Motile zygote of Plasmodium occurs in:
| 1. | Gut of female Anopheles |
| 2. | Salivary glands of Anopheles |
| 3. | Human RBCs |
| 4. | Human liver |
The human hindbrain comprises three parts, one of which is:
| 1. | Cerebellum | 2. | Hypothalamus |
| 3. | Spinal | 4. | Corpus callosum |
Which part of the human ear plays no role in hearing as such but is otherwise very much required?
1. Eustachian tube
2. Organ of Corti
3. Vestibular apparatus
4. Ear ossicles
The most abundant prokaryotes helpful to human in making curd from milk and in production of antibiotics are the ones categorized as:
| 1. | Cyanobacteria | 2. | Archaebacteria |
| 3. | Chemosynthetic autotrophs | 4. | Heterotrophic bacteria |
Which one of the following statements is false in respect of viability of mammalian sperm?
| 1. | Sperm is viable for only up to 24 hours |
| 2. | Survival of sperm depends on the pH of the medium and is more active in alkaline medium |
| 3. | Viability of sperm is determined by its motility |
| 4. | Sperms must be concentrated in a thick suspension |
Evolution of different species in a given area starting from a point and spreading to other geographical areas is known as:
| 1. | Adaptive radiation | 2. | Natural selection |
| 3. | Migration | 4. | Divergent evolution |
What is the figure given below showing in particular?

| 1. | Ovarian cancer | 2. | Uterine cancer |
| 3. | Tubectomy | 4. | Vasectomy |
In an area where DDT had been used extensively, the population of birds declined significantly because:
1. Birds stopped laying eggs
2. Earthworms in the area got eradicated
3. Cobras were feeding exclusively on birds
4. Many of the birds laid eggs, but the eggs did not hatch

The common bottle cork is a product of:
1. dermatogen
2. phellogen
3. xylem
4. vascular cambium
Widal test is carried out to test:
| 1. | Malaria | 2. | Diabetes mellitus |
| 3. | HIV/AIDS | 4. | typhoid fever |
Which part would be most suitable for raising virus-free plants for micro propagation?
1. Bark
2. Vascular tissue
3. Meristem
4. Node
Which one of the following is a wrong statement?
| 1. | Most of the forests have been lost in tropical areas |
| 2. | Ozone in upper part of atmosphere is harmful to animals |
| 3. | Greenhouse effect is a natural phenomenon |
| 4. | Eutrophication is a natural phenomenon in freshwater bodies |
Companion cells are closely associated with:
| 1. | Sieve elements | 2. | Vessel elements |
| 3. | Trichomes | 4. | Guard cells |
Common cold differs from pneumonia in, that:
| 1. | Pneumonia is a communicable disease, whereas common cold is a nutritional deficiency disease |
| 2. | Pneumonia can be prevented by a live attenuated bacterial vaccine, whereas the common cold has no effective vaccine |
| 3. | Common cold is caused by a virus, while Pneumonia is caused by a bacterium. |
| 4. | Pneumonia pathogen infects alveoli whereas common cold affects the nose and the respiratory passage but not the lungs |
Pheretima and its close relatives derive nourishment from:
1. Sugarcane roots
2. Decaying fallen leaves and soil organic matter
3. Soil insects
4. Small pieces of fresh fallen leaves of maize
Removal of RNA polymerase III from nucleoplasm will affect the synthesis of:
1. tRNA
2. hnRNA
3. mRNA
4. rRNA
A process that makes an important difference between C3 and C4 plants is:
1. Transpiration
2. Glycolysis
3. Photosynthesis
4. Photorespiration
PCR and Restriction Fragment Length Polymorphism are the methods for:
1. Study of enzymes
2. Genetic transformation
3. DNA sequencing
4. Genetic fingerprinting
Best defined function of manganese in green plants is :
1. Photolysis of water
2. Calvin cycle
3. Nitrogen fixation
4. Water absorption
Measuring Biochemical Oxygen Demand (BOD) is a method used for:
| 1. | Estimating the amount of organic matter in sewage water |
| 2. | Working out the efficiency of oil-driven automobile engines |
| 3. | Measuring the activity of Saccharomyces cerevisiae in producing curd on a commercial scale |
| 4. | Working out the efficiency of RBCs about their capacity to carry oxygen |
Which one of the following is not part of a transcription unit in DNA?
1. The inducer
2. A Terminator
3. A promoter
4. The structural gene
A certain road accident patient with an unknown blood group needs an immediate blood transfusion. His one doctor friend at once offers his blood. What was the blood group of the donor?
| 1. | Blood group B | 2. | Blood group AB |
| 3. | Blood group O | 4. | Blood group A |
Consumption of which one of the following foods can prevent the kind of blindness associated with vitamin-A deficiency?
| 1. | Flaver savr tomato | 2. | Canolla |
| 3. | Golden rice | 4. | Bt-brinjal |
The maximum amount of electrolytes and water (70-80%) from the glomerular filtrate is reabsorbed in which part of the nephron?
1. Ascending limb of the loop of Henle
2. Distal convoluted tubule
3. Proximal convoluted tubule
4. Descending limb of the loop of Henle
Both, autogamy and geitonogamy are prevented in:
1. Papaya
2. Cucumber
3. Castor
4. Maize
Placentation in tomato and lemon is:
| 1. | Parietal | 2. | free central |
| 3. | marginal | 4. | axile |
A person entering an empty room suddenly finds a snake right in front on opening the door. Which one of the following is likely to happen in his neurohormonal control system?
| 1. | Sympathetic nervous system activated releasing epinephrine and norepinephrine from adrenal medulla |
| 2. | Neurotransmitters diffuse rapidly across the cleft and transmit a nerve impulse |
| 3. | Hypothalamus activates the parasympathetic division of the brain |
| 4. | Sympathetic nervous system is activated releasing epinephrine and norepinephrine from the adrenal cortex |
Which one of the following is not a gaseous biogeochemical cycle in the ecosystem?
1. Sulphur cycle
2. Phosphorus cycle
3. Nitrogen cycle
4. Carbon cycle
Which one of the following options gives one correct example of convergent evolution and divergent evolution?
| Convergent Evolution | Divergent Evolution | |
| 1. | Eyes of octopus and mammals | Bones of forelimbs of vertebrates |
| 2. | Thorns of Bougainvillea and tendrils Cucurbita | Wings of butterflies and birds |
| 3. | Bones of forelimbs of vertebrates | Wings of butterfly and birds |
| 4. | Thorns of Bougainvillea and tendrils of Cucurbita | Eyes of Octopus and mammals |
Cycas and Adiantum resemble each other in having:
1. Seeds
2. Motile sperms
3. Cambium
4. Vessels
What was the most significant trend in the evolution of modem man (Homo sapiens) from his ancestors?
1. Shortening of jaws
2. Binocular vision
3. Increasing cranial capacity
4. upright posture
Cymose inflorescence is present in:
1. Solanum
2. Sesbania
3. Trifolium
4. Brassica
Ribosomal RNA is actively synthesized in:
1. Lysosomes
2. Nucleolus
3. Nucleoplasm
4. Ribosomes
During gamete formation, the enzyme recombinase participates during:
1. Metaphase-I
2. Anaphase-II
3. Prophase-I
4. Prophase-II
Identify the possible link A in the following food chain.
Green plantInsectFrogAEagle
1. Rabbit
2. Wolf
3. Cobra
4. Parrot
Phyllode is present in:
| 1. | Asparagus | 2. | Euphorbia |
| 3. | Australian Acacia | 4. | Opuntia |
Given below is an imaginary pyramid of numbers. What could be one of the possibilities about certain organisms at some of the different levels?
| 1. | Level PC is insects and level SC is small insectivorous birds |
| 2. | Level PP is phytoplanktons in the sea and Whale on top-level TC |
| 3. | Level PP is pipal trees and the level SC is sheep |
| 4. | Level PC is rats and level SC is cats |
Monascus purpureus is the yeast used commercially in the production of:
| 1. | Ethanol |
| 2. | Streptokinase for removing clots from the blood vessels |
| 3. | Citric acid |
| 4. | Blood cholesterol-lowering statins |
The correct sequence of cell organelles during photorespiration is:
1. chloroplast-Golgi bodies-mitochondria
2. chloroplast-rough endoplasmic reticulum-dictyosomes
3. chloroplast-mitochondria-peroxisome
4. chloroplast-vacuole-peroxisome
Which one of the following is correctly matched?
1. Passive transport of nutrients-ATP
2. Apoplast- Plasmodesmata
3. Potassium- Readily mobilized
4. Bakanae of rice seedlings-F Skoog
A normal-visioned man whose father was color blind marries a woman whose father was also colorblind. They have their first child as a daughter. What are the chances that this child would be colorblind?
1. 100%
2. 0%
3. 25%
4. 50%
Signals for parturition originate from:
1. Both placenta as well as fully developed foetus
2. Oxytocin released from maternal pituitary
3. Placenta only
4. Fully developed foetus only
A patient brought to a hospital with myocardial infarction is normally immediately given:
| 1. | penicillin | 2. | streptokinase |
| 3. | cyclosporin-A | 4. | statins |
Which one of the following is not a property of cancerous cells, whereas the remaining three are?
| 1. | They compete with normal cells for vital nutrients |
| 2. | They do not remain confined in the area of formation |
| 3. | They divide in an uncontrolled manner |
| 4. | They show contact inhibition |
The gynoecium consists of many free pistils in flowers of:
| 1. | Aloe | 2. | Tomato |
| 3. | Papaver | 4. | Michelia |
Which one of the following is not a functional unit of an ecosystem?
1. Energy flow
2. Decomposition
3. Productivity
4. Stratification
In a normal pregnant woman, the amount of total gonadotropin activity was assessed. The result expected was:
| 1. | High level of circulating FSH and LH in the uterus to stimulate implantation of the embryo |
| 2. | High level of circulating HCG to stimulate endometrial thickening |
| 3. | High levels of FSH and LH in uterus to stimulate endometrial thickening |
| 4. | High level of circulating HCG to stimulate oestrogen and progesterone synthesis |
Which one of the following areas in India is a hot spot of biodiversity?
1. Eastern Ghats
2. Gangetic plain
3. Sunder bans
4. Western Ghats
Which one of the following is the correct statement?
| 1. | Pteridophyte gametophyte has a protonemal and leafy stage |
| 2. | In gymnosperms, female gametophyte is free-living |
| 3. | Antheridiophores and protonema are present in pteridophytes |
| 4. | Origin of seed habit can be traced in pteridophytes |
Which one of the following does not differ in E.coli and Chlamydomonas?
1. Ribosomes
2. Chromosomal organization
3. Cell wall
4. Cell membrane
The cyanobacteria are also referred to as:
1. Protists
2. golden algae
3. Slime molds
4. Blue-green algae
The test-tube baby programme employs which one of the following techniques?
| 1. | Intra Cytoplasmic Sperm Injection (ICSI) |
| 2. | Intra Uterine Insemination (IUI) |
| 3. | Gamete Intra Fallopian Transfer (GIFT) |
| 4. | Zygote Intra Fallopian Transfer(ZIFT) |
Which one of the following is a case of wrong matching?
| 1. | Somatic hybridization- Fusion of two diverse cells |
| 2. | Vector DNA- Site for tRNA synthesis |
| 3. | Micropropagation- In vitro production of plants in large numbers |
| 4. | Callus - Unorganized mass of cells produced in tissue culture |
The highest number of species in the world is represented by:
| 1. | Fungi | 2. | Mosses |
| 3. | Algae | 4. | Lichens |
In which one of the following options the two examples are correctly matched with their particular type of immunity?
| Examples | Type of immunity |
|
| (a) | Polymorphonuclear leukocytes and monocytes | Cellular barriers |
| (b) | Anti-tetanus and anti-snake bite injection | Active immunity |
| (c) | Saliva in mouth and tears in eyes | Physical barriers |
| (d) | Mucus coating of the epithelium lining the urinogenital tract and HCl in the stomach | Physiological barriers |
Which one of the following is a wrong statement?
| 1. | Anabaena and Nostoc are capable of fixing nitrogen in a free-living state also |
| 2. | Root nodule-forming nitrogen fixers live as aerobes under free-living conditions |
| 3. | Phosphorus is a constituent of cell membranes, certain nucleic acids, and all proteins |
| 4. | Nitrosomonas and Nitrobacter are chemoautotrophs |
Anxiety and eating spicy food together in an otherwise normal human, may lead to:
1. Indigestion
2. Jaundice
3. Diarrhoea
4. vomiting
The Leydig cells as found in the human body are the secretory source of:
1. Progesterone
2. Intestinal mucus
3. Glocagon
4. Androgens
Compared to those of humans, the erythrocytes in frogs are:
1. Without nucleus but with haemoglobin
2. Nucleated and with haemoglobin
3. Very much smaller and fewer
4. Nucleated and without haemoglobin
In which one of the following, the genus name, its two characters and its phylum are not correctly matched, whereas the remaining three are correct?
| Genus Name | Two Characters | Phylum | ||
| 1. | Pila | (i) (ii) |
Body segmented Mouth with Radula |
Mollusca |
| 2. | Asterias | (i) (ii) |
Spiny skinned Water Vascular System |
Echinodermata |
| 3. | Sycon | (i) (ii) |
Pore bearing Canal system |
Porifera |
| 4. | Periplaneta | (i) (ii) |
Jointed appendages Chitinous exoskeleton |
Arthropoda |
What is true about ribosomes?
| 1. | The prokaryotic ribosomes are 80S, where S stands for sedimentation coefficient |
| 2. | These are composed of ribonucleic acid and proteins |
| 3. | These are found only in eukaryotic cells |
| 4. | These are self-splicing introns of some RNAs |
Cirrhosis of the liver is caused by the chronic intake of:
1. Opium
2. Alcohol
3. Tobacco (chewing)
4. Cocaine
Which one is a true statement regarding DNA polymerase used in PCR?
| 1. | It is used to ligate introduces DNA in recipient cells |
| 2. | It serves as a selectable marker |
| 3. | It is isolated from a virus |
| 4. | It remains active at high temperatures |
Which statement is wrong for viruses?
| 1. | All are parasites |
| 2. | All of them have helical symmetry |
| 3. | They have the ability to synthesize nucleic acids and proteins |
| 4. | Antibiotics have no effect on them |
Which one of the following is correctly matched?
1. Onion - Bulb
2. Ginger - Sucker
3. Chlamydomonas - Conidia
4. Yeast - Zoospores
How many plants in the list given below have composite fruits that develop from an inflorescence?
Walnut, poppy, radish, fig, pineapple, apple, tomato, mulberry
| 1. | Four | 2. | Five |
| 3. | Two | 4. | Three |
Given below is the diagrammatic representation of one of the categories of small molecular weight organic compounds in the living tissues. Identify the category shown and the one blank component X in it:
| 1. | Category: Cholesterol; Component: Guanine |
| 2. | Category: Amino acid; Component: NH2 |
| 3. | Category: Nucleotide; Component: Adenine |
| 4. | Category: Nucleoside; Component: Uracil |
Which one of the following microbes forms a symbiotic association with plants and helps them in their nutrition?
1. Azotobacter
2. Aspergillus
3. Glomus
4. Trichoderma
The extinct human who lived 100000 to 40000 years ago, in Europe, Asia, and parts of Africa, with short stature, heavy eyebrows, retreating for heads, large jaws with heavy teeth, stocky bodies, a lumbering gait, and stooped posture was:
1. Homo habilis
2. Neanderthal human
3. Cro-Magnon humans
4. Ramapithecus
If one strand of DNA has the nitrogenous base sequence as ATCTG, what would be the complementary RNA strand sequence?
1. TTAGU
2. UAGAC
3. AACTG
4. ATCGU
Which one of the following pairs of hormones are the examples of those that can easily pass through the cell membrane of the target cell and bind to a receptor inside it (mostly in the nucleus)?
1. Insulin and glucagon
2. Thyroxin and insulin
3. Somatosatin and oxytocin
4. Cortisol and testosterone
Nuclear membrane is absent in:
1. Penicillium
2. Agaricus
3. Volvox
4. Nostoc
Which one is the most abundant protein in the animal world?
| 1. | Trypsin | 2. | Haemoglobin |
| 3. | Collagen | 4. | Insulin |
Which one of the following is common to multicellular fungi, filamentous algae and protonema of mosses?
1. Diplontic life cycle
2. Members of kingdom-Plantae
3. Mode of nutrition
4. Multiplication by fragmentation
Which one single organism or the pair of organisms is correctly assigned to its or their named taxonomic group?
| 1. | Paramecium and Plasmodium belong to the same kingdom as that of Penicillium |
| 2. | Lichen is a composite organism formed from the symbiotic association of algae and a protozoan |
| 3. | Yeast is used in making bread and brew is a fungus |
| 4. | Nostoc and Anabaena are examples of Protista |
Even in the absence of pollinating agents, seed-setting is assured in:
1. Commelina
2. Zostera
3. Salvia
4. Fig
Yeast is used in the production of:
1. Citric acid and lactic acid
2. Lipase and pectinase
3. Bread and beer
4. Cheese and butter
Which one out of A-D given below correctly represents the structural formula of the basic amino acid?
| A | B |
.svg) |
.svg) |
| C | D |
.svg) |
.svg) |
| 1. | C | 2. | D |
| 3. | A | 4. | B |
The upright pyramid of number is absent in:
1. Pond
2. Forest
3. Lake
4. Grassland
What is correct to say about the hormone action in humans?
| 1. | Glucagon is secreted by β-cells of the islet of Langerhans and stimulates glycogenolysis |
| 2. | Secretion of thymosin is stimulates with ageing |
| 3. | In females, FSH first binds with specific receptors on ovarian cell membrane |
| 4. | FSH stimulates the secretion of oestrogen and progesterone |
Closed vascular bundles lack:
1. Ground tissue
2. Conjuctive tissue
3. Cambium
4. Pith
Which one of the following is the correct statement for respiration in humans?
| 1. | Cigarette smoking may lead to inflammation of the bronchi |
| 2. | Neural signals from the pneumotaxic centre in the pons region of the brain can increase the duration of inspiration |
| 3. | Workers in grinding and stone-breaking industries may suffer from lung fibrosis |
| 4. | About 90% of carbon dioxide (CO2) is carried by haemoglobin as carbaminohaemoglobin |
Removal of introns and joining of exons in a defined order during transcription is called:
1. Looping
2. Inducing
3. Slicing
4. Splicing
F2 generation in a Mendelian cross showed that both genotypic and phenotypic ratios are same as 1: 2: 1. It represents a case of:
1. Co-dominance
2. Dihybrid crosses
3. Monohybrid crosses with complete dominance
4. Monohybrid crosses with incomplete dominance
Given below is the representation of a certain event at a particular stage of a type of cell division. Which is this stage?
1. Prophase-I during meiosis
2. Prophase-II during meiosis
3. Prophase of mitosis
4. Both prophase and metaphase of mitosis
People who have migrated from the planes to an area adjoining Rohtang Pass about six months back:
| 1. | Have more RBCs and their haemoglobin has a lower binding affinity to 02 |
| 2. | Are not physically fit to play games like football |
| 3. | Suffer from altitude sickness with symptoms like nausea, fatigue, etc. |
| 4. | have the usual RBC count but their haemoglobin has a very binding affinity to 02 |
For transformation, microparticles coated with DNA to be bombarded with gene gun are made up of:
1. Silver or platinum
2. Platinum or zinc
3. Silicon or platinum
4. Gold or tungsten
A nitrogen-fixing microbe associated with Azolla in rice fields is:
1. Spirulina
2. Anabaena
3. Frankia
4. Tolypothrix
Select the correct statement regarding the specific disorder of muscular or skeletal system.
| 1. | Muscular dystrophy - age-related shortening of muscles |
| 2. | Osteoporosis- decrease in bone mass and higher chances of fractures with advancing age |
| 3. | Myasthenia gravis- an autoimmune disorder which inhibits the sliding of myosin filaments |
| 4. | Gout- inflammation of joints due to extra deposition of calcium |
Water containing cavities in vascular bundles are found in:
1. Sunflower
2. Maize
3. Cycas
4. Pinus
Select the correct statement from the following regarding cell membrane:
| 1. | Na+ and K+ ions move across cell membranes by passive transport |
| 2. | Proteins make up 60% to 70% of the cell membrane |
| 3. | Lipids are arranged in a bilayer with polar heads towards the inner part |
| 4. | Fluid mosaic model of the cell membrane was proposed by Singer and Nicolson |
Gymnosperms are also called softwood spermatophytes because they lack:
1. Cambium
2. Phloem fibres
3. Thick-walled tracheids
4. Xylem fibres
The coconut water and the edible part of coconut are equivalent to:
1. Endosperm
2. Endocarp
3. Mesocarp
4. Embryo
Vexillary aestivation is characteristic of the family:
1. Fabaceae
2. Asteraceae
3. Solanaceae
4. Brassicaceae
Which one of the following is an example of carrying out biological control of pests/diseases using microbes?
1. Trichoderma sp. against certain plant pathogens
2. Nucleopolyhedrovirus against white rust in Brassica
3. Bt-cotton to increase cotton yield
4. Ladybird beetle against aphids in mustard
Select the correct statement from the ones given below with respect to Periplaneta americana.
| 1. | Nervous system located dorsally, consists of segmentally arranged ganglia joined by a pair of longitudinal connectives |
| 2. | Males bear a pair of short thread-like anal styles |
| 3. | There are 16 very long Malpighian tubules present at the junctions of midgut and hindgut |
| 4. | Grinding of food is carried out only by the mouth parts |
Maximum nutritional diversity is found in the group:
1. Fungi
2. Animalia
3. Monera
4. Plantae
Aluminium is extracted from alumina Al2O3 by electrolysis of a molten mixture of:
1.
2.
3.
4.
pH of a saturated solution of Ba(OH)2 is 12. The value of solubility product Ksp of Ba (OH)2 is:
1.
2.
3.
4.
The change in oxidation number of chlorine when Cl2 gas reacts with hot and concentrated sodium hydroxide solution is:
1. Zero to +1 and Zero to –5
2. Zero to –1 and Zero to +5
3. Zero to –1 and Zero to +3
4. Zero to +1 and Zero to –3
Maximum number of electrons in a subshell with l = 3 and n = 4 is:
1. 14
2. 16
3. 10
4. 12
Which one of the following is an outer orbital complex and exhibits paramagnetic behavior?
1.
2.
3.
4.
In a reaction, A + B → Product, the rate is doubled when the concentration of B is doubled, and the rate increases by a factor of 8, when the concentrations of both the reactants (A and B) are doubled. The rate law for the reaction can be written as:
1. Rate = k[A][B]2
2. Rate = k[A]2[B]2
3. Rate = k[A][B]
4. Rate = k[A]2[B]
In which of the following reactions, the standard reaction entropy change
is positive, and standard Gibb's energy change
decreases sharply with increasing temperature?
| 1. | C(graphite) + \(\frac{1}{2}\)O2(g) → CO(g) |
| 2. | CO(g) + \(\frac{1}{2}\)O2(g) → CO2(g) |
| 3. | Mg(s) + \(\frac{1}{2}\)O2(g) → MgO(s) |
| 4. | \(\frac{1}{2}\)C(graphite) + \(\frac{1}{2}\)O2(g) → \(\frac{1}{2}\)CO2(g) |
Which one of the following is a mineral of iron?
1. Malachite
2. Cassiterite
3. Pyrolusite
4. Magnetite
In the Freundlich adsorption isotherm, the value of 1/n is:
1. Between 0 and 1 in all cases.
2. Between 2 and 4 in all cases.
3. 1 in the case of physical adsorption.
4. 1 in the case of chemisorption.
Equimolar solutions of the following substances were prepared separately. Which one of these will record the highest pH value?
| 1. | \(\mathrm{B a C l_{2}}\) | 2. | \(\mathrm{A l C l_{3}}\) |
| 3. | \(\mathrm{L i C l}\) | 4. | \(\mathrm{B e C l_{2}}\) |
50 mL of each gas A and of gas B takes 200 and 150 s respectively for effusing through a pinhole under the similar conditions. If the molecular mass of gas B is 36, the molecular mass of gas A will be:
1. 96
2. 128
3. 32
4. 64
The correct set of four quantum numbers for the valence electron of a rubidium atom (Z =37) is:
| 1. | \(5,1,1,+ {1 \over2}\) | 2. | \(6,0,0,+ {1 \over2}\) |
| 3. | \(5,0,0,+ {1 \over2}\) | 4. | \(5,1,0,+ {1 \over2}\) |
The highest oxidation number of nitrogen among the following compounds is:
| 1. | 2. | ||
| 3. | 4. |
Predict the products in the given reaction,
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Acetone is treated with excess ethanol in the presence of hydrochloric acid. The product obtained will be:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
A metal crystallizes with a fcc lattice. The edge of the unit cell is 408 pm. The diameter of the metal atom is:
1. 288 pm
2. 408 pm
3. 144 pm
4. 204 pm
Which one of the following statements about enzyme catalysis is incorrect?
| 1. | Enzymes are mostly proteinous in nature. |
| 2. | Enzyme action is specific. |
| 3. | Enzymes are denaturized by ultraviolet rays at high temperatures. |
| 4. | Enzymes are least reactive at an optimum temperature. |
In a zero-order reaction for every 10 °C rise of temperature, the rate is doubled.
If the temperature is increased from 10 °C to 100 °C, the rate of the reaction will become:
1. 256 times
2. 512 times
3. 64 times
4. 128 times
The disease caused by deficiency of vitamin B1 is:
| 1. | Convulsions | 2. | Beri-beri |
| 3. | Cheilosis | 4. | Sterility |
Among the following compounds the one that is most reactive towards electrophilic nitration is:
1. Benzoic acid
2. Nitrobenzene
3. Toluene
4. Benzene
Buffer solutions have constant acidity and alkalinity because:
| 1. | these give unionized acid or base on reaction with added acid or alkali. |
| 2. | acid and alkali in these solutions are shielded from attack by other ions |
| 3. | they have large excess of H+ or OH- ions |
| 4. | they have fixed value of pH |
The correct order of decreasing acid strength of
trichloroacetic acid (A), trifluoroacetic acid (B), acetic acid (C), and formic acid (D) is:
1. B > A > D > C
2. B > D > C > A
3. A > B > C > D
4. A > C > B >D
Sucrose can be formed by:
1. α–D–galactopyranose and α–D–glucopyranose
2. α–D–glucopyranose and β–D–fructofuranose
3. β–D–galactopyranose and α–D–fructofuranose
4. α–D–galactopyranose and β–D–fructopyranose
The enthalpy of fusion of water is 1.435 kcal/mol. The molar entropy change for the melting of ice at 0 oC is:
| 1. | 10.52 cal/(mol K) | 2. | 21.04 cal/(mol K) |
| 3. | 5.260 cal/(mol K) | 4. | 0.526 cal/(mol K) |
Isostructural pair among the following is:
1.
2.
3.
4.
The bond order of 1.5 is shown by:
| 1. | \(\text O^+_2\) | 2. | \(\text O^-_2\) |
| 3. | \(\text O^{2-}_2\) | 4. | \(\text O_2\) |
Which one of the following is not a condensation polymer?
1. Melamine
2. Glyptal
3. Dacron
4. Neoprene
The end product (C) in the below-mentioned reaction is:
\(H_3C-Br\xrightarrow{\bf KCN }A\xrightarrow{ \bf H_3O^+~~}B\xrightarrow[\bf\text{Ether}]{~~LiAlH_4~~}C\)
The incorrect IUPAC name among the following is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The number of octahedral void(s) per atom present in a cubic close-packed structure is:
1. 1
2. 3
3. 2
4. 4
In the extraction of copper from its sulphide ore, the metal is finally obtained by the reduction of cuprous oxide with:
1. Copper(l) sulphide (Cu2S)
2. Sulphur dioxide (SO2)
3. Iron Sulphide (FeS)
4. Carbon monoxide (CO)
An interstitial compound among the following is:
1. Invar
2. Steel
3. Bell metal
4. Bronze
A mixture of potassium chlorate, oxalic acid and sulphuric acid is heated. During the reaction which element undergoes maximum change in the oxidation number?Redox Reactions
1. S
2. H
3. Cl
4. C
Which one of the following alkali metals forms only the normal oxide M2O on heating in air?
1. Rb
2. K
3. Li
4. Na
The ease of adsorption of the hydrated alkali metal ions on ion-exchange resins follows the order:
1.
2.
3.
4.
The incorrect statement among the following regarding photochemical smog is:
| 1. | Carbon monoxide does not play any role in photochemical smog formation. |
| 2. | Photochemical smog is an oxidising agent in character. |
| 3. | Photochemical smog is formed through a photochemical reaction involving solar energy. |
| 4. | Photochemical smog does not cause irritation in the eyes and throat. |
CH3CHO and C6H5CH2CHO can be distinguished by:
1. Benedict test
2. Iodoform test
3. Tollen's reagent test
4. Fehling solution test
Which of the following statements is false?
| 1. | On passing H2S through acidified K2Cr2O7 solution, a milky colour is observed. |
| 2. | Na2Cr2O7 is preferred over K2Cr2O7 in volumetric analysis. |
| 3. | K2Cr2O7 solution in an acidic medium is orange. |
| 4. | K2Cr2O7 solution becomes yellow in increasing the pH beyond 7 |
The standard enthalpy of vaporization for water at 100 oC is 40.66 kJ mol–1. The internal energy of vaporization of water at 100 oC (in kJ mol–1) is:
(Assume water vapour behaves like an ideal gas.)
| 1. | +37.56 | 2. | –43.76 |
| 3. | +43.76 | 4. | +40.66 |
Identify the incorrect statement among the following:
| 1. | Amongst isoelectronic species, the smaller the positive charge on the cation, the smaller is the ionic radius |
| 2. | Amongst isoelectronic species, the greater the negative charge on the anion, the larger is ionic radius |
| 3. | Atomic radius of the elements increases as one moves down the first group of the periodic table |
| 4. | Atomic radius of the elements decreases as one moves across from left to right in the 2nd period of the periodic table |
The incorrect statement regarding oxoacids of phosphorus is:
| 1. | Orthophosphoric acid is used in the manufacture of triple superphosphate. |
| 2. | Hypophosphorous acid is a diprotic acid. |
| 3. | All oxoacids contain tetrahedral four coordinated phosphorus. |
| 4. | All oxoacids contain at least one P=O unit and one P-OH group. |
The protecting power of lyophilic colloidal sol is expressed in terms of:
1. Coagulation value
2. Gold number
3. Critical micelle concentration
4. Oxidation number
Sulphur trioxide can be obtained by which of the following reaction:
1.
2.
3.
4.
pA and pB are the vapour pressure of pure liquid components, A and B, respectively of an ideal binary solution.
If XA represents the mole fraction of component A, the total pressure of the solution will be:
| 1. | pA + XA (pB-pA) | 2. | pA + XA (pA-pB) |
| 3. | pB + XA (pB-pA) | 4. | pB + XA (pA-pB) |
1. Maleic acid
2. -amino acid
3. Lactic acid
4. Tartaric acid
Which of the following species contains three bond pairs and one lone pair around the central atom?
