A Carnot freezer takes heat from water at C inside it and rejects it to the room at a temperature of C. The latent heat of ice is J/kg. If 5 kg of water at C is converted into ice at C by the freezer, then the energy consumed by the freezer is closed to:
(1)
(2)
(3)
(4)
Which of the following shows the correct relationship between the pressure 'P' and density of an ideal gas at constant temperature?
(1)
(2)
(3)
(4)
are specific heats at constant pressure and constant volume respectively. It is observed that = a for hydrogen gas, for nitrogen gas.
The correct relation between a and b is:
(1) a = 28 b
(2)
(3) a = b
(4) a = 14b
An engine operates by taking n moles of an ideal gas through the cycle ABCDA shown in (Fig. 8.19). The thermal efficiency of the engine is:
(Take , where R is gas constant)
(1) 0.24
(2) 0.15
(3) 0.32
(4) 0.08
An ideal gas has molecules with 5 degrees of freedom. The ratio of specific heats at constant pressure and at constant volume is:
(1) 6
(2) 7/2
(3) 5/2
(4) 7/5
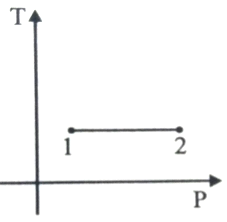
For the P-V diagram given for an ideal gas,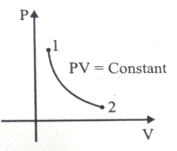
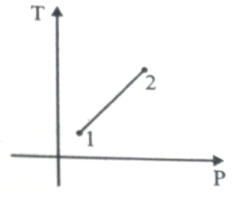
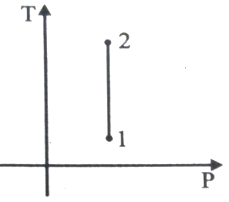
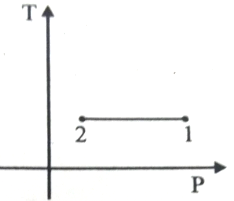
Out of the following which one correctly represents the T-P diagram?
1. 
2. 
3.
4.
N moles of a diatomic gas in a cylinder are at temperature T. Heat is supplied to the cylinder such that the temperature remains constant but n moles of the diatomic gas get converted into monoatomic gas. What is the change in the total kinetic energy of the gas?
(1)
(2) 0
(3)
(4)
Two moles of an ideal monoatomic gas occupies a volume V at 27. The gas expands adiabatically to a volume of 2V. Calculate final temperature of the gas .
1. 295 K
2. 189 K
3. 395 K
4. 298 K
A Carnot engine works like a refrigerator between 250 K and 300 K. It receives 500 cal heat from the reservoir at the lower temperature. The amount of work done in each cycle to operate the refrigerator is:
(1) 420 J
(2) 772 J
(3) 2100 J
(4) 2520 J
One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, .The work done on the gas will be:
(1) 300R
(2) 300R
(3) 300R
(4) 300R
Two Carnot engines A and B are operated in series. Engine A receives heat from a reservoir at 600 K and rejects heat to a reservoir at temperature T. Engine B receives heat rejected by engine A and in turn rejects it to a reservoir at 100K. If the efficiencies of the two engines A and B are represented by , respectively, then what is the value of ?
(1) 12/7
(2) 7/12
(3) 12/5
(4) 5/12
One mole of an ideal monoatomic gas is taken along the path ABCA as shown in the PV diagram (Fig. 8.26). The maximum temperature attained by the gas along the path BC is given by:
(1)
(2)
(3)
(4)
Two moles of helium are mixed with n moles of hydrogen. If for the mixture, then the value of n is :
(1) 1
(2) 3
(3) 2
(4) 3/2
A body takes 10 minutes to cool from . The temperature of the surroundings is constant at . Then, the temperature of the body after next 10 minutes will be approximately:
(1)
(2)
(3)
(4)
The value closest to the thermal velocity of a Helium atom at room temperature (300 K) in :
(1)
(2)
(3)
(4)