Which of the following is the connecting link between glycolysis and Krebs cycle?
1. Acetyl Co-A
2. Oxalosuccinic acid
3. Pyruvic acid
4. Citric acid
Photochemical phase does not include-
a. water splitting
b. oxygen release
c. CO2 assimilation
d. light absorption
Where does the splitting of water take place?
a. Stroma
b. Cytoplasm
c. Thylakoid lumen
d. Plasma membrane
What is the first member of TCA cycle?
(1) Acetyl CoA
(2) Oxaloacetate
(3) Citrate
(4) Pyruvate
Oxidation of two molecules of NADH gives
1. 6 ATPs
2. 3 ATPs
3. 1 ATP
4. 2 ATPs
Which of the following complex has got one Domain present in matrix and other domain in the inner membrane?
(1) Complex I
(2) Complex II
(3) Complex III
(4) Complex V
Tripalmitin
Which of the following is wrong?
1. A fatty acid
2. C51 H98 O6
3. C48 H96 O5
4. Both A and C
Which of the following is not a Physiological effect of Gibberellins?
1.Increase in the length of the grape stalks
2.They cause fruits like Apple to elongate and improve it's shape
3.They also delay senescence
4.They can shorten the market period for fruits
Which of the following hormones is synthesised in large amounts by tissues undergoing senescence and ripening fruits?
(1) Ethylene
(2) ABA
(3) Auxins
(4) GA3
Why photorespiration does not take place in C4 plants?
1. Do not contain RuBisCo.
2. Have a mechanism that increases the concentration of CO2 at the enzyme site.
3. Cells do not allow oxygen to accumulate in them.
4. Cells are impermeable to oxygen.
Why breakdown of proton gradient is essential during
photosynthesis
| 1. | It leads to production of NADPH. |
| 2. | It leads to production of O2 |
| 3. | It leads to production of ATP. |
| 4. | Both 1 and 3 |
| Statement - I : | Respiration is amphibolic pathway. |
| Statement - II : | Pure fat & pure protein can never be respiratory substrate. |
Option :
| 1. | Only Statement - II is correct. |
| 2. | Both Statements are correct. |
| 3. | Neither first nor second is correct. |
| 4. | Only first Statement is correct. |
Which of the following is not an action of ethylene in plants?
| 1. | Seedling triple response |
| 2. | Stimulates leaf and flower senescence |
| 3. | Inhibits senescence of mature xylem cells in preparation for plant use |
| 4. | Inhibits shoot growth except in some habitually flooded plants like rice |
The correct sequence of the organelles involved in photorespiration (passage of phosphoglycolate)are:
| 1. | Mitochondria, Peroxisomes and Chloroplasts |
| 2. | Chloroplasts, Peroxisomes and Mitochondria |
| 3. | Chloroplasts, Mitochondria and Peroxisomes |
| 4. | Peroxisomes, Chloroplasts and Mitochondria |
Which of the following plant growth regulators is derived from adenine but does not occur naturally in plants.
1. NAA
2. Zeatin
3. Kinetin
4. 2, 4-D
Phycoerythrin is the major pigment in :
| 1. | Red algae | 2. | Blue-green algae |
| 3. | Green algae | 4. | Brown algae |
In most seeds, the ratio of which of the following pair of PGRs determines whether the embryo remains dormant or germinates?
| 1. | ABA and GA | 2. | Auxin and Cytokinin |
| 3. | Auxin and GA | 4. | ABA and Cytokinin |
| Assertion (A): | The reaction centres in both photosystems in plants is the same. |
| Reason (R): | The reaction centres of both photosystems are formed by chlorophyll b molecules. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
| Assertion (A): | Spraying juvenile conifers with GAs hastens the maturity period. |
| Reason (R): | GAs lead to early seed production in conifers. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Consider the following statements:
I. A motor neuron along with the muscle fibres connected to it constitute a motor unit
II. The neurotransmitter released at the neuro-muscular junction is Acetylcholine.
III. Myosin head has ATPase activity.
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II, and III
Which of the following is dioecious?