1. \(\text H_{2} \text O\)
2. \(\text{BF}_{3}\)
3. \(\text{NH}^-_2\)
4. \(\text{PCl}_{3}\)
Limiting molar conductivity of NH4OH (i.e., is equal to -
1.
2.
3.
4.
The pair of species with the same bond order is:
1.
2.
3.
4.
Which of the following statement is false?
1.Artificial silk is derived from cellulose
2. Nylon-66 is an example of elastomer
3. The repeat unit in natural rubber is isoprene
4. Both starch and cellulose are polymers of glucose
| 1. |  |
2. |  |
| 3. |  |
4. |  |
When a string is divided into three segments of lengths \(l_1\), \(l_2\) and \(l_3\), the fundamental frequencies of these three segments are \(\nu_1\), \(\nu_2\) and \(\nu_3\) respectively. The original fundamental frequency (\(\nu\)) of the string is:
| 1. | \(\sqrt{\nu} = \sqrt{\nu_1}+\sqrt{\nu_2}+\sqrt{\nu_3}\) |
| 2. | \(\nu = \nu_1+\nu_2+\nu_3\) |
| 3. | \(\dfrac{1}{\nu} =\dfrac{1}{\nu_1} +\dfrac{1}{\nu_2}+\dfrac{1}{\nu_3}\) |
| 4. | \(\dfrac{1}{\sqrt{\nu}} =\dfrac{1}{\sqrt{\nu_1}} +\dfrac{1}{\sqrt{\nu_2}}+\dfrac{1}{\sqrt{\nu_3}}\) |
A \(200~\text{W}\) sodium street lamp emits yellow light of wavelength \(0.6~\mu\text{m}.\) Assuming it to be \(25\%\) efficient in converting electrical energy to light, the number of photons of yellow light it emits per second is:
1. \(1.5\times 10^{20}\)
2. \(6\times 10^{18}\)
3. \(62\times 10^{20}\)
4. \(3\times 10^{19}\)
Two ideal diodes are connected to a battery as shown in the circuit. The current supplied by the battery is:
| 1. | \(0.75~\text{A}\) | 2. | zero |
| 3. | \(0.25~\text{A}\) | 4. | \(0.5~\text{A}\) |
When a mass is rotating in a plane about a fixed point, its angular momentum is directed along:
| 1. | a line perpendicular to the plane of rotation |
| 2. | the line making an angle of \(45^{\circ}\) to the plane of rotation |
| 3. | the radius |
| 4. | the tangent to the orbit |
An electric dipole of moment \(p\) is placed in an electric field of intensity \(E.\) The dipole acquires a position such that the axis of the dipole makes an angle \(\theta\) with the direction of the field. Assuming that the potential energy of the dipole to be zero when \(\theta = 90^{\circ}\), the torque and the potential energy of the dipole will respectively be:
1. \(pE\text{sin}\theta, ~-pE\text{cos}\theta\)
2. \(pE\text{sin}\theta, ~-2pE\text{cos}\theta\)
3. \(pE\text{sin}\theta, ~2pE\text{cos}\theta\)
4. \(pE\text{cos}\theta, ~-pE\text{sin}\theta\)
In a CE transistor amplifier, the audio signal voltage across the resistance of 2 k is 2V. If the base resistance is 1 k and the current amplification of the transistor is 100, the input signal voltage is:
1. 0.1 V
2. 1.0 V
3. 1 mV
4. 10 mV
A coil of resistance \(400~\Omega\) is placed in a magnetic field. The magnetic flux \(\phi~\text{(Wb)}\) linked with the coil varies with time \(t~\text{(s)}\) as \(\phi=50t^{2}+4.\) The current in the coil at \(t=2~\text{s}\) is:
| 1. | \(0.5~\text{A}\) | 2. | \(0.1~\text{A}\) |
| 3. | \(2~\text{A}\) | 4. | \(1~\text{A}\) |

| 1. | \(2 {PV}\) | 2. | \(4{PV}\) |
| 3. | \(\frac{1}{2}{PV}\) | 4. | \(PV\) |
If the nuclear radius of \(^{27}\text{Al}\) is \(3.6\) Fermi, the approximate nuclear radius of \(^{64}\text{Cu}\) in Fermi is:
1. \(2.4\)
2. \(1.2\)
3. \(4.8\)
4. \(3.6\)
Two similar coils of radius \(R\) are lying concentrically with their planes at right angles to each other. The currents flowing in them are \(I\) and \(2I,\) respectively. What will be the resultant magnetic field induction at the centre?
| 1. | \(\sqrt{5} \mu_0I \over 2R\) | 2. | \({3} \mu_0I \over 2R\) |
| 3. | \( \mu_0I \over 2R\) | 4. | \( \mu_0I \over R\) |
| 1. | \(\dfrac{B}{A}\) | 2. | \(\dfrac{B}{2A}\) |
| 3. | \(\dfrac{2A}{B}\) | 4. | \(\dfrac{A}{B}\) |
When a biconvex lens of glass having a refractive index of \(1.47\) is dipped in a liquid, it acts as a plane sheet of glass. The liquid must have a refractive index:
| 1. | equal to that of glass. |
| 2. | less than one. |
| 3. | greater than that of glass. |
| 4. | less than that of glass. |
| 1. | \(\theta = \tan^{-1}\left(\frac{1}{4}\right)\) | 2. | \(\theta = \tan^{-1}(4)\) |
| 3. | \(\theta = \tan^{-1}(2)\) | 4. | \(\theta = 45^{\circ}\) |
In an electrical circuit \(R,\) \(L,\) \(C\) and an AC voltage source are all connected in series. When \(L\) is removed from the circuit, the phase difference between the voltage and the current in the circuit is \(\tan^{-1}\sqrt{3}\). If instead, \(C\) is removed from the circuit, the phase difference is again \(\tan^{-1}\sqrt{3}\). The power factor of the circuit is:
| 1. | \(\dfrac{1}{2} \) | 2. | \(\dfrac{1}{\sqrt{2}}\) |
| 3. | \(1 \) | 4. | \(\dfrac{\sqrt{3}}{2}\) |
| 1. | \(\dfrac{Q}{4\pi R^2\sigma}\) | 2. | \(\left(\dfrac{Q}{4\pi R^2\sigma}\right )^{\dfrac{-1}{2}}\) |
| 3. | \(\left(\dfrac{4\pi R^2 Q}{\sigma}\right )^{\dfrac{1}{4}}\) | 4. | \(\left(\dfrac{Q}{4\pi R^2 \sigma}\right)^{\dfrac{1}{4}}\) |
The current \((I)\) in the inductance is varying with time \((t)\) according to the plot shown in the figure.

| 1. |  |
2. |  |
| 3. |  |
4. |  |
A millivoltmeter of \(25~\text{mV}\) range is to be converted into an ammeter of \(25~\text{A}\) range. The value (in ohm) of the necessary shunt will be:
1. \(0.001\)
2. \(0.01\)
3. \(1\)
4. \(0.05\)
Two persons of masses \(55~\text{kg}\) and \(65~\text{kg}\) respectively, are at the opposite ends of a boat. The length of the boat is \(3.0~\text{m}\) and weighs \(100~\text{kg}.\) The \(55~\text{kg}\) man walks up to the \(65~\text{kg}\) man and sits with him. If the boat is in still water, the centre of mass of the system shifts by:
1. \(3.0~\text{m}\)
2. \(2.3~\text{m}\)
3. zero
4. \(0.75~\text{m}\)
A mixture consists of two radioactive materials A1 and A2 with half-lives of 20 s and 10 s respectively. Initially, the mixture has 40 g of A1 and 160 g of A2. The amount of the two in the mixture will become equal after:
1. 60 s
2. 80 s
3. 20 s
4. 40 s
| 1. | in the case of \(\mathrm{C},\) the valence band is not completely filled at absolute zero temperature. |
| 2. | in the case of \(\mathrm{C},\) the conduction band is partly filled even at absolute zero temperature. |
| 3. | the four bonding electrons in the case of \(\mathrm{C}\) lie in the second orbit, whereas in the case of \(\mathrm{Si},\) they lie in the third. |
| 4. | the four bonding electrons in the case of \(\mathrm{C}\) lie in the third orbit, whereas for \(\mathrm{Si},\) they lie in the fourth orbit. |
The height at which the weight of a body becomes \(\left ( \frac{1}{16} \right )^\mathrm{th}\) of its weight on the surface of the earth (radius \(R\)) is:
1. \(5R\)
2. \(15R\)
3. \(3R\)
4. \(4R\)
| 1. | \(\dfrac{24m}{25hR}\) | 2. | \(\dfrac{25hR}{24m}\) |
| 3. | \(\dfrac{25m}{24hR}\) | 4. | \(\dfrac{24hR}{25m}\) |
A compass needle which is allowed to move in a horizontal plane is taken to a geomagnetic pole. It:
1. will become rigid showing no movement
2. will stay in any position
3. will stay in north-south direction only
4. will stay in east-west direction only
In the circuit shown cells, \(A\) and \(B\) have negligible resistance. For \(V_A =12 ~\text{V},\) \(R_1 = 500 ~\Omega ,\) and \(R = 100 ~\Omega ,\) the galvanometer \((\text{G}) \) shows no deflection. The value of \(V_B\) is:
| 1. | \(4~\text V\) | 2. | \(2~\text V\) |
| 3. | \(12~\text V\) | 4. | \(6~\text V\) |
Four-point charges \(-Q, -q,~ 2q~\text{and}~2Q \) are placed, one at each corner of the square. The relation between \(Q\) and \(q\) for which the potential at the center of the square is zero is:
1. \(Q= -q\)
2. \(Q= -2q\)
3. \(Q= q\)
4. \(Q= 2q\)
A car of mass \(1000\) kg negotiates a banked curve of radius \(90\) m on a frictionless road. If the banking angle is of \(45^\circ,\) the speed of the car is:
| 1. | \(20\) ms–1 | 2. | \(30\) ms–1 |
| 3. | \(5\) ms–1 | 4. | \(10\) ms–1 |
A solid cylinder of mass \(3\) kg is rolling on a horizontal surface with a velocity of \(4\) ms-1. It collides with a horizontal spring of force constant \(200\) Nm-1. The maximum compression produced in the spring will be:
1. \(0.5\) m
2. \(0.6\) m
3. \(0.7\) m
4. \(0.2\) m
One mole of an ideal gas goes from an initial state \(A\) to the final state \(B\) with two processes. It first undergoes isothermal expansion from volume \(V\) to \(3V\) and then its volume is reduced from \(3V\) to \(V\) at constant pressure. The correct \((P-V)\) diagram representing the two processes is:
| 1. |  |
2. | 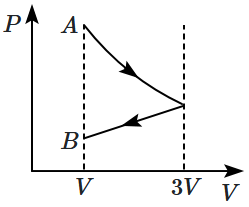 |
| 3. |  |
4. |  |
| 1. | same as that of \(B.\) |
| 2. | opposite to that of \(B.\) |
| 3. | \(\theta = \text{tan}^{-1}\left(\frac{1}{2} \right)\) to the positive \(x\)-axis. |
| 4. | \(\theta = \text{tan}^{-1}\left(\frac{-1}{2} \right )\) to the positive \(x\)-axis. |
Liquid oxygen at \(50~\text K\) is heated up to \(300~\text K\) at a constant pressure of \(1~\text{atm}.\) The rate of heating is constant. Which one of the following graphs represents the variation of temperature with time?
| 1. |  |
2. | 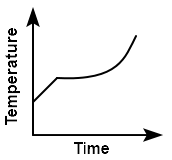 |
| 3. |  |
4. |  |
An alternating electric field of frequency , is applied across the dees (radius=R) of a cyclotron that is being used to accelerate protons (mass=m). The operating magnetic field B, used in the cyclotron and the kinetic energy (K) of the proton beam, produced by it, are given by:
1.
2.
3.
4.
A ray of light is incident at an angle of incidence, \(i\), on one face of a prism of angle \(A\) (assumed to be small) and emerges normally from the opposite face. If the refractive index of the prism is \(\mu,\) the angle of incidence \(i\), is nearly equal to:
| 1. | \(\mu A\) | 2. | \(\dfrac{\mu A}{2}\) |
| 3. | \(\dfrac{A}{\mu}\) | 4. | \(\dfrac{A}{2\mu}\) |
A spherical planet has a mass \(M_p\) and diameter \(D_p\). A particle of mass \(m\) falling freely near the surface of this planet will experience acceleration due to gravity equal to:
| 1. | \(\dfrac{4GM_pm}{D_p^2}\) | 2. | \(\dfrac{4GM_p}{D_p^2}\) |
| 3. | \(\dfrac{GM_pm}{D_p^2}\) | 4. | \(\dfrac{GM_p}{D_p^2}\) |
The damping force of an oscillator is directly proportional to the velocity. The units of the constant of proportionality are:
1. kg-msec-1
2. kg-msec-2
3. kg-sec-1
4. kg-sec
A concave mirror of the focal length \(f_1\) is placed at a distance of \(d\) from a convex lens of focal length \(f_2\). A beam of light coming from infinity and falling on this convex lens-concave mirror combination returns to infinity. The distance \(d\) must be equal to:
1. \(f_1+f_2\)
2. \(-f_1+f_2\)
3. \(2f_1+f_2\)
4. \(-2f_1+f_2\)
A geostationary satellite is orbiting the earth at a height of \(5R\) above the surface of the earth, \(R\) being the radius of the earth. The time period of another satellite in hours at a height of \(2R\) from the surface of the earth is:
1. \(5\)
2. \(10\)
3. \(6\sqrt2\)
4. \(\frac{6}{\sqrt{2}}\)
Transfer characteristics [output voltage () vs input voltage ()] for a base biased transistor in CE configurations are as shown in the figure. For using the transistor as a switch, it is used:
1. In region III
2. Both in the region (I) and (III)
3. In region II
4. In region I
If the voltage across a bulb rated \((220~\text{V}\text-100~\text{W})\) drops by \(2.5\%\) of its rated value, the percentage of the rated value by which the power would decrease is:
1. \(20\%\)
2. \(2.5\%\)
3. \(5\%\)
4. \(10\%\)
A particle has initial velocity \(\left(2 \hat{i} + 3 \hat{j}\right)\) and acceleration \(\left(0 . 3 \hat{i} + 0 . 2 \hat{j}\right)\). The magnitude of velocity after \(10\) s will be:
| 1. | \(9 \sqrt{2}~ \text{units}\) | 2. | \(5 \sqrt{2} ~\text{ units}\) |
| 3. | \(5~\text{units}\) | 4. | \(9~\text{units}\) |
Monochromatic radiation emitted when electron on hydrogen atom jumps from first excited to the ground state irradiates a photosensitive material. The stopping potential is measured to be \(3.57~\text{V}\). The threshold frequency of the material is:
1. \(4\times10^{15}~\text{Hz}\)
2. \(5\times10^{15}~\text{Hz}\)
3. \(1.6\times10^{15}~\text{Hz}\)
4. \(2.5\times10^{15}~\text{Hz}\)
\(\mathrm{ABC}\) is an equilateral triangle with \(O\) as its centre. \(F_1,\) \(F_2,\) and \(F_3\) represent three forces acting along the sides \({AB},\) \({BC}\) and \({AC}\) respectively. If the total torque about \(O\) is zero, then the magnitude of \(F_3\) is:
| 1. | \(F_1+F_2\) | 2. | \(F_1-F_2\) |
| 3. | \(\dfrac{F_1+F_2}{2}\) | 4. | \(2F_1+F_2\) |
The figure shows a logic circuit with two inputs \(A\) and \(B\) and the output \(C\). The voltage waveforms across \(A\), \(B\), and \(C\) are as given. The logic circuit gate is:

1. \(\text{OR}\) gate
2. \(\text{NOR}\) gate
3. \(\text{AND}\) gate
4. \(\text{NAND}\) gate
What is the flux through a cube of side \(a,\) if a point charge of \(q\) is placed at one of its corners?
| 1. | \(\dfrac{2q}{\varepsilon_0}\) | 2. | \(\dfrac{q}{8\varepsilon_0}\) |
| 3. | \(\dfrac{q}{\varepsilon_0}\) | 4. | \(\dfrac{q}{2\varepsilon_0}\) |
An \(\alpha\text-\)particle moves in a circular path of radius \(0.83~\text{cm}\) in the presence of a magnetic field of \(0.25~\text{Wb/m}^2.\) The de-Broglie wavelength associated with the particle will be:
1. \(1~\mathring{A}\)
2. \(0.1~\mathring{A}\)
3. \(10~\mathring{A}\)
4. \(0.01~\mathring{A}\)
The motion of a particle along a straight line is described by the equation \(x = 8+12t-t^3\) where \(x \) is in meter and \(t\) in seconds. The retardation of the particle, when its velocity becomes zero, is:
| 1. | \(24\) ms-2 | 2. | zero |
| 3. | \(6\) ms-2 | 4. | \(12\) ms-2 |
The magnifying power of a telescope is \(9\). When it is adjusted for parallel rays the distance between the objective and eyepiece is \(20~\text{cm}\). The focal length of the lenses is:
| 1. | \(10~\text{cm}, ~10~\text{cm}\) | 2. | \(15~\text{cm}, ~5~\text{cm}\) |
| 3. | \(18~\text{cm}, ~2~\text{cm}\) | 4. | \(11~\text{cm}, ~9~\text{cm}\) |
Two sources of sound placed close to each other, are emitting progressive waves given by,
\(y_1=4\sin 600\pi t\) and \(y_2=5\sin 608\pi t\).
An observer located near these two sources of sound will hear:
| 1. | \(4\) beats per second with intensity ratio \(25:16\) between waxing and waning |
| 2. | \(8\) beats per second with intensity ratio \(25:16\) between waxing and waning |
| 3. | \(8\) beats per second with intensity ratio \(81:1\) between waxing and waning |
| 4. | \(4\) beats per second with intensity ratio \(81:1\) between waxing and waning |
A ring is made of a wire having a resistance of \(R_0=12~\Omega.\). Find points \(\mathrm{A}\) and \(\mathrm{B}\), as shown in the figure, at which a current-carrying conductor should be connected so that the resistance \(R\) of the subcircuit between these points equals \(\frac{8}{3}~\Omega\)

| 1. | \(\dfrac{l_1}{l_2} = \dfrac{5}{8}\) | 2. | \(\dfrac{l_1}{l_2} = \dfrac{1}{3}\) |
| 3. | \(\dfrac{l_1}{l_2} = \dfrac{3}{8}\) | 4. | \(\dfrac{l_1}{l_2} = \dfrac{1}{2}\) |