One mole of diatomic ideal gas undergoes a cyclic process ABC as shown in the figure. The process of BC is adiabatic. The temperature at A, B and C are 400 K, 800 K, and 600 K respectively. Choose the correct statement:
1. The change in internal energy in the process AB is - 350R.
2. The change in internal energy in the process BC is - 500R.
3. The change in internal energy in the whole cyclic process is 250R.
4. The change in internal energy in the process CA is 700R.
The equation of state for a gas is given by PV=nRT+V, where n is number of moles and is positive constant. The initial temperature and pressure of one mole of the gas contained in a cylinder are respectively. The work done by the gas when its temperature doubles isobarically will be:
1.
2.
3.
4.
During an adiabatic compression, 830 J of work is done on 2 moles of a diatomic ideal gas to reduce its volume by 50%. The change in its temperature is nearly:
1. 40 K
2. 33 K
3. 20 K
4. 14 K
An ideal monoatomic gas confined in a cylinder by a spring loaded piston of cross-section . Initially, the gas is at 300 K and occupies a volume of and the spring is in its released state as shown in the figure. The gas is heated by a smaller heater until the piston moves out slowly by 0.1 m. The force constant of the spring is 8000 N/m and the atmospheric pressure is . The cylinder and the piston are thermally insulated. The piston and the spring are massless and there is no friction between the piston and cylinder. The final temperature of the gas will be:
(Neglect the heat loss through the lead wires of the heater. The heat capacity of the heater coil is also negligible).
1. 300 K
2. 800 K
3. 500 K
4. 1000 K
A Carnot engine absorbs 1000 J of heat energy from a reservoir at and rejects 600 J of heat energy during each cycle. The efficiency of the engine and temperature of the sink will be:
1.
2.
3.
4.
A gas is compressed from a volume of to a volume of at constant pressure of 100 N/. Then it is heated at constant volume by supplying 150 J of energy. As a result, the internal energy of the gas :
1. increases by 250 J
2. decreases by 250 J
3. increases by 50 J
4. decreases by 50 J
A gas molecule of mass M at the surface of the Earth has kinetic energy equivalent to that at . If it were to go up straight without colliding with any other molecule, how high it would rise? Assume that the height attained is much less than the radius of the earth:
1. 0
2.
3.
4.
Consider a spherical shell of radius R and temperature T. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume. If the shell now undergoes an adiabatic expansion, the relation between T and R is:
P-V curves for isothermal and adiabatic
processes of an ideal gas1.
2.
3.
4.
Consider an ideal gas confined in an isolated closed chamber. As the gas undergoes an adiabatic expansion, the average time of collision between molecules increases as , where V is the volume of gas. The value of q is :
1.
2.
3.
4.
A solid body of constant heat capacity is being heated by keeping it in contact with reservoirs in two ways:
a. Sequentially keeping in contact with 2 reservoirs such that each reservoir supplies the same amount of heat.
b. Subsequently keeping in contact with 8 reservoirs such that each reservoir supplies the same amount of heat.
In both cases, the body is brought from the initial temperature to the final temperature . Entropy change of the body in the two cases respectively is :
An ideal gas going through the reversible cycle , has the V-T diagram as shown below in the figure. Process are adiabatic.
The corresponding P-V diagram for the process is (all figures are schematic and not drawn to scale):
1.
2.
3.
4.
'n' moles of an ideal gas undergoes a process as shown in the figure. The maximum temperature of the gas during the process will be: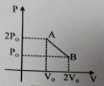
1.
2.
3.
4.
An ideal gas undergoes a quasi-static, reversible process in which the molar heat capacity C remains constant. If during this process the relation of pressure P and volume V is given by constant, then n is given by (Here are molar specific heat at constant pressure and constant volume, respectively).
1.
2.
3.
4.
200 g water is heated from . Ignoring the slight expansion of water, the change in internal energy is close to (Given specific heat of water=4184 J/Kg K) :
1. 8.4 kJ
2. 4.2 kJ
3. 16.7 kJ
4. 167.4 kJ
The ratio of work done by an ideal monoatomic gas to the heat supplied to it in an isobaric process is:
1. 3/5
2. 2/3
3. 3/2
4. 2/5