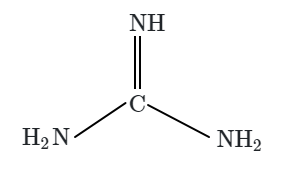
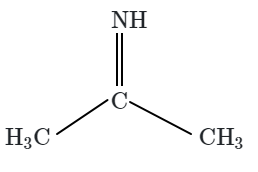
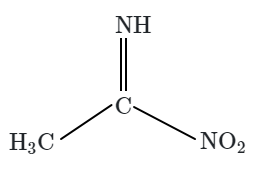
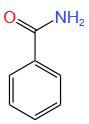
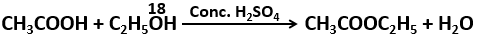Botany - Section A
1. Water hyacinth led to the decline of indigenous species when introduced in India. It grew massively because
| 1. |
It turned invasive and killed its predator |
| 2. |
Indigenous species created the favourable environment for it |
| 3. |
It requires O2 for photosynthesis which was provided by aquatic indigenous plants |
| 4. |
Its natural predator was not present |
2. The DNA sequence shown below is:
5’ – GAATTC – 3’
3’ – CTTAAG – 5’
| 1. |
the consensus sequence for prokaryotic promoter |
| 2. |
the most common VNTR in the human genome |
| 3. |
the recognition sequence of a common restriction endonuclease EcoR I |
| 4. |
an intron within the eukaryotic split gene |
3. In agar gel electrophoresis, the restriction fragments produced by restriction enzymes:
| 1. |
move towards cathode |
| 2. |
do not move at all |
| 3. |
are separated according to size with smaller fragments moving farther |
| 4. |
are transported onto a nitrocellulose membrane |
4. Consider the given two statements:
| Assertion (A): |
Measurement of biomass in terms of fresh weight is more accurate. |
| Reason (R): |
The standing crop is measured as the mass of living organisms
(biomass) or the number in a unit area. |
| 1. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 2. |
(A) is True but (R) is False |
| 3. |
(A) is False but (R) is True |
| 4. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
5. The chromosome, where the centromere is located in the centre and hence has two equal arms, is called as :
1. Metacentric
2. Sub-metacentric
3. Acrocentric
4. Telocentric
6. Like mitochondria, the chloroplasts:
| 1. |
are found only in plant cells |
| 2. |
do not have 70 S ribosomes |
| 3. |
are also double membrane bound |
| 4. |
are unable to carry out phosphorylation of ADP |
7. Identify the incorrectly matched pair:
|
Placentation |
Example |
| 1. |
Marginal |
Pea |
| 2. |
Axile |
China rose |
| 3. |
Parietal |
Primrose |
| 4. |
Basal |
Marigold |
8. A cell decides not to divide and enters the Quiescent Stage (G0) exiting the cell cycle from:
1. G1 phase
2. S phase
3. G2 phase
4. M phase
9. Which of the secondary metabolites are carbohydrate binding proteins and called lectins?
1. Morphine
2. Codeine
3. Concanavalin A
4. Vinblastin
10. How many of the given statements are correct:
| I: |
Endosperm development precedes embryo development. |
| II: |
The coconut water from tender coconut is cellular endosperm. |
| III: |
The white kernel in coconut is free nuclear endosperm. |
| IV: |
Endosperm is completely consumed by the developing embryo in pea. |
| V: |
Endosperm persists in the mature seed of castor. |
1. 2
2. 3
3. 4
4. 5
11. During which stage of the meiotic cell division does the centromere split leading to the separation of sister chromatids?
| 1. |
Metaphase I |
2. |
Metaphase II |
| 3. |
Anaphase I |
4. |
Anaphase II |
12. When a molecule of pyruvic acid undergoes lactic acid fermentation then
| 1. |
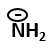
3 ATP are lost |
2. |
1 ATP is lost |
| 3. |
6 ATP are lost |
4. |
2 ATP are gained |
13. Select the incorrect statement with respect to photorespiration?
| 1. |
In C4-plants photorespiration does not occur |
| 2. |
The biological function of photorespiration is not known yet |
| 3. |
RuBP binds with O2 to form 2 molecules of 3-PGA |
| 4. |
There is no synthesis of ATP or NADPH |
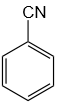
14. Identify the incorrectly matched pair:
| 1. |
Golden rice |
Vitamin B12 enriched rice |
| 2. |
Rosie |
First transgenic cow |
| 3. |
Bt Cotton |
Genetically modified pest resistant plant cotton variety |
| 4. |
Basmati controversy |
Biopiracy |
15. Which organization publishes the 'Red Data Book'?
| 1. |
IUCN |
2. |
UNEP |
| 3. |
WWF |
4. |
GEF |
16. Which plant growth regulators are terpenes?
| 1. |
Auxins |
2. |
GAs |
| 3. |
ABA |
4. |
Cytokinins |
17. Consider the given two statements:
| Assertion (A): |
Flowering plants have developed many devices to discourage self-pollination and to encourage cross-pollination. |
| Reason (R): |
Majority of flowering plants produce hermaphrodite flowers and pollen grains are likely to come in contact with the stigma of the same flower. |
| 1. |
Both (A) and (R) are True and (R) explains (A) |
| 2. |
Both (A) and (R) are True and (R) does not explain (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
(A) is False but (R) is True |
18. Consider the given two statements:
| Assertion (A): |
When resources in the habitat are unlimited, the population grows in an exponential or geometric fashion. |
| Reason (R): |
When resources in the habitat are unlimited, each species has the ability to realise fully its innate potential to grow in number. |
| 1. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 2. |
(A) is True but (R) is False |
| 3. |
(A) is False but (R) is True |
| 4. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
19. Which of the following is not a significance of mitosis?
| 1. |
Restoration of nuclear/cytoplasmic ratio |
| 2. |
Growth |
| 3. |
Reduction of chromosome number |
| 4. |
Replacement of lost cells |
20. You have a tall garden pea plant. A cross that will help you to determine whether the plant is homozygous tall or hybrid tall is:
| 1. |
A reciprocal cross |
| 2. |
A back cross with homozygous dominant parent |
| 3. |
Test cross |
| 4. |
Dihybrid cross |
21. The fruit in mango and coconut is called as:
1. Aril
2. Drupe
3. Berry
4. Samara
22. If you want to generate virus-free plant through micropropagation [plant tissue culture], the explant you should use is:
1. Meristem
2. Embryo
3. Anther
4. Ovule
23. About what per cent of the energy in the sunlight, available to them, primary producers can convert into Net Primary Productivity (NPP)?
1. 1
2. 10
3. 50
4. 90
24. Which of the following is not correct regarding the famous double helix model of DNA given by Watson and Crick?
| 1. |
The two strands are anti-parallel |
| 2. |
The two strands are complementary |
| 3. |
The strands are coiled in a right-handed fashion |
| 4. |
The pitch of the helix is 34 nm |
25. Sixth Extinction presently in progress is different from the previous episodes by some reasons except
1. Difference in the rates
2. Current rate is 100-1000 times faster than previous episodes
3. Human intrusions
4. Extinction of special bird species
26. To isolate DNA from a cell, the cell wall needs to be broken. Match the enzymes in Column I with the type of cell in Column II and select the correct match from the codes given:
|
Column I |
|
Column II |
| A. |
Lysozyme |
P. |
Fungal cell |
| B. |
Cellulase |
Q. |
Plant cell |
| C. |
Chitinase |
R. |
Bacterial cell |
Codes:
|
A |
B |
C |
| 1. |
P |
Q |
R |
| 2. |
P |
R |
Q |
| 3. |
R |
P |
Q |
| 4. |
R |
Q |
P |
27. Regarding various levels of protein structure, which of the following is not true?
| 1. |
The positional information of amino acids in a protein is called the primary structure. |
| 2. |
Alpha helix is a secondary structure and beta pleated sheet is a tertiary structure of a protein. |
| 3. |
Tertiary structure is absolutely necessary for the many biological activities of proteins. |
| 4. |
Quaternary structures are seen in proteins made of more than one polypeptide chain. |
28. Selaginella, a pteridophyte which belongs to the class Lycopsida, is:
| 1. |
heterosporous and has microphylls |
| 2. |
homosporous and has macrophylls |
| 3. |
homosporous and has microphylls |
| 4. |
heterosporous and has macrophylls |
29. Who is revered as ‘the father of ecology’ in India?
1. Gurdev Singh Khush
2. Panchanan Maheshwari
3. M. S. Swaminathan
4. Ramdeo Misra
30. The number of steps in the Kreb's cycle where oxidative decarboxylation takes place is:
31. Identify the incorrect statement:
| 1. |
Gymnosperms lack vessels in their xylem. |
| 2. |
In roots, the arrangement of primary xylem is endarch. |
| 3. |
Gymnosperms lack sieve tubes and companion cells. |
| 4. |
Phloem parenchyma is absent in most of the monocotyledons. |
32. A collective term for the parts of a flower that produce ovules and ultimately develop into the fruit and seeds is:
| 1. |
Pistil |
2. |
Megasporophyll |
| 3. |
Gynoecium |
4. |
Carpel |
33. All the following statements regarding diatoms are true except:
| 1. |
The two overlapping shells of cell wall fit together as in a soap box. |
| 2. |
They are the chief ‘producers’ in the oceans. |
| 3. |
Their cell walls are embedded with silica. |
| 4. |
Diatoms behave like heterotrophs when deprived of sunlight. |
34. Which of the following is true for trichomes
| 1. |
usually multicellular |
| 2. |
may be branched or unbranched |
| 3. |
prevent transpirational water loss |
| 4. |
All of the above |
35. Consider the given two statements:
| Assertion (A): |
Majority of insect-pollinated flowers are large, colourful, fragrant and rich in nectar. |
| Reason (R): |
Animals are attracted to flowers by colour and/or fragrance. |
| 1. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
| 2. |
(A) is True but (R) is False. |
| 3. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 4. |
(A) is False but (R) is True. |
Botany - Section B
36. Identify the correct statement(s) regarding the modifications seen in stems:
| I: |
Stem tendrils develop from axillary buds and help plants, such as pea, to climb |
| II: |
Opuntia modifies its stems into flattened structures to protect itself from browsing animals |
| III: |
Underground stems of some plants such as grass and strawberry spread to new niches and when older plants die, new plants are formed. |
| 1. |
I, II, and III |
2. |
Only II and III |
| 3. |
Only III |
4. |
Only I and III |
37. Which of the following is not an action of ethylene in plants?
| 1. |
Seedling triple response |
| 2. |
Stimulates leaf and flower senescence |
| 3. |
Inhibits senescence of mature xylem cells in preparation for plant use |
| 4. |
Inhibits shoot growth except in some habitually flooded plants like rice |
38. Consider the given two statements:
| Assertion (A): |
Unlike animals, plants retain the capacity for unlimited growth throughout their life. |
| Reason (R): |
Unlike animals, there is presence of meristems at certain locations in their body. |
| 1. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 2. |
(A) is True; (R) is False |
| 3. |
(A) is False; (R) is False |
| 4. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
39. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
Cyclic photophosphorylation results in the synthesis of ATP but not of NADPH + H+. |
| Reason (R): |
The excited electron does not pass on to NADP+ but is cycled back to the PS I complex through the electron transport chain. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
40. All the following are hotspots of biodiversity covering India except:
1. Eastern Ghats and Bangladesh
2. Western Ghats and Sri Lanka
3. Indo-Burma
4. Himalaya
41. Consider the given two statements:
| Assertion (A): |
The physical basis for segregation is the splitting of centromere during anaphase I of meiosis I. |
| Reason (R): |
At anaphase I, the sister chromatids separate and move towards opposite poles. |
| 1. |
Both (A) and (R) are False |
| 2. |
(A) is True but (R) is False |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
42. Given below are two statements : one is labelled as Assertion
(A) and the other is labelled as Reason
(R).
| Assertion (A): |
Most food chains or webs have only about four or five trophic levels. |
| Reason (R): |
Trophic efficiencies are generally only about 10% in different ecosystems. |
In the light of the above statements, choose the most appropriate answer from the options given below :
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
43. Regarding enzyme catalysis, all of the following will be true except:
| 1. |
The rate of reaction initially increases with an increase in substrate concentration |
| 2. |
It speeds up the rate of reaction by decreasing the requirement for activation energy |
| 3. |
If the associated co-factor is removed, the enzyme activity is enhanced |
| 4. |
Enzymes have an optimum temperature and pH, at which they act most efficiently |
44. Given below are two statements:
| Statement I: |
Chlorella is used as food supplement by space travellers |
| Statement II: |
Chlorella is a unicellular algae |
| 1. |
Both I and II are true and II correctly explains I |
| 2. |
I is true and II is false |
| 3. |
Both I and II are true but II does not explain I |
| 4. |
Both I and II are false |
45. Consider the two statements:
| Assertion (A): |
Current availability of CO2 levels is limiting to the C3 plants. |
| Reason (R): |
Carbon dioxide is the major limiting factor for photosynthesis. |
| 1. |
(A) is True but (R) is False |
| 2. |
Both (A) and (R) are True and (R) explains (A) |
| 3. |
Both (A) and (R) are True but (R) does not explain (A) |
| 4. |
(A) is False but (R) is True |
46. Polymerase chain reaction can amplify DNA, producing about 1 billion copies in 30 cycles. The correct sequence of the steps of this reaction is:
| 1. |
Denaturation → Annealing of primers → Extension of primers |
| 2. |
Denaturation → Extension of primers → Annealing of primers |
| 3. |
Extension of primers → Annealing of primers → Denaturation |
| 4. |
Annealing of primers → Extension of primers → Denaturation |
47. Column I shows characteristics of members of Red algae. Match each item in Column I with the one in Column II and select the correct match from the codes given:
|
Column I |
|
Column II |
| A. |
Pigment |
P. |
Fucoxanthin |
| B. |
Stored food |
Q. |
Phycoerythrin |
| C. |
Cell wall |
R. |
Floridean starch |
| D. |
Flagella |
S. |
Laminarin |
|
|
T. |
Polysulfate esters |
|
|
U. |
Algin |
|
|
V. |
2-8. equal, apical |
|
|
W. |
Absent |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
R |
T |
W |
| 2. |
P |
S |
U |
V |
| 3. |
Q |
R |
U |
W |
| 4. |
P |
S |
T |
V |
48. Identify the correct statements:
| I: |
Wheat, rice and oat plants represent C3 plants. |
| II: |
Sugarcane and corn represent C4 plants. |
1. Only
I
2. Only
II
3. Both
I and
II
4. Neither
I nor
II
49. Mechanical support to the growing parts of the plant such as young stem and petiole of leaf is provided by a living tissue called
| 1. |
Collenchyma |
2. |
Aerenchyma |
| 3. |
Chlorenchyma |
4. |
Sclerenchyma |
50. When proteins are respiratory substrates, the respiratory quotient is about:
| 1. |
0.7 |
2. |
0.9 |
| 3. |
1.0 |
4. |
4.0 |
Zoology - Section A
51. Memory cells:
| I: |
are produced in humoral immune response |
| II: |
are produced in cell mediated immune response |
| III: |
are responsible for passive immunity |
1. Only
I and
II are correct
2. Only
I and
III are correct
3. Only
II and
III are correct
4.
I,
II and
III are correct
52. Name a peptide hormone which acts mainly on hepatocytes, adipocytes and enhances cellular glucose uptake and utilisation.
1. Insulin
2. Glucagon
3. Secretin
4. Gastrin
53. Identify the incorrectly matched pair:
| 1. |
Down’s syndrome |
Trisomy 21 |
| 2. |
Sickle cell anaemia |
Inherited as an autosomal recessive condition |
| 3. |
Phenylketonuria |
Inherited as an autosomal dominant condition |
| 4. |
Klinefelter’s syndrome |
44 Autosomes + XXY |
54. Which of the following yields citric acid?
| 1. |
Penicillium citricum |
2. |
Aspergillus niger |
| 3. |
Saccharomyces |
4. |
Azospirilium |
55. Identify the incorrectly matched pair:
| 1. |
Myasthenia gravis |
Autoimmune disorder affecting neuro-muscular junction |
| 2. |
Tetany |
Spasms in muscle due to high Ca2+ in body fluids |
| 3. |
Osteoporosis |
Decreased bone mass is commonly due to decreased levels of estrogen |
| 4. |
Gout |
Inflammation of joints due to accumulation of uric acid crystals |
56. Identify the incorrect statement:
| 1. |
Lactational amenorrhoea can be effective for a maximum period of six months. |
| 2. |
Tubectomy and Vasectomy are easily reversible processes. |
| 3. |
Male barrier contraceptives protect against STIs. |
| 4. |
Oral contraceptive pills prevent ovulation. |
57. What event occurs during the joint diastole in the human heart?
1. Opening of AV valves
2. Closure of AV valves
3. Opening of semilunar valves
4. Closure of semilunar valves
58. The two polynucleotide strands in a dsDNA molecule are:
| 1. |
parallel and identical |
| 2. |
anti-parallel and identical |
| 3. |
parallel and complementary |
| 4. |
anti-parallel and complementary |
59. Which of the following would not be correct regarding vital capacity in human lungs?
| 1. |
It is the maximum volume of air a person can breathe in after a forced expiration. |
| 2. |
It includes ERV, TV, RV and IRV |
| 3. |
It can also be defined as the maximum volume of air a person can breathe out after a
forced inspiration. |
| 4. |
It can be calculated as the difference between total lung capacity and residual volume. |
60. Which of the following is wrongly matched in the given table?
|
|
Microbe
|
Product
|
Application
|
|
1.
|
Trichoderma polysporum
|
Cyclosporin A
|
Immunosuppressive drug
|
|
2.
|
Monascus purpureus
|
Statins
|
Lowering of blood cholesterol
|
|
3.
|
Streptococcus
|
Streptokinase
|
Removal of clot from blood vessel
|
|
4.
|
Clostridium butylicum
|
Lipase
|
Removal of oil stains
|
61. The second-most frequent mode of HIV transmission is:
| 1. |
through sexual contact with an infected person |
| 2. |
from mother to child during pregnancy, delivery, or breastfeeding |
| 3. |
via blood and blood products |
| 4. |
vector borne |
62. The human placenta does not secrete:
| 1. |
Oestrogen |
2. |
Progesterone |
| 3. |
Thyroxine |
4. |
Human placental lactogen |
63. Match each item in
Column-I with one given in
Column-II and select the correct match from the codes given:
Column-I
[Organism] |
Column-II
[Mechanism of sex determination] |
| A. |
Honey bees |
P. |
Genic balance |
| B. |
Grasshopper |
Q. |
ZZ-ZW, female heterogamety |
| C. |
Birds |
R. |
XX-XO male heterogamety |
| D. |
Fruit fly |
S. |
Haplo-diploidy |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
P |
S |
R |
| 2. |
R |
P |
Q |
S |
| 3. |
P |
Q |
R |
S |
| 4. |
S |
R |
Q |
P |
64. Identify the incorrectly matched pair:
| 1. |
Lysosomes |
Carry acid hydrolases |
| 2. |
Ribosomes |
Double membrane bound cell organelles |
| 3. |
Golgi apparatus |
Important site of formation of glycoproteins and glycolipids |
| 4. |
Amyloplasts |
Plastids that store carbohydrates |
65. Consider the given two statements:
| Statement I: |
Predators in nature are ‘prudent’. |
| Statement II: |
Prey species have evolved various defenses to lessen the impact of predation. |
1.
Statement I is correct;
Statement II is correct
2.
Statement I is correct;
Statement II is incorrect
3.
Statement I is incorrect;
Statement II is correct
4.
Statement I is incorrect;
Statement II is incorrect
66. All the following statements regarding histones, the proteins used in packaging of DNA, are correct except:
| 1. |
They are rich in amino acids - Lysine and Arginine. |
| 2. |
They carry a positive charge on the side chain. |
| 3. |
They are organized to form a unit of 8 molecules. |
| 4. |
The pH of histones is slightly acidic. |
67. DNA can be visualized through UV rays if it is stained with:
| 1. |
Ethidium bromide |
2. |
Polyethylene glycol |
| 3. |
Tritiated thymidine |
4. |
Colchicine
|
68. A chronic respiratory disorder, commonly caused by cigarette smoking but rarely due to deficiency of α-1 antitrypsin enzyme, is:
| 1. |
Bronchial asthma |
2. |
Tuberculosis |
| 3. |
Emphysema |
4. |
Pneumoconiosis |
69. Cr-Jacob disease (CJD) in humans is caused by:
| 1. |
Prions |
2. |
Viroids |
| 3. |
Virus |
4. |
Lichens |
70. The secondary oocyte completes the second meiotic division:
1. during the fetal life
2. before puberty
3. at the time of ovulation
4. at the time of fertilization
71. Tendons and ligaments are examples of:
1. Loose connective tissue
2. Specialized connective tissue
3. Dense regular connective tissue
4. Dense irregular connective tissue
72. Cu ions released by IUDs like Multiload 375 and CuT:
| 1. |
prevent ovulation |
| 2. |
suppress sperm motility and fertilising capacity |
| 3. |
prevent implantation |
| 4. |
make cervix hostile to sperms |
73. Inspiration is initiated by:
| 1. |
the contraction of diaphragm which increases the volume of thoracic chamber in the antero-posterior axis. |
| 2. |
the contraction of diaphragm which increases the volume of thoracic chamber in the dorso-ventral axis. |
| 3. |
the contraction of external inter-costal muscles which increases the volume of the thoracic chamber in the dorso-ventral axis. |
| 4. |
the contraction of external inter-costal muscles which increases the volume of the thoracic chamber in the antero-posterior axis. |
74. Variable number of tandem repeats (VNTR), a location in DNA where a short nucleotide sequence is organized as a tandem, repeats with variations in length between individuals, is useful in:
1. amplifying DNA
2. DNA fingerprinting
3. denaturing DNA
4. supercoiling of DNA
75. Ovary does not secrete:
1. Estrogen
2. Progesterone
3. Relaxin
4. Human chorionic gonadotropin
76. Chordates are not characterised by:
| 1. |
Presence of notochord |
| 2. |
Ventral, solid and double central nervous system |
| 3. |
Pharynx perforated by gill slits |
| 4. |
A post-anal tail |
77. In 'rivet popper hypothesis', Paul Ehrlich compared the rivets in an airplane to
| 1. | species within a genus | 2. | genetic diversity |
| 3. | ecosystem | 4. | genera within a family |
78. Frogs:
| I: |
have a constant body temperature and are warm blooded. |
| II: |
have the ability to change the colour to hide them from their enemies (camouflage). |
| III: |
are not seen during peak summer and winter. |
1. Only
I and
II are correct
2. Only
I and
III are correct
3. Only
II and
III are correct
4.
I,
II and
III are correct
79. Identify the correctly matched pairs:
|
Layer in the wall of artery/vein |
Made up of |
| I: |
Tunica intima |
Squamous epithelium |
| II: |
Tunica media |
Skeletal muscle/elastic fibres |
| III: |
Tunica externa |
Fibrous connective tissue |
1. Only
I and
II are correct
2. Only
I and
III are correct
3. Only
II and
III are correct
4.
I,
II and
III are correct
80. Which of the following is an egg laying mammal?
1. Ornithorhynchus
2. Ichthyophis
3. Hippocampus
4. Macropus
81. The resting axonal membrane is:
| 1. |
impermeable to the flux of both sodium and potassium ions |
| 2. |
freely permeable to the flux of both sodium and potassium ions |
| 3. |
relatively more permeable to the flux of potassium and nearly impermeable to the flux of sodium ions |
| 4. |
relatively more permeable to the flux of sodium and nearly impermeable to the flux of potassium ions |
82. One of the following biomolecules is neither a macromolecule nor a polymer. Identify it:
1. Proteins
2. Lipids
3. Carbohydrates
4. Nucleic acids
83. Cancer cells exhibit:
| 1. |
Contact inhibition |
2. |
Low division rate |
| 3. |
Metastasis |
4. |
High degree of differentiation |
84. Match each item in
Column-I with one in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Sertoli cells |
P. |
Secretion of androgens |
| B. |
Leydig cells |
Q. |
Spermiogenesis |
| C. |
Uterine Myometrium |
R. |
Implantation |
| D. |
Uterine Endometrium |
S. |
Parturition |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
R |
Q |
S |
| 2. |
R |
S |
P |
Q |
| 3. |
Q |
P |
S |
R |
| 4. |
S |
Q |
R |
P |
85. Identify the incorrectly matched pair:
| 1. |
Taylor |
Radioactive thymidine |
| 2. |
Meselson and Stahl |
15N |
| 3. |
Hershey and Chase |
32P and 35S |
| 4. |
Griffith |
14C |
Zoology - Section B
86. A feature of ‘r’ strategists would be:
| 1. | few offspring | 2. | little parental care |
| 3. | sigmoid growth curves | 4. | a longer generation time
|
87. Which of the following is/are cause(s) of biodiversity losses?
1. Over-exploitation, habitat loss and fragmentation
2. Climate change only
3. Over-exploitation only
4. Habitat loss and fragmentation only
88. Nissl’s granules are found in:
I: Soma of the neuron
II: Dendrites of the neuron
III: Axon of the neuron
| 1. |
Only I and II |
2. |
Only I and III |
| 3. |
Only II and III |
4. |
I, II and III |
89. The respiratory volume that averages 1100 mL to 1200 mL in a healthy adult male will be:
1. Tidal volume
2. Inspiratory Reserve Volume
3. Expiratory Reserve Volume
4. Residual Volume
90. In inspiration during pulmonary ventilation, the correct order of the given events is:
a. air flows into the lungs
b. alveolar volume increases
c. thoracic volume increases
d. pleural pressure decreases
e. alveolar pressure decreases
| 1. |
a, b, c, d, e |
2. |
b, e, a, c, d |
| 3. |
c, d, b, e, a |
4. |
d, e, b, a, c |
91. Why is a nucleic acid probe required in many genetic engineering experiments?
| 1. |
To clone genes |
| 2. |
To produce a large amount of DNA from a tiny amount of DNA |
| 3. |
To make DNA on the template of RNA |
| 4. |
To identify genes that have been separated by electrophoresis, or mRNA molecules through in-situ hybridization |
92. Which statements are correct regarding centriole?
| I: |
Centrioles are typically made up of nine sets of short microtubule triplets, arranged in a cylinder. |
| II: |
The main function of centrioles is to produce cilia during interphase and the aster and the spindle during cell division. |
| III: |
Centrioles start duplicating when DNA replicates. |
| 1. |
Only I and II |
2. |
Only I and III |
| 3. |
Only II and III |
4. |
I, II and III |
93. Which part of the nephron plays a significant role in the maintenance of high osmolarity of medullary interstitial fluid?
1. Proximal convoluted tubule
2. Ascending limb of Loop of Henle
3. Descending limb of Loop of Henle
4. Distal convoluted tubule
94. Identify the incorrect statement regarding human skeleton:
| 1. |
The total number of true ribs and floating ribs are 7 pairs and 2 pairs respectively |
| 2. |
Glenoid cavity and acetabulum are respectively present in scapula and coxal bone |
| 3. |
The number of cervical vertebrae in Giraffe are more than the cervical vertebrae in humans |
| 4. |
Sutures between cranial bones are examples of fibrous joints |
95. Correctly identify the match for A-G labelled in the given figure:

|
1 |
2 |
3 |
4 |
| A |
Atrial natriuretic peptide |
EPO |
Atrial natriuretic peptide |
EPO |
| B |
Vaso-
constriction |
Vaso-
dilation |
Vaso-
dilation |
Vaso-
constriction |
| C |
Increase in blood pressure |
Decrease in blood pressure |
Decrease in blood pressure |
Increase in blood pressure |
| D |
Decrease in renin |
Decrease in renin |
Decrease in renin |
Decrease in renin |
| E |
Decrease in Angiotensin II and aldosterone |
Decrease in Angiotensin II and aldosterone |
Decrease in Angiotensin II and aldosterone |
Decrease in Angiotensin II and aldosterone |
| F |
Diuresis |
Concentration of urine |
Diuresis |
Concentration of urine |
96. Potassium ions:
| I: |
are filtered at the glomerulus |
| II: |
are reabsorbed at the PCT |
| III: |
are secreted at DCT |
Correct statements are:
| 1. |
I & II only |
2. |
I & III only |
| 3. |
II & III only |
4. |
I, II & III |
97. Which one of the following statements is correct with respect to immunity?
| 1. |
Preformed antibodies need to be injected to treat the bite by a viper snake. |
| 2. |
The antibodies against smallpox pathogen are produced by T - lymphocytes. |
| 3. |
Antibodies are protein molecules, each of which has four light chains. |
| 4. |
Rejection of a kidney graft by the recipient is the function of B-lymphocytes. |
98. Regarding the stages in the life cycle of
Plasmodium:
| I: |
Sporozoite is the infective stage for man |
| II: |
In human body it first reproduces asexually in liver cells |
| III: |
fertilization takes place in the gut of the mosquito |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
99. During the elongation step of bacterial translation peptide bond formation, is catalyzed by a ribozyme which is:
| 1. |
the 23S ribosomal RNA in the 50S ribosomal subunit |
| 2. |
the 23S ribosomal RNA in the 30S ribosomal subunit |
| 3. |
the 16S ribosomal RNA in the 50S ribosomal subunit |
| 4. |
the 16S ribosomal RNA in the 30S ribosomal subunit |
100. Sucrose is converted into glucose and fructose by the enzyme:
1. Lactase
2. Zymase
3. Diastase
4. Invertase
Chemistry - Section A
101. Given below are two statements:
| Statement I: |
DDT (Dichloro‐diphenyl‐trichloroethane (\(\mathrm{C}_{14} \mathrm{H}_9 \mathrm{Cl}_5\)) is a herbicide. |
| Statement II: |
Triiodomethane (Iodoform) was used earlier as an antiseptic,
but the antiseptic properties are due to the liberation of free iodine and not to iodoform itself. |
1. Both
Statement I and
Statement II are true
2. Both
Statement I and
Statement II are false
3.
Statement I is true, but
Statement II is false
4.
Statement I is fals,e but
Statement II is true
102. The compound \(PbI_4\) does not exist because:
1. \(Pb^{2+}\) is more stable than \(Pb^{4+}\).
2. \(I^-\) is better reducing agent.
3. \(Pb^{4+}\) is more stable than \(Pb^{2+}\).
4. \(I^-\) is better oxidising agent.
103. The anion of acetylacetone (acac) forms Co(acac)3 chelate with Co3+. The rings of the chelate are:
1. Five membered
2. Four membered
3. Six membered
4. Three membered
104. The decreasing order of the rate of the electrophilic aromatic substitution reaction is:

1. B > C > A > D > E
2. A > B > C > D > E
3. E > D > C > B > A
4. A > B > D > C > E
105. Van't Hoff factor for \(SrCl_2\) at 0.01 M is 1.8. Percent dissociation of \(SrCl_2\) at this concentration is:
1. 70
2. 55
3. 40
4. 80
106. What is the correct order of basicity for the given compounds?
1. C > A > B > D
2. D > B > A > C
3. B > A > C > D
4. D > C > A > B
107. In Williamson’s synthesis, ethoxy ethane is prepared by:
| 1. |
Heating sodium ethoxide with ethyl bromide. |
| 2. |
Passing ethanol over heated alumina. |
| 3. |
Treating ethyl alcohol with an excess of conc. H2SO4 at 430-440 K. |
| 4. |
Heating ethanol with dry \(Ag_2O\) |
108. Match the complexes given in
Column-I with their characteristics given in
Column-II and assign the correct code:
|
Column-I |
|
Column-II |
| (P) |
\(\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}\) |
(I) |
Spin magnetic moment\(=\sqrt{8} \mathrm{BM}\) |
| (Q) |
\(\mathrm{Fe}(\mathrm{CO})_5 \) |
(II) |
\(s p^3\) hybridized state of central metal |
| (R) |
\(\left[\mathrm{MnBr}_4\right]^{2-} \) |
(III) |
\(d s p^2\) hybridized state of central metal |
| (S) |
\(\left[\mathrm{Ni}\left(\mathrm{NH}_3\right)_6\right]^{2+} \) |
(IV) |
Trigonal bipyramidal complex |
|
P |
Q |
R |
S |
| 1. |
III |
IV |
I |
II |
| 2. |
III |
IV |
II |
I |
| 3. |
I |
IV |
III |
II |
| 4. |
II |
IV |
III |
I |
109. The pair of species having similar magnetic moments is:
1. \(Ti^{3+} , V^{3+}\)
2. \(Cr^{3+} ,Mn^{2+}\)
3. \(Mn^{2+} , Fe^{3+}\)
4. \(Fe^{2+} ,Mn^{2+}\)
110. The conjugate base of hydrazoic acid is:
| 1. |
 |
2. |
\(N_3^-\) |
| 3. |
\(N_2^-\) |
4. |
\(N^{3-}\) |
111. The incorrect statement for lanthanides among the following statements is:
| 1. |
Yb2+ and Lu3+ are diamagnetic and paramagnetic, respectively. |
| 2. |
The most common stable oxidation state is +3. |
| 3. |
Many trivalent lanthanoid ions are coloured both in the solid state and in aqueous solutions. |
| 4. |
Mixed oxides of lanthanoids are employed as catalysts in petroleum cracking. |
112.
Which among the following has the greatest number of atoms at NTP?
| 1. |
5 ml CH4 |
2. |
20 ml N2 |
| 3. |
1 ml H2O |
4. |
10 ml CO2 |
113. Which compound is least reactive towards
\(S_N1\) reaction with aqueous
\(KOH\) ?
| 1. |
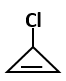 |
2. |
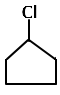 |
| 3. |
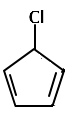 |
4. |
\((CH_3)_2CH-Cl\) |
114. Given below are two statements:
| Assertion (A): |
PCl3Br2 has zero dipole moment. |
| Reason (R): |
Br atoms occupy axial position while Cl atoms occupy equatorial position. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
115. Identify the incorrect statements regarding the importance of coordination compounds in biological systems.
| 1. |
Vitamin B-12 used to prevent anaemia, is a complex compound of zinc. |
| 2. |
Haemoglobin is the red pigment of blood and contains iron. |
| 3. |
Chlorophyll is the green pigment of plants and contains magnesium. |
| 4. |
All of the above are correct. |
116. Which conditions allow the hydrolysis of alkyl cyanides?
1. Acidic medium
2. Alkaline medium
3. Acidic as well as alkaline medium
4. Neither acidic nor alkaline medium
117. Which will have the largest number of atoms among the following?
| 1. |
1 g Au (s)
|
2. |
1 g Na (s)
|
| 3. |
1 g Li (s)
|
4. |
1 g of Cl2(g) |
118. For A+B C+D, H = -20 kJmol-1 the activation energy of the forward reaction is 85 kJ mol-1. The activation energy for backward reaction is...... kJ mol-1
119. pH of \(Ba(OH)_2\) is 11. Then the solubility product of \(Ba(OH)_2\) is:
1. \(10^{-6}\) M3
2. \(5 \times 10^{-7}\) M3
3. \(5 \times 10^{-10}\) M3
4. \(10^{-10}\) M3
120. The ion, from the ions given below, that has the same electron configuration as the noble gas argon is:
1. Na+
2. P2–
3. Al3+
4. Cl−
121. Coordination compound [Co(NH
3)
4(NO
2)
2]Cl exhibits:
| 1. |
Ionization isomerism, geometrical isomerism, and optical isomerism |
| 2. |
Linkage isomerism, geometrical isomerism, and optical isomerism |
| 3. |
Linkage isomerism, ionization isomerism, and optical isomerism |
| 4. |
Linkage isomerism, ionization isomerism, and geometrical isomerism |
122. S-S bond is present in:
1. \(H_2SO_5\)
2. \(H_2S_2O_6 \)
3. \(H_2S_2O_7\)
4. \(H_2S_2O_8\)
123. Two metals ‘A’ and ‘B’ have standard reduction potential (E
ored) values equal to 0.36 V and 1.23 V, respectively. The correct cell equation will be:
| 1. |
\(A+B^{2+} \rightarrow\)
\( A^{2+}+B \) |
2. |
\(A^{2+}+B \rightarrow\)
\( A+B^{2+} \) |
| 3. |
Both 1 & 2 |
4. |
None of the above. |
124. If the value of m for an electron is +3, it may be found in:
| 1. |
4s orbital |
2. |
4p orbital |
| 3. |
In any f-orbital |
4. |
In any d-orbital |
125. Which of the following species is not paramagnetic?
1. O2
2. B2
3. NO
4. CO
126. Benzene reacts with an excess of chlorine in the presence of ultraviolet light to produce:
1. Hexachloro benzene
2. p-Chlorotoluene
3. Benzene hexachloride
4. Chlorobenzene
127. The value of k at T→ ∞ in the Arrhenius equation would be:
| 1. |
\(A \) |
2. |
\(-\mathrm{A} \) |
| 3. |
\(\mathrm{e}^A \) |
4. |
\(\mathrm{e}^{-A}\) |
128. 
The IUPAC name of the above-given compound is:
1. 2,3,4-Trimethyl-3-(1-Methyl ethyl)pent-2-ene
2. 2,3,3-Trimethyl-3-(1-Methyl propyl)pent-2-ene
3. 2,3,4-Trimethyl-2-(1-Methyl ethyl)pent-2-ene
4. 2,4,4-Trimethyl-3-(1-Methyl ethyl)pent-2-ene
129. Consider the following compounds. The Kjeldahl method is not applicable for estimating nitrogen in how many of the given compounds?
130. The free energy change due to a reaction is zero when :
| 1. |
The reactants are initially mixed |
| 2. |
A catalyst is added |
| 3. |
The system is at an equilibrium |
| 4. |
The reactants are completely consumed |
131. In Cannizarro's reaction, we use 50% NaOH or KOH instead of dilute NaOH or KOH. The reason for using the 50% NaOH or KOH in Cannizarro's reaction is:
| 1. |
To convert monoanion to dianion |
| 2. |
To prevent the abstraction of hydrogen atom which is attached with carbonyl carbon |
| 3. |
To enhance the release of H+ ion |
| 4. |
None of the above |
132. Consider the half-cell reduction reaction:
\(\text{Mn}^{2+}+2e^-\rightarrow \text{Mn},\ E^{0}= -1.18~ \text V \)
\(\text{Mn}^{2+}\rightarrow \text{Mn}^{3+}+e^-,\ E^{0}= -1.51~ \text V \)
The
\(E^{0}\) for the reaction
\(\mathrm{3\ Mn^{2+}\rightarrow Mn^{0}+2Mn^{3+} }\) and possibility of the forward reaction are respectively:
| 1. |
–4.18 V and Yes |
2. |
+0.33 V and Yes |
| 3. |
+2.69 V and No |
4. |
–2.69 V and No |
133. The oxidation number of Cl in \(CaOCl_2\) is :
1. Zero
2. -1
3. +1
4. -1 as well as +1
134.
|
List-I |
|
List-II |
| A. |
\(XeF_4 \) |
(P) |
T-shape |
| B. |
\(SF_4 \) |
(Q) |
See-saw |
| C. |
\(NH^+_4 \) |
(R) |
Square planar |
| D. |
\(BrF_3 \) |
(S) |
Tetrahedral |
Match the species given in List I above with the geometries given in List II and identify the correct option from the following:
1. A → P, B →Q, C→R, D→S
2. A → R, B →Q, C→S, D→P
3. A → Q, B →P, C→S, D→R
4. A → S, B →R, C→P, D→Q
135. The most acidic hydrocarbon among the following is:
Chemistry - Section B
136. Which of the following aqueous solutions exhibits the highest boiling point?
| 1. |
1.0 M NaOH |
2. |
1.0 M \(Na_2SO_4~~~~~~\) |
| 3. |
1.0 M \(NH_4NO_3~~~~\) |
4. |
1.0 M KNO3 |
137. Given the reaction, \(A + B \rightarrow Products,\) , when concentration of A is fixed and concentration of B is increased 3 times then rate of reaction increases 27 times. Now, when the concentration of A and B both are doubled, then the rate of reaction increases 8 times. The rate law for the reaction will be:
1. \(Rate = K [A]^2[B] \)
2. \(Rate = K [B]^3 \)
3. \(Rate = K [A]^3[B] \)
4. \(Rate = K [A]^3[B]^2\)
138. The shape of IO2F2- ion is:
1. Linear
2. Trigonal bipyramidal
3. T-shaped
4. See-saw
139. Which of the following compounds is most reactive towards nucleophilic substitution reaction?
1. \(CH_3COCH_3\)
2. \(CH_3CHO\)
3. \(C_6H_5CHO\)
4. \(C_6H_5COCH_3\)
140. Which of the following order is incorrect?
| 1. |
\(HI > HBr > HCl > HF ~~\text{(Acidic strength)}\) |
| 2. |
\(F_2 > Cl_2 > Br_2 > I_2 ~~\text{(Bond Energy )}\) |
| 3. |
\(HF > HI > HBr> HCl ~~\text{(Boiling point)}\) |
| 4. |
\(F_2 > Cl_2 > Br_2 > I_2 ~~\text{(oxidising Power )}\) |
141.
| Assertion (A): |
Conductivity decreases with the decrease in the concentration of both the weak and strong electrolytes. |
| Reason (R): |
The number of ions per unit volume that carry the current in a solution decreases on dilution. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
142. The products formed after ozonolysis of Pent-2-ene are:
| 1. |
Ethanal and Methanal |
2. |
Ethanal and Propanal |
| 3. |
Ethanal and Butanal |
4. |
Ethanal and Ethanol |
143. The main reason for a larger number of oxidation states exhibited by the actinoids than the corresponding lanthanoids is:
| 1. |
The lesser energy difference between 5f and 6d orbitals than 4f and 5d orbitals |
| 2. |
Larger atomic size of actinoids than the lanthanoids |
| 3. |
More energy difference between 5f and 6d orbitals than between 4f and 5d orbitals |
| 4. |
Greater reactive nature of the actinoids than the lanthanoids |
144. What is the IUPAC name of the following compound?

1. 3-Bromo-1, 2-dimethylbut-1-ene
2. 4-Bromo-3-methylpent-2-ene
3. 2-Bromo-3-methylpent-3-ene
4. 3-Bromo-3-methyl-1, 2-dimethylprop-1-ene
145. At equilibrium, which of the following is always zero?
| 1. |
\(\Delta H\) |
2. |
\(\Delta S_{Total}\) |
| 3. |
\(\Delta S_{System}\) |
4. |
\(\Delta G^o\) |
146. Faraday’s laws of electrolysis are associated with:
1. The atomic number of the reactants.
2. The atomic number of the anion.
3. The equivalent weight of the electrolyte.
4. The speed of the cation
147. pH of a salt of a solution of weak acid and weak base depends upon:
1. Concentration of salt
2. \(K_a\) and \(K_b\) values of weak acid and weak base
3. Both 1 and 2
4. None of the above
148. Consider the following reaction
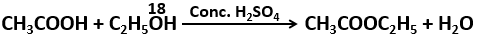
O
18 appears in:
1.
\(CH_3COOC_2H_5\)
2.
\(H_2O\)
3. Both 1 and 2
4. Cannot be predicted from the given reaction
149. A closed container has a mixture of 48 g of sodium hydroxide, 52 g of water, and 132 g of ammonium sulphate. The number of moles of oxygen atoms present in the container are:
1. 5
2. 7
3. 8
4. 10
150. The \(\Delta_0\) value of \([Ni(H_2O)_6]^{2+}\) is \(8500 ~cm^{-1}\) . The \(\Delta_0\) values for \([NiCl_6]^{4-}\) and \([Ni(NH_3)_6]^{2+}\) compared to \([Ni(H_2O)_6]^{2+}\)are:
1. Higher and lower, respectively
2. Lower and higher, respectively
3. Higher in both complex ions
4. Lowering both complex ions
Physics - Section A
151. A force of
\(100~\text N\) acts on a block, always acting opposite to its velocity. The block moves from
\(x=4~\text m\) to
\(x=10~\text m\) and then to
\(x=6~\text {m.}\) The work done by the force is:
1.
\(200~\text J\)
2.
\(-200~\text J\)
3.
\(1000~\text J\)
4.
\(-1000~\text J\)
152. If two identically shaped rods are joined end to end and compressive forces are applied to the system, the compressive strain will be:

| 1. |
larger in the rod with a larger Young's modulus |
| 2. |
larger in the rod with a smaller Young's modulus |
| 3. |
equal in both the rods |
| 4. |
negative in the rod with a smaller Young's modulus |
153. A particle moves in a circle with a uniform speed of \(3~\text{m/s}\) and its acceleration is \(6~\text{m/s}^2. \) The time period of its motion is (in seconds):
1. \(4\pi\)
2. \(2\pi\)
3. \(\pi\)
4. \(\pi/2\)
154. Two infinite positively charged wires lie in the
\(x\text-y\) plane, both carrying the same quantity of charge per unit length. One wire passes through
\(A(0,a)\) parallel to the
\(x\text-\)axis, while the other wire passes through
\(B(-a,0)\) parallel to the
\(y\text-\)axis. The electric field at the origin is along:
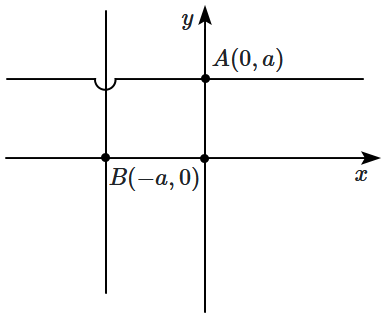 1. \(\hat i - \hat j\)
1. \(\hat i - \hat j\)
2. \(-(\hat i + \hat j)\)
3. \(-\hat i\)
4. \(-\hat j\)
155. A conducting circular wire of radius
\(r\) is moving with constant velocity
\(v\) towards the right in a uniform magnetic field
\(B.\) We consider two points
\(X,Y\) such that chord
\(XY\) is perpendicular to the velocity
\(v\) and is at a distance
\(x\) from the centre
\((O)\) of the circle. The EMF induced between
\(X,Y\) is
\(\varepsilon.\) Then,
\(\varepsilon\) is proportional to:

| 1. |
\(x\) |
2. |
\(\sqrt{r^2-x^2}\) |
| 3. |
\(r\) |
4. |
\(x\sqrt{r^2-x^2}\) |
156. The capacitance of the system (shown in the figure below) of parallel conducting plates, between the two outer plates
\((X)\) and the inner plate
\((Y)\) is (plate area=
\(A,\) plate separation
\(d,2d:\) small)

| 1. |
\(\dfrac{3\varepsilon_0A}{2d}\) |
2. |
\(\dfrac{4\varepsilon_0A}{3d}\) |
| 3. |
\(\dfrac{\varepsilon_0A}{3d}\) |
4. |
\(\dfrac{\varepsilon_0A}{2d}\) |
157. Two rods having coefficient of linear expansion
\(\alpha,3\alpha\) are connected end-on-end. The average coefficient of thermal expansion for the composite rod:
| 1. |
is \(2\alpha\) |
| 2. |
is \(4\alpha\) |
| 3. |
can be any value between \(\alpha\) and \(3\alpha\) |
| 4. |
can be any value between \(2\alpha\) and \(3\alpha\) |
158. Given below are two statements:
| Statement I: |
The gravitational force exerted by the Sun on the Earth is reduced when the Moon is between the Earth and the Sun. |
| Statement II: |
The gravitational force exerted by the Sun on the Earth is reduced when the Moon is opposite to the Sun, relative to the Earth. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
159. A current
\(I,\) flowing through a long straight wire produces a magnetic field of
\(4\times10^{-3}\) T at a point
\(P\), \(20\) cm away. If the wire is replaced by a circular wire of radius
\(20\) cm, centred at
\(P\) and carrying the same current
\(I,\) then the magnetic field at
\(P\) will be:
| 1. |
\(4\pi\times10^{-3}\) T |
| 2. |
\(\dfrac4\pi\times10^{-3}\) T |
| 3. |
\(4\times10^{-3} \) T |
| 4. |
\(2\times10^{-3}\) T |
160. A hydrogen atom
\((\mathrm{H})\) and a helium
\((\mathrm{He})\) atom, both have the same kinetic energy (they are non-relativistic). Their de-Broglie wavelengths are in the ratio:
\(\Large\frac{\lambda_\mathrm{H}}{\lambda_{\mathrm{He}}}=\)
| 1. |
\(4\) |
2. |
\(\dfrac14\) |
| 3. |
\(2\) |
4. |
\(\dfrac12\) |
161. Given below are two statements:
| Assertion (A): |
Parallel rays of light of different colours fail to converge to a point after reflection from a spherical mirror. |
| Reason (R): |
The refractive index of any material depends on the frequency of light. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
162. The minimum value of the moment of inertia for a uniform disc of mass
\(M\) and radius
\(R\) about any possible axis is:
| 1. |
\(\dfrac{MR^2}{2}\) |
2. |
\(\dfrac{MR^2}{4}\) |
| 3. |
\(\dfrac{MR^2}{8}\) |
4. |
\(\dfrac{MR^2}{6}\) |
163. Electrons
(mass \(m\)) moving with a velocity
\(v\) are incident normally onto a single slit of width
\(d,\) and are detected on a screen placed at a distance
\(D\) behind the slit. The central point on the screen where most of the electrons are detected is
\(O.\) The closest point to
\(O\) where no electrons are detected is
\(X.\) Then
\(OX\) equals:

| 1. |
\(\dfrac{hD}{mvd}\) |
2. |
\(\dfrac{hD}{2mvd}\) |
| 3. |
\(\dfrac{2hD}{mvd}\) |
4. |
\(\dfrac{3hD}{2mvd}\) |
164. Trains travel between station
\(A\) and station
\(B\): on the way up (from
\(A~\text{to}~B \)) - they travel at a speed of
\(80\text{ km/h},\) while on the return trip the trains travel at twice that speed. The services are maintained round the clock. Trains leave station
\(A\) every
\(30\text{ min}\) for station
\(B\) and reach
\(B\) in
\(2\text{ hrs.}\) All trains operate continuously, without any rest at
\(A\) or
\(B.\)
| 1. |
the frequency of trains leaving \(B\) must be twice as much as \(A\). |
| 2. |
the frequency of trains leaving \(B\) must be half as much as \(A\). |
| 3. |
the frequency of trains leaving \(B\) is equal to that at \(A\). |
| 4. |
the situation is impossible to maintain unless larger number of trains are provided at \(A\). |
165. An air bubble in soap water and a soap bubble in the air of the same size have excess pressure
\((\Delta P).\) Then:
| 1. |
\(\Delta P_\text{air bubble}>\Delta P_\text{soap bubble}\) |
| 2. |
\(\Delta P_\text{soap bubble}>\Delta P_\text{air bubble}\) |
| 3. |
\(\Delta P_\text{soap bubble}=\Delta P_\text{air bubble}\) |
| 4. |
\(\Delta P_\text{soap bubble}+\Delta P_\text{air bubble}=0\) |
166. A particle moving initially with a speed of \(3~\text{m/s}\) around a circle accelerates so as to increase its speed by \(3~\text{m/s}\) every half-revolution. If its initial kinetic energy is \(E,\) its kinetic energy after a complete revolution will be:
1. \(2E\)
2. \(3E\)
3. \(9E\)
4. \(16E\)
167. The potential on the surface of a uniform spherical volume charge distribution is \(10~\text{V}\); and it is observed that the potential at its centre is \(15~\text{V}\). If the radius of the sphere is halved, keeping the total charge constant, then the potential at its centre will be:
1. \(15~\text{V}\)
2. \(30~\text{V}\)
3. \(60~\text{V}\)
4. \(120~\text{V}\)
168. The current flowing through the resistance
\(R_1\), when
\(R_1=R_2\) and
\(R_3=R_4\) is:
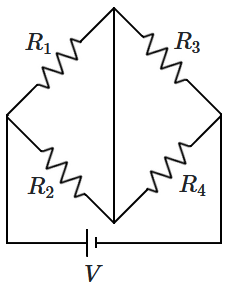
| 1. |
\(\frac{V}{4\left(R_1+R_3\right)}\) |
2. |
\(\frac{2 V}{\left(R_1+R_3\right)}\) |
| 3. |
\(\frac{V}{\left(R_1+R_3\right)}\) |
4. |
\(\frac{V}{2\left(R_1+R_3\right)}\) |
169. Given below are two statements:
| Assertion (A): |
Magnetic susceptibility is a pure number. |
| Reason (R): |
The value of magnetic susceptibility for vacuum is one. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
170. Exactly identical voltages are imposed on the system at
\(X, Y,\) and
\(Z:V_m \sin \omega t\). The peak voltage at
\(O\) is
\(V_o\). Then:

1.
\(V_o = V_m\)
2.
\(V_o < V_m \)
3.
\(V_o > V_m\)
4. any of the above can be possible.
171. The voltage across
\(AB\) i.e.,
\(V_{AB}\) is varied from
\(0\) V to
\(12\) V. The current through branch
\(XY~(i)\) is measured. Then:

| 1. |
\(i=0.05\) A if \(V_{AB}\geq10\) V and \(i=\dfrac{V_{AB}}{200~\Omega}\) otherwise |
| 2. |
\(i=0.05\) A if \(V_{AB}\leq10\) V and \(i=\dfrac{V_{AB}}{200~\Omega}\) otherwise |
| 3. |
\(i=0.05\) A |
| 4. |
\(i=\dfrac{V_{AB}}{200~\Omega}\) |
172. A waveform given by:
\(y=3A\sin(\omega t-kx)\)
is superposed with another waveform
\(y=4A\cos(\omega t-kx).\) The amplitude of the resulting waveform will be:
| 1. |
\(7A\) |
2. |
\(A\) |
| 3. |
\(3.5A\) |
4. |
\(5A\) |
173. On a rotating wheel with a moment of inertia,
\(2\) kg-m
2 about its vertical axis, a torque that can stop the wheel's rotation in one minute is
\(\dfrac{\mathit{\pi}}{15}~\text{N-m}\). The initial rotational speed of the wheel (in rpm) is:
| 1. |
\(20\) |
2. |
\(40\) |
| 3. |
\(60\) |
4. |
\(80\) |
174. Given below are two statements:
| Statement I: |
Given that the magnitude of the acceleration of a body is constant, the force acting on it must be constant. |
| Statement II: |
Newton's second law leads to the statement that the acceleration of a body is directly proportional to the net force acting on it. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
175. Given below are two statements:
| Statement I: |
Electromagnetic waves are radiated when a charged particle undergoes SHM. |
| Statement II: |
Electromagnetic waves propagate energy in the direction of the electric field of the wave. |
Choose the correct option from the given ones:
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
176. A \(3~\mu\text{F}\) capacitor is charged with \(6~\mu \text{C}\) and connected across a \(1~\text{mH}\) inductance. The rate of change of current is:
1. \(2\) A/s
2. \(2\times10^{-3}\) A/s
3. \(2\times10^{3}\) A/s
4. \(2\times10^{-6}\) A/s
177. Match the units mentioned in
List-I with the units in
List-II.
|
List-I |
|
List-II |
| (a) |
H/s |
(i) |
s2 |
| (b) |
H×A |
(ii) |
Wb |
| (c) |
H×F |
(iii) |
s |
| (d) |
\(\Omega\)×F |
(iv) |
\(\Omega\) |
| 1. |
(a)–(ii), (b)–(iv), (c)–(i), (d)–(iii) |
| 2. |
(a)–(iv), (b)–(ii), (c)–(i), (d)–(iii) |
| 3. |
(a)–(iv), (b)–(ii), (c)–(iii), (d)–(i) |
| 4. |
(a)–(ii), (b)–(iii), (c)–(iv), (d)–(i) |
178. The ratio of the
\(\text{RMS}\) speed
\((v_\text{RMS})\) of the molecules of an ideal gas to the speed of sound
\((v_s)\) in the gas, i.e.
\({\dfrac{v_\text{RMS}}{v_s}}{\small=}\)
| 1. |
\(\dfrac{3}{\gamma}\) |
2. |
\(\sqrt{\dfrac{3}{\gamma}}\) |
| 3. |
\(\dfrac{\gamma}{3}\) |
4. |
\(\sqrt{\dfrac{\gamma}{3}}\) |
179. A rod of mass
\(m\) length
\(L\) rotates about one of its ends, in its own plane. The angular speed of the rod is
\(\omega.\) The angular momentum of the rod is:
| 1. |
\(\dfrac{1}{4}mL^2\omega\) |
2. |
\(\dfrac{1}{3}mL^2\omega\) |
| 3. |
\(\dfrac{2}{3}mL^2\omega\) |
4. |
\(\dfrac{1}{12}mL^2\omega\) |
180. \(AB\) and
\(BC\) are a pair of mirrors inclined so that the angle between their planes is
\(60^{\circ}\), as shown in the figure. A ray of light
\(XY\) is incident on
\(AB\) and emerges as the ray
\(ZW\) after two reflections. If the incident ray is rotated so that
\(\angle AYX\) decreases by
\(15^{\circ}\), then
\(\angle WZC\):

| 1. |
increases by \(15^{\circ}\). |
2. |
increases by \(30^{\circ}\). |
| 3. |
decreases by \(15^{\circ}\). |
4. |
decreases by \(30^{\circ}\). |
181. A particle moves along a straight line with its velocity \((v)\) varying as the square root of its displacement \((x)\text:\) \(v\propto\sqrt x\)
Then its acceleration varies as:
1. \(\dfrac{1}{\sqrt x}\)
2. \(x^{3/2}\)
3. \(x^{-3/2}\)
4. \(x^0\)
182. The dynamic resistance of a diode is given by;
\(R=\dfrac{26}{i(\text{mA})}~\Omega,\) where
\(i\) is the diode current.
| Statement I: |
If the current through the diode increases, then the dynamic resistance decreases. |
| Statement II: |
If the temperature of the diode is increased, with the potential difference fixed, the dynamic resistance decreases. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
183. If
\(\Delta Q\) is the heat flowing out of a system,
\(\Delta W\) is the work done by the system on its surroundings, and
\(\Delta U\) is the decrease in internal energy of the system, then the first law of thermodynamics can be stated as:
| 1. |
\(\Delta Q=\Delta U+\Delta W\) |
| 2. |
\(\Delta U=\Delta Q+\Delta W\) |
| 3. |
\(\Delta U=\Delta Q-\Delta W\) |
| 4. |
\(\Delta U+\Delta Q+\Delta W=0\) |
184. \(\alpha \text-\)particles, \(\beta \text-\)particles and \(\gamma \text-\)rays, each have an energy of \(10~\text{MeV}.\) The increasing order of penetrating powers of the radiations will be:
1. \(\beta>~\alpha>~\gamma\)
2. \(\alpha<~\beta<~\gamma\)
3. \(\gamma=\beta<~\alpha\)
4. \(\alpha<~\beta=\gamma\)
185. Given below are two statements:
| Assertion (A): |
Bohr had to postulate that the electrons in stationary orbits around the nucleus do not radiate. |
| Reason (R): |
According to classical physics all accelerating electrons radiate. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
Physics - Section B
186. Four particles having identical charges are placed at the four corners of a square of side
\(2\) m. The potential at the centre of the square is
\(2\sqrt2\) V.
The field due to any charge at the location of its nearest neighbour is:
1.
\(1\) V/m
2.
\(0.5\) V/m
3.
\(0.25\) V/m
4.
\(0.125\) V/m
187. A string fixed at both ends is under tension
\(T.\) It has a length
\(L,\) and mass
\(m.\) The fundamental frequency of the vibration is:
| 1. |
\(\dfrac{ 1}{2L} \sqrt {\dfrac{T}{m}}\) |
2. |
\(\dfrac{1}{4 L} \sqrt{\dfrac{T}{m}}\) |
| 3. |
\(\dfrac{1}{2} \sqrt{\dfrac{TL}{2m}}\) |
4. |
\(\dfrac{1}{2} \sqrt{\dfrac{T}{m L}}\) |
188. A ray of light travelling in the air along the
\(x\)-axis encounters a medium, and the refracted ray is along
\(\hat i+\sqrt3\hat j.\) The refractive index of the medium is:
| 1. |
less than or equal to \(2\) |
| 2. |
greater than or equal to \(2\) |
| 3. |
equals \(2\) |
| 4. |
greater, less or equal to \(2\) |
189. Two particles of mass \(5~\text{kg}\) and \(10~\text{kg}\) respectively are attached to the two ends of a rigid rod of length \(1~\text{m}\) with negligible mass. The centre of mass of the system from the \(5~\text{kg}\) particle is nearly at a distance of:
1. \(50~\text{cm}\)
2. \(67~\text{cm}\)
3. \(80~\text{cm}\)
4. \(33~\text{cm}\)
190. An ideal monatomic gas and a diatomic gas, both undergo adiabatic expansion starting from the same point on the
\(P\)-\(V\) (indicator) diagram. The gases also undergo isothermal expansion. The curves are given by
\(a,b,c.\) Which of the following is correct?

| 1. |
\(a\)–isothermal, \(b\)–monatomic adiabatic, \(c\)–diatomic adiabatic |
| 2. |
\(a\)–monatomic adiabatic, \(b\)–diatomic adiabatic, \(c\)–isothermal |
| 3. |
\(a\)–diatomic adiabatic, \(b\)–monatomic adiabatic, \(c\)–isothermal |
| 4. |
\(a\)–isothermal, \(b\)–diatomic adiabatic, \(c\)–monatomic adiabatic |
191. A thin circular conducting wire is connected at
\(A,B\) where the smaller arc
\(AB\) represents
\(\dfrac14^{\text{th}}\) of the circumference. A current flows from
\(A\) to
\(B,\) dividing into two branches
\(i_1\) and
\(i_2\) at
\(A.\) The ratio
\(i_1:i_2\) equals:

| 1. |
\(3\) |
2. |
\(4\) |
| 3. |
\(\dfrac13\) |
4. |
\(1\) |
192. The energy of the block is \(E\), and the plane is smooth, the wall at the end \(B\) is smooth. Collisions with walls are elastic. The distance \(AB=l\), the spring is ideal and the spring constant is \(k\). The time period of the motion is:

| 1. |
\(2\pi\sqrt{\dfrac{m}{k}}\) |
| 2. |
\(\pi\sqrt{\dfrac{m}{k}}+l\sqrt{\dfrac{2m}{E}}\) |
| 3. |
\(2\pi\sqrt{\dfrac{m}{k}}+2l\sqrt{\dfrac{2m}{E}}\) |
| 4. |
\(\pi\sqrt{\dfrac{m}{k}}+l\sqrt{\dfrac{m}{2E}}\) |
193. A projectile is fired from the top of a cliff, the maximum range of the projectile being
\(1000\) m on level ground. The maximum range of the projectile, measured from the base of the cliff is:
| 1. |
greater than \(1000\) m |
| 2. |
less than \(1000\) m |
| 3. |
equal to \(1000\) m |
| 4. |
can be any of the above depending on the height of the cliff |
194. Water is under high pressure within a tank. When a hole is made at the top of the tank, a stream of water rises up vertically to a height
\(H\) above the hole. Assume the flow to be streamlined. The water pressure (gauge pressure) at the top of the tank, initially, was:
(\(\rho\) density of water)
| 1. |
\(H\rho g\) |
2. |
\(\dfrac{H\rho g}{2}\) |
| 3. |
\(2 H\rho g\) |
4. |
\(\sqrt 2 H \rho g\) |
195. A block of mass
\(5\) kg requires a minimum horizontal force of
\(20\) N to cause it to move. Take
\(g=10\) m/s2. Let
\(\mu_s\) be the coefficient of static friction, and
\(\mu\small{_ k}\) be that of kinetic friction.

1.
\(\mu_s=0.4\)
2.
\(\mu_s=\mu{\small_k}=0.4\)
3.
\(\mu{\small_ k}=0.4\)
4.
\(\mu_s\geq0.4\)
196. Consider a particle moving along a straight line under the action of a force which delivers constant power.
| Assertion (A): |
If the velocity of the particle is doubled, its acceleration is halved. |
| Reason (R): |
The power delivered by the force equals the force \(\times\) velocity which is a constant, and the acceleration is proportional to the force. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
197. In the circuit shown in the figure,
\(V_A>V_B.\) Which of the diodes conduct?

| 1. |
only \(D_3\) |
2. |
\(D_1\) and \(D_3\) |
| 3. |
all of the diodes |
4. |
none of the diodes |
198. In an experiment on the photoelectric effect, the maximum kinetic energy of the emitted electrons is plotted as a function of the frequency of incident radiation. The graph is:
| 1. |
a straight line with a positive intercept on the \(x\)-axis (frequency) |
| 2. |
a straight line with a positive intercept on the \(y\)-axis (kinetic energy) |
| 3. |
a parabola |
| 4. |
a hyperbola |
199. The output of the circuit given in the diagram is:

| 1. |
\(A\) AND \(B\) |
2. |
\(A\) OR \(B\) |
| 3. |
\(A\) |
4. |
\(B\) |
200. Given below are two statements:
| Statement I: |
A charged particle moving in a magnetic field experiences a force which is zero only when it moves in the direction of the field or against it. |
| Statement II: |
Whenever a charged particle moves in a uniform magnetic field, its trajectory may be a circle, a straight line or a helix. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course