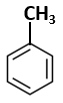
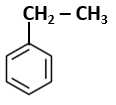
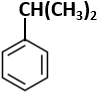
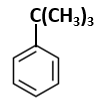
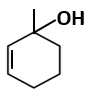
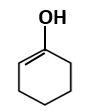
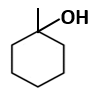
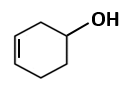
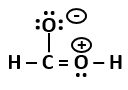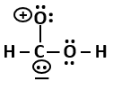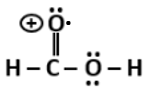Botany - Section A
1. At the root tip, some of the epidermal cells form very fine and delicate, thread-like structures called root hairs in the region of:
| 1. |
Root cap |
2. |
Meristematic activity |
| 3. |
Elongation |
4. |
Maturation |
2. A plasmid has genes for resistance against ampicillin and tetracycline. The tetracycline resistant gene is insertionally inactivated by inserting a foreign DNA within the gene. The bacterial cells are transformed. The recombinant transformants:
| 1. |
will be killed in a medium containing ampicillin but not in a medium containing tetracycline |
| 2. |
will be killed in a medium containing tetracycline but not in a medium containing ampicillin |
| 3. |
will be killed in a medium containing ampicillin and in a medium containing tetracycline |
| 4. |
will not be killed in a medium containing ampicillin and in a medium containing tetracycline |
3. Consider the two statements:
| Statement I: |
Plant growth and development is intimately linked to the water status of the plant. |
| Statement II: |
The plant cells grow in size by cell enlargement which in turn requires water. |
| 1. |
Statement I is correct; Statement II is correct |
| 2. |
Statement I is incorrect; Statement II is correct |
| 3. |
Statement I is correct; Statement II is incorrect |
| 4. |
Statement I is incorrect; Statement II is incorrect |
4. The term used to refer to the use of bio-resources by multinational companies and other organisations without proper authorisation from the countries and people concerned without compensatory payment is:
1. Bioprospecting
2. Biopiracy
3. Biomagnification
4. Bioaccumulation
5. Consider the given two statements:
| Assertion (A): |
In order to link the alien DNA, the vector needs to have very few, preferably single, recognition sites for the commonly used restriction enzymes. |
| Reason (R): |
Presence of more than one recognition sites within the vector will generate several fragments, which will complicate the gene cloning. |
| 1. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 2. |
(A) is True; (R) is False |
| 3. |
(A) is False; (R) is False |
| 4. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
6. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
The reaction center of PS I is known as P700 and the reaction center of PS II is known as P680. |
| Reason (R): |
PS I functions first in the light reactions. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
7. Splitting of centromere occurs in:
| 1. |
Mitotic anaphase and Anaphase II |
| 2. |
Mitotic anaphase and Anaphase I |
| 3. |
Mitotic metaphase and Metaphase II |
| 4. |
Mitotic metaphase and Metaphase I |
8. In Polymerase chain reaction:
| 1. |
the correct chronological sequence of steps is: Annealing of primer → Denaturation → Extension. |
| 2. |
Taq polymerase is an RNA polymerase derived from a fungus. |
| 3. |
millions to billions of copies of a specific DNA sample are made. |
| 4. |
viral antigens cannot be detected in a given sample. |
9. Plant species pollinated by water fall into two categories:
| (i) |
Those that distribute their pollen to the surface of water |
| (ii) |
Those that distribute their pollen beneath the surface of water |
The correct examples of the two categories will be:
|
(i) |
(ii) |
| 1. |
Vallisneria
|
Sea grasses
|
|
2.
|
Water hyacinth
|
Sea grasses
|
|
3.
|
Sea grasses
|
Water lily
|
|
4.
|
Sea grasses
|
Vallisneria
|
10. In a monocot seed, the aleurone layer describes:
1. a proteinaceous outermost layer of the endosperm
2. the shield shaped cotyledon
3. sheath enclosing the plumule
4. sheath enclosing the radicle
11. The decline in the population of Indian native fishes due to the introduction of Clarias gariepinus in river Yamuna can be categorised as:
1. Co-extinction
2. Habitat fragmentation
3. Over exploitation
4. Alien species invasion
12. The somatic cell of an organism has a diploid chromosome number of 16 [2N = 16]. The number of the chromosomes in this cell at the end of the S-phase will be:
13. Who popularised the term ‘Biodiversity’ to describe the combined diversity at all levels of biological organisation?
1. Robert May
2. Paul Ehrlich
3. Edward Wilson
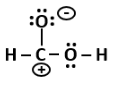
4. Alexander von Humboldt
14. Identify the incorrect statement:
| 1. |
Any calculations of energy content, biomass or numbers, need not include all organisms at that trophic level. |
| 2. |
No generalisations we make will be true if we take only a few individuals at any trophic level into account. |
| 3. |
A given organism may occupy more than one trophic level simultaneously. |
| 4. |
The trophic level represents a functional level, not a species as such. |
15. What is the correct order of the steps involved in the sexual cycle of organisms belonging to the Kingdom Fungi?
| 1. |
Meiosis in zygote — Plasmogamy — Karyogamy |
| 2. |
Karyogamy — Meiosis in zygote — Plasmogamy |
| 3. |
Plasmogamy — Karyogamy — Meiosis in zygote |
| 4. |
Karyogamy — Plasmogamy — Meiosis in zygote |
16. Agrobacterium tumefaciens infects the plant through its:
| 1. |
Chromosomal DNA |
2. |
Complementary DNA |
| 3. |
F plasmid |
4. |
Ti plasmid |
17. The correct sequence of layers [from the outside to the inside] of a mature woody stem will be:
1. Periderm – Vascular cambium – Phloem – Xylem
2. Periderm – Phloem – Vascular cambium – Xylem
3. Phellem – Phellogen – Phelloderm – Xylem – Phloem
4. Phellem – Phelloderm – Phellogen – Phloem – Xylem
18. Which of the following is true regarding the classes of Bryophytes?
| 1. |
The thallus of mosses is dorsiventral and closely appressed to the surface |
| 2. |
In the life cycle of liverworts, the predominant gametophytic stage is divided into two stages - the protonema and the leafy stage |
| 3. |
The sporophyte in liverworts is more elaborate than in mosses |
| 4. |
Asexual reproduction in liverworts takes place by fragmentation of thalli, or by the formation of specialised structures called gemmae |
19. Consider the given two statements:
| Statement I: |
Pollination plays a critical role in the sexual reproduction in flowering plants. |
| Statement II: |
As both types of gametes are non-motile, they have to be brought together for fertilisation to occur. |
| 1. |
Statement I is correct; Statement II is correct |
| 2. |
Statement I is incorrect; Statement II is correct |
| 3. |
Statement I is correct; Statement II is incorrect |
| 4. |
Statement I is incorrect; Statement II is incorrect |
20. Primary xylem is of two types – protoxylem and metaxylem.
| Statement I: |
In roots, the protoxylem lies towards the center (pith) and the metaxylem lies towards the periphery of the organ. This type of primary xylem is called endarch. |
| Statement II: |
In stems, the protoxylem lies towards the periphery and the metaxylem lies towards the center. Such an arrangement of the primary xylem is called an exarch. |
1.
Statement I is correct;
Statement II is correct
2.
Statement I is incorrect;
Statement II is correct
3.
Statement I is correct;
Statement II is incorrect
4.
