Botany - Section A
1. The first mitotic division of the zygote in angiosperms divides the fertilized egg into a basal cell and a terminal cell where:
| 1. |
the basal cell degenerates and the terminal cell gives rise to both the embryo and the suspensor |
| 2. |
the terminal cell degenerates and the basal cell gives rise to both the embryo and the suspensor |
| 3. |
the terminal cell gives rise to most of the embryo and the basal cell produces the suspensor |
| 4. |
the basal cell gives rise to most of the embryo and the terminal cell produces the suspensor |
2. Consider the two statements:
| I: |
Biochemical oxygen demand (BOD) is often used as an indicator of the degree of organic pollution of water. |
| II: |
An increase in BOD is used as a gauge of the effectiveness of wastewater treatment plants. |
| 1. |
Only I is correct |
2. |
Only II is correct |
| 3. |
Both I and II are correct |
4. |
Both I and II are incorrect |
3. The trophic level of an organism is the number of steps it is from the start of the food chain. The Level 2 typically will be occupied by:
| 1. |
Primary producer |
2. |
Herbivore |
| 3. |
Carnivore |
4. |
Apex predator |
4. In the given diagram, the correctly labelled parts are:

| 1. |
Only E |
2. |
Only D and E |
| 3. |
Only A and C |
4. |
Only B |
5. During RNA splicing 200-300 adenine residues are added:
| 1. |
at the 3’-end of RNA in a template independent manner |
| 2. |
at the 5’-end of RNA in a template independent manner |
| 3. |
at the 3’-end of RNA in a template dependent manner |
| 4. |
at the 5’-end of RNA in a template dependent manner |
6.
| Assertion (A): |
Thermostable DNA dependent DNA polymerase is used in Polymerase Chain Reaction |
| Reason (R): |
Such enzymes do not require primers and can initiate DNA synthesis |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
7.
| Assertion (A): |
Ex-situ conservation is advisable if the organism is facing a high threat of extinction. |
| Reason (R): |
Ex-situ conservation is an on-site conservation where we conserve and protect the whole ecosystem. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
8. Though their main body is sporophyte, yet, free-living photosynthetic gametophyte can be seen in:
| 1. |
Bryophytes |
2. |
Pteridophytes |
| 3. |
Gymnosperms |
4. |
Angiosperms |
9. Which of the following describes a male in birds?
10. In Oswald Avery, Colin MacLeod and Maclyn McCarty experiment, transformation was inhibited by the use of:
| 1. |
Protease |
2. |
Lipase |
| 3. |
RNase |
4. |
DNase |
11. RNA polymerase III is not responsible for transcription of:
| 1. |
tRNA |
2. |
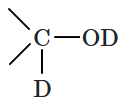
5srRNA |
| 3. |
snRNAs |
4. |
5.8srRNA |
12. All the following will be correct regarding mesosome in a bacterial cell except:
| 1. |
They are differentiated form of cell membrane |
| 2. |
They are essentially infoldings of cell membrane |
| 3. |
They can be in the form of vesicles, tubules and lamellae |
| 4. |
They contain photosynthetic pigments |
13. In mango and coconut:
| 1. |
the fruit is a drupe |
| 2. |
edible part is the mesocarp |
| 3. |
the fruit is parthenocarpic |
| 4. |
thalamus contributes maximum part in the development of the fruit |
14. Two organisms belonging to the same order may not belong to the same:
| 1. |
Class |
2. |
Phylum |
| 3. |
Family |
4. |
Kingdom |
15. In members of Basidiomycetes:
| 1. |
Mycelium is unbranched and coenocytic |
| 2. |
Sex organs are absent and plasmogamy does not take place |
| 3. |
Asexual spores are not found but vegetative reproduction by fragmentation is common |
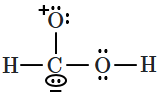
| 4. |
Basidiospores are produced endogenously on the basidium |
16. Match each item in
Column-I with one in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Dicot stem |
P. |
Vascular bundles are arranged in a ring |
| B. |
Monocot stem |
Q. |
Pith is small or inconspicuous |
| C. |
Dicot root |
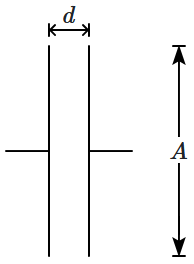
R. |
Vascular bundles are many and scattered in the ground tissue with no definite arrangement. |
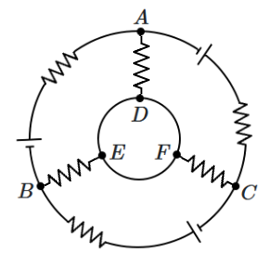
| D. |
Monocot root |
S. |
Pith is large and well developed |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
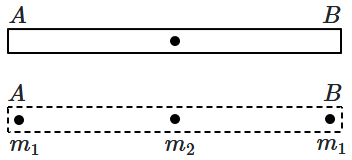
S |
P |
R |
| 2. |
S |
Q |
R |
P |
| 3. |
P |
R |
Q |
S |
| 4. |
R |
P |
S |
Q |
17. Different types of leaves in different phases of life in cotton, coriander and larkspur are an example of:
1. Pleiotropy
2. Phenotypic plasticity
3. Developmental noise
4. Polygenic trait
18. It is possible to induce mutations artificially through the use of radiations. Which of the following is especially suitable for inducing mutations in plants?
1. X rays
2. UV rays
3. γ rays
4. PEG
19. All the following are correct regarding Methanogens except:
| 1. |
They can grow anaerobically on cellulosic material |
| 2. |
They can produce large amounts of methane along with carbon dioxide and hydrogen |
| 3. |
They can be found in anaerobic sludge during sewage treatment and in rumen of cattle |
| 4. |
They are true bacteria and are chemoheterotrophs |
20. Identify the incorrect statement:
| 1. |
Amazon rain forest produces 20% of total oxygen in the Earth’s atmosphere |
| 2. |
All the biodiversity hotspots put together cover about 10 % of the Earth’s land area |
| 3. |
About 31 % of all gymnosperms species in the world face the threat of extinction |
| 4. |
India’s share of the global species diversity is about 8.1 % |
21. Which of the following organisms is not matched with the correct nature of the cell wall present in their cell/cells?
|
Organism |
Nature of cell wall |
| 1. |
True bacterium |
Polysaccharide + Amino acid |
| 2. |
Fungus |
With chitin |
| 3. |
Euglenoids |
Cellulosic |
| 4. |
Diatoms |
Embedded with silica |
22. Fucus is:
| 1. |
a brown alga having oogamous sexual reproduction and haplo-diplontic life cycle |
| 2. |
a red alga having anisogamous sexual reproduction and diplontic life cycle |
| 3. |
a brown alga having oogamous sexual reproduction and diplontic life cycle |
| 4. |
a red alga having isogamous sexual reproduction and diplontic life cycle |
23. In pea:
| 1. |
Flower is zygomorphic, aestivation is vexillary, stamens are diadelphous and placentation is marginal. |
| 2. |
Flower is actinomorphic, aestivation is vexillary, stamens are diadelphous and placentation is axile. |
| 3. |
Flower is zygomorphic, aestivation is twisted, stamens are monodelphous and placentation is parietal. |
| 4. |
Flower is actinomorphic, aestivation is imbricate, stamens are diadelphous and placentation is marginal. |
24. What given anatomical feature is not correct for a dicot stem?
| 1. |
Hypodermis |
Sclerenchymatous |
| 2. |
Endodermis |
Starch sheath |
| 3. |
Medullary rays |
Parenchyma cells |
| 4. |
Protoxylem |
Endarch |
25. In the mitochondrial electron transport system:
| 1. |
Electrons from NADH are oxidized by Complex II |
| 2. |
Ubiquinone is located on the outer surface of the inner membrane |
| 3. |
Complex IV refers to cytochrome c oxidase containing cytochromes a and a3, and two copper centres |
| 4. |
Cytochrome c is an electron carrier located within the inner membrane |
26. Calvin cycle is described under three stages – I: Carboxylation; II: Reduction and III: Regeneration. ATP is required during stage/s:
| 1. |
Only I |
2. |
Only I and III |
| 3. |
Only II and III |
4. |
Only IV |
27. Which of the following will always be true for an ecosystem?
| 1. |
Producers are more in number and biomass than the herbivores |
| 2. |
Pyramid of biomass is inverted |
| 3. |
Pyramid of energy is upright |
| 4. |
Energy at a lower trophic level is less than at a higher level |
28. Even if a plant is infected with a virus:
| I: |
the apical meristem is free of virus |
| II: |
the axillary meristem is free of virus |
| III: |
meristem can be grown in-vitro to obtain virus free plants |
| 1. |
Only I and III are correct |
2. |
Only II and III are correct |
| 3. |
Only I and II are correct |
4. |
I, II and III are correct |
29. What is not correct regarding apomixis?
1. It is production of seeds without fertilization
2. It is a form of asexual reproduction
3. It is fruit production without fertilization
4. Nucellar cells developing into embryos is an example
30. Which of the following is not correct regarding the decomposition of wastes?
| (a) |
Low temperature inhibits decomposition |
| (b) |
Warm and moist environment favors the process |
| (c) |
The process is anaerobic |
| (d) |
It is slower if detritus is rich in proteins and carbohydrates |
| (e) |
Detritus is degraded into simpler inorganic substance by fungal and bacterial enzymes |
Choose the correct answer from the options given below:
| 1. |
(b) and (c) only |
2. |
(c), (a) and (d) only |
| 3. |
(c) and (d) only |
4. |
(c), (d) and (e) only |
31. Give the correct descending order of organisms with reference to their estimated number found in Amazon forest.
| (a) |
Plants |
(b) |
Invertebrates |
| (c) |
Fishes |
(d) |
Mammals |
| (e) |
Birds |
|
|
Choose the correct answer from the options given below:
| 1. |
(a) > (b) > (e) > (d) > (c) |
2. |
(a) > (c) > (d) > (b) > (e) |
| 3. |
(b) > (a) > (e) > (d) > (c) |
4. |
(b) > (a) > (c) > (e) > (d) |
32. The World Summit on sustainable development held in 2002 in Johannesburg, South Africa pledged for:
| 1. |
A significant reduction in the current rate of biodiversity loss. |
| 2. |
Declaration of more biodiversity hotspots. |
| 3. |
Increase in agricultural production |
| 4. |
Collection and preservation of seeds of different genetic strains of commercially important plants. |
33. Some plants such as
Viola (common pansy),
Oxalis, and
Commelina produce two types of flowers:
| Statement I: |