1. Taenia
2. Fasciola
3. Ascaris
4. Earthworm
Dry, cornified skin along with epidermal scales or scutes is found in
1. Tortoise
2. Hyla
3. Bufo
4. All of the above
Which of the following is not associated with Impulse transmission across an electrical synapse?
| 1. | Faster than chemical synapse |
| 2. | Rare in our system |
| 3. | No synaptic cleft |
| 4. | Impulse transmission is not similar as that of transmission within a neuron |
Which of the following is the outermost mean of protection for brain?
1. Skull
2. Dura mater
3. Arachnoid
4. Pia mater
Which of the following is not true for cerebrum?
1. Forms the major part of the human brain
2. A deep cleft divides the cerebrum transversely into two halves
3. Cerebral hemispheres are longitudinal halves
4. The hemispheres are connected by a tract of nerve fibres called corpus callosum
Which of the following is not associated with midbrain?
1. Located between thalamus of the forebrain and pons of the hindbrain
2. A canal called the cerebral aqueduct passes through the midbrain
3. The ventral portion of the midbrain made up of mainly of four round swellings (lobes) called Corpora Quadrigemina
4. Midbrain is a part of the brain stem
In the given diagram of the human skull, the bones represented by the letters A, B, and C are respectively:
1. Sphenoid, Ethmoid and Lacrimal
2. Ethmoid, Sphenoid and Lacrimal
3. Sphenoid, Lacrimal and Ethmoid
4. Ethmoid, Lacrimal and Sphenoid
Maturation of lymphocytes takes place in
| 1. | thymus | 2. | Bone marrow |
| 3. | Liver | 4. | Both 1 and 2 |
The system which coordinate and regulate the physiological functions in the body is
| 1. | Nervous System and Excretory system |
| 2. | Endocrine System and Circulatory system |
| 3. | Nervous System and Endocrine System |
| 4. | Excretory and Circulatory system |
Hooks and Suckers are present in
1.All Flatworms
2.Parasitic flatworms
3.Non-parasitic flatworms
4.None of these
Nissl's granules are present in which part of a neuron?
1. Cyton
2. Synaptic knobs
3. Axon
4. Nerve endings
The hormone that reduces the destruction of bones also enhances the deposition of in bones thus making them solid and strong. This hormone is:-
1. Collips hormone
2. Thyrocalcitonin
3. Thyroxine
4. Vasopressin
Which of the following hormone binds with membrane-bound receptors?
1. Thyroxine
2. FSH
3. Estrogen
4. Cortisone
Like other animals with bilateral symmetry, flatworms have:
1. an internal body cavity
2. segmented bodies
3. three germ layers
4. specialized circulatory and respiratory organs
A snake has absence of
| 1. | Ear |
| 2. | External ear |
| 3. | Internal ear and columella auris |
| 4. | Both (2) & (3) |
| COLUMN I | COLUMN II | ||
| A. | Petromyzon | P | A jawless vertebrate |
| B. | Icthyophis | Q | A limbless amphibian |
| C. | Struthio | R | A flightless bird |
| A | B | C | |
| 1. | Q | R | P |
| 2. | Q | P | R |
| 3. | P | Q | R |
| 4. | P | R | Q |
| 1. | It influences menstrual cycle |
| 2. | It does not maintain body temperature |
| 3. | It is also known as vasopressin |
| 4. | Hyperproduction of this hormone causes cretinism |
| Assertion (A): | Ketonuria and glycosuria are the characteristic symptoms of diabetes insipidus |
| Reason (R): | Diabetes insipidus is caused due to deficiency of insulin |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Which one of the following can exhibit cis-trans-isomerism?
1. CH3-CHCl-COOH
2. H-C≡C-Cl
3. Cl-CH=CHCl
4. ClCH2-CH2Cl
A compound of molecular formula C7H16 shows optical isomerism, the compound will be
1. 2,3-dimethyl pentane
2. 2,2-dimethyl butane
3. 2-methyl hexane
4. None of the above
Which of the following is more basic than aniline?
1. Diphenylamine
2. Triphenylamine
3. p-nitroaniline
4. Benzylamine
Which of the following carbanions is the most stable?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Which of the following is the most stable carbonium ion?
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
In the following mechanism, the chain termination step is:
| (i) |  |
| (ii) |  |
| (iii) |  |
| (iv) |  |
1. (i)
2. (ii)
3. (iii)
4. (iv)
Which of the following statements about the inductive effect is correct?
| 1. | The inductive effect transfers electrons from one carbon atom to another. |
| 2. | The inductive effect operates in both \(\sigma\)- and \(\pi\)-bonds. |
| 3. | The inductive effect does not create any charge in the molecule. |
| 4. | The inductive effect creates partial charges and is distance-dependent. |
The IUPAC name of CH3-CH=CH-COOH is-
1. But-1-en-4-oic acid
2. 1-Hydroxybut-2-en-1-one
3. But-2-en-1-oic acid
4. But-2-en-4-oic acid
The IUPAC name of 
1. 2, 3-Dihydroxybutane-1, 4-carboxylic acid
2. 2, 3-Dihydroxybutane-1, 4-dioic acid
3. 1, 2-Dihydroxyethane dicarboxylic acid
4. None of the above
The IUPAC name of the below-given compound is:
1. 2-(Bromomethyl)-3-oxopentane carboxamide
2. 1-Bromo-2-carbamoylpentan-3-one
3. 5-Bromo-4-carbamoylpentan-3-one
4. 2-(Bromomethyl)-3-oxopentanamide
The IUPAC name of the following compound is:
(1) 4-Bromo-3-cyanophenol
(2) 2-Bromo-5-hydroxybenzonitrile
(3) 2-cyano-4-hydroxybromobenzene
(4) 6-Bromo-3-hydroxybenzonitrile
The stability of 2,3-dimethyl but-2-ene is more than 2-butene. This can be explained in terms of:
1. resonance
2. hyperconjugation
3. electromeric effect
4. inductive effect
The types of a functional group can be present in an amine with the formula C3H9N is-
1. 1
2. 2
3. 3
4. 4
Alkyl cyanide \(R-C \equiv N\) and alkyl isocyanides \(R- \overset{+}{N} \equiv C^-\) are:
| 1. | Tautomers | 2. | Metamers |
| 3. | Functional isomers | 4. | Geometrical isomers |
The carbocation among the following that doesn't get stabilized by resonance is :
| 1. | 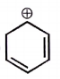 |
2. | 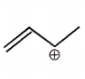 |
| 3. | 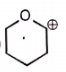 |
4. | 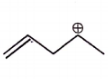 |
Two possible stereo-structures of CH3CHOH.COOH, that are optically active, called:
| 1. | Diastereomers | 2. | Atropisomers |
| 3. | Enantiomers | 4. | Mesomers |
The number of chiral carbon atoms in the given compound is are
1. 2
2. 3
3. 4
4. 1
What is the hybridization type of the \(\mathrm{C}_{4}- \mathrm{C}_5\) bond in the compound CH 2=CH-CH2-CH2-C≡CH?
1.
2.
3.
4.
The enolic form of an acetone contains:
1. 9 sigma bonds, 1 pi bond, and 2 lone pairs of electrons
2. 8 sigma bonds, 2 pi bonds, and 2 lone pairs of electrons
3. 10 sigma bonds, 1 pi bond, and 1 lone pair of electrons
4. 9 sigma bonds, 2 pi bonds, and 1 lone pair of electrons
Which of the following compounds will exhibit geometrical isomerism?
1. 1-Phenyl-2-butene
2. 3-Phenyl-1-butene
3. 2-Phenyl-1-butene
4. 1, 1-Dipheny]-1 -propene
Five molecules of a gas have speeds, 1, 2, 3, 4 and 5 km. The value of the root mean square speed of the gas molecules is:
1. 3 km
2. km
3. km
4. 3.5 km
Which one of the following graph is correct at constant pressure?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
A given mass of gas expands from state \(A\) to state \(B\) by
three paths \(1, 2~\text{and}~3\), as shown in the figure. If \(W_1, W_2~\text{and}~W_3\) respectively be the work done by the gas along the three paths, then:

| 1. | \(W_1 >W_2>W_3\) | 2. | \(W_1<W_2<W_3\) |
| 3. | \(W_1 =W_2=W_3\) | 4. | \(W_1 <W_2=W_3\) |
An ideal gas with adiabatic exponent y is heated at constant pressure and it absorbs Q heat. What fraction of this heat is used to perform external work?
1.
2.
3.
4.
Each of the two strings of length 51.6 cm and 49.1 cm are tensioned separately by 20N force. Mass per unit length of both the strings is same and equal to 1 When both the strings vibrate simultaneously the number of beats is :
1. 5
2. 7
3. 8
4. 3
A point performs simple harmonic oscillation of period T and the equation of motion is given by x= a sin .After the elapse of what fraction of the time period the velocity of the point will be equal to half to its maximum velocity?
1.
2.
3.
4
A plane wave is represented by
Where x and y are distances measured along in x and y direction in meters and t is time in seconds. This wave has
(1) A wavelength of 0.25 m and travels in + ve x-direction
(2) A wavelength of 0.25 m and travels in + ve y-direction
(3) A wavelength of 0.5 m and travels in – ve y-direction
(4) A wavelength of 0.5 m and travels in – ve x-direction
The length of two open organ pipes are l and respectively. Neglecting end correction, the frequency of beats between them will be approximate :
(1)
(2)
(3)
(4)
(Here v is the speed of sound)
The frequency of a stretched uniform wire under tension is in resonance with the fundamental frequency of a closed tube. If the tension in the wire is increased by 8 N, it is in resonance with the first overtone of the closed tube. The initial tension in the wire is
(1) 1 N
(2) 4 N
(3) 8 N
(4) 16 N
A metal wire of linear mass density of 9.8 g/m is stretched with a tension of 10 kg weight between two rigid supports 1 metre apart. The wire passes at its middle point between the poles of a permanent magnet, and it vibrates in resonance when carrying an alternating current of frequency n. The frequency n of the alternating source is :
(1) 25 Hz
(2) 50 Hz
(3) 100 Hz
(4) 200 Hz
If denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal PV curves at their point of intersection is
(1)
(2)
(3)
(4)
In an adiabatic expansion of a gas, if the initial and final temperatures are \(T_1\) and \(T_2\), respectively, then the change in internal energy of the gas is:
1. \(\frac{nR}{\gamma-1}(T_2-T_1)\)
2. \(\frac{nR}{\gamma-1}(T_1-T_2)\)
3. \(nR ~(T_1-T_2)\)
4. Zero
A diatomic gas initially at 18°C is compressed adiabatically to one-eighth of its original volume. The temperature after compression will be
(1) 10°C
(2) 887°C
(3) 668 K
(4) 144°C
The equation for an ideal gas is PV = RT, where V represents the volume of
1. 1 gm gas
2. Any mass of the gas
3. One gm mol gas
4. One litre gas
A vessel is partitioned in two equal halves by a fixed diathermic separator. Two different ideal gases are filled in left (L) and right (R) halves. The rms speed of the molecules in L part is equal to the mean speed of molecules in the R part. Then the ratio of the mass of a molecule in L part to that of a molecule in R part is
1.
2.
3.
4.
40 calories of heat is needed to raise the temperature of 1 mole of an ideal monoatomic gas from 20°C to 30°C at a constant pressure. The amount of heat required to raise its temperature over the same interval at a constant volume is
1. 20 calorie
2. 40 calorie
3. 60 calorie
4. 80 calorie
Two sound waves with wavelengths \(5.0~\text{m}\) and \(5.5~\text{m}\), respectively, propagate in gas with a velocity of \(330~\text{m/s}\). How many beats per second can we expect?
1. \(12\)
2. \(0\)
3. \(1\)
4. \(6\)
Maximum velocity in SHM is . The average velocity during the motion from one extreme point to the other extreme point will be
1.
2.
3.
4.
A particle under SHM takes 1.2 s to complete one vibration. Minimum time taken by it to travel from mean position to half of its amplitude is
1. 0.2 s
2. 0.1 s
3. 0.4 s
4. 0.3 s
A particle executes linear oscillation such that its epoch is zero. The ratio of the magnitude of its displacement in 1st second and 2nd second is (Time period = 12 seconds)
1.
2.
3.
4.