Statement I is incorrect;
Statement II is incorrect
21. Regarding factors affecting enzyme activity:
| I: |
Low temperature preserves the enzyme in a temporarily inactive state whereas high temperature destroys enzymatic activity. |
| II: |
With the increase in substrate concentration, the velocity of the enzymatic reaction rises at first. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
22. Identify the correct statement:
| 1. |
α amino acids are substituted methanes |
| 2. |
Only triglycerides are lipids that are both macromolecules as well as polymers |
| 3. |
Cellulose forms secondary helical structures and can hold iodine molecules in the helical portion |
| 4. |
Every virus will have both DNA and RNA as its genetic material |
23. “Bakane” – “Foolish seedling" disease in rice is caused by:
| 1. |
auxins |
2. |
gibberellins |
| 3. |
cytokinins |
4. |
ethylene |
24. Western Ghats have a large number of plant and animal species that are not found anywhere else. Which of the following terms will you use to notify such species?
| 1. |
Endemic |
2. |
Vulnerable |
| 3. |
Threatened |
4. |
Keystone |
25. The hilum in a typical angiosperm ovule represents the junction between -
1. Interguments and the embryo sac.
2. Embryo sac and the nucellus
3. Body of the ovule and the funicle
4. Nucellus and the funicle
26. In which of the following class of fungi, sex organs are absent and plasmogamy is brought about by fusion of two vegetative or somatic cells of different strains or genotypes?
1. Ascomycetes
2. Phycomycetes
3. Basidiomycetes
4. Deuteromycetes
27. For every CO
2 molecule entering the Calvin cycle:
| 1. |
3 molecules of ATP and 3 of NADPH are required. |
| 2. |
3 molecules of ATP and 2 of NADPH are required. |
| 3. |
3 molecules of ATP and 3 of NADPH are required. |
| 4. |
2 molecules of ATP and 2 of NADPH are required |
28. Identify the incorrect statement:
| 1. |
Somatic hybridization has been used to produce pomato, though it is not used commercially |
| 2. |
Methylophilus methylotropus [bacterium] and Spirulina [blue-green algae] are sources of single-cell protein |
| 3. |
Plant cells do not show cellular totipotency and hence cannot be cultured |
| 4. |
Gamma radiations are used to induce mutations in plants |
29. Apomictic embryos in Citrus arise from:
| 1. |
diploid Egg |
2. |
synergids |
| 3. |
nucellus |
4. |
antipodal cells |
30. Consider the given two statements:
| Assertion (A): |
Endosperm tissue of angiosperms can be a source of nutrition in animal diet. |
| Reason (R): |
Wheat endosperm is ground into flour for bread, while barley endosperm is the main source of sugars for beer production. |
| 1. |
Both (A) and (R) are True and (R) explains (A) |
| 2. |
Both (A) and (R) are True and (R) does not explain (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
(A) is False but (R) is True |
31. Match each item in Column I with one in Column II and choose the correct option from the codes given below:
|
Column I |
|
Column II |
| A. |
Psilopsida |
P. |
Selanginella |
| B. |
Lycopsida |
Q. |
Adiantum |
| C. |
Sphenopsida |
R. |
Equisetum |
| D. |
Pteropsida |
S. |
Psilotum |
|
A |
B |
C |
D |
| 1. |
R |
Q |
S |
P |
| 2. |
S |
P |
R |
Q |
| 3. |
P |
S |
Q |
R |
| 4. |
Q |
R |
P |
S |
32. Match the stage of Prophase I of Meiosis I given in
Column-I with the corresponding event in
Column-II and select the correct match from the codes given below:
|
Column-I |
|
Column-II |
| A. |
Zygotene |
P. |
Crossing over |
| B. |
Pachytene |
Q. |
Synapsis |
| C. |
Diplotene |
R. |
Terminalization of chiasmata |
| D. |
Diakinesis |
S. |
Appearance of chiasmata |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
P |
S |
R |
| 2. |
P |
Q |
S |
R |
| 3. |
P |
Q |
R |
S |
| 4. |
Q |
P |
R |
S |
33. Which of the following is matched incorrectly:
1. Mangifera : indica
2. Panthera : tuberosum
3. Solanum : melongena
4. Solanum : nigrum
34. Identify the incorrect match:
|
mRNA codon |
Amino acid |
| 1. |
AUG |
Methionine |
| 2. |
GGG |
Glycine |
| 3. |
UGA |
Tryptophan |
| 4. |
UAG |
None; it is a stop codon |
35. Identify the incorrectly matched pair:
| 1. |
Primary constriction |
Satellite chromosome |
| 2. |
Cilia/flagella |
9 + 2 arrangement of microtubules |
| 3. |
Centriole |
9 + 0 arrangement of microtubules |
| 4. |
Lysosomes |
Acid hydrolases |
Botany - Section B
36. Consider the two statements:
| I: |
Predators in nature are ‘prudent’. |
| II: |
Prey species have evolved defenses to lessen the impact of predation. |
1. Both
I and
II are correct and
II explains
I.
2. Both
I and
II are correct but
II does not explain
I.
3. Only
I is correct.
4. Both
I and
II are incorrect.
37. The primary role in abscission in majority of the plants is played by:
1. ABA and ethylene
2. Auxin and ethylene
3. Auxin and ABA
4. Cytokinin and auxin
38. What is the total number of NADH and molecules produced for each molecule of glucose metabolized by glycolysis and the citric acid cycle?
39. A heterozygous pea plant with violet flowers was crossed with a homozygous pea plant with white flowers. Violet is dominant over white. Which one of the following represents the expected combinations among 40 progenies formed?
1. 30 produced violet and 10 produced white flowers
2. 20 produced violet and 20 produced white flowers.
3. All 40 produced violet flowers
4. All 40 produced white flowers
40. Plants restore to the air whatever breathing animals and burning candles remove, it was hypothesised by:
1. Cornelius van Niel
2. Joseph Priestley
3. Julius von Sachs
4. T.W. Engelmann
41. Integuments of an ovule encircle the nucellus except at the tip where a small opening is organised, called the:
| 1. | germ pore | 2. | chalaza |
| 3. | micropyle | 4. | filiform apparatus |
42. Regarding cell membrane:
| I: |
The lipid content mainly is phosphoglycerides |
| II: |
Proteins can move laterally within the overall bilayer |
| III: |
The membrane is selectively permeable |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
43. Consider the given two statements:
| Assertion (A): |
Global efforts to conserve biodiversity largely concentrate on the hot biodiversity spots located all over the world. |
| Reason (R): |
Biodiversity hotspots’ regions have very high levels of species richness and high degree of endemism. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
44. Which of the following biomolecule is not a biomacromolecule but present in the acid-insoluble fraction
1. Protein
2. Lipid
3. Nucleic acid
4. Polysaccharide
45. Kingdom Protista has brought together Chlamydomonas, Chlorella with Paramoecium and Amoeba. On what basis were these organisms separated under previous classification systems?
1. Cell wall
2. Cell type
3. Body organisation
4. Mode of nutrition
46. The tRNA molecule:
| 1. |
looks like an inverted L in its secondary structure |
| 2. |
is the largest of the major cellular RNAs |
| 3. |
acts as an adapter molecule |
| 4. |
has catalytic functions |
47. The large holes in ‘Swiss cheese’ are due to production of a large amount of CO2 by a bacterium named:
1. Staphylococcus aureus
2. Propionibacterium shermanii
3. Penicillium roqueforti
4. Bacillus subtilis
48. The following diagram shows the biome distribution with respect to annual temperature and precipition. Grassland, Temperate forest and Coniferous forest are represented respectively by:

1. A, C and F
2. B, D and E
3. B, E and F
4. A, D and F
49. Nitrogen is often a limiting nutrient in terrestrial ecosystems because:
| 1. |
the percentage of nitrogen in the atmosphere is low |
| 2. |
atmospheric nitrogen is primarily in the stratosphere |
| 3. |
dinitrogen cannot be used directly by the organisms |
| 4. |
there are regional fluctuations in nitrogen levels in the atmosphere |
50. In a plant cell:
| 1. |
there are multiple vacuoles, each small in size |
| 2. |
centrioles play prominent role in organization of microtubules |
| 3. |
cytokinesis occurs through cleavage furrow |
| 4. |
Secondary cell wall is formed on the inner [towards membrane] side of the cell |
Zoology - Section A
51. Which of the following sexually transmitted infection is caused by a bacterium and is curable if diagnosed early?
| 1. |
Syphilis |
2. |
HIV |
| 3. |
Genital herpes |
4. |
Trichomoniasis |
52. The karyotype of a person suffering from Down’s syndrome will show:
| 1. |
44 Autosomes + XXY |
| 2. |
44 Autosomes + XO |
| 3. |
45 Autosomes [trisomy #21] + XY |
| 4. |
43 Autosomes [monosomy #21] + XY |
53. Streptokinase is a:
1. thrombolytic medication and enzyme
2. thrombolytic medication and an ion channel blocker
3. anticoagulant and an anti-platelet agent
4. anticoagulant and vasodilator
54. The sporozoite of the malarial parasite is present in ----
| 1. |
saliva of infected female Anopheles mosquito. |
| 2. |
RBC of human suffering from malaria. |
| 3. |
Spleen of infected humans. |
| 4. |
Gut of female Anopheles mosquito. |
55. Which of the following is not aneuploidy?
1. Down’s syndrome
2. Klinefelter’s syndrome
3. Turner’s syndrome
4. Phenylketonuria
56. Blood calcium level can be increased by the administration of?
1. glucagon
2. parathormone
3. thyroxine
4. calcitonin
57. Identify the incorrect statement:
| 1. |
Metamerism appeared for the first time in annelida |
| 2. |
Arthropods have jointed appendages and chitinous exoskeleton |
| 3. |
Reptiles are endotherms |
| 4. |
Forelimbs of birds are modifies into wings |
58. On an average a female cockroach produces:
1. 9-10 oothecae , each containing 14-16 eggs
2. 14-16 oothecae , each containing 9-10 eggs
3. 5-6 oothecae , each containing 9-10 eggs
4. 9-10 oothecae , each containing 5-6 eggs
59. Generally, the shape not seen in a bacterial cell is:
1. Rod like
2. Comma shaped
3. Spherical
4. Icosahedral
60. How many of the given statements are correct?
| I: |
The left and right cerebral hemispheres are connected by a tract of nerve fibres called corpus spongiosum. |
| II: |
Association areas are neither clearly sensory nor motor in function. |
| III: |
Thalamus is a major coordinating centre for sensory and motor signaling. |
| IV: |
The hindbrain comprises pons, cerebellum and medulla. |
| V: |
Three major regions make up the brain stem: mid brain, pons and medulla oblongata. |
61. Angiotensin:
| I: |
is a vasoconstrictor |
| II: |
stimulates the adrenal cortex to secrete aldosterone |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
62. Persons with AB blood type can:
| 1. |
donate blood to persons with all blood types as neither A nor B antigens are present on their RBCs |
| 2. |
receive blood from persons with all blood types as both A and B antigens are present on their RBCs |
| 3. |
receive blood from persons with all blood types as neither A nor B antigens are present on their RBCs |
| 4. |
receive blood from persons with all blood types as neither anti-A nor anti-B antibodies are present on his blood plasma |
63. Identify the incorrect statement regarding thalassemia:
| 1. |
There is reduced synthesis of one of the globin chains. |
| 2. |
It is an autosome-linked dominant blood disease. |
| 3. |
Alpha thalassemia is controlled by two closely linked genes on chromosome 16. |
| 4. |
Beta thalassemia is controlled by a single gene on chromosome 11. |
64. Foetal ejection reflex is initiated by:
| 1. |
Foetal hypothalamus |
| 2. |
Maternal pituitary |
| 3. |
Fully developed foetus and placenta |
| 4. |
Maternal hypothalamus |
65. The given age pyramid represents a:

| 1. |
Fast expanding population |
2. |
Slowly expanding population |
| 3. |
Stable population |
4. |
Declining population |
66. Identify the incorrect statement regarding transgenic animals:
| 1. |
α-1 antitrypsin is being produced in a transgenic sheep |
| 2. |
The first transgenic cow, Rosie, produced human protein-enriched milk (2.4 grams per litre). |
| 3. |
Transgenic mice are being used to test the safety of the polio vaccine. |
| 4. |
Over 95 percent of all existing transgenic animals are cattle. |
67. A characteristic 9 + 2 arrangement of axonemal microtubules is seen in:
1. Cilia and flagella
2. Centriole
3. Filaments in axons of neurons
4. Actin myofilaments
68. The first restriction endonuclease, whose functioning depended on a specific DNA nucleotide sequence was:
| 1. |
EcoR I |
2. |
BamH I |
| 3. |
Hind II |
4. |
Sma I |
69. After getting into the body of the person, the first cells that HIV uses as HIV factory are:
1. T cytotoxic cells
2. T helper cells
3. Macrophages
4. Dendritic cells
70. Urea can be used in humans to increase the osmolarity primarily in:
| 1. |
Blood |
2. |
Renal cortex |
| 3. |
Renal medulla |
4. |
Urinary bladder |
71. In a continuous culture system:
| I: |
The used medium is drained out from one side while the fresh medium is added from the other. |
| II: |
The cells are maintained in their physiologically most active log/exponential phase. |
| III: |
A larger biomass is produced leading to higher yields of desired proteins. |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
72. Which age group is considered as the most vulnerable for the development of drug addiction?
| 1. |
Pre-pubertal |
2. |
Adolescence |
| 3. |
Late adulthood |
4. |
Geriatric |
73. By process of leaching during decomposition of detritus, water soluble :
| 1. |
organic nutrients go down the soil horizon and get precipitated as unavailable salts |
| 2. |
inorganic nutrients go down the soil horizon and get precipitated as unavailable salts |
| 3. |
organic nutrients go down the soil horizon and get precipitated as available salts |
| 4. |
inorganic nutrients go down the soil horizon and get precipitated as available salts |
74. The length of which of the following structures remains same during muscle contraction by sliding filament mechanism?
| 1. |
H zone |
2. |
A band |
| 3. |
I band |
4. |
Sarcomere |
75. In the human genome:
| 1. |
the total number of genes are about 60000 |
| 2. |
less than 2 percent of genome does not code for proteins |
| 3. |
repeated sequences are rarely found |
| 4. |
SNPa occur at about 1.4 million locations |
76. Communication junctions called as intercalated discs are seen in:
1. Cardiac muscle
2. Bone
3. Cartilage
4. Neuron
77. Match List-I with List-II
| List-I |
List-II |
| (a) Filariasis |
(i) Haemophilus influenzae |
| (b) Amoebiasis |
(ii) Trichophyton |
| (c) Pneumonia |
(iii) Wuchereria bancrofti |
| (d) Ringworm |
(iv) Entamoeba histolytica |
Choose the correct answer from the options given below.
|
(a) |
(b) |
(c) |
(d) |
| 1. |
(i) |
(ii) |
(iv) |
(iii) |
| 2. |
(ii) |
(iii) |
(i) |
(iv) |
| 3. |
(iv) |
(i) |
(iii) |
(ii) |
| 4. |
(iii) |
(iv) |
(i) |
(ii) |
78. Fimbriae in bacterial cells:
| 1. |
play an important role in motility |
| 2. |
attach the cell to the rocks in a stream and also to the host tissues |
| 3. |
provide surface area for the location of enzymes |
| 4. |
form a tube like structure for passage of DNA during bacterial conjugation |
79. If the gene coding for permease enzyme in the lac operon is inactivated by a mutation:
| 1. |
the cell will not be able to metabolize glucose |
| 2. |
the cell will not be able to breakdown lactose |
| 3. |
lactose will not be able to enter the cell |
| 4. |
the catabolite activator protein will increase the rate of transcription of lac operon |
80. The members of which of the following phylum are bilaterally symmetrical, triploblastic and pseudocoelomates?
| 1. |
Platyhelminthes |
2. |
Annelids |
| 3. |
Mollusc |
4. |
Aschelminthes |
81. The binding of Hb to oxygen is primarily related to
1. Partial pressure of oxygen
2. Partial pressure of carbon dioxide
3. Hydrogen ion concentration
4. Temperature
82. The amount of CO2 that can diffuse through the diffusion membrane per unit difference in partial pressure is much higher as compared to that of O2. This is because:
| 1. |
Solubility coefficient of CO2 is higher |
| 2. |
Solubility coefficient of CO2 is lesser |
| 3. |
Amount of gases in blood is independent of partial pressures of the gases in the atmosphere |
| 4. |
Arterial blood contains more O2 than CO2 |
83. Identify the correct statement:
| 1. |
In spermiogenesis, spermatids are formed, while in spermiation, spermatozoa are formed. |
| 2. |
Spermatogonia are diploid and primary spermatocytes are haploid. |
| 3. |
Testes descend into the scrotum after birth but before puberty while ovaries do not descend at all. |
| 4. |
The Infundibulum of fallopian tube possesses fimbriae while fertilization occurs in the ampulla of the fallopian tube. |
84. Which vitamin, important for effective erythropoiesis, is added by LAB when it is used to convert milk into curd?
1. Vitamin D
2. Vitamin A
3. Vitamin B12
4. Vitamin E
85. A method used in molecular biology for the detection of a specific DNA sequence in DNA samples is:
| 1. |
Northern blotting |
2. |
Western blotting |
| 3. |
Southern blotting |
4. |
Eastern blotting |
Zoology - Section B
86. The bacterium
Escherichia coli was not used by:
| 1. |
Fred Griffith, when he discovered bacterial transformation |
| 2. |
Alfred Hershey and Martha Chase, when they conclusively proved that DNA is the molecule that stores genetic information |
| 3. |
Matthew Meselson and F. Stahl, when they experimentally proved that DNA replicates semi-conservatively |
| 4. |
Francois Jacob and Jacques Monod, when they proposed the working of lactose operon |
87. The Monarch butterfly is highly distasteful to its predator (bird) because:
| 1. |
it accumulates TMAO as an osmolyte. |
| 2. |
it accumulates silica on its wings. |
| 3. |
the butterfly acquires a chemical during its caterpillar stage by feeding on a poisonous weed. |
| 4. |
it is covered with toxin mucilage. |
88. The cumulative thickness of the three layers of the diffusion [respiratory] membrane is:
1. less than 1 µm
2. less than 1mm
3. between 2 µm and 5 µm
4. between 2 mm and 5 mm
89. What prevents the development of new follicles during the post-ovulatory luteal phase of the menstrual cycle?
| 1. |
A very low level of progesterone and oestrogen secretion by the corpus luteum |
| 2. |
Secretion of high levels of FSH by the corpus luteum |
| 3. |
LH surge just before ovulation |
| 4. |
Low levels of both FSH and LH levels |
90. Genes that code for a pair of contrasting traits or slightly different forms of the same gene are known as:
| 1. |
Alleles |
2. |
Loci |
| 3. |
Cistrons |
4. |
Introns |
91. The coxal bone in the human skeleton:
| 1. |
is a part of the pectoral girdle, has a glenoid cavity that articulates with the head of the femur. |
| 2. |
is a part of the pelvic girdle, has an acetabulum that articulates with the head of the humerus. |
| 3. |
is a part of the pelvic girdle and is formed by the fusion of three bones. |
| 4. |
is a part of pectoral girdle and is formed by the fusion of three bones. |
92. Match the pathogens that can cause disease in humans in
Column-I with their correct biological names in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| a. |
Filariasis |
(i) |
Haemophilus influenza |
| b. |
Amoebiasis |
(ii) |
Trichophyton |
| c. |
Pneumonia |
(iii) |
Wuchereria bancrofti |
| d. |
Ringworm |
(iv) |
Entamoeba histolytica |
|
(a) |
(b) |
(c) |
(d) |
| 1. |
(i) |
(ii) |
(iv) |
(iii) |
| 2. |
(ii) |
(iii) |
(i) |
(iv) |
| 3. |
(iv) |
(i) |
(iii) |
(ii) |
| 4. |
(iii) |
(iv) |
(i) |
(ii) |
93. The volume of blood pumped out by each ventricle per minute is called as the:
1. stroke volume
2. ejection fraction
3. cardiac output
4. end diastolic volume
94. In an operon:
| I: |
a polycistronic structural gene is regulated by a common promoter and regulatory genes |
| II: |
the regulation is always negative and inducible |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
95. Identify the incorrect statement regarding fallopian tubes:
| 1. |
Infundibulum possesses fimbriae |
| 2. |
Fertilization takes place in the narrowest part of fallopian tube called ampulla |
| 3. |
Isthmus joins the uterus |
| 4. |
It is lined with ciliated epithelium |
96. Some hormones can act on their target cells through second messengers. Identify the one that does not:
| 1. |
cortisol |
2. |
adrenaline |
| 3. |
FSH |
4. |
calcitonin |
97. Identify the incorrectly match pair:
| 1. |
RuBisCO |
Most abundant protein in the whole of the biosphere |
| 2. |
Insulin |
A polymer of fructose |
| 3. |
Vinblastin |
A secondary metabolite used as a drug |
| 4. |
Adenine |
A substituted purine |
98. The trachea is surrounded by 16 to 20 incomplete and C-shaped rings of:
1. Hyaline cartilage
2. Elastic cartilage
3. Fibrous cartilage
4. Calcified cartilage
99. Which of the following part of human brain is also called emotional brain?
1. Corpus callosum
2. Limbic system
3. Epithalamus
4. Broca's area
100. According to the 2017 MTP Amendment, the maximum gestation age till which MTP is allowed is:
1. 16 weeks
2. 20 weeks
3. 24 weeks
4. 28 weeks
Chemistry - Section A
101. Which is not a conjugate acid/base pair?
1. H
2CO
3 and CO
32-
2. HSO
4- and SO
42-
3. H
2PO
4- and HPO
42-
4. H
3O
+ and H
2O
102. The three cells with their \(E^\circ_{\text{(cell)}}\) values are given below:
|
Cells |
\(E^\circ_{\text{(cell)}}/V\) |
| (a) |
Fe|Fe2+||Fe3+|Fe |
0.404 |
| (b) |
Fe|Fe2+||Fe3+, Fe2+|Pt |
1.211 |
| (c) |
Fe|Fe3+||Fe3+, Fe2+|Pt |
0.807 |
The standard Gibbs free energy change values for three cells are, respectively:
(F represents the charge on 1 mole of electrons.)
| 1. |
–1.212 F, –1.211 F, –0.807 F |
| 2. |
+2.424 F, +2.422 F, +2.421 F |
| 3. |
–0.808 F, –2.422 F, –2.421 F |
| 4. |
–2.424 F, –2.422 F, –2.421 F |
103. An ideal solution is made by mixing 2 moles of benzene
\((P^o = 266 ~mm) \) and 3 moles of another liquid
\((P^o= 236 ~mm) \).
The total vapor pressure of the solution at the same temperature would be:
| 1. |
502 mm |
2. |
248 mm |
| 3. |
600 mm |
4. |
298 mm |
104. The angular shape of ozone molecule (O3) consists of:
1. 1 sigma and 2 pi bonds
2. 2 sigma and 2 pi bonds
3. 1 sigma and 1 pi bonds
4. 2 sigma and 1 pi bonds
105. Which of the following statements is/are incorrect?
| 1. |
When CO is converted into CO+ then bond length of CO decreases |
| 2. |
When O2 is converted into \(O^+_2\) then bond length of O2 increases |
| 3. |
When N2 is converted into \(N^+_2\) then bond length of N2 increases |
| 4. |
Both (1) and (2) |
106. The set of molecules having zero dipole moment is:
1. \(\mathrm{NF}_3, \mathrm{CO}_2, \mathrm{CCl}_4 \)
2. \(\mathrm{BF}_3, \mathrm{H}_2 \mathrm{S}, \mathrm{CCl}_4 \)
3. \(\mathrm{BF}_3, \mathrm{CO}_2, \mathrm{CCl}_4\)
4. \(\mathrm{BF}_3, \mathrm{CO}_2, \mathrm{CHCl}_3 \)