Cleistogamous flowers which are similar to flowers of other species with exposed anthers and stigma |
| Statement II: |
Chasmogamous flowers which do not open at all. |
| 1. |
Statement I is correct; Statement II is correct |
| 2. |
Statement I is incorrect; Statement II is correct |
| 3. |
Statement I is correct; Statement II is incorrect |
| 4. |
Statement I is incorrect; Statement II is incorrect |
34. The science of changing the traits of plants in order to produce desired characteristics is known as:
| 1. | Plant breeding | 2. | Horticulture |
| 3. | Plantlet regeneration | 4. | Organogenesis |
35. Regarding Kranz anatomy, the bundle sheath cells:
| 1. |
are rich in an enzyme Ribulose bisphosphate carboxylase-oxygenase (RuBisCO) but lack PEPcase. |
| 2. |
are rich in enzymes Ribulose bisphosphate carboxylase-oxygenase (RuBisCO) and PEPcase. |
| 3. |
lack enzymes Ribulose bisphosphate carboxylase-oxygenase (RuBisCO) and PEPcase. |
| 4. |
lack enzyme Ribulose bisphosphate carboxylase-oxygenase (RuBisCO) but are rich in PEPcase. |
Botany - Section B
36. In seagrasses:
| 1. |
female flowers remain submerged in water and pollen grains are released inside the water |
| 2. |
pollen grains are released on the surface of the water, which are passively carried away by water currents; some of them eventually reach the female flower |
| 3. |
pollination is by wind |
| 4. |
pollination is by bats |
37. In a cross WwYy X WWyy [assuming independent assortment], what proportion of the progeny will be WWyy?
38. The basic structural unit of DNA packaging in eukaryotes [the basic repeating unit of eukaryotic chromatin] is called as:
| 1. |
Nucleosome |
2. |
Solenoid |
| 3. |
Euchromatin |
4. |
Heterochromatin |
39. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
Spraying sugarcane crops with gibberellins increase the yield by as much as 20 tonnes per acre. |
| Reason (R): |
Sugarcane stores carbohydrates such as sugar in their stems. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
40. The PGR ethylene does not:
| 1. |
promote senescence and abscission of plant organs |
| 2. |
affect fruit ripening |
| 3. |
stimulate closure of stomata in times of water stress |
| 4. |
help in initiating flowering in pineapple |
41. In Electron Transport System in mitochondria, UQH
2 passes electrons to:
| 1. |
FMN |
2. |
Cytochrome b |
| 3. |
Fe-S |
4. |
FAD |
42. Both Dinoflagellates and Euglenoids:
1. have stiff cellulosic cell wall
2. have two flagella
3. can behave as heterotrophs when deprived of sunlight
4. are mostly marine and photosynthetic
43. What percent of F
2 progeny of a typical Mendelian monohybrid cross are expected to be true-breeding?
44. The names for Complex III and Complex IV in the mitochondrial electron transport chain respectively are:
1. NADH dehydrogenase and Succinic dehydrogenase
2. NADH dehydrogenase and ATP synthase
3. Cytochrome bc1 and Cytochrome c oxidase
4. Cytochrome bc1 and Cytochrome b6f
45. Match the Scientists given in
Column-I with their contribution in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Jan Ingenhousz |
P. |
Provided evidence for production of glucose when plants grow |
| B. |
T. W Engelmann |
Q. |
Described the first action spectrum for photosynthesis |
| C. |
Julius von Sachs |
R. |
Showed that sunlight is essential for photosynthesis |
| D. |
Cornelius van Neil |
S. |
Inferred that oxygen evolved in photosynthesis comes from water and not from carbon dioxide |
Codes
|
A |
B |
C |
D |
| 1. |
R |
Q |
P |
S |
| 2. |
Q |
R |
S |
P |
| 3. |
P |
S |
R |
Q |
| 4. |
S |
P |
Q |
R |
46. In plant cell division, the formation of the new cell wall begins with the formation of a simple precursor called:
| 1. |
the cell plate that represents the middle lamella |
| 2. |
the cell plate that represents the primary cell wall |
| 3. |
the cleavage furrow that represents the middle lamella |
| 4. |
the contractile ring that represents the primary cell wall |
47. The members of Family Liliaceae have six:
| I: |
Tepals |
| II: |
Fused carpels in the ovary |
| III: |
Stamens |
1. Only
I and
II
2. Only
I and
III
3. Only
II and
III
4.
I,
II and
III
48. Genera like
Selaginella and
Salvinia:
| 1. |
do not possess vascular tissues |
| 2. |
have a dominant gametophyte in their life cycle |
| 3. |
produce two kinds of spores |
| 4. |
have macrophylls |
49. In the logistic growth of a population, the acceleration phase is followed by a/an:
| 1. |
Lag phase |
2. |
Deceleration phase |
| 3. |
Asymptote |
4. |
Sudden crash phase |
50. Who predicted that plants give off oxygen as a result of splitting of water molecules during photosynthesis, not of carbon dioxide molecules as thought before?
| 1. |
Preistley |
2. |
C. B. van Neil |
| 3. |
T. W. Engelmann |
4. |
Arnon and his co-workers |
Zoology - Section A
51. The first meiotic division in the primary oocyte:
1. occurs after the onset of puberty
2. is completed before the birth
3. begins in foetal life
4. occurs at the time of fertilization
52. The last of the germ layers to appear in the human embryonic development is the:
1. Ectoderm
2. Endoderm
3. Mesoderm
4. All the three germ layers appear simultaneously
53.
| Assertion (A): |
The female anopheles mosquito is not considered a parasite |
| Reason (R): |
The bite of the female anopheles mosquito injects infective stage of malarial parasite into human blood |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
54. Pure-breeding red-flowering snapdragons [
Antirrhinum sp.] are crossed with pure-breeding white-flowering snapdragons. The proportion of progeny with pink flowers from this cross is expected to be:
| 1. |
25 % |
2. |
50 % |
| 3. |
75 % |
4. |
100 % |
55. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
Hemichordates are not chordates. |
| Reason (R): |
They do not have a notochord. |
In the light of the above statements, choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
56. A diploid cell has a chromosome number of 8 and DNA content as 8 units. At the end of the S phase, the number of chromosomes and the DNA content respectively will be:
| 1. | 8 and 8 | 2. | 16 and 8 |
| 3. | 8 and 16 | 4. | 16 and 16 |
57. Most birds and mammals:
| 1. |
breed many times during their lifetime and produce a small number of large-sized offspring. |
| 2. |
breed only once during their lifetime and produce a small number of large-sized offspring. |
| 3. |
breed many times during their lifetime and produce a large number of small-sized offspring. |
| 4. |
breed only once during their lifetime and produce a large number of small-sized offspring. |
58. The fragments created by cutting DNA with restriction endonucleases can be separated by gel electrophoresis. What is incorrect regarding this procedure?
| 1. |
The fragments move towards anode. |
| 2. |
Agar gel is used for sieving effect. |
| 3. |
DNA fragments can be visualized moving in the medium if stained with ethidium bromide followed by exposure to UV radiation. |
| 4. |
The larger fragments move farther in the medium. |
59. Mood swings, increased aggressiveness and reduced fertility in males can be the result of long term abuse of:
1. Narcotic analgesics
2. Anabolic steroids
3. Diurectics
4. Cannabinoids
60. Darwin finches and Australian marsupials are excellent examples of:
1. Saltation
2. Catastrophism
3. Adaptive radiation
4. Anthropogenic artificial selection
61. Glucose transporter type 4 [GLUT-4]:
1. is widely distributed in fetal tissues.
2. is expressed at highest levels in erythrocytes
3. is expressed mostly in neurons
4. enables glucose transport into cells
62. Under normal physiological conditions:
| 1. |
Inspiratory reserve volume is less than expiratory reserve volume |
| 2. |
Residual volume in a healthy male is about 2500 to 3000 ml |
| 3. |
Functional residual capacity should be more than expiratory capacity |
| 4. |
Total lung capacity + residual volume = Vital capacity |
63. The stroke volume of left ventricle in a person is 75 ml. His cardiac output is 6000 ml. What will be his heart rate?
| 1. |
72 per minute |
2. |
75 per minute |
| 3. |
80 per minute |
4. |
100 per minute |
64. What would be true about the role of collecting duct in the concentration of urine by the kidneys?
| I: |
Allows passage of small amounts of urea into the medullary interstitium |
| II: |
Large amounts of water could be reabsorbed from this part to produce a concentrated urine. |
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
65. Transport of sodium and potassium ions across axonal membrane:
| 1. |
is active through sodium-potassium pump and passive through sodium and potassium channels |
| 2. |
is active through both sodium-potassium pump through sodium and potassium channels |
| 3. |
is passive through both sodium-potassium pump through sodium and potassium channels |
| 4. |
is passive through sodium-potassium pump and active through sodium and potassium channels |
66. Neurohypophysis:
1. secretes large number of tropins
2. synthesizes oxytocin and vasopressin
3. merges with pars distalis in humans
4. stores and releases oxytocin and vasopressin
67. Identify the incorrectly matched pair:
| 1. |
Ciliated epithelium |
Fallopian tubes |
| 2. |
Dense irregular connective tissue |
Skin |
| 3. |
Nissl’s granules |
Neuron |
| 4. |
Smooth muscle |
Intercalated disc |
68. All enzymes are:
| 1. |
Proteins |
| 2. |
Thermostable |
| 3. |
Capable of speeding up a biochemical reaction |
| 4. |
Capable of working in alkaline pH |
69. Which of the following is not a correctly matched pair?
| 1. |
Ear ossicles |
Total 6 in number |
| 2. |
Pubic symphysis |
Fibrous cartilage |
| 3. |
Gliding joint |
Between atlas and axis vertebrae |
| 4. |
Scapula |
Glenoid cavity |
70. Which structure enables the communication between the left and right cerebral hemispheres in the human brain?
1. Foramen of Monroe
