| 1. | Epicotyl | 2. | Shoot apical meristem |
| 3. | Young leaves | 4. | Hypocotyl |
Which of the following is a primary metabolite?
| 1. | Carotenoid | 2. | Glucose |
| 3. | Morphine | 4. | Cellulose |
| Assertion (A): | Sporopollenin preserves pollen in fossil deposits |
| Reason (R): | Sporopollenin is resistant to physical and biological decomposition |
| 1. | (A) is True; (R) is False |
| 2. | (A) is False; (R) is True |
| 3. | Both (A) and (R) are not True |
| 4. | Both (A) and (R) are True. |
In which one of the following options, the two given taxonomic categories are correctly matched with their organism?
| Taxonomic Categories | Organism | ||
| 1. | Diptera and Felidae | - | Musca |
| 2. | Solanaceae and Polymoniales | - | Datura |
| 3. | Canidae and Primate | - | Gibbon |
| 4. | Triticum and Sapindales | - | Wheat |
Which of the following statements is correct?
| 1. | Organisms that depend on living plants are called saprophytes. |
| 2. | Some of the organisms can fix atmospheric nitrogen in specialized cells called sheath cells. |
| 3. | The fusion of two cells is called Karyogamy. |
| 4. | Fusion of protoplasms between two motile or non-motile gametes is called plasmogamy. |
Which non-radioactive isotope was used by Meselson and Stahl to label DNA in their experiment?
1. \(^{35} S\)
2. \(^{15} N\)
3. \(^{32} P\)
4. None
Which one of the following pairs of codons are correctly matched with their function or the signal for the particular amino acid?
1. GUU, GCU – Alanine
2. UAG, UGA – Stop
3. AUG, ACG – Start/methionine
4. UUA, UCA – Leucine
With reference to protein synthesis, consider the following events
| a. | Binding of aminoacyl - tRNA complex with mRNA |
| b. | Activation of amino acid |
| c. | Polypeptide formation |
| d. | Formation of aminoacyl - tRNA complex |
What is the correct sequence of the above events in protein synthesis?
| 1. | b, d, a & c |
| 2. | a, b, d & c |
| 3. | d, b, a & c |
| 4. | a, d, b & c |
A test cross is carried out to:
| 1. | Predict whether two traits are linked |
| 2. | Assess the number of alleles of a gene |
| 3. | Determine whether two species or varieties will breed successfully |
| 4. | Determine the genotype of a plant at F2 |
Stellar distances are measured in :
1. Meters
2. Nanometers
3. Light years
4. Kilometers
Select the incorrect statement with respect to plant growth regulators:
| 1. | 2, 4-D (2, 4-dichlorophenoxyacetic acid) is a synthetic auxin |
| 2. | Spraying sugarcane crop with gibberellins increases the length of the stem |
| 3. | Cytokinins promote the apical dominance |
| 4. | Ethylene is a gaseous hormone |
The region of meristematic activity has cells of:
1. Small size, thin walled and with dense protoplasm
2. Large size, thick walled and with the loose protoplasm
3. Small size, thin walled with loose protoplasm
4. Large size, thin walled and with dense protoplasm
Which of the following is true for vacuole?
| 1. | It is present in both plants and animals |
| 2. | Central vacuole is present in plants |
| 3. | It occupies 90% volume of the plants |
| 4. | All of these |
Which of the following plants are grown in green house by exploiting the fact that higher CO2 leads to more productivity?
1. Tomatoes
2. Papaya
3. Cycas
4. Pomegranate
How many ATPs are net gained till the completion of Krebs’ cycle during breakdown of one molecule of Glucose?
| 1. | 4 ATPs in Glycolysis and 1 GTP in Krebs’ Cycle |
| 2. | 4 ATPs in Glycolysis and 2 GTP in Krebs’ Cycle |
| 3. | 2 ATPs in Glycolysis and 2 GTP in Krebs’ Cycle |
| 4. | 2 ATPs in Glycolysis and 1 GTP in Krebs’ Cycle |
Lichens are mutually beneficial associations between:
1. Autotrophic and heterotrophic members
2. Two autotrophic partners
3. Two heterotrophic partners
4. Fungi and roots of higher plants
Which of the following is false?
| I. | Endosperm formation starts prior to first division of zygote |
| II. | Angiospermic endosperm is mostly 3N while gymnospermic one is N. |
| III. | The most common type of endosperm is nuclear. |
| IV. | Coconut has both liquid nuclear (multinucleate) and cellular endosperm. |
| V. | Milky water of green tender coconut is liquid female gametophyte. |
| 1. | I and II only |
| 2. | III only |
| 3. | V only |
| 4. | II only |
According to extreme estimates, which say that there is 20 to 50 million global species diversity is based on:
1. Species richness of insects
2. Species richness of Arthropods
3. Species evenness of insects
4. Species evenness of Arthropods
The cylindrical body or curved base of a bioreactor actually facilitates:
1. The handling and maintenance of bioreactor
2. The mixing of the reactor content
3. Better oxygen transport
4. More accumulation of product
Isotopes used in proving semiconservative replication of DNA were:
| 1. | 14N 14C | 2. | 14N 15N |
| 3. | 14N 31P | 4. | 14C 31P |
Which of the following is released into natural water bodies like rivers and streams?
1. Effluent of primary treatment
2. Effluent of secondary treatment
3. Settling component of secondary treatment
4. Settling component of primary treatment
Mycorrhiza is an example of:
1. Symbiotic relationship
2. Ectoparasitism
3. Endoparasitism
4. Decomposers
Which of the following shows whorled phyllotaxy?
| 1. | Mustard | 2. | China rose |
| 3. | Alstonia | 4. | Calotropis |
| 1. | pollen exine where sporopollenin is present |
| 2. | pollen exine where sporopollenin is absent |
| 3. | pollen intine where hemicellulose is present |
| 4. | pollen intine where hemicellulose is absent |
| 1. | 1 | 2. | 2 |
| 3. | 4 | 4. | 7 |
Most favourite and ideal material for researchers in genetics is:
| 1. | Housefly | 2. | Mosquito |
| 3. | Frog | 4. | Fruitfly. |
Which of the following are the important floral rewards to the animal pollinators?
1. Nectar and pollen grains
2. Floral fragrance and calcium crystals
3. Protein pellicle and stigmatic exudates
4. Colour and large size of flower
Commonly used vectors for human genome sequencing are:
1. T - DNA
2. BAC and YAC
3. Expression Vectors
4. T/A Cloning Vectors
| I: | The sex organs in bryophytes are multicellular. |
| II: | The male sex organ is called antheridium and they produce biflagellate antherozoids. |
| III: | The female sex organ called archegonium is flask-shaped and produces a single egg. |
| IV: | The antherozoids are released into water where they come in contact with archegonium. |
| V: | Zygotes undergo reduction division immediately. |
| 1. | Palms | 2. | Soybean |
| 3. | Potato | 4. | Barley |
| Assertion (A): | The use of baculovirus as biocontrol agents is especially desirable when an ecologically sensitive area is being treated. |
| Reason (R): | Baculoviruses are species-specific, narrow spectrum insecticides. |
| 1. | Both (A) and (R) are True and (R) correctly explains (A). |
| 2. | (A) is True; (R) is False |
| 3. | (A) is False; (R) is False |
| 4. | Both (A) and (R) are True but (R) does not correctly explain (A) |
| I: | The conversion of glucose into glucose 6-phosphate. |
| II: | The conversion of fructose 6-phosphate to fructose 1, 6-bisphosphate. |
Which of the following plastid store fats?
| 1. | Elaioplast | 2. | Aleuroplast |
| 3. | Proteinoplast | 4. | Amyloplast |
Consider the given two statements:
| I: | The term lenticel is usually associated with the breakage of periderm tissue; however, lenticels also refer to the lightly colored spots found on apples. |
| II: | Both structures have similar functions in gas exchange. |
1. Both I and II are correct and II explains I
2. Both I and II are correct but II does not explain I
3. I is correct but II is incorrect
4. I is incorrect but II is correct
In the internodal segments of tobacco stems, the callus is actually:
| 1. | An undifferentiated cell |
| 2. | A mass of differentiated cell |
| 3. | A mass of undifferentiated cell |
| 4. | A differentiated cell |
Mark the incorrect one:
| 1. | Pollen consumption has been claimed to increase the performance of athletes and race horses |
| 2. | Viability of pollen grain depends upon temperature, not humidity |
| 3. | Pollen grains of a large number of species can be stored in liquid nitrogen for years |
| 4. | Pollen grains of rice and wheat lose viability within half an hour |
Which of the following are true for cyanobacteria?
| (I) | They are freshwater/marine or terrestrial algae. |
| (II) | Often form blooms in unpolluted water bodies. |
| (III) | Maybe unicellular, colonial, or filamentous. |
| (IV) | Some of them fix atmospheric nitrogen. |
The lateral branches originate from the basal and underground portion of the main stem, grow horizontally beneath the soil, and then come out obliquely upward to give rise to leafy shoot. Which of the following examples are associated with such modifications?
1. Banana, Mint, Jasmine
2. Banana, Pineapple, Chrysanthemum
3. Pineapple, Mint, Citrus
4. Papaya, Citrus, Banana
Select the two correct statements out of the four (a-d) given below about gymnosperms:
| a: | Roots in some non-vascular archegoniate have a fungal association in the form of mycorrhiza |
| b: | Leaves are well adapted to withstand extremes of temperature, humidity, and wind |
| c: | The nucellus is protected by the ovary wall and the composite structure is called an ovule |
| d: | Multicellular female gametophyte is retained within the megasporangium |
| 1. | a and c | 2. | b and c |
| 3. | c and d | 4. | b and d |
Select the incorrect statement
| 1. | All living organisms have enzymatic machinery to partially oxidise glucose without oxygen |
| 2. | In plants, glucose is derived from sucrose which is the end product of photosynthesis |
| 3. | 2ATP are produced as net gain by direct phosphorylation during glycolysis |
| 4. | One dehydrogenation and one decarboxylation step occur during EMP pathway |
Read the following statements with respect to antenna molecules:
| a: | The LHC are made up of hundreds of pigment molecules bound to proteins. |
| b: | Water splitting complex is associated with the PS-ll. |
| 1. | Only b is correct |
| 2. | Both a & b are incorrect |
| 3. | Only a is correct |
| 4. | Both a & b are correct |
Which of the given plant produces cardiac glycosides?
| 1. | Calotropis | 2. | Acacia |
| 3. | Nepenthes | 4. | Utricularia |
The motion of sister Chromatids towards opposite poles of the cell occurs by:
| 1. | Shortening of microtubules attached to centromere |
| 2. | Shortening of microtubules attached to kinetochores |
| 3. | Shortening of microtubules attached to Metaphase plate |
| 4. | Shortening of microtubules attached to other homologous chromosomes |
| I: | is a type of apomixes |
| II: | occurs in many citrus varieties |
| Assertion (A): | The underlying mechanism of pleiotropy in most cases is the effect of a gene on metabolic pathways which contribute towards different phenotypes. |
| Reason (R): | Pleiotropy occurs when one gene influences two or more seemingly unrelated phenotypic traits. |
| 1. | (A) is False but (R) is True |
| 2. | (A) is True but (R) is False |
| 3. | Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 4. | Both (A) and (R) are True but (R) is not the correct explanation of (A) |
| 1. | Antibiotics are chemical substances, which are produced by some microbes and can kill or retard the growth of other (disease-causing) microbes. |
| 2. | Penicillin was the first antibiotic to be discovered, and it was a chance discovery. |
| 3. | Alexander Fleming while working on Staphylococci bacteria, discovered Penicillin in the mould Penicillium chrysogenum. |
| 4. | Penicillin’s full potential as an effective antibiotic was established much later by Ernest Chain and Howard Florey. |
For a given lead configuration, the ECGs obtained from _________ individuals have _________ shape.
1. different, different
2. different, same
3. different, roughly same
4. Twins, same
High pCO2 and low pO2 can be found in:
| 1. | alveoli | 2. | tissues |
| 3. | blood | 4. | heart |
Which structures perform the function of mitochondria in bacteria ?
1. Nucleoid
2. Ribosomes
3. Cell wall
4. Mesosomes
Given below is the structure of a monosaccharide. The homopolymer of this monosaccharide is used in:
| 1. | Storage of carbohydrates in plants |
| 2. | Storage of carbohydrates in animals |
| 3. | Making the medium for microbial culture |
| 4. | Formation of the structure of the plant cell wall |
Identify the option where all the columns are not correctly matched:
|
Animal |
Phylum |
Features |
|
|
1. |
Pleurobrachia |
Ctenophora |
Comb plates, |
|
2. |
Loligo |
Mollusca |
Radula, |
|
3. |
Balanoglossus |
Hemichordata |
Respiration by gills, |
|
4. |
Ascidia |
Chordata |
Marine, |
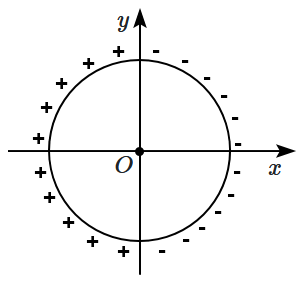
Which of the following symbols represent consanguineous mating?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Choose the incorrect statement with respect to natural selection theory:
| 1. | The unit of natural selection is an individual |
| 2. | Darwin implied natural selection as a method of evolution |
| 3. | Reproductively fit organisms will survive more and hence are selected by nature |
| 4. | Increase in the number of melanised moths during post-industrialisation is not an example of natural selection |
SAN is present in __________ while AVN is present in__________.
| 1. | Right upper corner and left lower corner of both the atria. |
| 2. | Right upper corner and left lower corner of right atrium. |
| 3. | Right upper corner and left lower corner of left atrium. |
| 4. | Right upper corner and left median corner of left atrium. |
In the given diagram, the columns of Bertini are represented by the letter:
| 1. | A | 2. | B |
| 3. | C | 4. | D |
Given below are the statements related to the functioning of different parts of the human brain with respect to the labelled structure in the diagram showing sagital section of human brain.
| a. | A is a major coordinating centre for sensory and motor signalling. |
| b. | B controls body temperature, urge for eating and drinking. |
| c. | C along with amygdala and hippocampus involved in regulation of sexual behaviour, expression of emotional reaction. |
| d. | D converts short term memories into long term memories. |
How many statements are correct?
| 1. | One | 2. | Two |
| 3. | Three | 4. | Four |
Select the correct match:
1. Zona reticularis – inner layer of adrenal medulla
2. Zona fasciculata – middle layer of adrenal cortex
3. Zona glomerulosa – outer layer of adrenal medulla
4. Zona fasciculata – middle layer of adrenal medulla
Restriction endonucleases are enzymes that:
| 1. | make cuts at specific positions within the DNA molecule |
| 2. | recognize a specific nucleotide sequence for binding of DNA ligase |
| 3. | restrict the action of the enzyme DNA polymerase |
| 4. | remove nucleotides from the ends of the DNA molecule |
Which of the following genes control the cotton bollworm?
1. cry IAc and cry IIAb
2. cry IIAb and cry IIAc
3. cry IAb and cry IAc
4. cry IAb and cry IIAc
| 1. | ori | 2. | rop |
| 3. | ampR | 4. | tetR |
| 1. | RDT | 2. | Urine analysis |
| 3. | PCR | 4. | ELISA |
Pelvic girdle is made up of:
1. Two coxal bones
2. Two sacral bones
3. Two Femur bones
4. Two glenoid cavity
In which of the chromosomes two equal arms are present?
1. sub-metacentric chromosome
2. metacentric chromosome
3. acrocentric chromosome
4. telocentric chromosome
| 1. | Black | 2. | White |
| 3. | Green | 4. | Orange |
In a growing population of a country:
| 1. | pre-reproductive individuals are more than reproductive individuals. |
| 2. | reproductive individuals are less than post-reproductive individuals. |
| 3. | reproductive and pre-reproductive individuals are equal in number. |
| 4. | pre-reproductive individuals are less than reproductive individuals. |
Which of the following cell will undergo meiosis?
1. Sertoli cells
2. Spermatogonia
3. Leydig cell
4. Immunologically competent cells
Which of the following is true?
| 1. | Gills are used by most of the aquatic animals |
| 2. | lungs are used by most of the terrestrial animals |
| 3. | Mammals have a well developed respiratory system |
| 4. | All of the above |
Which of the following hormones is responsible for both the milk ejection reflex and the foetal ejection reflex?
| 1. | Estrogen | 2. | Prolactin |
| 3. | Oxytocin | 4. | Relaxin |
Match the items in Column-I with those in Column-II:
| Column-I | Column-II | ||
| (a) | Podocytes | (i) | Crystallised oxalate |
| (b) | Protonephridia | (ii) | Annelids |
| (c) | Nephridia | (iii) | Amphioxus |
| (d) | Renal calculi | (iv) | Filtration slits |
Select the correct option from the following:
1. (a)-(iii), (b )-(iv), (c)-(ii), (d)-(i)
2. (a)-(iii), (b)-(ii), (c)-(iv), (d)-(i)
3. (a)-(iv), (b)-(iii), (c)-(ii), (d)-(i)
4. (a)-(iv), (b)-(ii), (c)-(iii), (d)-(i)
A selectable marker is used to :
| 1. | help in eliminating the non-transformants so that the transformants can be regenerated |
| 2. | identify the gene for the desired trait in an alien organism |
| 3. | select a suitable vector for transformation in a specific crop |
| 4. | mark a gene on a chromosome for isolation using restriction enzyme |
Which of the following sexually transmitted diseases is not completely curable?
| 1. | Chlamydiosis | 2. | Gonorrhoea |
| 3. | Genital warts | 4. | Genital herpes |
Drug called 'Heroin' is synthesized by:
| 1. | nitration of morphine | 2. | methylation of morphine |
| 3. | acetylation of morphine | 4. | glycosylation of morphine |
Variations caused by mutation, as proposed by Hugo de Vries are:
1. small and directionless
2. random and directional
3. random and directionless
4. small and directional
Match the following columns and select the correct option:
| Column-I | Column-II | ||
| (1) | Eosinophils | (i) | lmmune response |
| (2) | Basophils | (ii) | Phagocytosis |
| (3) | Neutrophils | (iii) | Release histaminase, destructive enzymes |
| (4) | Lymphocytes | (iv) | Release granules containing histamine |
| (1) | (2) | (3) | (4) | |
| 1. | (iv) | (i) | (ii) | (iii) |
| 2. | (i) | (ii) | (iv) | (iii) |
| 3. | (ii) | (i) | (iii) | (iv) |
| 4. | (iii) | (iv) | (ii) | (i) |
Match the following diseases with the causative organism and select the correct option:
| Column I | Column II |
| (a) Gregarious, polyphagous pest | (i) Asterias |
| (b) Adult with radial symmetry and larva with bilateral symmetry | (ii) Scorpion |
| (c) Book lungs | (iii) Ctenoplana |
| (d) Bioluminescence | (iv) Locusta |
| (a) | (b) | (c) | (d) | |
| 1. | (iv) | (i) | (ii) | (iii) |
| 2. | (iii) | (ii) | (i) | (iv) |
| 3. | (ii) | (i) | (iii) | (iv) |
| 4. | (i) | (iii) | (ii) | (iv) |
To induce uterine contractions for parturition which of the following hormones can be injected to the female?
1. hCG
2. Estrogen
3. Progesterone
4. Oxytocin
Which one of the following is False?
1. Fatty acids and glycerol are soluble in water
2. Phospholipids are found in the cell membrane
3. Oils have lower melting point
4. In lipids fatty acids are found esterified with glycerol
| Assertion (A): | Morphine should not be routinely used in patients with mild to moderate pain. |
| Reason (R): | Morphine increases sensitivity to pain. |
| 1. | Both (A) and (R) are True and (R) correctly explains (A). |
| 2. | (A) is True; (R) is False |
| 3. | (A) is False; (R) is True |
| 4. | Both (A) and (R) are True but (R) does not correctly explain (A). |
| Assertion (A): | Hormones act only on their target cells. |
| Reason (R): | Hormones are transported to their target organs by ducts. |
| 1. | Both (A) and (R) are True and (R) correctly explains (A). |
| 2. | (A) is True but (R) is False |
| 3. | (A) is False but (R) is False |
| 4. | Both (A) and (R) are True but (R) does not correctly explain (A). |
| Assertion (A): | Deficiency of ADH causes diabetes insipidus. |
| Reason (R): | ADH promotes the expulsion of water from the collecting duct, a portion of the nephron. |
| 1. | Both (A) and (R) are True and (R) correctly explains (A). |
| 2. | (A) is True; (R) is False |
| 3. | Both (A) and (R) are False |
| 4. | Both (A) and (R) are True but (R) does not correctly explain (A). |
Pneumotaxic centre which can moderate the functions of the respiratory rhythm centre is present at:
1. Pons region of brain
2. Thalamus
3. Spinal cord
4. Right cerebral hemisphere
| 1. | A thin muscular wall | 2. | A thick muscular wall |
| 3. | A thin fibrous tissue | 4. | A thick fibrous tissue |
The muscle can directly use which of the following as a source of energy?
| 1. | Creatine phosphate | 2. | Glucose |
| 3. | ATP | 4. | Fatty acids |
Which of the following is true about the Endocrine system?
1. It is not directly connected with the organs
2. Response is slow
3. Coordinates through secretion of hormones
4. All of the above
How many seminiferous tubules will be there per testis?
| 1. | 1 to 3 | 2. | 500 |
| 3. | 1000 | 4. | At maximum, 4 |
Which of the following is correct regarding colostrum ?
| 1. | Rich in antigens |
| 2. | Rich in antibodies |
| 3. | Produced upto 6 months (lactation period) |
| 4. | lts release is blocked by low levels of prolactin |
Choose the incorrect option with respect to assisted reproductive technologies:
| 1. | In IUT, embryos with upto 8 blastomeres are transferred into the uterus |
| 2. | In ZIFT, zygote can be transferred into the fallopian tube |
| 3. | In AI, the semen collected either from husband or healthy donor is artificially introduced either into the vagina or into the uterus |
| 4. | During IVF, fertilization occurs outside the body in almost similar conditions as that in the body |
Select the correct statement:
| 1. | Human cell divide once approximately every 24 hours |
| 2. | Centrioles duplicates in the cytoplasm during G2 - phase |
| 3. | In the quiescent stage, cells remain metabolically inactive |
| 4. | During G1 - stage cell is metabolically active but does not grow |
Mendelian disorders are mainly determined by:
1. Alteration or mutation in a single gene.
2. Chromosomal gross structural changes.
3. Recombination between linked genes.
4. Jumping genes
Which of the following is not the example of evolution by anthropogenic actions?
1. Herbicide resistant varieties
2. Antibiotic resistant microbes
3. Darwin Finches
4. Different breeds of dogs
Read the following four statements A, B, C and D which are the descriptions of some events in the life cycle of Plasmodium and select the correct option having a set of correct statements
| A. | Parasite reproduces asexually in red blood cells. The rupture of RBCs is associated with the release of a toxic substance, haemozoin, which is responsible for chills and high fever |
| B. | Sexual stages (gametocytes) develop in the intestine of man |
| C. | Fertilisation and development take place in the mosquito's intestine |
| D. | Female mosquito takes up the gametocyte with blood meal |
1. A & B
2. B & C
3. A & D
4. C & D
The linking of antibiotic resistance gene with the plasmid vector became possible due to:
| 1. | DNA polymerase | 2. | Exonucleases |
| 3. | DNA ligase | 4. | Endonucleases |
| I: | All living members are ectoparasites on some fishes. |
| II: | They have an elongated body bearing 6-15 pairs of gill slits for respiration. |
| III: | They have a sucking and circular mouth without jaws. |
| IV: | Their body has scales and paired fins. |
| V: | Circulation is of open type. |
| VI: | Cyclostomes are fresh water animals but migrate for spawning to marine water. |
| I: | facilitates water reabsorption from latter parts of the tubule, thereby preventing diuresis. |
| II: | is a potent vasodilator. |
| 1. | corpus striatum | 2. | corpus callosum |
| 3. | corpus luteum | 4. | corpus unigemina |
Parazoa is a taxon at the base of the phylogenetic tree of the animal kingdom and includes the most primitive forms, characterized by not having proper tissues. The phylum/phyla included in this taxon will be:
| I. Porifera | II. Cnidaria | III. Ctenophora |
| 1. | Only I | 2. | Only II and III |
| 3. | Only I and II | 4. | I, II and III |
Metallic sodium is fused with organic compounds for testing nitrogen, sulphur, and halogens. The correct reason behind this is:
| 1. | To convert all compounds to their ionic form. |
| 2. | Sodium reduces the compounds. |
| 3. | Sodium converts all compounds in their covalent form. |
| 4. | None of the above. |
Chloromethane on treatment with excess of ammonia yields mainly:
| 1. | N, N-dimethylmethanamine |
| 2. | N-methylmethanamine (CH3 —NH—CH3 ) |
| 3. | Methanamine (CH3 NH2 ) |
| 4. | Mixture containing all these in equal proportion. |
What is the product formed when methylamine reacts with nitrous acid?
| 1. | CH3-O-N=O | 2. | CH3-O-CH3 |
| 3. | CH3-OH | 4. | CH3CHO |
Proteins are found to have two different types of secondary structures via -helix and -pleated sheet structure. The -helix structure of a protein is stabilized by-
1. Peptide bonds
2. Van der waals forces
3. Hydrogen bonds
4. Dipole-dipole interactions
The degenerate orbitals of \(\left[\right. C r \left(H_{2} O\right)_{6} ]^{3 +}\) are:
1. \(d_{z^2}\) and \(d_{x z}\)
2. \(d_{y z}\) and \(d_{z^2}\)
3. \(d_{xz}\) and \(d_{y z}\)
4. \(d_{x^{2} - y^{2}}\) and \(d_{x y}\)
On heating compound (A) gives a gas (B) which is a constituent of air. This gas when treated with H2 in the presence of a catalyst gives another gas (C) which is basic in nature. (A) should not be-
1. (NH4)2Cr2O7
2. Pb(NO3)2
3. NaN3
4. NH4NO2
| 1. | 5 | 2. | 4 |
| 3. | 6 | 4. | 8 |
The number of chiral centers present in [B] is-
| 1. | 5 | 2. | 6 |
| 3. | 4 | 4. | 2 |
Electromagnetic radiation of wavelength 663 nm is just sufficient to ionize the atom of metal A. The ionization energy of metal A in kJ mol–1 is: (Rounded-off to the nearest integer)
[h = 6.63 × 10–34 Js, c = 3.00 × 108 ms–1 , NA = 6.02 × 1023 mol–1]
| 1. | 189 | 2. | 175 |
| 3. | 181 | 4. | 185 |
Identify A in the given reaction:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
1 molal aqueous solution of an electrolyte A2B3 is 60% ionised. The boiling point of the solution at 1 atm is-. (Rounded-off to the nearest integer)
[Given Kb for (H2O) = 0.52 K kg mol–1]
| 1. | 373 K | 2. | 380 K |
| 3. | 375 K | 4. | 377 K |
1 gram of a carbonate (M2CO3) on treatment with excess HCl produces 0.01186 moles of CO2. The molar mass of M2CO3 in g mol–1 is:
1. 118.6
2. 11.86
3. 88.6
4. 84.3
The \(p K_a\) of a weak acid (HA) and \(p K_b\) of a weak base (BOH) are 3.2 and 3.4, respectively.
The pH of their salt (AB) solution is:
1. 7.0
2. 1.0
3. 7.2
4. 6.9
Which of the following reactions is an example of a redox reaction?
1. \(XeF_6+H_2O~\longrightarrow~XeOF_4+2HF\)
2. \(XeF_6+2H_2O~\longrightarrow~XeO_2F_2+4HF\)
3. \(XeF_4+O_2F_2~\longrightarrow~XeF_6+O_2\)
4. \(XeF_2+PF_5~\longrightarrow~[XeF]^{+1}+PF_6~^-\)
The element having the highest boiling point among the following is:
| 1. | He | 2. | Ne |
| 3. | Kr | 4. | Xe |
A reactant (A) forms two products :
If Ea2 = 2 Ea1, then k1 and k2 are related as:
| 1. | 2. | ||
| 3. | 4. |
In context with the transition elements, which of the following statements is incorrect?
| 1. | In addition to the normal oxidation state, the zero oxidation state is also shown by these elements in complexes. |
| 2. | In the highest oxidation states, the transition metal shows basic character and forms cationic complexes. |
| 3. | In the highest oxidation states of the first five transition elements (Sc to Mn), all the 4s and 3d electrons are used for bonding. |
| 4. | Once the d5 configuration is exceeded, the tendency to involve all the 3d electrons in bonding decreases. |
Among the ligands NH3, en, CN- and CO, the correct order of their increasing field strength is:
1. CO < NH3 < en < CN-
2. NH3 < en < CN- < CO
3. CN- < NH3 < CO < en
4. en < CN- < NH3 < CO
Diamond and graphite both are made of carbon atoms. Diamond is extremely hard whereas graphite is soft. This is because :
| 1. | Diamond has carbon-carbon double bond while graphite has carbon-carbon single bond |
| 2. | Diamond is ionic whereas graphite is covalent |
| 3. | Diamond has a strong covalent bond with regular tetrahedron pattern |
| 4. | Certain atoms in diamond are smaller in size |
An alkene on reductive ozonolysis gives 2-molecules of CH2(CHO)2. The alkene is-
1. 2,4-Hexadiene
2. 1,3-Cyclohexadiene
3. 1,4-Cyclohexadiene
4. 1-Methyl-1, 3-cyclopentadiene
The equilibrium constant for the reaction
\(\mathrm{N}_{2}(g)+\mathrm{O}_{2}(g) \rightleftharpoons 2 \mathrm{NO}(g)\)
at temperature T is \(4 \times 10^{-4}\) . The value of Kc for the reaction,
\(\mathrm{{NO}}(g) \rightleftharpoons \frac{1}{2} \mathrm{{~N}_{2}}(g)+\frac{1}{2} \mathrm{O}_{2}(g)\)at the same temperature) is:
| 1. | \(2.5 \times 10^{2}\) | 2. | \(5 \times 10^{1}\) |
| 3. | \(4 \times 10^{-4}\) | 4. | \(2 \times 10^{-2}\) |
The standard emf of a cell, involving one electron change is found to be 0.591 V at 25°C.
The equilibrium constant of the reaction is :
(Given F = 96500 C mol–1)
| 1. | 1.0×101 | 2. | 1.0×105 |
| 3. | 1.0×1010 | 4. | 1.0×1030 |
\((\Delta H-\Delta U)\) for the formation of carbon monoxide (CO) from its elements at 298 K is :
(R = 8.314 JK-1 mol-1)
| 1. | -1238.78 J mol-1 | 2. | 1238.78 J mol-1 |
| 3. | -2477.57 J mol-1 | 4. | 2477.57 J mol-1 |
Identify Z in the given sequence:
CH3COONH4 \(\xrightarrow[]{∆}\) 𝑋 \(\xrightarrow[∆]{P_2O_5}\) 𝑌 \(\xrightarrow{H_2O/H^+}\) 𝑍
1. CH3CH2CONH2
2. CH3CN
3. CH3COOH
4. (CH3CO)2O
The number of π-bonds in the formula given below, NC—CH=CH—CN are:
| 1. | 5 | 2. | 4 |
| 3. | 3 | 4. | 2 |
Given the following benzyl/allyl system:
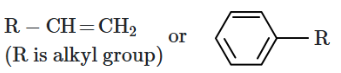
1. (CH3)3C- > (CH3)2CH- > CH3CH2-
2. CH3CH2- >(CH3)2CH- > (CH3)3C-
3. (CH3)2CH- > CH3CH2- > (CH3)3C-
4. (CH3)3C- > CH3CH2- > (CH3)2CH-
| 1. | F | 2. | Cl |
| 3. | Br | 4. | I |
For the reaction, 2A → B, rates= k[A]2. If the concentration of reactant is doubled, then the:
| (a) | rate of reaction will be doubled. |
| (b) | rate constant will remain unchanged, however rate of reaction is directly proportional to the rate constant. |
| (c) | rate constant will change since the rate of reaction and rate constant are directly proportional to each other. |
| (d) | rate of reaction will increase by four times. |
Identify the set of correct statements & choose the correct answer from the options given below:
| 1. | (a) and (c) only | 2. | (a) and (b) only |
| 3. | (b) and (d) only | 4. | (c) and (d) only |
| Statement I: | Order is applicable only for elementary reactions whereas molecularity is applicable for both elementary as well as complex reactions. |
| Statement II: | Order of a reaction is an experimental quantity. |
Arrange the following ligands in order of increasing field strength:
\(\mathrm{S}^{2-}, \mathrm{C}_2 \mathrm{O}_4^{2-}, \mathrm{NH}_3, \text { en, } \mathrm{CO} \)
| 1. | NO, ZnO, CO2, CaO | 2. | ZnO, NO, CaO, CO2 |
| 3. | NO, CO2, ZnO, CaO | 4. | NO, CO2, CaO, ZnO |
An anticancer agent among the following is:
| 1. | \(\text { mer} - \left[\mathrm{Co}\left(\mathrm{NH}_3\right)_3 \mathrm{Cl}\right] \) | 2. | \(\text { cis -} \left[\mathrm{PtCl}_2\left(\mathrm{NH}_3\right)_2\right] \) |
| 3. | \(\text {cis -} \mathrm{K}_2\left[\mathrm{Pt} \mathrm{Cl}_2 \mathrm{Br}_2\right] \) | 4. | \(\mathrm{NH}_2 \mathrm{CoCl}_4\) |
bond is present in:
| 1. | XeO3 | 2. | POCl3 |
| 3. | SO3 | 4. | All |
The electrode potential for Mg electrode varies according to the equation
\(E_{Mg^{2+}/Mg}\ = \ E_{Mg^{2+}/Mg}^{o} \ - \ \frac{0.059}{2}log\frac{1}{[Mg^{2+}]}\)
The graph of EMg2+ / Mg vs log [Mg2+] among the following is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
can be converted into by:
1. Catalytic hydrogenation
2. Treatment with LiAI
3. Treatment with pyridinium chlorochromate
4. Treatment with KMn
Compound can be prepared by the reaction of-
1. Phenol and benzoic acid in the presence of NaOH
2. Phenol and benzoyl chloride in the presence of pyridine
3. Phenol and benzoyl chloride in the presence of
4. Phenol and benzaldehyde in the presence of palladium
For a first-order reaction A → B the reaction rate at a reactant concentration of 0.01M is found to be . The half-life period of the reaction is:
| 1. | 300s | 2. | 30s |
| 3. | 220s | 4. | 347s |
Sulphuric acid reacts with sodium hydroxide as follows
When 1L of 0.1 M sulphuric acid solution is allowed to react with 1 L of 0.1 M sodium hydroxide solution, the amount of sodium sulphate formed and its molarity in the solution obtained are respectively-
| 1. | 0.1 M, 7.10 g | 2. | 7.10 g, 0.025 M |
| 3. | 0.025 M, 3.55 g | 4. | 3.55 g, 0.25 M |
| 1. | 2. | O2 | |
| 3. | 4. |
The osmotic pressure of a dilute solution of an ionic compound XY in water is four times that of a solution of 0.01 M BaCl2 in water. Assuming complete dissociation of the given ionic compound in water, the concentration of XY (in mol L–1 ) in solution is:
| 1. | 4 × 10–4 | 2. | 4 × 10–2 |
| 3. | 16 × 10–4 | 4. | 6 × 10–2 |
| Assertion (A): | When Cu (II) and sulphide ions are mixed, they react together extremely quickly to give a solid. |
| Reason (R): | The equilibrium constant of Cu2+(aq) + S2- (aq) ⇋ CuS(s) is high because the solubility product is low. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. | Both (A) and (R) are False. |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are True but (R) is not the correct explanation of (A) |
Five moles of an ideal gas at 1 bar and 298 K are expanded into a vacuum till the volume doubles. The work done is:
1. –RT ln V2/V1
2. CV(T2 – T1)
3. zero
4. – RT(V2 – V1)
Which of the following species has unequal bond lengths?
| 1. | BF4- | 2. | XeF4 |
| 3. | SF4 | 4. | SiF4 |
The product A in the following reaction is:

| 1. |  |
2. |  |
| 3. |  |
4. |  |
Use the provided information in the following paper chromatogram.

Figure: Paper chromatography for compounds A and B.
The Rf value of A is Y×10–1. The value of Y is:
| 1. | 2 | 2. | 3 |
| 3. | 4 | 4. | 8 |
The correct structural formula of 5-nitro-3-methoxy-3-methylhexanoyl chloride among the following is-
| 1. | 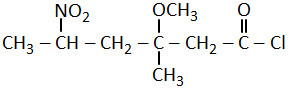 |
| 2. | 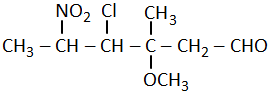 |
| 3. | 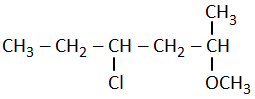 |
| 4. | 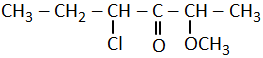 |
The moment of inertia of a body about a given axis is \(1.5\text{ kg.m}^2.\) Initially, the body is at rest. In order to produce rotational kinetic energy of \(1200\text{ J},\) the angular acceleration of \(20\text{ rad/s}^2\) must be applied about the axis for a duration of:
| 1. | \(5\text{ s}\) | 2. | \(3\text{ s}\) |
| 3. | \(2.5\text{ s}\) | 4. | \(2\text{ s}\) |
In the given figure, the energy levels of hydrogen atoms have been shown along with some transitions marked \(A, B, C, D\text{ and }E.\) Transitions \(A\) and \(B\) represent, respectively:
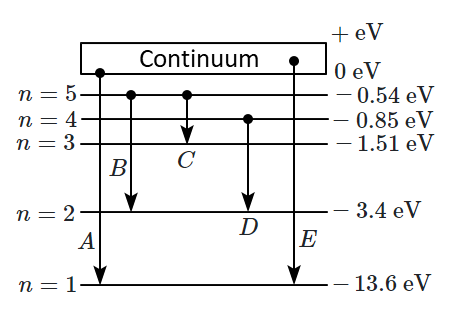
| 1. | The ionization potential of hydrogen and the second member of the Balmer series. |
| 2. | The first member of the Lyman series and the second member of the Paschen series. |
| 3. | The series limit of the Lyman series and the third member of the Balmer series. |
| 4. | The series limit of the Lyman series and the second member of the Paschen series. |
A sinusoidal voltage of peak value \(283~\text{V}\) (assuming that the frequency of the source can be varied) is applied to a series \(LCR\) circuit in which \(R=3~\Omega\), \(L=25.48~\text{mH},\) and \(C= 796~\mu\text{F}.\) The current in the circuit at the resonance is:
1. \(60~\text{A}\)
2. \(66.7~\text{A}\)
3. \(65~\text{A}\)
4. \(63.3~\text{A}\)
A metal cube is exposed to a hydrostatic pressure of \(4\) GPa. The bulk modulus of the metal is given as \(B=8\times 10^{10}~\text{Pa}.\) What is the percentage change in the volume of the cube?
1. \(2.5\)
2. \(5\)
3. \(7.5\)
4. \(10\)
Consider a two particle system with particles having masses \(m_1\) and \(m_2\). If the first particle is pushed towards the centre of mass through a distance \(d\), by what distance should be second particle be moved, so as to keep the centre of mass at the same position?
| 1. | \( \frac{m_2}{m_1} d\) | 2. | \( \frac{m_1}{m_1+m_2} d \) |
| 3. | \( \frac{m_1}{m_2} d \) | 4. | \(d\) |
From spring, a body of mass \(4~\text{kg}\) hangs and oscillates with a period \(0.5\) seconds. On the removal of the body, the spring is shortened by: [ Take \(\pi^2 = 10 \) ]
| 1. | \( 6.4 \text{ cm} \) | 2. | \(6.25~\text{cm}\) |
| 3. | \(6.8~\text{cm}\) | 4. | \(7.1~\text{cm}\) |
| 1. | straight lines | 2. | planar |
| 3. | spherical | 4. | ellipsoidal |
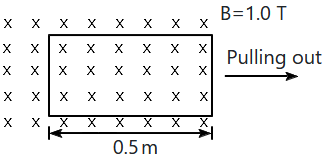
| 1. | \({3.125}\times{10}^{{-}{3}}~\text{J} \) | 2. | \({6.25}\times{10}^{{-}{4}}~\text{J}\) |
| 3. | \({1.25}\times{10}^{{-}{2}}~\text{J}\) | 4. | \({5.0}\times{10}^{{-}{4}}~\text{J}\) |
| 1. | \(\dfrac{|\mu_1|}{|\mu_2|}=1 \) | 2. | \(\mu_1\) and \(\mu_2\) are in the same direction |
| 3. | \(\dfrac{|\mu_1|}{|\mu_2|}=\sqrt2 \) | 4. | \(\dfrac{|\mu_1|}{|\mu_2|}=\dfrac{1}{\sqrt2}\) |
| 1. | Radio wave | 2. | Infrared wave |
| 3. | Microwave | 4. | Matter wave |
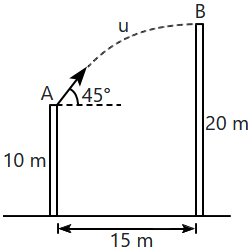
| 1. | \(20~\text{m/s}\) | 2. | \(40~\text{m/s}\) |
| 3. | \(15\sqrt{2}~\text{m/s}\) | 4. | \(50~\text{m/s}\) |
| 1. | 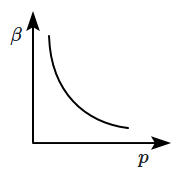 |
2. | 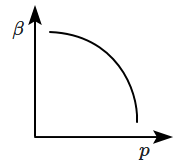 |
| 3. | 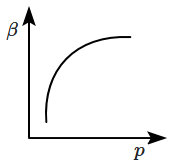 |
4. | 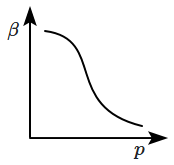 |
| 1. | \( 360000 \) | 2. | \( 72000 \) |
| 3. | \( 36000 \) | 4. | \( 129600\) |
| 1. | \(20~\text{min}\) | 2. | \(5~\text{min}\) |
| 3. | \(\frac{20}{3}~\text{min}\) | 4. | \(40~\text{min}\) |
| Statement I: | Molar heat capacity at constant pressure for all diatomic gases is the same. |
| Statement II: | The specific heat capacity at constant pressure of all diatomic ideal gases is the same. |
| 1. | only (I) is correct |
| 2. | only (II) is correct |
| 3. | both (I) and (II) are correct |
| 4. | none of them are correct |
| 1. | Flight of a jet plane |
| 2. | A cricket player lowering his hands while catching a cricket ball |
| 3. | Walking on the floor |
| 4. | Rebounding of a rubber ball |
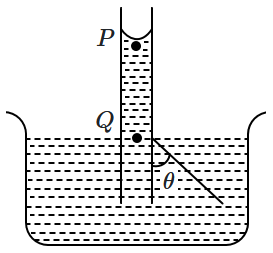
| 1. | \(\left({\dfrac{2T}{r}}\right)\cos\mathit{\theta}\) | 2. | \(\dfrac{T}{{r}\cos\mathit{\theta}}\) |
| 3. | \(\dfrac{2T}{{r}\cos\mathit{\theta}}\) | 4. | \(\left({\dfrac{4T}{r}}\right)\cos\mathit{\theta}\) |
| 1. | 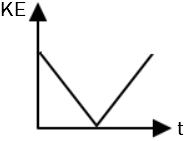 |
2. | 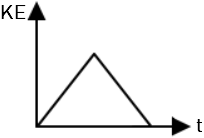 |
| 3. | 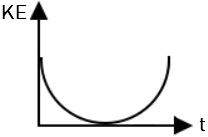 |
4. | 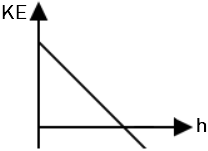 |
| 1. | It will go very far in the space. |
| 2. | It will fall down on the earth gradually. |
| 3. | It will move with the same speed, tangentially to the spacecraft. |
| 4. | It will continue to move with the same speed along the original orbit of the spacecraft. |
| 1. | \(\dfrac{\sqrt{3}}{2}\) | 2. | \(\dfrac{2}{\sqrt{3}}\) |
| 3. | \(2\) | 4. | data not sufficient |
| 1. | \(8~\text{cm}\) | 2. | \(20.3~\text{cm}\) |
| 3. | \(13.3~\text{cm}\) | 4. | \(16~\text{cm}\) |
| 1. | only the proton number changes. |
| 2. | both the neutron number and the proton number change. |
| 3. | there is no change in the proton number and the neutron number. |
| 4. | only the neutron number changes. |
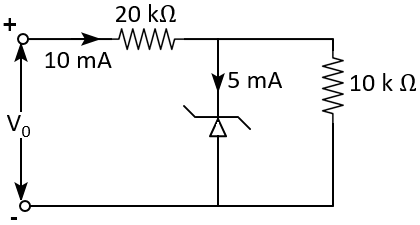
The circuit represents a full wave bridge rectifier when switch \(S\) is open. The output voltage \((V_0)\) pattern across \(R_L\) when \(S\) is closed:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
An \(\alpha\)-particle and a proton are accelerated from rest by a potential difference of \(200~\text{V}\). After this, their de-Broglie wavelengths are \(\lambda_{\alpha}\)
| 1. | \(3.8\) | 2. | \(8\) |
| 3. | \(7.8\) | 4. | \(2.8\) |
A small block starts slipping down from a point \(\mathrm{B}\) on an inclined plane \(\mathrm{AB}\), which is making an angle \(\theta\) with the horizontal section. \(\mathrm{BC}\) is smooth and the remaining section \(\mathrm{CA}\) is rough with a coefficient of friction \(\mu\). It is found that the block comes to rest as it reaches the bottom (point \(\mathrm{A}\)) of the inclined plane. If \(BC=2AC\), the coefficient of friction is given by \(\mu=k\tan\theta \). The value of \(k\) is:
| 1. | \(1\) | 2. | \(2\) |
| 3. | \(3\) | 4. | \(4\) |
Starting from the origin at a time \(t = 0,\) with an initial velocity \(5\hat j~\text{ms}^{-1},\) a particle moves in the \((x\text-y)\) plane with a constant acceleration of \((10\hat i+4\hat j)~\text{ms}^{-2}.\) At time \(t,\) its coordinates are \((20~\text{m}, y_0~\text m).\) The value of \(t\) is:
| 1. | \(2~\text s\) | 2. | \(4~\text s\) |
| 3. | \(6~\text s\) | 4. | \(8~\text s\) |
A body is moving in a low circular orbit about a planet of mass \(M\) and radius \(R\). The radius of the orbit can be taken to be \(R\) itself. The ratio of the speed of this body in the orbit to the escape velocity from the planet is:
1. \(\sqrt{2}\)
2. \(\dfrac{1}{\sqrt{2}}\)
3. \(2\)
4. \(1\)
If the terminal speed of a sphere of gold (density = \(19.5~\text{kg/m}^3\)) is \(0.2~\text{m/s}\) in a viscous liquid (density = \(1.5~\text{kg/m}^3\)), the terminal speed of a sphere of silver (density = \(10.5~\text{kg/m}^3\)) of the same size in the same liquid will be:
1. \(0.4~\text{m/s}\)
2. \(0.133~\text{m/s}\)
3. \(0.1~\text{m/s}\)
4. \(0.2~\text{m/s}\)
A particle executes simple harmonic motion with a frequency of \(2\) Hz. The frequency with which its potential energy oscillates is:
1. \(4\) Hz
2. \(2\) Hz
3. \(6\) Hz
4. \(8\) Hz
| 1. | Body of metal is equipotential. |
| 2. | Field lines are perpendicular to the metal surface. |
| 3. | Electrostatic potential inside a metal body is zero. |
| 4. | Electrostatic field inside metal is zero. |
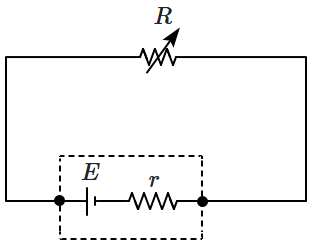
| 1. | Increases |
| 2. | Decreases |
| 3. | First increase then decreases |
| 4. | None of these |
| 1. | becomes zero | 2. | increases |
| 3. | decreases | 4. | remains unchanged |
| 1. | \({\lambda}={\pi}\times{10}^{-2} \) m | 2. | \(\lambda=10^{-3} \) m |
| 3. | \({f=10}^{-3}/\left({{2}{\pi}}\right) \) Hz | 4. | \(f=10^{4} \) Hz |
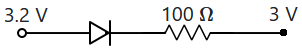
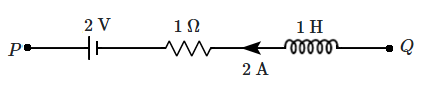
| 1. | \(12\) N | 2. | \(8\) N |
| 3. | \(6\) N | 4. | \(4\) N |