107. Which of the compounds with molecular formula C5H10 yields acetone on ozonolysis?
1. 2-methyl-2-butene
2. 2-methyl-1-butene
3. Cyclopentane
4. 3-methyl-1-butene
108. The number of moles of
\(K_2Cr_2O_7\) required to oxidise one mole of
\(FeSO_4\) in acidic medium is:
| 1. |
6 |
2. |
\(1 \over 2\) |
| 3. |
\(1 \over 3\) |
4. |
\(1 \over 6\) |
109. The best reducing agent among the following is:
1. \(GeCl_2\)
2. \(SnCl_2\)
3. \(PbCl_2\)
4. \(GeCl_4\)
110. Which of the following statements is not correct regarding galvanic cells?
| 1. |
Oxidation occurs at the anode. |
| 2. |
Ions carry current inside the cell. |
| 3. |
Electrons flow in the external circuit from cathode to anode. |
| 4. |
When the cell potential is positive, the cell reaction is spontaneous. |
111. The X and A in the following reaction is:
CH3–CH2–Cl + Ethanolic NaCN→ X \(\xrightarrow[]{A}\) CH3–CH2–CH2–NH2
1. X = CH3CH2CN; A = LiAlH4
2. X = CH3CH2NC; A = KMnO4
3. X = CH3CH2CN; A = CrO3
4. X = CH3CH2NC; A = LiAlH4
112. Consider the given reaction:
\(2A(g) + B(g) + C(s) \rightarrow 2D(g) \)
For this reaction,
\(\Delta \mathrm{U}^{\circ}=-10.5 \mathrm{~kJ} \text { and } \Delta \mathrm{S}^{\circ}=-44.1 \mathrm{JK}^{-1} \)
The value of
\(\Delta G^o \) for the above reaction at 25 °C will be:
| 1. |
1.2 kJ |
2. |
0.16 kJ |
| 3. |
3.2 kJ |
4. |
1.6 kJ |
113. The correct formula of Roult's law for non-volatile and electrolyte solute is:
(P = Vapour pressure of pure solvent
P
s = vapour pressure of solution
i = Van't Hoff Factor
n = Moles of solute
N = Moles of solvent)
| 1. |
\(P-P_s \over P \) = i (\(n \over n+N\)) |
2. |
\(P-P_s \over P_s\) = i (\(n \over n+N\)) |
| 3. |
\(P-P_s \over P \) = \(i n \over in+N\) |
4. |
\(P-P_s \over P_s\) = \(in \over in+N\) |
114. Which of the following element does not show a variable oxidation state?
1. \(Zn\)
2. \(Fe\)
3. \(Mn\)
4. \(Cu\)
115. What will be the nature of an aqueous solution of sodium bisulfite (\(NaHSO_3 \))?
1. Acidic
2. Alkaline
3. Slightly alkaline
4. Slightly acidic
116. Given the reaction:
R-OH + PCl
5 \( \longrightarrow \) R-Cl + POCl
3 + HCl
The most reactive alcohol in the above reaction will be:
| 1. |
1o Alcohol |
2. |
2o Alcohol |
| 3. |
3o Alcohol |
4. |
Cannot be predicted |
117. Beryllium has higher ionization enthalpy than boron. This can be explained as:
| 1. |
Beryllium has a higher size than boron, hence its ionization enthalpy is higher. |
| 2. |
Penetration of 2p-electrons to the nucleus is more than the 2s electrons. |
| 3. |
It is easier to remove electrons from 2p-orbital as compared to 2s-orbital due to more penetration of s electron. |
| 4. |
Ionization energy increases in a period. |
118. The bond order of C2 molecule is two. Therefore, it can be concluded that it has a double bond. Consequently, it can also be concluded that C2 molecule contains:
1. Two sigma bonds
2. Two pi bonds
3. One sigma and one pi bond
4. Half sigma and one and half pi bond
119. For the reaction, \(\mathrm{N}_2+3 \mathrm{H}_2 \rightarrow 2 \mathrm{NH}_3,\) if, \(\dfrac{d[NH_{3}]}{dt} \ = \ 2\times 10^{-4} \ mol \ L^{-1} \ s^{-1}\), the value of \(\dfrac{-d[H_{2}]}{dt}\) would be:
1. \(3 \times 10^{-4} \mathrm{~mol} \mathrm{~L}^{-1} \mathrm{~s}^{-1} \)
2. \(4 \times 10^{-4} \mathrm{~mol} \mathrm{~L}^{-1} \mathrm{~s}^{-1} \)
3. \(6 \times 10^{-4} \mathrm{~mol} \mathrm{~L}^{-1} \mathrm{~s}^{-1} \)
4. \(1 \times 10^{-4} \mathrm{~mol} \mathrm{~L}^{-1} \mathrm{~s}^{-1}\)
120. Which compound is most reactive towards electrophilic substitution reaction?
121. How many nodal planes are present in \(4~dz^2\) ?
1. 1
2. 2
3. 3
4. Zero
122. The products obtained by the electrolysis of an aqueous solution of \(CuCl_2 \) with platinum electrodes will be:
1. Anode: \(O_2\) , Cathode: \(H_2\)
2. Anode: \(Cl_2\) , Cathode: \(H_2\)
3. Anode: \(Cl_2\) , Cathode: \(Cu\)
4. Anode: \(O_2\) , Cathode: \(Cu\)
123. When sodium thiosulphate is reacted with I2 , then the product of sulphur will be:
1.
2.
3.
4.
124. Formula of
\(bis~\text {(ethylenediamine) }~\text {dithiocyanatoplatinum} \text{(IV)}\) ion is:
1. \(Pt[(en)_2S_2CN]^{4-}\)
2. \(Pt[(en)_2S_2CN]^{4+}\)
3. \([Pt(en)(SCN)_2]^{4-}\)
4. \([Pt(en)_2(SCN)_2]^{2+}\)
125. The number of hydrogen bonded water molecules(s) associated with \(\mathrm{C u S O_{4} . 5 H_{2} O}\) is:
126. Which of the following lanthanoid ions is diamagnetic?
(At. No. Ce = 58, Sm = 62, Eu = 63, Yb = 70)
| 1. |
|
2. |
|
| 3. |
|
4. |
|
127. The correct order of increasing C-X bond reactivity toward nucleophiles among the following is:
| I |
 |
II |
 |
| III |
\(\left(\mathrm{CH}_3\right)_3 \mathrm{C}-\mathrm{X}\) |
IV |
\(\left(\mathrm{CH}_3\right)_2 \mathrm{CH}-\mathrm{X}\) |
| 1. |
I < II < IV < III |
2. |
II < III < I < IV |
| 3. |
IV < III < I < II |
4. |
III < II < I < IV |
128. When toluene is oxidised with chromyl chloride, benzaldehyde is formed. This reaction is known as:
1. Gattermann Koch Synthesis
2. Etard's reaction
3. Schmidt's reaction
4. Friedel Craft reaction
129. 10 gm carbon reacts with 100 gm \(Cl_2\) to form \(CCl_4.\) The maximum weight of \(CCl_4\) formed will be:
1. 128 gm
2. 94.22 gm
3. 108.45 gm
4. 54.22 gm
130. The correct order of electron gain enthalpy is:
1. S > Se > Te > O
2. Te > Se > S > O
3. O > S > Se > Te
4. S > O > Se > Te
131. 3-Methylpentane forms all the possible monohalogenated products. Among all monohalogenated products, how many products can show optical isomerism?
