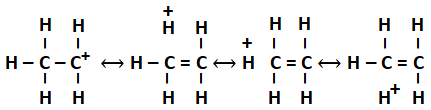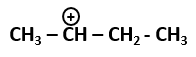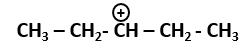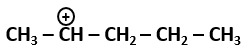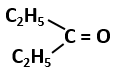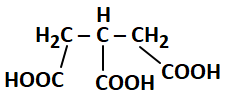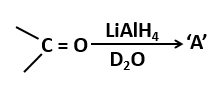Botany - Section A
1. Identify the incorrect statement:
| 1. |
LAB check disease causing microbes in our stomach. |
| 2. |
Dough for making foods like dosa and idli are fermented by yeast. |
| 3. |
Swiss cheese is fermented by Propionibacterium shermanii |
| 4. |
Roquefort cheese is an unripened cheese |
2. In the given diagram showing the TS of wall of an anther, identify the layer where cells can be binucleate:

3. What is incorrect regarding the tRNA molecule?
1. There are no tRNAs for stop codons
2. It is the smallest of the major cellular RNAs
3. The actual structure of tRNA looks like a clover leaf
4. Its 3’ end is the amino acid acceptor end
4. Identify the correct chronological sequence in the stages of embryonic development in a dicot:

| 1. |
A → B → C |
2. |
C → A → B |
| 3. |
B → A → C |
4. |
C → B → A |
5.
| Assertion (A): |
In order to link the alien DNA, the vector needs to have very few, preferably single, recognition sites for the commonly used restriction enzymes. |
| Reason (R): |
Presence of more than one recognition sites within the vector will generate several fragments, which will complicate the gene cloning. |
| 1. |
Both (A) and (R) are True and the (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but the (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
6. In Australian acacia:
| 1. |
The leaves are converted into spine for defence |
| 2. |
The leaves are converted into tendrils for climbing |
| 3. |
The petiole expands, becomes green, and photosynthesizes food |
| 4. |
The leaves become fleshy and store food |
7. Among the two nucleic acids, the DNA is a better genetic material because:
| 1. |
It is able to generate its replica (Replication). |
| 2. |
It is more stable chemically and structurally. |
| 3. |
It provides the scope for rapid changes (mutation). |
| 4. |
It is able to express itself in the form of 'Mendelian Characters’. |
8. If we say that the genetic code is ‘degenerate’, it essentially means:
1. Some amino acids are coded by more than one codon.
2. The codon is read in mRNA in a contiguous fashion.
3. The code is nearly universal.
4. AUG has dual functions.
9. Intangible benefits like aesthetic pleasures of walking through thick woods is a strong argument for the need to conserve biodiversity. This type of argument is called as:
1. Narrowly utilitarian
2. Ethical
3. Broadly utilitarian
4. Sustainable
10. Identify the incorrectly matched pair:
| 1. |
Linus Pauling |
Discovered the triple helix structure of collagen |
| 2. |
Rudolf Virchow |
Omnis cellula-e-cellula |
| 3. |
Singer and Nicolson |
Fluid mosaic model |
| 4. |
Georges Palade |
First observed ribosome |
11. Identify the incorrect statement:
1. Viruses are obligate parasites
2. Prions are abnormally folded infectious proteins
3. Viroids are free RNA and lack a protein coat
4. Lichens grow very well in polluted areas
12. Identify the incorrectly matched pair:
| 1. |
Pneumatophores |
Rhizophora |
| 2. |
Leaf tendrils |
Gourds |
| 3. |
Stilt roots |
Maize |
| 4. |
Palmately compound leaves |
Silk cotton |
13. All the following statements regarding collenchyma in plants is true except:
| 1. |
Occurs in layers below the epidermis in most of the dicotyledonous plants |
| 2. |
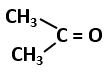
Cells are thickened at the corners due to deposition of cellulose, hemicelluloses and pectin |
| 3. |
There is considerable amount of intercellular space between cells |
| 4. |
Provide mechanical support to the growing parts of the plants |
14. Which of the following is a dominant trait in Pisum sativum?
| 1. |
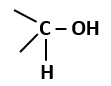
Yellow pod colour |
2. |
Green seed colour |
| 3. |
Axial flower position |
4. |
Constricted pod shape |
15. Regarding energy flow in an ecosystem:
| 1. |
About 2 to 10 % of incident solar radiation is PAR |
| 2. |
An ecosystem is exempt from the Second Law of Thermodynamics |
| 3. |
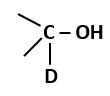
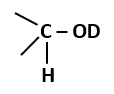
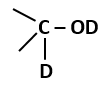
The amount of energy increases at successive trophic levels |
| 4. |
The number of trophic levels in a grazing food chain is limited |
16. Sacred groves of Khasi and Jaintia hills are located in:
| 1. |
Assam |
2. |
Meghalaya |
| 3. |
Manipur |
4. |
Arunachal Pradesh |
17. Morels and truffles:
| 1. |
are pathogenic fungi causing rust and smut |
| 2. |
do not reproduce sexually |
| 3. |
do not produce asexual spores |
| 4. |
are delicacies belonging to ascomycetes |
18. In palmately compound leaves:
| 1. |
Leaf lamina is entire |
| 2. |
Leaf lamina is incised but the incisions do not touch the mid rib |
| 3. |
Leaf lamina is incised, lamina reach up to the midrib and leaflets are present on a common axis |
| 4. |
Leaf lamina is incised, lamina reach up to the midrib and leaflets are attached at a common point |
19. Ethylene, a PGR, is not involved in:
| 1. |
fruit ripening |
| 2. |
breaking seed and bud dormancy |
| 3. |
closure of stomata in water stress |
| 4. |
respiratory climactic |
20. In the Citric acid cycle the reduction of NAD+ to NADH + H+ takes place at all the following steps except:
1. Conversion of Citric acid to α-ketoglutaric acid
2. Conversion of α-ketoglutaric acid to Succinyl CoA
3. Conversion of Malic acid to Oxaloacetic acid
4. Conversion of Succinic acid to Fumaric acid
21. Identify the incorrect statement regarding photosynthesis in higher plants:
| 1. |
Chlorophyll a is the chief pigment associated with photosynthesis. |
| 2. |
Splitting of water is associated with PS II |
| 3. |
Cyclic flow of electrons results only in the synthesis of NADH |
| 4. |
Biosynthetic phase is dependent on the products of the light reaction |
22. In a sample of water polluted with sewage discharge, if you measure BOD [Biochemical Oxygen Demand] and DO [Dissolved Oxygen], you expect to find that:
| 1. |
The levels of both BOD and DO are high |
| 2. |
The levels of both BOD and DO are low |
| 3. |
The levels of BOD will be high and the levels of DO will be low |
| 4. |
The levels of BOD will be low and the levels of DO will be high |
23. In an ecosystem, during decomposition, water soluble inorganic nutrients go down into the soil horizon and get precipitated as unavailable salts by the process of:
| 1. |
Fragmentation |
2. |
Leaching |
| 3. |
Catabolism |
4. |
Humification |
24. One of the crosses conducted by Mendel was a ‘test cross’. This cross can be used to:
| 1. |
determine whether the individual is heterozygous or homozygous dominant |
| 2. |
determine whether a gene is located on a sex chromosome or an autosome |
| 3. |
differentiate between incomplete dominance and codominance |
| 4. |
identify linkage between two genes |
25. Regarding pollination:
| 1. |
Xenogamy is the only type which brings genetically different types of pollen grains to the stigma |
| 2. |
Majority of plants use abiotic agents for pollination |
| 3. |
All aquatic plants are pollinated by water |
| 4. |
Geitonogamy is genetically similar to cross pollination |
26. Regarding pteridophytes:
| I: |
They are the first terrestrial plants to possess true vascular tissue |
| II: |
Main plant is sporophyte |
| III: |
Water is required for transfer of antherozoids |
1. Only
II is incorrect
2. Only
I is correct
3.
I and
II are incorrect
4.
I,
II and
III are correct
27. Gymnosperms lack:
| 1. |
Secondary growth |
2. |
Vessels in their xylem |
| 3. |
Albuminous cells |
4. |
A dominant sporophyte |
28. In non-cyclic photo-phosphorylation:
| 1. |
only PS I is functional |
| 2. |
both photosystems work in series, first PS I and then PS II |
| 3. |
only ATP synthesis takes place and NADPH is not synthesized |
| 4. |
both ATP and NADPH are synthesized |
29. How many of the given statements are correct?
I: Vallisneria and
Hydrilla grow in fresh water
II: Zostera is a marine sea-grass
III: All aquatic plants use water for pollination.
IV: Water lily is pollinated by insect.
30. Self-incompatibility:
| I: |
prevents inbreeding |
| II: |
is a genetic mechanism |
| III: |
prevents self-pollen from fertilising the ovules by inhibiting pollen germination or pollen tube growth in the pistil. |
| 1. |
Only I and II are correct |
2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
4. |
I, II and III are correct |
31. Consider the given two statements:
| Assertion (A): |
Although Mendel published his work on inheritance of characters in 1865 but it remained unrecognised till 1900. |
| Reason (R): |
In 1900, three Scientists, Avery, Macleod, McCarty, independently rediscovered Mendel’s results on the inheritance of characters. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
32. The given floral formula represents plants belonging to the family:

| 1. |
Gramineae |
2. |
Malvaceae |
| 3. |
Cruciferae |
4. |
Leguminosae |
33. All the following are ex-situ methods for conservation of biodiversity except:
1. Botanical Gardens
2. Seed banks
3. Plant tissue culture
4. Biosphere Reserves
34. Which of the following is the home for greatest biodiversity on Earth?
| 1. |
The Amazon Forest |
2. |
The Western Ghats |
| 3. |
The African grasslands |
4. |
The Australian Continent |
35. Maximum global diversity amongst the following is exhibited by:
| 1. |
Mollusks |
2. |
Echinoderms |
| 3. |
Annelids |
4. |
Crustaceans |
Botany - Section B
36. The need for pollinators is not obligatory in:
1. chasmogamous flowers
2. cleistogamous flowers
3. geitonogamy
4. xenogamy
37. Identify the option that includes all dominant traits seen in Pisum sativum?
| 1. |
Tall stem height, Violet flower, Green pod colour, Axial flower position, Inflated pod shape, Round seed shape and Yellow seed colour |
| 2. |
Tall stem height, Violet flower, Yellow pod colour, Axial flower position, Inflated pod shape, Round seed shape and Green seed colour |
| 3. |
Dwarf stem height, Violet flower, Green pod colour, Axial flower position, Inflated pod shape, Round seed shape and Yellow seed colour |
| 4. |
Tall stem height, Violet flower, Green pod colour, Terminal flower position, Inflated pod shape, Round seed shape and Yellow seed colour |
38. In Hershey and Chase experiment:
| I: |
Radioactive sulfur-35 was used to label the DNA contained in the T2 phage |
| II: |
Radioactive phosphorus-32 was used to label the protein sections of the T2 phage |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
39. 2, 4 – D, widely used to kill dicotyledonous weeds, is a derivative of:
1. Auxin
2. Cytokinin
3. Gibberellin
4. Abscisic acid
40. For each molecule of glucose, the number of molecules of Acetyl CoA entering the Citric Acid Cycle is:
41. Mycelium branched and septate, asexual spores conidia produced exogenously and sexual spores produced endogenously are the features of fungi belonging to:
| 1. |
Phycomycetes |
2. |
Ascomycetes |
| 3. |
Basidiomycetes |
4. |
Deuteromycetes |
42. Which law is based on the fact that the alleles do not show ant blending and that both the characters are recovered as such in the F2 generation though one of these is not seen at the F1 stage?
1. Law of dominance
2. Law of segregation
3. Law of independent assortment
4. Law of linkage
43. What is incorrect about CF
0 part of ATP synthase?
| 1. |
It is embedded in the thylakoid membrane |
| 2. |
It forms a transmembrane channel |
| 3. |
It carries out facilitated diffusion of protons |
| 4. |
It protrudes on the outer surface of the thylakoid membrane on the side that faces the stroma |
44. All the following plants belong to the family Solanaceae, except:
| 1. |
Belladonna |
2. |
Tobacco |
| 3. |
Indigofera |
4. |
Petunia |
45. The cell wall of the cells of an alga has cellulose and algin. Another feature expected in this alga would be:
| 1. |
Floridean starch as stored food |
| 2. |
Phycoerythrin in addition to chlorophyll a, c |
| 3. |
2, unequal, lateral flagellar insertions |
| 4. |
Polysulphate esters in cell wall |
46. Accessory pigments of photosynthesis in higher plants:
| I: |
include chlorophyll b, xanthophylls and carotenoids. |
| II: |
absorb light and transfer electrons to chlorophyll a. |
| III: |
protect chlorophyll a from photo-oxidation. |
1. Only
I and
II are correct
2. Only
I and
III are correct
3. Only
II and
III are correct
4.
I,
II and
III are correct
47. Which of the following would be the oldest in the given periods of geological time scale?
| 1. |
Carboniferous |
2. |
Permian |
| 3. |
Triassic |
4. |
Jurassic |
48. The number of carbon atoms are 3 in all of the following molecules except:
1. Dihydroxyacetone phosphate
2. Glyceraldehyde 3-phosphate
3. Acetyl CoA
4. Pyruvic acid
49. A population with finite resources shows a logistic growth curve where the correct sequence of events will be:
| 1. |
Stationary phase → Acceleration phase → Lag phase → Asymptote |
| 2. |
Acceleration phase → Deceleration → Asymptote |
| 3. |
Acceleration phase → Leg phase → Stationary phase |
| 4. |
Lag phase → Acceleration phase → Deceleration → Asymptote |
50. A plant hormone least likely to be classified a growth promoter in plants will be:
| 1. |
auxins |
2. |
gibberellins |
| 3. |
cytokinins |
4. |
abscisic acid |
Zoology - Section A
51. Identify the correct statements:
| I: |
Fertilization occurs in the ampulla of the fallopian tube |
| II: |
Implantation occurs at the stage of blastocyst |
| III: |
First movements of the foetus are usually observed during the fifth month |
1. Only
II
2. Only
I and
II
3. Only
II and
III
4.
I,
II and
III
52. Consider the two statements:
| I: |
During pregnancy, there is no menstruation. |
| II: |
Corpus luteum degenerates early in the event of fertilization |
1. Both I and II are correct and II explains I
2. Both I and II are correct but II does not explain I
3. I is correct but II is incorrect
4. I is incorrect but II is correct
53.
| Assertion (A): |
Competition can be avoided even if two species compete for the same resource |
| Reason (R): |
The competition species can partition their resources to avoid competition |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is False but (R) is True |
| 4. |
(A) is True but (R) is False |
54. Which of the following is not an example of commensalism?
1. An orchid growing as an epiphyte on a mango branch
2. Barnacles growing on the back of a whale
3. Cuckoo laying eggs in the nests of crow
4. Clown fish living among the tentacles of sea anemone
55. The discipline of biotechnology was founded on the breakthrough work of:
| 1. |
Bolivar and Rodriquez |
2. |
Cohen and Boyer |
| 3. |
Nathans and Daniels |
4. |
Redi and Spallanzani |
56. Which of the following drugs has a potent stimulating action on CNS, producing a sense of euphoria and increased energy?
| 1. |
Cocaine |
2. |
Morphine |
| 3. |
Barbiturates |
4. |
Benzodiazepines |
57. That embryos never pass through the adult stages of other animals was proposed by:
| 1. |
Karl Ernst von Baer |
2. |
Hugo de Vries |
| 3. |
Eldredge and Gould |
4. |
Ernst Haeckel |
58. Which class of vertebrates bears jaw but does not bear limbs?
| 1. |
Cyclostomata |
2. |
Chondrichthyes |
| 3. |
Amphibia |
4. |
Reptilia |
59. Regarding polysaccharides:
| I: |
Cellulose does not contain complex helices. |
| II: |
Inulin is a polymer of fructose. |
| III: |
Left end of glycogen is the reducing end |
1. Only
I and
II are correct
2. Only
I and
III are correct
3. Only
II and
III are correct
4.
I,
II and
III are correct
60. Which of the following is not associated with mitosis?
| 1. |
Growth of multicellular living organisms |
| 2. |
Restoration of effective nuclear/cytoplasmic ratio |
| 3. |
Cell repair |
| 4. |
Pairing of homologous chromosomes and crossing over |
61. In a healthy human, under normal physiological conditions the partial pressure of oxygen in systemic [oxygenated blood] circulation is:
1. about 104 mm Hg
2. about 40 mm Hg
3. about 95 mm Hg
4. about 159 mm Hg
62. Approximately what amount of carbon dioxide is removed by lungs per hour in a healthy resting individual?
| 1. |
200 ml |
2. |
12 L |
| 3. |
122 L |
4. |
288 L |
63. Medulla oblongata:
| I: |
is a part of the brain stem |
| II: |
is a part of the hind brain |
| III: |
is a part of the limbic system |
| 1. |
Only I and II |
2. |
Only I and III |
| 3. |
Only II and III |
4. |
I, II and III |
64. What is correct regarding iodothyronines?
1. They are derivatives of tryptophan amino acid
2. They are peptides
3. They interact with intracellular receptors
4. They act both as hormones neurotransmitters
65. During the ventricular systole:
| I: |
The atria also undergo systole |
| II: |
The atrioventricular valves get closed |
| III: |
The semilunar valves are forced open |
1. Only
I and
II are correct
2. Only
I and
III are correct
3. Only
II and
III are correct
4.
I,
II and
III are correct
66. A fluid-filled cavity called as ‘antrum’ is characteristically seen in a :
| 1. |
Secondary follicle |
2. |
Tertiary follicle |
| 3. |
Morula |
4. |
Blastocyst |
67. A person suffering from Klinefelter’s syndrome:
1. has 45 autosomes
2. is likely to have short stature and webbed neck
3. can develop Gynecomastia
4. has normal fertility
68. Km value of an enzyme signifies the substrate concentration at which the enzyme catalyzed reaction:
1. achieves Vmax
2. achieves ½ Vmax
3. is not affected by the presence of a competitive inhibitor
4. stops
69. At the alveoli:
| I: |
The factors are all favourable for the formation of oxyhemoglobin. |
| II: |
Carbon dioxide trapped as bicarbonate at the tissue level is released out as carbon dioxide. |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
70. Diminished ability of kidney to concentrate urine leading to dehydration can be due to:
1. Increased secretion of ADH
2. Decreased secretion of ADH
3. Increased secretion of Insulin
4. Decreased secretion of ANF
71. In the lac operon:
1. the repressor of the operon is synthesized constitutively
2. glucose and galactose act as inducers
3. the lac y gene codes for beta galactosidase
4. regulation by repressor is positive regulation
72. Which of the following steps would be most important to check the population growth rate of our country?
| 1. |
Statutory ban on amniocentesis |
| 2. |
Increasing age of marriage for females |
| 3. |
Allowing MTP till 28 weeks of pregnancy |
| 4. |
Motivating couples for using various contraceptive methods |
73. Which of the following animals belongs to the subphylum Vertebrata?
1. Doliolum
2. Branchistoma
3. Petromyzon
4. Salpa
74. Fallopian tubes are lined with:
| 1. |
Cuboidal ciliated epithelium |
| 2. |
Cuboidal brush bordered epithelium |
| 3. |
Columnar ciliated epithelium |
| 4. |
Columnar brush bordered epithelium |
75. Octopus has:
| 1. |
Tetrameric radial symmetry |
| 2. |
Hexameric radial symmetry |
| 3. |
Octomeric radial symmetry |
| 4. |
Bilateral symmetry |
76. Match blood cells given in Column I with their most important function in Column II and select the correct match from the codes given:
|
Column I |
|
Column II |
| A. |
Neutrophils |
P. |
Phagocytosis |
| B. |
Basophils |
Q. |
Inflammatory reactions |
| C. |
Lymphocytes |
R. |
Immune responses |
| D. |
Thrombocytes |
S. |
Coagulation of blood |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
Q |
P |
S |
R |
| 3. |
R |
S |
P |
Q |
| 4. |
S |
R |
Q |
P |
77. A motor neuron, at the neuromuscular junction, releases a neurotransmitter that is:
1. Epinephrine
2. Acetylcholine
3. Dopamine
4. Serotonin
78. Identify the hormone that is not correctly matched with its function:
| 1. |
Parathyroid hormone |
Increases calcium levels in the blood |
| 2. |
Aldosterone |
Stimulates reabsorption of sodium at the renal tubules |
| 3. |
Insulin |
Enhances cellular glucose uptake and utilization |
| 4. |
Cortisol |
Enhances cellular amino acids uptake and utilization |
79. Treatment with divalent cations, ice, and heat shock are used in the transformation of certain:
1. Dicot plant cells
2. Monocot plant cells
3. Bacterial cells
4. Animal cells
80. To produce recombinant human insulin, the pharmaceutical company Eli Lily:
| 1. |
produced chains A and B together, cleaved them and combined them by disulfide bonds |
| 2. |
produced chains A and B separately, extracted and combined by creating disulfide bonds |
| 3. |
produced chains A and B together, cleaved them and combined them by hydrogen bonds |
| 4. |
produced chains A and B separately, extracted and combined by creating hydrogen bonds |
81. The thymus:
| 1. |
is located dorsal to the heart and aorta |
| 2. |
is very small in size at birth and gradually increases in size after puberty |
| 3. |
provides the site for interaction of lymphocytes with the antigen |
| 4. |
is a primary lymphoid organ in the human body |
82. Pouched mammals of Australia survived because:
| 1. |
they all are top predators in a food chain |
| 2. |
they are all herbivores and hence get to feed on large amounts of available primary production |
| 3. |
of lack of competition from any other mammals |
| 4. |
the Australian environment is not conducive for the survival of true placental mammals |
83. Consider the given two statements:
| Assertion (A): |
Atrial Natriuretic Factor (ANF) mechanism acts as a check on the renin-angiotensin mechanism. |
| Reason (R): |
An increase in blood flow to the atria of the heart can cause the release of ANF. |
| 1. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 2. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
| 3. |
(A) is True; (R) is False. |
| 4. |
Both (A) and (R) are False. |
84. Steroid hormones are produced in human body by:
1. GIT and Adrenal medulla
2. Adrenal cortex and Thyroid
3. Adrenal cortex and Gonads
4. Gonads and Thymus
85. An autoimmune disorder that targets the alpha cells of pancreas will cause:
I: an increase in secretion of insulin
II: a decrease in secretion of glucagon
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
Zoology - Section B
86. What is correct regarding both spermatogenesis and oogenesis?
1. Begin after the onset of puberty
2. The resultant cells of meiosis I are equal in size
3. The resultant cells of meiosis I are unequal in size
4. The resultant gametes are haploid
87. Absence of a menstrual period in a woman of reproductive age is called as:
| 1. |
Menopause |
2. |
Dysmenorrhoea |
| 3. |
Menorrhagia |
4. |
Amenorrhoea |
88. Consider the following two statements:
| I: |
Plasmid can be used as a vector in recombinant DNA technology experiments. |
| II: |
Plasmid is an extrachromosomal, circular, double-stranded DNA found in some bacteria. |
| 1. |
Both I and II are correct and II explains I |
| 2. |
Both I and II are correct but II does not explain I |
| 3. |
I is correct but II is incorrect |
| 4. |
Both I and II are incorrect |
89. Match each item in
Column-I with one in
Column-II and select the correct match from the codes given:
|
Column-I
[Ancestors of Home sapiens] |
|
Column-II
[Cranial capacity] |
| A. |
Homo erectus |
P. |
1400 cc |
| B. |
Homo habilis |
Q. |
450 cc |
| C. |
Australopithecus |
R. |
650-800 cc |
| D. |
Neanderthal man |
S. |
900 cc |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
P |
S |
R |
| 2. |
R |
P |
Q |
S |
| 3. |
P |
Q |
R |
S |
| 4. |
S |
R |
Q |
P |
90. Consider the given two statements:
| I: |
When a neuron is not conducting any impulse, i.e., resting, the axonal membrane is comparatively more permeable to potassium ions (K+ ) and nearly impermeable to sodium ions (Na+). |
| II: |
Similarly, the membrane is permeable to negatively charged proteins present in the axoplasm. |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
91. Which of the following glands do not secrete a protein hormone?
| 1. |
Pancreas |
2. |
Thyroid |
| 3. |
Adenohypophysis |
4. |
Gonads |
92. Identify the incorrectly matched pair:
1. Radula – Molluscs
2. Malpighian tubules– Insects
3. Choanocytes– Sponges
4. Cnidocytes – Ctenophora
93.
| Assertion (A): |
Living state is a non-equilibrium steady state. |
| Reason (R): |
Living organisms work continuously. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
94. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
The amount of carbon dioxide that can diffuse through the diffusion membrane per unit difference in partial pressure is much higher compared to that of oxygen. |
| Reason (R): |
The solubility of carbon dioxide is 20 – 25 times lower than that of oxygen. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
95. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
Rh incompatibility related complication does not arise in first pregnancy even if an Rh negative mother is carrying an Rh positive foetus. |
| Reason (R): |
Rh antigens are proteins. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
96. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
The filtrate gets concentrated as it moves down the descending limb of loop of Henle in the human nephron. |
| Reason (R): |
The descending limb of loop of Henle is impermeable to water but allows transport of electrolytes actively or passively. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
97. The given diagram shows a representation of a synovial joint found between atlas and axis vertebrae. What is this type of synovial joint called?

| 1. |
Hinge |
2. |
Ball and socket |
| 3. |
Pivot |
4. |
Saddle |
98. HIV:
| I: |
uses phagocytic neutrophils as factory to produce more virus particles. |
| II: |
destroys B lymphocytes selectively. |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
99. Which of the following is a free-living non-pathogenic nematode, frequently used in the study of developmental genetics, and whose genome has been sequenced?
1. Arabidopsis thaliana
2. Caenorhabditis elegans
3. Drosophila melanogaster
4. Meloidogyne incognitia
100. Forebrain in frogs does not include:
| 1. |
olfactory lobes |
| 2. |
paired cerebral hemispheres |
| 3. |
unpaired diencephalon |
| 4. |
a pair of optic lobes |
Chemistry - Section A
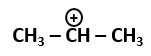
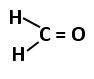
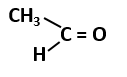
101. The most stable carbocation among the following is:
102. For the reaction,
\(H^{+}+BrO^-_3+3Br^- \rightarrow~5Br_2+H_2O\) the correct representation of the consumption & formation of reactants and products is:
1.
2.
3.
4.
103. The correct order of bond strength of H2O, H2S, H2Se, H2Te is:
1. \(\mathrm{{H}_2 {O}>{H}_2 {~S}>{H}_2 {Se}>{H}_2 {Te}} \)
2. \(\mathrm{{H}_2 {~S}>{H}_2 {O}>{H}_2 {Se}>{H}_2 {Te}} \)
3. \(\mathrm{{H}_2 {Te}>{H}_2 {Se}>{H}_2 {~S}>{H}_2 {O}} \)
4. \(\mathrm{{H}_2 {Te}>{H}_2 {~S}>{H}_2 {O}>{H}_2} \mathrm{Se} \)
104. Given below are two statements:
| Assertion (A): |
On heating PCl5, it readily converts into PCl3 and Cl2 |
| Reason (R): |
The axial P-Cl bond is larger than the equitorial P-Cl bond |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
105. The vapour pressures of benzene and toluene in pure states are 700 mm of Hg and 600 mm of Hg respectively. If equal masses of benzene and toluene are mixed together then the total vapour pressure of the solution will be approximately:
| 1. |
620 mm of Hg |
2. |
640 mm of Hg |
| 3. |
654 mm of Hg |
4. |
674 mm of Hg |
106. Aqueous NaCl solution is electrolyzed using platinum electrodes. It can be concluded that the pH of the solution:
| 1. |
Increases |
2. |
Decreases |
| 3. |
Unchanged |
4. |
Can't be predicted |
107. The colourless ion from among the following is:
1. Mn2+
2. Cu+
3. Cr3+
4. Fe2+
108. Consider the given reaction:

Compound 'B' is:
| 1. |
Crotonaldehyde |
2. |
Acrolein |
| 3. |
Mesityl oxide |
4. |
Propanal |
109. The Hell Volhard Zelinsky (HVZ) reaction is not given by:
1. Acetic acid
2. 2,2-Dimethyl propanoic acid
3. 2-Methyl propanoic acid
4. 2,3-Dimethyl butanoic acid
110. Consider the reaction, A → Products. When the concentration of A increases 4 times then its rate increases 2 times. The order with respect to A will be:
| 1. |
0.5 |
2. |
1.0 |
| 3. |
2.0 |
4. |
3 .0 |
111. The most basic oxide is:
| 1. |
P2O3 |
2. |
Bi2O5 |
| 3. |
As2O3 |
4. |
Bi2O3 |
112. In [Cr(NH
3)
6]Cl
3, the spin-only magnetic moment of central atom will be:
| 1. |
1.732 BM |
2. |
2.45 BM |
| 3. |
3.87 BM |
4. |
4.9 BM |
113. Case Study
A halogen is more electronegative than carbon. Consequently, the two atoms do not share their bonding electrons equally, because a partial negative charge is formed on halogen and a partial positive charge on the carbon to which it is bonded. It is the polar-carbon halogen bond that causes alkyl halides to undergo substitution reactions.
There are two important mechanisms for the substitution reaction:
| 1. |
SN1: The carbon-halogen bond breaks heterolytically without any assistance from the nucleophile, forming a carbocation. The carbocation- an electrophile-then reacts with the nucleophile to form the substitution product |
| 2. |
SN2: A nucleophile is attracted to the partially positively charged carbon (an electrophile).As the nucleophile approaches the carbon and forms a new bond, the carbon-halogen bond breaks heterolytically (the halogen takes both of the bonding electrons) |
Which solvent, from the following, favors the S
N2 mechanism?
| 1. |
Non-polar solvent |
2. |
Polar protic solvent |
| 3. |
Polar aprotic solvent |
4. |
All of the above |
114. When aniline is reacted with concentrated HNO3, then which, of the following, nitro derivative of aniline has the highest percentage?
1. o-nitroaniline
2. m-nitoaniline
3. p-nitroaniline
4. All have equal percentages
115. The correct representation for the formation of CH4(g) is:
1. \(\mathrm{C} (diamond )+2 \mathrm{H}_{2}(g) \longrightarrow \mathrm{CH}_{4}(g)\)
2. \(C(graphite) +4 \mathrm{H}{(g)} \longrightarrow \mathrm{CH}_{4}(g)\)
3. \(\mathrm{C} (graphite) +2 \mathrm{H}_{2}(g) \longrightarrow \mathrm{CH}_{4}(g)\)
4. \(C( diamond \left.)+4 \mathrm{H}{\mathrm{(g}}\right) \rightarrow \mathrm{CH}_{4}(g)\)
116. The strongest conjugate base is:
\(1.~CH_3COO^-\\ \\ 2.~Cl^-\\ \\ 3. \ NO^-_3\\ \\ 4. ~HSO^-_4 \)
117. When borax is red-heated then a transparent glassy solid is formed. The composition of this glassy solid is:
1. B2O3
2. NaBO2 + B2O3
3. Na2B4O7
4. NaBO2
118. The hyperconjugation effect is a permanent effect in which localization of
\(\sigma\) electrons of the C-H bond of an alkyl group directly attached to an atom of the unsaturated system or to an atom with an unshared p orbital takes place.
Hydrogen attaches to alpha carbon in an allylic compound and alpha carbon of carbon-containing positive charge show hyperconjugation. The orbital diagram showing hyperconjugation in ethyl cation is as follows:
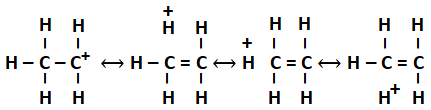
In general, the greater the number of alkyl groups attached to a positively charged carbon atom, greater is the hyperconjugation interaction and stabilization of the cation. The more hyperconjugation in the structure more is the stability.
The most stable carbocation is:
119. During the preparation of Mohr's salt solution (Ferrous ammonium sulphate), which of the following acid is added to prevent hydrolysis of Fe2+ ion?
1. Concentrated sulphuric acid
2. Dilute nitric acid
3. Dilute sulphuric acid
4. Dilute hydrochloric acid
120. The most acidic compound among the following, is:
1. p-Nitrophenol
2. o-Nitrophenol
3. o-Cresol
4. m-Nitrophenol
121. When glucose is reduced with red phosphorous and HI, then n-hexane is formed. This reaction shows that glucose has:
1. A ring structure
2. A linear chain of six carbon atoms
3. Five -OH groups
4. \(-CHO\) group
122. Which of the following species has the highest bond energy?
| 1. |
\(O_2\) |
2. |
\(O^-_2\) |
| 3. |
\(O^+_2\) |
4. |
\(O^{2+}_2\) |
123. Consider the reaction given below:
\(xMnO_2+yHCl\xrightarrow{~~~~~~~~}xMnCl_2+ZH_2O+aCl_2 \)
The value of \([(x+y)-(a+z)] \) will be:
1. 1.5
2. 3
3. 2
4. 1
124. When but-2-yne reacts with \(Na/Liq.NH_3\), then the product formed is:
1. Butane
2. Cis-but-2-ene
3. Trans-but-2-ene
4. But-1-yne
125. For the reaction,
\(2A+B\xrightarrow{~~~~~~~~}3C,8\) moles of
\(A\) reacts with
\(5\) moles of
\(B\) to form
\(C.\)
Calculate the maximum number of moles that are formed.
| 1. |
\(13\) mole |
2. |
\(15\) mole |
| 3. |
\(12\) mole |
4. |
\(8\) mole |
126. The de-Broglie wavelength of a particle of mass 6.63 g moving with a velocity of 100 \(ms^{-1}\) is:
1. \(10^{-33} \mathrm{~m}\)
2. \(10^{-35} \mathrm{~m}\)
3. \(10^{-31} \mathrm{~m}\)
4. \(10^{-25} \mathrm{~m}\)
127. Amongst the given trioxides of the nitrogen family, the amphoteric oxide is:
1. \(P_2O_3 \)
2. \(Sb_2O_3 \)
3. \(Bi_2O_3 \)
4. None of the above
128. The formal charge of N in \(NO^-_3\) is:
1. Zero
2. -1
3. +1
4. -3
129. The number of bridging carbonyl groups in [Co2(CO)8] and [Mn2(CO)10], respectively are:
| 1. |
2 and 0 |
2. |
2 and 2 |
| 3. |
2 and 4 |
4. |
0 and 2 |
130. Consider the following facts:
| A. |
Copper solution can be stirred with a silver spoon. |
| B. |
Zinc solution can be stirred with a silver spoon. |
| C. |
Copper solution cannot be stored in a zinc pot. |
Based on the above facts, choose the correct order of the standard reduction potential of
\(Cu^{2+}/Cu ; ~Zn^{2+} /Zn \) and
\(Ag^+ /Ag \):
| 1. |
\(E^o_{Cu^{2+}/Cu } > E^o_{Ag^{+}/Ag } > E^o_{Zn^{2+}/Zn } \) |
| 2. |
\(E^o_{Cu^{2+}/Cu } < E^o_{Ag^{+}/Ag } < E^o_{Zn^{2+}/Zn } \) |
| 3. |
\(E^o_{Ag^{+}/Ag } > E^o_{Cu^{2+}/Cu } > E^o_{Zn^{2+}/Zn } \) |
| 4. |
\(E^o_{Zn^{2+}/Zn } > E^o_{Cu^{2+}/Cu } > E^o_{Ag^{+}/Ag } \) |
131. The values of \(K_{sp}\) of CaCO3 and CaC2O4 are 4.7 × 10–9 and 1.3 × 10–9 respectively at 25°C. If the mixture of these two is washed with water, what is the concentration of Ca2+ ions in the water?
1. 5.8 × 10–5 M
2. 6.8 × 10–5 M
3. 3.6 × 10–5 M
4. 7.7 × 10–5 M
132. Consider the following molecule:
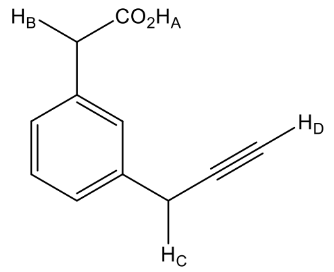
The correct order of the acidic strength of marked hydrogen is:
| 1. |
\(\mathrm{H}_{\mathrm{A}}>\mathrm{H}_{\mathrm{D}}>\mathrm{H}_{\mathrm{B}}>\mathrm{H}_{\mathrm{C}} ~ \) |
2. |
\(\mathrm{H}_{\mathrm{B}}>\mathrm{H}_{\mathrm{A}}>\mathrm{H}_{\mathrm{D}}>\mathrm{H}_{\mathrm{C}} ~\) |
| 3. |
\(\mathrm{H}_{\mathrm{A}}>\mathrm{H}_{\mathrm{B}}>\mathrm{H}_{\mathrm{C}}>\mathrm{H}_{\mathrm{D}} ~\) |
4. |
\(\mathrm{H}_{\mathrm{C}}>\mathrm{H}_{\mathrm{B}}>\mathrm{H}_{\mathrm{D}}>\mathrm{H}_{\mathrm{A}} ~\) |
133. The \(E^{\ominus}\) values for
\(\begin{aligned} & \mathrm{Al}^+ / \mathrm{Al}=+0.55 \mathrm{~V} \text { and } \mathrm{Tl}^{+} / \mathrm{Tl}=-0.34 \mathrm{~V} \\ & \mathrm{Al}^{3+} / \mathrm{Al}=-1.66 \mathrm{~V} \text { and } \mathrm{Tl}^{3+} / \mathrm{Tl}=+1.26 \mathrm{~V} \end{aligned} \)
The incorrect statement among the following is:
1. Al is more electropositive than TI.
2. Tl3+ is a good reducing agent than Tl1+.
3. Al+ is unstable in solution.
4. Tl can be easily oxidized to Tl+ as compared to Tl3+.
134. In which of the following processes does the value of the magnetic moment not change?
1. \(\mathrm{N}_2 \rightarrow \mathrm{N}_2^{-}\)
2. \(\mathrm{N}_2 \rightarrow \mathrm{N}_2^{+}\)
3. \(\mathrm{O}_2 \rightarrow \mathrm{O}_2^{2-} \)
4. \(\mathrm{O}_2^{+} \rightarrow \mathrm{O}_2^{-} \)
135. Addison’s disease is:
| 1. |
hypothyroidism |
2. |
hyperthyroidism |
| 3. |
hypoadrenalism |
4. |
hypopituitarism |
Chemistry - Section B
136. In the following reaction carried out at 500K, 18 moles of N2 gas is mixed with 24 moles of H2 gas. What is the percentage yield of NH3 if the reaction produces 13.5 moles of NH3?
N2(g) + 3 H2 (g) \(\rightarrow\) 2NH3 (g)
1. 16%
2. 66%
3. 72%
4. 84%
137. A hydrocarbon has the molecular formula C5H8 . On ozonolysis, it gives formaldehyde & 2-keto propanal only. The name of hydrocarbon is:
1. 2-Methylbuta-1,3-diene
2. 3-Methylbut-1-yne
3. 3-Methylbuta-1,2-diene
4. 2-Methylbut-2-ene
138. The molarity of NaNO3 solution is 2M. The density of solution is 1.2 gram/ml. The molality of the solution will be:
(Molar weight of NaNO3 = 85)
1. 1.94 m
2. 2 m
3. 2.06 m
4. 1.85 m
139. For the reaction, A + B → Products
| Experiment |
[A] |
[B] |
Rate (mole litre-1 min-1) |
| 1. |
0.01 |
0.01 |
2 × 10-4 |
| 2. |
0.02 |
0.01 |
4 × 10-4 |
| 3. |
0.01 |
0.02 |
8 × 10-4 |
The rate law and unit of rate constant respectively, are:
| 1. |
Rate = K [A]2 [B] and litre2 mole-2 min-1 |
| 2. |
Rate = K [A]3 and litre2 mole-2 min-1 |
| 3. |
Rate = K [A] [B]2 and litre mole-1 min-1 |
| 4. |
Rate = K [A] [B]2 and litre2 mole-2 min-1 |
140. Gold does not react with concentrated HNO3 but dissolves in aqua regia. The product formed, which gets dissolved in the aqua regia solution is:
1. AuCl3
2. Au(NO3)3
3. HAuCl4
4. HAu(NO3)4
141. Which carbonyl compound does not react with NaHSO
3?
142. The correct relation for the work done in isothermal expansion will be:
1. wreversible < wirreversible
2. wreversible > wirreversible
3. wreversible = wirreversible
4. All of the above
143. For the following compound, IUPAC name is:
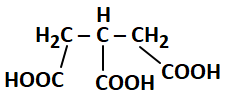
1. 3- Carboxypentan-1,5-dioic acid
2. 1,2,3-tricarboxypropane
3. Propane-1,2,3-tricarboxylic acid
4. All of these
144. Two compartments, A and B, are separated by a semipermeable membrane. Compartment A contains 0.01 M Na₂SO₄ solution, and compartment B contains 0.02 M BaCl₂ solution. In which compartment will the precipitate of BaSO₄ be formed?
1. Compartment A
2. Compartment B
3. No precipitate is formed in any compartment
4. Precipitate forms in both compartments A and B
145. Which of the following solutions acts as a buffer?
1. \(HCl+NH_4Cl\)
2. \(CH_3COONH_4+H_2O\)
3. \(NaOH+CH_3COONa\)
4. \(HNO_3+NaNO_3\)
146. In
\([Cr(NH_3)_6]~[Co(CN)_6]\) , the oxidation state of Cr and Co, respectively, are :
| 1. |
0 and +6 |
2. |
+2 and +4 |
| 3. |
+3 and +3 |
4. |
+4 and +2 |
147. When aluminium is made to react with an excess of \(NaOH\) , a soluble complex of aluminium is formed. In the soluble complex of aluminium , aluminium exists in:
1. Anionic part
2. Cationic part
3. Both cationic as well as anionic part
4. Neutral complex
148. Acetone and methanol are separated by which of the following methods?
| 1. |
Fractional distillation |
2. |
Steam distillation |
| 3. |
Vacuum distillation |
4. |
Azeotropic distillation |
149. In which of the following cells, the e.m.f of the cell will be the highest?
| 1. |
\(\large {Zn | Zn^{2+} ||Cu^{2+} | Cu \atop (1M)(0.1M)}\) |
2. |
\(\large{Zn | Zn^{2+} ||Cu^{2+} | Cu \atop (0.1M)(1M)}\) |
| 3. |
\(\large{Zn | Zn^{2+} ||Cu^{2+} | Cu \atop (0.01M)(2M)}\) |
4. |
\(\large{Zn | Zn^{2+} ||Cu^{2+} | Cu \atop (0.2M)(1M)}\) |
150. Given the following reaction
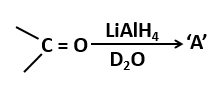
The compound 'A' in the above reaction is:
Physics - Section A
151. The magnitude of the vector
\(\widehat i+\widehat i\times\widehat j+(\widehat i\times\widehat j)\times\widehat i+((\widehat i\times\widehat j)\times\widehat i)\times\widehat j\):
1. \(1\)
2. \(\sqrt2\)
3. \(\sqrt3\)
4. \(2\)
152. Assume that a space shuttle flies in a circular orbit very close to the Earth's surface. Taking the radius of the space shuttle's orbit to be equal to the radius of the earth \((R)\) and the acceleration due to gravity to be \(g,\) the time period of one revolution of the space shuttle is (nearly):
| 1. |
\(\sqrt{\dfrac{2R}{g}}\) |
2. |
\(\sqrt{\dfrac{\pi R}{g}}\) |
| 3. |
\(\sqrt{\dfrac{2\pi R}{g}}\) |
4. |
\(\sqrt{\dfrac{4\pi^2 R}{g}}\) |
153. Bernoulli's theorem in fluid mechanics is based on:
| 1. |
conservation of momentum. |
| 2. |
conservation of force. |
| 3. |
conservation of angular momentum. |
| 4. |
work-energy theorem. |
154. The internal energy of a gas is given by \(U=2pV.\) The gas expands from \(100\) cc to \(200\) cc against a constant pressure of \(10^{5}\) Pa. The heat absorbed by the gas is:
| 1. |
\(10\) J |
2. |
\(20\) J |
| 3. |
\(30\) J |
4. |
\(40\) J |
155. Two ideal gases contained in vessels \(A\) and \(B\) are connected by means of a pipe, and the plug is opened so that the gases mix. The final pressure is \(P\) (atm) and the temperature is \(T\) (kelvin). Then, \(\dfrac{P}{T}=\) (numerically)

| 1. |
\(\dfrac{1}{100}\) |
2. |
\(\dfrac{1}{200}\) |
| 3. |
\(\dfrac{1}{400}\) |
4. |
none of these |
156. The pressure exerted by a gas enclosed within a room is due to:
| 1. |
collisions of the gas molecules with the walls of the room |
| 2. |
the repulsive force between molecules of the gas |
| 3. |
weight of the molecules of the gas |
| 4. |
angular momentum of the molecules |
157. The maximum speed and acceleration of a particle undergoing SHM are \(v_0\) and \(a_0,\) respectively. The time period of the SHM is:
| 1. |
\(\dfrac{2\pi v_0}{a_0}\) |
2. |
\(\dfrac{2\pi a_0}{v_0}\) |
| 3. |
\(\dfrac{v_0}{a_0}\) |
4. |
\(\dfrac{2v_0}{a_0}\) |
158. Given below are two statements:
| Assertion (A): |
Sound waves in a gas are pressure waves, but these are also accompanied by changes in local temperature. |
| Reason (R): |
Sound waves are propagated in gas through an adiabatic process and hence accompanied by temperature variations. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
159. A charged particle of charge \(q\) and mass \(m\) is projected vertically upward with a speed \(u\). Acceleration due to gravity \((g)\) acts downwards, while a uniform electric field \(E\) acts along the horizontal. The speed of the projectile's impact on the horizontal is \(v\), while the horizontal range is \(R.\) Then:
1. \(v^2=u^2+2gR\)
2. \(v^2=u^2+2gR\left(\frac{qE}{mg}\right)\)
3. \(v^2=u^2+gR\)
4. \(v^2=u^2+gR\left(\frac{qE}{mg}\right)\)
160. Initially, uncharged capacitors are connected in a circuit, as shown in the diagram. The potentials at
\(C,\) \(D\) satisfy:
\(V_C=V_D.\) Then:
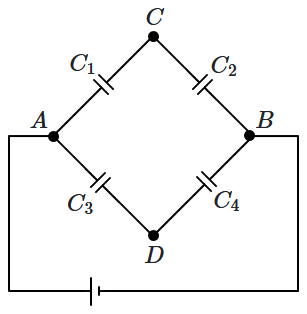
| 1. |
\(C_1C_2 = C_3C_4\) |
2. |
\(\dfrac{C_1}{C_4}=\dfrac{C_2}{C_3}\) |
| 3. |
\(\dfrac{C_1}{C_3}=\dfrac{C_2}{C_4}\) |
4. |
\(C_1C_3 = C_2C_4\)
|
161. Two very long wires of length
\(L\) are placed parallel to each other separated by a distance
\(r(r << L) . \) The wires carry equal currents
\(i .\) The force between the two wires is nearly:
| 1. |
\(\dfrac{\mu_{0} i^{2} L}{2 \pi r}\) |
2. |
\(\dfrac{\mu_{0} i^{2} L}{4 \pi r}\) |
| 3. |
\(\dfrac{\mu_{0} i^{2} L}{2 r}\) |
4. |
\(\dfrac{\mu_{0} i^{2} L}{4 r}\) |
162. A straight long current-carrying wire carrying a current
\(i\) is placed in a uniform magnetic field, and it is observed that the field vanishes at a point which is at a distance
\(r\) from the wire. The force on the wire, per unit length, is:
| 1. |
\(\dfrac{\mu_{0} i^{2}}{2 \pi r}\) |
2. |
\(\dfrac{\mu_{0} i^{2}}{4 \pi r}\) |
| 3. |
\(\dfrac{\sqrt{2} \mu_{0} i^{2}}{2 \pi r}\) |
4. |
\( \dfrac{\mu_{0} r^{2}}{2 \pi r \sqrt{2}}\) |
163. A \(100\)-turn coil of wire of size \(2~\text{cm}\times 1.5~\text{cm}\) is suspended between the poles of a magnet producing a field of \(1~\text T,\) inside a galvanometer. Calculate the torque on the coil due to a current of \(0.1~\text{A}\) passing through the coil.
1. \(3 \times 10^{-5}\) N-m
2. \(30\) N-m
3. \(3 \times 10^{-3}\) N-m
4. \(3 \times 10^{-2}\) N-m
164. The current through the primary of a step-down transformer with a turns-ratio of \(4\) is \(1.6~\text{A}\). The current in the secondary circuit is:
1. \(0.4~\text{A}\)
2. \(0.8~\text{A}\)
3. \(6.4~\text{A}\)
4. \(12.8~\text{A}\)
165. Given below are two statements:
A rectangular loop of resistance
\(R\) is placed in a region where there is a magnetic field
\(B\), passing perpendicularly through the plane of the loop, as shown in the figure. The loop is pulled with a constant velocity
\(v\) so that it is partially within the field.

| Assertion (A): |
An external force \(F\) is needed to be applied in the direction of the velocity \(v\) so that the loop can move with constant velocity \(v\). |
| Reason (R): |
As the loop moves towards the right, the magnetic flux decreases inducing an emf and a corresponding current. This current causes a retarding force to be exerted on the wire. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
166. An electromagnetic wave is incident onto a surface and delivers an energy
\(E\) and a momentum
\(p.\) Then:
| 1. |
\(E\) and \(p\) are both zero. |
| 2. |
\(E\) and \(p\) are both non-zero. |
| 3. |
\(E\) is zero and \(p\) is non-zero. |
| 4. |
\(E\) is non-zero, \(p\) is zero. |
167. Two parallel mirrors
\(M_1\) and
\(M_2\) are placed facing each other on opposite sides of a room. The separation of the mirrors, as shown in the figure below, is
\(4~\text{m}.\) A point object
\(P\) is placed at a distance of
\(1~\text{m}\) from
\(M_1.\) The separation between the images formed after a single reflection is:

| 1. |
\(2~\text{m}\) |
2. |
\(6~\text{m}\) |
| 3. |
\(8~\text{m}\) |
4. |
\(4~\text{m}\) |
168. Atomic number increases in:
| 1. |
\(\alpha\text-\)decay. |
| 2. |
\(\beta^{-}\text-\)decay. |
| 3. |
\(\beta^{+}\text{-}\)decay. |
| 4. |
\(K\text{-}\)electron capture. |
169. The equivalent resistance between
\(A\) and
\(B\) of the network shown below is:
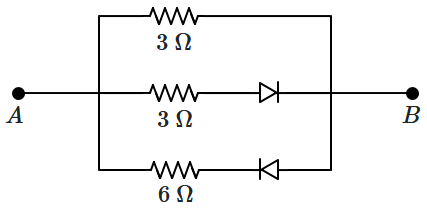
| 1. |
\(1.2~ \Omega\) |
| 2. |
\(3 ~\Omega\) |
| 3. |
\(1.5~\Omega\) if \(V_{A}>V_{B}\) and \(2~\Omega\) if \(V_{A}<V_{B}\) |
| 4. |
\(1.5~\Omega\) if \(V_{A}<V_{B}\) |
170. The end
\('B'\) of the circuit is earthed
\((V_B = 0)\) while a sinusoidal voltage is applied at
\('A';\) \(V_{A}=V_{0} \sin \omega t.\) The rms voltage across the capacitor
\(C\) equals that across the upper resistor
\(R\) (as shown in the figure). What voltage should be applied at
\(X\) \((V_X)\) so that the current flowing into both the resistors is equal?
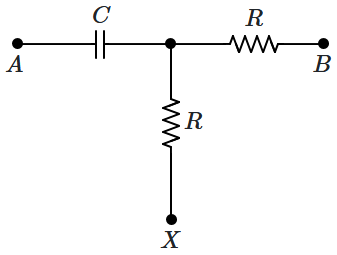
1.
\(V_{X}=\dfrac{V_{0}}{2} \sin \omega t\)
2.
\(V_{X}=\dfrac{V_{0}}{2} \cos\omega t\)
3.
\(V_{X}= V_0 (\sin \omega t + \cos \omega t)\)
4.
\(V_X = 0\)
171. Young's double-slit experiment is conducted with the light of wavelength \(700~\text{nm}\). A thin strip of a glass of refractive index \(\mu=1.7\) is placed in front of one of the slits and the fringe system is displaced by \(10\) fringes. The thickness of the glass strip is:
1. \(10~\mu \text{m}\)
2. \(1~\mu \text{m}\)
3. \(17~\mu \text{m}\)
4. \(1.7~\mu \text{m}\)
172. A monochromatic source of light emits \(50~\text{W}\) at a wavelength of \(400~\text{nm}.\) The number of photons emitted per second is approximately (take \({hc} = 1240~\text{eV-nm}\)):
1. \(10^{20}\)
2. \(10^{25}\)
3. \(10^{30}\)
4. \(10^{40}\)
173. The quantity "Action" (
\(S\)) is defined by the equation:
\(S=\int_{0}^{t}(K-U)^{} d t\)
where
\(K\) is the kinetic energy,
\(U\) is the potential energy and the integral is over the time,
\(t\) during the motion.
The proper unit of action will be:
| 1. |
kg-m/s |
2. |
kg-m2 |
| 3. |
kg-m2-s |
4. |
kg-m2/s |
174. Which of the following statements is true about the motion depicted in the diagram?

| 1. |
The acceleration is constant and non-zero. |
| 2. |
The velocity changes suddenly during the motion. |
| 3. |
The velocity is positive throughout. |
| 4. |
All of the above are true. |
175. A block of mass
\(1~\text{kg}\) moving with a velocity of
\(4~\text{m/s}\) strikes a second block of mass
\(2~\text{kg}\) at rest. The first block is brought to rest by the collision. The coefficient of restitution is:
| 1. |
\(1 \over2 \) |
2. |
\(1 \over 3 \) |
| 3. |
\(2 \over 3 \) |
4. |
\(1 \over 4 \) |
176. Two identical blocks are connected by an ideal spring and the system is allowed to oscillate, when undergoing horizontal displacements in opposite directions, with the centre-of-mass at rest.
\(O\) is the mid-point of the spring,
\(A\) is the left end point,
\(B\) is the right end-point. The motion of
\(A\) is described by:
\(x_{\Large_A} = A_0 \sin ~\omega t\) (displacement is taken to be positive rightward).
Then, the motion of the point
\(B\) is described by:
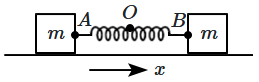
| 1. |
\(x_{\Large_B}=A_{0} \sin \omega t\) |
2. |
\(x_{\Large_B}=A_{0} \cos \omega t\) |
| 3. |
\(x_{B}=A_{0} \sin (\omega t+\pi)\) |
4. |
\(x_{\Large_B}=A_{0} \cos (\omega t+\pi)\) |
177. All the resistances in circuit are
\(2~\Omega\) each,
\(E_1=4~\text{V}\) and
\(E_2=1~\text{V}\). The potential difference between
\(C\) and
\(D\) (i.e.
\(V_{C}-V_{D}\)) is:

1.
\(1.5~\text{V}\)
2.
\(-1.5~\text{V}\)
3.
\(1.2~\text{V}\)
4.
\(-1.2~\text{V}\)
178. A small solenoid is kept inside a much larger solenoid, with their axes parallel to each other. The small solenoid has a cross-sectional radius
\(r_1,\) length
\(l_1\) and the total number of turns
\(N_1.\) The corresponding quantities for the larger solenoid are:
\(r_2,~ l_2,~ N_2\) respectively.
Their mutual inductance is (nearly) given by:
| 1. |
\(\dfrac{\mu_0\pi r^2_1N_1N_2}{l_2}\) |
2. |
\(\dfrac{\mu_0\pi r^2_1N_1N_2}{\sqrt{l_1l_2}}\) |
| 3. |
\(\dfrac{\mu_0\pi r^2_1N_1N_2}{l_1}\) |
4. |
\(\dfrac{\mu_0~\pi r_1r_2N_1N_2}{\sqrt{l_1}}\) |
179. Which of the following has the same orbital radius as the Bohr radius?
| 1. |
\(n=2\) state of \(\mathrm{He}^{+}\) \(\left(Z=2\right)\) ion |
| 2. |
\(n=4\) state of \(\mathrm{He}^{+}\) \(\left(Z=2\right)\) ion |
| 3. |
\(n=2 \) state of \(\mathrm{Be}^{3+}\)\(\left(Z=4\right)\) ion |
| 4. |
\(n=3\) state of \(\mathrm{Li}^{2+}\) \(\left(Z=3\right)\) ion |
180. The band gap between the valence band and the conduction band in a certain material is \(3~\text{eV}.\) What is the longest wavelength of light that can create an electron-hole pair in the material, with the electron in the conduction band and the hole in the valence band?
(take \(hc=1240~\text{eV-nm}\) )
1. \(414~\text{nm}\)
2. \(300~\text{nm}\)
3. \(830~\text{nm}\)
4. \(207~\text{nm}\)
181. Raindrops fall from the sky making an angle of
\(30^\circ\) with the vertical. If a man runs at
\(2\) m/s, he finds that the drops fall vertically. If he were to run in the opposite direction with the same
speed\(,\) the raindrops will fall with a vertical speed of:
| 1. |
\(2\) m/s |
2. |
\(4\) m/s |
| 3. |
\(2\sqrt3 \) m/s |
4. |
\(4\sqrt3 \) m/s |
182. A small block of mass
\(m\) is released as shown in the figure against the vertical part of the smooth curved surface of a wedge of mass
\(M\), initially at rest. When the block reaches the bottom, it travels horizontally with a kinetic energy
\(E_m,\) while the wedge recoils towards left with a kinetic energy
\(E_M.\) Then:

| 1. |
\(\dfrac{E_m}{m}=\dfrac{E_M}{M}\) |
2. |
\(mE_m=ME_M\) |
| 3. |
\(\dfrac{E_m}{m^2}=\dfrac{E_M}{M^2}\) |
4. |
\(m^2E_m=M^2E_M\) |
183. A uniform solid hemisphere of mass
\(m\) and radius
\(R\) is rotated about an axis passing through its center
\(O\) along a diameter of its flat surface. The moment of inertia of the hemisphere, about this axis, is:
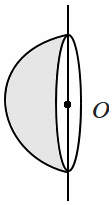
| 1. |
\(\dfrac15mR^2\) |
2. |
\(\dfrac25mR^2\) |
| 3. |
\(\dfrac13mR^2\) |
4. |
\(\dfrac23mR^2\) |
184. A body cools from \(52^\circ \text{C}\) to \(48^\circ \text{C}\) in \(6\) minutes. How much time will the same body take to cool from \(53^\circ \text{C}\) to \(47^\circ \text{C}?\) Assume cooling is linear with time.
1. \(12\) minutes
2. \(9\) minutes
3. \(8\) minutes
4. \(7\) minutes
185. All the resistances in the circuit shown below are
\(2~\Omega.\) The equivalent resistance between
\(A\) and
\(C\) is:

| 1. |
\(4~\Omega\) |
2. |
\(2~\Omega\) |
| 3. |
\(\dfrac43~\Omega\) |
4. |
\(\dfrac{10}3~\Omega\) |
Physics - Section B
186. A particle sits on the periphery of the wheel of a car, which is being driven along a straight road at a speed \(v.\) The radius of the wheel of the car is \(R.\) The instantaneous acceleration of the particle, as observed by a passenger, is:
| 1. |
\(\frac{4v^2}{R}\) |
2. |
\(\frac{2v^2}{R}\) |
| 3. |
\(\frac{v^2}{R}\) |
4. |
\(\frac{v^2}{2R}\) |
187. A wire connects two blocks of masses \(M\), \(2M\); both lying on a smooth horizontal plane.

When a force \(F_1\) is applied to \(2M\) as shown in figure 1, the wire just breaks. On the other hand, when \(F_2\) is applied to \(M\) as shown in figure 2,

the wire just breaks. Assume that the mass of the wire is negligible. Then:
| 1. |
\(F_1 =F_2\) |
| 2. |
\(F_1>F_2\) |
| 3. |
\(F_1<F_2\) |
| 4. |
Any of the above is possible |
188. Given below are two statements:
| Assertion (A): |
Insects are observed to walk on the surface of water. |
| Reason (R): |
This is due to the viscous force of water acting on the insect's legs. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
189. A graph is plotted with the \(x \text-\)axis representing the position and the \(y\text-\)axis representing the velocity of a particle undergoing SHM along a straight line. The shape of the graph is:
| 1. |
a straight line with a positive slope |
| 2. |
a straight line with a negative slope |
| 3. |
a circle |
| 4. |
an ellipse |
190. Consider a uniformly charged hemispherical shell of radius
\(R,\) carrying a surface charge of density
\(\sigma.\) The potential at a point
\(P\) on the inner rim of the hemisphere is
\(V_P,\) while that at the center is
\(V_O,\) and the potential at the mid-point (
\(A\)) of
\(OP\) is
\(V_A.\) Then:
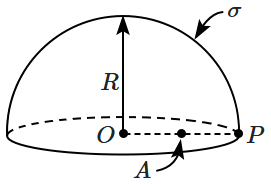
1.
\(V_O>V_A>V_P\)
2.
\(V_O<V_A<V_P\)
3.
\(V_A>V_O, V_A>V_P\)
4.
\(V_O=V_A=V_P\)
191. A current
\(i\) is distributed uniformly over the cross-section of a cylindrical wire of radius
\(R,\) as shown in the diagram. The magnetic field at the surface is
\(B_s.\) The magnetic field at the point
\(P\) inside the cross-section equals:
\(\left(OP =\dfrac{R}{2}\right )\)

| 1. |
\(\dfrac{B_s}{2}\) |
2. |
\(2 B_s\) |
| 3. |
\(\dfrac{B_s}{4}\) |
4. |
\(4 B_s\) |
192. A thin equi-convex lens of refractive index
\(1.2\) is dipped in oil of index
\(1.44.\) The lens has a power of
\(2\) D (in air). When it is immersed in the oil, the focal length of the lens becomes:
| 1. |
\(50\) cm |
2. |
\(-50\) cm |
| 3. |
\(-50/1.2\) cm |
4. |
\(-50 \times1.2\) cm |
193. The frequency of light in a photoelectric experiment is tripled. The stopping potential will:
| 1. |
be tripled |
2. |
be more than tripled |
| 3. |
be less than tripled |
4. |
become one-third |
194. A load resistance is connected to a full-wave rectifier and the peak current through the load is
\(I_0.\) The RMS current through the load resistor is:
| 1. |
\(\dfrac{I_{0}}{\sqrt{2}}\) |
2. |
\(2\left(\dfrac{I_{0}}{\sqrt{2}}\right)\) |
| 3. |
\(I_{0}\) |
4. |
None of the above |
195. A ball is released with a velocity
\((2 \hat{\imath}+2 \hat{\jmath})~ \text{m/s}\) on the rectangular pool table from the point
\((3, 0)\text{ m.}\) All the collisions of the ball are elastic.
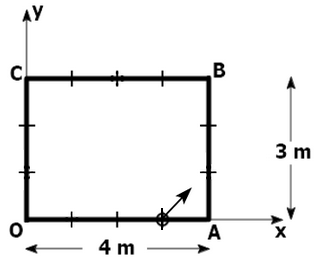
After the
\(4^{\text{th}}\) collision with the edges of the board, the location and velocity of the ball will be:
| 1. |
\((3,0)\text{ m and} ~(2 \hat{\imath}+2 \hat{\jmath}) ~\text{m/s}\) |
| 2. |
\((0,2)~ \text{m and} ~(2 \hat{\imath}-2 \hat{\jmath}) ~\text{m/s}\) |
| 3. |
\((1,0) ~\text{m and} ~(2 \hat{\imath}+2 \hat{\jmath}) ~\text{m/s}\) |
| 4. |
\((2,2)~\text{m and }(-2 \hat{\imath}-2 \hat{\jmath}) ~\text{m/s}\) |
196. The current passing through a certain device doubles when the temperature rises by
\(10^{\circ}\text{C}\), in the temperature range from
\(0^{\circ}\text{C}\) to
\(500^{\circ}\text{C}\). This effect is used to measure the temperature of a sample. The current is measured to be
\(5~\text{mA}\) at
\(15^{\circ}\text{C}\) and then the next measurement shows a current of
\(50~\text{mA}\). The temperature of the new measurement is nearly: (take
\(\text{log}_{10}2 \simeq 0.3\), if required)
| 1. |
\(150^{\circ}\text{C}\) |
2. |
\(50^{\circ}\text{C}\) |
| 3. |
\(450^{\circ}\text{C}\) |
4. |
\(25^{\circ}\text{C}\) |
197. A piece of alloy of mass
\(250~\text{g}\) (specific heat capacity
\(=0.1 \times\) that of water) is placed in a furnace and then put into a calorimeter containing
\(240~\text{g}\) of water at
\(20^\circ\text{C}.\) The water equivalent of the calorimeter is
\(10~\text{g}\). The final temperature of the mixture is
\(50^\circ\text{C}.\) The temperature of the furnace is (nearly):
| 1. |
\(250^\circ\text{C}\) |
2. |
\(350^\circ\text{C}\) |
| 3. |
\(600^\circ\text{C}\) |
4. |
\(800^\circ\text{C}\) |
198. A small thin tube is connected to two soap bubbles, one of radius
\(R\) and the other radius
\(r\) — so that the air in the two bubbles can flow through the tube. Then, air will flow:
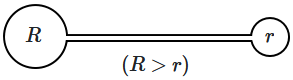
| 1. |
from the larger bubble to the smaller bubble, until the two are of equal size. |
| 2. |
from the large bubble to the smaller bubble initially, and the smaller one becomes larger; then the flow is reversed. |
| 3. |
from the smaller bubble to the larger one until the smaller bubble becomes very small. |
| 4. |
from the larger to the smaller bubble and it bursts the smaller bubble. |
199. Sinusoidal sound waves of the same frequency travelling in air along the
\(x\)-axis and the
\(y\)-axis arrive in phase with each other at the origin. Their amplitudes are equal to
\(A\) (each). The amplitude of the vibration at the origin is:

| 1. |
\(A\) |
2. |
\(\sqrt 2A\) |
| 3. |
\(2A\) |
4. |
\((2+\sqrt2)A\) |
200. An ideal gas undergoes a thermodynamic process described by the equation:
\(PV^2=C,\)
where \(C\) is a constant. The gas transitions from an initial state \((P_1, V_1, T_1)\) to a final state \((P_2, V_2, T_2).\) Which of the following statements is correct?
| 1. |
\(\text{If}~P_1>P_2,~\text{then}~T_1<T_2 \) |
| 2. |
\(\text{If}~V_2>V_1,~\text{then}~T_2>T_1\) |
| 3. |
\(\text{If}~V_2>V_1,~\text{then}~T_2<T_1\) |
| 4. |
\(\text{If}~P_1>P_2,~\text{then}~V_1>V_2\) |
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course