Which of the following cell organelles is present in the highest number in secretory cells?
| 1. | Mitochondria | 2. | Golgi complex |
| 3. | Endoplasmic reticulum | 4. | Lysosomes |
Non-membranous nucleoplasmic structures in the nucleus, are the sites for active synthesis of:
| 1. | protein | 2. | mRNA |
| 3. | rRNA | 4. | tRNA |
Which of the following nucleic acids is present in an organism having 70 S ribosomes only?
| 1. | Single-stranded DNA with a protein coat |
| 2. | Double-stranded circular naked DNA |
| 3. | Double-stranded DNA enclosed in nuclear membrane |
| 4. | Double-stranded circular DNA with histone proteins |
After meiosis I, the resultant daughter cells have
| 1. | same amount of DNA as in the parent cell in S |
| 2. | twice the amount of DNA in comparison to haploid |
| 3. | same amount of DNA in comparison to haploid gamete |
| 4. | four times the amount of DNA in comparison to haploid gamete |
Which of the following organic compounds is the main constituent of Lecithin?
1. Arachidonic acid
2. Phospholipid
3. Cholesterol
4. Phosphoprotein
The main difference between active and passive transport across cell membrane is :
| 1. | passive transport is non-selective whereas active transport is selective |
| 2. | passive transport requires a concentration gradient across a biological membrane whereas active transport requires energy to move solutes. |
| 3. | passive transport is confined to anionic carrier proteins whereas active transport is confined to cationic channel. |
| 4. | active transport occurs more rapidly than passive |
Match the items given in Column - I with those in Column - II and choose the correct option:
| Column-I | Column-II | ||
| (a) | Rennin | (i) | Vitamin B12 |
| (b) | Enterokinase | (ii) | Facilitated transport |
| (c) | Oxyntic cells | (iii) | Milk proteins |
| (d) | Fructose | (iv) | Trypsinogen |
| Options: | (a) | (b) | (c) | (d) |
| 1. | iii | iv | ii | i |
| 2. | iv | iii | i | ii |
| 3. | iv | iii | ii | i |
| 4. | iii | iv | i | ii |
Kwashiorkor disease is due to:
| 1. | simultaneous deficiency of proteins and fats |
| 2. | simultaneous deficiency of proteins and calories |
| 3. | deficiency of carbohydrates |
| 4. | protein deficiency not accompanied by calorie deficiency |
Select the correct sequence of events.
| 1. | Gametogenesis → Gamete transfer → Syngamy → Zygote → Cell division (Cleavage) → Cell differentiation → Organogenesis |
| 2. | Gametogenesis → Gamete transfer → Syngamy → Zygote → Cell division (Cleavage) → Organogenesis → Cell differentiation |
| 3. | Gametogenesis → Syngamy → Gamete transfer → Zygote → Cell division (Cleavage) → Cell differentiation → Organogenesis |
| 4. | Gametogenesis → Gamete transfer → Syngamy → Zygote → Cell differentiation → Cell division (Cleavage) → Organogenesis |
Which of the following hormones is responsible for both the milk ejection reflex and the foetal ejection reflex?
| 1. | Estrogen | 2. | Prolactin |
| 3. | Oxytocin | 4. | Relaxin |
No new follicles develop in the luteal phase of the menstrual cycle because :
| 1. | Follicles do not remain in the ovary after ovulation |
| 2. | FSH levels are high in the luteal phase |
| 3. | LH levels are high in the luteal phase |
| 4. | Both FSH and LH levels are low in the luteal phase |
In Australia, marsupials and placental mammals have evolved to share many similar characteristics. This type of evolution may be referred to as:
| 1. | Adaptive Radiation | 2. | Divergent Evolution |
| 3. | Cyclical Evolution | 4. | Convergent Evolution |
| Column-I | Column-II | ||
|---|---|---|---|
| (a) | XX–XO method of sex determination | (i) | Turner’s syndrome |
| (b) | XX–XY method of sex determination | (ii) | Female heterogametic |
| (c) | Karyotype – 45 | (iii) | Grasshopper |
| (d) | ZW–ZZ method of sex determination | (iv) | Female homogametic |
Select the correct answer
| Option | (a) | (b) | (c) | (d) |
|---|---|---|---|---|
| 1. | (ii) | (iv) | (i) | (iii) |
| 2. | (i) | (iv) | (ii) | (iii) |
| 3. | (iii) | (iv) | (i) | (ii) |
| 4. | (iv) | (ii) | (i) | (iii) |
What will be the sequence of mRNA produced by the following stretch of DNA?
3' ATGCATGCATGCATG 5' TEMPLATE STRAND
5' TACGTACGTACGTAC 3' CODING STRAND
1. 3' AUGCAUGCAUGCAUG 5'
2. 5' UACGUACGUACGUAC 3'
3. 3' UACGUACGUACGUAC 5'
4. 5' AUGCAUGCAUGCAUG 3'
Select the incorrect statement regarding inbreeding:
| 1. | Inbreeding helps in elimination of deleterious alleles from the population |
| 2. | Inbreeding is necessary to evolve a pure-line in any animal |
| 3. | Continued inbreeding reduces fertility and leads to inbreeding depression. |
| 4. | Inbreeding depression cannot be overcome by out-crossing. |
For a biocontrol agent to be part of an integrated pest management, it should be:
| 1. | species-specific and symbiotic |
| 2. | free living and broad spectrum |
| 3. | narrow spectrum and symbiotic |
| 4. | species-specific and inactive on non-target organisms |
Match the following enzymes with their functions:
|
Column-I |
Column-II |
||
|
(a) |
Restriction endonuclease |
(i) |
joins the DNA fragments |
|
(b) |
Exonuclease |
(ii) |
extends primers on genomic DNA template |
|
(c) |
DNA ligase |
(iii) |
cuts DNA at a specific position |
|
(d) |
Tag polymerase |
(iv) |
removes nucleotides from the ends of DNA |
Select the correct option from the following:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iii) | (i) | (iv) | (ii) |
| 2. | (iii) | (iv) | (i) | (ii) |
| 3. | (iv) | (iii) | (i) | (ii) |
| 4. | (ii) | (iv) | (i) | (iii) |
The two antibiotic resistance genes on vector pBR 322 are for:
1. Ampicillin and Tetracycline
2. Ampicillin and Chloramphenicol
3. Chloramphenicol and Tetracycline
4. Tetracycline and Kanamycin
The exploitation of bioresources of a nation by multinational companies without authorization from the concerned country is referred to as:
| 1. | Bioweapon | 2. | Biopiracy |
| 3. | Bioethics | 4. | Biowar |
Carnivorous animals - lions and leopards, occupy the same niche but lions predate mostly larger animals and leopards take smaller ones. This mechanism of competition is referred to as:
| 1. | Character displacement | 2. | Altruism |
| 3. | Resource partitioning | 4. | Competitive exclusion |
The decline in the population of Indian native fishes due to the introduction of Clarias gariepinus in river Yamuna can be categorised as:
1. Co-extinction
2. Habitat fragmentation
3. Over exploitation
4. Alien species invasion
Match the following RNA polymerases with their transcribed products:
|
Column-I |
Column-II |
||
|
(a) |
RNA polymerase I |
(i) |
tRNA |
|
(b) |
RNA polymerase II |
(ii) |
rRNA |
|
(c) |
RNA polymerase III |
(iii) |
hnRNA |
Select the correct option from the following:
| Options: | (a) | (b) | (c) |
| 1. | i | iii | ii |
| 2. | i | ii | iii |
| 3. | ii | iii | i |
| 4. | iii | ii | i |
In a marriage between a male with blood group A and a female with blood group B, the progeny had either blood group AB or B. What could be the possible genotype of the parents?
1. IA i (Male) ; IB IB (Female)
2. IA IA (Male) ; IB IB (Female)
3. IA IA (Male) ; IB i (Female)
4. IA i (Male) ; IB i(Female)
A population of a species invades a new area. Which of the following condition will lead to adaptive radiation?
| 1. | Area with a large number of habitats having very low food supply |
| 2. | Area with a single type of vacant habitat |
| 3. | Area with many types of vacant habitats |
| 4. | Area with many habitats occupied by a large number of species |
Identify A, B and C in the diagrammatic representation of the mechanism of hormone action.
Select the correct option from the following:
| 1. | A = Steroid Hormone; B = Hormone receptor Complex; C = Protein |
| 2. | A = Protein Hormone; B = Receptor; C = Cyclic AMP |
| 3. | A = Steroid Hormone; B = Receptor; C = Second Messenger |
| 4. | A = Protein Hormone; B = Cyclic AMP; C = Hormone-receptor Complex |
Humans have acquired an immune system that produces antibodies to neutralize pathogens. Still innate immune system is present at the time of birth because it:
| 1. | is very specific and uses different macrophages |
| 2. | produces memory cells for mounting a fast secondary response |
| 3. | has natural killer cells that can phagocytose and destroy microbes |
| 4. | provides passive immunity |
Which of the following statements is not correct?
| 1. | An action potential in an axon does not move backward because the segment behind is in a refractory phase. |
| 2. | Depolarization of hair cells of cochlea results in the opening of the mechanically gated Potassium- ion channels. |
| 3. | Rods are very sensitive and contribute to daylight vision. |
| 4. | In the knee-jerk reflex, stimulus is the stretching of muscle and response is its contraction. |
Match the following joints with the bones involved:
| Column-I | Column-II | ||
| (a) | Gliding joint | (i) | Between carpal and metacarpal of thumb |
| (b) | Hinge joint | (ii) | Between Atlas and Axis |
| (c) | Pivot joint | (iii) | Between the Carpals |
| (d) | Saddle joint | (iv) | Between Humerus and Ulna |
Select the correct option from the following:
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (iv) | (ii) | (i) |
| 2. | (iv) | (i) | (ii) | (iii) |
| 3. | (iv) | (ii) | (iii) | (i) |
| 4. | (i) | (iii) | (ii) | (iv) |
Which of the following diseases is an auto-immune disorder?
| 1. | Myasthenia gravis | 2. | Arthritis |
| 3. | Osteoporosis | 4. | Gout |
Artificial light, extended work-time and reduced sleep-time disrupt the activity of:
| 1. | Thymus gland | 2. | Pineal gland |
| 3. | Adrenal gland | 4. | Posterior pituitary gland |
Which of the following conditions will stimulate parathyroid gland to release parathyroid hormone?
