Which one of the following has the shortest carbon-carbon bond length?
1. Benzene
2. Ethene
3. Ethyne
4. Ethane
What is the correct order of increasing acidic strength among (I) p-methoxyphenol, (II) p-methylphenol, and (III) p-nitrophenol?
1. III < I < II
2. II < I < III
3. III < II < I
4. I < II < III
Which order for basic character of amine is correct for following compounds?
1. 3>1>2>5>4 2. 3>2>1>5>4
3. 3>1>2>4>5 4. 3>2>1>4>5
The most acidic compound among the following is:
| 1. | o-Cresol | 2. | p-Nitrophenol |
| 3. | Phenol | 4. | m-Cresol |
The correct order of basicities of the following compounds is:
(1)
(2)
(3)
(4)
1. 2>1>3>4
2. 1>3>2>4
3. 3>1>2>4
4. 1>2>3>4
In the above compound, arrange the nitrogen according to their decreasing basic strength.
1. 1>2>3>4
2. 4>3>1>2
3. 2>4>1>3
4. 3>4>1>2
Which of the following does not represent the resonating structure of
1.
2.
3.
4.
If 
1. P
2. Q
3. R
4. S
The correct acidic strength order of acidic hydrogen x, y and z is respectively.
(1) x > z > y
(2) x > y > z
(3) z > y > x
(4) y > z > x
The correct order of acid and basic strength for the following pair of compounds should be?
(1) I > II ; III > IV ; V > VI ; VII < VIII
(2) I < II ; III > IV ; V < VI ; VII > VIII
(3) I > II ; III > IV ; V > VI ; VII > VIII
(4) I < II ; III > IV ; V < VI ; VII < VIII
Electrophiles are:
1. electron loving species
2. electron hating species
3. nucleus loving reagents
4. nucleus hating reagents
Who pointed out the concept of hyperconjugation?
(1) Nathan and Baker
(2) Mulliken
(3) Kekule
(4) Kolbe
Which one among the following is most reactive towards nucleophilic addition reaction?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The correct increasing order of basic strength of the given structures is:
\((1) \mathrm{H}_3 \mathrm{C}-\mathrm{CH}_2^{-}~~~~ (2) \mathrm{H}_2 \mathrm{C}=\mathrm{CH}^{-}~~~ (3) H-C \equiv C^{-}\)
| 1. | (2) > (1) > (3) | 2. | (1) > (3) > (2) |
| 3. | (3) > (2) > (1) | 4. | (1) > (2) > (3) |
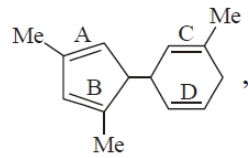
The double bond in the above-mentioned compound that accepts proton fastest is:
| 1. | A | 2. | B |
| 3. | C | 4. | D |
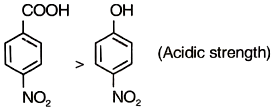
Which of the following order is incorrect regarding the indicated property?
1. 
2. 
3. 
4. 
Which of the following carboxylic acids will be the most acidic?
1.
2.
3.
4.
A: Inductive effect is a permanent effect.
R: group shows +l effect.
1. Both Assertion & Reason are true and the reason is the correct explanation of the assertion.
2. Both Assertion & Reason are true but the reason is not the correct explanation of the assertion
3. Assertion is a true statement but Reason is false.
4. Both Assertion and Reason are false statements.
The aromatic structure(s) out of given structures is/are-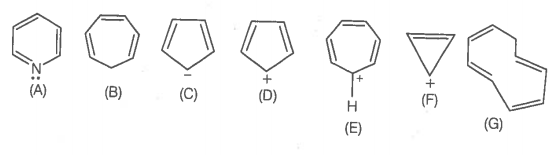
| 1. | A, C, D, F & G only | 2. | A & D only |
| 3. | A, C, E, F only | 4. | All are aromatic |
The Huckel's rule-based aromaticity is shown by:
| (A) |  |
(B) |  |
| (C) |  |
(D) |  |
| (E) |  |
(F) |  |
| 1. | A, B, D only | 2. | B, D only |
| 3. | B, D, E and F only | 4. | A, B, D, E & F only |
Match the ions given in Column I with their nature given in Column II.
| Column I (Ions) |
Column II (Corresponding nature) |
||
| A. |  |
I. | Stable due to resonance |
| B. |  |
II. | Destabilised due to inductive effect |
| C. |  |
III. | Stabilised by hyperconjugation |
| D. |  |
IV. | A secondary carbocation |
Codes:
| Options: | A | B | C | D |
| 1. | I,II | II | II | III,IV |
| 2. | I | II,IV | III | IV |
| 3. | I | IV | III | II |
| 4. | IV | I | III | II |
The species among the following that is not an electrophile is:
1.
2.
3.
4.
Which amongst the following is the most stable carbocation:
| 1. | 2. | ||
| 3. |  |
4. |  |
ion is a-
1. carbocation
2. nucleophile
3. electrophile
4. carboanion
The species present below is
1. nucleophile
2. carbocation
3. electrophile
4. carboanion
The incorrect order regarding the – 𝐼 effect of the substituents among the following is:
| 1. | \(-\mathrm{I}<-\mathrm{Br}<-\mathrm{Cl}<-\mathrm{F}\) |
| 2. | \(-\stackrel{+}{\mathrm{NR}}_3<-\stackrel{+}{\mathrm{OR}}_2\) |
| 3. | \(-\stackrel{+}{\mathrm{NR}}_3<-\mathrm{OR}<-\mathrm{F}\) |
| 4. | \(-\mathrm{SR}<-\mathrm{OR}<-\stackrel{+}{\mathrm{OR}}_2\) |
The strongest ortho/para directing group is:
1. −NH2
2. −CH3
3. −Cl
4. −C2H5
| 1. |  |
2. |  |
| 3. |  |
4. |  |

| 1. | 12 | 2. | 13 |
| 3. | 15 | 4. | 16 |
| Statement I: | Cycloheptatrienyl cation is an aromatic species. |
| Statement II: | Cycloheptatrienyl cation contains \(6 \pi\) electrons in conjugation. |
| 1. | Statement I is incorrect and Statement II is correct. |
| 2. | Both Statement I and Statement II are correct. |
| 3. | Both Statement I and Statement II are incorrect. |
| 4. | Statement I is correct and Statement II is incorrect. |
| Statement I: | Inductive effect and resonance are permanent effects. |
| Statement II: | The energy of the actual structure of the molecule (the resonance hybrid) is higher than that of any of the canonical structures. |
| 1. | Statement I is correct, but Statement II is incorrect. |
| 2. | Both Statement I and Statement II are correct. |
| 3. | Both Statement I and Statement II are incorrect. |
| 4. | Statement I is incorrect, but Statement II is correct. |

| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |

| 1. | I > III > II | 2. | II > I > III |
| 3. | I > II > III | 4. | III > II > I |


| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. |  |
2. |  |
| 3. |  |
4. |  |
