the amount of heat required to raise temperature of 1g of water by 1°C and it is defined under which of the following conditions
(1) From 14.5°C to 15.5°C at 760 mm of Hg
(2) From 95.5°C to 99.5°C at 760 mm of Hg
(3) From 13.5°C to 14.5°C at 76 mm of Hg
(4) From 3.5°C to 4.5°C at 760 mm of Hg
A solid substance is at 30°C. To this substance heat energy is supplied at a constant rate. Then the temperature versus time graph is as shown in the figure. The substance is in a liquid state for the portion (of the graph)
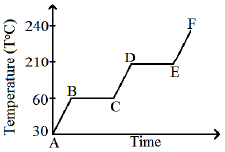
(1) BC
(2) CD
(3) ED
(4) EF
A stationary object at 4°C and weighing 3.5 kg falls from a height of 2000 m on a snow mountain at 0°C. If the temperature of the object just before hitting the snow is 0°C and (the latent heat of ice = 3.5x joule/s), then the mass of ice that will melt is
(1) 2 kg
(2) 200 g
(3) 20 g
(4) 2 g
A substance of mass m kg requires a power input of P watts to remain in the molten state at its melting point. When that power is turned off, the sample completely solidifies in time t sec. What is the latent heat of fusion of the substance
1. Pm/t
2. Pt/m
3. m/Pt
4. t/Pm
80 gm of water at 30°C is poured on a large block of ice at 0°C. The mass of ice that melts
(1) 30 gm
(2) 80 gm
(3) 1600 gm
(4) 150 gm
An experiment took 10 minutes to raise the temperature of water for 0°C to 100°C and another 55 minutes to convert it totally into steam by a stabilized heater. The latent heat of vaporization comes out to be
(1) 530 cal/g
(2) 540 cal/g
(3) 550 cal/g
(4) 560 cal/g
1 of water at its boiling point absorbs 540 calories of heat to become steam with a volume of 1671cm? If the atmospheric pressure = 1.013 x and the mechanical equivalent of heat = 4.19J / calorie, the energy spent in this process in overcoming intermolecular forces is
(1) 540 cal
(2) 40 cal
(3) 500 cal
(4) Zero
Two conducting rods A and B of the same length and cross-sectional area are connected (i) In series (ii) In parallel as shown. In both combinations, a temperature difference of 100°C is maintained. If the thermal conductivity of A is 3K and that of B is K then the ratio of heat current flowing in parallel combination to that flowing in series combination is
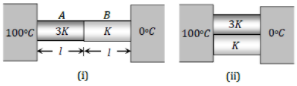
1. 16/3
2. 3/16
3. 1/1
4. 1/3
A black body is heated from 27°C to 127° C. The ratio of their energies of radiations emitted will be
(1) 3:4
(2) 9:16
(3) 27: 64
(4) 81: 256
A body takes T minutes to cool from 62° C to 61°C when the surrounding temperature is 30°C. The time taken by the body to cool from 46°C to 45°C is
(1) Greater than T minutes
(2) Equal to T minutes
(3) Less than T minutes
(4) Equal to T/2 minutes
A partition wall has two layers A and B in contact, each made of a different material. They have the same thickness but the thermal conductivity of layer A is twice that of layer B. If the steady-state temperature difference across the wall is 60K, then the corresponding difference across the layer A is
(1) 10 K
(2) 20K
(3) 30K
(4) 40 K
Water and turpentine oil (specific heat less than that of water) are both heated to the same temperature. Equal amounts of these placed in identical calorimeters are then left in the air
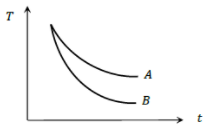
(1) Their cooling curves will be identical
(2) A and B will represent cooling curves of water and oil respectively
(3) B and A will represent cooling curves of water and oil respectively
(4) None of the above
A bimetallic strip is made of aluminum and steel (). On heating, the strip will
(1) Remain straight
(2) Get twisted
(3) Will bend with aluminum on the concave side
(4) Will bend with steel on the concave side
An aluminum sphere is dipped into the water. Which of the following is true?
(1) Buoyancy will be less in the water at 0°C than that in the water at 4°C
(2) Buoyancy will be more in the water at 0°C than that in the water at 4°C
(3) Buoyancy in the water at 0°C will be the same as that in the water at 4°C
(4) Buoyancy may be more or less in the water at 4°C depending on the radius of the sphere
The temperature of a body on the Kelvin scale is found to be \(x^\circ~\text K.\) When it is measured by a Fahrenheit thermometer, it is found to be \(x^\circ~\text F,\) then the value of \(x\) is:
1. \(40\)
2. \(313\)
3. \(574.25\)
4. \(301.25\)
A beaker is completely filled with water at 4°C. it will overflow if
(1) Heated above 4°C
(2) Cooled below 4°C
(3) Both heated and cooled above and below at 4°C respectively
(4) None of the above
If the length of a cylinder on the heating increase by 2%, the area of its base will increase by
(1) 0.5%
(2) 2%
(3) 1%
(4) 4%
The coefficient of apparent expansion of a liquid when determined using two different vessels A and B are respectively. If the coefficient of linear expansion of the vessel A is , the coefficient of linear expansion of the vessel B is
1.
2.
3.
4.
A clock with an iron pendulum keeps correct time at 15° C. what will be the error, in seconds per day, if the room temperature is 20°C? (The coefficient of linear expansion of iron is 0.000012/°C.)
(1) 2.6 s
(2) 6.2 s
(3) 1.3 s
(4) 3.1 s