1. 1
2. 2
3. 3
4. 4
132. Which of the following does not illustrate resonance between the structures?
133. What is the relative order of basicity for the given-below compounds?
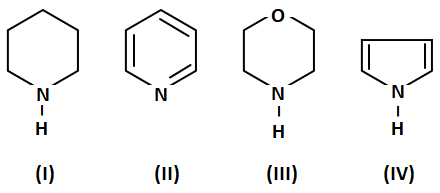
1. IV>I>III>II
2. III>I>IV>II
3. II>I>III>IV
4. I>III>II>IV
134. The most basic hydride of Group 15 is:
1. \(NH_3 \)
2. \(PH_3 \)
3. \(AsH_3 \)
4. \(BiH_3 \)
135. Which of the following ions has the maximum magnetic moment?
1. \(Mn^{3+}\)
2. \(Cu^{2+}\)
3. \(Fe^{3+}\)
4. \(V^{3+}\)
Chemistry - Section B
136. The element 'Re' belongs to which group and period, respectively?
1. 7 and 6
2. 7 and 7
3. 6 and 7
4. 6 and 6
137. At 298 K the Emf
of the following cell is:
\(\small{Pt|H_{2}(1 \ atm)|H^{+}(0.02 \ M) \ || \ H^{+}(0.01 \ M)|H_{2}(1 \ atm)|Pt}\)
1. - 0.017 V
2. 0.0295 V
3. 0.1 V
4. 0.059 V
138. What is the activation energy for reverse reaction on the basis of given data ?
N2O4(g) 2NO2(g) ΔH = +54kJ
Ea(forward) = +57.2 kJ
1. -54 kJ
2. +3.2 kJ
3. 60.2 kJ
4. 111.2 kJ
139. For the reversible reaction
\(N_2(g)+3H_2(g)⇌2NH_3(g)\), what is the effect on the equilibrium position when an inert gas is added at constant volume?
| 1. |
Shifts in the forward reaction |
| 2. |
Shifts in backward reaction |
| 3. |
Remains unaffected |
| 4. |
Initially in the forward direction and then in the backward direction |
140. Among acetic acid, phenol and n-hexanol which one of the following compound will react with NaHCO3 solution to give sodium salt and CO2?
1. Acetic acid
2. n-Hexanol
3. Acetic acid and Phenol
4. Phenol
141. Match column I with column II:
|
Column-I (Compounds) |
|
Column-II(number of lone pairs of a central atom) |
| A. |
\(IF_7 \) |
P. |
0 |
| B. |
\(ICl_4^- \) |
Q. |
1 |
| C. |
\(XeF_2 \) |
R. |
2 |
| D. |
\(XeF_6 \) |
S. |
3 |
Choose the correct answer from the options given below:
1. A – P, B – Q, C – R, D – S
2. A – P, B – R, C – S, D - Q
3. A – R, B – S, C – P, D - Q
4. A – S, B – R, C – Q, D - P
142. Which, of the following alcohols, is most reactive toward dehydration?
143. Given the reaction:
\(A + B \rightarrow C+D - q~ cal. \) is found to have a positive entropy change. The reaction will be:
| 1. |
Possible at high temperature. |
| 2. |
Possible only at low temperatures. |
| 3. |
Not possible at any temperature. |
| 4. |
Possible at any temperature. |
144. The most stable resonating structure of HCOOH is:
145. Which of the following mixtures can act as an acidic buffer?
1. 50 ml 0.1 M CH3COOH + 50 ml 0.1 M NaOH
2. 100 ml 0.1 M CH3COOH + 50 ml 0.1 M NaOH
3. 50 ml 0.1 M CH3COOH + 100 ml 0.1 M NaOH
4. None of the above
146. The hybridisations of atomic orbitals of nitrogen in \(\mathrm{NO}^{+}\), \(N O_3^{-}\) and NH3 respectively are:
1.
2.
3.
4.
147.
| Assertion (A): |
Osmosis is the movement of solvent molecules (e.g., water) across a semipermeable membrane from a region of lower solute concentration to a region of higher solute concentration.
|
| Reason (R): |
Solutions having the same osmotic pressure are called isotonic solutions. |
| 1. |
Both (A) and (R) are True, and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True, but (R) is not the correct explanation of (A). |
| 3. |
(A) is True, but (R) is False. |
| 4. |
(A) is False, but (R) is True. |
148. The most stable carbocation is:
149. The magnetic moment of a metal ion is 3.87 B.M. The metal ion is:
| 1. |
\(V^{3+} \) |
2. |
\(\mathrm{Cr}^{3+} \) |
| 3. |
\(\mathrm{Mn}^{2+} \) |
4. |
\(T i^{2+} \) |
150. Neutral oxide, among the following, is:
| 1. |
\(\text{CO}\) |
2. |
\(\text{SnO}_2\) |
| 3. |
\(\text{ZnO}\) |
4. |
\(\text{SiO}_2\) |
Physics - Section A
151. A calorimeter contains \(270\) g of ice at \(0^\circ\)C (specific latent heat \(80\) cal/g). Steam (specific latent heat \(540\) cal/g) at \(100^\circ\)C is continuously passed through it, and the excess steam is allowed to escape. Assume negligible loss of heat to the surroundings, except due to excess steam being allowed to escape. Also, ignore the heat capacity of the calorimeter. The final mass of water in the calorimeter is:
1. \(40\) g
2. \(90\) g
3. \(310\) g
4. \(360\) g
152. The product of the angular momentum and the kinetic energy of an electron in the
\(n^\text{th}\) Bohr orbit in a hydrogen atom is proportional to:
| 1. |
\(n\) |
2. |
\(n^2\) |
| 3. |
\(\dfrac1n\) |
4. |
\(\dfrac{1}{n^3}\) |
153. Three charged particles having charges equal to
\(+1~\text C,\) \(-2~\text C\) and
\(+3~\text C\) are placed on the
\(x\text-\)axis at
\(x=1~\text{cm},\) \(2~\text{cm},\) \(3~\text{cm}\) respectively. Consider a sphere of radius
\(10~\text{cm}\) centred at the origin. The magnitude of the flux of the electric field of these charges through the sphere is, numerically:
| 1. |
slightly greater than \(\dfrac{2}{\varepsilon_0}\) |
| 2. |
slightly less than \(\dfrac{2}{\varepsilon_0}\) |
| 3. |
exactly equal to \(\dfrac{2}{\varepsilon_0}\) |
| 4. |
exactly equal to \(-\dfrac{2}{\varepsilon_0}\) |
154. Two long solenoids have the same total length and the same total number of turns, but their cross-sectional areas are different: \(A_1,A_2\) where \(A_1>A_2.\) Their self-inductances \(L_1,L_2\) are in the ratio (approximately):
1. \(A_1:A_2\)
2. \(A^2_1:A_2^2\)
3. \(A_2:A_1\)
4. \(A^2_2:A_1^2\)
155. A sample of magnetic material is placed in a magnetic field. As the magnetic field is increased, its magnetisation increases and finally, it becomes constant — even if the field is increased further.
Now, the temperature of the sample is decreased. The magnetisation:
| 1. |
increases |
| 2. |
decreases |
| 3. |
remains unchanged |
| 4. |
decreases first and then increases |
156. Given below are two statements:
| Assertion (A): |
If two particles move with uniform accelerations in different directions, then their relative velocity changes in direction. |
| Reason (R): |
Since the acceleration are in different directions, there is a relative acceleration and hence the relative velocity changes. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
157. A block is placed on a smooth horizontal surface, and forces are applied to it as shown in the diagram. Take
\(g=10~\text{m/s}^2.\) The normal reaction acting on the block is:

1.
\(100~\text N\)
2.
\(60~\text N\)
3.
\(40~\text N\)
4.
\(20~\text N\)
158. A solid sphere (centre
\(O\)) of homogeneous transparent material kept upright has rays parallel to its vertical diameter falling on it. A ray that is aimed at
\(P,\) where the horizontal distance
\(OP=\dfrac{\sqrt3}{2}R\) (
\(R\) being the sphere's radius), undergoes refraction at the sphere's surface and strikes the lowest point
\(A.\)
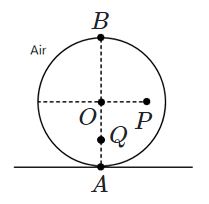
Rays falling close to the highest point
\((B)\) of the sphere are directed towards a point
\(Q\) on the vertical diameter
\(AB,\) after refraction. The distance
\(BQ\) is:
| 1. |
\(\dfrac{R}{2}\) |
2. |
\(\dfrac{R\sqrt3}{\sqrt3 +1}\) |
| 3. |
\(\dfrac{R\sqrt3}{2}\) |
4. |
\(\dfrac{R\sqrt3}{\sqrt3 -1}\) |
159. The output
\((Y)\) of the logic circuit given above is:

1.
