Three perfect gasses at absolute temperatures are mixed. The masses of molecules are and the number of molecules are respectively. Assuming no loss of energy, the final temperature of the mixture is :
1.
2.
3.
4.
A container with insulated walls is divided into two equal parts by a partition fitted with a valve. One part is filled with an ideal gas at a pressure P and temperature T, whereas the other part is completely evacuated. If the valve is suddenly opened, the pressure and temperature of the gas will be
1.
2.
3.
4.
Air is pumped into an automobile tube up to a pressure of 200 kPa in the morning when the air temperature is 22C. During the day, the temperature rises to 42C and the be expands by 2%. The pressure of the air in the tube at this temperature will be, approximately :
1. 212 kPa
2. 209 kPa
3. 206 kPa
4. 200 kPa
An ideal gas is expanding such that = constant. The coefficient of volume expansion of the gas is:
1.
2.
3.
4.
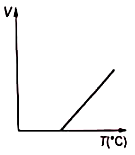
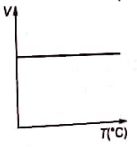
Volume-temperature graph at atmospheric pressure for a monoatomic gas is :
1. 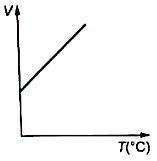 2.
2. 
3. 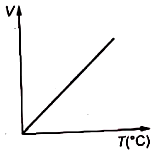 4.
4. 
The equation of state for 5g of oxygen at a pressure P and temperature T, when occupying a volume V, will be
1. PV = (5/32)RT
2. PV = 5RT
3. PV = (5/2)RT
4. PV = (5/16)RT
Temperature remaining constant, the pressure of the gas is decreased by 20%. The percentage change in volume :
1. increases by 20%
2. decreases by 20%
3. increases by 25%
4. decreases by 25%
The figure below shows the plot of versus p for oxygen gas at two different temperatures.
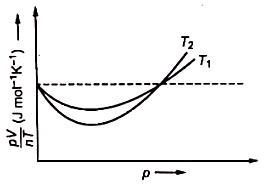
Read the following statements concerning the above curves.
(i) The dotted line corresponds to the ideal gas behavior
(ii)
(iii) The value of at the point where the curve meets on the y-axis is the same for all gases.
1. (i) only
2. (i) and (ii)
3. All of these
4. None of these
The root mean square velocity of gas molecules at 27C is 1365 . The gas is :
1.
2. He
3.
4.
Two identical vessels A and B with frictionless pistons contain the same ideal gas at the same temperature and the same volume V. The masses of gas in A and B are and respectively. The gases are allowed to expand isothermally to the same final volume 2 V. The change in pressures of the gas in A and B are found to be P and 1.5P respectively. Then :
1. 9 = 4
2. 3 = 2
3. 2 = 3
4. 4 = 9
A bubble is at the bottom of the lake of depth h. As the bubble comes to sea level, its radius increases to three times. If atmospheric pressure is equal to l meter of the water column, then h is equal to :
1. 26l
2. l
3. 25l
4. 30l
Calculate the ratio of RMS speeds of oxygen gas molecules to that of hydrogen gas molecules kept at the same temperature :
1. 1:4
2. 1:8
3. 1:2
4. 1:6
The root mean square speed of the molecules of an ideal gas is v. If the pressure is increased to two times at a constant temperature, the RMS speed will become :
1.
2. v
3. 2v
4. 4v