An action spectrum of photosynthesis was first described by:
1. Jan Ingenhousz
2. Julius von Sachs
3. T.W. Engelmann
4. Cornelius van Niel
Dark reactions of photosynthesis are actually not totally independent of light as:
1. The initial reactions occur in the presence of light.
2. The reaction will be inhibited by the presence of light.
3. They can occur during the day as well.
4. They utilize the products of the light reaction.
All the following regarding the role of xanthophylls and carotenoids in photosynthesis are correct except:
| 1. | They act as accessory pigments |
| 2. | Their peak absorption is in blue and red regions of VIBGYOR |
| 3. | They enable a wider range of wavelength of incoming light to be utilized for photosynthesis |
| 4. | They protect chlorophyll a form of photo-oxidation. |
The chloroplast stroma lamella membrane:
| 1. | lacks PS II as well as NADP reductase enzyme. |
| 2. | lacks PS II but has NADP reductase enzyme. |
| 3. | lacks NADP reductase enzyme but has PS II. |
| 4. | has PS II as well as NADP reductase enzyme. |
Which stage of the Calvin Cycle utilizes ATP?
1. Carboxylation
2. Reduction
3. Regeneration
4. Both 2 and 3
Which of the following are special features of the C4 plants?
| I: | They have a special type of leaf anatomy. |
| II: | They tolerate higher temperatures and show a response to high light intensities. |
| III: | They lack photorespiration and have greater productivity of biomass. |
| 1. | Only I and II |
| 2. | Only I and III |
| 3. | Only II and III |
| 4. | I, II and III |
Plants can get along without respiratory organs because of the following except:
| 1. | Each plant part takes care of its own gas exchange needs |
| 2. | Plants do not present great demands for gas exchange. |
| 3. | Gas diffusion in plants occurs easily over long distances in plants. |
| 4. | Each living cell in a plant is located quite close to the surface of the plant. |
Sucrose is converted into glucose and fructose by the enzyme:
1. Maltase
2. Zymase
3. Isomerase
4. Invertase
The co-factor required for the activity of pyruvate dehydrogenase is:
1. Zinc
2. Magnesium
3. Manganese
4. Copper
In Kreb's cycle, a molecule of GTP is produced during the conversion of:
1. Citrate into Ketoglutarate
2. Succinyl-CoA into succinate
3. Succinate into malate
4. Malate into oxaloacetate
In the mitochondrial electron transport system, the term Complex III refers to:
1. NADH dehydrogenase
2. Cytochrome c
3. FADH2
4. Cytochrome bc1 complex
For each ATP produced, how many H passes through F0 from the intermembrane space to the matrix down the electrochemical proton gradient?
1. 1
2. 2
3. 3
4. 4
What is the net gain of the number of ATP molecules produced during aerobic respiration of one molecule of glucose?
1. 30
2. 36
3. 38
4. 40
If fatty acids were to be respired, they would first be degraded to:
1. Glucose-6 phosphate
2. Pyruvate
3. Acetyl-CoA
4. Alpha ketoglutarate
The formation of interfascicular cambium in plants is due to:-
1. Non-differentiation
2. Re-differentiation
3. Differentiation
4. De-differentiation
Heterophyllous development due to environment is an example of:-
1. Developmental noise
2. Norm of the reaction
3. Convergence
4. Plasticity
Gibberellic acid is a/an:-
1. lndole compound
2. Terpene
3. Adenine derivative
4. Carotenoid
The PGR used to initiate flowering and for synchronizing fruit-set in pineapples is:
1. Abscisic acid
2. Cytokinin
3. Auxin
4. Ethylene
Cytokinin does not help in:
1. Lateral shoot growth
2. Overcoming apical dominance
3. Adventitious shoot formation
4. Promoting leaf senescence
In most situations, which of the following acts as an antagonist to GAs?
1. ABA
2. IAA
3. Kinetin
4. Ethylene
Which of the following is the site for perception of light/dark in plants?