\(A\) AND
\(C\)
2.
\(A\) OR
\(C\)
3. always TRUE
4. always FALSE
160. One mole of an equimolar mixture of monoatomic \((He)\) and diatomic \((H_2)\) gases is heated to raise the temperature by \(1\) K under constant pressure. The amount of heat used in this process is (nearly):
1. \(8.3\) J
2. \(16.6\) J
3. \(25\) J
4. \(29\) J
161. Given below are two statements:
| Statement I: |
If the acceleration of a particle is directed towards a fixed point, and proportional to the distance from that point – the motion is SHM. |
| Statement II: |
During SHM, the kinetic energy of the particle oscillates at twice the frequency of the SHM. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
162. The circuit shown in the figure has ideal batteries wired up along with the resistances.
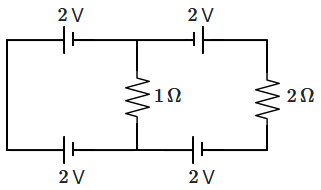
The current through the
\(2~\Omega\) resistance is:
1.
\(0\) A
2.
\(1\) A
3.
\(1.5\) A
4.
\(2\) A
163. Match the following quantities in
List-I with their dimensions in
List-II.
| List-I |
List-II |
| (a) |
acceleration |
(i) |
\([M^0L^0T^0 ]\) |
| (b) |
torque |
(ii) |
\([ML^{-1}T^{-2} ]\) |
| (c) |
absorptive power |
(iii) |
\([LT^{-2} ]\) |
| (d) |
pressure |
(iv) |
\([ML^2T^{-2} ]\) |
| 1. |
(a)-(iii), (b)-(iv), (c)-(i), (d)-(ii) |
| 2. |
(a)-(iii), (b)-(ii), (c)-(i), (d)-(iv) |
| 3. |
(a)-(iii), (b)-(i), (c)-(ii), (d)-(iv) |
| 4. |
(a)-(ii), (b)-(iv), (c)-(iii), (d)-(i) |
164. Given below are two statements:
| Statement I: |
If a galvanometer is connected with a high resistance in series with it, it can be used as an ammeter. |
| Statement II: |
If a galvanometer is connected with a low resistance in parallel with it, it can be used as a voltmeter. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
165. The moment of inertia of a uniform solid hemisphere, about a tangent to the circumference of the hemisphere's flat circular surface, is:
(mass of hemisphere: \(m,\) radius: \(R\))
| 1. |
\(\dfrac75mR^2\) |
2. |
\(\dfrac7{10}mR^2\) |
| 3. |
\(\dfrac15mR^2\) |
4. |
\(\dfrac65mR^2\) |
166. A
\(10:1\) step-down transformer has an ideal diode and a
\(10~\Omega\) resistance connected to its secondary circuit while
\(220~\text{V}\) AC mains is applied to the primary.
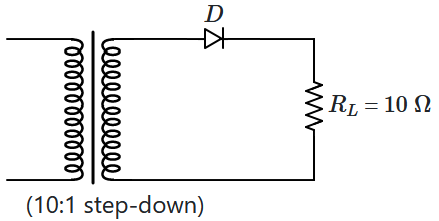
The peak reverse voltage across the diode is:
| 1. |
\(220~\text V\) |
2. |
\(22\sqrt 2~\text V\) |
| 3. |
\(\dfrac{22 }{\sqrt 2}~\text V\) |
4. |
\(44\sqrt 2~\text V\) |
167. Which, of the following, shows the correct graph of the de-Broglie wavelength
\((\lambda)\) of a particle and its kinetic energy
\((E_K)?\)
168. Given below are two statements:
| Assertion (A): |
Faraday's law of electromagnetic induction is not consistent with the law of conservation of energy. |
| Reason (R): |
Lenz's law is consistent with energy conservation. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
169. An inductor \(\left(L = 200~\text{mH}\right)\) is connected to an AC source of peak emf \(210~\text{V}\) and frequency \(50~\text{Hz}\). The peak current is:
1. \(2.7~\text{A}\)
2. \(1.2~\text{A}\)
3. \(2.0~\text{A}\)
4. \(3.3~\text{A}\)
170. Given below are two statements:
| Statement I: |
The kinetic energy of a planet is maximum when it is closest to the sun. |
| Statement II: |
The time taken by a planet to move from the closest position (perihelion) to the farthest position (aphelion) is larger for a planet that is farther from the sun. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
171. During a
\(\beta\)-decay, fast electrons are found to have been emitted. These electrons come from:
| 1. |
the outermost atomic shell |
| 2. |
the innermost atomic shell |
| 3. |
the conduction band |
| 4. |
the nucleus |
172. Three capacitors are connected in the configuration shown, with
\(C_1=C_3=C\) and
\(C_2=2C.\) If a charge
\(Q\) is passed through the circuit from
\(A\) to
\(B\) (with the capacitors initially uncharged), the energies stored in the capacitors,
\(C_1,C_2,C_3\) are in the ratio:

| 1. |
\(1:2:1\) |
2. |
\(1:\dfrac12:1\) |
| 3. |
\(1:4:1\) |
4. |
\(1:\dfrac14:1\) |
173. If an AND-gate and an OR-gate are OR-ed together, the resulting gate is:
1. AND
2. OR
3. XOR
4. none of the above
174. A uniform disc of mass
\(m\) and radius
\(r\) is rotating freely about its own axis, which is kept vertical. There is a small insect of mass
\(\dfrac m2\) sitting on the periphery of the disc, also rotating with the disc. The initial angular speed of the disc is
\(\omega_0.\) The insect moves radially inward and finally reaches the centre. The final angular speed of the disc is:
| 1. |
\(\omega_0\) |
2. |
\(2\omega_0\) |
| 3. |
\(\dfrac32\omega_0\) |
4. |
\(\dfrac52\omega_0\) |
175. In a Young's double-slit experimental setup,
\(240\) fringes are observed to be formed in a region of the screen when light of wavelength
\(450\) nm is used. If the wavelength of light is changed to
\(600\) nm, the number of fringes formed in the same region will be:
| 1. |
\(135\) |
2. |
\(180\) |
| 3. |
\(320\) |
4. |
\(428\) |
176. A plane electromagnetic waveform given by:
\(\vec {E}_1=E_0\hat j\sin(\omega t-kx)\)
propagates along the
\(x\)-axis. A second waveform given by:
\(\vec {E}_2=E_0\hat k\sin(\omega t-kx)\)
is also allowed to propagate. The magnetic field has the amplitude:
(Assume speed of light in vacuum is \(c\))
| 1. |
\(\dfrac{E_0}{c}\) |
2. |
\(\dfrac{E_0}{2c}\) |
| 3. |
\(\dfrac{\sqrt2E_0}{c}\) |
4. |
Zero |
177. A negative point charge
\((-q)\) is placed at the centre of a spherical ball of charge distributed uniformly over its volume. The radius of the ball is
\(R.\) The electric field on its surface is zero. The potential on its surface is:
| 1. |
zero |
2. |
\(\dfrac{kq }{2R}\) |
| 3. |
\(\dfrac{-kq }{ 2R}\) |
4. |
\(\dfrac{2kq }{ R}\) |
178. A gas
\((\gamma = 1.5)\) undergoes a process in which its volume is doubled, but the speed of sound in the gas remains unchanged. Then,
| 1. |
the pressure is halved |
| 2. |
the pressure decreases by a factor of \(2\sqrt 2\) |
| 3. |
the temperature is halved |
| 4. |
the temperature decreases by a factor of \(2 \sqrt 2\) |
179. Two organ pipes closed at one end produce \(5\) beats per second in fundamental mode. If the ratio of their lengths is \(10:11\), then their frequencies (in Hz) are:
| 1. |
\(55,50\) |
2. |
\(105,100\) |
| 3. |
\(75,70\) |
4. |
\(100,95\) |
180. Given below are two statements:
| Statement I: |
The total energy of a particle falling freely under gravity increases with time. |
| Statement II: |
The law of conservation of mechanical energy does not apply to the motion of a projectile under gravity. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
181. The elastic energy density in a stretched wire is:
| 1. |
\(\text{(stress)}^2 × \text{strain}\) |
2. |
\(\text{stress} × \text{strain}\) |
| 3. |
\(\dfrac12\times \text{stress} × \text{strain}\) |
4. |
\(\text{stress} × \text{(strain)}^2\) |
182. The velocity-time graph of a particle moving along a straight line is shown in the adjacent figure. The average velocity is:

| 1. |
\(1~\text{ms}^{-1}\) |
2. |
\(2~\text{ms}^{-1}\) m/s |
| 3. |
less than \(1~\text{ms}^{-1}\) |
4. |
greater than \(2~\text{ms}^{-1}\) |
183. Given below are two statements:
| Statement I: |
In an elastic collision, the relative velocity of approach equals the relative velocity of separation. |
| Statement II: |
In an elastic collision, the kinetic energy before the collision is equal to the kinetic energy after the collision. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
184. The capacitance of the system of capacitors connected in the circuit, between
\(A\) and
\(B,\) equals:

1.
\(4~\mu\text{F}\)
2.
\(2.5~\mu \text{F}\)
3.
\(2.4~\mu \text{F}\)
4.
\(1.5~\mu \text{F}\)
185. If a convex lens
\((\mu=1.5)\) is immersed in water
\(\left(\mu=\dfrac{4}{3}\right)\) then its focal length in water is:
| 1. |
\(\dfrac14\) of that in air |
2. |
\(\dfrac89\) of that in air |
| 3. |
\(\dfrac98\) of that in air |
4. |
\(4\) times that in air |
Physics - Section B
186. The string and pulley are ideal, and there is no friction anywhere. If the block
\(A\) is moving down at
\(2\) m/s with the string taut, the rate at which work is done on the
\(2\) kg block
\(B\) is:
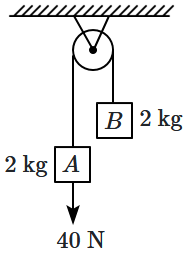
1.
\(80\) W
2.
\(40\) W
3.
\(20\) W
4.
\(0\) W
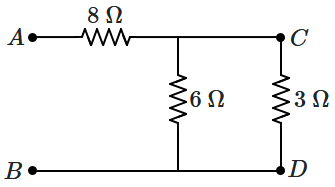
187. The resistance between
\(A\) and
\(B\) is

| 1. |
\(100~\Omega\) if \(V_A>V_B\) and \(200~\Omega\) if \(V_A<V_B\) |
| 2. |
\(100~\Omega\) if \(V_A<V_B\) and \(200~\Omega\) if \(V_A>V_B\) |
| 3. |
\(100~\Omega\) |
| 4. |
\(200~\Omega\) |
188. Select the correct option based on the statements given below:
| Statement I: |
When a projectile is at its highest point, its tangential acceleration is zero. |
| Statement II: |
When a projectile is at the highest point of its trajectory, its speed is minimum. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
189. Two blocks are joined by a light spring of spring constant,
\(k=1000\) N/m, and are placed at rest on a horizontal frictionless table. A horizontal force of
\(6\) N acts on the
\(2\) kg block. The acceleration of the centre-of-mass of the system is:

| 1. |
\(3\) m/s2 |
2. |
\(2\) m/s2 |
| 3. |
\(1.5\) m/s2 |
4. |
\(4.5\) m/s2 |
190. Consider a tank full of water with an outlet at the bottom. As the outlet valve is opened, and water flows out, the pressure inside the tank near the outlet:
| 1. |
increases. |
| 2. |
decreases. |
| 3. |
remains unchanged. |
| 4. |
increases and then quickly returns to normal. |
191. A block
\(A\) is pushed on a smooth horizontal plane by applying a horizontal force
\(F ,\) which causes an acceleration of
\({\dfrac g 4}\) (\(g\): acceleration due to gravity). The block does not topple, even though the force acts at its highest point. The normal reaction shifts forward by:

| 1. |
\({\dfrac b 2}\) |
2. |
\({ \dfrac b 4}\) |
| 3. |
\({\dfrac b 8}\) |
4. |
\(\dfrac b 3\) |
192. Photons of frequency
\(\nu\) fill a room. A metallic plate having a work function
\(W\) \((<h\nu)\) is moved with a velocity
\(v\), in this room. The maximum energy of the emitted photoelectrons: (in the plate's frame)
| 1. |
does not depend on \(v\) |
| 2. |
increases as \(v\) increases |
| 3. |
decreases as \(v\) increases |
| 4. |
first increases and then decreases as \(v\) is increased |
193. The surface tension of soapy water is
\(S.\) When bubbles are blown with soapy water, one bubble of radius
\(r\) is formed within another of radius
\(3r.\) The excess pressure within the smaller bubble, relative to the atmospheric pressure is:
| 1. |
\(\dfrac{4S}{r}\) |
2. |
\(\dfrac{8S}{3r}\) |
| 3. |
\(\dfrac{8S}{r}\) |
4. |
\(\dfrac{16S}{3r}\) |
194. The system of blocks is pulled up by force as shown in the figure. The force exerted on the
\(3\) kg block by the connecting string is:

| 1. |
\(80~\text{N}\) |
2. |
\(60~\text{N}\) |
| 3. |
\(40~\text{N}\) |
4. |
\(100~\text{N}\) |
195. A parallel beam is incident on to a lens of focal length
\(f\) (positive), parallel to its principal axis. A thin prism of vertex angle
\(A\) and refractive index
\(\mu\) is inserted in the path of the parallel beam before it falls on the lens. The focal point of the image:

| 1. |
remains unchanged |
| 2. |
is displaced along \(+y\) by \((\mu-1)Af\) |
| 3. |
is displaced along \(-y\) by \((\mu-1)Af\) |
| 4. |
is displaced along \(+x\) by \((\mu-1)Af\) |
196. A potential difference
\(V_{AB}\) is impressed across
\(AB\) and the potential difference across
\(CD\) \((V_{CD})\) is measured. Assume,
\(V_{AB}=300~\text{V}.\)
The current through the
\(8~\Omega\) resistance is:
1.
\(\dfrac{300}{17}~\text A\)
2.
\(\dfrac{300}{8.5}~\text A\)
3.
\(30~\text A\)
4.
\(\Big(\dfrac{300}{14}+\dfrac{300}{11}\Big)~\text A~~\)
197. A spring-mass system is undergoing horizontal oscillations on a frictionless surface. During the oscillation, the block picks up a particle, when it is at its extreme position. As a result of this,

| 1. |
the amplitude increases. |
| 2. |
the amplitude decreases. |
| 3. |
the frequency increases. |
| 4. |
the frequency decreases. |
198. Two long parallel wires carrying currents
\(I_1\) and
\(I_2\) give a magnetic field of
\(3\) G at a point exactly mid-way between the two wires. When one of the currents is reversed, the field becomes
\(5\) G. The ratio of the large current to the smaller one is:
| 1. |
\(2\) |
2. |
\(\dfrac43\) |
| 3. |
\(\dfrac32\) |
4. |
\(4\) |
199. A sinusoidal waveform whose displacement is given
by: \(y(x, t)=(5 \mathrm{~mm}) \sin 2 \pi\left(\frac{x}{2 \mathrm{~m}}+\frac{t}{0.01 \mathrm{~s}}\right)\)
propagates along the
\(x\)-axis.
The frequency of the wave is:
| 1. |
\(0.01~\text{Hz}\) |
2. |
\(\large\frac{0.01}{2\pi}\)\(~\text{Hz}\) |
| 3. |
\(100~\text{Hz}\) |
4. |
\(200~\text{Hz}\) |
200. Given below are two statements:
| Assertion (A): |
In an isothermal process, whole of the heat energy supplied to the body is converted into internal energy. |
| Reason (R): |
According to the first law of thermodynamics, \(\Delta Q = \Delta U + \Delta W \). |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course