VNTR belongs to a class of satellite DNA and its size in a genome varies from 0.1 to ___
1. 20 kb
2. 30 kb
3. 40 kb
4. 50 kb
All the following statements about cellulose are correct but one is wrong. Which one is wrong?
| 1. | Cellulose is homopolymer |
| 2. | It is the most abundant organic molecule in the biosphere |
| 3. | It has -1-4 glycosidic bonds |
| 4. | It is a branched polymer of - glucose, can hold iodine in the helical portion giving blue black colour with it |
Arrange the layers of anther wall from locus to periphery:
1. Epidermis, middle layers, tapetum, endothecium
2. Tapetum, middle layers, epidermis, endothecium
3. Endothecium, epidermis, middle layers, tapetum
4. Tapetum, middle layers, endothecium, epidermis
Read the following statements:
| a. | As we go higher from species to kingdom, the number of common characteristics goes on decreasing |
| b. | Lower the taxa, less are the characteristics that the members within the taxon share |
| c. | Higher the category, greater is the difficulty of determining the relationship to other taxa at the same level |
| 1. | Two cells called synergids flank the egg at the chalazal end |
| 2. | Three antipodal cells guide the entry of the pollen tube into the embryo sac |
| 3. | at maturity there are 8 cells and 7 nuclei |
| 4. | polar nuclei share the cytoplasm of the large central cell |
Which one of the following is the start codon?
| 1. | UGA | 2. | UAA |
| 3. | UAG | 4. | AUG |
If a length of DNA has 45,000 base pairs, how many complete turns will the DNA molecule take?
| 1. | 45000 | 2. | 450 |
| 3. | 4500 | 4. | 45 |
Find the odd one (with respect to dominant traits in garden pea):
| 1. | Yellow pod color | 2. | Violet flower color |
| 3. | Yellow seed color | 4. | Full pod shape |
Match List - I with List - II
| List - I | List - II |
| (a) Aspergillus Niger | (i) Acetic Acid |
| (b) Acetobacter aceti | (ii) Lactic Acid |
| (c) Clostridium butylicum | (iii) Citric Acid |
| (d) Lactobacillus | (iv) Butyric Acid |
Choose the correct answer from the options given below.
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iii) | (i) | (iv) |
| 2. | (iv) | (ii) | (i) | (iii) |
| 3. | (iii) | (i) | (iv) | (ii) |
| 4. | (i) | (ii) | (iii) | (iv) |
Read the following paragraph with blanks A, B, C, and D.
Hugo de Vries based his work on A brought forth the idea of mutations. Mutations are random and B while Darwinian variations are small and C . Evolution for Darwin was gradual while de Vries believed mutation caused speciation and hence called it D.
Choose the option which correctly fills in the blanks A, B, C, and D.
| A | B | C | D | |
| 1. | Evening primrose | Directional | Directionless | Saltation |
| 2. | Evening primrose | Directional | Directionless | Natural selection |
| 3. | Garden pea | Directionless | Directional | Saltation |
| 4. | Evening primrose | Directionless | Directional | Saltation |
The historic convention on biological diversity held in Rio de Janeiro in 1992 is known as:
1. CITES Convention
2. The Earth Summit
3. G-16 Summit
4. MAB Programme
Consider the following statements:
| I. | Whorled phyllotaxy is seen in Alstonia. |
| II. | Flowers of mustard, datura and chilli are actinomorphic |
| III. | The ovary in plum, peach and rose is epigynous. |
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II and III
The main arena of various types of activities of a cell is –
1. Plasma membrane
2. Mitochondrian
3. Cytoplasm
4. Nucleus
How much O2 is used in the respiration of two molecules of Tripalmitin?
1. 145
2. 102
3. 98
4. 6
The following processes occur during photosynthesis
| (i) | Reduction of carbon dioxide |
| (ii) | The splitting of water |
| (iii) | The synthesis of glucose |
| (iv) | Release of oxygen |
| (v) | Formation of ATP |
Which one of the following combinations is correct for the light phase?
| 1. | (i), (ii) and (iii) |
| 2. | (iii), (iv) and (v) |
| 3. | (i), (iii) and (iv) |
| 4. | (ii), (iv) and (v) |
Bark includes a variety of tissue types like:
| 1. | Phellem, secondary xylem, and secondary phloem |
| 2. | Periderm and all kinds of secondary tissues |
| 3. | Phellogen, secondary phloem, phellem, and secondary cortex |
| 4. | Phelloderm, secondary phloem, and primary xylem |
The classification given by Bentham and Hooker is:
| 1. | Artificial | 2. | Natural |
| 3. | Phylogenetic | 4. | Numerical |
Absence of sex organs, general absence of asexual spores, and plasmogamy by means of somatogamy are features of:
| 1. | Ascomycetes | 2. | Deuteromycetes |
| 3. | Basidiomycetes | 4. | Phycpmycetes |
Consider the following statements:
| I. | Ori is responsible for copy number of the linked DNA |
| II. | Transformation is process through which a piece of DNA is introduced into a host bacterium |
| III. | Bacteriophages have very high copy numbers of their genome within the bacterial cell. |
| 1. | I and II only | 2. | I and III only |
| 3. | II and III only | 4. | I, II and III |
The following figure represents :
1. Transverse section of young anther
2. 3-D cut section of mature anther
3. Cut section of filament
4. Transverse section of mature filament
According to Darwin, organic evolution is due to:
| 1. | Interspecific competition |
| 2. | Competition within closely related species. |
| 3. | Reduced feeding efficiency in one species due to the presence of interfering species. |
| 4. | Intraspecific competition |
How many of these statements are true for polymorphism?
| (A) | It is inheritable |
| (B) | It arises due to mutation |
| (C) | It varies at the genetic level |
| (D) | It is the basis of genetic mapping |
| (E) | It is important for evolution and speciation |
| (F) | It varies from single nucleotide to large scale |
1. 6
2. 5
3. 4
4. 3
Decomposers like fungi and bacteria are:
(i) autotrophs
(ii) heterotrophs
(iii) saprotrophs
(iv) chemo-autotrophs
Choose the correct answer:
1. (i) and (iii)
2. (i) and (iv)
3. (ii) and (iii)
4. (i) and (ii)
After about how many years of the formation of the earth, life appeared on this planet?
1. 500 billion years
2. 50 million years
3. 500 million years
4. 50 billion years
Which of the following is a mustard family?
| 1. | Brassicaceae | 2. | Solanaceae |
| 3. | Liliaceae | 4. | Fabaceae |
The frequency of recombination between gene pairs on the same chromosome as a measure of the distance between genes was explained by:
1. Sutton Boveri
2. T.H. Morgan
3. Gregor J. Mendel
4. Alfred Sturtevant
In some plants, the thalamus contributes to fruit formation. Such fruits are termed as:
1. False fruits
2. Aggregate fruits
3. True fruits
4. Parthenocarpic fruit
Which of the following was presented as evidence in favor of “the RNA world” hypothesis?
| 1. | The fact that DNA can encode genetic information |
| 2. | The fact that enzymes, made of protein, can catalyze biological reactions |
| 3. | The discovery that some enzymes are composed of RNA rather than protein |
| 4. | The discovery of new life forms that encode their genetic information in RNA rather than DNA |
| I: | are commonly found in the anaerobic sludge during sewage treatment. |
| II: | are also present in the rumen (a part of stomach) of cattle. |
| 1. | 5 per cent. | 2. | 13 per cent. |
| 3. | 20 per cent. | 4. | 45 per cent. |
| 1. | Acetyl CoA, a 2 carbon molecule |
| 2. | G3P (Glyceraldehyde-3-phosphate), a 3 carbon molecule |
| 3. | RuBP (Ribulose-1,5-bisphosphate), a 5 carbon molecule |
| 4. | Citrate, a 6 carbon molecule |
| 1. | gibberellins | 2. | cytokinins |
| 3. | abscisic acid | 4. | auxins |
| 1. | 3 | 2. | 25 |
| 3. | 34 | 4. | 98 |
The part of chromosome beyond secondary constriction is known as:
| 1. | Chromomere | 2. | Satellite |
| 3. | Kinetochore | 4. | Centromere |
C2H4 or Ethylene are …...or derivative of….
1. Indol compounds
2. Gases
3. carotenoids
4. Terpenes
The following diagram shows:
1. Multicarpellary, syncarpus pistil of Papaver
2. Multicarpellary, apocarpus pistil of Papaver
3. Multicarpellary, apocarpus gynoecium of Michelia
4. Multicarpellary, syncarpus gynoecium of Michelia
Match each item in Column-I with one item in Column-II and choose your answer from the codes given below:
| Column-I (Disease) |
Column-II (causative organism) |
||
| I. | Potato spindle tuber disease | 1. | Viroid |
| II. | Creutzfeldt Jakob Disease | 2. | Prion |
| III. | Mosaic disease of tobacco | 3. | Virus |
| IV. | Rust of wheat | 4. | Fungus |
Codes:
| I | II | III | IV | |
| 1. | 1 | 2 | 3 | 4 |
| 2. | 2 | 1 | 4 | 3 |
| 3. | 2 | 1 | 3 | 4 |
| 4. | 1 | 2 | 4 | 3 |
Which kind of modification is seen in the given diagrams?
1. Vegetables for storage
2. Roots for absorption
3. Roots for storage
4. Stem for protection
The hypodermis of dicot stem and monocot stem is made up of:
1. collenchyma and sclerenchyma respectively
2. collenchyma in both
3. sclerenchyma in both
4. sclerenchyma and collenchyma respectively
Certain marine brown and red algae produce large amounts of:
| 1. | Carrageen and algin respectively |
| 2. | Fixed nitrogen |
| 3. | Sulphated phycocolloids |
| 4. | Water holding substances |
Pyruvate dehydrogenase complex, responsible for Pyruvate decarboxylation in cellular respiration in eukaryotes, is found in:
1. Cytoplasm
2. Perimitochondrial space
3. Mitochondiral matrix
4. lnner mitochondiral membrane
All given statements are correct with respect to chemiosmotic hypothesis of photosynthesis, except:
| 1. | Photolysis of water produces H+ ions that accumulate within the lumen of thylakoid |
| 2. | Protons are transported across the membrane from lumen of thylakoid to stroma side of chloroplast, which is carried out by the primary acceptor of electrons |
| 3. | Conversion of NADP+ to NADPH+ H+ removes protons from the stroma side of chloroplast |
| 4. | ATP production occurs towards stroma side of chloroplast due to movement of protons from lumen side through CF0-CF1 complex |
A non-proteinaceous enzyme is:
1. lysozyme
2. ribozyme
3. ligase
4. deoxyribonuclease
Which stage of meiotic prophase shows terminalisation of chiasmata as its distincitve feature?
1. Diakinesis
2. Pachytene
3. Leptotene
4. Zygotene
| I: | is production of seeds without fertilisation |
| II: | means that fruits develop without fertilisation |
| III: | can be induced through the application of growth hormones |
| IV: | leads to production of seedless fruits |
| Assertion (A): | Cyclosporin produced by Trichoderma has been commercialised as blood-cholesterol lowering agents. |
| Reason (R): | Cyclosporin acts by competitively inhibiting the enzyme responsible for synthesis of cholesterol. |
| 1. | Both (A) and (R) are True and (R) correctly explains (A). |
| 2. | (A) is True; (R) is False |
| 3. | (A) is False; (R) is False |
| 4. | Both (A) and (R) are True but (R) does not correctly explain (A). |
| 1. | Carbon dioxide concentration | 2. | Light intensity |
| 3. | Temperature | 4. | Oxygen |
What does the P wave represent in ECG?
1. Atrial Diastole
2. Atrial systole
3. ventricular Diastole
4. Joint Diastole
How many of the following factors are favourable for shifting the Oxyhaemoglobin dissociation curve to the left side: (low pO2, high pCO2, more H+ concentration, higher temperature, high pO2, low pCO2, less H+ concentration, lower temperature)
1. 3
2. 4
3. 5
4. 6
Match the columns and identify the correct option:
| Column-I | Column-II | ||
| A. | Thylakoids | 1. | Disc-shaped sacs in Golgi apparatus |
| B. | Cristae | 2. | Condensed structure of DNA |
| C. | Cisternae | 3. | Flat membranous sacs in stroma |
| D. | Chromatin | 4. | Infolding in mitochondria |
Options:
| A | B | C | D | |
| 1. | 4 | 3 | 1 | 2 |
| 2. | 3 | 4 | 1 | 2 |
| 3. | 3 | 1 | 4 | 2 |
| 4. | 3 | 4 | 2 | 1 |
Which of the following is incorrect?
| 1. | The substrate binds to the active site of the enzyme |
| 2. | The binding of the substrate induces the enzyme to alter its shape. |
| 3. | The active site of the enzyme when it is in close proximity of the substrate breaks the chemical bonds of the substrate. |
| 4. | None of these |
Select the incorrect match with respect to group of animals and their taxon:
| 1. | Ichthyophis, Bufo, Hyla | – Amphibia |
| 2. | Hippocampus, Exocoetus, Pterophyllum | – Osteichthyes |
| 3. | Corvus, Chelone, Calotes | – Reptilia |
| 4. | Pteropus, Equus, Delphinus | – Mammalia |
What is the genetic disorder in which an individual has an overall masculine development gynaecomastia and is sterile?
1. Down's syndrome
2. Turner's syndrome
3. Klinefelter's syndrome
4. Edward syndrome
A Hominid fossil discovered in Java in 1891, now extinct, having a cranial capacity of about 900 ccs was:
1. Homo erectus
2. Neanderthal man
3. Homo sapiens
4. Australopithecus
| 1. | erythroblasts | 2. | megakaryocytes |
| 3. | myeloblasts | 4. | thrombocytes |
Which of the following stimulates the secretion of gastric juice?
| 1. | Gastrin | 2. | Enterogasterone |
| 3. | Secretin | 4. | Hepatocrinin |
What is true regarding A, B, and C in the given diagrammatic representation of rDNA technology?
| I: | At A same restriction enzyme is used to cut both foreign and vector DNA |
| II: | The enzyme used at B is DNA ligase |
| III: | Step C can be called as transformation |
| 1. | I and II only | 2. | I and III only |
| 3. | II and III only | 4. | I,II and III |
The DNA molecule to which the gene of interest is integrated for cloning is called:
| 1. | Vector | 2. | Template |
| 3. | Carrier | 4. | Transformer |
Which kind of therapy was given in 1990 to a four-year-old girl with adenosine deaminase (ADA) deficiency?
| 1. | Radiation therapy | 2. | Gene therapy |
| 3. | Chemotherapy | 4. | Immunotherapy |
Which of the following sequences of PCR is correct?
| (a) | (b) | (c) | |
| 1. | Extension of primer |
Annealing | Denaturation |
| 2. | Denaturation | Annealing | Extension of primer |
| 3. | Annealing | Extension of primer |
Denaturation |
| 4. | Denaturation | Extension of primer |
Annealing |
With regard to insulin, choose the correct statements:
| (a) | C-peptide is not present in mature insulin. |
| (b) | The insulin produced by rDNA technology has C-peptide. |
| (c) | The pro-insulin has C-peptide. |
| (d) | A-peptide and B-peptide of insulin are interconnected by disulphide bridges. |
| 1. | (a), (c) and (d) only | 2. | (a) and (d) Only |
| 3. | (b) and (d) only | 4. | (b) and (c) only |
The calcium ions released in sarcoplasm will bind to:
| 1. | Tropomyosin | 2. | Troponin |
| 3. | Actin | 4. | Meromyosin |
A __________is obtained when percentage saturation of Hb with O2 is plotted against pO2.
| 1. | J shaped curve | 2. | Sigmoid Curve |
| 3. | Exponential curve | 4. | None of these |
Which of the following is true about Inclusion bodies?
1. membraneless
2. present freely in cytoplasm
3. store phosphate, glycogen
4. All of the above
Which of the following increases when there is depolarisation of a nerve fibre?
1. Diffusion of sodium ions into the neuron
2. Diffusion of sodium ions out of the neuron
3. Diffusion of potassium ions into the neuron
4. Both 1 & 3
Which of the following organisms is used in the production of beverages?
1. Penicillium notatum
2. Saccharomyces cerevisiae
3. Aspergillus niger
4. Clostridium butylicum
Parturition is induced by:
1. Neural mechanism
2. Endocrine mechanism
3. Both
4. Local mechanism
Consider the statements given below regarding contraception and answer as directed thereafter:
| (i) | Medical Termination of Pregnancy (MTP) during the first trimester is generally safe |
| (ii) | Generally, chances of conception are nil until the mother breast-feeds the infants up to two year |
| (iii) | Intrauterine devices like copper-T are effective contraceptives |
| (iv) | Contraception pills may be taken up to one week after coitus to prevent conception |
Which two of the above statements are correct?
1. (ii) and (iii)
2. (iii) and (iv)
3. (i) and (iii)
4. (i) and (ii)
Atherosclerosis affects:
| 1. | the vessel which supply blood to kidney |
| 2. | the vessel which supply blood to brain |
| 3. | the vessel which supply blood to heart muscle |
| 4. | the vessel which supply blood to lungs |
Which of the following is not true about testosterone?
1. stimulates spermatogenesis
2. influence libido
3. Catabolic effects on protein metabolism
4. responsible for aggressiveness in male
The illustration below shows the skull of two different mammals. Use the illustration to answer the question that follows:
Which of the following accurately describes the differences between these skulls?
| 1. | Skull A has more teeth than skull B |
| 2. | Skull A has more brain capacity than skull B |
| 3. | Skull A is of a primate and skull B is not of a primate |
| 4. | Skull A is the skull of an ape and skull B is the skull of a human |
| Column-I | Column-II | ||
| (a) | Restriction endonuclease | (i) | joins the DNA fragments |
| (b) | Exonuclease | (ii) | extends primers on genomic DNA template |
| (c) | DNA ligase | (iii) | cuts DNA at specific position |
| (d) | Taq polymerase | (iv) | removes nucleotides from the ends of DNA |
Select the correct option from the following:
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (i) | (iv) | (ii) |
| 2. | (iii) | (iv) | (i) | (ii) |
| 3. | (iv) | (iii) | (i) | (ii) |
| 4. | (ii) | (iv) | (i) | (iii) |
Match the following parts of a nephron with their function:
| Column-I | Column-II | ||
| (a) | Descending limb of Henle's loop | (i) | Reabsorption of salts |
| (b) | Proximal convoluted tubule | (ii) | Reabsorption of water |
| (c) | Ascending limb of Henle's loop | (iii) | Conditional reabsorption of sodium ions and water |
| (d) | Distal convoluted tubule | (iv) | Reabsorption of ions, water and organic nutrients |
Select the correct option from the following:
| (a) | (b) | (c) | (d) | |
| 1. | (i) | (iii) | (ii) | (iv) |
| 2. | (ii) | (iv) | (i) | (iii) |
| 3. | (i) | (iv) | (ii) | (iii) |
| 4. | (iv) | (i) | (iii) | (ii) |
"Ramachandran plot" is used to confirm the structure of
| 1. | RNA | 2. | Proteins |
| 3. | Triacylglycerides | 4. | DNA |
Identify the correct pair representing the causative agent of typhoid fever and the confirmatory test for typhoid.
1. Salmonella typhi/Widal test
2. Plasmodium vivax/UTI test
3. Streptococcus pneumoniae/Widal test
4. Salmonella typhi/Anthrone test
Consider the following features:
| (a) | Organ system level of organization |
| (b) | Bilateral symmetry |
| (c) | True coelomates with the segmentation of the body |
Select the correct option of animal groups that possess all the above characteristics:
1. Annelida, Mollusca and Chordata
2. Annelida, Arthropoda, and Chordata
3. Annelida, Arthropoda, and Mollusca
4. Arthropoda, Mollusca, and Chordata
Choose the correct option wherein the correct stages of the development of human embryo takes place:
| Ovary | Fallopian Tube | Uterus | |
| 1. | Morula | Fertilized egg | Blastocyst |
| 2. | Unfertilized egg | Fertilized egg | Morula |
| 3. | Unfertilized egg | Fertilized egg | Blastocyst |
| 4. | Fertilized egg | Morula | Blastocyst |
| 1. | Thermoregulation | 2. | Osmoregulation |
| 3. | Endocrine functions | 4. | Gastric secretions |
| 1. | Tryptophan | 2. | Tyrosine |
| 3. | Methionine | 4. | Glycine |
In a marriage between a male with blood group A and a female with blood group B, the progeny had either blood group AB or B. What could be the possible genotype of parents?
| 1. | IA i (Male) ; IB IB (Female) |
| 2. | IA IA (Male) ; IB IB (Female) |
| 3. | IA IA (Male) ; IB i (Female) |
| 4. | IA i (Male) ; IB i(Female) |
Given below is a list of animals. What is the number of animals in the list that have a true coelom but not a segmented body?
| Meandrina, Chaetopleura, Cucumaria, Ancylostoma, Bombyx, Aplysia, Amphioxus, Antedon |
| 1. | 2 | 2. | 3 |
| 3. | 4 | 4. | 5 |
Which of the following cell/cells will secrete histamine, serotonin & heparin?
| 1. | Eosinophil | 2. | Basophil |
| 3. | Lymphocyte | 4. | Neutrophils |
What amount of carbon dioxide is delivered by every 100 ml of deoxygenated blood to the alveoli?
1. 4 ml
2. 5 ml
3. 15 ml
4. 20 ml
Which of the following is not important for maintaining the increasing osmolarity in the medullary interstitium?
| 1. | The proximity between Henle's loop and vasa recta |
| 2. | Counter current flow in two limbs of tubule |
| 3. | The differential permeability of different segments of nephron |
| 4. | Counter current flow in two limbs of Vasa recta |
Which of the following is not a part of pectoral girdle?
1. Scapula
2. Clavicle
3. Sternum
4. Glenoid cavity
Which of the following is not true about catecholamines?
1. secreted by adrenal medulla
2. Inhibits breakdown of glucose
3. increases the rate of respiration
4. increase heartbeat
Which of the following chemical changes is responsible for repolarisation of the membrane of axons?
1. Opening of Na+ channels and rapid influx of Na+ ions
2. Opening of K+ channels and rapid influx of K+ ions
3. Opening of Na+ channels and closing of K+ channels
4. Closing of Na+ channels and opening of K+ channels
In the given diagram, the part of the female reproductive system that undergoes cyclical changes with changes in the secretion of gonadotropins and gonadal steroids during the menstrual cycle is represented by:
1. A
2. B
3. C
4. D
Select the correct statement regarding spermiogenesis:
| 1. | lnvolves transformation of spermatids into spermatozoa |
| 2. | lnvolves release of spermatozoa from seminiferous tubule into the lumen |
| 3. | lt is followed by meiosis ll that restores the haploid number of chromosomes in spermatozoa |
| 4. | All of these |
Read the following given statements and choose the correct option:
| A: | All ART require extremely high precision handing by specialized professionals and expensive instrumentation. |
| B: | Emotional, religious and social factors are also deterrents in the adoption of these methods |
1. Both A and B are incorrect
2. Both A and B are correct
3. Only A is correct
4. Only B is correct
Find the odd one out with respect to meiotic cell cycle:
1. DNA replication occurs once only i.e., before Gap-2
2. Karyokinesis occurs twice
3. Reduction of ploidy at metaphase-l
4. Crossing over in tetrad stage
The naturalist who had the similar observations as that of Darwin was…
| 1. | Alfred Wallace | 2. | Oparin |
| 3. | Haldane | 4. | L. Miller |
To which type of barriers under innate immunity do the saliva in the mouth and the tears from the eyes belong?
1. Cellular barriers
2. Physiological barriers
3. Physical barriers
4. Cytokine barriers
What is the criterion for DNA fragments movement on agarose gel during gel electrophoresis?
1. The larger the fragment size, the farther it moves
2. The smaller the fragment size, the farther it moves
3. Positive charged fragment moves to farther end.
4. Negatively charged fragments do not move
Cellulose upon acetylation with excess acetic anhydride/H2SO4 (catalytic) gives cellulose triacetate whose structure is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Arrange the following species in increasing order of their dipole moments:
NH3,NF3,BF3,H2O
1.
2.
3.
4.
Arrange the following compounds in the increasing order of their densities:
 |
 |
 |
 |
| (i) | (ii) | (iii) | (iv) |
1. (i) < (ii) < (iii) < (iv)
2. (i) < (iii) < (iv) < (ii)
3. (iv) < (iii) < (ii) < (i)
4. (ii) < (iv) < (iii) < (i)
The correct increasing order of basic strength for the following compounds is:
 |
 |
 |
| (I) | (II) | (III) |
1. II < III < I
2. III < I < II
3. III < II < I
4. II < I < III
The oxoacid of sulphur that does not contain a bond between sulphur atoms is:
1. H2S2O7
2. H2S2O3
3. H2S4O6
4. H2S2O4
The correct statements among the following are:
| (I) | Valence bond theory cannot explain the color exhibited by transition metal complexes. |
| (II) | Valence bond theory can predict quantitatively the magnetic properties of transition metal complexes. |
| (III) | Valence bond theory cannot distinguish ligands as weak and strong field ones. |
1. (I), (II), and (III)
2. (II), and (III) only
3. (I), and (II) only
4. (I), and (III) only
Consider the reaction sequence given below :
The correct statement among the following is:
| 1. | Changing the concentration of base will have no effect on reaction (1) |
| 2. | Changing the concentration of base will have no effect on reaction (2) |
| 3. | Changing the base from -OH to -OR will have no effect on reaction (2) |
| 4. | Doubling the concentration of base will double the rate of both the reactions. |
The work function of sodium metal is 4.41 × 10–19 J. If the photons of wavelength 300 nm are incident on the metal, the kinetic energy of the ejected electrons will be:
(h = 6.63 × 10–34 Js ; c = 3 × 108 m/s)
1. 111 × 10–21
2. 333 × 10–21
3. 222 × 10–21
4. 555 × 10–21
An organic compound (A) (molecular formula C6H12O2) was hydrolyzed with dil. H2SO4 to give a carboxylic acid (B) and alcohol (C). 'C' gives white turbidity immediately when treated with anhydrous ZnCl2 and conc. HCl. The organic compound (A) is:
| 1. | 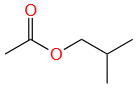 |
2. |  |
| 3. | 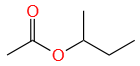 |
4. |  |
The set that contains atomic numbers of only transition elements is:
1. 21, 32, 53, 64
2. 9, 17, 34, 38
3. 21, 25, 42, 72
4. 37, 42, 50, 64
Identify the major products (A) and (B) respectively in the following reactions of phenol:

| [A] | [B] | |
| 1. |  |
 |
| 2. |  |
 |
| 3. | 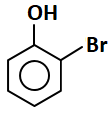 |
 |
| 4. |  |
 |
Number of bridging CO ligands in [Mn2(CO)10] is:
| 1. | 2 | 2. | 0 |
| 3. | 4 | 4. | 1 |
The NaNO3 weighed out to make 50 mL of an aqueous solution containing 70.0 mg Na+ per ml is:
(Rounded off to the nearest integer) [Given: Atomic weight in g mol–1 – Na: 23; N: 14; O: 16]
| 1. | 13 g | 2. | 26 g |
| 3. | 18 g | 4. | 22 g |
When 12.2 g of benzoic acid is dissolved in 100 g of water, the freezing point of the solution was found to be
–0.93 °C. The number (n) of benzoic acid molecules associated (assuming 100% association) is:
(Kf(H2O) = 1.86K kg mol–1)
| 1. | 3 | 2. | 1 |
| 3. | 2 | 4. | 5 |
In a mildly alkaline medium, thiosulphate ion is oxidized by to "A". The oxidation state of sulphur in "A" is:
| 1. | +4 | 2. | +2 |
| 3. | –2 | 4. | +6 |
The pH of an ammonium phosphate solution, if the pKa of phosphoric acid and the pKb of ammonium
hydroxide are 5.23 and 4.75 respectively, is:
| 1. | 8 | 2. | 6 |
| 3. | 7 | 4. | 10 |
Which of the following is the strongest reducing agent?
[Given: \(E_{Cl_{2}/Cl^{-}}^{o} = 1.36 V\); \(E_{Cr^{3+}/Cr}^{o} = -0.74 V\);
\(E_{Cr_20_7^{2-}/Cr^{3+}}^{o} = 1.33 V\) and \(E_{MnO_4^{-}/Mn^{2+}}^{o} = 1.51 V\)]
| 1. | Cr3+ | 2. | Cl– |
| 3. | Cr | 4. | Mn2+ |
The most reactive species among the following is:
1.
2.
3.
4. ICI
The time for the half-life period of a certain reaction A → Products is 1 hour. When the initial concentration of the reactant 'A' is 2.0 mol L-1, the time taken for its concentration to come from 0.50 to 0.25 mol L-1,if it is a zero-order reaction, is:
| 1. | 1h | 2. | 4 h |
| 3. | 0.5 h | 4. | 0.25 h |
| Assertion (A): | Nitrogen and oxygen are the main components in the atmosphere but these do not react to form oxides of nitrogen. |
| Reason (R): | The reaction between nitrogen and oxygen requires high temperature. |
| 1. | (A) is False but (R) is True. |
| 2. | Both (A) and (R) are False. |
| 3. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
Aluminium oxide may be electrolysed at 1000 °C to furnish aluminium metal.
The cathode reaction is Al3+ + 3e- → Al.
To prepare 5.12 kg of aluminium metal by this method would require:
(Atomic mass = 27 amu; 1 faraday=96,500 Coulombs)
1. 5.49×101 C of electricity
2. 5.49×104 C of electricity
3. 1.83×107 C of electricity
4. 5.49×107 C of electricity
In which of the following will the Kharasch effect operate?
1. CH3CH2CH = CH2 + HCI
2. CH3CH2—CH = CH2 + HBr
3. CH3CH = CH—CH3 + HBr
4. CH3CH2CH = CH2 + HI
Among the following acids which has the lowest pKa value?
1. CH3CH2COOH
2. (CH3)2 CH—COOH
3. HCOOH
4. CH3COOH
The conjugate base of \(H_2PO_{4}^{-}\) is:
1. \(PO_{4}^{3-}\)
2. \(P_2O_5\)
3. \(H_3PO_4\)
4. \(HPO_{4}^{2-}\)
Which of the following is the standard enthalpy of formation of carbon monoxide (CO), if the standard enthalpies of combustion of carbon (C) and carbon monoxide (CO) respectively are −393.5 kJ mol⁻¹ and −283 kJ mol⁻¹?
1. 110.5 kJ
2. 676.5 kJ
3. -676.5 kJ
4. -110.5 kJ
The compound that does not have sp2 hybridized carbon among the following is:
1. Acetone
2. Acetic acid
3. Acetonitrile
4. Acetamide
According to Fajan's rule polarization is more with:
1. Small cation and large anion
2. Small cation and small anion
3. Large cation and large anion
4. Large cation and small anion
| List-I (Molecules) |
List-II (Shape) |
||
| (a) | NH3 | (i) | Square pyramidal |
| (b) | ClF3 | (ii) | Trigonal bipyramidal |
| (c) | PCl5 | (iii) | Trigonal pyramidal |
| (d) | BrF5 | (iv) | T-shape |
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iii) | (iv) | (i) |
| 2. | (iii) | (iv) | (ii) | (i) |
| 3. | (iv) | (iii) | (i) | (ii) |
| 4. | (iii) | (iv) | (i) | (ii) |
| 1. | 0.36 | 2. | 3.6 × 10–2 |
| 3. | 3.6 × 10–3 | 4. | 3.6 |
| 1. |  |
2. |  |
| 3. |  |
4. |  |
 is:
is:
| 1. | sp2 | 2. | spd2 |
| 3. | sp2d | 4. | sp3 |
| 1. | \( Rate =k[A]^0[B]^2 \) | 2. | \( Rate =k[A][B] \) |
| 3. | \(Rate=k[A]^{1 / 2}[B]^2 \) | 4. | \(Rate =k[A]^{-1 / 2}[B]^{3 / 2}\) |
| 1. | 0.05 M NaCl | 2. | 0.1 M KCl |
| 3. | 0.1 M MgSO4 | 4. | 0.1 M NaCl |
| I: | Cr2+ and Mn3+ have the same electronic configuration. |
| II: | Cr2+ is a reducing agent, while Mn3+ is an oxidising agent. |
| III: | Cr2+ is an oxidising agent, while Mn3+ is a reducing agent. |
| IV: | Both Cr and Mn are oxidising agents. |
The sum of the coordination number and oxidation number of the metal M in the complex [M(en)2(C2O4)]Cl is:
| 1. | Nine (9) | 2. | Six (6) |
| 3. | Seven (7) | 4. | Eight (8) |
A substance that gives a brick red flame and breaks down
on heating to give oxygen and a brown gas is:
1. Calcium carbonate
2. Magnesium carbonate
3. Calcium nitrate
4. None of the above
The cell that will measure the standard electrode potential of a copper electrode is:
| 1. | Pt(s) | H2(g, \(1 \over 10\) bar) | H+(aq, 1M) || Cu2+(aq, 1M) | Cu |
| 2. | Pt(s) | H2(g, 1 bar) | H+(aq, 1M) || Cu2+ (aq, 2M) | Cu |
| 3. | Pt(s) | H2(g, 1 bar) | H+(aq, 1M)|| Cu2+ (aq, 1M) | Cu |
| 4. | Pt(s) | H2(g, \(1 \over 10\) bar) | H+(aq, \(1 \over 10\)M) || Cu2+(aq, 1M) | Cu |
|
Column I
(Thermodynamic/Kinetic Principle)
|
Column II
(Impact on Reaction Dynamics)
|
||
| A. | Catalyst alters the rate of reaction | 1. | Proper orientation is not always there |
| B. | 2. | By lowering the activation energy | |
| C. | Energetically favorable reactions are sometimes slow | 3. | Total probability is one |
| D. | The area under the Maxwell-Boltzmann curve is constant | 4. | Refers to the fraction of molecules with energy equal to or greater than the activation energy |
Codes:
| A | B | C | D | |
| 1. | 2 | 4 | 1 | 3 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 3 | 4 | 1 | 2 |
Monochlorination of toluene in sunlight followed by hydrolysis with aq. NaOH yields:
1. o-Cresol
2. m-Cresol
3. 2, 4- Dihydroxytoluene
4. Benzyl alcohol
The most reactive compound among the following toward nucleophilic addition reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
One mole of oxygen gas at STP is equal to:
| (a) | 6 .022× 1023 molecules of oxygen |
| (b) | 6.022 × 1023 atoms of oxygen |
| (c) | 16 g of oxygen molecule |
| (d) | 32 g of oxygen |
Choose the correct answer from the option given below:
1. (a) and (b)The ion that has sp3d2 hybridization for the central atom is:
1. [ICl4]–
2. [ICl2]–
3. [BrF2]–
4. [IF6]–
1 g of a non-volatile non-electrolyte solute is dissolved in 100 g of two different solvents A and B whose ebullioscopic constants are in the ratio of 1 : 5. The ratio of the elevation in their boiling points, , is:
1. 5:1
2. 1:2
3. 10:1
4. 1:5
In which one of the following equilibria, Kp \(\ne\) KC?
1.
2.
3.
4.
For one mole of an ideal gas, which of these statements must be true?
(I) U and H each depend only on temperature.
(II) Compressibility factor z is not equal to 1.
(III) CP, m – CV, m = R
(IV) dU = CVdT for any process.
1. (I), (III) and (IV)
2. (II), (III) and (IV)
3. (III) and (IV)
4. (I) and (III)
The compound that has the largest H–M–H bond angle (M=N, O, S, C), is:
| 1. | H2O | 2. | CH4 |
| 3. | NH3 | 4. | H2S |
Given the following reaction:

| 1. | 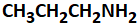 |
2. |  |
| 3. |  |
4. |  |
The false statement among the following is:
| 1. | Carius tube is used in the estimation of sulphur in an organic compound |
| 2. | Carius method is used for the estimation of nitrogen in an organic compound |
| 3. | Phosphoric acid produced on oxidation of phosphorus present in an organic compound is precipitated as Mg2P2O7 by adding magnesia mixture. |
| 4. | Kjeldahl's method is used for the estimation of nitrogen in an organic compound |
The IUPAC name of the following compound is:
1. 4-Bromo-3-cynaophenol
2. 2-Bromo-5-hydroxybenzonitrile
3. 2-Cyano-4-hydroxybromobenzene
4. 6-Bromo-3-hydroxybenzonitrile
The diagram shows stress v/s strain curve for materials \(A\) and \(B\). From the curves, we infer that:
| 1. | \(A\) is brittle but \(B\) is ductile. |
| 2. | \(A\) is ductile and \(B\) is brittle. |
| 3. | Both \(A\) and \(B\) are ductile. |
| 4. | Both \(A\) and \(B\) are brittle. |
A thin smooth rod of length \(L\) and mass \(M\) is rotating freely with angular speed \(\omega_0\)
| 1. | \( \dfrac{M \omega_0}{M+3 m} \) | 2. | \(\dfrac{M \omega_0}{M+2 m} \) |
| 3. | \(\dfrac{M \omega_0}{M+m} \) | 4. | \(\dfrac{M \omega_0}{M+6 m}\) |
| 1. | \(3 \over 2\) | 2. | \(2 \over 3\) |
| 3. | \(4 \over 9\) | 4. | \(9 \over 4\) |
Each side of a cubic box made of metal sheet is '\(a\)' at room temperature '\(T\)'. The coefficient of linear expansion of the metal sheet is '\(\alpha\)'. The metal sheet is heated uniformly by a small temperature\(\Delta T\) so that its new temperature is \(T+\Delta T\). Calculate the coefficient of volume expansion of the metal sheet:
1. \(\alpha\)
2. \(2\alpha\)
3. \(3\alpha\)
4. \(4\alpha\)
The electric field in a region is given \(\vec{E}=\dfrac{4}{5}\hat{j}~\text{N/C}\). The electric flux (in SI units) through the rectangular surface of area \(5~\text{m}^2\) (parallel to \(XZ\)-plane) is:
1. \(1\)
2. \(2\)
3. \(3\)
4. \(4\)
\(2\) moles of a diatomic gas are enclosed in a vessel. When a certain amount of heat is supplied, \(50\%\) of the gas molecules get dissociated, but there is no rise in temperature. What is the heat supplied, if the temperature is \(T\)?
| 1. | \(RT\) | 2. | \(\frac{RT}{2}\) |
| 3. | \(\frac{11RT}{2}\) | 4. | \(5RT\) |
If the speed of sound in air is \(v,\) then the minimum possible length of the closed-end organ pipe which resonates to frequency \(f\) will be:
1. \(\dfrac{v}{2f}\)
2. \(\dfrac{v}{4f}\)
3. \(\dfrac{v}{3f}\)
4. \(\dfrac{v}{f}\)
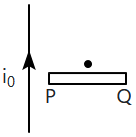
| 1. | clockwise | 2. | anticlockwise |
| 3. | along \(PQ\) | 4. | does not rotate |
| 1. | \(\dfrac{\varepsilon{B}}{l}\) | 2. | \(\dfrac{\varepsilon}{Bl}\) |
| 3. | \(\dfrac{{3}\varepsilon}{2Bl}\) | 4. | \(\dfrac{{2}\varepsilon}{3Bl}\) |
Rings are rotated and translated in a uniform magnetic field as shown in the figure. Arrange the magnitude of emf induced across \(AB\):

| 1. | \(\mathrm{emf_{a}<emf_{b}<emf_{c}}\) |
| 2. | \(\mathrm{emf_{a}=emf_{b}<emf_{c}}\) |
| 3. | \(\mathrm{emf_{a}={emf}_{c}<{emf}_{b}}\) |
| 4. | \(\mathrm{emf_{a}<emf_{b}={emf}_{c}}\) |
| 1. | The force between the proton beams will be attractive. |
| 2. | The magnetic force between proton beams will be attractive. |
| 3. | Repulsive forces will be smaller than attractive forces. |
| 4. | The magnetic and electrostatic forces will cancel each other. |
| 1. | \({I}_{C}~ {=} ~{0}{,}\;{I}_{d}\ne{0}\) |
| 2. | \({I}_{C}\ne{0}{,}\;{I}_{d}~{=}~{0}\) |
| 3. | \(I_{C}=I_{D}\) but in the same direction |
| 4. | \(I_{C}=I_{D}\) but in the opposite direction |
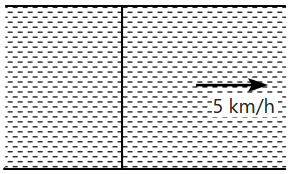
A uniform cable of mass \(M\) and length \(L\) is placed on a horizontal surface such that its \(\left ( \dfrac{1}{n} \right )^\text{th}\) part is hanging below the edge of the surface. To lift the hanging part of the cable up to the surface, the work done should be:
| 1. | \(nMgl\) | 2. | \(\dfrac{MgL}{2n^2}\) |
| 3. | \(\dfrac{2MgL}{n^2}\) | 4. | \(\dfrac{4MgL}{n^2}\) |
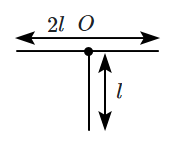
| 1. | \(\dfrac{2}{3}{Ml}^{2}\) | 2. | \({Ml}^{2}\) |
| 3. | \(\dfrac{{Ml}^{2}}{3}\) | 4. | \(\dfrac{{Ml}^{2}}{2}\) |
| 1. | \(0\) unit | 2. | \(2\) unit |
| 3. | \(4\) unit | 4. | \(3\) unit |
| 1. | \(16~\text{J}\) | 2. | \(32~\text{J}\) |
| 3. | \(64~\text{J}\) | 4. | \(128~\text{J}\) |
| 1. | \( \sin ^{-1}\left(\dfrac{1}{4}\right) \) | 2. | \( \sin ^{-1}\left(\dfrac{1}{2}\right) \) |
| 3. | \( \sin ^{-1}\left(\dfrac{2}{3}\right) \) | 4. | \( \sin ^{-1}\left(\dfrac{3}{4}\right)\) |
| 1. | \(\mathit{\gamma} {:}\mathit{\gamma}{-}{1}{:}\frac{1}{\mathit{\gamma}} \) | 2. | \({1}{:}{1}{:}\mathit{\gamma}{-}{1}\) |
| 3. | \(\mathit{\gamma}{:}\mathit{\gamma}{-}{1}{:}{1}\) | 4. | \(\mathit{\gamma}{:}{1}{:}\mathit{\gamma}{-}{1}\) |
| 1. | \({2}\mathit{\pi}{L}\) | 2. | \(\dfrac{L}{\sqrt{{2}\mathit{\pi}}}\) |
| 3. | \(L\) | 4. | \(\dfrac{L}{{2}\mathit{\pi}}\) |
| 1. | the depletion region is reduced and barrier height is increased. |
| 2. | the depletion region is widened and barrier height is reduced. |
| 3. | both the depletion region and barrier height are reduced. |
| 4. | both the depletion region and barrier height are increased. |
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. | \(\frac{\lambda}{{\lambda}'}=\frac{1}{2}\) | 2. | \(1>\frac{\lambda}{{\lambda}{'}}>\frac{1}{2}\) |
| 3. | \(\frac{\lambda}{{\lambda}{'}}<\frac{1}{2}\) | 4. | \(\frac{\lambda}{{\lambda}{'}}>1\) |
| 1. | \({r}_{n}\propto{n}^{1/3}\) | 2. | \({r}_{n}\propto{n}^{2/3}\) |
| 3. | \({r}_{n}\propto{n}^{}\) | 4. | \(r_{n}\) is independent of \(n\) |
| Assertion (A): | A biconvex lens of focal length \(10\) cm is split into two equal parts by a plane parallel to its principal axis. The focal length of each part will be \(20\) cm. |
| Reason (R): | Focal length does not depend on the radii of curvature of two surfaces. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
| 1. | \(f_o=45~\text{cm}~\text{and}~f_e=-9 ~\text{cm}\) |
| 2. | \(f_o=7.2~\text{cm}~\text{and}~f_e=5 ~\text{cm}\) |
| 3. | \(f_o=50~\text{cm}~\text{and}~f_e=10 ~\text{cm}\) |
| 4. | \(f_o=30~\text{cm}~\text{and}~f_e=6 ~\text{cm}\) |
A current-carrying wire is placed below a coil in its plane, with current flowing as shown in the figure. If the current increases,:

| 1. | no current will be induced in the coil |
| 2. | an anticlockwise current will be induced in the coil |
| 3. | a clockwise current will be induced in the coil |
| 4. | the current induced in the coil will be first anticlockwise and then clockwise |
| 1. | \(e^{-\omega t} \) | 2. | \(\text{sin}\omega t\) |
| 3. | \(\text{sin}\omega t+\text{cos}\omega t\) | 4. | \(\text{sin}(\omega t+\pi/4) \) |
| 1. | decrease continuously with mass number. |
| 2. | first decreases and then increases with an increase in mass number. |
| 3. | first increases and then decreases with an increase in mass number. |
| 4. | increases continuously with mass number. |
| Statement I: | Photodiodes are operated in reverse biased. |
| Statement II: | Current in forward bias is more than current in reverse bias in \(\mathrm{p\text-n}\) diode. |
| 1. | Both Statements are true. |
| 2. | Statement I is true and Statement II is false. |
| 3. | Statement I is false and Statement II is true. |
| 4. | Both Statements are false. |
An LED is constructed from a \(\mathrm{p\text{-}n}\) junction diode using \(\mathrm{GaAsP}.\) The energy gap is \(1.9~\text{eV}.\) The wavelength of the light emitted will be equal to:
1. \(10.4 \times 10^{-26}~ \text{m}\)
2. \(654~ \text{nm}\)
3. \(654~ \text{m}\)
4. \(654\times 10^{-11}~\text{m}\)
For a uniform rectangular sheet shown in the figure, if \(I_O\) and \(I_{O'}\) be moments of inertia about the axes perpendicular to the sheet and passing through \(O\) (the centre of mass) and \(O'\) (corner point), then:
| 1. | \(I_{O'}=I_O\) | 2. | \(I_{O'}<I_O\) |
| 3. | \(I_{O'}>I_O\) | 4. | can't say |
\(512\) Identical drops of mercury are each charged to a potential of \(2~\text{V}.\) When these drops are joined to form a single larger drop, the potential of this combined drop will be:
1. \(32~\text{V}\)
2. \(64~\text{V}\)
3. \(96~\text{V}\)
4. \(128~\text{V}\)
The potential energy \((U)\) of a diatomic molecule is a function dependent on \(r\) (interatomic distance) as \(\begin{equation} U=\dfrac{\alpha}{r^{10}}-\dfrac{\beta}{r^{5}}-3 \end{equation}\) where, \(\alpha\) and \(\beta\) are positive constants. The equilibrium distance between two atoms will be:
| 1. | \( \left(\dfrac{2 \alpha}{\beta}\right)^{1 / 7} \) | 2. | \( \left(\dfrac{2 \alpha}{\beta}\right)^{1 / 3} \) |
| 3. | \( \left(\dfrac{2 \alpha}{\beta}\right)^{1 / 4} \) | 4. | \( \left(\dfrac{2 \alpha}{\beta}\right)^{1 / 5}\) |
A planet revolving in elliptical orbit has:
| (A) | a constant velocity of revolution. |
| (B) | the least velocity when it is nearest to the sun. |
| (C) | its areal velocity directly proportional to its velocity. |
| (D) | its areal velocity inversely proportional to its velocity. |
| (E) | to follow a trajectory such that the areal velocity is constant. |
Choose the correct answer from the options given below:
| 1. | (A) only | 2. | (D) only |
| 3. | (C) only | 4. | (E) only |
The trajectory of a projectile in a vertical plane is \(y=\alpha x-\beta x^2\), where \(\alpha\) and \(\beta\) are constants and \(x\) & \(y\) are respectively the horizontal and vertical distances of the projectile from the point of projection. The angle of projection \(\theta\) will be:
1. \(
\tan ^{-1} \alpha
\)
2. \( \tan ^{-1} \beta
\)
3. \( \tan ^{-1} 2 \beta
\)
4. \( \tan ^{-1} 2 \alpha\)
A beam of protons with speed \(4\times 10^5~\text{ms}^{-1}\) enters a uniform magnetic field of \(0.3~\text{T}\) at an angle of \(60^\circ\) to the magnetic field. The pitch of the resulting helical path of protons is close to: (Mass of the proton \(=1.67\times 10^{-27}~\text{kg}\), charge of the proton \(=1.69\times 10^{-19}~\text{C}\))
1. \(12~\text{cm}\)
2. \(4~\text{cm}\)
3. \(5~\text{cm}\)
4. \(2~\text{cm}\)
A spherical solid ball of volume \(V\) is made of a material of density \(\rho_1.\) It is falling through a liquid of density \(\rho_2(\rho_2<\rho_1)\) Assuming that the liquid applies a viscous force on the ball that is proportional to the square of its speed \(v,\) i.e., \(F_{\text{viscous}}=-kv^2(k>0),\) the terminal speed of the ball is:
| 1. | \( \dfrac{{Vg} \rho_1}{k} \) | 2. | \( \sqrt{\dfrac{{Vg} \rho_1}{k}} \) |
| 3. | \( \dfrac{{Vg}\left(\rho_1-\rho_2\right)}{k} \) | 4. | \( \sqrt{\dfrac{{Vg}\left(\rho_1-\rho_2\right)}{k}} \) |
A spring pendulum is taken from the equator to the pole. Its time period:
| 1. | first increases, then decreases |
| 2. | increases |
| 3. | decreases |
| 4. | remains same |
The figure shows the snapshot of a travelling sine wave in a string. Four elemental portions \(a,b,c\) and \(d\) are indicated on the string. The elemental portion with maximum potential energy is/are:
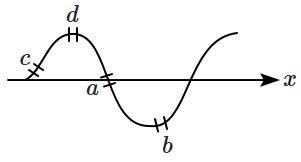
1. \(a\)
2. \(b\)
3. \(c\)
4. \(b \text{ and } d\)
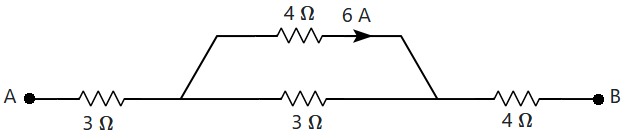
| 1. | \(122\) V | 2. | \(60\) V |
| 3. | \(100\) V | 4. | \(30\) V |
| 1. | \(3~\text{N}\) | 2. | \(2~\text{N}\) |
| 3. | \(1~\text{N}\) | 4. | zero |

| 1. | \(10~\text{J}\) | 2. | \(12~\text{J}\) |
| 3. | \(36~\text{J}\) | 4. | \(6~\text{J}\) |