Botany - Section A
1. Identify the correct relationship of an ovary and an ovule in angiosperms:
| 1. |
ovary is the enlarged apical portion of the pistil; the ovary contains ovules, which develop into seeds upon fertilization |
| 2. |
ovary is the enlarged basal portion of the pistil; the ovary contains ovules, which develop into seeds upon fertilization |
| 3. |
ovary is the enlarged basal portion of the pistil; the ovary contains ovules, which develop into fruits upon fertilization |
| 4. |
ovary is the enlarged apical portion of the pistil; the ovary contains ovules, which develop into fruits upon fertilization |
2. In angiosperms:
| I: |
Each anther develops four microsporangia |
| II: |
Each microspore mother cell undergoes meiosis and produces four haploid microspores |
| III: |
Each microspore gives rise to a male gametophyte |
1. Only
I and
II are correct
2. Only
I and
III are correct
3. Only
II and
III are correct
4.
I,
II and
III are correct
3. In contrast to
Rhizobium species,
Azotobacter species are:
| 1. |
lacking nitrogenase |
| 2. |
free-living, nitrogen-fixing bacteria |
| 3. |
obligate symbionts |
| 4. |
Gram-positive bacteria |
4. Lady beetles, and in particular their larvae, are voracious predators of:
| 1. | Aphids | 2. | Mosquito |
| 3. | Spider mites | 4. | Nematodes |
5. Match each item in Column I with one in Column II and select the correct match from the codes given:
|
Column I
[Flower] |
|
Column II
[Pollination by] |
| A. |
Brassica |
P. |
Water |
| B. |
Zostera |
Q. |
Moth |
| C. |
Grasses |
R. |
Wind |
| D. |
Yucca |
S. |
Honey bees |
Codes:
|
A |
B |
C |
D |
| 1. |
S |
P |
R |
Q |
| 2. |
Q |
R |
P |
S |
| 3. |
S |
P |
Q |
R |
| 4. |
Q |
R |
S |
P |
6. Which RNA molecule plays structural and catalytic role during translation?
| 1. |
miRNA |
2. |
rRNA |
| 3. |
tRNA |
4. |
mRNA |
7. During the decomposition of detritus, the correct chronological sequence of steps involved is:
| 1. |
Fragmentation → Leaching → Catabolism |
| 2. |
Catabolism → Leaching → Fragmentation |
| 3. |
Leaching → Fragmentation → Catabolism |
| 4. |
There is no sequence as all steps occur simultaneously |
8.
| Assertion (A): |
It makes sense to make concentrated efforts to conserve hotspots of biodiversity. |
| Reason (R): |
They cover majority of the area of the planet earth. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
9. The initial carboxylation reaction:
| 1. |
occurs in mesophyll cells in C3 plants and in bundle sheath cells in C4 plants |
| 2. |
occurs in bundle sheath cells in C3 plants and in mesophyll cells in C4 plants |
| 3. |
occurs in mesophyll cells in both C3 plants and in C4 plants |
| 4. |
occurs in bundle sheath cells in both C3 plants and in C4 plants |
10. Select the genotype amongst the given genotypes which will result in lightest skin colour in humans:
| 1. |
AAbbCC |
2. |
AABBCc |
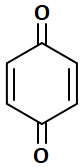
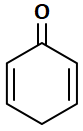
| 3. |
aaBBCC |
4. |
AaBbCc |
11. To form a nucleosome:
| 1. |
the negatively charged DNA is wrapped around the positively charged histone octamer |
| 2. |
the negatively charged DNA is wrapped around the positively charged histone tetramer |
| 3. |
the positively charged DNA is wrapped around the negatively charged histone octamer |
| 4. |
the positively charged DNA is wrapped around the negatively charged histone tetramer |
12. A segment of DNA coding for a polypeptide is a/an:
1. Recon
2. Exon
3. Cistron
4. Intron
14. Which of the following is/are a surface structure of the bacterial cell but do not play a role in motility?
I. Flagella
II. Pili
III. Fimbriae
| 1. |
Only I |
2. |
Only II |
| 3. |
Only II and III |
4. |
Only III |
15. Identify the incorrectly matched pair:
| 1. |
Having the sepals united |
Gamosepalous |
| 2. |
Sterile stamen |
Staminate |
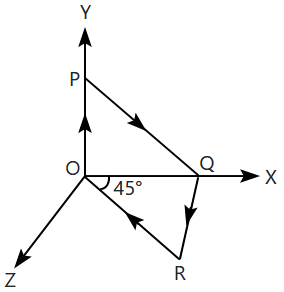
| 3. |
Stamens attached to perianth |
Epiphyllous |
| 4. |
Having the carpels of the gynoecium fused in a compound ovary |
Syncarpous |
16. Both Mycoplasma and Euglenoids:
1. can survive without oxygen
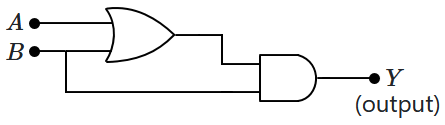
2. are unicellular eukaryotes
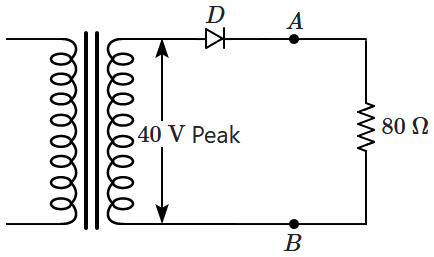
3. lack cell wall
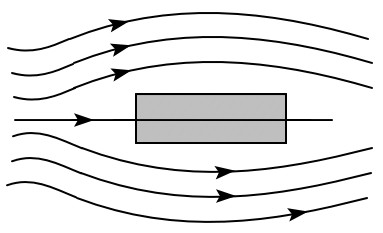
4. have two flagella
17. Regarding the epidermal tissue system of the plant:
| 1. |
Cuticle is present in almost all roots. |
| 2. |
Only guard cells lack chloroplasts. |
| 3. |
Guard cells are bean-shaped in grasses. |
| 4. |
Trichomes are usually multicellular. |
18. Abnormally folded infectious proteins can, in humans, cause:
1. Mad cow disease
2. Scrapie
3. Creutzfeldt-Jacob disease
4. MERS
19. When the ratio of carbon dioxide and oxygen in the vicinity of RuBisCO is 1, the affinity of the enzyme:
| 1. |
is nearly nil for oxygen |
| 2. |
is about equal for both carbon dioxide and oxygen |
| 3. |
is much greater for oxygen than carbon dioxide |
| 4. |
is much greater for carbon dioxide than oxygen |
20. The number of carbon atoms in all the following molecules is 4 except in:
| 1. | α ketoglutaric acid | 2. | Oxaloacetic acid |
| 3. | Succinic acid | 4. | Malic acid |
21. PGRs can be broadly divided into two groups – plant growth promoters and plant growth inhibitors. Ethylene:
| 1. |
belongs to the group of plant growth promoters |
| 2. |
belongs to the group of plant growth inhibitors |
| 3. |
could fit either of the groups, but is largely an inhibitor of growth activities |
| 4. |
could fit either of the groups, but is largely a promoter of growth activities |
22. Antipodal is an adjective that means ‘diametrically opposite’. Where are the antipodal cells located in an embryosac of an angiosperm?
1. At the micropylar end
2. At the chalazal end
3. In the middle
4. There are three – one at micropylar and two at chalazal end
23. Study the given diagram and select the best option:

| 1. |
The mating between I-1 and I-II is consanguineous |
| 2. |
The first born child of I-1 and I-II is a male |
| 3. |
The first born child of I-1 and I-II is a female |
| 4. |
The first born child of I-1 and I-II is affected by a genetic disease |
24. Identify the correct statement:
| 1. |
Roquefort cheese is ripened by a growing a specific bacterium on them |
| 2. |
LAB improves the nutritional quality of curd by increasing Vitamin A |
| 3. |
Aspergillus niger is used for commercial production of citric acid |
| 4. |
Statins are used to lower blood glucose levels in IDDM |
25. Identify the option where the features are correctly matched to the types with respect to mustard:
|
Flower symmetry |
Ovary position |
Androecium |
Gynoecium |
| 1. |
Zygomorphic |
Inferior |
Variable length stamens within a flower |
Parietal placentation |
| 2. |
Zygomorphic |
Inferior |
Polyadelphous |
Marginal placentation |
| 3. |
Actinomorphic |
Superior |
Polyadelphous |
Axile placentation |
| 4. |
Actinomorphic |
Superior |
Variable length stamens within a flower |
Parietal placentation |
26. Both Xylem and Phloem are complex tissues. At maturity, the living component of xylem and the dead component of phloem respectively are:
| 1. |
Xylem parenchyma and Phloem parenchyma |
| 2. |
Xylem parenchyma and Phloem fibres |
| 3. |
Xylem fibres and Phloem fibres |
| 4. |
Xylem fibres and Phloem parenchyma |
27. Consider the following statements:
| I: |
Among invertebrates maximum species richness is seen in insects |
| II: |
Among vertebrates maximum species richness is seen in mammals |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
28. A dominant gametophyte will be seen in the life cycle of:
1. Marchantia
2. Sequoia
3. Adiantum
4. Eucalyptus
29. Given below are two statements:
| Statement I: |
Pyramid of energy is always upright and is the most efficient. |
| Statement II: |
Pyramid of biomass in sea is generally inverted. |
In the light of the above statements, choose the most appropriate answer from the options given below:
1. Statement I is correct but Statement II is incorrect.
2. Statement I is incorrect but Statement II is correct.
3. Both Statement I and Statement II are correct.
4. Both Statement I and Statement II are incorrect.
30. Match
List-I with
List-II:
|
List-I |
|
List-II |
| (a) |
Sacred groves |
(i) |
Alien species |
| (b) |
Zoological park |
(ii) |
Release of large quantity of oxygen |
| (c) |
Nile perch |
(iii) |
Ex-situ conservation |
| (d) |
Amazon forest |
(iv) |
Khasi Hills in Meghalaya |
Choose the correct answer from the options given below:
|
(a) |
(b) |
(c) |
(d) |
| 1. |
(iv) |
(iii) |
(i) |
(ii) |
| 2. |
(ii) |
(iv) |
(i) |
(iii) |
| 3. |
(iv) |
(i) |
(ii) |
(iii) |
| 4. |
(iv) |
(iii) |
(ii) |
(i) |
31. What is the expected percentage of F
2 progeny with yellow and inflated pod in dihybrid cross experiment involving pea plants with green coloured, inflated pod and yellow coloured constricted pod?
| 1. |
100% |
2. |
56.25% |
| 3. |
18.75 % |
4. |
9% |
32. Consider the given two statements:
| Statement I: |
Relationship between a species of moth and the plant Yucca is obligatory. |
| Statement II: |
The moth deposits its eggs in the locule of the ovary and the flower, in turn, gets pollinated by the moth. |
| 1. |
Statement I is correct; Statement II is correct |
| 2. |
Statement I is incorrect; Statement II is correct |
| 3. |
Statement I is correct; Statement II is incorrect |
| 4. |
Statement I is incorrect; Statement II is incorrect |
33. All the following are floral characteristics of plants belonging to the Gramineae family except:
1. Flowers are characteristically arranged in spikelets
2. The flowers are usually unisexual
3. The perianth is reduced to two scales, called lodicules
4. The fruit is a caryopsis
34. Consider the given two statements:
| Assertion (A): |
The protons or hydrogen ions that are produced by the splitting of water accumulate within the lumen of the thylakoids. |
| Reason (R): |
Splitting of the water molecule takes place on the inner side of the thylakoid membrane |
| 1. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 2. |
(A) is True; (R) is False |
| 3. |
(A) is False; (R) is False |
| 4. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
35. Who gave the rivet-popper hypothesis?
| 1. |
Humboldt |
2. |
Ehlrich |
| 3. |
Tilman |
4. |
Tansley |
Botany - Section B
36. If you consider the overall flow of electrons in the light reactions, it can best be described as from:
| 1. |
Glucose to oxygen |
| 2. |
PS I to PS II |
| 3. |
Antenna complex to the reaction centre |
| 4. |
Water to NADPH |
37. In some species, such as coffee, the endosperm does not develop. Instead, the nucellus produces a nutritive tissue termed:
| 1. |
Perisperm |
2. |
Cotyledon |
| 3. |
Albumen |
4. |
Scutellum |
38. Match the terms given in
Column-I with their given definition in
Column-II and select the correct match from the codes given:
| Column-I |
Column-II |
| A. |
Homozygous |
P. |
having two different alleles of a particular gene or genes |
| B. |
Heterozygous |
Q. |
having two identical alleles of a particular gene or genes |
| C. |
Homologous |
R. |
not having the same alleles or genes in the same order of arrangement |
| D. |
Heterologous |
S. |
having the same alleles or genes in the same order of arrangement |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
S |
R |
Q |
P |
| 3. |
Q |
P |
S |
R |
| 4. |
P |
Q |
S |
R |
39. What is incorrect about the Double helix structure of DNA molecule?
1. The two strands are antiparallel
2. The two strands are complementary
3. The pitch is 34 nm
4. The two strands are coiled in a right-handed fashion
40. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
Growth, at a cellular level, is measured by a variety of parameters such as
increase in fresh weight, dry weight, length, area, volume and cell number. |
| Reason (R): |
Such parameters are more or less proportional to the increase in protoplasm
which is primarily responsible for growth at cellular level. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
41. Nostoc:
| 1. |
is a green alga |
| 2. |
is unicellular eukaryote capable of forming filamentous colonies |
| 3. |
can fix atmospheric nitrogen |
| 4. |
carry out anoxygenic photosynthesis |
42. In
Dianthus or
Primrose, the ovule is:
| 1. |
borne on a ridge along the ventral suture of the ovary |
| 2. |
attached to axial placenta in a multilocular ovary |
| 3. |
borne on central axis and septa are absent |
| 4. |
single and attached to placenta at the base of the ovary |
43. During Mendel’s investigations into inheritance patterns it was for the first time that:
| 1. |
experiments were conducted on garden pea |
| 2. |
artificial pollination experiments were carried out |
| 3. |
statistical analysis and mathematical logic were applied to problems in biology |
| 4. |
all characteristics were passed to the next generation through blending inheritance |
44. In glycolysis, which of the following conversions is an energy yielding process?
1. Glucose to Glucose-6-phosphate
2. Fructose-6-phosphate to Fructose 1,6- biphopshate
3. BPGA to 3-phosphoglyceric acid
4. 3-phosphoglyceric acid to 2-phosphoglycerate
45. RuBP is a:
| 1. |
Aldopentose sugar |
2. |
Ketopentose sugar |
| 3. |
Aldohexose sugar |
4. |
Ketohexose sugar |
46. What would be true for both
Cycas and
Pinus?
| 1. |
Cycas is a gymnosperm and Pinus is a pteridophyte |
| 2. |
The gametophyte is dominant in the life cycle of Cycas and the sporophytes is dominant in the life cycle of Pinus |
| 3. |
Roots of Pinus have mycorrhiza association and coralloid roots of Cycas are associated with nitrogen-fixing cyanobacteria |
| 4. |
In Pinus, the male and female cones are borne on different trees and in Cycas on the same tree |
47. Which relationship given in column I is not correctly matched with the partner apparently not deriving any benefit out of the relationship [Column II]?
|
Column I |
Column II |
| 1. |
Orchid growing on a mango branch |
Mango |
| 2. |
Barnacles growing on back of a whale |
Whale |
| 3. |
Sea anemone and clown fish |
Sea anemone |
| 4. |
Lichen |
Algal partner |
48. Consider the given two statements:
| Assertion (A): |
Regarding Down's syndrome, parents of the affected individual are usually genetically normal. |
| Reason (R): |
The probability increases from low in 20-year-old mothers to higher in those of age 45. |
| 1. |
(A) is True but (R) is False |
| 2. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 3. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 4. |
(A) is False but (R) is True |
49. Consider the two statements:
| Statement I: |
In C4 plants, photorespiration does not occur. |
| Statement II: |
C4 plants have a mechanism that increases the concentration of CO2 at the RuBisCO enzyme site. |
| 1. |
Statement I is correct; Statement II is correct |
| 2. |
Statement I is incorrect; Statement II is correct |
| 3. |
Statement I is correct; Statement II is incorrect |
| 4. |
Statement I is incorrect; Statement II is incorrect |
50. Which of the following plant growth regulators enhances the respiration rate during ripening of the fruits?
| 1. |
ABA |
2. |
Ethylene |
| 3. |
Auxins |
4. |
Cytokinins |
Zoology - Section A
51. The testes of a male child aged four years will have:
| 1. |
primary spermatocytes |
2. |
immature sperms |
| 3. |
spermatogonia |
4. |
spermatids |
52. Predators:
| 1. |
act as conduits for energy transfer across trophic levels |
| 2. |
do not include herbivores in broad ecological context |
| 3. |
indiscriminately kill their prey |
| 4. |
have a very high biotic potential |
53. Consider the two statements
| I: |
Phenylketonuria is caused by mutation in a pleiotropic gene |
| II: |
Sickle cell anemia is inherited as an autosomal-recessive condition |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
54. Which of the following human diseases is not transmitted by faecal-oral route ?
1. Ascariasis
2. Viral Diarrhoea
3. Filariasis
4. Typhoid
55. Identify the incorrectly matched pair:
| 1. |
Length of Testis |
4 to 5 cm |
| 2. |
Width of testis |
2 to 3 cm |
| 3. |
Length of ovary |
5 to 6 cm |
| 4. |
Length of fallopian tube |
10 to 12 cm |
56. Which of the following animals is not matched to its correct scientific name?
| 1. |
Penguin: Aptenodytes |
2. |
Tortoise: Testudo |
| 3. |
Platypus: Macropus |
4. |
Angel fish: Pterophyllum |
57. Which of the following is not a function of adrenaline and noradrenaline?
1. Piloerection
2. Pupilary constriction
3. Increase in strength of heart contraction
4. Breakdown of glycogen, lipids, and proteins.
58. Which of the following stages of meiosis is not correctly matched with the event of cell division?
| 1. |
Metaphase I |
Bivalent alignment on the equatorial plate |
| 2. |
Anaphase I |
Splitting of centromere |
| 3. |
Prophase II |
Nuclear membrane disappears by the end |
| 4. |
Metaphase II |
Microtubules from the opposite poles of the spindle get attached to the kinetochores of sister chromatids |
59. To increase renal medullary interstitial osmolality, NaCl passes to the interstitium from:
| 1. |
The thick segment of ascending limb of the loop of Henle |
| 2. |
The collecting tubule |
| 3. |
Descending limb of vasa recta |
| 4. |
The Proximal Convoluted Tubule |
60. The main challenge for production of insulin using rDNA techniques was:
1. getting insulin assembled into a mature form.
2. isolating DNA in pure form from human cell
3. selecting the vector for cloning the gene
4. purification of the human insulin
61. The first transgenic cow produced:
| 1. |
enzyme to treat emphysema |
| 2. |
human protein enriched milk |
| 3. |
enzyme to treat phenylketonuria |
| 4. |
organs for xenotransplantation |
62. What controls the copy number of linked DNA in a transformed bacterial cell?
| 1. |
Ori |
2. |
Cloning sites |
| 3. |
Selectable marker |
4. |
Reporter gene |
63. Modern Homo sapiens arose:
1. 75000-10000 years ago
2. 100000-40000 years ago
3. 1.5 mya
4. 3-4 mya
64. At present, the legal age for marriage in India:
| 1. |
has been raised to 21 years for females |
| 2. |
has been reduced to 18 years for males |
| 3. |
is 18 years for females and 21 years for males |
| 4. |
is 18 years for males and 21 years for females |
65. Occurrence of which of the following is unlikely?
| 1. |
Ciliated epithelium in fallopian tube |
| 2. |
Cuboidal brush bordered epithelium in PCT |
| 3. |
Columnar brush bordered epithelium in small intestine |
| 4. |
Compound epithelium at the diffusion boundaries |
66. Which secondary metabolite is used as a drug in the treatment of certain cancers?
1. Abrin
2. Digoxin
3. Concanavalin A
4. Vinblastin
67. The conducting part of the human respiratory system transports atmospheric air to alveoli and also performs all the following functions except:
1. Clears the inhaled air of foreign particles
2. Humidifies the air
3. Brings the air to body temperature
4. Provides site for diffusion of gases into the blood
68. If an Rh –ve mother is carrying an Rh +ve foetus, she is likely to be exposed to the Rh antigen:
1. At the time of implantation of the blastocyst
2. At the time when the placenta becomes functional
3. When the first movements of the baby can be felt
4. At the time of delivery
69. The central nervous system in chordates is not:
| 1. |
Single |
| 2. |
Hollow |
| 3. |
Ventral |
| 4. |
Derived from embryonic ectoderm |
70. The number of:
| 1. |
Aromatic proteinogenic amino acids is 5 |
| 2. |
Carbon atoms in arachidonic acid including carboxyl carbon is 20 |
| 3. |
Fatty acids in cholesterol is 3 |
| 4. |
Polypeptide chains in quaternary structure of haemoglobin is 2 |
71. In the diffusion membrane in human lungs, a basement membrane supports:
| I. Alveolar epithelium |
II. Vascular endothelium |
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
72. A cardiovascular centre that can moderate the cardiac function through the autonomic neural system is located in the:
| 1. |
Pons varoli |
2. |
Medulla oblongata |
| 3. |
Hypothalamus |
4. |
Cerebrum |
73. ADH:
| I: |
is released when an excessive loss of fluid from the body activates osmoreceptors in the body. |
| II: |
prevents dieresis by facilitating water reabsorption from the initial and proximal parts of the tubules. |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
74. Identify the incorrect statement regarding the ultra-structure of a skeletal muscle:
| 1. |
Z lines bisect the I band |
| 2. |
G actin is a polymer of F actins |
| 3. |
A subunit of troponin masks active binding sites for myosin on the actin filament. |
| 4. |
Globular head of myosin has ATPase activity. |
75. Consider the given two statements:
| I: |
In a neuron, Nissl’s granules are present in dendrites as well as cell body |
| II: |
Unmyelinated neurons in peripheral neural system are not enclosed by a Schwann cell |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
76. Identify the correctly matched pair:
| 1. |
Hyposecretion of growth hormone in adults |
Acromegaly |
| 2. |
Lack of insulin |
Diabetes insipidus |
| 3. |
Hyperthyroidism |
Graves’ disease |
| 4. |
Hypocortisolism |
Cushing’s disease |
77. The frequency of a recessive allele in a population at genetic equilibrium is 0.3. What is the frequency of homozygous dominant genotype in this population?
1. 0.49
2. 0.09
3. 0.21
4. 0.7
78. While cutting dsDNA the progression of a restriction enzyme digestion can be checked by employing:
1. Agarose gel electrophoresis
2. FISH
3. Ethidium bromide
4. Autoradiography
79. Meselson and Stahl were able to distinguish heavy DNA molecules from the normal DNA by:
1. growing them in radioactive 15NH4Cl environment
2. centrifugation in a CsCl density gradient
3. radioactive tagging with 32P
4. agarose gel electrophoresis
80. In bacteria, the formation of peptide bond is catalyzed by a ribozyme that is a part of the:
| 1. |
50S (23S component) ribosome subunit |
| 2. |
30S (28S component) ribosome subunit |
| 3. |
60S (28S component) ribosome subunit |
| 4. |
40S (23S component) ribosome subunit |
81. Which of the following drugs is a very effective sedative and painkiller, acts on its receptors in CNS and GIT and is also a CNS suppressant?
1. Morphine
2. Marijuana
3. Cocaine
4. Atropine
82. Identify the correct statements:
| I: |
Macrophages in body and leucocytes in blood exhibit amoeboid movement. |
| II: |
Passage of ova through the female reproductive tract is facilitated by the ciliary movement. |
| III: |
Human sperm moves with the help of flagellum |
1. Only
I and
II
2. Only
I and
III
3. Only
II and
III
4.
I,
II and
III
83. The anterior pituitary gland in humans:
| I: |
is the storage and release centre for neurohormones produced elsewhere |
| II: |
is connected to hypothalamus by a portal circulation |
| III: |
is also called as adenohypophysis |
1. Only
II is correct
2. Only
III is correct
3. Only
I and
III are correct
4. Only
II and
III are correct
84. Thyroid gland in humans play an important role in:
I: Regulation of basal metabolic rate.
II: Mental development
III: Calcium homeostasis
| 1. |
Only I and II |
2. |
Only I and III |
| 3. |
Only II and III |
4. |
I, II and III |
85. Consider the given two statements:
| Assertion (A): |
The tiger census in our national parks and tiger reserves is often based on pug marks and fecal pellets. |
| Reason (R): |
For ecological investigations, it is mandatory to know the absolute population densities in all instances. |
| 1. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 2. |
(A) is True but (R) is False |
| 3. |
(A) is False but (R) is True |
| 4. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
Zoology - Section B
86. The partial pressures are not equal:
1. for oxygen in deoxygenated blood and tissues
2. for oxygen in alveoli and oxygenated blood
3. for carbon dioxide in alveoli and oxygenated blood
4. for carbon dioxide in deoxygenated blood and tissues
87. In haemodialysis, the cleared blood is pumped back to the body:
1. through a vein after adding heparin
2. through a vein after adding anti-heparin
3. through an artery after adding heparin
4. through an artery after adding anti-heparin
88. Dwarfism can be caused by the deficiency, during childhood, of:
| I. |
Growth hormone |
| II. |
Thyroid hormones |
| 1. |
Only I and the dwarf will have normal mental development |
| 2. |
Only II and the dwarf will also be mentally retarded |
| 3. |
Both I and II and in both cases the mental development will be affected |
| 4. |
Both I and II and mental development will be effected only in II. |
89. The arrow in the figure shows:

| 1. |
Rete testis |
2. |
Caput epididymis |
| 3. |
Cauda epididymis |
4. |
Vasa efferentia |
90. Which hormone, also called the ‘birth hormone’, initiates and sustains labour contractions during parturition?
| 1. |
Progesterone |
2. |
Oxytocin |
| 3. |
Estrogen |
4. |
Relaxin |
91. Copper releasing IUDs:
| I: |
are not effective as emergency contraceptives |
| II: |
damage sperms and disrupt their motility |
1. Both
I and
II are correct
2. Only
I is correct
3. Only
II is correct
4. Both
I and
II are incorrect
92. Which of the following tools of recombinant DNA technology is incorrectly paired with its use?
| 1. |
DNA ligase |
cutting DNA, creating sticky ends of restriction fragments |
| 2. |
DNA polymerase |
polymerase chain reaction to amplify sections of DNA |
| 3. |
Electrophoresis |
separation of DNA fragments |
| 4. |
Reverse transcriptase |
production of cDNA from mRNA |
93. Match each item in Column I with one in Column II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Number of genes on chromosome 1 |
P. |
2968 |
| B. |
Number of genes on Y chromosome |
Q. |
Less than 2 |
| C. |
Percent of genome that codes for proteins |
R. |
Over 50 |
| D. |
Percent of discovered genes without known functions |
S. |
231 |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
S |
Q |
R |
| 2. |
S |
P |
Q |
R |
| 3. |
P |
S |
R |
Q |
| 4. |
S |
P |
R |
Q |
94. All the following are functions of lymph in the human body except:
| 1. |
Helps in digestion of fats |
| 2. |
Returns proteins and excess interstitial fluid to the bloodstream |
| 3. |
Transports nutrients and hormones |
| 4. |
Helps in immunity against disease-causing agents |
95. Consider the given statements regarding sponges:
| I: |
They have a cellular level of organisation |
| II: |
Digestion is intracellular |
| III: |
Fertilization is external and development is direct |
1. Only
I and
II are correct
2. Only
I and
III are correct
3. Only
II and
III are correct
4.
I,
II and
III are correct
96. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
Lipids are macromolecules |
| Reason (R): |
They have a molecular weight range of ten thousand Daltons and above. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
97. In a healthy human adult, what contributes maximum to the total body weight?
1. Muscle
2. Bone
3. Fat
4. Hair
98. Which of the following conditions can be caused by exposure to allergens?
1. Asthma
2. Rheumatoid arthritis
3. Japanese encephalitis
4. Leukaemia
99. The frog has:
| I: |
Hepatic portal system |
| II: |
Renal portal system |
| 1. |
Only I is correct |
2. |
Only II is correct |
| 3. |
Both I and II are correct |
4. |
Both I and II are incorrect |
100. Consider the given two statements:
| Assertion (A): |
Cortisol increases blood glucose levels. |
| Reason (R): |
Cortisol increases the glucose uptake by the body cells. |
| 1. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 2. |
(A) is True; (R) is False |
| 3. |
(A) is False; (R) is False |
| 4. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
Chemistry - Section A
101.
Consider the following reactions:

‘A’ and ‘B’ ,respectively, in the above reactions are:
1. CH3COOC2H5, C2H5OH
102. How is the volume of \(\mathrm{KMnO_4}\) solution determined during titration?
1. By measuring the mass of \(\mathrm{KMnO_4}\) crystals used
2. By monitoring the colour change of the solution
3. By titrating with a standard solution of oxalic acid
4. By measuring the pH of the solution
103.
| Statement I: |
Boiling point increases in the order of
HBr < HI < HCl< HF |
| Statement II: |
Intermolecular hydrogen bonding exists in HF molecules. |
| 1. |
Statement I is incorrect but Statement II is correct |
| 2. |
Both Statement I and Statement II are correct |
| 3. |
Both Statement I and Statement II are incorrect |
| 4. |
Statement I is correct but Statement II is incorrect |
104. The vapour pressure of a solution is 2% less than the vapour pressure of pure water. The molality of the solution is approximately
1. 0.92
2. 1.11
3. 1.26
4. 1.52
105. Consider the reaction, A + B → Products. On keeping the concentration of B fixed and increasing the concentration of A 3 times, the rate of reaction increases 27 times. Now the concentration of A and B are both doubled and it is found that the rate of reaction becomes 8 times. The orders with respect to A and B, respectively, are:
1. 3 and 1
2. 2 and 1
3. 3 and 0
4. 2 and 0
106. The molecule that is not hydrolyzed by water at 25oC is:
1. SF4
2. SF6
3. BF3
4. SiCl4
107. The spin-only magnetic moment of the
[\(ZCl_4\)]²⁻complex is 3.87 BM.
Identify the metal ion represented by Z:
108. The Crystal Field Stabilization Energy (CFSE) and the spin-only magnetic moment in Bohr Magneton (BM) for the complex K3[Fe(CN)6] are, respectively.
1. \(-2.0 \Delta_0 ~and ~ \sqrt {35}~ BM\)
2. \(-2.0 \Delta_0 ~and ~ \sqrt {3}~ BM\)
3. \(-0.4 \Delta_0 ~and ~ \sqrt {3} ~BM\)
4. \(-2.4 \Delta_0 ~and ~ \sqrt {35}~ BM\)
109. The correct order of acidity among nitrophenols is:
1. o- > m- > p-
2. o- > p- > m-
3. p- > m- > o-
4. p- > o- > m-
110. The compound that would react with Tollen's reagent is:
| 1. |
CH3CHO |
| 2. |
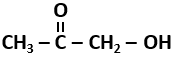 |
| 3. |
HCOOH |
| 4. |
All of the above. |
111. Lactose is a disaccharide composed of:
| 1. |
\(\alpha - D-\)Glucose and \(\beta-D-\)Galactose |
| 2. |
\(\beta-D-\)Glucose and \(\beta-D-\)Galactose |
| 3. |
\(\beta - D-\)Glucose and \(\alpha-D-\)Galactose |
| 4. |
\(\alpha - D-\)Glucose and \(\alpha-D-\)Galactose |
112. The number of unpaired electrons in Ni and Ni2+, respectively, are (Z of Ni = 28)
1. 2, 2
2. 2, 4
3. 2, 0
4. 4, 2
113. The bond order of B
2 is one. The type of bond and magnetic behavior of B
2, respectively, are:
| 1. |
\(\sigma \) and diamagnetic |
2. |
\(\pi\) and diamagnetic |
| 3. |
\(\sigma \) and paramagnetic |
4. |
\(\pi\) and paramagnetic |
114. How many Faradays are required to deposit 127 grams of copper from a copper sulfate solution?
(Atomic weight of copper = 63.5)
1. 1 F
2. 2 F
3. 3 F
4. 4 F
115. What is the total count of isomers, encompassing both geometrical and optical types, for the coordination compound [Pt(en)₂Cl₂]?
| 1. |
Two (2) |
2. |
Three (3) |
| 3. |
Four (4) |
4. |
Five (5) |
116. When alkyl halide is reacted with AgCN, then alkyl isocyanide is formed as a major product.
The reason for this reaction is:
1. AgCN is an electrovalent compound
2. AgCN is a covalent compound
3. In cyanide ion, nitrogen is more nucleophilic
4. Alkyl isocyanide is more stable than alkyl cyanide
117. Which of the following processes is endothermic?
1. \(O + e^- \rightarrow O^-\)
2. \(N + e^- \rightarrow N^-\)
3. \(Cl + e^- \rightarrow Cl^-\)
4. None of the above
118. For which of the following reaction,
\(\Delta H < \Delta E \) ?
| 1. |
\(N_2 (g) + 3H_2(g) \rightarrow 2NH_3 (g) \) |
| 2. |
\(PCl_5 (g) \rightarrow PCl_3(g) + Cl_2(g) \) |
| 3. |
\(H_2 (g) + I_2(g) \rightarrow 2HI(g) \) |
| 4. |
\(CaCO_3 (s) \rightarrow CaO (s) + CO_2 (g) \) |
119. Given below are two statements:
| Statement I: |
\(HCOOH\) is more acidic than \(C_6H_5COOH\) |
| Statement II: |
\(CH_3COOH\) is less acidic than \(C_6H_5COOH\) |
| 1. |
Statement I is correct and Statement II is incorrect |
| 2. |
Statement I is incorrect and Statement II is incorrect |
| 3. |
Statement I is correct and Statement II is correct |
| 4. |
Statement I is incorrect and Statement II is correct |
120. When but-2-yne is reacted with hydrogen in the presence of Lindlar's catalyst, the product formed is:
| 1. |
Butane |
2. |
But-1-ene |
| 3. |
Cis-but-2-ene |
4. |
Trans-but-2-ene |
121. The increasing order (lowest first) for the values of charge/mass ratio for electron (e-), proton (p+), neutron (n), and alpha particle (α), respectively, is:
1. \(e, ~p,~n,~\alpha\)
2. \(n,~p,~e~,~\alpha\)
3. \(n,~p,~\alpha,~e\)
4. \(n,~\alpha,~p,~e\)
122. The carboxylic acid among the following that can give the Hell Volhard Zelinsky (HVZ) reaction is:
1. HCOOH
2. 2,2-Dimethyl propanoic acid
3. Benzoic acid
4. Propanoic acid
123. The most basic compound among the following is:
1. \(C_6H_5NH_2 \)
2. \(p-NO_2-C_6H_4NH_2 \)
3. \(m-NO_2-C_6H_4NH_2 \)
4. \(C_6H_5CH_2NH_2 \)
124. 10.6 gm \(Na_2CO_3 \) is dissolved in 250 ml \(H_2O \) . The molarity of \(Na^+ \) ions and \(CO^{2-}_3\) in this solution, respectively, are:
1. 0.4 M and 0.2 M
2. 0.8 M and 0.4 M
3. 0.4 M and 0.4 M
4. 0.4 M and 0.8 M
125. The molecular geometry of XeF5⁺ will be:
1. Square pyramidal
2. Square planar
3. Trigonal bipyramidal
4. Octahedral
126. 100 ml of \(HCl\) (pH=1) is mixed with 150 ml of \(HCl\) (pH =2). The pH of the resulting solution will be:
(log 4.6 = 0.65)
1. 1.35
2. 1.5
3. 1.8
4. 1.9
127. In Caro's acid \((H_2SO_5)\), the oxidation number of 'S' will be?
1. +8
2. +7
3. +6
4. +5
128. The monomer unit of cellulose is:
1. \(\alpha -\mathrm{D~ Glucose }\)
2. \(\beta -\mathrm{D~ Glucose }\)
3. \(\alpha -\text{D Glucose} + \beta-\text{D Glucose}\)
4. Fructose
129. Consider the given two statements:
| Statement I: |
1 amu is defined as the mass exactly equal to one-twelfth of the mass of one carbon-12 atom. |
| Statement II: |
Number of atoms in 8 u of He is 2NA. |
In the light of above statements, choose the correct option from the following :
| 1. |
Both Statement I and Statement II are correct. |
| 2. |
Both Statement I and Statement II are incorrect. |
| 3. |
Statement I is correct but Statement II is incorrect. |
| 4. |
Statement I is incorrect but Statement II is correct |
130.
| Assertion (A): |
Ionization enthalpy increases along each series of transition elements from left to right. However, small variations occur. |
| Reason (R): |
There is a corresponding increase in nuclear charge which accompanies the filling of electrons in the inner d-orbitals. |
| 1. |
(A) is True but (R) is False |
| 2. |
(A) is False but (R) is True |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
131. T-shaped molecule among the following is:
1. SF4
2. NH3
3. BrCl3
4. FO3−
132. What is the molecular mass of 'X', if its 2% (w/v) solution exhibits the same osmotic pressure as a 4% (w/v) solution of sucrose?
| 1. |
570 g/mol |
2. |
205.2 g/mol |
| 3. |
171 g/mol |
4. |
None of these |
133.
| Assertion (A): |
Zinc is not a transition metal. |
| Reason (R): |
Zinc has a full d10 configuration in its ground state as well as in its common oxidation states. |
In light of the above statements, choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
(A) is False but (R) is True |
134. Match the isolated gaseous ions given in column I with the number of unpaired electrons given in column II and assign the correct code:
|
Column I |
|
Column II |
| (A) |
\(\mathrm{Cr}^{3+}\) |
(P) |
2 |
| (B) |
\(\mathrm{Fe}^{3+}\) |
(Q) |
3 |
| (C) |
\(\mathrm{Ni}^{2+}\) |
(R) |
4 |
| (D) |
\(\mathrm{Mn}^{3+}\) |
(S) |
5 |
|
A |
B |
C |
D |
| 1. |
R |
P |
Q |
S |
| 2. |
Q |
S |
P |
R |
| 3. |
Q |
S |
R |
P |
| 4. |
S |
Q |
R |
P |
135. During electrolysis of aqueous NaCl solution using 'Pt' electrodes, the product(s) among the following formed during electrolysis is/are:
(a) \(H_2\)
(b) \(Cl_2\)
(c) NaOH
| 1. |
Only (a) and (b) |
2. |
Only (b) and (c) |
| 3. |
Only (a) and (c) |
4. |
(a), (b) and (c) |
Chemistry - Section B
136. A compound with the formula C₄H₁₀O (x) can react with sodium. Upon vigorous oxidation, this compound yields a carboxylic acid with the formula C₄H₈O₂ (y). The compound (x) could be:
1. Ether
2. 2° Alcohol
3. 2° Alcohol
4. 1° Alcohol
137.
| Assertion (A): |
On hydrolysis, PCl3 produces a dibasic oxyacid of phosphorous.
|
| Reason (R): |
H3PO2 is a dibasic acid. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
138. Which of the following compounds can show tautomerism?
1. I, II, III, IV
2. I and II
3. III and IV
4. Only II
139. The correct order of stability of the following carbocations is:
1. I > II > III > IV
2. IV > III > II > I
3. I > IV > III > II
4. I > IV > II > III
140. If A (volatile solute) and B (volatile solvent) form an azeotropic mixture then the correct statement regarding the above azeotropic mixture will be -
| 1. |
Mole fraction of A in the vapour phase is more than the mole fraction of A or B in liquid phase |
| 2. |
Mole fraction of A or B in the vapour phase is more than the mole fraction of A or B in liquid phase |
| 3. |
Mole fraction of A or B in the vapour phase is same as the mole fraction of A or B in liquid phase |
| 4. |
Mole fraction of A and B in the vapour phase is less than the mole fraction of A or B in liquid phase |
141. 9.5 gram MgCl
2 is dissolved in 500 g of water. The difference in boiling point and freezing point of the solution will be (K
b for
H2O = 0.52 K/m, K
f for
H2O = 1.86 K/m, Molar weight of
MgCl2 = 95) (assume that MgCl
2 is 100% dissociated)
| 1. |
103.57 oC |
2. |
101.428 oC |
| 3. |
374.428 K |
4. |
372.196 K |
142. Which metal evolves hydrogen gas when reacted with dilute HNO₃?
143. In a hydrogen concentration cell using solutions with pH 3 and pH 5, which pH belongs at the cathode to make the reaction spontaneous, and what is the resulting cell EMF respectively? ( Given:\(2.303 RT \over F\)=0.059)
1. Solution of pH = 3 and +0.118 V
2. Solution of pH = 5 and +0.118 V
3. Solution of pH = 3 and +0.059 V
4. Solution of pH = 5 and +0.059 V
144. A first-order reaction completes 20% in 10 minutes.
The time required for 99.9% completion of the reaction will be:
1. 100 minutes approximately
2. 200 minutes approximately
3. 300 minutes approximately
4. 400 minutes approximately
145. The nature of an aqueous solution of NaHCO₃ is:
1. Acidic
2. Alkaline
3. Slightly alkaline
4. Slightly acidic
146.
| Assertion (A): |
In freezing of water, \(\Delta H = -ve~~and ~~\Delta S = -ve \) |
| Reason (R): |
For the spontaneity of freezing of water, \(\Delta H < T \Delta S \) |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
147. When an excess of Cl2 reacts with CH4 in the presence of sunlight, the major product formed is:
1. \(CH_3Cl\)
2. \(CH_2Cl_2\)
3. \(CHCl_3\)
4. \(CCl_4\)
148. Acetic acid dimerises in benzene solution. The value of Van't Hoff factor (i) for the dimerization of acetic acid is 0.6. Therefore, the percentage of dimerization of acetic acid will be:
| 1. |
20% |
2. |
40% |
| 3. |
60% |
4. |
80% |
149. Which of the following solutions has the highest osmotic pressure?
1. 1% glucose solution
2. 1% urea solution
3. 1% sucrose solution
4. All of the above have the same osmotic pressure
150. Given below are two statements:
| Statement I: |
Cr2+ is oxidising agent and Mn3+ is a reducing agent in nature. |
| Statement II: |
Sc3+ compounds are repelled by the applied magnetic field. |
In light of the above statements, choose the most appropriate answer from the options given below:
| 1. |
Both I and II are correct. |
| 2. |
Both I and II are incorrect. |
| 3. |
I is correct but II is incorrect. |
| 4. |
I is incorrect but II is correct. |
Physics - Section A
151. A block of mass \(m\) is suspended by means of a string, which passes over a massless pulley and is connected to a second block of mass, \({2m},\) sitting on a smooth horizontal table as shown. The system is released. The acceleration of the blocks (acceleration due to gravity is \(g\)) is:
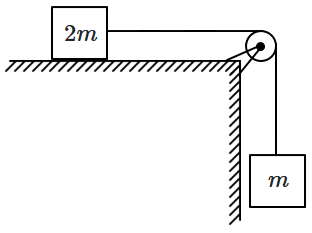
| 1. |
\(\dfrac g3\) |
2. |
\(\dfrac {2g}3\) |
| 3. |
\(\dfrac {g}2\) |
4. |
\(g\) |
152. The ratio \(C_P/C_V=1.5\) for a certain ideal gas. The gas is taken at an initial pressure of \(2\) kPa and compressed suddenly to \(\dfrac14\) of its initial volume. The final pressure is:
1. \(\dfrac12\) kPa
2. \(4\) kPa
3. \(8\) kPa
4. \(16\) kPa
153. Two rods of identical dimensions are joined end-to-end, and the ends of the composite rod are kept at \(0^\circ\text{C}\) and \(100^\circ\text{C}\) (as shown in the diagram). The temperature of the joint is found to be \(40^\circ\text{C}.\) Assuming no loss of heat through the sides of the rods, the ratio of the conductivities of the rods \(\dfrac{K_1}{K_2}\) is:

| 1. |
\(\dfrac32\) |
2. |
\(\dfrac23\) |
| 3. |
\(\dfrac11\) |
4. |
\(\dfrac{\sqrt3}{\sqrt2}\) |
154. The pressure of an ideal gas is written as \(p=\dfrac{2E}{3V},\) where \(E\) is the total kinetic energy, and \(V\) is the volume.
This statement is:
| 1. |
always true. |
| 2. |
true for mono-atomic gases. |
| 3. |
always false. |
| 4. |
true for diatomic gases. |
155. A particle of mass \(m\) executes SHM along a straight line with an amplitude \(A\) and frequency \(f.\)
| Assertion (A): |
The kinetic energy of the particle undergoes oscillation with a frequency \(2f.\) |
| Reason (R): |
Velocity of the particle, \(v = {\dfrac{dx}{dt}}\), its kinetic energy equals \({\dfrac 12}mv^2\) and the particle oscillates sinusoidally with a frequency \(f\). |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
156. If the absolute temperature of a gas is increased by \(1\text{%},\) the speed of sound in the gas:
1. increases by \(\sqrt{1} \text{%}\)
2. decreases by \(\sqrt{1} \text{%}\)
3. increases by \(1\text{%}\)
4. increases by \(0.5\text{%}\)
157. Identical point charges (
\(q\) each), are placed at the eight corners of a cube of side
\(a.\) When one of the charges is removed, the electric field at the center becomes
\(E_c.\)
Now, identical point charges (same magnitude
\(q\) each), are placed at the four corners of a square of side
\(a.\) When one of the charges is removed, the electric field at the center becomes
\(E_s.\) Then,
| 1. |
\(\dfrac{E_s}{2}=\dfrac{E_C}{3}\) |
2. |
\(\dfrac{E_s}{3}=\dfrac{E_C}{2}\) |
| 3. |
\(\dfrac{E_s}{\sqrt2}=\dfrac{E_C}{\sqrt3}\) |
4. |
\(\dfrac{E_s}{\sqrt3}=\dfrac{E_C}{\sqrt2}\) |
158. The capacitors are initially uncharged. The three points
\(A,B,C\) are maintained at potentials
\(V_A=4~\text{V},\) \(V_B=1~\text{V}\) and
\(V_C=1~\text{V}.\) The potential at
\(X,\) \(V_X=\)
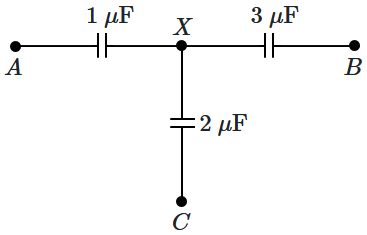
1.
\(2~\text V\)
2.
\(1.5~\text V\)
3.
\(3~\text V\)
4.
\(0.5~\text V\)
159. A rod
\(XY\) of length
\(l\) is placed in a uniform magnetic field
\(B\), as shown in the diagram. The rod moves with a velocity
\(v\), making an angle of
\(60^\circ\) with its length. The EMF induced in the rod is:

| 1. |
\(vBl\) |
2. |
\(\dfrac{vBl}{2}\) |
| 3. |
\(\dfrac{\sqrt 3}{2}vBl\) |
4. |
\(\dfrac{1}{\sqrt 3}vBl\) |
160. A horizontal square loop of area
\(A\) has
\(n\) turns of wire. It is immersed in a uniform, rotating magnetic field
\(B\) which is initially perpendicular to the plane of the loop. The field rotates with an angular speed
\(\omega\) about a diagonal of the loop. The EMF induced across the loop is:

| 1. |
constant, of magnitude \(n\omega BA\). |
| 2. |
increasing with time \(t\), of magnitude \(n\omega^2BAt\). |
| 3. |
decreasing with time \(t\), of magnitude \(\dfrac{nBA}{t}\). |
| 4. |
sinusoidal with time \(t\), of amplitude \(n\omega BA\). |
161. In the given scenario, the voltage, \(V_2 > V_1,\) and no current flow through the source on the left. The phase difference between the two sources is \(\phi.\)

Which of the following expressions correctly relates
\(\phi\text{?}\)
| 1. |
\(R\sin\phi= \dfrac{1}{\omega C}\) |
2. |
\(R\cos\phi= \dfrac{1}{\omega C}\) |
| 3. |
\(R\tan\phi= \dfrac{1}{\omega C}\) |
4. |
\(R\cot\phi= \dfrac{1}{\omega C}\) |
162. Find the minimum order of a green fringe (\(\lambda = 500\) nm) which overlaps a dark fringe of violet (\(\lambda = 400\) nm) in a Young's double-slit experiment conducted with these two colours.
1. \(4\)
2. \(2\)
3. \(5\)
4. \(2.5\)
163. All the resistances to the left of the vertical line
\(PQ\) are
\(1~\Omega\), while those on the right of line
\(PQ\) are
\(2~\Omega\) as shown in the figure above. The equivalent resistance between
\(A\) and
\(B\) is:

| 1. |
\(18~\Omega\) |
2. |
\(9~\Omega\) |
| 3. |
\(4.5~\Omega\) |
4. |
\(2.25~\Omega\) |
164. An electromagnetic waveform given by
\(\vec{E}=E_{0} \hat{j} \sin \omega t\cos k x\) is set up in a certain region of space, where
\(\vec{E}\) represents the electric field. The magnetic field associated with this waveform oscillates along the direction of:
| 1. |
\(\hat {i}\) |
2. |
\(\hat {j}\) |
| 3. |
\(\hat{k} \) |
4. |
\(\hat{j} + \hat{k}\) |
165. If the ratio of magnifications produced by a simple microscope in near point adjustment and far point adjustment is \(\dfrac{6}{5},\) then the focal length of the lens is:
(take \(D=25\text{cm} \))
1. \(5\text{cm}\)
2. \(10\text{cm}\)
3. \(55\text{cm}\)
4. \(0.2\text{cm}\)
166. A parallel beam of light is incident onto a convex lens and after passing through the lens, it gets reflected from a plane mirror and passes through the lens again. The focal length of the lens is
\(f.\) Where should the mirror be placed so that the final beam emerges parallel?
| 1. |
Just behind the lens. |
| 2. |
At the focus of the lens. |
| 3. |
At a distance of \(2f\) from the lens. |
| 4. |
At a distance of \(3f\) from the lens. |
167. A muon is approximately \(200\) times heavier than an electron, but it is negatively charged, just like the electron. If a 'muonic'
\(\mathrm H\)-like atom is formed by a proton and a negative muon, assuming everything else except the mass to be the same, what will be the radius of the orbit for the ground state?
(assume the Bohr radius (\(\mathrm H\)-atom) to be \(r_\mathrm H\))
1. \(r_\mathrm{H}\)
2. \(r_\mathrm{H} \times 200\)
3. \(\dfrac{r_\mathrm{H} }{200}\)
4. \(r_\mathrm{H}(200)^2\)
168. The dimension of which group of quantities is the same?
\(h\) : Planck's constant, \(K\) : kinetic energy, \(\omega\) : angular speed/frequency, \(F\): force, \(L\) : inductance, \(i\) : current, \(q\) : charge, \(t\) : time, \(x\): distance
1. \(h, ~Ftx,~ Liq\)
2. \(K , h\omega , \omega Li\)
3. \(Fx, ~Li^2,~K \omega\)
4. \(\dfrac{Fx}{t} ,~ Kx, ~ ht\)
169. A particle is projected vertically upwards with a speed \(u\) and moves under the force of gravity. The distance travelled by the particle during its entire motion (until it returns) is \(d_1.\) If the force of gravity were to be switched off, and the particle travelled for the same length of time, then the distance travelled is \(d_2.\) Then,
1. \(d_2 = d_1\)
2. \(d_2 = 2d_1\)
3. \(d_2 = 3d_1\)
4. \(d_2 = 4 d_1\)
170. A particle starts to move along a straight line under a force that delivers constant power \(P,\) starting from rest. The velocity of the particle \(v\) varies with time \(t,\) as:
1. \(v \mathcal{} \propto t\)
2. \(v \propto \dfrac{1}{t}\)
3. \(v \propto \sqrt{t}\)
4. \(v \propto \dfrac{1}{\sqrt{t}}\)
171. A uniform rod
\(AB\) of mass m and length
\(L\) is replaced by three particles – two particles of masses
\(m_1\) each at the ends and another particle of mass
\(m_2\) at its centre. The new system of particles has the same total mass, the same center-of-mass and the same moment of inertia about an axis through its C.M. and perpendicular to
\(AB.\)
If the moment of inertia of the new system of particles is measured about an axis passing through the end
\(A,\) and perpendicular to
\(AB\) then it is found to be
\(I_2.\) For the rod, the same quantity is found to be
\(I_1.\) Then
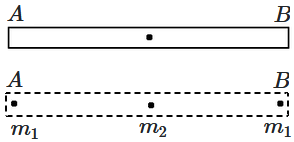
1.
\(I_1 = 2I_2\)
2.
\(I_2 = 2I_1\)
3.
\(I_1 = I_2\)
4.
\(I_1 = 3I_2\)
172. Two particles of masses
\(m_1,~m_2\) are placed on the axis of a uniform circular ring of mass
\(M\) and radius
\(R,\) on opposite sides of the centre of the ring. The distances of
\(m_1,~m_2\) from the centre of the ring are
\(x_1,~x_2\) respectively, and
\(x_1~ x_2 \ll R.\) The net force on the ring vanishes. Then,

| 1. |
\(\dfrac{m_{1}}{x_{1}}=\dfrac{m_{2}}{x_{2}} \) |
2. |
\(\dfrac{m_{1}}{x_{1}^{2}}=\dfrac{m_{2}}{x_{2}^{2}} \) |
| 3. |
\(\dfrac{m_{1}}{x_{1}^{3}}=\dfrac{m_{2}}{x_{2}^{3}} \) |
4. |
\(m_{1} x_{1}=m_{2} x_{2} \) |
173. A wooden block floats submerged at the interface of two liquids, the upper one of specific gravity \(0.4\) and the lower one being water (density: \(1\) g/cc). It is observed that the block has \(\dfrac{1}{3}\) of its volume in water and \(\dfrac{2}{3}\) of it in the upper liquid. The density of the block(in g/cc) is:
1. \(0.5\)
2. \(0.6\)
3. \(0.8\)
4. \(0.9\)
174. A current carrying circular loop of wire is placed in a magnetic field
\(B\), which makes an angle
\(\theta\) with the normal to the loop. The radius of the loop is
\(r\), and the loop carries a current
\(i\). The magnetic interaction energy of the current carrying loop is
\(E_B\) and the torque on the loop has the magnitude
\(\tau_B\).
Which of the following, is independent of
\(\theta?\)
| 1. |
\(E_B\cdot\tau_B\) |
2. |
\(\dfrac{E_B}{\tau_B}\) |
| 3. |
\(E_B^2+\tau_B^2\) |
4. |
\(E_B^2-\tau_B^2\) |
175. A square loop of side length
\(a\) carrying a current
\(i\) is placed as shown in the figure. The magnetic moment of the loop is:
1.
\({ia^2\over2}{[\hat i-\hat k]}\)
2.
\(ia^2{[\hat i-\hat j]}\)
3.
\({ia^2\over\sqrt 2}{[\hat j-\hat k]}\)
4.
\({ia^2\over\sqrt 2}{[\hat i-\hat k]}\)
176. The magnetic field at a point (
\(P\)) on the axis of a circular current carrying wire is
\(\dfrac18\) of the field at its centre. The radius of the circular curve is
\(R.\) The distance between
\(P\) and the cente of the circle
\((OP).\) is:

Then,
| 1. |
\(OP=R\) |
2. |
\(OP=\dfrac R2\) |
| 3. |
\(OP=\sqrt3R\) |
4. |
\(OP=8R\) |
177. The average energy of a neutron produced in fission of \(^{235}_{92}\mathrm{U}\) in MeV is:
1. \(2\) MeV
2. \(3\) MeV
3. \(4\) MeV
4. \(6\) MeV
178. A boat is rowed across a
\(400~\text m\) wide river so that it can reach the opposite bank in a minimum of
\(10\) minutes. No matter which direction the boat is rowed in, it cannot reach a point exactly opposite on the other bank, unless it is rowed at a slightly higher speed. The speed of flow of the river is:
| 1. |
\(2.4~\text{km/h}\) |
2. |
\(4.8~\text{km/h}\) |
| 3. |
\(2.4\sqrt2~\text{km/h}\) |
4. |
\(\dfrac{2.4}{\sqrt2}~\text{km/h}\) |
179. The speed of sound in a gas at temperature
\(T\) is
\(v_s\) while the RMS speed of its molecules is
\(v_r.\) The ratio of specific heats
\((C_p/C_v)\) is equal to:
| 1. |
\(\sqrt{3}\left(\dfrac{v_s}{v_r}\right )\) |
2. |
\(\dfrac{1}{\sqrt3}\Big(\dfrac{v_s}{v_r}\Big)\) |
| 3. |
\(3\Big(\dfrac{v_s}{v_r}\Big)^{2}\) |
4. |
\(\dfrac13\Big(\dfrac{v_s}{v_r}\Big)^2\) |
180. A block of mass
\(m\) slides down the smooth inclined surface of a wedge of
mass \(M\) starting from rest. The wedge is at rest on the horizontal surface beneath it, due to friction. The acceleration of the center of mass of the system of the block and the wedge is:

| 1. |
\(\dfrac{mg~\text{sin}\theta}{m+M}\) |
2. |
\(\dfrac{mg~\text{cos}\theta}{m+M}\) |
| 3. |
\(g~\text{sin}\theta\) |
4. |
zero |
181. Four identical point charges
(\(q\) each) are placed at the four corners of a square of
diagonal \(d.\) The potential at a point which is at a
distance \(\dfrac{d}{2}\) above the centre of the square is:
\(\Big(k=\dfrac{1}{4\pi\varepsilon_0}\Big)\)
| 1. |
\(\dfrac{8~kq}{d}\) |
2. |
\(\dfrac{4~kq}{d}\) |
| 3. |
\(\dfrac{4\sqrt2~kq}{d}\) |
4. |
\(\dfrac{\sqrt2~kq}{d}\) |
182. Ultraviolet photons, each of energy \(20~\text{eV},\) are incident onto a gas of \(\mathrm {H}\)-atoms, causing the emission of electrons. The kinetic energy of the emitted electrons has the value:
1. \(6.4~\text{eV}\)
2. \(7.2~\text{eV}\)
3. \(3.2~\text{eV}\)
4. \(13.6~\text{eV}\)
183. The input signals at
\(A\) and
\(B\) are as follows:
At \(A:\) \(0011010\)
At \(B:\) \(1001101\)
The output at
\(Y\) is:
1.
\(1001101\)
2.
\(0011010\)
3.
\(1101001\)
4.
\(0110010\)
184. The diode shown in the adjacent circuit is ideal and connected to the secondary of an ideal transformer.
The average voltage across the diode (forward direction is taken as positive) is:
| 1. |
zero |
| 2. |
positive, greater than \(20~\text V\) |
| 3. |
positive, less than \(20~\text V\) |
| 4. |
negative |
185. A material when placed in a uniform magnetic field affects the magnetic lines as shown below.
The material may be:
| 1. |
ferromagnetic |
| 2. |
diamagnetic |
| 3. |
paramagnetic |
| 4. |
either ferromagnetic or paramagnetic |
Physics - Section B
186. The loss in weight of a lead block is \(3\) times in liquid \(A\) compared to that in liquid \(B\). In both cases, the block is half immersed. If \(\rho_A, \rho_B\) be the densities of the liquids and \(\rho_L\) the density of lead, then:
| 1. |
\(3\rho_A=\rho_B\) |
2. |
\(\dfrac32\rho_A=\rho_B\) |
| 3. |
\(\rho_A=\dfrac32\rho_B\) |
4. |
\(\rho_A=3\rho_B\) |
187. A metal ball of mass \(2~\text{kg}\) is heated by a \(30~\text{W}\) heater, in a room at \(20^{\circ}\text{C}.\) The temperature of the metal becomes steady at \(50^{\circ}\text{C}.\) If the ball was kept in a room at \(20^{\circ}\text{C}\) while maintaining a temperature of \(10^{\circ}\text{C},\) the rate at which heat must be removed from the ball is:
1. \(20~\text{W}\)
2. \(10~\text{W}\)
3. \(5~\text{W}\)
4. \(1~\text{W}\)
188. A simple pendulum of time period \(T_0\) is taken in a rocket which is accelerating upwards initially and then, after some time, it moves with uniform velocity upward. The time period of the pendulum is observed within the rocket and is found to be \(2T_0.\) The rocket, at that time, must be at a distance (above the earth's surface) of (radius of earth = \(R\)) is:
1. \(\dfrac{R}{2}\)
2. \(\dfrac{R}{4}\)
3. \(R\)
4. \(4R\)
189. A rope of uniform mass per unit length
\(\mu\) is suspended from the ceiling, hanging under its own weight. If a small transverse pulse is formed at its lower end
\(A\), it travels upward with a local speed
\(v=\sqrt {\dfrac{\text{tension}}{\text{mass/length}}}\).