2. Cerebral aqueduct of Sylvius
3. Corpus callosum
4. Cerebellar peduncles
71. A steroid hormone when released increases blood glucose level, causes lipolysis and proteolysis, retards cellular uptake and utilization of amino acids by body cells and has a potent anti-inflammatory effect. This hormone is:
1. Thyroxin
2. Adrenaline
3. Growth hormone
4. Cortisol
72. Regarding the male accessory sex ducts:
| 1. |
About 200 seminiferous tubules are present in testis lobule |
| 2. |
Epididymis is located along the anterior surface of each testis |
| 3. |
Vas deferens ascends to the abdomen and loops over the bladder |
| 4. |
Ducts of seminal vesicles open at the urethral meatus |
73. Regarding ‘Saheli’:
| I: |
It is a steroidal oral contraceptive pill developed by CDRI |
| II: |
It has very few side effects and high contraceptive value |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
74. Under normal physiological conditions, what percent of haemoglobin is saturated with oxygen when the partial pressure of oxygen is 40 mm Hg?
1. 50
2. 75
3. 90
4. 98
75. Biopiracy is:
| 1. |
the exploration of natural sources for small molecules, macromolecules and biochemical and genetic information that could be developed into commercially valuable products. |
| 2. |
exploitation of a region's biological resources or indigenous knowledge unethically and without providing fair compensation. |
| 3. |
the use of living organisms, like microbes and bacteria, in the removal of contaminants, pollutants, and toxins from soil, water, and other environments |
| 4. |
the process of developing a crop with bioavailable micronutrients in its edible parts. |
76. Identify hominids who were cave dwellers, used hides to protect their bodies and buried their dead:
1. Homo erectus
2. Homo sapiens
3. Homo neanderthalensis
4. Homo habilis
77. In a cardiac cycle, joint diastole state correctly means:
1. only the two atria are in a relaxed state
2. only the two ventricles are in a relaxed state
3. ventricles and atria are in a relaxed state
4. only the left atrium and the left ventricle are in a relaxed state
78. During the concentration of urine by human kidneys:
| I: |
Small amounts of urea is transported back to medullary interstitium by ascending portion of vasa recta |
| II: |
NaCl is returned to the medullary interstitium by the descending portion of vasa recta |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
79. In Eukaryotes, during transcription, the introns:
1. represent the coding sequence of eukaryotic split gene
2. are loosely defined as regulatory genes
3. do not appear in mature or processed RNA
4. provide site of attachment for RNA polymerase
80. Identify the incorrect statement:
| 1. |
Notochord is derived from embryonic mesoderm |
| 2. |
Stomochord in hemochordates is actually notochord |
| 3. |
Chordates have a post-anal tail |
| 4. |
Pharynx is perforated by gill slits in chordates |
81. In an animal cell, hydrolases active at acidic pH are richly found in:
| 1. |
Lysosomes |
2. |
Centrosome |
| 3. |
Peroxisomes |
4. |
Glyoxysomes |
82. Sticky ends produced by certain restriction endonucleases are so named because:
| 1. |
they form hydrogen bonds with their complementary cut counterparts |
| 2. |
they allow a host cell to retain a large number of copies of vector DNA |
| 3. |
they act as selectable markers to isolate transformed cells |
| 4. |
they form covalent phosphodiester bonds with their complementary cut counterparts |
83. A person with HIV infection starts suffering from infections, that could have been otherwise overcome, when the virus significantly destroys:
1. Macrophages
2. Helper T cells
3. Cytotoxic T cells
4. B cells
84. Which hormone produced by the intestinal mucosa causes the secretion of enzymes from the pancreas and the contraction of gall bladder?
| 1. |
cholecystokinin |
2. |
gastric inhibitory peptide |
| 3. |
secretin |
4. |
gastrin |
85. Blood doping is the use of synthetic EPO [erythropoietin] by some athletes to boost their:
1. blood calcium levels
2. secretion of growth hormone
3. blood oxygen levels
4. muscle mass
Zoology - Section B
86. Match the disorder given in Column-I with the feature most closely related to the disorder in Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Asthma |
P. |
Inflammation of bronchi and bronchioles |
| B. |
Emphysema |
Q. |
Damage to alveolar walls |
| C. |
Occupation lung disease |
R. |
Pulmonary fibrosis |
Codes
|
A |
B |
C |
| 1. |
P |
Q |
R |
| 2. |
Q |
P |
R |
| 3. |
Q |
R |
P |
| 4. |
R |
Q |
P |
87. Hypothalamus:
| 1. |
is a basal part of the telencephalon, forebrain. |
| 2. |
contains several groups of neurosecretory cells called nuclei which produce hormones |
| 3. |
is connected to the neurohypophysis by a portal circulation |
| 4. |
is the location of the respiratory rhythm center |
88. During puberty, the increased secretion of which of the following causes the male secondary sexual characters to be manifested?
1. GnRH from the hypothalamus
2. FSH from the anterior pituitary
3. LH from the anterior pituitary
4. Testosterone from the testes
89. The foetus is actually differentiated from:
| 1. |
the inner cell mass of the blastocyst |
| 2. |
the trophoblast cells of the blastocyst |
| 3. |
partly the trophoblast cells but mainly the inner cell mass of the blastocyst |
| 4. |
theca cells and granulosa cells of the antral follicle |
90. As their primary mechanism of action, combined oral contraceptive pills:
| 1. |
inhibit follicular development and prevent ovulation |
| 2. |
increase sperm penetration of cervix by making the cervical secretions thick |
| 3. |
make uterine endometrium hostile to implantation |
| 4. |
slow down tubal motility and ova transport interfering with fertilization |
91. Consider the two statements:
| I: |
Agar Gel electrophoresis is useful in analysis of DNA. |
| II: |
DNA is generally positively charged due to its nitrogenous bases so an electrical field forces it to move toward a negative pole. |
| 1. |
Both I and II are correct and II explains I |
| 2. |
Both I and II are correct but II does not explain I |
| 3. |
I is correct but II is incorrect |
| 4. |
Both I and II are incorrect |
92. What is true regarding the experiment carried out by S. L. Miller?
| 1. |
The flask contained methane, ammonia and oxygen |
| 2. |
The temperature was kept at 4°C |
| 3. |
Electric discharge was used as a source of energy |
| 4. |
He did not observe formation of any amino acids |
93. What feature on ECG represents the return of ventricles from excited to normal state?
| 1. |
P wave |
2. |
R wave |
| 3. |
S wave |
4. |
T wave |
94. Platyhelminthes, rotifers and lancelets:
1. are pseudocoelomates
2. have protonephridia as excretory structure
3. do not reproduce by sexual method
4. possess remarkable powers of true regeneration
95. Which of the following is not a feature of animals belonging to Phylum Annelida?
1. Metameric segmentation
2. An open circulatory system
3. Nephridia for osmoregulation and excretion
4. Bilateral symmetry
96. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
Dietary proteins are the source of essential amino acids. |
| Reason (R): |
All essential amino acids are aromatic. |
In the light of the above statements, choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
97. The relaxation of muscle after contraction is related to:
1. Hydrolysis of ATP by the myosin head
2. binding of calcium ions to Troponin C
3. Pumping of calcium ions back to the sarcoplasmic cisternae
4. Release of acetylcholine at the neuro-muscular junction
98. Match each item regarding
Periplaneta americana in
Column-I with one in
Column-II and select the correct match from the codes given:
|
Column-I
(Morphological feature) |
|
Column-I
(Location) |
| A. |
Sternites |
P. |
Ventral sclerites |
| B. |
Tegmina |
Q. |
Dorsal sclerites |
| C. |
Tergites |
R. |
Mesothorax |
| D. |
Hind wings |
S. |
Metathorax |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
S |
P |
R |
| 2. |
S |
Q |
R |
P |
| 3. |
P |
R |
Q |
S |
| 4. |
R |
P |
S |
Q |
99. One of the strands of a dsDNA molecule has 5 adenine and 5 cytosine bases. The number of hydrogen bonds in this dsDNA molecule is expected to be:
100. Identify the incorrectly matched pair:
| 1. |
CT |
Computed Tomography |
| 2. |
MRI |
Magnetic Radiation Imaging |
| 3. |
NACO |
National AIDS Control Organization |
| 4. |
MALT |
Mucosa Associated Lymphoid Tissue |
Chemistry - Section A
101. Given the cell reaction
\(Pb+Sn^{2+}~\longrightarrow~Pb^{2+}+Sn\) . Also, given that,
\(Pb~\longrightarrow~Pb^{2+},E^0=0.13~V\) and
\(Sn^{2+}+2e^-~\longrightarrow~Sn;E^0=-0.14~V\). What would be the ratio of log of concentration of cation of lead to the conc of cation of tin for which
\(E = 0?\)
1. 1/4
2. 1/2
3. 1/3
4. 1/1
102. Tetrahedral and diamagnetic compound among the following is:
| 1. |
[NiCl4]2- |
2. |
[Ni(CN)4]2- |
| 3. |
Ni(CO)4 |
4. |
[Ni(H2O)6]2+ |
103. The number of structural isomers of (with one double bond) and (with one triple bond) is:
1. \(\mathrm{C}_4 \mathrm{H}_8=3 ; \mathrm{C}_5 \mathrm{H}_8=3 \)
2. \(\mathrm{C}_4 \mathrm{H}_8=4 ; \mathrm{C}_5 \mathrm{H}_8=4 \)
3. \(\mathrm{C}_4 \mathrm{H}_8=3 ; \mathrm{C}_5 \mathrm{H}_8=4\)
4. \(\mathrm{C}_4 \mathrm{H}_8=4 ; \mathrm{C}_5 \mathrm{H}_8=3\)
104. The hybridised state of carbon, number of hexagons and number of pentagons in C60 (Fullerene), respectively, are:
1. sp2 , 12 & 20
2. sp3 , 20 & 12
3. sp3 , 12 & 20
4. sp2 , 20 & 12
105. Two students, X and Y, reported the mass of the same substance as 7.0 g and 7.00 g, respectively. Determine the correct statement from the following options:
1. Both are equally accurate.
2. X is more accurate than Y.
3. Y is more accurate than X.
4. Both are inaccurate scientifically.
106. The value of the rate constant depends on:
(a) Concentration
(b) Catalyst
(c) Temperature
1. (a) only
2. (b), (c) only
3. (a), (b) only
4. (c) only
107. The total number of geometrical isomers possible for an octahedral complex of the type [\( Ma_2b_2c_2\)] are:
[M = transition metal; a, b, and c are monodentate ligands]
1. 3
2. 4
3. 5
4. 6
108. Consider the complex, [NiCl4]2-, [Ni(NH3)6]2+, and [Ni(CN)4]2-.
These complexes absorb Red, Yellow, and Blue light respectively.
The colours that they emit, respectively, are:
1. Green, Violet, and orange
2. Blue, Red, and Yellow
3. Green, Orange, and Violet
4. Cannot be predicted
109. A carbonyl compound reacts with HCN followed by hydrolysis to give lactic acid. The carbonyl compound is:
| 1. |
HCHO |
2. |
CH3CHO |
| 3. |
CH3COCH3 |
4. |
CH3CH2CHO |
110. When Grignard reagent reacts with HCN or RCN followed by hydrolysis, carbonyl compounds are formed. The carbonyl compound which cannot be formed in the above reaction is:
1. HCHO
2. CH3CHO
3. CH3COCH3
4. All carbonyl compounds can formed.
111. Diazo group coupling reaction is an example of:
1. Nucleophilic substitution reaction
2. Electrophilic substitution reaction
3. Elimination reaction
4. Free radical substitution reaction
112. What is the Van't Hoff factor (i) for Al
2(SO
4)
3 if it dissociates to 40%?
| 1. |
2.0 |
2. |
2.6 |
| 3. |
3.6 |
4. |
5.0 |
113. Example(s) of a pseudo-unimolecular reaction among the following is/are:
| 1. |
\(\small{CH_3COOC_2H_5 + NaOH \rightarrow CH_3COONa + C_2H_5OH}\) |
| 2. |
\(\small{CH_3COOC_2H_5 + H_2O \xrightarrow{H^+} CH_3COOH + C_2H_5OH}\) |
| 3. |
\(C_{12}H_{22}O_{11}+ H_2O \xrightarrow{H^+} C_6H_{12}O_6+ C_6H_{12}O_6\\ (cane~ sugar)~~~~~~~~~~~~~~~~(glucose)~~~~~(Fructose)\) |
| 4. |
Both 2 and 3 |
114. Phosphinic acid and Phosphonic acid, respectively, are:
1. H3PO2 and H3PO3
2. H3PO3 and H3PO2
3. H3PO2 and H3PO4
4. H3PO3 and H3PO4
115. Eu2+ and Ce4+ behave, respectively, as:
1. Reducing agent and Reducing agent
2. Oxidising agent and Reducing agent
3. Reducing agent and oxidising agent
4. Oxidising agent and oxidising agent
116. The compounds A and B in the following reaction are:
(CH3)3C–O–CH3 + HI → A + B
1mole
1. (CH3)3C–OH + CH3I
2. (CH3)3C–I + CH3I
3. (CH3)3C–OH + CH3OH
4. (CH3)3C–I + CH3OH
117. Which of the following, has greatest number of atoms at NTP?
1. 1 ml N2
2. 1 ml CH4
3. 1 ml H2O
4. 1 ml NH3
118. For which of the following equations, \(\Delta H > \Delta E \)?
\(1.~~\mathrm{N}_{2}(g)+3 \mathrm{H}_{2}(\mathrm{~g}) \longrightarrow 2 \mathrm{NH}_{3}(\mathrm{g})\\ \\ 2.~~\mathrm{H}_{2}(g)+\frac{1}{2} \mathrm{O}_{2}(\mathrm{~g}) \longrightarrow \mathrm{H}_{2} \mathrm{O}(\mathrm{l})\\ \\ 3.~~\mathrm{PCl}_{5}(g) \rightarrow \mathrm{PCl}_{3}(g)+\mathrm{Cl}_{2}(g)\\ 4.~~\mathrm{H}_{2}(\mathrm{~g})+\mathrm{I}_{2}(\mathrm{~g}) \rightarrow 2 \mathrm{HI}(\mathrm{g}) \)
119. Equal volumes of pH=4 and pH =6 solutions are mixed together. The pH of the resulting solution will be:
1. 4.3
2. 4.7
3. 5.3
4. 5.7
120. Th solubility of AgCl is minimum in:
1. 0.01 M NaCl
2. 0.02 M CaCl2
3. 0.05 M AgNO3
4. 0.01 M NH4OH
121. Aqueous \(NaCl\) solution is electrolysed using platinum electrodes. The incorrect statement regarding this electrolysis is:
1. pH of solution increases
2. \(Cl_2\) gas is evolved at the anode
3. \(H_2\) gas is evolved at the cathode
4. pH of solution remains unchanged
122. The hybridised state of Xe and shape of \(XeOF_4\) , respectively, will be:
1. \(sp^3d\) and Square pyramidal
2. \(sp^3d^2\) and Octahedral
3. \(sp^3d^2 \) and Square pyramidal
4. \(sp^3d\) and Trigonal bipyramidal
123. 3 gm \(H_2\) reacts with 20 gm \(O_2\) to form \(H_2O\). The maximum mass of \(H_2O\) formed is:
1. 27 gm
2. 22.5 gm
3. 18 gm
4. 9.0 gm
124. If the value of m = +3 for an electron, then the electron must be present in:
| 1. |
s subshell |
2. |
p subshell |
| 3. |
d subshell |
4. |
f subshell |
125. When toluene is oxidised with chromyl chloride, then the product formed is:
| 1. |
Benzaldehyde |
2. |
Benzoic acid |
| 3. |
Cumene |
4. |
Benzene |
126. Which of the following represents the strongest bond?
| 1. |
\(sp^3\frac{\sigma}{}sp^3\) |
2. |
\(sp^2\frac{\sigma}{}sp^2\) |
| 3. |
\(sp\frac{\sigma}{}sp\) |
4. |
\(p\frac{\pi}{}p\) |
127. What is the oxidation number of phosphorus (
\(P\)) in the compound
\(\text{Ca}(\text{H}_2\text{PO}_2)_2\)?
128. Which of the following solvents favours \(S_N2\) mechanism?
1. Non polar solvent
2. Polar protic solvent
3. Polar aprotic solvent
4. All of these
129. Which of the following carbohydrates is laevorotatory in nature?
1. Glucose
2. Fructose
3. Mannose
4. Sucrose
130. Which one of the following elements has the highest 3rd ionization potential?
1. Na
2. Mg
3. Al
4. Si
131. Consider the following statements:
| I: |
The radius of an anion is larger than that of the parent atom. |
| II: |
The electronegativity of any given element is a constant, and it is a measurable quantity. |
| III: |
The electronegativity of an element is the tendency of an isolated atom to attract an electron. |
Which of the above statements is/are correct?
1. Only
I
2. Only
II
3. Only
I and
III
4. Only
II and
III
132. Among the following pairs of complexes, in which case is the ∆0 value higher for the first species:
1. \(\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right]^{3+} \text { and }\left[\mathrm{Co}(\mathrm{CN})_6\right]^{3-} \)
2. \(\left[\mathrm{CoF}_6\right]^{3-} \text { and }\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right]^{3+} \)
3. \(\left[\mathrm{Co}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+} \text { and }\left[\mathrm{Co}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{3+} \)
4. \(\left[\mathrm{Rh}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{3+} \text { and }\left[\mathrm{Co}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{3+} \)
133. A pair of carbocations that has a more stable second carbocation than the first is :
134. An amphoteric oxide among the following is:
| 1. |
V2O5 |
2. |
CrO3 |
| 3. |
Mn2O7 |
4. |
V2O3 |
135. The rate law for the dimerisation of \(NO_2\) is:
\(-\frac{\mathrm{d}\left[\mathrm{NO}_2\right]}{\mathrm{dt}}=\mathrm{k}\left[\mathrm{NO}_2\right]^2\)
The value of the specific rate constant, k can be changed by:
1. Doubling the total pressure on the system.
2. Doubling the temperature.
3. Both of (1) and (2)
4. None of the above.
Chemistry - Section B
136. Ammonia evolved due to complete conversion of nitrogen from 2.24 g sample of protein was absorbed in 50 mL of 0.5 M HNO3. The excess acid required 40mL of 0.2 M NaOH. The percentage of nitrogen in the sample is:
137. Lassaigne's test for nitrogen is not given by:
| 1. |
Nitrobenzene |
2. |
Hydroxyl Amine |
| 3. |
Aniline |
4. |
Pyridine |
138. Consider the following resonating structures of formic acid:
The correct order of the stability of the above resonating structures is:
| 1. |
I > II > III > IV |
2. |
III > I > II > IV |
| 3. |
I > III > II > IV |
4. |
I > II > IV > III |
139. If the half-life of a radioactive substance is 69.3 minutes, how long, approximately, will it take for it to decay to 20% of its initial quantity?
| 1. |
80.5 min |
2. |
161 min |
| 3. |
192 min |
4. |
235 min |
140. Compound 'A' in the following reaction is:
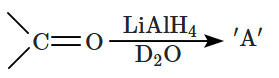
141. \(CH_3 -CH-CH_2-NH_2 + HNO_2 \rightarrow A (Major),\\ \\ ~~~~~~~~~~~~~|\\ ~~~~~~~~~~~~CH_3\)
The compound 'A' is
| 1. |
\(CH_3 - CH-CH_2 -OH \\ ~~~~~~~~~~~~~~|\\ ~~~~~~~~~~~~~CH_3\) |
| 2. |
(CH3)3C-OH |
| 3. |
\(CH_3 - CH-CH_2 -CH_3 \\ ~~~~~~~~~~~~~~|\\ ~~~~~~~~~~~~~OH\) |
| 4. |
\(CH_3 - CH-CH_2 -NO_2 \\ ~~~~~~~~~~~~~~|\\ ~~~~~~~~~~~~~CH_3\) |
142. In a nitrating mixture (HNO3 + H2SO4), H2SO4 can act as
1. Acid
2. Base
3. Neither acid nor base
4. Both acid as well as base
143. A mixture of ethanol and water shows a positive deviation from Raoult's law. This mixture also forms an azeotropic mixture. The boiling point of this azeotropic mixture will be
| 1. |
Less than the boiling point of water but more than the boiling point of ethanol |
| 2. |
More than the boiling point of water but less than the boiling point of ethanol |
| 3. |
More than the boiling point of ethanol as well as water |
| 4. |
Less than the boiling point of ethanol as well as water |
144. The ratio of number of atoms in 1 gm
\(NH_3\) and 1 gm
\(N_2\) is:
| 1. |
\(\dfrac {28}{ 17}\) |
2. |
\(\dfrac {28 }{ 34} \) |
| 3. |
\(\dfrac {56 }{ 17}\) |
4. |
\(\dfrac {17 }{ 28}\) |
145. Aqueous solution of which of the following salts is not a neutral solution?
| 1. |
\(NaCl\) |
2. |
\(CH_3COONH_4\) |
| 3. |
\(Na_2SO_4 \) |
4. |
None of the above |
146. Which of the following solutions has the minimum freezing point?
1. 0.02 M NaCl
2. 0.05 M Urea
3. 0.01 M \(MgCl_2\)
4. 0.01 M KCl
147. What happens to the specific conductivity of a solution upon dilution?
1. It decreases.
2. It increases.
3. It remains the same.
4. It cannot be predicted.
148. In the boiling of egg, entropy
1. decreases
2. increases
3. remains unchanged
4. cannot be predicted
149. Which one of the following statements is true about the structure of \(\mathrm{CO^{2-}_3}\) ion?
| 1. |
It can be explained by considering sp3 hybridization. |
| 2. |
Out of the three C–O bonds, two are longer and one is shorter. |
| 3. |
It has three sigma and three \(\pi\)-bonds. |
| 4. |
All three C–O bonds are equal in length with a bond order in between 1 and 2. |
150. Which of the following is not isoelectronic with H
2S?
| 1. |
F2 gas |
2. |
Oxide ion |
| 3. |
Ca+2 |
4. |
Sc+3 |
Physics - Section A
151. Water flows out of a conical funnel, with a small bore, and also out of a rectangular tank with a small bore: the respective speeds being \(v_1, v_2.\) The cross-sectional areas are the same at the top, while the water levels are also the same in both.

Then:
1. \(v_1=v_2\)
2. \(v_1=2v_2\)
3. \(\frac{v_1}{v_2}=\) extremely large
4. \(\frac{v_1}{v_2}=\) extremely small
152. A gas thermometer measures the temperature by measuring the pressure of a constant volume of gas (considered to be ideal). The pressure is directly proportional to the absolute temperature. The pressure at \(27^\circ\text{C}\) is found to be \(15\) kPa. When the pressure is \(20\) kPa, the temperature is:
1. \(20.25^\circ\text{C}\)
2. \(127^\circ\text{C}\)
3. \(225^\circ\text{C}\)
4. \(36^\circ\text{C}\)
153. Hydrogen gas is contained in a vessel and the RMS speed of the gas molecules is \(v\). The gas is heated isobarically so that its volume doubles, then it is compressed isothermally so that it returns to the same volume. The final RMS speed of the molecules will be:
| 1. |
2\(v\) |
2. |
\(v\)/2 |
| 3. |
\(v\)\(\sqrt2\) |
4. |
\(v\)/\(\sqrt2\) |
154. A block is connected to a spring and the system is suspended from the ceiling. The extension in the spring in equilibrium is \(x.\) If the system is allowed to oscillate vertically, then its time period is:

1. \(\sqrt{\dfrac{2x}{g}}\)
2. \(\sqrt{\dfrac{x}{2g}}\)
3. \(\sqrt{\dfrac{\pi^2x}{g}}\)
4. \(\sqrt{\dfrac{4\pi^2x}{g}}\)
155. The separation between a node and the next antinode in a vibrating air column is \(20\) cm. The speed of sound is \(320\) m/s. The frequency of sound is:
1. \(128\) Hz
2. \(256\) Hz
3. \(400\) Hz
4. \(800\) Hz
156. Two infinitely large plane parallel sheets carry uniform surface charge densities
\(+\sigma,~-\sigma\) and are placed a distance
\(d\) apart. The electric fields in the regions
①, ②, ③ are
\(E_1, E_2, E_3\) along the direction
\(x\) which is perpendicular to the two planes.

Then:
| 1. |
\(E_1 < 0, E_2 > 0, E_3 < 0\) |
| 2. |
\(E_1 < 0, E_2 = 0, E_3 < 0\) |
| 3. |
\(E_1 = 0, E_2 > 0, E_3 = 0\) |
| 4. |
\(E_1 < 0, E_2 > 0, E_3 > 0\) |
157. Consider a parallel plate capacitor of plate area '
\(A\)', plate separation '
\(d\)'. Suppose that the plates are given charges
\(+Q,-Q\) respectively. The force between the two plates is proportional to:
| 1. |
\(\dfrac{Q^2}{d^2}\) |
2. |
\(\dfrac{Q^2}{A}\) |
| 3. |
\(\dfrac{Q^2}{d\sqrt A}\) |
4. |
\(\dfrac{Q^2\sqrt A}{d^3}\) |
158. All the resistances shown in the network are equal to
\(1~\Omega.\) Find the resistance of the network measured between
\(A\) and
\(D.\)

| 1. |
\(1~\Omega\) |
2. |
\(2~\Omega\) |
| 3. |
\(\dfrac{1}{2}~\Omega\) |
4. |
\(\dfrac{1}{4}~\Omega\) |
159. All the resistances shown in the network are equal to
\(1~\Omega\), while the EMFs of the cells are
\(2~\text{V}\). The current flowing through the branch
\(AC\) is:
1.
\(1~\text{A}\)
2.
\(2~\text{A}\)
3.
\(4~\text{A}\)
4.
\(6~\text{A}\)
160. An
\(\alpha\)-particle and a proton of the same kinetic energy move along circular paths of radii
\(r_{\alpha}\) and
\(r_p\) respectively, in the same magnetic field. The ratio
\((r_{\alpha} / r_p) \) equals:
| 1. |
\(2\) |
2. |
\( \dfrac{1} {2}\) |
| 3. |
\(1\) |
4. |
\(4\) |
161. The mutual inductance between the two circuits can be determined by simply letting a current
\(i\) flow through one circuit and finding the flux of the magnetic field through the second circuit:
\(\phi_{2}=M_{12} i_{1}\), where
\(M_{12}\) is the mutual inductance. Using this method, or otherwise determine the mutual inductance
\((M)\) between a long straight wire, and a small coplanar loop of the area
\(A\), located at a distance
\(l\) from the wire. The value of
\(M\) is:
| 1. |
\( \dfrac{\mu_{0} l}{2 \pi}\) |
2. |
\(\dfrac{\mu_{0} A}{2 \pi l}\) |
| 3. |
\(\dfrac{\mu_{0} l^{3}}{4 \pi A}\) |
4. |
\(\dfrac{\mu_{0} A^{2}}{2 \pi l^{3}}\) |
162. In the system shown in the figure, the horizontal rod falls vertically down under its own weight while retaining electrical contact with parallel rails. There is no resistance in the circuit, and there is a uniform horizontal magnetic field into the plane. The acceleration of the rod
\(PQ\), as it falls down is '
\(a\)'.

Then:
| 1. |
\(a=g\) |
| 2. |
\(a>g\) |
| 3. |
\(a<g\) |
| 4. |
\(a\) is initially less than \(g\), but later it is greater than \(g\). |
163. An AC source of emf
\(100~\text{V}\) and frequency
\(f= \dfrac{50}{\pi}\) Hz has an internal resistance
\(100~\Omega\). A load resistance
\(R_L\) is connected across the source. Maximum power is dissipated in the load, when:
| 1. |
\(R_L = 100\sqrt 2~\Omega\) |
2. |
\(R_L = \dfrac{100}{\sqrt 2}~\Omega \) |
| 3. |
\(R_L = 100~\Omega\) |
4. |
\(R_L = 200~\Omega\) |
164. A ray of light passes through three parallel media, with the angles of incidence and emergence as shown in the figure. The refractive indices of the media are
\(\mu_{1}\) = 1,
\(\mu_{2}\) and
\(\mu_{3}\).

Then:
| 1. |
\(\mu_{2}=\dfrac{1}{3},~\mu_{3}=\dfrac{1}{2}\) |
2. |
\(\mu_{2}=3,~\mu_{3}=\dfrac{3}{2}\) |
| 3. |
\(\mu_{2}=\dfrac{1}{3},~\mu_{3}=\dfrac{2}{3}\) |
4. |
\(\mu_{2}=3,~\mu_{3}=2\) |
165. The electric field associated with a light beam incident on a metallic surface with work-function
\(3.0\) eV, is given by;
\(E = E_o~\sin (\pi \times 10^7 \text{m}^{-1}) (x-ct)\). The stopping potential for the emitted photoelectrons is: (take
\(hc=1240\) eV-nm, if required)
| 1. |
\(0.1\) |
2. |
\(3.2\) |
| 3. |
\(9.4\) |
4. |
none of the above |
166. Which of the following gates, may be used to form all other gates?
| 1. |
AND |
2. |
OR |
| 3. |
NOT |
4. |
NAND |
167. A thin prism of refractive index \(\mu = 1.5\) introduces a deviation of \(+1^\circ\) into the path of an incident beam. When this prism is immersed in a liquid of refractive index \(\mu = 2\), the deviation introduced will be:
1. \( \left ( \dfrac{7}{6} \right )^\circ\)
2. \( \left ( \dfrac{3}{2} \right )^\circ\)
3. \(- \left ( \dfrac{1}{2} \right )^\circ\)
4. \( \left ( \dfrac{1}{2} \right )^\circ\)
168. The width of the central maximum of the diffraction pattern of a single slit of width
\(1~\text{mm}\) equals the width of the slit itself, when the screen is
\(1~\text m\) away from it. The wavelength of light used equals:
| 1. |
\(250~\text{nm}\) |
2. |
\(500~\text{nm}\) |
| 3. |
\(1000~\text{nm}\) |
4. |
\(2000~\text{nm}\) |
169. Given below are two statements:
| Assertion (A): |
When light consisting of wavelengths corresponding to the Balmer series is incident on a gas containing \(\mathrm{He}^{+}\) ions in the first three excited states - it can be absorbed by the \(\mathrm{He}^{+}\) ions. |
| Reason (R): |
All the energy levels of the \(\mathrm{He}^{+}\) ions are the same as those of the \(\mathrm{H}\) atoms. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
170. The system is at rest initially, due to the force of friction acting on
\(A\). If the string connecting the
lower blocks is cut, the accelerations of the blocks
\(A\),
\(B\) &
\(C\) will be, respectively,

| 1. |
\(\dfrac g 3\) to left, \(g\) upward, \(g\) downward. |
| 2. |
zero, zero, \(g\) downward. |
| 3. |
zero, \(g\) upward, \(g\) downward. |
| 4. |
\(g\) to right, zero, \(g\) downward. |
171. A uniform rod
\(AB\) of mass m and length
\(L\) is replaced by three particles – two particles of masses
\(m_1\) each at the ends and another particle of mass
\(m_2\) at its centre. The new system of particles has the same total mass, the same center-of-mass and the same moment of inertia about an axis through its C.M. and perpendicular to
\(AB.\)
Which of the following is true?
| 1. |
\(m_1=\frac{m}{3},~m_2=\frac{m}{3}~~~~~~~\) |
| 2. |
\(m_1=\frac{m}{4},~m_2=\frac{m}{2}\) |
| 3. |
\(m_1=\frac{m}{6},~m_2=\frac{2m}{3}\) |
| 4. |
\(m_1=\frac{m}{5},~m_2=\frac{3m}{5}\) |
172. Point charges
\(q\), each, are placed at the eight vertices of a cube of edge
\(l\). The flux of the net electric field through a face of the cube is:
| 1. |
\(\dfrac{4q}{3\varepsilon_0}\) |
2. |
\(\dfrac{2q}{3\varepsilon_0}\) |
| 3. |
\(\dfrac{q}{3\varepsilon_0}\) |
4. |
\(\dfrac{q}{6\varepsilon_0}\) |
173. Whenever a photon is emitted by a hydrogen atom in the Paschen series, it is followed by further emissions of photons, in the Balmer series or the Lyman series.
These photons can have:
| 1. |
2 possible energy values. |
| 2. |
3 possible energy values. |
| 3. |
4 possible energy values. |
| 4. |
5 possible energy values. |
174. A \(\mathrm{p\text-n}\) junction has an electric field of \(6\times 10^{5}~\text{V/m}\) in the junction and the junction width is \(500~\text{nm}.\) The height of the potential barrier is:
1. \(0.6~\text V\)
2. \(0.3~\text V\)
3. \(0.5~\text V\)
4. \(0.25~\text V\)
175. What is the unit of the quantity represented by: \(\dfrac{\text{(Angular momentum)}}{\text{(electric charge)}^2}\)
1. \(\Omega\) (ohm)
2. s/m
3. \(\text{H }\)(henry)
4. \(\text{F }\) (farad)
176. 
Consider the two situations shown in the figures. In figure (A), the particle
\(P\) is given a velocity
\(u\) up a smooth horizontal incline and it reaches a maximum vertical height
\(h_A\). In figure (B), the same particle
\(P\) is projected with a velocity
\(u\) at an angle
\(\theta\) (parallel to the previous incline) and reaches a maximum height
\(h_B\).
Then,
| 1. |
\(h_A=h_B~\text{sin}\theta\) |
2. |
\(h_A~\text{sin}\theta=h_B\) |
| 3. |
\(h_A~\text{sin}^2\theta=h_B\) |
4. |
\(\dfrac{h_A}{\text{sin}^2\theta}=h_B\) |
177. A particle undergoes an elastic collision with an identical particle at rest. After the collision, the two particles move with velocities \(\vec{v_1},~\vec{v_2}.\) Final velocities \(\vec{v_1},~\vec{v_2}\) are in different directions. The angle between \(\vec{v_1}\) and \(\vec{v_2}\) is:
1. \(\dfrac{\pi}{6}\)
2. \(\dfrac{\pi}{3}\)
3. \(\dfrac{\pi}{2}\)
4. \(\dfrac{2\pi}{3}\)
178. A thin uniform hemispherical shell
(radius: \(R,\) center: \(O\)) is cut into two symmetric quarter spheres by means of a vertical plane, as shown. The centre-of-mass of a quarter sphere is at a distance
\(d\) from
\(O\). Then
\(d\)=

| 1. |
\(\dfrac R2\) |
2. |
\(\dfrac R{\sqrt2}\) |
| 3. |
\(\dfrac R{4}\) |
4. |
\(\dfrac R{2\sqrt2}\) |
179. The angular momentum of a planet of mass
\(m,\) moving around the sun
(mass: \(M\gg m\)) in an orbit of radius
\(r\) is proportional to:
| 1. |
\(mr\) |
2. |
\(\dfrac{m}{r}\) |
| 3. |
\(m\sqrt r\) |
4. |
\(\dfrac{m}{\sqrt r}\) |
180. The internal energy of a gas is given by
\(U=\dfrac32PV.\) The gas expands in such a way that its internal energy
(initially \(U_0\)) remains constant throughout the process, but its
volume changes from
\(V_0\) to
\(2V_0.\) The heat supplied to the gas equals:
| 1. |
\(U_0\mathrm{ln}(2)\) |
2. |
\(\dfrac12U_0~\mathrm{ln}(2)\) |
| 3. |
\(\dfrac13U_0~\mathrm{ln}(2)\) |
4. |
\(\dfrac23U_0~\mathrm{ln}(2)\) |
181. A plane electromagnetic wavefront is incident at an angle of
\(30^{\circ}\) onto a flat surface. The difference between the arrival times of the wave at the points
\(A\) and
\(B\) is
\(T,\) where
\(AB=L.\) Then, the speed of the wavefront in the medium is:

| 1. |
\(\dfrac{L}{T}\) |
2. |
\(\dfrac{2L}{T}\) |
| 3. |
\(\dfrac{L}{2T}\) |
4. |
\(\dfrac{\sqrt3L}{T}\) |
182. A mango falling from a
height \(H\) from a tree is observed from a car moving with a uniform
velocity \(u,\) along a road, that passes underneath. The mango hits the car. Assume the size of the car to be negligible, and the acceleration due to gravity to be
\(g.\)

The relative speed of the mango when it hits the car is:
| 1. |
\(u+\sqrt{2gH}\) |
2. |
\(u-\sqrt{2gH}\) |
| 3. |
\(\sqrt{u^2+{2gH}}\) |
4. |
\(\sqrt{u^2-{2gH}}\) |
183. Antineutrinos are emitted during:
1. \(\alpha\)-decay
2. \(\beta\)-decay
3. \(\gamma\)-decay
4. neutron emission
184. Given below are two statements:
| Assertion (A): |
The density of the nucleus is much higher than that of ordinary matter. |
| Reason (R): |
Most of the mass of the atom is concentrated in the nucleus while the size of this nucleus is almost \(10^5\) times smaller. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
185. Two identical magnets are placed along the
\(x\text-\)axis, equidistant from the origin and opposing each other. If a third magnet be placed at the origin along the
\(y\text-\)axis, with its north-pole towards the positive
\(y\text-\)axis, it will experience:

| 1. |
force along +ve \(y\text-\)axis |
| 2. |
force along -ve \(y\text-\)axis |
| 3. |
zero force but a clockwise torque |
| 4. |
zero force but an anticlockwise torque |
Physics - Section B
186. A boat is rowed across a river so that its velocity with respect to water is directed perpendicular to the river's flow. The speed of the boat with respect to the water is \(2~\text{km/h} \) and the speed of the river's flow is \(3~\text{km/h}. \) If the river is \(1~\text{km}\) wide, how far down the river will the passengers have to disembark, with respect to their starting point?
1. \(\dfrac{2}{3}~\text{km}\)
2. \(1~\text{km}\)
3. \(\dfrac{3}{2}~\text{km}\)
4. None of the above is correct.
187. In a reversible process, the change in internal energy \(U\) of an ideal gas \((C_P/C_V=\gamma)\) is zero, while the volume increases from \(V\) to \(2V\). If the initial pressure is \(P\), the final pressure will be:
| 1. |
\(2P\) |
2. |
\(\dfrac P2\) |
| 3. |
\(P\) |
4. |
\(\dfrac{P}{2^\gamma}\) |
188. The equation of motion of a particle that starts moving at \(t=0 \) s is given by; \({x}=5 \sin \left(\dfrac{\pi t}{2}+\dfrac{\pi}{3}\right) \) where \(x\) is in cm and time \(t\) is in second. The time, when the particle first comes to rest, is:
1. \(\dfrac{1}{3}\text{ s}\)
2. \(\dfrac{7}{6}\text{ s}\)
3. \(\dfrac{2}{3}\text{ s}\)
4. \(\dfrac{13}{6}\text{ s}\)
189. The intensity \((I)\) of a sound wave depends on the distance \((x)\) from the source as:
| 1. |
\(I\propto x\) |
2. |
\(I\propto \dfrac1x\) |
| 3. |
\(I\propto \dfrac1{x^2}\) |
4. |
none of the above |
190. A dielectric slab is inserted between the plates of an isolated charged capacitor. Which of the following remains unchanged?
| (I) |
The charge on the plates. |
| (II) |
The potential difference between the plates. |
| (III) |
The energy stored in the capacitor. |
Choose the correct option from given ones:
| 1. |
(I) only |
2. |
(I) and (II) |
| 3. |
(I) and (III) |
4. |
(I), (II) and (III) |
191. Given below are two statements:
Several resistances
\(R_1, R_2, .........R_n\) are connected in parallel. The equivalent resistance of the combination is
\(R\).
| Assertion (A): |
The fractional error in \(R\) is most affected by that of the smallest resistance in the combination, other things being equal. |
| Reason (R): |
In parallel, the conductances add. The contribution to the overall error in the conductance is largest for the largest conductance or the smallest resistance. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
192. A lens is placed as shown in the figure, with the
\(x\)-axis along the principal axis and the origin at the optical centre. A ray of light incident on the lens at
\(P~(0, y)\) undergoes a deviation
\(\delta.\) A graph of
\(\delta\) vs
\(y\) is shown:


The focal length of the lens is nearly:
1.
\(2~\text{cm}\)
2.
\(20~\text{cm}\)
3.
\(115~\text{cm}\)
4.
\(230~\text{cm}\)
193. A fraction
\(f\) of the incident energy in a beam of light of wavelength
\(\lambda\) is absorbed by a metallic surface and causes photoemission. If the power of the beam falling on the surface is
\(P\), then the maximum photocurrent is:
(
\(e\) is electronic charge,
\(h\) is Planck's constant,
\(c\) is the velocity of light in vacuum)
| 1. |
\(\dfrac{\lambda{P}}{h c} f\) |
2. |
\(\dfrac{2\lambda{P}}{h c} f\) |
| 3. |
\(\dfrac{\lambda{P}}{h c} f e\) |
4. |
\(\dfrac{2\lambda{P}}{h c} f e\) |
194. A
\(10:1\) step-down transformer has an ideal diode and a
\(10\) \(\Omega\) resistance connected to its secondary circuit while
\(220~\text{V}\) AC mains is applied to the primary.
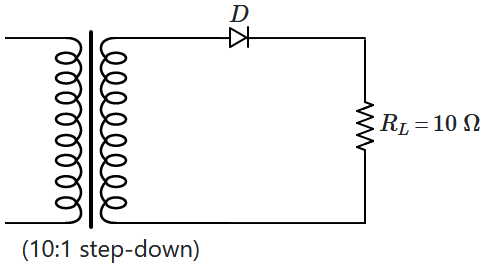
Assuming the transformer to be ideal, the current in the primary circuit when the diode is reverse-biased is:
| 1. |
\(0.22~\text{A}\) |
2. |
\({0.22 \over \sqrt 2}~\text{A}\) |
| 3. |
\(0.22\sqrt 2~\text{A}\) |
4. |
zero |
195. A ball is released with a velocity
\((2 \hat{\imath}+2 \hat{\jmath})~ \mathrm{m} / \mathrm{s}\) on the rectangular pool table from the point
\((3, 0)\text{ m.}\) All the collisions of the ball are elastic.
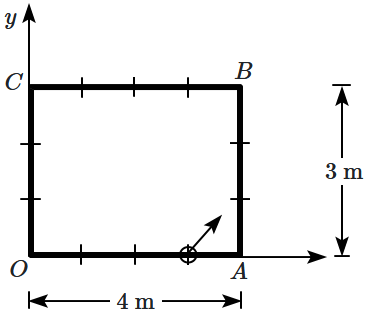
After
\(4\) seconds of being released, the location of the ball will be:
1.
\((2, 2)\) m
2.
\((0, 1)\) m
3.
\((2, 1)\) m
4.
\((3, 2)\) m
196. As water flows out slowly from a tap, under gravity, it is observed that the cross-section of the stream decreases as it falls down (figure). This effect is due to:
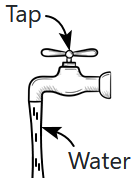
| 1. |
viscosity of water |
| 2. |
the surface tension of water |
| 3. |
incompressibility and Bernoulli's principle |
| 4. |
increase in atmospheric pressure at lower levels |
197. When the temperature difference between a body and its surroundings is \(20^{\circ}\text{C}\), it loses heat to the surroundings at a rate of \(40\) W. If the temperature difference increases to \(25^{\circ}\text{C}\), the rate of loss of heat is:
1. \(45\) W
2. \(50\) W
3. \(60\) W
4. \(80\) W
198. A wire carrying a current
\(i\) is bent into the form of an arc of a circle with center
\(O\), joined smoothly to two long, straight wires at its ends. The magnetic field at the centre
\(O\) is twice that due to the straight portions. The angle subtended at the centre
\(O\) by the arc is:

| 1. |
\(\theta=\dfrac{\pi}{3}\) |
2. |
\(\theta=\dfrac{\pi}{2}\) |
| 3. |
\(\theta=1\) rad |
4. |
\(\theta=2\) rad |
199. A block of mass
\(m\) is placed between two springs connected to the ends of a railroad car. The surface supporting the block is horizontal, and the spring are initially relaxed. The car is given an acceleration
\(a\) and the mass
\(m\) finally comes to equilibrium within the car. Let
\(x\) be the compression (or extension) in the two springs. Assume friction to be negligible. Then:

| 1. |
\(k_1x-k_2x=ma \) |
| 2. |
\(\dfrac{k_1k_2}{k_1+k_2}x=ma \) |
| 3. |
\(k_1x+k_2x=ma \) |
| 4. |
\(\dfrac{k_1k_2}{k_1-k_2}=ma \) |
200. A liquid of density
\(\rho\) flows through a bent tube of cross-section
\(A,\) with a speed
\(v.\) The liquid enters at point
\(A\) and exits at
\(B\) in the opposite direction. The radius of the bend is
\(R.\) The tube lies on a horizontal table. The force required to hold the tube equals:

1.
\(\rho Av^2\)
2.
\(2\rho Av^2\)
3.
\(\sqrt2\rho Av^2\)
4.
\(\rho v^2\pi R^2\)
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course

.svg)
.svg)
.svg)
.svg)




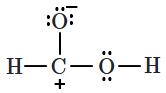
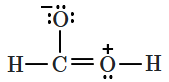
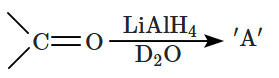









 Consider the two situations shown in the figures. In figure (A), the particle \(P\) is given a velocity \(u\) up a smooth horizontal incline and it reaches a maximum vertical height \(h_A\). In figure (B), the same particle \(P\) is projected with a velocity \(u\) at an angle \(\theta\) (parallel to the previous incline) and reaches a maximum height \(h_B\).
Consider the two situations shown in the figures. In figure (A), the particle \(P\) is given a velocity \(u\) up a smooth horizontal incline and it reaches a maximum vertical height \(h_A\). In figure (B), the same particle \(P\) is projected with a velocity \(u\) at an angle \(\theta\) (parallel to the previous incline) and reaches a maximum height \(h_B\).





 Assuming the transformer to be ideal, the current in the primary circuit when the diode is reverse-biased is:
Assuming the transformer to be ideal, the current in the primary circuit when the diode is reverse-biased is:




