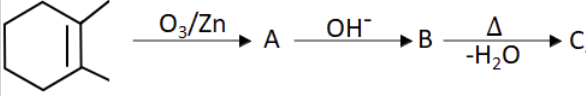Botany - Section A
1. Match each item in
Column-I with one in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Groundnut |
P. |
Lack endosperm |
| B. |
Coconut |
Q. |
Endosperm persists in mature seeds |
| C. |
Orchids |
R. |
Endosperm is completely consumed by developing embryo |
Codes
|
A |
B |
C |
| 1. |
P |
Q |
R |
| 2. |
R |
Q |
P |
| 3. |
R |
P |
Q |
| 4. |
P |
R |
Q |
2. Match the Mendel’s Laws given in
Column-I with their correct definition in
Column-II and select the correct match from the codes given:
| Column-I |
Column-II |
| A. |
Law of
dominance |
P. |
During gamete formation, the alleles for each gene segregate from each other so that each gamete carries only one allele for each gene. |
| B. |
Law of
segregation |
Q. |
In a heterozygote, one trait will conceal the presence of another trait for the same characteristic. |
| C. |
Law of independent assortment |
R. |
Genes of different traits can segregate independently during the formation of gametes. |
Codes:
|
A |
B |
C |
| 1. |
Q |
R |
P |
| 2. |
Q |
P |
R |
| 3. |
P |
Q |
R |
| 4. |
R |
Q |
P |
3. In a dihybrid cross, AaBb X AaBb, assuming independent assortment, what proportion of progeny are expected to express either of the dominant phenotype?
4. Consider the two statements:
| I: |
Baculoviruses are considered a safe option for use in research applications. |
| II: |
They have a broad range of organisms that can act as their hosts. |
| 1. |
Both I and II are correct and II explains I |
| 2. |
Both I and II are correct but II does not explain I |
| 3. |
I is correct but II is incorrect |
| 4. |
I is incorrect but II is correct |
5. If plants convert about 1% of the sunlight they receive into chemical energy, the total energy originally present in the incident sunlight that is finally embodied in a tertiary consumer is about:
1. 0.1%
2. 0.01%
3. 0.001%
4. 0.0001%
6. The genus
Citrus has a number of species that undergo polyembryony, where alongside sexually-derived embryos:
| 1. |
multiple nucellar-cell-derived embryos exist |
| 2. |
multiple integument derived embryos exist |
| 3. |
only one nucellar-cell-derived embryo exists |
| 4. |
multiple antipodal-cell-derived embryos exist |
7. In the given diagram, identify A, B, C and D:

|
A |
B |
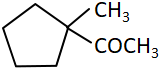
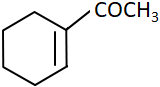
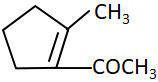
C |
D |
| 1. |
Terminator |
Promoter |
Template Strand |
Coding Strand |
| 2. |
Promoter |
Terminator |
Template Strand |
Coding Strand |
| 3. |
Promoter |
Terminator |
Coding Strand |
Template Strand |
| 4. |
Terminator |
Promoter |
Coding Strand |
Template Strand |
8. Currently. what is the most important cause that is driving animals and plants to extinction?
1. Habitat loss and fragmentation
2. Alien species invasion
3. Over-exploitation
4. Co-extinction
9.
| Assertion (A): |
Cyclosporin A has been commercialized as a blood- cholesterol lowering agent |
| Reason (R): |
Cyclosporin A is a competitive inhibitor of an enzyme responsible for the biosynthesis of cholesterol |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
10. Match each item in Column I with the one in Column II and select the correct match from the codes given:
|
Column-I
[Kingdom] |
|
Column-II
[Cell wall] |
| A. |
Monera |
P. |
Absent |
| B. |
Fungi |
Q. |
Chitinous |
| C. |
Plantae |
R. |
Polysaccharide + amino acid |
| D. |
Animalia |
S. |
Cellulosic |
Codes:
|
A |
B |
C |
D |
| 1. |
R |
S |
Q |
P |
| 2. |
R |
Q |
S |
P |
| 3. |
Q |
S |
R |
P |
| 4. |
P |
Q |
R |
S |
11. Apomixis is:
1. the development of fruit without fertilization
2. production of seeds without fertilization
3. unfertilized eggs developing into males
4. unfertilized eggs developing into females
12. To form a nucleoside a nitrogenous base is linked to the OH of:
1. 1' C pentose sugar through an O-glycosidic linkage
2. 1' C pentose sugar through an N-glycosidic linkage
3. 5' C pentose sugar through an N-glycosidic linkage
4. 5' C pentose sugar through an O-glycosidic linkage
13. You start with 15N15N (heavy) DNA of E.coli. After TWO generations in the 14N medium, you expect E. coli cells will contain:
| 1. |
25% 15N15N DNA, 50% 15N14N DNA, and 25% 14N14N DNA. |
| 2. |
50% 15N15N DNA and 50% 14N14N DNA. |
| 3. |
50% 15N15N DNA and 50% 15N14N DNA. |
| 4. |
50% 15N14N DNA and 50% 14N14N DNA. |
14. Name the scientist who conducted long-term ecosystem experiments to understand correlation between species richness with ecosystem stability:
1. Paul Ehlrich
2. David Tilman
3. Robert May
4. MacArthur
15. Which PGR promotes flowering in pineapples, induces parthenocarpy in tomatoes and also used as herbicides?
| 1. |
Auxins |
2. |
Cytokinins |
| 3. |
Gibberellins |
4. |
Ethylene |
16. You do not expect to see in a plant cell:
1. A tonoplast
2. An elaioplast
3. 80 S ribosomes
4. A centrosome
17. Consider the given statements:
| I: |
In flowers of guava and cucumber, the flower is epigynous and the ovary is inferior |
| II: |
In flowers of mustard, the flower is hypogynous and the ovary is superior |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
18. Regarding bacteria:
1. The rod shaped are called as coccus
2. Majority are photosynthetic autotrophs
3. As a group show the maximum metabolic diversity
4. All are pathogenic to humans
19. Match each item in
Column-I with one in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Companion cells |
P. |
Absent in most monocots |
| B. |
Albuminous cells |
Q. |
Lacking in gymnosperms |
| C. |
Phloem parenchyma |
R. |
Generally absent in primary phloem |
| D. |
Phloem fibers |
S. |
Parenchyma cell adjacent to the sieve cell in gymnosperm wood |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
S |
P |
R |
| 2. |
S |
Q |
R |
P |
| 3. |
P |
R |
Q |
S |
| 4. |
R |
P |
S |
Q |
20. An intervening dikaryophase is seen during the sexual reproduction in members of the fungal class:
1. Phycomycetes and Ascomycetes
2. Ascomycytes and Basidiomycetes
3. Basidiomycetes and Deuteromycetes
4. Phycomycetes and Deuteromycetes
21. Which of the following is/are a correct match?
|
|
Fabaceae |
Solanaceae |
| I. |
Ovary |
Inferior |
Monocarpellary unilocular |
| II. |
Fruit |
Endospermous |
Non-endospermic |
1. Only
I
2. Only
II
3. Both
I and
II
4. Neither
I nor
II
22. Which of the following is/are a correct match?
| I: |
Bulliform cells |
large, bubble-shaped epidermal cells that occur in groups on the upper surface of the leaves of many monocots. |
| II: |
Lenticels |
a porous tissue consisting of cells with large intercellular spaces in the periderm of the secondarily thickened organs and the bark of woody stems and roots of dicotyledonous flowering plants |
1. Only
I
2. Only
II
3. Both
I and
II
4. Neither
I nor
II
23. The reducing power used in Calvin cycle for reduction of carbon dioxide is:
| 1. |
NADH |
2. |
NADPH |
| 3. |
ATP |
4. |
Water |
24. During glycolysis in plant cells, the enzyme hexokinase catalyzes the phosphorylation of:
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
25. The strength of linkage between two genes will be:
1. higher if the genes are farther from each other
2. higher if the genes are closer to each other
3. higher if gametic coupling is present
4. higher if gametic repulsion is present
26. Select the option where the given characters are correct match for members of Rhodophyceae:
|
Major pigments |
Stored food |
Cell wall |
Flagella |
| 1. |
Chlorophyll a, d; phycoerythrin |
Mannitol. Laminarin |
Cellulose, pectin, polysulphate esters |
2-8, equal, apical |
| 2. |
Chlorophyll a, d; phycoerythrin |
Floridean starch |
Cellulose, pectin, polysulphate esters |
Absent |
| 3. |
Chlorophyll a, c; Fucoxanthin |
Mannitol. Laminarin |
Cellulose and algin |
2, unequal, lateral |
| 4. |
Chlorophyll a, b; |
Starch |
Cellulose |
2-8, equal, apical |
27. During the formation of the female gametophyte in angiosperms, cell walls are laid when the developing female gametophyte reaches:
1. 2 nucleate stage
2. 4 nucleate stage
3. 6 nucleate stage
4. 8 nucleate stage
28. Select the correct statements with respect to pleiotropism:
| (a) |
A gene is said to be pleiotropic if it affects more than one trait |
| (b) |
Phenylketonuria is an example of pleiotropy |
| (c) |
A condition where one gene has several alleles is referred to as pleiotropism |
| (d) |
A trait is said to be pleiotropic if several genes control it |
Choose the correct answer from the options given below:
| 1. |
(a) and (b) only |
2. |
(a) and (d) only |
| 3. |
(a), (b) and (c) only |
4. |
(b), (c) and (d) only |
29. The largest tiger reserve in India is:
| 1. |
Valmiki |
2. |
Nagarjunsagar - Srisailam |
| 3. |
Periyar |
4. |
Nagarhole |
30. Which of the following has maximum genetic diversity in India?
| 1. |
Mango |
2. |
Wheat |
| 3. |
Groundnut |
4. |
Rice |
31. In angiosperms:
| I: |
the seed is the final product of sexual reproduction. |
| II: |
the seed is often described as a fertilised ovule. |
| III: |
seeds are formed inside fruits. |
| 1. |
Only I and II are correct |
2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
4. |
I, II and III are correct |
32. Consider the given two statements:
| Assertion (A): |
Pollen is one part of the plant that is more likely to be preserved as fossil. |
| Reason (R): |
Pollen intine is made of sporopollenin, one of the most chemically inert biological polymers. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
33. Each restriction endonuclease:
| I: |
binds to the DNA after identifying its specific recognition sequence |
| II: |
cuts one of the two strands of the double helix at random points in their sugar-phosphate backbones |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
34. Consider the two statements:
| Statement I: |
Current availability of CO2 levels is limiting to the C3 plants. |
| Statement II: |
C3 plants show saturation at about \(360 ~\mu lL^{-1}\) |
| 1. |
Statement I is correct; Statement II is correct |
| 2. |
Statement I is incorrect; Statement II is correct |
| 3. |
Statement I is correct; Statement II is incorrect |
| 4. |
Statement I is incorrect; Statement II is incorrect |
35. Consider the given two statements:
| Assertion (A): |
The number of trophic levels in the grazing food chain is restricted. |
| Reason (R): |
Based on the source of their nutrition or food, organisms occupy a specific
place in the food chain that is known as their trophic level. |
| 1. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 2. |
(A) is True but (R) is False |
| 3. |
(A) is False but (R) is True |
| 4. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
Botany - Section B
36. The embryo of grass seed is enclosed within:
| 1. |
a single protective sheath: the coleoptile |
| 2. |
a single protective sheath: the coleorrhiza |
| 3. |
two protective sheaths: a coleoptile covering the young shoot and the coleorrhiza covering the young root |
| 4. |
two protective sheaths: a coleoptile covering the young root and the coleorrhiza covering the young shoot |
37. Consider the given two statements:
| I: |
In a test cross, the allele the individual in question [one that expresses the dominant phenotype] passes on, determines the phenotype of the offspring. |
| II: |
The homozygous recessive individual can only pass on recessive alleles. |
| 1. |
Both I and II are correct and II explains I |
| 2. |
Both I and II are correct but II does not explain I |
| 3. |
I is correct but II is incorrect |
| 4. |
I is incorrect but II is correct |
38. Identify the correctly matched pair:
| I. |
Binding of nitrogenous bases to a pentose sugar to form a nucleoside |
N-glycosidic bond at C1 of the pentose sugar |
| II. |
Binding of phosphate to a nucleoside to form a nucleotide |
Ester bond at C5 of the pentose sugar |
| III. |
Joining of two nucleotides to form a dinucleotide |
5’-3’ phosphodiester bond |
1. Only
I and
II
2. Only
I and
III
3. Only
II and
III
4.
I, II and
III
39. In a test cross AaBb X aabb [assuming independent assortment], the ratio of progeny with genotypes AaBb and aabb will be:
1. 1 : 1
2. 3 : 1
3. 1 : 1 : 1 : 1
4. 9 : 1
40. Given below are two statements: one is labeled Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
Alternaria, Colletotrichum and Trichoderma are kept in the fungal class- deuteromycetes. |
| Reason (R): |
Asexual and vegetative phases in the life cycle of these fungi are not known. |
In the light of the above statements, choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
41. Identify the incorrect statement regarding photosynthetic pigments in higher plants:
| 1. |
Chlorophyll a is the chief pigment associated with photosynthesis. |
| 2. |
In the chromatogram, carotenoids appear yellow to yellow-orange. |
| 3. |
Accessory pigments protect chlorophyll a from photo-oxidation. |
| 4. |
The maximum absorption of chlorophyll a is in blue and green wavelength region of light. |
42. Gynoecium is situated in the centre and other parts of the flower are located on the rim of the thalamus almost at the same level in:
1. Mustard
2. China rose
3. Plum
4. Guava
43. Krebs cycle takes place in the part labeled:

44. During oxidation within a cell, all the energy contained in respiratory substrates:
1. is released free into the cell
2. is released in a single step
3. is released in a series of slow stepwise reactions
4. gets converted to usable energy
45. Bundle sheath cells, part of Kranz anatomy in a C
4 plant, are not characterized by:
| 1. |
May form several layers around the vascular bundle |
| 2. |
Have large number of chloroplasts |
| 3. |
Have thick walls impervious to gaseous exchange |
| 4. |
Large intercellular spaces are seen |
46. Identify the incorrect statement:
| 1. |
Insulin is a polymer of fructose. |
| 2. |
Cellulose is a homopolymer |
| 3. |
Starch is a homopolymer |
| 4. |
Chitin is nitrogen containing complex polysaccharide |
47. Match the alga with the type of life cycle and select the correct match from the codes given:
|
Alga |
|
Type of life cycle |
| A. |
Spirogyra |
P. |
Haplontic |
| B. |
Fucus |
Q. |
Diplontic |
| C. |
Polysiphonia |
R. |
Haplo-diplontic |
Codes:
|
A |
B |
C |
| 1. |
P |
Q |
R |
| 2. |
P |
R |
Q |
| 3. |
Q |
P |
R |
| 4. |
R |
Q |
P |
48. RNA interference involves silencing of a specific mRNA which essentially means:
| 1. |
the transcription of mRNA is stopped by inactivating the promoter |
| 2. |
the hnRNA is not allowed to undergo post-transcriptional modification |
| 3. |
the mRNA cannot cross the nuclear pore complex |
| 4. |
translation of the mRNA molecule is prevented |
49. In PS I, the reaction centre chlorophyll a has an absorption peak at:
| 1. |
660 nm |
2. |
680 nm |
| 3. |
700 nm |
4. |
730 nm |
50. Formation of interfascicular cambium and cork cambium in certain plants is an example of:
| 1. |
Differentiation |
2. |
De-differentiation |
| 3. |
Re-differentiation |
4. |
Anti-differentiation |
Zoology - Section A
51. Consider the given two statements:
| I: |
Secondary oocyte is fertilized by the sperm. |
| II: |
At fertilization, the secondary oocyte completes Meiosis-II |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
52. The mean annual precipitation is the most variable in:
1. Tropical forest
2. Desert
3. Grassland
4. Arctic tundra
53.
| Assertion (A): |
Transgenic animals can be specifically designed to allow the study of how genes are regulated, and how they affect the normal functions of the body and its development, e.g., study of complex factors involved in growth such as insulin-like growth factor. |
| Reason (R): |
By introducing genes from other species that alter the formation of this factor and studying the biological effects that result, information is obtained about the biological role of the factor in the body. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
54. In the absence of fertilization:
1. the zona pellucid gets hardened
2. the secondary oocyte completes meiosis II
3. the corpus luteum degenerates
4. menstruation does not occur
55. According to the MTP [Amendment] Act 2017, the gestational age till which MTP can be performed is a maximum of:
| 1. |
12 weeks |
2. |
16 weeks |
| 3. |
20 weeks |
4. |
24 weeks |
56. Study the given diagram and select the correct option:

| 1. |
The karyotype is of an individual suffering from Down’s syndrome |
| 2. |
The number of autosomes seen is 43 |
| 3. |
The genetic sex of this individual must be female |
| 4. |
The fertility will be maintained in this individual |
57. An environment eliminates individuals at the extremes of a population. This type of natural selection will be called as:
1. Stabilizing
2. Directional
3. Disruptive
4. Dispersive
58. When a retrovirus infects an animal cell:
| 1. |
c DNA is introduced into cell |
| 2. |
Viral RNA is produced by reverse transcription |
| 3. |
Viral RNA incorporates into host genomes |
| 4. |
Infected cell can survive while viruses are being replicated and released |
59. Match each item in
Column-I with the one in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Cephalochordata |
P. |
Swim bladder |
| B. |
Cyclostomata |
Q. |
Notochord – head to tail |
| C. |
Chondrichthyes |
R. |
Males bear claspers on pelvic fins |
| D. |
Osteichthyes |
S. |
Marine but migrate to freshwater for spawning |
Codes:
|
A |
B |
C |
D |
| 1. |
S |
Q |
P |
R |
| 2. |
P |
Q |
R |
S |
| 3. |
Q |
S |
R |
P |
| 4. |
R |
P |
S |
Q |
60. Given below are two statements: one is labelled as
Assertion (A) and the other is labelled as
Reason (R):
| Assertion (A): |
Meiosis conserves specific chromosome number of each species across generations in sexually reproducing organisms. |
| Reason (R): |
Meiosis, per se, results in reduction of chromosome number by half. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
61. Aldosterone acts mainly at renal tubules and stimulates the:
| 1. |
Reabsorption of Na+ and K+ |
| 2. |
Excretion of Na+ and K+ |
| 3. |
Excretion of K+ and phosphate ions |
| 4. |
Reabsorption of Na+ and phosphate ions |
62. To maintain the pH and ionic balance of the body fluids, the proximal convoluted tubule selectively secretes all the following into the filtrate except:
| I. |
Hydrogen ion |
II. |
Potassium ion |
| III. |
Ammonia |
IV. |
Bicarbonate ion |
1. I & II
2. II & III
3. II & IV
4. Only IV
63. A person has the following values of respiratory volumes:
| Tidal Volume |
400 ml |
| Expiratory reserve volume |
1000 ml |
| Inspiratory reserve volume |
2500 ml |
| Residual volume |
1000 ml |
His functional residual capacity and vital capacity respectively are:
1. 2000 ml and 3900 ml
2. 2000 ml and 4900 ml
3. 1400 ml and 3900 ml
4. 1400 ml and 4900 ml
64. Identify the incorrect statement:
| 1. |
PCR is now routinely used to detect HIV in suspected AIDS patients. |
| 2. |
PCR is being used to detect mutations in genes in suspected cancer patients. |
| 3. |
ELISA is based on the principle of antigen-antibody interaction. |
| 4. |
ELISA does not detect antibodies and can detect only antigens. |
65. Identify the correct statement:
| 1. |
Agrobacterium tumefaciens is able to transform large number of monocot plants. |
| 2. |
Micro-injection is the only way to transform animal cells. |
| 3. |
Competence in bacterial cells can be induced by trivalent anions like phosphates. |
| 4. |
Biolistics or gene gun is a suitable method to transform plant cells. |
66. If we compare percentage weight of elements present in Earth’s crust and the Human body, which of the following would be present in the largest percentage in both?
1. Carbon
2. Oxygen
3. Hydrogen
4. Nitrogen
67. Which of the following values will not be normal for a healthy person?
1. 10 gms of haemoglobin per 100 ml of blood
2. 5 million RBC per mm3
3. 7000 WBC per mm3
4. 200000 platelets per mm3
68. Identify the gland that is not matched correctly to its location in the body:
| 1. |
Thymus |
Dorsal side of aorta |
| 2. |
Parathyroid |
Back side of thyroid |
| 3. |
Pineal |
Dorsal side of forebrain |
| 4. |
Adrenal |
Superior pole of kidney |
69. The total number of bones in the skeleton of girdles in adult humans is:
70. Which biomolecule makes up maximum percent of the total cellular mass in an average cell?
1. Proteins
2. Carbohydrates
3. Lipids
4. Nucleic acids
71. Match each item in
Column-I with one in
Column-II and select the best match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Comb plates |
P. |
Cnidaria |
| B. |
Metagenesis |
Q. |
Mollusca |
| C. |
Canal system |
R. |
Sponges |
| D. |
Radula |
S. |
Ctenophora |
Codes:
|
A |
B |
C |
D |
| 1. |
S |
P |
R |
Q |
| 2. |
P |
S |
R |
Q |
| 3. |
P |
S |
Q |
R |
| 4. |
S |
P |
Q |
R |
72. What type of chromosomes will not have a short and a long arm?
1. Acrocentric
2. Metacentric
3. Sub-metacentric
4. Both 1 and 2
73. Inspiration during breathing occurs when the pressure in lungs is:
1. Zero
2. Negative with respect to atmospheric pressure
3. Equal to atmospheric pressure
4. Positive with respect to atmospheric pressure
74. What is incorrect regarding human heart?
1. It is neurogenic
2. It is derived from embryonic mesoderm
3. It is a muscular pump
4. It is located in thorax between the lungs
75. Nephrons closest to the renal medulla are called:
1. Cortical
2. Medullary
3. Juxta-medullary
4. Proto-nephridia
76. Karl Ernst von Baer:
| 1. |
conducted a control experiment to disprove spontaneous generation |
| 2. |
proposed the concept of pangenesis as the physical basis of heredity |
| 3. |
disapproved the proposal of Ernst Haeckel [Biogenetic law] |
| 4. |
was one of the rediscoverers of Mendel’s laws |
77. The strain of Escherechia coli from which the restriction enzyme EcoR I has been isolated is:
1. DH5α
2. RY 13
3. NC101
4. K1
78. Identify the incorrect statement:
| 1. |
Both AIDS and Hepatitis B can be transmitted through sexual contact or infected blood. |
| 2. |
Chronic use of alcohol can lead to cirrhosis of liver. |
| 3. |
Nicotine stimulates adrenal cortex to secrete corticosteroids. |
| 4. |
Morphine is a very effective sedative and painkiller. |
79. If a molecule that can act as a genetic material should be able to generate its replica, which of the following molecules can act as a genetic material?
| I. Proteins |
II. DNA |
III. RNA |
| 1. |
Only I and II |
2. |
Only I and III |
| 3. |
Only II and III |
4. |
I, II and III |
80. The number of cranial nerves in frogs are:
| 1. |
8 pairs |
2. |
10 pairs |
| 3. |
12 pairs |
4. |
31 pairs |
81. Consider the two statements:
| Assertion (A): |
Carbon dioxide (CO2) diffuses faster than oxygen (O2) across the respiratory membrane. |
| Reason (R): |
CO2 is 20–25 times more soluble in blood and alveolar fluids than O2. |
| 1. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
| 2. |
(A) is True but (R) is False. |
| 3. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 4. |
(A) is False but (R) is True. |
82. A patient suffering from hyperparathyroidism may have:
I: increased bone deposition
II: increased risk of fractures
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
83. Deaf-mutism and mental retardation is seen in the deficiency of:
| 1. |
Iron |
2. |
Calcium |
| 3. |
B12 |
4. |
Iodine |
84. Aldosterone causes:
| I: |
Reabsorption of sodium ions by the distal parts of the tubules. |
| II: |
Secretion of potassium ions by the distal parts of the tubules. |
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
85. If N is the population density at time t, then its density at time t +1 is [B is births; I is immigration;
D is deaths; E is emigration]:
1. Nt+1 = Nt + [(B + I) – (D + E)]
2. Nt+1 = Nt + [(B + I) + (D + E)]
3. Nt+1 = Nt + [(B + I) (D + E)]
4. Nt+1 = Nt + [(B + I) / (D + E)]
Zoology - Section B
86. The part of the human respiratory tract that is not included in the conducting zone is the:
1. Pharynx
2. Bronchi
3. Larynx
4. Respiratory bronchiole
87. The regulation of GFR involves hormonal feedback mechanisms involving all the following except:
1. Heart
2. Lungs
3. Hypothalamus
4. Kidney
88. All the following are related to the limbic system in the human brain except:
| 1. |
Amygdala |
2. |
Hippocampus |
| 3. |
Olfactory bulb |
4. |
Corpus callosum
|
89. Which of the following hormones is not a steroid and yet does not act on the target cells through a second messenger?
1. Corticotropin
2. Cortisol
3. Iodothyronines
4. Estrogen
90. Consider the given two statements:
| I: |
The presence or absence of hymen is not a reliable indicator of virginity or sexual experience. |
| II: |
It is a membrane that often covers the opening of vagina partially. |
1. Both
I and
II are correct and
II explains
I
2. Both
I and
II are correct and
II does not explain
I
3.
I is correct and
II is incorrect
4. Both
I and
II are incorrect
91. The mammary ampulla in the given diagram is shown by:

92. Vasectomy:
| I: |
is the most effective permanent form of contraception available to men. |
| II: |
protects against sexually transmitted infections. |
| 1. |
Both I and II are correct |
| 2. |
Only I is correct |
| 3. |
Only II is correct |
| 4. |
Both I and II are incorrect |
93. PCR does not require:
1. A thermostable DNA polymerase
2. Dideoxy-dNTPs (ddNTPs)
3. Template DNA
4. Primers
94.
| Assertion (A): |
VNTRs are the basis of DNA fingerprinting by RFLP |
| Reason (R): |
VNTRs are polymorphic and are inherited |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is false |
| 4. |
Both (A) and (R) are False |
95.
| Assertion (A): |
The genetic sex of an individual with Klinefelter's syndrome is female |
| Reason (R): |
Only X-chromosome is present in their genotype |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
96. Match each item in
Column-I with the one in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Plasma |
P. |
45 % of blood |
| B. |
Formed elements |
Q. |
55 % of blood |
| C. |
Neutrophils |
R. |
60-65 % of total WBCs |
| D. |
Lymphocytes |
S. |
20-25 % of total WBCs |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
Q |
P |
S |
R |
| 3. |
Q |
P |
R |
S |
| 4. |
P |
Q |
S |
R |
97. At a place where a ‘diffusion boundary’ is present in the human body, you are most likely to find
1. Simple squamous epithelium
2. Cuboidal brush bordered epithelium
3. Columnar ciliated epithelium
4. Compound epithelium
98. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
Post-menopausal women are at higher risk to develop osteoporosis. |
| Reason (R): |
Level of oestrogen in post-menopausal women is decreased. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
99. Nicotine:
| 1. |
is a CNS depressant and can cause respiratory depression |
| 2. |
commonly leads to development of cirrhosis of liver |
| 3. |
stimulates adrenal gland to release adrenaline and nor-adrenaline |
| 4. |
is very effective analgesic and sedative |
100. Droplets can be the mode of transmission of pathogens causing all the following diseases except:
1. Ascariasis
2. Pneumonia
3. Common cold
4. Covid-19
Chemistry - Section A
101. The oxidation of benzene by V2O5 in the presence of air produces:
| 1. |
Benzoic anhydride |
2. |
Maleic anhydride |
| 3. |
Benzoic acid |
4. |
Benzaldehyde |
102. The correct order for capturing polarity among the following is:
1. NH3 < H2O < HF < H2S
2. H2S < NH3 < H2O < HF
3. H2O < NH3 < H2S < HF
4. HF < H2O < NH3 < H2S
103. \(CH_3CH_2CHCl_2\) upon reaction with \(NaNH_2\) gives:
104. Amylopectin is a branched chain polymer of \(\alpha-D\) glucose units in which chain is formed by C1-C4 glycosidic linkage while branching occurs by
1. C2-C5 glycosidic linkage
2. C1-C5 gycosidic linkage
3. C1-C6 glycosidic linkage
4. C2-C6 glycosidic linkage
105. Generally, water-soluble vitamins (i.e., vitamins B and C) cannot be stored in our body, except for which of the following vitamins?
1. Vitamin B1
2. Vitamin B6
3. Vitamin B12
4. Vitamic C
106. Case Study Question:
The relative location of the bond pair and the lone pair is decided by Bent's rule. The rule states that the more electronegative atom prefers to stay in the orbital having less s character while the lone pair prefers to stay in the orbital having more s character. In molecules with trigonal bipyramidal (TBP) geometry, more electronegative atom prefers axial position while the lone pais prefers equatorial position.
The hybridised state of Br in CsBr3 and shape of \(Br^-_3\) are, respectively:
1. sp3d and Linear
2. No hybridisation and Linear
3. sp3d and Trigonal bipyramidal
4. No hybridisation and Trigonal bipyramidal
107. Determine the percetage dimerisation of acetic acid if it dimerises in benzene.
[Given: The value of Van't Hoff factor (i) for the dimerisation of acetic acid is 0.7]
| 1. |
30% |
2. |
60% |
| 3. |
70% |
4. |
90% |
108. The standard reduction potential of Zn
2+/Zn and Cu
2+/Cu is -0.76 and +0.34 V respectively. The oxidising agent and reducing agent, respectively, are
| 1. |
Cu & Zn2+ |
2. |
Zn & Cu |
| 3. |
Cu2+ & Zn2+ |
4. |
Cu2+ & Zn |
109. In salt analysis, group V radicals
\(\mathrm{(Ba^{2+}, Ca^{2+} ~and ~Sr^{2+} )}\) are precipitated as their carbonates by adding solid
\(\mathrm{NH_4Cl}\) and excess of
\(\mathrm{NH_4OH}\) solution followed by solid
\(\mathrm{(NH_4)_2CO_3}\)
Choose the correct reagent used for the confirmation of
\(\mathrm{Ca^{2+}}\) ion from the following:
| 1. |
Ammonium sulphate |
2. |
Potassium chromate |
| 3. |
Ammonium oxalate |
4. |
Ammonium nitrate |
110. MnO2 when fused with KOH and oxidized in the air gives a dark green compound X. In an acidic solution, X undergoes disproportionation to give an intense purple compound Y and MnO2.
The compounds X and Y, respectively are:
1. Mn2O7 and KMnO4
2. K2MnO4 and KMnO4
3. KMnO4 and K2MnO4
4. KMnO4 and Mn2O7
111. The oxidising ability of the given anions follows the order :
\(\begin{aligned} &\text {1. } \mathrm{CrO}_{4}^{2-}<\mathrm{MnO}^-_{4}<\mathrm{VO}^{3-}_{4}<TiO^{4-}_4\\ &\text {2. } \mathrm{MnO}_{4}^{-}<\mathrm{CrO}_{4}^{2-}< \mathrm{VO}_{4}^{3-}<\mathrm{TiO}_{4}^{4-}\\ &\text {3. } \mathrm{TiO}_{4}^{4-}<\mathrm{VO}_{4}^{3-}<\mathrm{CrO}_{4}^{2-}<\mathrm{MnO}_{4}^{-}\\ &\text {4.} \mathrm{Ti} \mathrm{O}_{4}^{4-}<\mathrm{VO}_{4}^{3-}<\operatorname{Mn}{O^-_{4}}< CrO^{2-}_4 \end{aligned}\)
112. The number of hydrate isomers of CoCl
3.6H
2O is/are:
| 1. |
One(1) |
2. |
Two(2) |
| 3. |
Three(3) |
4. |
Four(4) |
113. Which alcohol is most reactive in an esterification reaction?
| 1. |
CH3OH |
2. |
CH3CH2OH |
| 3. |
CH3CH(CH3)CH2OH |
4. |
(CH3)3C-OH |
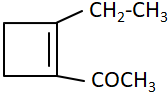
114. 
The compound 'C' in the above reaction is:
115. [Cu(NH
3)
4]SO
4 is a blue soluble complex.
The blue colour of this complex is due to the presence of:
| 1. |
[Cu(NH3)4]2+ |
2. |
Cu2+ |
| 3. |
\(SO^{2-}_4\) |
4. |
[Cu(NH3)6]2+ |
116. The radioactive lanthanoid among the following is:
| 1. |
Ce(58) |
2. |
Nd(60) |
| 3. |
Pm(61) |
4. |
Eu(63) |
117. \(\psi_{3,2,0}\) is the representation of the orbital:
1. 3px
2. 3dxy
3. 3dz2
4. 3dx2-y2
118. On adding inert gas at constant volume, equilibrium shifts in the forward direction will take place in -
1. \(N_2 (g) + 3H_2 (g)\rightleftharpoons 2NH_3 (g)\)
2. \(PCl_5 (g) \rightleftharpoons PCl_3 (g) + Cl_2 (g)\)
3. \(H_2(g) + I_2 (g) \rightleftharpoons 2HI (g)\)
4. None of the above.
119. 2-Butyne is hydrogenated in presence of Lindlar's catalyst, The major product formed will be
1. Cis-but-2-ene
2. Trans-but-2-ene
3. 1-Butyne
4. Butane
120. What types of π bonds are present in the structure of ClO4-, which contains three π bonds?
1. \(3 ~ d\pi-p\pi\)
2. \(2 ~ d\pi-p\pi ~and ~1 ~ p\pi-p\pi \)
3. \(1 ~ d\pi-p\pi ~and ~2 ~ p\pi-p\pi \)
4. \(3 ~ p\pi-p\pi \)
121. Which of the following compounds does not act as an oxidising agent?
| 1. |
\(SO_2\) |
2. |
\(HCl\) |
| 3. |
\(KMnO_4\) |
4. |
\(MnO_2 \) |
122. The compound, among the following, that has the highest \(pK_a\) is:
1. \(CH_3COOH\)
2. \(HCOOH\)
3. \(C_6H_5COOH\)
4. \((CH_3)_2CHCOOH\)
123. Which of the following contains the highest mass?
1. 2.24 \(l\) \(\text{SO}_2\) gas at NTP
2. 0.4 mole \(\text{NO}_2\) gas
3. 3.01×1022 molecules of \(\text{CO}_2\) gas
4. 5 gram \(\text{Fe}\)
124. Inversion of cane sugar is an example of:
| 1. |
Unimolecular reaction |
| 2. |
Pseudounimolecular reaction |
| 3. |
Bimolecular reaction |
| 4. |
Second order reaction |
125. When formaldehyde reacts with ethyl magnesium bromide followed by hydrolysis,
the product formed will be:
1. Methanol
2. Ethanol
3. Propanol
4. Propan-2-ol
126. Heat at constant volume and heat at constant pressure are, respectively:
1. \(\Delta G ~~and ~~\Delta H \)
2. \(\Delta H ~~and ~~\Delta E \)
3. \(\Delta E ~~and ~~\Delta H \)
4. \(\Delta E ~~and ~~\Delta G \)
127. The hybridised state of boron in diborane \((B_2H_6)\) is:
1. \(sp\)
2. \(sp^2\)
3. \(sp^3\)
4. \(sp^2\) as well as \(sp^3\)
128. When aqueous solution of \(\text{NaCl}\) is electrolyzed using inert electrodes then the ion discharged at cathode and anode, respectively, are:
1. \(\mathrm{Na^+ ~and ~~OH^-}\)
2. \(\mathrm{H^+ ~and ~~OH^-}\)
3. \(\mathrm{H^+ ~and ~~Cl^-}\)
4. \(\mathrm{Na^+ ~and ~~Cl^-}\)
129. In thin layer chromatography, which of the following is not used to visualise the separated layer?
| 1. |
Visualisation agent in the mobile phase |
| 2. |
Spraying of suitable reagent |
| 3. |
UV light |
| 4. |
Solid I2 |
130.
| Assertion (A): |
SeCl4 does not have a tetrahedral structure. |
| Reason (R): |
Se has two lone pairs in SeCl4 . |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
131. Select the correct option based on statements below:
| Assertion (A): |
The metal carbon bond in metal carbonyls possesses both \(\sigma\) and \(\pi\) character. |
| Reason (R): |
The ligand to metal bond is a \(\pi\) bond and metal to ligand bond is a \(\sigma\) bond. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
132. Which one of the following statements is incorrect related to Molecular Orbital Theory?
| 1. |
The \(\pi^*\) antibonding molecular orbital has a node between the nuclei. |
| 2. |
In the formation of a bonding molecular orbital, the two electron waves of the bonding atoms reinforce each other. |
| 3. |
Molecular orbitals obtained from \(2P_x\) and \(2P_y\) orbitals are symmetrical around the bond axis. |
| 4. |
A \(\pi-\)bonding molecular orbital has larger electron density above and below the internuclear axis. |
133. When two liquids A and B mix together, the boiling point of solution is found higher than that of the individual liquids. The nature of the solution is:
| 1. |
Ideal solution. |
| 2. |
Positive deviation with a non-ideal solution. |
| 3. |
Negative deviation with a non-ideal solution. |
| 4. |
Normal solution. |
134. The incorrect statement among the following is:
| 1. |
Ionic bonds are non-directional, while covalent bonds are directional. |
| 2. |
The formation of \(\pi\)- bond shortens the distance between the two concerned atoms. |
| 3. |
An ionic bond is possible between similar and dissimilar atoms. |
| 4. |
Linear overlapping of atomic p-orbitals leads to a sigma bond. |
135. The electrolytes (A) and (B) in the below graph, respectively, may be:
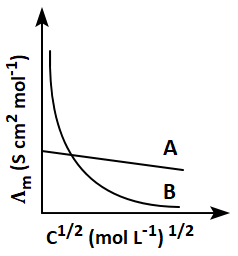
1. A = KCl; B = NH
4OH
2. A = NH
4OH; B = CH
3COOH
3. A = CH
3COOH; B= CH
3COONa
4. A = NH
4OH; B = NaCl
Chemistry - Section B
136. The boiling point of 1M Urea solution is 101
oC. At the same temperature, the boiling point of 1M KCl solution will be:
1. 101
oC
2. 102
oC
3. 202
oC
4. Cannot be predicted (Insufficient Data)
137. When a CuSO₄ solution using copper electrodes is electrolyzed, the incorrect statement among the following is:
1. The weight of the cathode increases.
2. O2 gas is evolved at the anode.
3. The weight of the anode decreases.
4. pH of the solution remains the same.
138. The molar conductivity of 0.05 M NH
4Cl is 20 S cm
2 mol
-1. The molar conductivities of
\(NH^+_4\) and
\(Cl^-\) ions at infinite dilution are 74 S cm
2 mol
-1 and 26 S cm
2 mol
-1, respectively. The dissociation constant of NH
4Cl will be:
| 1. |
\(2.5 \times 10^{-3}\) |
2. |
\(2 \times 10^{-3}\) |
| 3. |
\(2.5 \times 10^{-4}\) |
4. |
\(2 \times 10^{-4}\) |
139. A catalyst accelerates a reaction primarily by stabilizing the:
| 1. |
Substrate |
2. |
Product |
| 3. |
Intermediate |
4. |
Transition state |
140. Based on the hyperconjugation effect described above,
identify the compound in which the double bond is the longest:
1. CH3- CH = CH - CH3
2. (CH3)3 C-CH = CH2
3. CH3-CH = CH-CH(CH3)2
4. CH2 = CH- CH2 -CH3
141. Which, of the following solutions has the highest freezing point?
1. 1 M Urea solution
2. 1 M KCl solution
3. 1 M MgCl2 solution
4. All have the same freezing point
142. If equal volumes of pH=8 and pH=13 solutions are mixed together, then the pH of the resulting solution will be:
| 1. |
8.3 |
2. |
9.3 |
| 3. |
10.3 |
4. |
12.7 |
143. In graphite, the hybridized state of carbon and nature of graphite, respectively, are
1. sp3 and aromatic
2. sp2 and nonaromatic
3. sp2 and aromatic
4. sp3 and nonaromatic
144. The total number of structural isomers of the molecular formula
\(C_4H_8\) are:
145. The most acidic compound among the following is:
| 1. |
o-Cresol |
2. |
m-Cresol |
| 3. |
p-Cresol |
4. |
o-Xylene |
146. In which of the following cases, value of the Van't Hoff factor (i) is the highest?
1. \(NaCl~ (dissociation = 90\%)\)
2. \(CaF_2~ (dissociation = 80\%)\)
3. \(K_4[Fe(CN)_6]~ (dissociation = 50\%)\)
4. \(AlCl_3~ (dissociation = 70\%)\)
147. The correct order of spin-only magnetic moment for the given complexes is:
1. \([Co(H_2O)_6]^{2+} > [MnCl_6]^{3-} > [Fe(CN)_6]^{3-} \)
2. \([Fe(CN)_6]^{3-} >[Co(H_2O)_6]^{2+} > [MnCl_6]^{3-} \)
3. \([MnCl_6]^{3-} >[Fe(CN)_6]^{3-} >[Co(H_2O)_6]^{2+} \)
4. \([MnCl_6]^{3-} > [Co(H_2O_6]^{2+} > [Fe(CN)_6]^{3-} \)
148. The molar conductance of an electrolyte increases with dilution according to the equation:
\(\Lambda_{\mathrm{m}}=\Lambda_{\mathrm{m}}^{\circ}-\mathrm{A} \sqrt{\mathrm{c}} \)
Consider the following four statements:
| A: |
This equation applies to both strong and weak electrolytes. |
| B: |
The value of the constant A depends upon the nature of the solvent. |
| C: |
The value of constant A is the same for both \(BaCl_2\) and \(MgSO_4\) |
| D: |
The value of constant A is the same for both \(BaCl_2\) and \(Mg(OH)_2\) |
Which of the above statements are correct?
| 1. |
(A) and (B) only |
2. |
(A), (B), and (C) only |
| 3. |
(B) and (C) only |
4. |
(B) and (D) only |
149. The coloured ion among the following is :
1. \(\mathrm{Cu}^{+} \)
2. \(\mathrm{Cu}^{2+} \)
3. \(\mathrm{Ti}^{4+} \)
4. \(\mathrm{V}^{5+} \)
150. Given are ions in
Column-I and ionic radii in
Column-II:
|
Column-I (Ion) |
|
Column-II (Ionic Size) |
| (p) |
\(\mathrm{Mg}^{2+}\) |
(I) |
1.19 Å |
| (Q) |
\(\mathrm{O}^{2-}\) |
(II) |
0.72 Å |
| (R) |
\(\mathrm{Na}^{+}\) |
(III) |
1.16 Å |
| (S) |
\(\mathrm{F}^{-}\) |
(IV) |
1.26 Å |
Identify the option capturing the correct match of information given in Column I and Column II above:
|
P |
Q |
R |
S |
| 1. |
III |
I |
II |
IV |
| 2. |
II |
IV |
I |
III |
| 3. |
II |
IV |
III |
I |
| 4. |
IV |
II |
III |
I |
Physics - Section A
151. Given below are two statements:
| Assertion (A): |
It is observed that when a car brakes suddenly, the passengers are thrown forward. |
| Reason (R): |
(Newton's 1st Law of Motion) Everybody continues in its state of rest or of uniform motion in a straight line except in so far as it be compelled by an externally impressed force to act otherwise. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
152. A beaker containing water, is placed in an elevator and the absolute pressure is measured at a depth of \(10~\text{cm}\) below the water level:
| 1. |
If the elevator accelerates up, the pressure decreases. |
| 2. |
If the elevator moves up with uniform velocity, the pressure decreases. |
| 3. |
If the elevator accelerates down, the pressure decreases. |
| 4. |
If the elevator moves down with uniform velocity, pressure increases. |
153. The ice-point reading on a thermometer scale is found to be \(20^\circ,\) while the steam point is found to be \(70^\circ.\) When this thermometer reads \(100^\circ ,\) the actual temperature is:
1. \(80^\circ\text{C}\)
2. \(130^\circ\text{C}\)
3. \(160^\circ\text{C}\)
4. \(200^\circ\text{C}\)
154. 
A pair of field lines are drawn, connecting the charges
\(q_1, q_2\) in addition to the straight field line connecting them. From the above diagram,
| 1. |
\(q_1>0, q_2<0~\text{and}~|q_1|>|q_2|\) |
| 2. |
\(q_1<0, q_2>0~\text{and}~|q_1|>|q_2|\) |
| 3. |
\(q_1>0, q_2<0~\text{and}~|q_1|<|q_2|\) |
| 4. |
\(q_1<0, q_2>0~\text{and}~|q_1|<|q_2|\) |
155. How will the voltage
\((V)\) between the two plates of a parallel plate capacitor depend on the distance
\((d)\) between the plates, if the charge on the capacitor remains the same?
156. A parallel plate capacitor
\((C)\) is charged by connecting it to a battery (EMF
\(E\)). A dielectric slab is inserted into the space between the plates.
| (I) |
The charge on the plates increases. |
| (II) |
The energy stored in the capacitor increases. |
| (III) |
Work is done by the battery as the slab is inserted. |
Choose the correct answer from the options given below:
| 1. |
(I), and (II) are True. |
| 2. |
(I) and (III) are True. |
| 3. |
only (I) is True. |
| 4. |
(I), (II), and (III) are True. |
157. A long solenoid has a square cross-section of side \(a\). It has turn-density n (number of turns per unit axial length). A current \(i\) is passed through this solenoid. The magnetic field at the centre of the solenoid is \(B_c\). Then, \(B_c\) is proportional to:
(I) \(a\)
(II) \( \dfrac{1} {a}\)
(III) \(n\)
(IV) \(i\)
Choose the correct option from the given ones:
1. I, III, IV
2. II, III, IV
3. III, IV
4. IV Only
158. The coercive force for a certain magnet is \(3 \times 10^3 ~\text{A/m}. \) This magnet is placed within a solenoid having \(40~\text{turns/cm}. \) What current should be passed through the solenoid so that the magnet is demagnetized?
1. \(0.75~\text{A}\)
2. \(75~\text{A}\)
3. \(1.33~\text{A}\)
4. \(133~\text{A}\)
159. A thin metallic plate is allowed to fall through the space between two magnetic poles creating a horizontal magnetic field. The plate is vertical, and its face is perpendicular to the field lines as it falls. While it is entering the region of the magnetic field,

| 1. |
The acceleration of the plate is equal to \(g.\) |
| 2. |
The acceleration of the plate is greater than \(g.\) |
| 3. |
The acceleration of the plate is less than \(g.\) |
| 4. |
The plate comes to a stop and rebounds upward. |
160. A convex lens forms a real image of the same size as that of the object, but when it is moved closer by \(15~\text{cm}\) it forms a real image that is twice as large. The focal length of the lens is:
1. \(15~\text{cm}\)
2. \(30~\text{cm}\)
3. \(45~\text{cm}\)
4. \(90~\text{cm}\)
161. A ray of light is incident normally onto the surface
\(AB\) of a right-angled prism
\(ABC\) (
\(\angle B=90^\circ\)) and emerges from the surface
\(AC\). The refractive index of the material of the prism is
\(\mu = \sqrt{2}\).
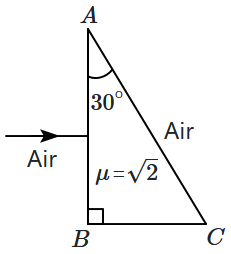
At what other angle of incidence on the surface
\(AB\) will the ray undergo the same total deviation as before?
1.
\(15^{\circ}\)
2.
\(30^{\circ}\)
3.
\(45^{\circ}\)
4.
\(60^{\circ}\)
162. A double-slit experiment is performed with one slit four times as wide as the other. Assuming that the amplitude of light coming from a slit is proportional to the slit-width, the ratio of the maximum and minimum intensities on the screen,
\(\dfrac{I_{max}}{I_{min}}=\)
| 1. |
\(\dfrac{5}{3}\) |
2. |
\(\dfrac{3}{1}\) |
| 3. |
\(\dfrac{25}{9}\) |
4. |
\(\dfrac{9}{1}\) |
163. Photons of energy
\(h\nu\) (
\(\nu=\) frequency) incident onto a photocathode cause the emission of photoelectrons, and a current flows in the circuit. Assume the photoelectrons are emitted normal to the photocathode. A uniform magnetic field
\(B\) is switched on parallel to the plates (into the plane). The work function is
\(W\) and the separation between the cathode and anode is
\(d.\) The minimum value of
\(B\) for which the ammeter shows zero current satisfies:
(where
\(m\) is the mass of the electron and
\(e\) is the charge of the electron)

| 1. |
\(Bed=(h\nu-W)\) |
2. |
\((Bed)^2=m(h\nu+W)\) |
| 3. |
\((Bed)^2=2m(h\nu-W)\) |
4. |
\((Bed)^2=2m(h\nu+W)\) |
164. Let
\(n_h,~n_e\) be the number of holes and electrons in a sample of silicon
\((\mathrm{Si})\) doped with phosphorous
\((\mathrm{P})\). Then:
| 1. |
\(n_h=n_e\) |
| 2. |
\(n_h>n_e\) |
| 3. |
\(n_e>n_h\) |
| 4. |
any of the above may be true depending on the temperature of the sample. |
165. Two semi-circular current-carrying wires are placed in the same plane so that they share a common centre. The magnetic field due to any one of them at the common centre has the magnitude, \(B_O\). When one of them is tilted so that it is in a perpendicular plane, with the same centre, the magnetic field at the common centre is \(B\). Then,
1. \(B =2B_O\)
2. \(B~=\dfrac{B_O}{2}\)
3. \(B=\sqrt 2 B_O\)
4. \(B=\dfrac{B_O}{\sqrt 2}\)
166. Given below are two statements:
| Assertion (A): |
As the drift velocity decreases, the current flowing through the conductor decreases. |
| Reason (R): |
The current flowing through a conductor is directly proportional to the drift velocity. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
167. The end
\('B'\) of the circuit is earthed
\((V_B = 0)\) while a sinusoidal voltage is applied at
\('A';\) \(V_{A}=V_{0} \sin \omega t.\) The rms voltage across the capacitor
\({C}\) equals that across the upper resistor
\({R}\) (as shown in the figure). What is the phase difference between the current through the capacitor and the voltage across the capacitor when no current flows out at
\(X?\)
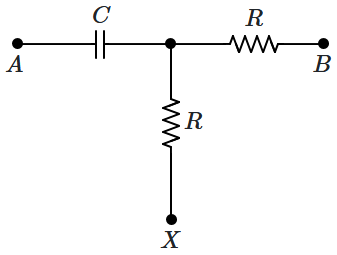
| 1. |
\(0^{\circ}\) |
2. |
\(45^{\circ}\) |
| 3. |
\(90^{\circ}\) |
4. |
\(180^{\circ}\) |
168. Let the average nuclear force exerted by a proton on a proton be \(\mathrm{F_{pp}},\) by a proton on a neutron be \(\mathrm{F_{pn}}\) and by a neutron on a neutron be \(\mathrm{F_{nn}}.\) When their separations are nearly \(1~\text{fm},\) then:
1. \( \mathrm{F}_{\mathrm{pp}}<\mathrm{F}_{\mathrm{pn}}<\mathrm{F}_{\mathrm{nn}} \)
2. \( \mathrm{F}_{\mathrm{pn}}>\mathrm{F}_{\mathrm{pp}}>\mathrm{F}_{\mathrm{nn}} \)
3. \( \mathrm{F}_{\mathrm{pp}}>\mathrm{F}_{\mathrm{pn}}>\mathrm{F}_{\mathrm{nn}} \)
4. \(\mathrm{F}_{\mathrm{pp}}=\mathrm{F}_{\mathrm{pn}}=\mathrm{F}_{\mathrm{nn}}\)
169. A particle is projected with a speed \(u\) so that it has the maximum horizontal range under gravity. The speed is increased to \(v\) (without changing the direction of its projection), so that, after projection, it passes above its previous point of impact but at a height which is equal to its previous maximum height. Then,
1. \(2u=v\)
2. \(3u=2v\)
3. \(2u = \sqrt 3 v\)
4. \(3u =\sqrt 2 v\)
170. During an experiment, an ideal gas is observed to obey the law:
\(PV^3= \text{constant}\), as the process is conducted. If the volume of the gas doubles during this process, the absolute temperature of the gas changes from
\(T\) to:
| 1. |
\(2T\) |
2. |
\(4T\) |
| 3. |
\(\dfrac T 2\) |
4. |
\(\dfrac T 4\) |
171. Two identical springs are joined end-to-end to form a single spring and a block is suspended from the combination. The time period of oscillation is
\(T_1.\) Alternatively, if the springs were joined in parallel, the time period is
\(T_2.\) Then:
| 1. |
\(T_1 = T_2\) |
| 2. |
\(T_1 = 2T_2 \) |
| 3. |
\(T_2 = 2T_1\) |
| 4. |
none of the above is true. |
172. A
\(100\) cm wire of mass
\(40\) g is fixed at both ends. A tuning fork, vibrating at a frequency of
\(50\) Hz, sets the wire into resonance in its fundamental mode. Then, the tension in the wire is:
| 1. |
\(400\) N |
2. |
\(100\) N |
| 3. |
\(25\) N |
4. |
\(1600\) N |
173. If the absolute temperature increases by
\(1\%\), the frequency of an organ pipe will:
| 1. |
increase by \(1\%\) |
2. |
decrease by \(1\%\) |
| 3. |
increase by \(0.5\%\) |
4. |
decrease by \(0.5\%\) |
174. Two short bar magnets are placed at large distances from each other, with their axes aligned along the same line but in opposite directions to each other. A null point is formed between them at a distance
\(r_1\) from the first magnet and
\(r_2\) from the second. If the dipole moments of the magnets are
\(P_1\) and
\(P_2\), then:
| 1. |
\(\dfrac{r_1}{r_2}=\dfrac{P_1}{P_2}\) |
| 2. |
\(\left(\dfrac{r_1}{r_2}\right)^2=\dfrac{P_1}{P_2} \) |
| 3. |
\(\left(\dfrac{r_1}{r_2}\right)^3=\dfrac{P_1}{P_2} \) |
| 4. |
none of the above is true. |
175. A small solenoid is kept inside a much larger solenoid, with their axes parallel to each other. The small solenoid has a cross-sectional radius \(r_1,\) length \(l_1\) and the total number of turns \(N_1.\) The corresponding quantities for the larger solenoid are: \(r_2,~ l_2,~ N_2\) respectively.
The smaller solenoid, which had a mutual inductance \(M_0\) with the larger one, is now rotated so that it makes an angle \(\theta\), with the larger one: the angle being measured between their axes. The mutual inductance is (nearly):
1. \(M_0\)
2. \(M_0\cos\theta\)
3. \(M_0\sin\theta\)
4. \(M_0\cos^2\theta\)
176. In stimulated emission, an incoming photon interacts with an excited atom (e.g.
\(\mathrm{H}^{*}\)) and brings the atom to its ground state, while an extra photon is emitted - as it happens in a laser. When a photon stimulates the emission of another photon, the two photons have:
| 1. |
the same phase. |
| 2. |
the same energy. |
| 3. |
the same direction. |
| 4. |
the same phase, energy, and direction. |
177. The velocity-time graph of a particle, moving along a straight time, is shown in the figure. The curve, when plotted, takes the form of a 'circle'. The magnitude of the average acceleration of the particle is:

| 1. |
\(1\) m/s2 |
| 2. |
\(2\) m/s2 |
| 3. |
less than \(1\) m/s2 |
| 4. |
greater than \(2\) m/s2 |
178. A small block of mass
\(m\) lies on a frictionless wedge of mass
\(M,\) which is pushed horizontally to the right by means of a constant force
\(F\). There is no relative motion between the block and the wedge. Let the work done by
\(F\) on
\(M\) be
\(W_F\). The work done by the normal force (between
\(M\) &
\(m\)) on
\(m\) be
\(W_m\). Both are measured for the same time interval.

Then:
| 1. |
\(\dfrac{W_F}{M}=\dfrac{W_m}{m}\) |
2. |
\(W_F\cdot M=W_m\cdot m\) |
| 3. |
\(\dfrac{W_F}{M+m}=\dfrac{W_m}{m}\) |
4. |
\(\dfrac{W_F}{M}=\dfrac{W_m}{m+M}\) |
179. A uniform rod of mass
\(M\) and length
\(L\) lies at rest on a smooth horizontal plane, as shown in the figure. A particle of mass
\(m,\) moving with an initial velocity
\(u\) strikes one end
\((A)\) of the rod and stops. The initial velocity
\(u\) is perpendicular to the length
\((AB)\) of the rod.
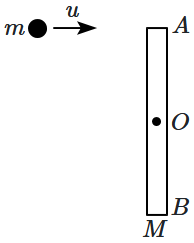
The angular momentum of the system, about
\(O,\) before the collision is:
| 1. |
zero |
2. |
\(muL\) |
| 3. |
\(\dfrac{muL}2\) |
4. |
\(\dfrac{muL}2+\dfrac{1}{12}ML^2\Big(\dfrac{u}{L}\Big)\) |
180. The quantity \(\eta\) is defined by: \(\eta=\dfrac{1}{\mu_0\sigma}\)
where \(\mu_0\) is the permeability of free space and \(\sigma\) is the electrical conductivity (of a plasma). \(\eta\) is referred to as the magnetic diffusivity. Its SI unit is:
1. m2/s
2. m/s2
3. C-m2/s
4. T-m/s
181. A uniform solid sphere lying on a smooth horizontal surface is acted upon by a horizontal force, which is impulsive and transmits to it a
momentum \(mv,\) where \(m\) is the mass of the sphere (see figure). The angular velocity of the sphere is:
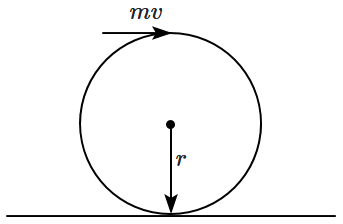
| 1. |
\(\dfrac{v}{r}\) |
2. |
\(\dfrac{2v}{r}\) |
| 3. |
\(\dfrac32\dfrac vr\) |
4. |
\(\dfrac52\dfrac vr\) |
182. Given below are two statements:
| Assertion (A): |
As a gas bubble rises from the bottom of a lake, its volume decreases. |
| Reason (R): |
As the gas bubble rises from the bottom of a lake, the pressure of the gas within decreases. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
183. A straight horizontal wire of
mass \(m\) and
length \(l,\) and having a negligible resistance can slide freely on a pair of conducting parallel rails, placed vertically. The rails are connected at the top by a capacitor
\(C.\) A uniform magnetic field
\(B\) exists in the region, perpendicular to the plane of the rails. The wire:

| 1. |
falls with uniform velocity. |
| 2. |
accelerates down with acceleration less than \(g\). |
| 3. |
accelerates down with acceleration equal to \(g\). |
| 4. |
moves down and eventually comes to rest. |
184. Which of the following situation(s), can cause the radiation of an electromagnetic wave?
| (A) |
a varying sinusoidal current flowing through a capacitor |
| (B) |
an electric dipole, whose size (and magnitude) is oscillating with time |
| (C) |
a steady current flowing through a toroid |
Choose the correct option from the given ones:
| 1. |
only (A) |
| 2. |
only (B) |
| 3. |
only (A) & (B) |
| 4. |
(A), (B), (C) |
185. Consider the circuit shown in the above diagram. The output
\((Y)\) of the circuit does not depend on
\(A.\) Then,
\(Y=\)

1.
\(B\) AND
\(C\)
2.
\(B\) OR
\(C\)
3.
\(B\)
4.
\(C\)
Physics - Section B
186. The net force acting on a projectile at the highest point of its trajectory, moving with a speed \(v,\) at a height \(H\) is:
(\(m\) is the mass of the projectile)
1. zero
2. \(mg\)
3. \(mg+\dfrac{mv^2}{H}\)
4. \(mg-\dfrac{mv^2}{H}\)
187. A water pump lifts water from a well which is \(20~\text{m}\) deep at a rate of \(60~\text{kg/min}\) – with negligible kinetic energy Take \(g = 10~\text{m/s}^2\). The losses in the pump are \(20\%\). The power of the pump is:
1. \(200~\text{W}\)
2. \(250~\text{W}\)
3. \(2000~\text{W}\)
4. \(500~\text{W}\)
188. The first overtone of a closed organ pipe of length \(l_1\) matches the fundamental frequency of an open pipe of length \(l_2\). Then,
1. \(l_1 = 2l_2\)
2. \(l_2 = 2l_1\)
3. \(2l_1 = 3l_2\)
4. \(2l_2 = 3l_1\)
189. A metallic sphere of radius
\(R\) is given a charge
\(Q.\) The energy stored in the sphere due to this charge is:
| 1. |
\(\dfrac{Q^2}{4\pi\varepsilon_0R}\) |
2. |
\(\dfrac{2Q^2}{4\pi\varepsilon_0R}\) |
| 3. |
\(\dfrac12 \left ( \dfrac{Q^2}{4\pi\varepsilon_0R} \right )\) |
4. |
\(\dfrac{Q^2}{16\pi\varepsilon_0R}\) |
190. The potential difference between the ends of a
\(12~\text{V}\) battery when it is being charged by a
\(2~\text{A}\) charger is found to be
\(13.2~\text{V}\). If this battery is connected in a circuit with a
\(6~\Omega\) resistance, the current will be nearly:
| 1. |
\(2~\text{A}\) |
2. |
\(1~\text{A}\) |
| 3. |
\(1.8~\text{A}\) |
4. |
\(2.2~\text{A}\) |
191. A current-carrying loop of wire in the shape of a square of side
\(a\) lies in the
\(x\text-y\) plane. A uniform magnetic field
\(B\) acts in the plane. Then:

| 1. |
The force on the loop is \(4iaB\). |
| 2. |
The torque on the loop is \(ia^2B\). |
| 3. |
The force on the loop is \(\sqrt {2} iaB\). |
| 4. |
The torque on the loop is \(\sqrt{2}ia^2B\). |
192. A small object is placed on the principal axis of a convex lens and it forms a real image with a (transverse) magnification of
\(2.\) If the object were to move perpendicular to the principal axis with a speed
\(u,\) the speed of the image will be:
| 1. |
\(u\) |
2. |
\(u/2\) |
| 3. |
\(2u\) |
4. |
\(4u\) |
193. Given below are two statements:
| Assertion (A): |
The resistance of a photodiode decreases when light having photons of sufficient energy is incident on it. |
| Reason (R): |
When energetic photons fall on the \(\mathrm{p\text{-}n}\) junction of a photodiode, electron-hole pairs are created due to the breaking of the valence bonds. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
194. A metallic ball (work function: \(2\) eV) is irradiated with light consisting of photons of wavelength \(200\) nm. The ball has an initial charge, giving it a potential \(1\) V. Take the product of Planck's constant and velocity of light, hc as \(1240\) eV-nm. The final potential of the ball, when photoemission practically stops, is:
1. \(2\) V
2. \(3.2\) V
3. \(4.2\) V
4. \(5.2\) V
195. There is no friction anywhere, and the string and the pulley are ideal. Assume that
\(m_1 < m_2. \) The acceleration of
\(m_2\) down the plane is
\(a.\) Then,
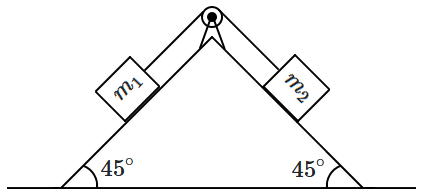
| 1. |
as \(\dfrac{m_1 }{m_2} \rightarrow 1 , a \rightarrow 0\) |
| 2. |
as \(\dfrac{m_1 }{m_2} \rightarrow 0 , a \rightarrow g\) |
| 3. |
\(a\) varies linearly with \(\dfrac{m_1 }{m_2}\) |
| 4. |
all the above are true. |
196. Which of the following diagrams correctly illustrates the streamline flow of a liquid out of a tank?
197. The arrangement shows two pistons
\(P_1,P_2\) with a rigid connecting rod
\(C\) so that they can slide together with respect to the two fixed cylinders of cross-sectional areas
\(A_1,A_2\) respectively. The two cylinders are connected by means of two pipes to a small cylinder (of area
\(A\)) with a piston at the bottom on which is applied a force
\(F.\) The interior of the pipes and cylinder is filled with an incompressible oil. Ignore any pressure variations due to gravity. The net force on the two pistons
\(P_1, P_2\) is:

1.
\(\dfrac{F}{A}(A_1+A_2)\) to right.
2.
\(\dfrac{F}{A}(A_1+A_2)\) to left.
3.
\(\dfrac{F}{A}(A_2-A_1)\) to right.
4.
\(\dfrac{F}{A}(A_2-A_1)\) to left.
198. The pressure of an ideal gas
\(\left(\gamma=\dfrac32\right)\) is increased by
\(1\%\) in an adiabatic process. The temperature of the gas:
| 1. |
increases by \(1.5\%\) |
| 2. |
decreases by \(1.5\%\) |
| 3. |
increases by \(\frac13\%\) |
| 4. |
increases by \(\frac23\%\) |
199. A block
\(A\) is placed on a spring and causes it to be compressed; when another
block \(B\) is placed on top of the first one the compression increases by
\(125\%.\) The time period of small oscillations is
\(T_A\) when
\(A\) is present, and is
\(T_{AB},\) when both
\(A~\&~B\) are present. Then,

| 1. |
\(T_{AB}=\dfrac{3}{2}~T_A\) |
2. |
\(T_{AB}=\dfrac{\sqrt5}{2}~T_A\) |
| 3. |
\(T_{AB}=\dfrac{1}{2}~T_A\) |
4. |
\(T_{AB}=\dfrac{2}{3}~T_A\) |
200. The fundamental frequencies of a closed pipe and an open pipe are identical. The first overtone for the closed pipe is
\(f_c\) and for the open pipe is
\(f_o.\) Their ratio
\(\dfrac{f_c}{f_o}\) is:
| 1. |
\(1\) |
2. |
\(1/2\) |
| 3. |
\(2/3\) |
4. |
\(3/2\) |
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course