1. Root apex
2. Shoot apex
3. Leaves
4. Flower
Histamine in the blood is secreted by:
| 1. | Mast cells | 2. | Macrophages |
| 3. | Eosinophils | 4. | Basophils |
A person with AB blood group:
| 1. | Has A and B antigens in his plasma |
| 2. | Has antibodies on RBC surface |
| 3. | Can receive blood transfusion from a person of his own group. |
| 4. | Can donate blood to all other group recipients |
The inter-atrial septum in the human heart can be best described as:
1. A thin muscular wall
2. A thick muscular wall
3. A thin fibrous tissue
4. A thick fibrous tissue
The sino-atrial node is the normal pacemaker of the human heart because:
| 1. | It is located in the right atrium. |
| 2. | It is the only part of the conducting system that can generate impulse. |
| 3. | It can generate the maximum number of action potentials in the conducting system. |
| 4. | It produces action potentials that are more in size than the other parts of the conducting system. |
What percentage of ventricular filling is achieved by atrial contraction?
1. 30
2. 50
3. 70
4. 90
The second heart sound during the cardiac cycle is produced by the:
1. Simultaneous opening of the atrioventricular valves
2. Simultaneous closure of the atrioventricular valves
3. Simultaneous opening of the semilunar valves
4. Simultaneous closure of the semilunar valves
In a surface ECG the QRS complex represents:
1. Atrial depolarization
2. Atrial repolarization
3. Ventricular depolarization
4. Ventricular repolarization
Normal activities of the human heart are regulated:
1. Intrinsically
2. By the autonomic nervous system
3. By the brain stem
4. By the diencephalon
A common symptom produced by myocardial ischemia is:
1. Hypertension
2. Vertigo
3. Angina
4. Hyperventilation
The blood from the glomerulus is carried away by:
1. Afferent arteriole
2. Efferent arteriole
3. Peritubular capillary network
4. Renal vein
Vasa recta in cortical nephrons :
1. Arises from afferent arteriole rather than efferent arteriole
2. Does not get involved in counter current exchange with loop of Henle
3. Carries deoxygenated blood rich in urea
4. Is absent or highly reduced
The proximal convoluted tubule is lined by the:
1. Simple cuboidal epithelium
2. Simple columnar epithelium
3. Simple cuboidal brush bordered epithelium
4. Simple columnar brush bordered epithelium
During the concentration of urine by the human kidneys, NaCl is returned to the medullary interstitium by the:
1. Descending limb of the loop of Henle
2. Ascending limb of the loop of Henle
3. Descending limb of the vasa recta
4. Ascending limb of the vasa recta
An increase in the blood flow to the atria of the heart can cause the release of:
1. Erythropoeitin
2. Atrial natriuretic factor
3. Renin
4. Aldosterone
On average, what amount of urea is excreted out per day by a healthy human being?
1. 5-10 gm
2. 15-20 gm
3. 25-30 gm
4. 50-60 gm
Skeletal muscle bundles [fascicles] are held together by a common connective tissue layer called:
1. Perimysium
2. Endomysium
3. Fascia
4. Aponeurosis
The neurotransmitter released at the neuromuscular junction is:
1. Adrenaline
2. Nor-adrenaline
3. Dopamine
4. Acetylcholine
During muscle contraction, the length of all the following gets reduced except:
1. H-Zone
2. A-Band
3. I-Band
4. Sacromere
What is true about the white muscle fibers?
1. Myoglobin content is high
2. They have a large number of mitochondria
3. They depend on anaerobic process for energy
4. They are adapted for slow sustained activities
Which of the following is not a cranial bone?
| 1. | Zygomatic | 2. | Sphenoid |
| 3. | Ethmoid | 4. | Parietal |
The human ribs are termed as bicephalic because:
| 1. | They have two articulations surfaces on their ventral end. |
| 2. | They have two articulations surfaces on their dorsal end. |
| 3. | They have two articulations surfaces on their ventral end and two on the dorsal end. |
| 4. | They have two articulations surfaces on their ventral end and one on the dorsal end. |
The clavicle is articulated with the:
1. Xiphoid process of the sternum
2. Acromion of the scapula
3. Glenoid cavity of the scapula
4. Head of the humerus
Hypocalcemia may lead to:-
1. Kidney stones
2. Cholelithiasis
3. Stronger muscle contractions
4. Tetany
| 1. | Not enclosed by a Schwann cell and hence not enclosed by a myelin sheath. |
| 2. | Enclosed by a Schwann cell that does not form a myelin sheath. |
| 3. | Not enclosed by a Schwann cell but enclosed by a myelin sheath formed by oligodendrocytes. |
| 4. | Not found in the autonomous and the somatic neural system. |
The brain stem does not include :
1. Mid-brain
2. Medulla oblongata
3. Pons Varolii
4. Cerebellum
Which of the following is a major coordinating center for sensory and motor signaling for the cerebrum?