The speed of the pulse is:
| 1. |
maximum at \(A,\) minimum at \(O\) |
| 2. |
minimum at \(A,\) maximum at \(O\) |
| 3. |
uniform |
| 4. |
minimum at \(A\) and \(O,\) maximum in the middle |
190. Two capacitors, one of
\(3~\mu\text{F}\) and the other of
\(1~\mu\text{F}\) are charged to
\(2~\text{V},\) \(4~\text{V}\) and are connected to each other in either of the two ways: their like terminals together
\((A)\) or unlike (i.e. oppositely charged) terminals together
\((B).\) The potential differences are
\(V_A, V_B\) in these cases. Then:
| 1. |
\(V_A= 2V_B\) |
2. |
\(V_A= 4V_B\) |
| 3. |
\(V_A=5V_B\) |
4. |
\(V_A = 6V_B\) |
191. In the circuit shown in the diagram, the resistance
\(r\) is unknown but fixed. The EMF
\(E\) can be varied and the current
\((i)\) passing through the ammeter
\({(A)}\) can be measured in magnitude only.
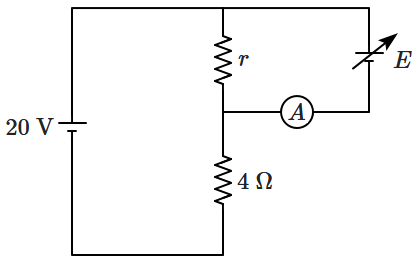
When the EMF
\(E\) is negligible (i.e. practically zero) the ammeter reading will:
| 1. |
be \(5~\text{A},\) independent of \(r\) |
| 2. |
be \(2.5~\text{A},\) independent of \(r\) |
| 3. |
be less than \(5~\text{A},\) decreasing with \(r\) |
| 4. |
be more than \(5~\text{A},\) increasing with \(r\) |
192. The inputs
\(A\) and
\(B\) can be set to
\(+5~\text V\) (representing logic
\(1\)) or
\(0~\text V\) (representing logic
\(0\)). Then, the circuit in the figure below represents:

| 1. |
OR - gate |
2. |
AND - gate |
| 3. |
NOR - gate |
4. |
NAND - gate |
193. A long current-carrying solenoid produces a magnetic field
\(B\) at its centre,
\(O\). When a current-carrying wire is placed parallel to the axis of the solenoid, the field at
\(O\) has the magnitude
\(2B\). The field due to wire has the magnitude (at
\(O\)) of:
| 1. |
\(B\) |
2. |
\(3B\) |
| 3. |
\(\dfrac {B} {\sqrt3}\) |
4. |
\(\sqrt 3~ B\) |
194. Photons of light of wavelength,
\(\lambda=400\) nm
are incident on a composite photocathode consisting of multiple regions with metals having work functions of
\(2.1\) eV and
\(1.1\) eV. The emitted photoelectrons are sent through a retarding potential difference,
\(V_0\). What is the minimum value of
\(V_0\) required to stop all electrons? (take:
\(hc=1240\) eV-nm)
| 1. |
\(1\) V |
2. |
\(1.5\) V |
| 3. |
\(2\) V |
4. |
\(5.2\) V |
195. Two identical equilateral triangular prisms, each of which gives a minimum deviation of
\(60^{\circ}\) are taken: call these prisms
\(A,B\). These are placed as shown in the figure, and a ray of light is incident on prism
\(A\) at minimum deviation. Now prism
\(B\) is cut in half, along the dotted line, and the right half is removed. The deviation of the emerging ray is:

| 1. |
\(90^{\circ}\) |
2. |
\(45^{\circ}\) |
| 3. |
\(60^{\circ}\) |
4. |
\(30^{\circ}\) |
196. Two particles
\(A\),
\(B\) are projected simultaneously from the base of a triangle
\(ABC\). Particle
\(A\) is projected from vertex
\(A\) along
\(AC,\) and particle
\(B\) is projected from vertex
\(B\) along
\(BC\). Their respective velocities are
\(v_A\) &
\(v_B\) and they move with uniform velocities. For the particles to collide:

| 1. |
\(v_A~\text{cos}A=v_B~\text{cos}B\) |
| 2. |
\(v_A~\text{sin}A=v_B~\text{sin}B\) |
| 3. |
\(\dfrac{v_A}{\text{sin}A}=\dfrac{v_B}{\text{sin}B}\) |
| 4. |
\(v_A~\text{tan}A=v_B~\text{tan}B\) |
197. A simple pendulum of length
\(L\) is suspended from a point
\(O\) in a rocket which is ready to be launched from earth. The rocket takes off with an upward acceleration equal to
\(g.\) Immediately after take off, the bob of the pendulum is given a horizontal velocity so that it just completes a vertical circle. The bob's speed (relative to the rocket) at its highest point is
\(v_H,\) where:
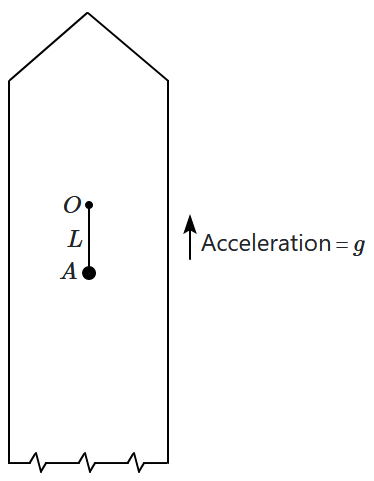
1.
\(v_H=\sqrt{gL}\)
2.
\(v_H=\sqrt{2gL}\)
3.
\(v_H=\sqrt{3gL}\)
4.
\(v_H=\sqrt{5gL}\)
198. \(7\) mole of a certain monoatomic ideal gas undergoes a temperature increase of \(40~\text{K}\) at constant pressure. The increase in the internal energy of the gas in this process is:
(Given: \(R=8.3~ \text{J-K}^{-1} \text{mol}^{-1}\))
1. \(5810~\text{J}\)
2. \(3486~\text{J}\)
3. \(11620~\text{J}\)
4. \(6972~\text{J}\)
199. The two blocks
\(A,B\) have identical masses and are placed on a smooth horizontal plane. The
pulley \(P\) is light, and is rigidly connected to the
block \(A\) by means of a horizontal light rod. The string connecting
block \(B\) is also horizontal. A
force \(F\) is acting vertically on the string, and the two blocks move horizontally. The force on
\(A\) due to rod is:

1.
\(F\)
2.
\(2F\)
3.
\(\sqrt2F\)
4.
\(\dfrac{F}{\sqrt2}\)
200. The bob (mass :
\(m\)) of a simple pendulum is suspended from a long wire of
length \(L.\) When this bob is submerged in a fluid of
density \(\rho,\) it is observed by means of a careful measurement that the extension in the wire is halved. The volume of the bob is:

| 1. |
\(\dfrac{m}{\rho}\) |
2. |
\(\dfrac{2m}{\rho}\) |
| 3. |
\(\dfrac{m}{2\rho}\) |
4. |
\(\dfrac{3m}{2\rho}\) |
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course
.svg)
.svg)
.svg)



















 When the EMF \(E\) is negligible (i.e. practically zero) the ammeter reading will:
When the EMF \(E\) is negligible (i.e. practically zero) the ammeter reading will:





