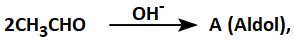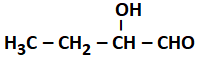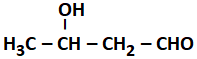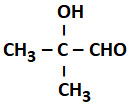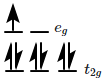Botany - Section A
1. Consider the given two statements:
| I: |
At maturity, none of the components of xylem tissue is living. |
| II: |
At maturity, none of the components of phloem tissue is dead. |
The correct statement/s is/are:
1. Only I
2. Only II
3. Both I and II
4. Neither I nor II
2. Like mitochondria, the chloroplasts:
| I: |
have their own DNA |
| II: |
have an analogous inner membrane |
1. Only I is correct
2. Only II is correct
3. Both I and II are correct
4. Both I and II are incorrect
3. Identify the correct statements:
| I: |
Cleistogamous flowers are invariably autogamous. |
| II: |
Geitonogamy is genetically similar to autogamy. |
1. Only
I
2. Only
II
3. Both
I and
II
4. Neither
I nor
II
4. A very low level of expression of lac operon is present in E.coli:
1. Only when lactose is present
2. Only when glucose is absent
3. Only when allolactose is present
4. all the time
5.
| Assertion (A): |
When constructing rDNA, the vector DNA should never be cut by the same restriction endonuclease that was used to cut the donor DNA |
| Reason (R): |
Use of same restriction endonuclease will generate same kind of sticky ends retarding the function of DNA ligase. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
6.
| Assertion (A): |
The organic farmer holds the view that eradication of pest is undesirable |
| Reason (R): |
Biocontrol is not a good strategy to control pest |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 3. |
(A) is True but (R) is False |
| 4. |
Both (A) and (R) are False |
7. Abiotic pollination is most commonly brought about by:
| 1. |
Wind |
2. |
Water |
| 3. |
Soil |
4. |
Bees |
8. In the semi-conservative scheme of DNA replication:
| I: |
The two strands separate and act as a template for the synthesis of new complementary strands. |
| II: |
After the completion of replication, each DNA molecule would have one parental and one newly synthesised strand. |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
9. In an mRNA, untranslated regions are present:
| 1. |
only at the 5’ end |
| 2. |
only at the 3’ end |
| 3. |
At both 5' -end (before start codon) and at 3' -end (after stop codon) |
| 4. |
At both 5' -end (after start codon) and at 3' -end (before stop codon) |
10. India’s share of world’s land area and global species diversity respectively is:
| 1. |
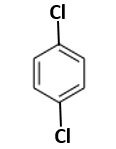
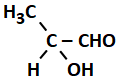
2.4 % and 8.1 % |
2. |
1.7 % and 12 % |
| 3. |
2.1 % and 9.5 % |
4. |
3.4 % and 16 % |
11. Identify the incorrect statement regarding heterotrophic bacteria?
| 1. |
They are most abundant bacteria in nature |
| 2. |
Many of them are important decomposers |
| 3. |
They play a great role in recycling nutrients like nitrogen, phosphorus, iron and sulphur |
| 4. |
They are helpful in making curd from milk, production of antibiotics, fixing nitrogen in legume roots, etc. |
12. Identify the incorrect statement:
| 1. |
Ray florets of sunflower have epigynous flowers with inferior ovary. |
| 2. |
In vexillary aestivation, keel are the two largest posterior petals. |
| 3. |
Stamens are united into one bunch in China rose. |
| 4. |
Placentation is marginal in Pea. |
13. Palisade parenchyma:
| 1. |
are prominent in isobilateral leaf |
| 2. |
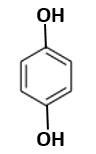
is abaxially placed in dorsi-ventral leaf |
| 3. |
are loosely arranged oval or round cells extending to the lower epidermis in dorsi-ventral leaf |
| 4. |
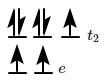
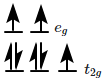
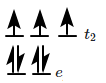
is made of elongated cells vertically arranged parallel to each other in dorsi-ventral leaf |
14. A pure tall plant is crossed with a hybrid tall plant [tall completely dominant over dwarf]. What proportion of progeny is expected to be dwarf?
1. 0
2. ½
3. ¼
4. ¾
15. The net primary productivity of oceans is about:
| 1. |
25 % of the annual net primary productivity of the whole biosphere |
| 2. |
33 % of the annual net primary productivity of the whole biosphere |
| 3. |
66 % of the annual net primary productivity of the whole biosphere |
| 4. |
70 % of the annual net primary productivity of the whole biosphere |
16. A hierarchy is an arrangement of items that are represented as being "above", "below", or "at the same level as" one another. The correct ascending order of taxonomic hierarchy is:
| 1. |
Species → Genus → Family → Order → Class → Division → Kingdom |
| 2. |
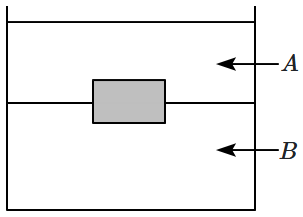
Species → Genus → Family → Class → Order → Division → Kingdom |
| 3. |
Species → Genus → Order → Family → Class → Division → Kingdom |
| 4. |
Genus → Species → Family → Order → Class → Kingdom → Division |
17. Both prokaryotic and eukaryotic cells share all the given features except:
| 1. |
presence of ribosomes |
| 2. |
arising from pre-existing cells |
| 3. |
compartmentalization through membrane bound organelles |
| 4. |
presence of a cell membrane |
18. Trichoderma polysporum is a fungal species:
1. extensively used in biocontrol of nematode pests
2. and is a source of an immunosuppressive agent
3. used for commercial production of citric acid
4. that causes ringworm in humans
19. In a dihybrid cross for seed colour and seed shape, the F₂ generation shows the phenotypic segregation of seed colour (Yellow : Green) in the ratio:
| 1. |
1 : 1 |
2. |
3 : 1 |
| 3. |
2 : 1 |
4. |
1 : 2 |
20. Name the scientist:
| I. |
He studied the tobacco mosaic virus. |
| II. |
He succeeded in extracting the virus in the form of pure crystals in 1935. |
| III. |
He was able to show that the tobacco mosaic virus is composed of protein and RNA. |
| 1. |
D. J. Ivanovsky |
2. |
W. M. Stanley |
| 3. |
M. W. Beijerinck |
4. |
Louis Pasteur |
21. In plants:
| 1. |
Both growth and differentiation are ‘open’ |
| 2. |
Root, elongating at a constant rate, exhibit geometric growth |
| 3. |
Extension growth, with the help of water, does not take place |
| 4. |
Development is independent of any external factors |
22. In both lactate fermentation and alcohol fermentation:
| 1. |
glucose is completely oxidized under aerobic conditions |
| 2. |
carbon dioxide is produced |
| 3. |
about 70 percent of energy trapped in glucose is released |
| 4. |
NADH + H+ is reoxidized to NAD+ |
23. Select the statements that will be true for C
4 plants:
| I: |
Calvin cycle takes place in Bundle sheath cells |
| II: |
Number of carbon atoms in the primary carbon dioxide acceptor is 5 |
| III: |
Initial carboxylation reaction occurs in mesophyll cells |
| IV: |
Primary carbon dioxide fixation product is oxaloacetic acid |
| 1. |
Only I, II and III |
2. |
Only I, III and IV |
| 3. |
Only II, III and IV |
4. |
I, II, III and IV |
24. Identify the incorrectly matched row:
|
Extinct animal |
Category |
Country/Continent |
| 1. |
Dodo |
Bird |
Mauritius |
| 2. |
Quagga |
Subspecies of Zebra |
India |
| 3. |
Thylacine |
Marsupial |
Australia |
| 4. |
Stellar’s sea cow |
Mammal |
Russia |
25. Which of the following pteridophytes does not belong to the same class as the other three?
| 1. |
Dryopteris |
2. |
Pteris |
| 3. |
Adiantum |
4. |
Equisetum |
26. Liliaceae:
| 1. |
is a family of dicotyledonous plants |
| 2. |
is distributed in a highly restricted rand of specific temperate climates |
| 3. |
is characterized by floral characters such as tepal, six [3+3]; stamen, six [3+3], epitepalous |
| 4. |
includes economically important plants such as sunhemp, lupin and indigofera |
27. Identify the incorrect statement regarding the secondary growth seen in plants:
| 1. |
Secondary growth does not occur in monocotyledons |
| 2. |
The cells of secondary cortex are parenchymatous |
| 3. |
Bark refers to all the tissues exterior to the vascular cambium, therefore it does not include secondary phloem |
| 4. |
In the dicot root, the vascular cambium originates from the tissue located just below the phloem bundles, a portion of pericycle tissue, above the protoxylem forming a complete and continuous wavy ring, which later becomes circular |
28. Identify the correct set of statements with regard to the properties of humus.
| (a) |
Highly resistant to microbial action |
| (b) |
Dark-colored amorphous substance |
| (c) |
End product of detritus food chain |
| (d) |
Reservoir of nutrients |
| (e) |
Undergoes decomposition very fast |
Choose the correct answer from the options given below:
1. (a), (b), and (d) only
2. (a),(b) and (e) only
3. (a) and (b) only
4. (b), (c), and (a)
29. A certain plant homozygous for yellow seeds and red flowers was crossed with a plant homozygous for green seeds and white flowers. The F
1 plants had yellow seeds and pink flowers. The F
1 plants were selfed to get F
2 progeny. Assuming independent assortment of the two characters, how many phenotypic categories are expected for these characters in the F
2 generation?
30. There are many ovules in the ovary in all the following angiosperms, except:
| 1. | orchids | 2. | paddy |
| 3. | papaya | 4. | water melon |
31. Photosynthesis is carried out by green plants most effectively in what wavelength range of light?
| 1. |
Green |
2. |
Infrared |
| 3. |
Blue and red |
4. |
Ultraviolet |
32. Identify the correct statements:
| I: |
The primary carbon dioxide acceptor in C3 plants is Phospho enol pyruvate [PEP]. |
| II: |
The primary carbon fixation product in C3 plants is Oxalo acetic acid [OAA]. |
1. Only
I
2. Only
II
3. Both
I and
II
4. Neither
I nor
II
33. A stable biological community:
| I: |
should not show too much variation in productivity from year to year |
| II: |
must be either resistant or resilient to occasional disturbances (natural or man-made) |
| III: |
must also be resistant to invasions by alien species |
1. Only
I and
II are correct
2. Only
I and
III are correct
3. Only
II and
III are correct
4.
I,
II and
III are correct
34. What caused the extinction of Steller’s sea cow and passenger pigeon?
1. Habitat loss and fragmentation
2. Over exploitation
3. Invasion by alien species
4. Co-extinction
35. The historic Convention on Biological Diversity (‘The Earth Summit’) was held in Rio de Janeiro in:
| 1. |
1986 |
2. |
1987 |
| 3. |
1992 |
4. |
2002 |
Botany - Section B
36. Electrons carried by NADH enter the electron transport system when they are transferred to:
| 1. |
FMN |
2. |
Ubiquinone |
| 3. |
Fe-S |
4. |
FAD |
37. In the electron transport system in the mitochondria, oxygen acts as a:
I: final acceptor of electrons
II: final acceptor of protons
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
38. Identify the correct statements:
| I: |
In castor and maize, autogamy is prevented but not geitonogamy |
| II. |
In papaya, both autogamy and geitonogamy are prevented |
| 1. |
Both I and II |
2. |
Only I |
| 3. |
Only II |
4. |
Neither I nor II |
39. In pea seeds, starch synthesis is controlled by one gene with two alleles [B/b]. A pea seed with genotype Bb will be:
1. Round and Large
2. Wrinkled and large
3. Round and intermediate in size
4. Round and small in size
40. The molecule shown in the given figure:

| I: |
serve as substrate for DNA replication |
| II: |
provide energy for polymerisation reaction |
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
41. How is a prion different from viroids, viruses, bacteria, fungi, and parasites?
1. It is non-infectious
2. It is obligate intracellular parasite
3. It is devoid of nucleic acids
4. It is a low molecular weight RNA molecule
42. In a cross AaBb X aabb [assuming independent assortment], what fraction of progeny are expected to be AaBb?
1. ¼
2. 1/3
3. ½
4. 2/3
43. Consider the two statements:
| I: |
Current availability of carbon dioxide levels is limiting to the C4 plants. |
| II: |
C3 plants have a much lower temperature optimum than C4 plants. |
| 1. |
Only I is correct |
2. |
Only II is correct |
| 3. |
Both I and II are correct |
4. |
Both I and II are incorrect |
44. The ratio of the volume of CO
2 evolved to the volume of O
2 consumed in respiration is called as the respiratory quotient [RQ]. Calculate RQ when albumin is used as a respiratory substrate[see the given equation]:
C
77H
112N
2O
22S + 77O
2 → 63 CO
2 + 38 H
2O + SO
2 + 9 CO(NH
2)
2
| 1. |
0.70 |
2. |
0.82 |
| 3. |
0.90 |
4. |
1.00 |
45. China rose:
1. belongs to Brassicaceae
2. has parietal placentation
3. has monoadelphous stamen
4. has valvate aestivation
46. The sporophyte of mosses is more elaborate than that in:
| 1. |
Pteridophytes |
2. |
Gymnosperms |
| 3. |
Angiosperms |
4. |
Liverworts |
47. What provides the electrons needed to replace those removed from photosystem II during non cyclic photophosphorylation?
| 1. |
PS I |
2. |
Oxygen |
| 3. |
Water |
4. |
Carbon dioxide |
48. As a method of cellular defence, the RNA interference takes place in all:
| 1. |
nematodes |
2. |
bacteria and archaea |
| 3. |
eukaryotic cells |
4. |
plant cells |
49. The process of individuals of the same species that have come into the habitat from elsewhere during the time period under consideration is referred as:
1. Association
2. Emigration
3. Competition
4. Immigration
50. Auxins:
I: promote flowering in tomatoes.
II: induce parthenocarpy in pineapples.
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
Zoology - Section A
51. Consider the two statements
| I: |
Sertoli cells divide meiotically to produce sperms |
| II: |
Leydic cells secrete androgens when stimulated by FSH |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
52. Identify the incorrect statement:
| 1. |
Zinc is a cofactor for carboxypeptidase |
| 2. |
NADP contains the vitamin cyanocobalamin |
| 3. |
Malonate is a competitive inhibitor of Succinic dehydrogenase |
| 4. |
Haem is the prosthetic group in peroxidase |
53. Which of the following statements is not correct?
| 1. |
Every 100 ml of oxygenated blood can deliver around 5 ml of oxygen to the tissue under normal physiological conditions. |
| 2. |
Oxygen gas has most potent effect on the central chemoreceptors and plays most vital role in regulation of respiration |
| 3. |
Nearly 70 percent of carbon dioxide is carried as bicarbonate in the blood. |
| 4. |
Every 100 ml of deoxygenated blood delivers about 4 ml of carbon dioxide to the alveoli. |
54. Identify A, B, C and D in the diagrammatic representation of reflex action, showing knee jerk reflex:

|
A |
B |
C |
D |
| 1. |
Muscle spindle |
Motor end plate |
Afferent pathway |
Efferent pathway |
| 2. |
Muscle spindle |
Motor end plate |
Efferent pathway |
Afferent pathway |
| 3 |
Motor end plate |
Muscle spindle |
Efferent pathway |
Afferent pathway |
| 4. |
Motor end plate |
Muscle spindle |
Afferent pathway |
Efferent pathway |
55. Identify the incorrectly matched pair:
| 1. |
Acromegaly |
Excess of growth hormone in adulthood |
| 2. |
Diabetes insipidus |
Deficiency of insulin |
| 3. |
Graves’ disease |
Hyperthyroidism |
| 4. |
Addison’s disease |
Underproduction of adrenal cortex hormones |
56. Identify the incorrect statement:
| 1. |
Glomerulus is a tuft of capillaries formed by afferent arteriole. |
| 2. |
Blood from the glomerulus is carried away by an efferent arteriole. |
| 3. |
Vasa recta is well developed in cortical nephrons. |
| 4. |
The DCTs of many nephrons open into a straight tube called collecting duct. |
57. Study the given diagram regarding the alimentary canal of cockroach and select the correct statements:

| I: |
A is used for storing food |
| II: |
B helps in grinding the food particles |
| III: |
C help in osmoregulation |
| IV: |
D secretes digestive enzymes |
1. Only
I and
II
2. Only
I,
III and
IV
3. Only
II
4. Only
III and
IV
58. Triploblastic, bilaterally symmetrical animals with true coelom but no segmentation are:
| 1. |
Aschelminthes |
2. |
Molluscs |
| 3. |
Annelids |
4. |
Chordates |
59. Thrombokinase:
| 1. |
is an enzyme formed in and secreted by the platelets |
| 2. |
is an enzyme complex required to convert prothrombin into thrombin |
| 3. |
dissolves the intravascular clots |
| 4. |
prevents coagulation of the blood |
60. Who amongst the following was an American scientist?
1. J. B. S. Haldane
2. Alexander Oparin
3. Charles Darwin
4. Stanley L. Miller
61. The polymerase chain reaction:
| I: |
is used to synthesize multiple copies of the gene [or DNA] in vitro. |
| II: |
uses a thermostable DNA dependent DNA polymerase. |
| III: |
is now routinely used to detect HIV in suspected AIDS patient. |
| IV: |
is being used to detect mutations in genes in suspected cancer patients. |
| 1. |
All except I are true |
2. |
Only II and III are true |
| 3. |
Only IV is not true |
4. |
I, II, III and IV are true |
62. Which of the following is not correct regarding Variable Number of Tandem Repeats in a DNA sample?
| 1. |
Show very high degree of polymorphism |
| 2. |
Belong to a class of satellite DNA referred to as mini-satellites |
| 3. |
Vary in size from 100 kb to 1000 kb |
| 4. |
Are used in a technique of DNA fingerprinting |
63. Predators play very important roles in an ecosystem that include:
| I: |
They act as conduit of energy |
| II: |
They keep prey population under check |
| III: |
They maintain species diversity in a community |
1. Only
I and
II
2. Only
I and
III
3. Only
II and
III
4.
I,
II and
III
64. Which of the following will not be seen in any Cnidarian?
1. Metagenesis
2. Cnodicytes
3. Bilateral symmetry in larva
4. Tentacles
65. Intercalated discs are:
| 1. |
flattened interconnected smooth ER in muscle fibre |
| 2. |
communication junctions between cardiac muscle cells |
| 3. |
cell junctions that hold smooth muscle cells together in a unit |
| 4. |
dorsoventrally flattened mitochondria in a skeletal muscle cell |
66. Which of the following is not a lipid?
| 1. |
Palmitic acid |
2. |
Glutamic acid |
| 3. |
Cholesterol |
4. |
Lecithin |
67. Identify the stage of meiotic cell division, which is followed by cytokinesis, leading to the formation of a dyad of cells:
| 1. |
Anaphase I |
2. |
Telophase I |
| 3. |
Anaphase II |
4. |
Telophase II |
68. The partial pressure of oxygen is expected to be least in:
1. Atmospheric air
2. Alveoli
3. Oxygenated blood
4. Tissues
69. Identify the incorrect statement:
| 1. |
Mollusks and arthropods have closed circulatory system |
| 2. |
All vertebrates possess a muscular chambered heart |
| 3. |
In fishes, the heart pumps out deoxygenated blood |
| 4. |
The left atrium receives oxygenated blood in amphibians and reptiles |
70. Regarding the human kidney:
| 1. |
glomerulus is a tuft of capillaries formed by the efferent arteriole |
| 2. |
vasa recta is highly reduced in juxtamedullary nephrons |
| 3. |
DCTs of many nephrons open into a straight tube called collecting duct |
| 4. |
columns of Bertini are extensions of medulla into the cortex |
71. Osteoporosis, commonly seen in post menopausal women, is commonly caused by:
1. low levels of calcium ions in blood
2. decreased levels of oestrogens
3. autoimmune destruction of joints and cartilages
4. accumulation of uric acid crystals
72. Each time the sodium-potassium pump works, it pumps:
1. the anions as well with sodium to the outside
2. the anions as well with potassium to the inside
3. 3 sodium ions outwards
4. 3 potassium ions inwards
73. During oogenesis:
| 1. |
meiosis I is completed before birth |
| 2. |
meiosis I, when completed results in equal sized daughter cells |
| 3. |
meiosis II results in the unequal sized cells |
| 4. |
meiosis II is completed just before ovulation |
74. Thermus aquaticus is the source bacterium of:
1. a heat-resistant enzyme used in PCR
2. an acid-resistant enzyme used in biological detergents
3. a reporter enzyme used in recombinant procedures
4. a key enzyme involved in ELISA
75. With the help of ELISA, infection by pathogen can be detected by the presence in a sample of:
I. antigens of the pathogen
II. antibodies synthesized against the pathogen
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
76. Both AIDS and Hepatitis B infections in humans:
| 1. |
are acute fatal infections |
| 2. |
are easily cured if detected early |
| 3. |
can be transmitted by a mosquito vector |
| 4. |
can be transmitted through sexual contact or infected blood |
77. Identify the incorrect statement regarding allergy:
| 1. |
It is an exaggerated response of the immune system |
| 2. |
Antibodies produced in response to allergens are of IgA type |
| 3. |
It can be caused by release of chemicals like histamine by mast cells |
| 4. |
Steroids can quickly reduce the symptoms of allergy |
78. The lineage Synapsids → Pelycosaurs → Therapsids evolved into:
| 1. |
Crocodiles |
2. |
Turtles |
| 3. |
Birds |
4. |
Mammals |
79. All the following are examples of genetic disorders with aneuploidy except:
1. Down’s syndrome
2. Cushing’s syndrome
3. Klinefelter syndrome
4. Turner’s syndrome
80. Which of the following hormones does not generate second messenger in their target cells?
1. Insulin
2. Parathyroid hormone
3. Triiodothyronine
4. Adrenaline
81. Identify the incorrectly matched pair:
| 1. |
Golgi apparatus |
Formation of glycoproteins and glycolipids |
| 2. |
Rough ER |
Synthesis of steroidal hormones in animal cells |
| 3. |
Lysosomes |
Carry acid hydrolases |
| 4. |
Nucelolus |
Site for active rRNA synthesis |
82. Which of the following is the correct equation of exponential growth?
1. \(N_t = N_0 ~e^{rnt}\)
2. \(N_t = N_0 ~e^{rpt}\)
3. \(N_t = N_0 ~e^{rst}\)
4. \(N_t = N_0 ~e^{rt}\)
83. Toxoplasma, an organism commonly causing opportunistic infection in AIDS patients is a/an:
1. autotrophic bacterium
2. fungus
3. protozoan
4. helminth
84. All the following functions will be carried out by adrenaline except:
1. an increase in heart rate and strength of heart beat
2. pupil dilation
3. stimulation of gastrointestinal activity
4. increased blood glucose level
85. Identify the correct statement:
1. Pineal is stimulated by bright light
2. Pineal secretes melanin
3. Pineal is present in the mediastinum
4. Pineal plays a very important role in diurnal rhythm
Zoology - Section B
86. What would be a normal finding on semen analysis of a normal fertile male?
| I: |
A count of 10 million sperms per ml of semen |
| II: |
60 percent sperms have normal shape and size |
| III: |
40 percent sperms show vigorous motility |
| 1. |
Only II |
2. |
Only I and II |
| 3. |
Only II and III |
4. |
I, II and III |
87. Although estrogen levels vary through the menstrual cycle, the highest level will be seen:
| 1. |
in the middle of the follicular phase after the appearance of LH. |
| 2. |
near the end of the follicular phase just before ovulation. |
| 3. |
in the middle of the secretory phase when the corpus luteum is most developed. |
| 4. |
near the end of the secretory phase just before the onset of the next menstrual cycle. |
88. Identify the incorrect statement:
| 1. |
CDRI, located at Lucknow, has developed ‘Saheli’ – a non-steroidal oral contraceptive pill |
| 2. |
Amniocentesis is statutorily banned in India for Pre Natal Sex Determination |
| 3. |
An ideal contraceptive should both be effective and reversible |
| 4. |
Increasing birth rate is the main cause of the increase in the population of India today |
89. The economist whose book ‘Essay on the Principle of Populations’ influenced Darwin was:
1. Georges Cuvier
2. Alfred Lyell
3. T. R. Malthus
4. A. R. Wallace
90. The medulla oblongata contains centres which control all the following except:
| 1. |
respiration |
2. |
cardiovascular reflexes |
| 3. |
gastric secretions |
4. |
body temperature |
91. In Chordates:
| I: |
Heart is dorsal |
| II: |
Central nervous system is ventral |
| III: |
A post-anal tail is present |
1. Only
I and
II are correct
2. Only
III is correct
3. Only
II is correct
4. Only
I and
III are correct
92. Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R):
| Assertion (A): |
Oxytocin helps in parturition and causes milk ejection from the mammary glands. |
| Reason (R): |
Oxytocin acts on the smooth muscles of our body and stimulates their contraction. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
93. What is correct regarding the atrial natriuretic factor?
| 1. |
It causes vasodilation and reduces blood pressure |
| 2. |
It acts on the atrial wall of our heart and reduces cardiac output |
| 3. |
It stimulates angiotensin leading to vasoconstriction |
| 4. |
It acts on the distal tubule and lead to diuresis. |
94. What can act as a competitive inhibitor of enzyme Succinic dehydrogenase?
| 1. | ATP | 2. | FADH2 |
| 3. | Malonate | 4. | Pyruvate |
95. At 40 mm Hg partial pressure of oxygen, the percentage saturation of haemoglobin with oxygen is expected to be about:
1. 50 %
2. 75 %
3. 95 %
4. 99 %
96. Given below are two statements: one is labelled as
Assertion (A) and the other is labelled as
Reason (R):
| Assertion (A): |
The sino-atrial node is normally the pacemaker of the human heart. |
| Reason (R): |
It is the only auto-excitable part of the nodal tissue of the human heart. |
In the light of the above statements choose the correct answer from the options given below:
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
97. What is true regarding Downstream Processing?
| I: |
This stage is after the biosynthetic stage. |
| II: |
The processes include separation and purification. |
| 1. |
Only I |
2. |
Only II |
| 3. |
Both I and II |
4. |
Neither I nor II |
98. Which of the following bones is not a part of human cranium?
| 1. |
Sphenoid |
2. |
Maxilla |
| 3. |
Ethmoid |
4. |
Occipital |
99. Which of the following is not true regarding spleen?
| 1. |
It is a primary lymphoid organ. |
| 2. |
It has a large reservoir of erythrocytes. |
| 3. |
It mainly contains lymphocytes and phagocytes. |
| 4. |
It is a large bean shaped organ in the left upper quadrant of the abdomen. |
100. The ratios between Adenine and Thymine and Guanine and Cytosine are constant and equal one for a:
| 1. |
ss RNA |
2. |
dsRNA |
| 3. |
dsDNA |
4. |
ssDNA |
Chemistry - Section A
101. An organic compound ‘
X’ on treatment with pyridinium chloro chromate in dicholoromethane gives compound ‘
Y’.
Compound ‘
Y’ reacts with I
2 and alkali to form triiodomethane.
The compound ‘
X’ is:
1. C2H5OH
102. Al2O3 is reduced by electrolysis at low potentials and high currents. If 4.0 x 104 A of current is passed through molten Al2O3 for 6 hours, the mass of aluminum produced is:
(Assume 100 % current efficiency, the atomic mass of Al = 27 g mol-1)
| 1. |
9.0 x 103 g |
2. |
8.1 x 104 g |
| 3. |
2.4 x 105 g |
4. |
1.3 x 104 g |
103. Match the items of column I with items of column II and mark the correct option:
|
Column I
(Reaction) |
|
Column II
(Reagents and uses) |
| A. |
Kolbe's reaction |
1. |
Heated copper at 573 K |
| B. |
Reimer-Tiemann reaction |
2. |
Conversion of phenol to salicylic acid |
| C. |
Williamson's synthesis |
3. |
Conversion of phenol to salicylaldehyde |
| D. |
Conversion of alcohol to ketone |
4. |
Reaction of alkyl halide with sodium alkoxide |
1. A-3 , B-1 , C-4 , D-2
2. A -2 , B-3, C-4 , D-1
3. A-4 , B-3 , C-2 , D-1
4. A-1 , B-2, C-3 , D-4
104. Given below are two statements:
| Assertion (A): |
Mixture of nitric acid and water forms maximum boiling azeotropes. |
| Reason (R): |
Interaction between nitric acid and water is stronger than the interaction
between nitric acid and nitric acid or water and water. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
106. The chain growth of a silicone is stopped by adding:
1. Me3SiCl
2. Me2SiCl2
3. MeSiCl3
4. SiCl4
107. When a 10-ampere current is passed through acidulated water (H2O containing some H2SO4 ), 2.24 litres H2 gas is evolved at NTP. The duration for which current is passed through acidulated water is:
1. 965 seconds
2. 1930 seconds
3. 3860 seconds
4. 7720 seconds
108. Which molecule is not produced during either partial or complete hydrolysis of XeF
6?
| 1. |
XeOF2 |
2. |
XeOF4 |
| 3. |
XeO2F2 |
4. |
XeO3 |
109. Which of the following species is both tetrahedral and diamagnetic?
| 1. |
\(\left[\mathrm{Ni}(\mathrm{CN})_{4}\right]^{2-} \) |
2. |
\(\left[\mathrm{Ni}(\mathrm{CO})_{4}\right] \) |
| 3. |
\(\left[\mathrm{NiCl}_{4}\right]^{2-}\) |
4. |
\(\left[\mathrm{Cu}\left(\mathrm{NH}_{3}\right)_{4}\right]^{2+}\) |
110. The reason behind formation of isocyanide in the reaction between alkyl halide and AgCN, is:
| 1. |
AgCN is an electrovalent compound |
| 2. |
AgCN is a covalent compound |
| 3. |
In -CN, lone pair of e-s is present on carbon only |
| 4. |
None of the above |
111. Case study
Aldol Condensation
When two carbonyl compounds having \(\alpha-\)hydrogen atoms are condensed in presence of dilute alkali to form a compound that has the properties of alcohol as well as carbonyl compound, the compound is known as ALDOL. Aldol is unstable and on heating, it eliminates one H2O molecule and is converted into \(\alpha , \beta-\) unsaturated aldehyde or ketone
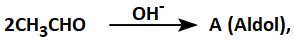
Compound 'A' is:
112. The molarity of 28% KOH solution (d = 1.2 g/ml) is:
1. 3 M
2. 6 M
3. 9 M
4. 12 M
113. [Cr(NH
3)
5Br]SO
4 and [Cr(NH
3)
5(SO
4)]Br are:
| 1. |
Geometrical isomers |
2. |
Ionisation Isomers |
| 3. |
Linkage isomers |
4. |
Optical isomers |
114. But-1-en-3-yne contains:
| 1. |
6σ and 3π bonds |
2. |
7σ and 2π bonds |
| 3. |
7σ and 3π bonds |
4. |
5σ and 3π bonds |
115. For a given reaction scheme;
\(x \mathrm{Cl}_{2}+y~\mathrm{NaOH} \longrightarrow \mathrm{zNaCl}+a~\mathrm{} \mathrm{NaClO}_{3} + bH_2O\)
The ratio of
\({ x + y \over z + a+b}\)will be:
| 1. |
One (1) |
2. |
Two (2) |
| 3. |
Five (5) |
4. |
Three (3) |
116. The molecular formula C
4H
10O can show:
| 1. |
Chain isomerism |
2. |
Position isomerism |
| 3. |
Metamerism |
4. |
All of these |
117.
| Assertion (A): |
The boiling point of neopentane is less than that of isopentane |
| Reason (R): |
The higher the branching, the lower the boiling point of a compound. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
118. Which of the following is not a neutral oxide?
1. NO
2. NO2
3. N2O
4. CO
119. When \(K_2Cr_2O_7\) is reacted with \(SnCl_2\) in presence of \(HCl\) , then the reduced product of \(K_2Cr_2O_7\) and oxidized product of \(SnCl_2\) , respectively, are:
1. \([CrCl_4]^- ~~~\text{and} ~~~Cl_2~\)
2. \(CrCl_3 ~~~\text{and} ~~~Cl_2~\)
3. \(CrCl_3 ~~~\text{and} ~~~SnCl_4~\)
4. \(CrCl_6 ~~~\text{and} ~~~SnCl_4~\)
120. In the Lucas test for alcohols, which intermediate is formed?
| 1. |
Carbocation |
2. |
Carbanion |
| 3. |
Free Radical |
4. |
No intermediate |
121. When \(HCHO \) and \(C_6H_5CHO\) are reacted in the presence of 50% \(NaOH\), then the products formed are:
1. \(HCOONa ~~\text{and} ~~ C_6H_5COONa\)
2. \(CH_3OH ~~\text{and} ~~ C_6H_5CH_2OH \)
3. \(HCOONa ~~\text{and} ~~C_6H_5CH_2OH\)
4. \(CH_3OH ~~\text{and} ~~ C_6H_5COONa\)
122. The non-reducing sugar amongst the following is:
1. Glucose
2. Fructose
3. Sucrose
4. Mannose
123. The molarity of 40% \(NaOH \) (by mass) is:
(d = 1.2 g/ml)
1. 10 M
2. 12 M
3. 5 M
4. 6 M
124. The magnetic moment of \(Ni^{2+}\) (Z=28) is:
1. 1.732 BM
2. 2.82 BM
3. 3.87 BM
4. 4.89 BM
125. The most acidic oxide amongst the following is
| 1. |
NO |
2. |
\(N_2O\) |
| 3. |
\(NO_2\) |
4. |
\(N_2O_5\) |
126. Which of the following is not a state function?
| 1. |
H (Enthalpy) |
2. |
Q (Heat) |
| 3. |
S (Entropy) |
4. |
E (Internal Energy) |
127. For the reaction
\(CO_2 (g) \rightleftharpoons CO(g) + { 1 \over 2}O_2(g) ,\) the value of
\(K_P \over K_C\) will be:
| 1. |
RT |
2. |
\(\sqrt {RT}\) |
| 3. |
\({1 \over RT}\) |
4. |
\({ 1 \over \sqrt {RT}}\) |
128. An amine reacts with benzene sulphonyl chloride to form solid compound which is soluble in alkali. The amine may be:
1. \(\mathrm{(CH_3)_2NH}\)
2. \(\mathrm{(CH_3)_3N}\)
3. \(\mathrm{CH_3 - CH_2 - NH_2}\)
4. \(\mathrm{CH_3 - NH - CH_2 - CH_3}\)
129. For the reaction, \(N_2 + 3H_2 \rightarrow 2NH_3\), the rate of disappearance of \(H_2\) is \(6 \times 10^{-4}\) mole litre-1 min-1. The rate of appearance of \(NH_3\) will be:
1. \(2 \times 10^{-4}~ mole ~litre^{-1} ~min^{-1}\)
2. \(4 \times 10^{-4}~ mole ~litre^{-1} ~min^{-1}\)
3. \(6 \times 10^{-4}~ mole ~litre^{-1} ~min^{-1}\)
4. \(3 \times 10^{-4}~ mole ~litre^{-1} ~min^{-1}\)
130.
| I: |
The product of the reaction of phenol with bromine depends on the nature of the solvent. |
| II: |
The reaction of phenol with bromine in CHCl3 gives a monosubstituted bromo derivative whereas the reaction of phenol with bromine water yields a trisubstituted bromo derivative of phenol. |
In light of the above statements, choose the most appropriate answer from the options given below:
| 1. |
I is correct and II is incorrect |
| 2. |
I is incorrect and II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
131. The oxidation state of cobalt in
\([Co(en)_2FBr]Cl \) is:
132. The number of moles of hydrogen gas liberated when 39 g of potassium reacts with 7.8 g of water is:
1. 0.22 mol
2. 0.43 mol
3. 0.50 mol
4. 1.0 mol
133. Match the molecules in
List-I with their corresponding bond angles in
List-II and choose the correct option.
|
List-I
(Molecule) |
|
List-II
(Bond Angle) |
| A. |
H2O |
(I) |
\(107.8^{\circ}\) |
| B. |
NH3 |
(II) |
\(93.6^{\circ}\) |
| C. |
CH4 |
(III) |
\(104^{\circ}\) |
| D. |
PH3 |
(IV) |
\(109.5^{\circ}\) |
1. A ‐ III, B ‐ I, C ‐ IV, D ‐ II
2. A ‐ III, B ‐ I, C ‐ II, D ‐ IV
3. A ‐ III, B ‐ II, C ‐ IV, D ‐ I
4. A ‐ II, B ‐ I, C ‐ IV, D ‐ III
134. Identify the correct energy diagram that shows the electron distribution according to the crystal field model of 3d-electrons in
\(\left[\mathrm{CoCl}_4\right]^{2-} \).
135. Consider the given two statements:
| Assertion (A): |
Tropics have greater biodiversity than other regions on Earth. |
| Reason (R): |
Tropics are subjected to larger seasonal variations than other regions on Earth. |
| 1. |
Both (A) and (R) are True and (R) correctly explains (A). |
| 2. |
(A) is True but (R) is False |
| 3. |
(A) is False but (R) is True |
| 4. |
Both (A) and (R) are True but (R) does not correctly explain (A). |
Chemistry - Section B
136. The volume occupied by ten molecules of water (density 1g cm-3 ) is:
| 1. |
18 cm3 |
2. |
22400 cm3 |
| 3. |
6.023 \(\times\) 10-23 cm3 |
4. |
3.0 \(\times\) 10-22 cm3 |
137. Among isomeric pentanes, which of the following has the highest melting point and the lowest boiling point?
1. n-Pentane
2. Isopentane
3. Neopentane
4. None of the above
138. How many cyclic isomers are possible for the molecular formula C5H10?
1. 2
2. 3
3. 4
4. 5
139. A volatile solute (A) is mixed with a volatile solvent (B). For this solution, vapour pressure is represented as P = 125 X
A + 45 torr (X
A = Mole fraction of A in liquid phase) The vapour pressures of A and B in a pure state, respectively, are:
| 1. |
125 torr and 45 torr |
2. |
170 torr and 45 torr |
| 3. |
125 torr and 170 torr |
4. |
45 torr and 170 torr |
140. When an electrolytic solution is diluted, then the incorrect statement from the following is:
1. Specific conductivity increases
2. Molar conductivity increases
3. Equivalent conductivity increases
4. Specific conductivity decreases
141. The reaction that does not produce nitrogen is:
1. Heating of NH4NO2
2. Heating of (NH4)2Cr2O7
3. Reaction of excess of NH3 with Cl2
4. Heating of NH4NO3
142. In the first step, in esterification reaction, H+ attacks on:
1. Oxygen atom of an alcohol
2. Doubly bonded oxygen of carboxylic acid
3. Singly bonded oxygen of carboxylic acid
4. Any oxygen atom
143. When phenol is reacted with concentrated HNO
3 and concentrated H
2SO
4, 2,4,6-trinitrophenol is formed. The correct statement from the following with respect to this reaction is:
| 1. |
Firstly we added concentrated H2SO4 and then we added concentrated HNO3 |
| 2. |
Firstly we added concentrated HNO3 and then we added concentrated H2SO4 |
| 3. |
Both concentrated HNO3 and concentrated H2SO4 are added together |
| 4. |
Here, concentrated H2SO4 acts as dehydrating agent |
144. The correct order of reactivity, amongst the given arrangements, towards nucleophilic addition, is:
1. \(\mathrm{{HCHO}>{CH}_{3} {CHO}_{}>{CH}_{3} {COCH}_{3}>{C_6H}_{5} {CHO}_{}}\)
2. \(\mathrm{{C_6H}_{5} {CHO}>{HCHO}>{CH}_{3} {CHO}_{}>{CH}_{3} {COCH}_{3}}\)
3. \(\mathrm{{HCHO}>{CH}_{3} {CHO}>{C_6H}_{5} {CHO}>{CH}_{3} {COCH}_{3}}\)
4. \(\mathrm{{HCHO}>{C}_{6} {H}_{5} {CHO}_{}>{CH}_{3} {CHO}>{CH}_{3} {COCH_3}}\)
145. In which complex, central atom is dsp2 hybridised and paramagnetic?
1. [NiCl4]2-
2. [Ni(CN)4]2-
3. [Cu(NH3)4]2+
4. [PtCl4]2-
146. The heats of combustion of C, H
2, and C
2H
6 are -94 Kcal, -68 Kcal, and -372 Kcal, respectively. The enthalpy of the formation of C
2H
6 will be:
| 1. |
+20 K cal |
2. |
-40 K cal |
| 3. |
-20 K cal |
4. |
+40 K cal |
147. pH of
\(10^{-7} ~M~NaOH\) solution at 25°C will be:
| 1. |
7 |
2. |
6.7 |
| 3. |
7.3 |
4. |
8.3 |
148. The maximum value of the rate constant can be achieved by:
| 1. |
Decreasing the activation energy to zero |
| 2. |
Increasing the temperature up to infinity |
| 3. |
Both 1 and 2 |
| 4. |
Decreasing the Arrhenius constant |
149. Which one of the following ions has highest hydration energy?
| 1. |
\(Mg^{2+} \) |
2. |
\(Li^+ \) |
| 3. |
\(Be^{2+} \) |
4. |
\(Na^+ \) |
150. How many alcohol structural isomers are possible of molecular formula
\(C_4H_{10}O\) ?
Physics - Section A
151. A block of mass
\(m\) slides down a smooth plane inclined at an angle of
\(60^\circ\) with the horizontal. The normal reaction of the incline acting on the block equals:
| 1. |
\(mg\sin60^\circ\) |
2. |
\(mg\cos60^\circ\) |
| 3. |
\(mg\tan60^\circ\) |
4. |
\(mg\cot60^\circ\) |
152. Two wires \((A, B)\) of identical lengths have breaking stresses in the ratio \(1:2,\) while their cross-sectional areas are the same. When a block of mass \(M\) is placed on the horizontal light rod, it is observed that wire \(A\) breaks if \(M\) is placed slightly to the left, while \(B\) breaks if \(M\) is placed slightly to right.

Then,
\(\dfrac{x_1}{x_2}=\)
| 1. |
\(\dfrac12\) |
2. |
\(\dfrac14\) |
| 3. |
\(\dfrac21\) |
4. |
\(\dfrac41\) |
153. A metal block floats at the interface of two liquids with \(\frac13\) of its volume in the upper liquid \((A)\) and \(\frac23\) in the lower liquid \((B).\) The densities of the metal, liquid \(A,\) and liquid \(B\) are \(\rho,~ \rho_{\Large_A}~, \rho_{\Large_B}\) respectively. Then:
| 1. |
\(\rho_{\Large_B}=2\rho_{\Large_A}\) |
2. |
\(\rho=\frac13\rho_{\Large_A}+\frac23\rho_{\Large_B}\) |
| 3. |
\(\rho=\frac23\rho_{\Large_A}+\frac13\rho_{\Large_B}\) |
4. |
\(\rho=\frac13\sqrt{\rho_{\Large_A}\rho_{\Large_B}}\) |
154. The temperature at which the Celsius and Fahrenheit thermometers agree (to give the same numerical value) is:
| 1. |
\(-40^\circ\) |
2. |
\(40^\circ\) |
| 3. |
\(0^\circ\) |
4. |
\(50^\circ\) |
155. Two metallic plates are placed parallel to each other, at a separation \(d.\) A dielectric (\(K=2\)) of width \(d/3\) is inserted into space between the plates, parallel to plates: the separation between the plates being \(d.\) The capacitance of the plates:
1. increases by \(20\)%
2. decreases by \(20\)%
3. increases by \(33\)%
4. decreases by \(25\)%
156. \(N\) capacitances, \(C\) each, are available to be connected in series or in parallel. The ratio of the minimum \((C_\text{min})\) and maximum \((C_\text{max})\) capacitance that can be formed from these is \(\dfrac{C_\text{max}}{C_\text{min}}=\)
1. \(N\)
2. \(N^2\)
3. \(N^3\)
4. \(\sqrt N\)
157. When a particle of charge \(q\) and mass \(m\) is projected perpendicular to a magnetic field, it moves in a circle of radius \(r.\) When the particle is projected upward with the same kinetic energy in a uniform gravitational field \((g)\), it rises to a height \(h\). The magnetic field is:
| 1. |
\(\dfrac{m}{q r} \sqrt{\dfrac{g h}{2}}\) |
2. |
\(\dfrac{2m}{q r} \sqrt{\dfrac{g h}{2}}\) |
| 3. |
\(\dfrac{m}{2q r} \sqrt{\dfrac{g h}{2}}\) |
4. |
none of the above. |
158. The magnetic field, at a point
\(10\) cm away, from a short bar magnet is
\(3 \times 10^{-4}\) T, when the magnet is placed in an end-on position. If the magnet is in a broadside-on position, the field will be:
| 1. |
\(6 \times 10^{-4}\) T |
2. |
\(1.5 \times 10^{-4}\) T |
| 3. |
\(3 \sqrt2 \times 10^{-4}\) T |
4. |
\({\dfrac 3 {\sqrt 2}}\times 10^{-4}\) T |
159. An inductor
\((L)\) and a resistor
\((R)\) are connected in series across a battery of emf
\(E,\) and the circuit is switched on. The current rises steadily. The rate of increase of the current
\(\left(\text{i.e.,}\dfrac {di} {dt}\right),\) when the voltage drops across the resistor is
\(\dfrac{E}{2}\), is given by:
\(\dfrac {di} {dt}\) =
| 1. |
\(\dfrac{E}{L}\) |
2. |
\(\dfrac{E}{2L}\) |
| 3. |
\(\dfrac{2E}{L}\) |
4. |
\(\dfrac{E}{L}e^{-1}\) |
160. A transformer has
\(100\) turns in its primary. It has two secondary circuits: one with
\(10\) turns and the other with
\(20\) turns. The RMS voltage across the primary is
\(30~\text{V}.\) The secondaries are connected to
\(10~\Omega\) loads, as shown. Assuming no power loss, the RMS current in the primary is:

1.
\(0.2~\text{A}\)
2.
\(0.15~\text{A}\)
3.
\(0.45~\text{A}\)
4.
\(0.9~\text{A}\)
161. The maximum electric field of a plane electromagnetic wave traveling through a vacuum is \(300~\text{V/m}.\) The maximum magnetic field of this wave is:
1. \(300~\text{T}\)
2. \(10^{-6}~\text{T}\)
3. \(9 \times 10^{10}~\text{T}\)
4. \(300\sqrt {2}~\text{T}\)
162. The Sun has a diameter of \(1.4\times10^6~\text{km}\) and is at a distance of \(150\times10^6~\text{km}\) from the Earth. An image of the Sun is formed by a convex lens of focal length \(30~\text{cm}.\) The diameter of the image is:
1. \(2.8~\text{cm}\)
2. \(2.8~\text{mm}\)
3. \(1.4~\text{mm}\)
4. \(0.7~\text{mm}\)
163. In Young's double-slit experiment conducted with the light of an unknown wavelength, it is found that the fringe width is twice the separation between the slits,
\(d,\) which is
\(0.5~\text{mm}.\) The slit to screen distance is
\(1~\text{m}.\) The wavelength of light used is:
| 1. |
\(125~\text{nm}\) |
2. |
\(250~\text{nm}\) |
| 3. |
\(500~\text{nm}\) |
4. |
\(1000~\text{nm}\) |
164. Given below are two statements:
| Assertion (A): |
In the Wheatstone Bridge shown in the figure, if the resistances in opposite arms are switched (i.e. \(Q, R \) are exchanged) then the bridge remains balanced if it was initially balanced. |
| Reason (R): |
The balance condition \(\dfrac P Q\) = \(\dfrac R S\) is not affected if resistances in opposite arms are switched. |

| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
165. A circular loop of radius
\(R,\) enters a region of uniform magnetic field
\(B\) as shown in the diagram. The field
\((B)\) is perpendicular to the plane of the loop while the velocity of the loop,
\(v\) is along its plane. The induced EMF:

| 1. |
increases continuously. |
| 2. |
decreases continuously. |
| 3. |
first increases and then decreases. |
| 4. |
remains constant throughout. |
166. The nuclear radius of a \({ }_{3}^{8} \mathrm{L i}\) nucleus is \(2.2~\text{fm}\). The nuclear radius of \({ }_{30}^{64} \mathrm{Zn}\) is nearly:
1. \(4.4~\text{fm}\)
2. \(7~\text{fm}\)
3. \(22~\text{fm}\)
4. \(17.6~\text{fm}\)
167. The two inputs
\((A)\) and
\((B)\) of a logic circuit are shown along with the output
\((Y)\) as functions of time. The 'highs' represent logic
\(1\) and the 'lows' represents logic
\(0.\) The correct truth table for this circuit is:

| 1. |
| \(A\) |
\(B\) |
\(Y\) |
| \(0\) |
\(0\) |
\(0\) |
| \(0\) |
\(1\) |
\(0\) |
| \(1\) |
\(0\) |
\(1\) |
| \(1\) |
\(1\) |
\(0\) |
|
2. |
| \(A\) |
\(B\) |
\(Y\) |
| \(0\) |
\(0\) |
\(0\) |
| \(0\) |
\(1\) |
\(1\) |
| \(1\) |
\(0\) |
\(0\) |
| \(1\) |
\(1\) |
\(0\) |
|
| 3. |
| \(A\) |
\(B\) |
\(Y\) |
| \(0\) |
\(0\) |
\(1\) |
| \(0\) |
\(1\) |
\(0\) |
| \(1\) |
\(0\) |
\(0\) |
| \(1\) |
\(1\) |
\(1\) |
|
4. |
| \(A\) |
\(B\) |
\(Y\) |
| \(0\) |
\(0\) |
\(0\) |
| \(0\) |
\(1\) |
\(1\) |
| \(1\) |
\(0\) |
\(1\) |
| \(1\) |
\(1\) |
\(0\) |
|
168. Given below are two statements:
| Assertion (A): |
The centre-of-mass of an isolated system of particles remains at rest if it is initially at rest. |
| Reason (R): |
Internal forces acting within a system cannot change the velocity of the centre-of-mass which is proportional to the total momentum of the system. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
169. Two particles of equal
mass \(m\) and
charge \(q\) move in a circular orbit of
radius \(r\) under the influence of a magnetic field
\(B.\) The kinetic energy of the particles is proportional to (assume that the particles don't exert electrostatic forces on each other):
| 1. |
\(q^{2}\) |
2. |
\(B^{2}\) |
| 3. |
\(r^{2}\) |
4. |
All of the above |
170. In this
\(LCR\) circuit, it is observed that the current and the voltage are in phase with each other. When the capacitor is replaced by a short circuit, the phase difference becomes
\(60^{\circ}.\)

Then:
| 1. |
\(\dfrac{L}{C}=R^2 \) |
2. |
\(\dfrac{L}{C}=2R^2\) |
| 3. |
\(\dfrac{L}{C}=3R^2\) |
4. |
\(\dfrac{L}{C}=\dfrac13R^2\) |
171. Wind blows towards north with a speed of
\(20~\text{m/s}.\) A plane flies in air with a velocity relative to ground of
\(100~\text{m/s},\) towards east. Then, the velocity of the plane relative to air is:
| 1. |
\(100\) m/s towards east and \(20\) m/s towards north. |
| 2. |
\(100\) m/s towards east and \(20\) m/s towards south. |
| 3. |
\(100\) m/s towards west and \(20\) m/s towards north. |
| 4. |
\(100\) m/s towards west and \(20\) m/s towards south. |
172. What should be the mass of a uniform sphere of radius
\(R\) so that the escape velocity from its surface equals
\(c,\) the velocity of light in vacuum? (Assume Newton's theory of gravitation to hold true)
| 1. |
\(\dfrac{Rc^2}{G}\) |
2. |
\(\dfrac{Rc^2}{2G}\) |
| 3. |
\(\dfrac{2Rc^2}{G}\) |
4. |
\(\sqrt2\dfrac{Rc^2}{G}\) |
173. A block is suspended from a spring and allowed to oscillate vertically, giving a time period
\(T.\) If the block is suspended from the same spring, the elongation produced will be:
| 1. |
\(\dfrac12gT^2\) |
2. |
\(\dfrac14gT^2\) |
| 3. |
\(\dfrac{1}{2\pi}gT^2\) |
4. |
\(\dfrac{1}{4\pi^2}gT^2\) |
174. The equation of vibration of a taut string, fixed at both ends, is given by:
\(y=(3~\text{mm})~\text{cos}\Big(\dfrac{\pi x}{10~\text{cm}}\Big)~\text{sin}(800\pi~\text{s}^{-1}~\text{t})\)
The speed of waves on the string is:
1. \(20\) m/s
2. 40 m/s
3. \(80\) m/s
4. \(160\) m/s
175. A ball of mass
\(m\) is projected with a
speed \(u,\) at an angle of
\(\theta\) with the horizontal. At its highest point, it moves on a smooth horizontal platform with a spring of spring
constant \(k\) attached, and the ball compresses the spring. The maximum compression in the spring is
\(x.\) Then:

| 1. |
\(\frac12mu^2=\frac12kx^2\) |
2. |
\(\frac12mu^2cos^2\theta=\frac12kx^2\) |
| 3. |
\(\frac12mu^2=\frac12kx^2cos^2\theta\) |
4. |
\(\frac12mu^2sin^2\theta=\frac12kx^2\) |
176. An ideal gas obeys the law
\(P^2V\) = constant during a reversible thermodynamic process. The relationship between
volume \((V)\) and absolute
temperature \((T)\) during this process is:
| 1. |
\(\dfrac {V}{T}\) = constant |
2. |
\(\dfrac {V^2}{T}\) = constant |
| 3. |
\(\dfrac {T^2}{V}\) = constant |
4. |
\(TV^2\) = constant |
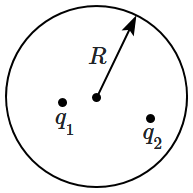
177. An uncharged spherical conducting shell of outer
radius \(R\) has two point charges
\(q_1,q_2\) placed within it. The flux of the electric field due to all charges from the outer surface of the sphere is
\(\phi_0\) while the potential of the sphere is
\(V_0.\) The ratio
\(\dfrac{\phi_0}{V_0}\) is proportional to:
| 1. |
\(\sqrt R\) |
2. |
\(R\) |
| 3. |
\(R^2\) |
4. |
\(\dfrac{1}{R^2}\) |
178. A ray of light, incident at
\(45^{\circ}\) onto the surface
\(AB\) of the right-angled prism, is reflected from
\(AC\) and retraces its path backwards. The refractive index of the material of the prism is:

1.
\(\sqrt2\)
2.
\(2\)
3.
\(2\sqrt2\)
4.
\(2+\sqrt2\)
179. The total energy of an electron in the
\(n^{\text{th}}\) orbit of a hydrogen atom is
\((-E_n). \) The de-Broglie wavelength of the electron in this orbit is:
(mass of electron = \(m\))
| 1. |
\(\dfrac{h}{\sqrt{2mE_n}}\) |
2. |
\(\dfrac{h}{\sqrt{mE_n}}\) |
| 3. |
\(\dfrac{h}{\sqrt{4mE_n}}\) |
4. |
\(\dfrac{hc}{\sqrt{E_n}}\) |
180. Given below are two statements:
| Assertion (A): |
The magnetic moment of a hydrogen-like atom is higher when it is in a state of higher quantum number \(n.\) |
| Reason (R): |
The magnetic moment of hydrogen-like atom, as calculated from Bohr's theory, is directly proportional to the principal quantum number \(n.\) |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
181. An input signal to a logic circuit's inputs is represented by a
\(0,\) if the voltage is low (true) and by a
\(1,\) if the voltage is high (false). The truth table representing the operation of the circuit is given by:
\(\begin{array}{|c|c|c|} \hline A & B & Y~\text{(output)} \\ \hline 0 & 0 & 0 \\ \hline 0 & 1 & 1 \\ \hline 1 & 0 & 0 \\ \hline 1 & 1 & 0 \\ \hline \end{array}\)
An input consisting of the following is presented to the pins
\(A\) and
\(B\):
| 1. |
pin A: \(0110101001\) |
| 2. |
pin B: \(1000011100\) |
The output
\(Y\) is:
| 1. |
\(0110100001\) |
| 2. |
\(1000010100\) |
| 3. |
\(0111100011\) |
| 4. |
\(1001010010\) |
182. Which of the following, is dimensionless?
| 1. |
\(\dfrac{\text{pressure}}{\text{density}\times\text{acceleration}}\) |
2. |
\(\dfrac{\text{density}\times\text{pressure}}{\text{speed}}\) |
| 3. |
\(\dfrac{\text{viscosity}\times\text{speed}}{\text{surface tension}}\) |
4. |
\(\dfrac{\text{surface tension}}{\text{(speed)}^2}\) |
183. A mango falling from a
height \(H\) from a tree is observed from a car moving with a uniform
velocity \(u,\)\(g.\) along a road, that passes underneath. The mango hits the car. Assume the size of the car to be negligible, and the acceleration due to gravity to be

Initially, the distance of the car from the tree was
\(d.\) Then:
| 1. |
\(\dfrac{u^2}{g}=\dfrac{d^2}{2H}\) |
2. |
\(\dfrac{u^2}{g}=\dfrac{d^2}{4H}\) |
| 3. |
\(\dfrac{u^2}{g}=\dfrac{H^2}{2d}\) |
4. |
\(\dfrac{u^2}{g}=\dfrac{H^2}{4d}\) |
184. What is the value of linear velocity, if the angular velocity vector is \(\vec{\omega}=3 \hat{i}-4 \hat{j}+\hat{k}\) and the position vector is \(\vec {r}=5 \hat{i}-6 \hat{j}+6 \hat{k}?\)
1. \(-18 \hat{i}-13 \hat{j}+2 \hat{k}\)
2. \(18 \hat{i}+13 \hat{j}-2 \hat{k}\)
3. \(6 \hat{i}+2 \hat{j}-3 \hat{k}\)
4. \(6 \hat{i}-2 \hat{j}+8 \hat{k}\)
185. The block, shown in the figure, is suspended as shown from two identical steel wires. The extension in the wires due to the tension is
\(\Delta l_1.\) If the block is suspended by one of the wires the extension in it is
\(\Delta l_2.\) Then
\(\dfrac{\Delta l_1}{\Delta l_2}\) equals:

| 1. |
\(1\) |
2. |
\(2\) |
| 3. |
\(\sqrt 2\) |
4. |
\(\dfrac12\) |
Physics - Section B
186. A man \((A)\) has to throw a ball vertically up to a partner \((B)\) who is standing up, above his level by \(15~\text{m}.\) The \((B)\) partner can catch the ball only when it comes downwards with a maximum speed of \(10~\text{m/s}\)
(take acceleration due to gravity as \(10~\text{m/s}^{2}\))
The minimum and maximum speeds of the throw are: (nearly)
1. \(10~\text{m/s}~\text{and}~20~\text{m/s}\)
2. \(10~\text{m/s}~\text{and}~30~\text{m/s}\)
3. \(20~\text{m/s}~\text{and}~20\sqrt{3}~\text{m/s}\)
4. \(10\sqrt{3}~\text{m/s}~\text{and}~20~\text{m/s}\)
187. Two vessels containing gases at pressures \(P_1,P_2~\)are connected by a \(U\text-\)tube containing two liquids \(A,B\) of densities \(3\rho, \rho\). The levels of the liquids and their contact surfaces are shown in the diagram above. Then,
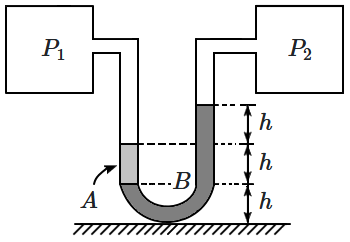
1. \(P_1-P_2=\rho gh\)
2. \(P_2-P_1=\rho gh\)
3. \(P_1=P_2\)
4. \(P_1+P_2=7\rho gh\)
188. A metal ball of mass \(2\) kg is heated by a \(30\) W heater, in a room at \(20^\circ \text{C}.\) The temperature of the metal becomes steady at \(50^\circ \text{C}.\) If the same ball was heated by a \(20\) W heater in a room at \(30^\circ \text{C},\) the steady temperature of the ball will be:
| 1. |
\(40^\circ \text{C}\) |
2. |
\(50^\circ \text{C}\) |
| 3. |
\(60^\circ \text{C}\) |
4. |
\(70^\circ \text{C}\) |
189. An ideal gas is enclosed in a volume by means of a piston-cylinder arrangement as shown in the adjacent diagram. The piston as well as the walls of the cylinder are non-conducting. The cross-sectional area of the piston is \(A.\) Gravity \(g\) is acting downward. A small block of mass \(m\) is placed on top of the piston. There is no atmospheric pressure outside. An amount of thermal energy \(\Delta Q\) is slowly supplied to the gas, and its temperature rises. Then, the gas:

| 1. |
expands continuously, making the volume infinite. |
| 2. |
first expands and then contracts slightly. |
| 3. |
expands and then reaches a steady-state. |
| 4. |
expands and then contracts to return to its initial volume. |
190. In the circuit shown in the diagram, the resistance
\(r\) is unknown but fixed. The EMF
\(E\) can be varied and the current
\((i)\) passing through the ammeter
\({(A)}\) can be measured in magnitude only.
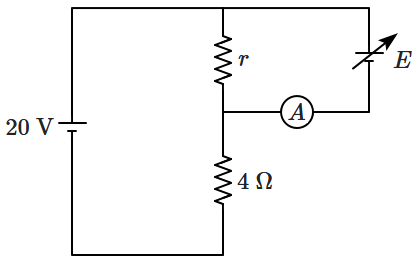
When the EMF
\(E\) is adjusted so that the current in the ammeter
\({(A)}\) is zero, then the value of this EMF is
\(E_0.\)
| 1. |
\(E_0= 16~\text{V}~\text{if}~r = 8~\Omega\) |
| 2. |
\(E_0= 10~\text{V}~\text{if}~r = 2~\Omega\) |
| 3. |
\(E_0= 5~\text{V}~\text{if}~r = 1~\Omega\) |
| 4. |
\(E_0< 20~\text{V}~\text{no matter what the value of}~r.\) |
191. An equi-convex lens of focal length
\(50~\text{cm}\) and an equi-concave lens of the same focal length are placed
\(50~\text{cm}\) apart, with a common principal axis. A point object is placed on the principal axis of the system, at a distance of
\(100~\text{cm}\) in front of the convex lens (see figure). The final image is formed at:

| 1. |
\(100~\text{cm}\) in front of the concave lens |
| 2. |
\(50~\text{cm}\) in front of the concave lens |
| 3. |
\(50~\text{cm}\) behind the concave lens |
| 4. |
infinity |
192. A uniform beam
\(3.0~\text m\) long, of weight
\(100~\text N\) has a
\(300~\text N\) weight placed
\(0.5~\text m\) from one end. The beam is suspended by a string
\(1.0~\text m\) from the same end. A diagram of the weights placed on the beam is given below:

How far from the other end must a weight of
\(80~\text N\) be placed for the beam to be balanced?
| 1. |
\(0.75~\text m\) |
2. |
\(2.25~\text m\) |
| 3. |
\(1.25~\text m\) |
4. |
\(1.875~\text m\) |
193. If a simple pendulum be suspended in an elevator which is moving upward, its time period is found to decrease by \(2\%.\) The acceleration of the elevator is (in magnitude):
1. \(2\%\) of \(g\)
2. \(1\%\) of \(g\)
3. \(4\%\) of \(g\)
4. \(102\%\) of \(g\)
194. A man, swimming with a speed
\(u_1,\) can cross a river fastest in a time,
\(T\). His friend, who swims with a speed
\(u_2,\) reaches the opposite bank in the same time when he swims at an angle of
\(30^{\circ}\) with the bank. Then:
| 1. |
\(u_1=\dfrac{\sqrt3}{2}u_2\) |
2. |
\(u_1=\dfrac{1}{2}u_2\) |
| 3. |
\(u_1=\dfrac{1}{\sqrt2}u_2\) |
4. |
\(u_1=\dfrac{1}{\sqrt3}u_2\) |
195. An Atwood's machine with blocks of masses
\(3\) kg and
\(2\) kg is set up in a laboratory. The string is taut and the blocks start moving at
\(t=0.\)

The work done by tension on the
\(3\) kg block has a magnitude
\(W_1\) while the work done by gravity on the same block has a magnitude
\(W_2,\) since the beginning of motion.
| 1. |
\(W_1=W_2\) |
| 2. |
\(W_1>W_2\) |
| 3. |
\(W_1<W_2\) |
| 4. |
Any of the above can be true |
196. The magnetic field at the center
\(O\) of the semi-circular part of the current-carrying wire, due to the curved and the straight wires (very long), is:

| 1. |
\({\Large\frac{\mu_0i}{4R}}\) |
| 2. |
\({\Large\frac{\mu_0i}{4R}}+{\Large\frac{\mu_0i}{2\pi R}}\) |
| 3. |
\({\Large\Big(\frac{\mu_0i}{4R}+\frac{\mu_0i}{4\pi R}\Big)} \) |
| 4. |
\({\Large\Big[\Big(\frac{\mu_0i}{4R}\Big)^2+\Big(\frac{\mu_0i}{2\pi R}\Big)^2\Big]^{1/2}} \) |
197. A photon of energy
\(E_p\) has the same wavelength as an electron of kinetic energy
\(E_e.\) The rest energy
(i.e., \(mc^2\)) of the electron is:
| 1. |
\(\dfrac{E^2_p}{E_e}\) |
2. |
\(\dfrac{E^2_e}{E_p}\) |
| 3. |
\(\dfrac{E^2_e}{2E_p}\) |
4. |
\(\dfrac{E^2_p}{2E_e}\) |
198. A bullet of mass \(20\) g and moving with \(600\) m/s collides with a block of mass \(4\) kg hanging with the string. What is the velocity of a bullet when it comes out of the block, if the block rises to height \(0.2\) m after collision?
1. \(200\) m/s
2. \(150\) m/s
3. \(400\) m/s
4. \(300\) m/s
199. The diode shown in the adjacent circuit is ideal and connected to the secondary of an ideal transformer.
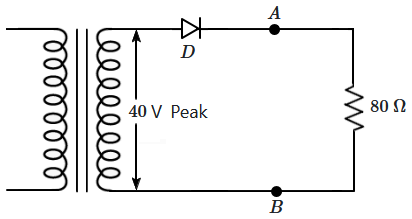
The average current flowing through the diode is: (forward is taken as positive)
| 1. |
zero |
| 2. |
positive and greater than \(0.5~\text A\) |
| 3. |
positive, but less than \(0.5~\text A\) |
| 4. |
negative |
200. The SI unit of current density is:
| 1. |
\(\text{A/m}^3\) |
2. |
\(\text{A/m}^2\) |
| 3. |
\(\text{A/m}\) |
4. |
\(\text{A-m}\) |
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course