1. Hypothalamus
2. Thalamus
3. Corpora quadrigemina
4. Amygdala
The sensory neuron enters the spinal cord through:
| 1. | The dorsal root of the spinal nerve |
| 2. | The ventral root of the spinal nerve |
| 3. | Either dorsal or ventral root depending on the point of origin |
| 4. | Both dorsal and ventral roots of the spinal nerves |
From inside to outside the correct order of the following cells in the retina would be:
1. Ganglion cells - Photoreceptor cells - Bipolar cells
2. Ganglion cells - Bipolar cells - Photoreceptor cells
3. Bipolar cells - Photoreceptor cells - Ganglion cells
4. Photoreceptor cells - Ganglion cells - Bipolar cells
In the human eye, the visual acuity is highest at:
1. Macula lutea
2. Optic disc
3. Fovea centralis
4. Optic chiasma
Reissner's membrane separates:
1. Scala vestibuli and Scala media
2. Tympanic cavity and Pharynx
3. Fenestra rotundus and Fenestra ovalis
4. Scala media and Scala tympani
Glucocorticoids do not:
1. Stimulate gluconeogenesis
2. Cause lipolysis
3. Cause proteolysis
4. Stimulate cellular uptake and utilization of amino acids
The development of secondary sexual characters in females is primarily controlled by:
1. Estrogen
2. Progesterone
3. LH
4. FSH
Which hormone stimulates exocrine pancreas to secrete water and bicarbonate ions?
1. Gastrin
2. Cholecystokinin
3. Secretin
4. Gastric inhibitory peptide
A hormone that has intracellular receptors would be
1. ACTH
2. ADH
3. Melatonin
4. Aldosterone
A hormone that is antagonist to the parathyroid hormone is secreted from :
1. Thyroid
2. Adrenal cortex
3. Hypothalamus
4. Parathyroid itself
The immune responses in older people are weak due to the degeneration of:
1. Thyroid
2. Pineal
3. Adrenal
4. Thymus
Which of the following endocrine glands is under the direct neural regulation of the hypothalamus?
1. Pineal
2. Thymus
3. Posterior pituitary
4. Adrenal medulla
Which of the following is true?
1. Hormones regulate cellular functions, and generally negative feedback regulates hormone levels.
2. The circulating level of a hormone is held constant through a series of positive feedback loops.
3. Both lipid-soluble hormones and water soluble hormones bind to intracellular protein receptors.
4. The ducts of endocrine organs release their contents into the bloodstream.
The resting membrane potential is established primarily due to:
1. Sodium-potassium pump
2. Efflux of potassium
3. Influx of sodium
4. Influx of chloride
Identify the correct statement regarding the excretion of acids by the human kidney :
1. The filtered bicarbonate is absorbed by anion transport
2. The intercalated cells of the distal tubule secrete hydrogen ions to reabsorb bicarbonate from the tubular fluid.
3. All of the filtered bicarbonate is normally reabsorbed in the first half of the distal tubule.
4. Urine pH is normally greater than that of plasma.
Blood doping means use of banned substances by athletes that have a favorable effect on erythrocyte count by stimulating the bone marrow. The source of such a hormone can be the human:
1. Spleen
2. Heart
3. Kidney
4. Lungs
The main disadvantage of urea as an excretory product for animals eliminating it would be:
1. its high toxicity
2. Needs lot of water to be eliminated
3. It being an osmolyte
4. Energy expenditure to produce it
Match each item in Column I with one in Column II and choose the correct option from the codes given below:
| Column I | Column II | ||
| A. | Filtration fraction | a. | 19% |
| B. | Normal Blood Urea Level | b. | 23% |
| C. | GFR | c. | 70-100 mg % |
| d. | 15-40 mg % | ||
| e. | 180 ml/min | ||
| f. | 125 ml/min |
| A | B | C | |
| 1. | b | d | f |
| 2. | a | d | f |
| 3. | a | c | e |
| 4. | b | d | e |
The steps of the cardiac cycle in sequence are...
1. isovolumic contraction, isovolumic relaxation, ejection, passive ventricular filling, active ventricular filling.
2. isovolumic relaxation, isovolumic contraction, ejection, passive ventricular filling, active ventricular filling.
3. isovolumic contraction, ejection, isovolumic relaxation, passive ventricular filling, active ventricular filling.
4. isovolumic contraction, ejection, isovolumic relaxation, active ventricular filling, passive ventricular filling
A characteristic of human cornea is :
1. absence of blood circulation
2. cause cataract in old age
3. has lacrimal glands for secreting tears
4. secreted by conjunctiva and glandular layer
Most sensory receptors function by producing depolarising potentials. Which of the following function by hyperpolarization rather than depolarization ?
1. proprioceptors
2. nociceptors
3. olfactory receptors
4. rods and cones
A flash of red light followed by a flash of far-red light given during the middle of the night to a short-day plant will likely
1. cause increased flower production.
2. have no effect upon flowering.
3. inhibit flowering.
4. stimulate flowering.
Both red and blue light are involved in?
1. stem elongation.
2. photoperiodism.
3. positive phototropism.
4. tracking seasons.
Which of the following statements about plant hormones is false?
| 1. | The growth of plants in nature is probably regulated by a combination of growth-stimulating and growth-inhibiting hormones. |
| 2. | Abscisic acid generally promotes growth. |
| 3. | Gibberellins stimulate cell enlargement. |
| 4. | Cytokinins promote cell division. |
Compared to retinal, chlorophyll can be described as a pigment that has a
| 1. | narrow absorption range but high efficiency |
| 2. | narrow apsorption range but low efficiency |
| 3. | wide absorption range but high effeciency |
| 4. | wide absorption range but low efficiency |
In the absence of oxygen, the primary purpose of fermentation is to:
| 1. | produce amino acids for protein synthesis |
| 2. | generate a proton gradient for ATP synthesis |
| 3. | oxidize glucose to generate reduce electron carriers |
| 4. | regenerate NAD+ from NADH allowing glycolysis to continue |
Consider the situation where photosynthesis is going on at a steady pace in a typical experiment with a light on, and carbon dioxide is being combined with ribulose-bisphosphate (RuBP) to produce 3-phosphoglycerate (3PG). Then suddenly the source of carbon dioxide is eliminated. What changes in the concentrations of 3PG and RuBP would occur?
| 1. | 3PG levels rise, RuBP levels fall. |
| 2. | 3PG levels fall, RuBP levels rise. |
| 3. | 3PG levels rise, RuBP levels rise. |
| 4. | 3PG levels fall, RuBP levels fall. |
Identify the statement that is true for C4 plants but not for C3 plants?
| 1. | Initially fix carbon dioxide in mesophyll cells as 4-carbon long compounds. |
| 2. | Initially fix carbon dioxide in bundle sheath cells as 3-carbon long compounds. |
| 3. | Initially fix carbon dioxide in mesophyll cells as 3-carbon long compounds. |
| 4. | Initially fix carbon dioxide in bundle sheath cells as 4-carbon long compounds. |
Fatty acids enter cellular respiration as:
1. One carbon fragments
2. Two carbon fragments
3. Three carbon fragments
4. Long chains of 16 to 20 carbon atoms
During glycolysis, when glucose is catabolized to pyruvate, most of the energy of glucose is
| 1. | transferred to ADP, forming ATP. |
| 2. | transferred directly to ATP. |
| 3. | retained in the pyruvate. |
| 4. | stored in the NADH produced. |
In which reaction hydrogen is not formed ?
(1) Copper and hydrochloric acid
(2) Iron and sulphuric acid
(3) Magnesium and steam
(4) Sodium and alcohol
Which salt does not cause hardness of water ?
(1) Mg(HCO3)2
(2) CaCl2
(3) MgSO4
(4) MgCO3
Pick the correct statement :
(1) Borax is white crystalline solid containing [B4O5(OH)4]2- units
(2) Aqueous solution of borax is acidic in nature.
(3) Cobalt gives green colour in borax bead test.
(4) All are incorrect.
An amphoteric oxide among the following is-
1. CO2
2. CO
3. SnO2
4. CaO
The incorrect statement among the following is-
| 1. | Lithium is the strongest reducing agent. |
| 2. | Density of potassium is more than sodium. |
| 3. | Cs are used in devising photoelectric cells. |
| 4. | All alkali metals give a blue solution in liquid ammonia. |
Property of the alkaline earth metals that increases with their atomic number is
(1) ionisation energy
(2) solubility of their hydroxides
(3) solubility of their sulphates
(4) electronegativity
Which salt will not impart color to flame?
1. LiCl
2. MgCl2
3. CaCl2
4. KI
In B2H6
(1) there is direct boron-boron bond
(2) the structure is similar to that of C2H6
(3) the boron atoms are linked through hydrogen bridges
(4) all the atoms are in one plane
H3BO3 is
(1) monobasic and weak Lewis acid
(2) monobasic and weak bronsted acid
(3) monobasic and strong Lewis acid
(4) tribasic and weak Bronsted acid
Which order for basic character of amine is correct for following compounds?
1. 3>1>2>5>4 2. 3>2>1>5>4
3. 3>1>2>4>5 4. 3>2>1>4>5
What is the decreasing order of acidity for the compounds given below?

| 1. | III>IV>I>II | 2. | I>IV>III>II |
| 3. | II>I>III>IV | 4. | IV>III>I>II |
Which of the provided alkenes exhibits the greatest stability?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
What is major product of following reaction ?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The correct increasing order of carbon-carbon bond length in the given compounds is:
1. III<II<I<IV
2. IV<I<II<III
3. I<II<III<IV
4. I<IV<III<II
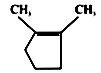
1.
2.
3. 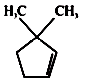
4. 
Consider the given reaction below:
Which of these is true regarding the reaction shown above?
| 1. | The configuration of the chiral carbon remains the same. |
| 2. | The configuration of the chiral carbon gets inverted |
| 3. | The compound formed as a product must be a dextro isomer |
| 4. | The reactant is optically inactive but the product is obtained as a levo isomer. |

A is:
1. 3-keto butanol
2. Butan-1,4-dial
3. 1-aldo-2-butanone
4. 2-keto butan-1,4-dial
Which of the following happens to be the carbanion with highest stability?
The IUPAC name of the following compounds is:

1. 1-Bromocyclohexane carboxylic acid
2. 3-Bromocyclohexanoic acid
3. 3-Bromoheptanoic acid
4. 3-Bromocyclohexanecarboxylic acid
A compound 'A'
1. Decolourises Baeyer's reagent
2. On hydroxylation and futher oxidation with gave formic acid and acetone
3. On ozonolysis gave one molecule of acetone and one of formaldehyde 'A' is
1. Propylene 2. Ethylene
3. n-butylene 4. Isobutylene
Trans-2-butene on bromination using CS2 gives which dibromoderivative
1. (R), (R)-2,3-dibromobutane
2. (S),(S)-2,3-dibromobutane
3. Racemic-2,3-dibromobutane
4. Meso-2,3-dibromobutane
The most reactive of these compounds towards sulphonation is
1. Toluene 2. Chlorobenzene
3. Nitrobenzene 4. m-Xylene

Product 2 is
Gas A
Gas B
Gas C
Gas (A+B+C)
gas mixture Gas X
Gas X is-
1. Gas A + Gas B 2. Only Gas B
3. Gas B + Gas C 4. Only Gas A
What is the product formed when n-heptane is heated with a mixture of Cr2O3 and Al2O3 at 600°C?
| 1. | Cyclohexane | 2. | Cyclohexene |
| 3. | Benzene | 4. | Toluene |
The ease of dehydrohalogen of alkyl halide with alcoholic KOH is
1. 3º > 2º > 1º
2. 3º < 2º < 1º
3. 3º > 2º < 1º
4. 3º < 2º > 1º
The intermediate during the addition of HCl to propene in the presence of peroxide is
1.
2.
3.
4.
Which of the following carbocations is expected to be most stable?
1.
2.
3.
4.
A group that deactivates the benzene ring towards electrophilic substitution but which directs the incoming group principally to the o- and p-position is.
1. -NH2
2. -Cl
3. -NO2
4. -C2H5
In the given conformation C2 is rotated about C2-C3 bond anticlockwise by an angle of 120° then the conformation obtained is
1. Fully eclipsed conformation
2. Partially eclipsed conformation
3. Gauche conformation
4. Staggered conformation
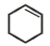
Which of the following is formed by the thermal decomposition of the hydroxide of:
1. 
2. 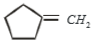
3. 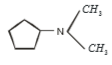
4. 
| 1. | the motion is oscillatory but not SHM. |
| 2. | the motion is SHM with an amplitude \(a\sqrt{2}\). |
| 3. | the motion is SHM with an amplitude \(\sqrt{2}\). |
| 4. | the motion is SHM with an amplitude \(a\). |
| 1. | \(2A,A\) | 2. | \(4A,0\) |
| 3. | \(A,A\) | 4. | \(0,2A\) |
The displacement of a particle executing SHM is given by y = 0.25 (sin 200t) cm. The maximum speed of the particles is:
1. 200 cm/sec
2. 100 cm/sec
3. 50 cm/sec
4. 0.25 cm/sec
Which of the following figure represents damped harmonic motion?
| (i) |  |
| (ii) |  |
| (iii) |  |
| (iv) |  |
1. (i) and (ii)
2. (iii) and (iv)
3. (i), (ii), (iii), and (iv)
4. (i) and (iv)
If P is the pressure of the gas then the KE per unit volume of the gas is:
1.
2.
3.
4.
A gas mixture consists of 2 moles of oxygen and 4 moles of argon at temperature T. Neglecting all vibrational modes, the total internal energy of the system is
1. 4RT
2. 15RT
3. 9RT
4. 11RT
In a given process, dW = 0, dQ < 0, then for the gas:
1. Temperature increases
2. Volume decreases
3. Pressure decreases
4. Pressure increases
A given mass of gas expands from state \(A\) to state \(B\) by
three paths \(1, 2~\text{and}~3\), as shown in the figure. If \(W_1, W_2~\text{and}~W_3\) respectively be the work done by the gas along the three paths, then:

| 1. | \(W_1 >W_2>W_3\) | 2. | \(W_1<W_2<W_3\) |
| 3. | \(W_1 =W_2=W_3\) | 4. | \(W_1 <W_2=W_3\) |
The ratio of the relative rise in pressure for adiabatic compression to that for isothermal compression is
1.
2.
3. 1-
4.
A sink, that is, the system where heat is rejected, is essential for the conversion of heat into work. From which law does the above inference follow?
1. Zeroth
2. First
3. Second
4. Third
For the indicator diagram given below, which of the following is not correct?

| 1. | Cycle II is a heat engine cycle. |
| 2. | Net work is done on the gas in cycle I. |
| 3. | Work done is positive for cycle I. |
| 4. | Work done is positive for cycle II. |
A plane progressive wave cannot be represented by
1.
2.
3.
4.
The phase difference between the prongs of a tuning fork is :
1.
2.
3.
4.
The isothermal elasticity of a medium is and the adiabatic elasticity in . The velocity of sound in the medium is proportional to
1.
2.
3.
4.
When a wave is reflected from a denser medium the change in phase is :
1. 0
2.
3.
4.
In a stationary wave along a string, the strain is:
1. zero at the antinodes
2. maximum at the antinodes
3. zero at the nodes
4. maximum at the nodes
A source of sound moves away with the velocity of sound from a stationary observer. The frequency of the sound heard by the observer:
1. remains the same
2. is doubled
3. is halved
4. becomes infinity
The variation of pressure versus temperature of an ideal gas is shown in the given diagram. From this diagram, one can conclude that

| 1. | Volume increases continuously |
| 2. | Volume decreases continuously |
| 3. | Volume first increases then decreases |
| 4. | Volume first decreases, then increase |
The variation of density (p) of gas with its absolute temperature (T) at constant pressure is best represented by the graph
| 1. |  |
2. |  |
| 3. |  |
4. |  |
An ideal gas with adiabatic exponent y is heated at constant pressure and it absorbs Q heat. What fraction of this heat is used to perform external work?
1.
2.
3.
4.
A Carnot engine working between 400K and 800K has a work output of 900J per cycle. The amount of heat energy supplied to engine from the source per cycle is:
1. 900 J
2. 1800 J
3. 450 J
4. 2700 J
Temperature is defined by
1. First Law of thermodynamics
2. Second Law of thermodynamics
3. Third Law of thermodynamics
4. Zeroth Law of thermodynamics
The fastest mode of heat transfer is
1. Conduction
2. Radiation
3. Convection
4. all are equally fast
The P-V diagram for a thermodynamic system is shown in the figure. The work done by the system during the cyclic process ABCA is

1. Zero
2. 90 J
3. 30 J
4. 60 J
A temperature of \(100^{\circ}\text {F}\) (Fahrenheit scale) is equal to \(T~\text{K}\) (Kelvin scale). The value of \(T\) is:
1. \(310.9\)
2. \(37.8\)
3. \(100\)
4. \(122.4\)
A body cools down from \(80^{\circ}\mathrm{C}\) \(70^{\circ}\mathrm{C}\)
| 1. | less than 5 minutes. |
| 2. | equal to 5 minutes. |
| 3. | more than 5 minutes. |
| 4. | can't say anything as the temperature of the surroundings is not known. |
| 1. | \( \frac{T}{12} \) | 2. | \(\frac{5 T}{12} \) |
| 3. | \( \frac{7 T}{12} \) | 4. | \(\frac{2 T}{3}\) |
The longitudinal waves can travel :
1. only in liquids
2. only in solids
3. only in gases
4. In liquids, solids, and gases
If \(\lambda_m\) is the wavelength, corresponding to which the radiant intensity of a block is at its maximum and its absolute temperature is \(T,\) then which of the following graphs correctly represents the variation of \(T?\)
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The time period of a spring mass system at the surface of the earth is \(2~\text{s}.\) What will be the time period of this system on the moon where the acceleration due to gravity is \(\frac{1}{16}^\text{th}\) of the value of \(g\) on the earth's surface?
| 1. | \(\frac{1}{\sqrt{6}} ~\mathrm{s} \) | 2. | \(2 \sqrt{6}~ \mathrm{s} \) |
| 3. | \(2~ \mathrm{s} \) | 4. | \( 12~\mathrm{ s}\) |
The equation of a wave pulse travelling along x-axis is given by , x and y are in meters and t is in seconds. The amplitude of the wave pulse is
1. 5 m
2. 20 m
3. 15 m
4. 30 m
| 1. | \(100~\text{times}\) | 2. | \(50~\text{times}\) |
| 3. | \(200~\text{times}\) | 4. | \(400~\text{times}\) |
| 1. | \(100~\text{and}~50\) | 2. | \(44~\text{and}~22\) |
| 3. | \(80~\text{and}~40\) | 4. | \(72~\text{and}~30\) |
| 1. | \(275~\text{Hz}\) | 2. | \(175~\text{Hz}\) |
| 3. | \(525~\text{Hz}\) | 4. | \(575~\text{Hz}\) |
The sound intensity level at a point 10 m away from a point source is 20dB, then the sound level at a distance 1m from the same source would be
1. 40 dB
2. 30 dB
3. 200 dB
4. 100 dB
In the phenomenon of interference of sound by two coherent sources if difference of intensity at maxima to intensity at minima is 20dB, then the ratio of intensities of the two sources is
1.
2.
3.
4.
The maximum concentration of alcohol in beverages that are naturally fermented is:
1. 5 - 10%
2. 10 - 15%
3. 20 - 25%
4. 45 - 50%
Which of the following is the most stable carbocation
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
In the following compounds,
The order of basicity is
1. IV>I>III>II
2. III>I>IV>II
3. II>I>III>IV
4. I>III>II>IV