1. Fall in active Vitamin D levels
2. Fall in blood Ca+2 levels
3. Fall in bone Ca+2 levels
4. Rise in blood Ca+2 levels
Which of the following is a correct statement?
| 1. | IUDs once inserted need not be replaced. |
| 2. | IUDs are generally inserted by the user herself. |
| 3. | IUDs increase phagocytosis of sperms in the uterus. |
| 4. | IUDs suppress gametogenesis. |
Which of the following sexually transmitted diseases do not specifically affect reproductive organs?
| 1. | Genital warts and Hepatitis-B |
| 2. | Syphilis and Genital herpes |
| 3. | AIDS and Hepatitis B |
| 4. | Chlamydiosis and AIDS |
Match the following genera with their respective phylum:
| Column-I | Column-II | ||
| (a) | Ophiura | (i) | Mollusca |
| (b) | Physalia | (ii) | Platyhelminthes |
| (c) | Pinctada | (iii) | Echinodermata |
| (d) | Planaria | (iv) | Coelenterata |
| Options: | (a) | (b) | (c) | (d) |
| 1. | iv | i | iii | ii |
| 2. | iii | iv | i | ii |
| 3. | i | iii | iv | ii |
| 4. | iii | iv | ii | i |
Which of the following animals are true coelomates with bilateral symmetry?
| 1. | Adult Echinoderms | 2. | Aschelminthes |
| 3. | Platyhelminthes | 4. | Annelids |
The contrasting characteristics generally in a pair used for identification of animals in Taxonomic Key are referred to as:
1. Lead
2. Couplet
3. Doublet
4. Alternate
Match the following cell structure with its characteristic feature:
| Column-I | Column-II | ||
| (a) | Tight junctions | (i) | Cement neighboring cells together to form a sheet |
| (b) | Adhering junctions | (ii) | Transmit information through chemicals to another cell |
| (c) | Gap junctions | (iii) | Establish a barrier to prevent leakage of fluid across epithelial cells |
| (d) | Synaptic junctions | (iv) | Cytoplasmic channels to facilitate communication between adjacent cells |
Select the correct option from the following:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (ii) | (iv) | (i) | (iii) |
| 2. | (iv) | (ii) | (i) | (iii) |
| 3. | (iii) | (i) | (iv) | (ii) |
| 4. | (iv) | (iii) | (i) | (ii) |
Which of the following statements is INCORRECT?
| 1. | Cockroaches exhibit mosaic vision with less sensitivity and more resolution |
| 2. | A mushroom-shaped gland is present in the 6th-7th abdominal segments of male |
| 3. | A pair of spermatheca is present in the 6th segment of female cockroach |
| 4. | Female cockroach possesses sixteen ovarioles in the ovaries |
Select the correct statement:
| 1. | Expiration occurs due to external intercostal muscles. |
| 2. | Intrapulmonary pressure is lower than atmospheric pressure during inspiration. |
| 3. | Inspiration occurs when atmospheric pressure is less than intrapulmonary pressure. |
| 4. | Expiration is initiated due to the contraction of the diaphragm. |
The maximum volume of air a person can breathe in after a forced expiration is known as:
1. Expiratory Capacity
2. Vital Capacity
3. Inspiratory Capacity
4. Total Lung Capacity
All the components of the nodal tissue are autoexcitable. Why does the SA node act as the normal pacemaker?
| 1. | SA node has the lowest rate of depolarisation |
| 2. | SA node is the only component to generate the threshold potential. |
| 3. | Only SA node can convey the action potential to the other |
| 4. | SA node has the highest rate of depolarisation. |
A specialised nodal tissue embedded in the lower corner of the right atrium, close to atrio-ventricular septum, delays the spreading of impulses to heart apex for about 0.1 sec.
This delay allows -
1. blood to enter aorta.
2. the ventricles to empty completely.
3. blood to enter pulmonary arteries.
4. the atria to empty completely.
Match the following parts of a nephron with their function:
| (a) | Descending limb of Henle's loop | (i) | Reabsorption of salts only |
| (b) | Proximal convoluted tubule | (ii) | Reabsorption of water only |
| (c) | Ascending limb of Henle's loop | (iii) | Conditional reabsorption of sodium ions |
| (d) | Distal convoluted tubule | (iv) | Reabsorption of ions, water, and organic nutrients |
Select the correct option from the following:
| Options: | (a) | (b) | (c) | (d) |
| 1. | i | iii | ii | iv |
| 2. | ii | iv | i | iii |
| 3. | i | iv | ii | iii |
| 4. | iv | i | iii | ii |
Match the items in Column - I with those in Column - II:
| Column I | Column II | ||
| (a) | Podocytes | (i) | Crystallised oxalate |
| (b) | Protonephridia | (ii) | Annelids |
| (c) | Nephridia | (iii) | Amphioxus |
| (d) | Renal calculi | (iv) | Filtration slits |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (iv) | (ii) | (i) |
| 2. | (iii) | (ii) | (iv) | (i) |
| 3. | (iv) | (iii) | (ii) | (i) |
| 4. | (iv) | (ii) | (iii) | (i) |
Which of the following receptors are specifically responsible for maintenance of balance of body and posture?
1. Basilar membrane and otoliths
2. Hair cells and organ of corti
3. Tectorial membrane and macula
4. Crista ampullaris and macula
Which of the following is against the rules of ICBN?
| 1. | Handwritten scientific names should be underlined separately. |
| 2. | Every species should have a generic name and a specific epithet. |
| 3. | Scientific names are in Latin and should be italicized. |
| 4. | Generic and specific names should be written starting with small letters. |
Mad cow disease in cattle is caused by an organism which has:
1. Inert crystalline
2. Abnormally folded protein
3. Free RNA without protein coat
4. Free DNA without protein coat
Which of the following statements is correct?
1. Lichens do not grow in polluted areas.
2. Algal component of lichens is called mycobiont.
3. Fungal component of lichens is called phycobiont.
4. Lichens are not good pollution indicators.
Match the organisms in Column-I with habitats in Column-II.
| Column-I | Column-II | ||
| (a) | Halophiles | (i) | Hot springs |
| (b) | Thermoacidophiles | (ii) | Aquatic environment |
| (c) | Methanogens | (iii) | Guts of ruminants |
| (d) | Cyanobacteria | (iv) | Salty areas |
Select the correct answer from the options given below:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iv) | (i) | (iii) | (ii) |
| 2. | (i) | (ii) | (iii) | (iv) |
| 3. | (iii) | (iv) | (i) | (i) |
| 4. | (ii) | (iv) | (iii) | (i) |
In the dicot root, the vascular cambium originates from:
| 1. | Tissue located below the phloem bundles and a portion of pericycle tissue above protoxylem |
| 2. | Cortical region |
| 3. | Parenchyma between endodermis and pericycle |
| 4. | Intrafascicular and interfascicular tissue in a ring |
Which of the following shows whorled phyllotaxy?
| 1. | Mustard | 2. | China rose |
| 3. | Alstonia | 4. | Calotropis |
Regeneration of damaged growing grass following grazing is largely due to:
| 1. | Lateral meristem | 2. | Apical meristem |
| 3. | Intercalary meristem | 4. | Secondary meristem |
Bicarpellary ovary with obliquely placed septum is seen in:
1. Brassica
2. Aloe
3. Solanum
4. Sesbania
Which is the most common type of embryo sac in angiosperms?
1. Tetrasporic with one mitotic stage of division
2. Monosporic with three sequential mitotic divisions
3. Monosporic with two sequential mitotic divisions
4. Bisporic with two sequential mitotic divisions
From the following, identify the correct combination of salient features of the Genetic Code:
| 1. | Universal, Non-ambiguous, Overlapping |
| 2. | Degenerate, Overlapping, Commaless |
| 3. | Universal, Ambiguous, Degenerate |
| 4. | Degenerate, Non-overlapping, Non-ambiguous |
Which scientist experimentally proved that DNA is the sole genetic material in bacteriophage?
1. Beadle and Tatum
2. Messelson and Stahl
3. Hershey and Chase
4. Jacob and Monod
In the process of transcription in Eukaryotes, the RNA polymerase I transcribe -
1. mRNA with additional processing, capping and tailing
2. tRNA, 5 S rRNA and snRNAs
3. rRNAs - 28 S, 18 S and 5.8 S
4. Precursor of mRNA, hnRNA
In which genetic condition, each cell in the affected person, has three sex chromosomes XXY?
1. Thalassemia
2. Klinefelter's Syndrome
3. Phenylketonuria
4. Turner's Syndrome
What initiation and termination factors are involved in transcription in prokaryotes?
1. , respectively
2. , respectively
3. , respectively
4. , respectively
Which of the following statements is correct about the origin and evolution of men?
| 1. | Agriculture came around 50,000 years back. |
| 2. | The Dryopithecus and Ramapithecus primates existing 15 million years ago, walked like men. |
| 3. | Homo habilis probably ate meat. |
| 4. | Neanderthal men lived in Asia between 100000 and 40000 years back. |
The production of gametes by the parents the formation of zygotes, the F1 and F2 plants can be understood using
| 1. | Pie diagram | 2. | A pyramid diagram |
| 3. | Punnett square | 4. | Wenn diagram |
Match Column-I with Column-II.
| Column-I | Column-II | ||
| (a) | Golgi apparatus | (i) | Synthesis of protein |
| (b) | Lysosomes | (ii) | Trap waste and excretory products |
| (c) | Vacuoles | (iii) | Formation of glycoproteins and glycolipids |
| (d) | Ribosomes | (iv) | Digesting biomolecules |
Choose the right match from the options given below:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iii) | (iv) | (ii) | (i) |
| 2. | (iv) | (iii) | (i) | (ii) |
| 3. | (iii) | (ii) | (iv) | (i) |
| 4. | (i) | (ii) | (iv) | (iii) |
Prosthetic groups differ from co-enzymes, in which:
| 1. | they require metal ions for their activity. |
| 2. | they (prosthetic groups) are tightly bound to apoenzymes. |
| 3. | their association with apoenzymes is transient. |
| 4. | they can serve as co-factors in a number of enzyme-catalyzed reactions. |
Crossing over takes place between which chromatids and in which stage of the cell cycle?
| 1. | Non-sister chromatids of non-homologous chromosomes at Zygotene stage of prophase I. |
| 2. | Non-sister chromatids of homologous chromosomes at Pachytene stage of prophase I. |
| 3. | Non-sister chromatids of homologous chromosomes at Zygotene stage of prophase I. |
| 4. | Non-sister chromatids of non-homologous chromosomes at Pachytene stage of prophase I. |
"Ramachandran plot" is used to confirm the structure of:
1. RNA
2. Proteins
3. Triacylglycerides
4. DNA
Which of the following is not a feature of the active transport of solutes in plants?
1. Occurs against the concentration gradient
2. Non-selective
3. Occurs through membranes
4. Requires ATP
Which of the following bacteria reduce nitrate in soil into nitrogen?
1. Nitrobacter
2. Thiobacillus
3. Nitrococcus
4. Nitrosomonas
What will be the direction of flow of water when a plant cell is placed in a Hypotonic solution?
1. Water will flow in both directions.
2. Water will flow out of the cell.
3. Water will flow into the cell.
4. No flow of water in any direction.
Where is the respiratory electron transport system (ETS) located in plants?
1. Mitochondrial matrix
2. Outer mitochondrial membrane
3. Inner mitochondrial membrane
4. Intermembrane space
In Hatch and Slack pathway, the primary CO2 acceptor is -
1. Oxaloacetic acid
2. Phosphoglyceric acid
3. Phosphoenol pyruvate
4. Rubisco
Removal of shoot tips is a very useful technique to boost the production of tea leaves. This is because:
| 1. | Gibberellins prevent bolting and are inactivated. |
| 2. | Auxins prevent leaf drop at early stages. |
| 3. | Effect of auxins is removed and the growth of lateral buds is enhanced. |
| 4. | Gibberellins delay the senescence of leaves. |
One scientist cultured Cladophora in a suspension of Azotobacter and illuminated the culture by splitting light through a prism. He observed that bacteria accumulated mainly in the region of:
| 1. | Violet and green light | 2. | Indigo and green light |
| 3. | Orange and yellow light | 4. | Blue and red light |
In order to increase the yield of sugarcane crops, which of the following plant growth regulators should be sprayed?
| 1. | Ethylene | 2. | Auxins |
| 3. | Gibberellins | 4. | Cytokinins |
What type of pollination takes place in Vallisneria?
| 1. | Pollination occurs in submerged conditions by water. |
| 2. | Flowers emerge above the surface of water and pollination occurs by insects. |
| 3. | Flowers emerge above the water surface and pollen is carried by the wind. |
| 4. | Male flowers are carried by water currents to female flowers at the surface of the water. |
In which one of the following, both autogamy and geitonogamy are prevented?
1. Wheat
2. Papaya
3. Castor
4. Maize
Match the placental types Column-I with their examples Column-II.
| Column-I | Column-II | ||
| (a) | Basal | (i) | Mustard |
| (b) | Axile | (ii) | China rose |
| (c) | Parietal | (iii) | Dianthus |
| (d) | Free central | (iv) | Sunflower |
Choose the correct answer from the following options:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (ii) | (iii) | (iv) | (i) |
| 2. | (i) | (ii) | (iii) | (iv) |
| 3. | (iv) | (ii) | (i) | (iii) |
| 4. | (iii) | (iv) | (i) | (ii) |
A selectable marker is used to:
| 1. | help in eliminating the non-transformants so that the transformants can be regenerated. |
| 2. | identify the gene for the desired trait in an alien organism. |
| 3. | select a suitable vector for transformation in a specific crop. |
| 4. | mark a gene on a chromosome for isolation using a restriction enzyme. |
Western Ghats have a large number of plant and animal species that are not found anywhere else. Which of the following terms will you use to notify such species?
| 1. | Endemic | 2. | Vulnerable |
| 3. | Threatened | 4. | Keystone |
Which of the following statements about ozone is correct?
1. Tropospheric ozone protects us from UV radiation
2. Stratospheric ozone is 'bad'
3. Tropospheric ozone is 'good'
4. Stratospheric ozone protects us from UV radiation
Which of the following is an innovative remedy for plastic waste?
1. Burning in the absence of oxygen
2. Burying 500 m deep below the soil surface
3. Polyblend
4. Electrostatic precipitator
Between which of the following, the relationship is not an example of commensalism?
| 1. | Orchid and the tree on which it grows |
| 2. | Cattle Egret and grazing cattle |
| 3. | Sea Anemone and Clownfish |
| 4. | Female wasp and fig species |
If an agricultural field is liberally irrigated for a prolonged period of time, it is likely to face the problem of:
| 1. | Metal Toxicity | 2. | Alkalinity |
| 3. | Acidity | 4. | Salinity |
Which of the following statements about methanogens is not correct?
| 1. | They can be used to produce biogas. |
| 2. | They are found in the rumen of cattle and their excreta. |
| 3. | They grow aerobically and breakdown cellulose-rich food. |
| 4. | They produce methane gas. |
In mung bean, resistance to yellow mosaic virus, and powdery mildew was brought about by:
1. Mutation breeding
2. Biofortification
3. Tissue culture
4. Hybridization and selection
Coca alkaloid or cocaine is obtained from:
| 1. | Papaver somniferum | 2. | Atropa belladonna |
| 3. | Erythroxylum coca | 4. | Datura |
Among the following pairs of microbes, which pair has both the microbes that can be used as biofertilizers?
1. Aspergillus and Rhizopus
2. Rhizobium and Rhizopus
3. Cyanobacteria and Rhizobium
4. Aspergillus and Cyanobacteria
Given below are four statements pertaining to the separation of DNA fragments using gel electrophoresis. Identify the incorrect statements:
| (a) | DNA is a negatively charged molecule and so it is loaded on gel towards the anode terminal. |
| (b) | DNA fragments travel along the surface of the gel, whose concentration does not affect the movement of DNA. |
| (c) | The smaller the DNA fragment, the greater the distance it travels through it. |
| (d) | Pure DNA can be visualized directly by exposing it to UV radiation. |
Choose the correct answer from the options given below:
| 1. | (a), (c) and (d) | 2. | (a), (b) and (c) |
| 3. | (b), (c) and (d) | 4. | (a), (b) and (d) |
An enzyme catalysing the removal of nucleotides from ends of DNA is:
| 1. | DNA ligase | 2. | Endonuclease |
| 3. | Exonuclease | 4. | Protease |
In RNAi, the genes are silenced using:
| 1. | dsRNA | 2. | ssDNA |
| 3. | ssRNA | 4. | dsDNA |
Mark the reaction among the following that does not give benzoic acid as the major product:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Which structure(s) of proteins remains(s) intact during denaturation process?
1. Both secondary and tertiary structures
2. Primary structure only
3. Secondary structure only
4. Tertiary structure only
The polymer that is used as a substitute for wool in making commercial fibres is -
1. Melamine
2. Nylon-6, 6
3. Polyacrylonitrile
4. Buna-N
The artificial sweetener stable at cooking temperature that does not provide calories is-
1. Saccharin
2. Aspartame
3. Sucralose
4. Alitame
The liquefied gas that is used in dry cleaning along with a suitable detergent is-
1. Water gas
2. Petroleum gas
3. NO2
4. CO2
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Which of the following is produced when the vapours of secondary alcohol are passed over heated copper at 573 K?
| 1. | A carboxylic acid | 2. | An aldehyde |
| 3. | A ketone | 4. | An alkene |
The major products C and D formed in the following reaction respectively are:
\(CH_3CH_2CH_2OC{(CH_3)}_3 \xrightarrow[\Delta]{\text{Excess}\ HI}\ C + D\)
1. H3C-CH2-CH2-I and I-C(CH3)3
2. H3C-CH2-CH2-OH and I-C(CH3)3
3. H3C-CH2-CH2-I and HO-C(CH3)3
4. H3C-CH2-CH2-OH and HO-C(CH3)3
Match the oxide given in Column-I with its property given in Column-II:
| Column-I | Column-II | ||
| (i) | (a) | Neutral | |
| (ii) | (b) | Basic | |
| (iii) | (c) | Acidic | |
| (iv) | (d) | Amphoteric |
Which of the following options is correct?
| (i) | (ii) | (iii) | (iv) | |
| 1. | (b) | (a) | (d) | (c) |
| 2. | (c) | (b) | (a) | (d) |
| 3. | (a) | (d) | (b) | (c) |
| 4. | (b) | (d) | (a) | (c) |
Match the catalyst with the process:
| Catalyst | Process | ||
| (i) | \(V_2O_5\) | (a) | The oxidation of ethylene to ethanal |
| (ii) | \(TiCl_4 + Al(CH_3)_3\) | (b) | Polymerisation of alkynes |
| (iii) | \(PdCl_2\) | (c) | Oxidation of \(SO_2\) in the manufacture of \(H_2SO_4\) |
| (iv) | Nickel complexes | (d) | Polymerisation of ethylene |
Which of the following is the correct matching of Catalyst & Process?
| Options: | (i) | (ii) | (iii) | (iv) |
| 1. | c | d | a | b |
| 2. | a | b | c | d |
| 3. | a | c | b | d |
| 4. | c | a | d | b |
1. (i)-(c), (ii)-(d), (iii)-(a), (iv)-(b)
2. (i)-(a), (ii)-(b), (iii)-(c), (iv)-(d)
3. (i)-(a), (ii)-(n), (iii)-(b), (iv)-(d)
4. (i)-(c), (ii)-(a), (iii)-(d), (iv)-(b)
The most stable carbocation among the following is:
| 1. |  |
2. |  |
| 3. | 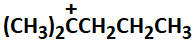 |
4. |
The alkane that gives only one monochloro product on chlorination with \(\text{Cl}_2\) in presence of diffused sunlight is-
| 1. | 2,2,-Dimethylbutane | 2. | Neopentane |
| 3. | n-Pentane | 4. | Isopentane |
In the following reaction,
\(\mathrm{ CH_{3}C\equiv CH\xrightarrow[Red \ hot \ iron \ tube]{873 K} \ A}\)
The number of () bonds present in the product (A) is:
| 1. | 21 | 2. | 9 |
| 3. | 24 | 4. | 18 |
Aluminium chloride in acidified aqueous solution forms a complex 'A'.
The formula of A and hybridisation state of Al in 'A' is respectively:
1. [Al(H2O)6]3+, sp3d2
2. [Al(H2O)4]3+, sp3
3. [Al(H2O)4]3+, dsp2
4. [Al(H2O)]3+, d2sp3
The compound among the following that used in cosmetic surgery is:
1. Silica
2. Silicates
3. Silicones
4. Zeolites
Identify the incorrect statement:
| 1. | The scientific and technological process used for isolation of the metal from its ore is known as metallurgy |
| 2. | Minerals are naturally occurring chemical substances in the earth's crust |
| 3. | Ores are minerals that may contain a metal |
| 4. | Gangue is an ore contaminated with undesired materials |
A compound "X", upon reaction with H2O, produces a colourless gas "Y" with a rotten fish smell. Gas 'Y' is absorbed in a solution of CuSO4 to give Cu3P2 as one of the products. The compound 'X' will be:
1. Ca3P2
2.
3.
4. Ca3(PO4)2
| 1. | H2S2O7 | 2. | H2SO3 |
| 3. | H2SO4 | 4. | H2S2O8 |
When neutral or faintly alkaline is treated with potassium iodide, iodide ion is converted into 'X'. 'X' is:
1.
2.
3.
4.
The crystal field stabilization energy (CFSE) for [CoCl6]4– is 18000 cm–1.
The CFSE for [CoCl4]2– will be:
| 1. | 2. | ||
| 3. | 4. |
|
Limiting molar conductivities, for the given solutions, are:
\(\lambda_{m}^{0} \left(\right. H_{2} S O_{4} \left.\right) = x\) \(S c m^{2}\) \(m o l^{- 1}\)
\(\lambda_{m}^{0} \left(\right. K_{2} S O_{4} \left.\right) = y\) \(S c m^{2}\) \(m o l^{- 1}\)
\(\lambda_{m}^{0} \left(\right. C H_{3} C O O K \left.\right) = z\) \(S c m^{2}\) \(m o l^{- 1}\)
From the data given above, it can be concluded that \(\lambda_m^0 \) in (\(S\ cm^2\ mol^{-1}\)) for CH3COOH will be:
| 1. | \(\mathrm{x-y+2z}\) | 2. | \(\mathrm{x+y+z}\) |
| 3. | \(\mathrm{x-y+z}\) | 4. | \(\mathrm{{(x-y) \over 2}+z}\) |
A first-order reaction has a rate constant of 2.303 × 10¯3 s¯1. The time required for 40 g of this reactant to reduce to 10 g will be
[Given that ]
| 1. | 230.3 s | 2. | 301 s |
| 3. | 2000 s | 4. | 602 s |
| 1. | 3.2 × 104 s-1 | 2. | 1.6 × 106s-1 |
| 3. | 1.6 × 103 s-1 | 4. | 3.2 × 106 s-1 |
The equation that represents a Freundlich adsorption isotherm is:
1.
2.
3.
4.
Which of the following is a paramagnetic compound?
1.
2.
3.
4.
Arrange the following species in increasing order of their dipole moments:
NH3,NF3,BF3,H2O
1.
2.
3.
4.
Crude sodium chloride obtained by crystallization of brine solution does not contain-
1.
2.
3.
4.
Which of the alkali metal chloride (MCl) forms its dehydrate (MCl.) easily?
1. LiCl
2. CsCl
3. RbCl
4. KCl
| 1. | 7.01 | 2. | 2 |
| 3. | 12 | 4. | 9 |
Which of the following cannot act both as a Bronsted acid and as a Bronsted base?
| 1. | \(\mathrm{H C O_{3}^{-}}\) | 2. | \(\mathrm{NH_3}\) |
| 3. | \(\mathrm{HCl}\) | 4. | \(\mathrm{H S O_{4}^{-}}\) |
The molar solubility of in 0.1 M solution of NaF will be:
| 1. | 2. | ||
| 3. | 4. |
The oxidation state of Cr in is:
| 1. | –6 | 2. | +12 |
| 3. | +6 | 4. | +4 |
The number of hydrogen bonded water molecules(s) associated with \(\mathrm{C u S O_{4} . 5 H_{2} O}\) is:
| 1. | 3 | 2. | 1 |
| 3. | 2 | 4. | 5 |
The formula of nickel oxide with metal deficiency defect in its crystal is . The crystal contains ions. The fraction of nickel existing as ions in the crystal is-
1. 0.96
2. 0.04
3. 0.50
4. 0.31
The correct statement regarding a solution of two components A and B exhibiting positive deviation from ideal behaviour is :
| 1. | Intermolecular attractive force between A-A and B-B are stronger than those between A-B |
| 2. | ∆mixH = 0 at constant T and P |
| 3. | ∆mixV = 0 at constant T and P |
| 4. | Intermolecular attractive forces between A-A and B-B are equal to those between A-B |
In water-saturated air, the mole fraction of water vapor is 0.02. If the total pressure of the saturated air is 1.2 atm, the partial pressure of dry air is-
1. 1.17 atm
2. 1.76 atm
3. 1.27 atm
4. 0.98 atm
The standard electrode potential (E°) values of Al3+/ Al, Ag+ / Ag, K+ / K, and Cr3+ / Cr are –1.66 V, 0.80 V, –2.93 V, & –0.79 V respectively. The correct decreasing order of the reducing power of the metals is:
| 1. | Ag > Cr > Al > K | 2. | K > Al > Cr > Ag |
| 3. | K > Al > Ag > Cr | 4. | Al > K > Ag > Cr |
The density of a 2 M aqueous solution of NaOH is 1.28 g/. The molality of the solution is:
[molecular mass of NaOH = 40 g mol-1]
| 1. | 1.20 m | 2. | 1.56 m |
| 3. | 1.67 m | 4. | 1.32 m |
Orbital having 3 angular nodes and 3 total nodes is:
| 1. | 5 p | 2. | 3 d |
| 3. | 4 f | 4. | 6 d |
In hydrogen atom, what is the de Broglie wavelength of an electron in the second Bohr orbit is: [Given that Bohr radius, ]
1. 211.6 pm
2. 211.6 pm
3. pm
4. 105.8 pm
The volume occupied by 1.8 g of water vapour at 374 and 1 bar pressure will be -
[Use R=0.083 bar ]
1. 96.66 L
2. 55.87 L
3. 3.10 L
4. 5.31 L
What is the amount of work done by an ideal gas, if the gas expands isothermally from \(10^{-3}~m^3\) to \(10^{-2}~m^3\) at \(300~K\)against a constant pressure of \(10^{5}~Nm^{-2}\)?
| 1. | \(+270 ~kJ\) | 2. | \(–900 ~J\) |
| 3. | \(+900 ~kJ\) | 4. | \(–900~ kJ\) |
Reversible expansion of an ideal gas under isothermal and adiabatic conditions are shown in the figure:
ABIsothermal expansion
ACAdiabatic expansion
Which of the following options is not correct?
| 1. | \(\Delta S_{\text {isothermal }}>\Delta S_{\text {adiabatic }} \) | 2. | \(T_A=T_B \) |
| 3. | \(W_{\text {isothermal }}>W_{\text {adiabatic }} \) | 4. | \(T_C>T_A\) |
| 1. | \(\sigma_{1}=\dfrac{5}{6}\sigma ,~\sigma_{2}=\dfrac{5}{6}\sigma\) |
| 2. | \(\sigma_{1}=\dfrac{5}{2}\sigma ,~\sigma_{2}=\dfrac{5}{6}\sigma\) |
| 3. | \(\sigma_{1}=\dfrac{5}{2}\sigma ,~\sigma_{2}=\dfrac{5}{3}\sigma\) |
| 4. | \(\sigma_{1}=\dfrac{5}{3}\sigma ,~\sigma_{2}=\dfrac{5}{6}\sigma\) |
The distance covered by a particle undergoing SHM in one time period is: (amplitude \(= A\))
1. zero
2. \(A\)
3. \(2 A\)
4. \(4 A\)
A mass falls from a height \(h\) and its time of fall \(t\) is recorded in terms of time period \(T\) of a simple pendulum. On the surface of the earth, it is found that \(t=2T\). The entire setup is taken on the surface of another planet whose mass is half of that of the Earth and whose radius is the same. The same experiment is repeated and corresponding times are noted as \(t'\) and \(T'\). Then we can say:
| 1. | \(t' = \sqrt{2}T\) | 2. | \(t'>2T'\) |
| 3. | \(t'<2T'\) | 4. | \(t' = 2T'\) |
| 1. | \(500\) m/s | 2. | \(156\) m/s |
| 3. | \(344\) m/s | 4. | \(172\) m/s |
An object flying in the air with velocity \((20 \hat{i}+25 \hat{j}-12 \hat{k})\) suddenly breaks into two pieces whose masses are in the ratio of \(1:5.\) The smaller mass flies off with a velocity \((100 \hat{i}+35 \hat{j}+8 \hat{k})\). The velocity of the larger piece will be:
1. \( 4 \hat{i}+23 \hat{j}-16 \hat{k}\)
2. \( -100 \hat{i}-35 \hat{j}-8 \hat{k} \)
3. \( 20 \hat{i}+15 \hat{j}-80 \hat{k} \)
4. \( -20 \hat{i}-15 \hat{j}-80 \hat{k}\)
An object kept in a large room having an air temperature of \(25^\circ \text{C}\) takes \(12 ~\text{min}\) to cool from \(80^\circ \text{C}\) to \(70^\circ \text{C}.\) The time taken to cool for the same object from \(70^\circ \text{C}\) to \(60^\circ \text{C}\) would be nearly:
| 1. | \(10 ~\text{min}\) | 2. | \(12 ~\text{min}\) |
| 3. | \(20 ~\text{min}\) | 4. | \(15 ~\text{min}\) |
Two small spherical metal balls, having equal masses, are made from materials of densities \(\rho_1\) and \(\rho_2\) such that \(\rho_1=8\rho_2\)
| 1. | \(\dfrac{79}{72}\) | 2. | \(\dfrac{19}{36}\) |
| 3. | \(\dfrac{39}{72}\) | 4. | \(\dfrac{79}{36}\) |
| 1. | \( \dfrac{v_0}{n} ~\text{rad} / \text{s}^2\) | 2. | \( \dfrac{v_0^2}{2 \pi {nr}^2}~ \text{rad} / \text{s}^2 \) |
| 3. | \( \dfrac{v_0^2}{4 \pi {n}{r}^2}~ \text{rad} / \text{s}^2 \) | 4. | \( \dfrac{v_0^2}{4 \pi {nr}} ~\text{rad} / \text{s}^2 \) |
| 1. | \(t_1<t_2 \) or \(t_1>t_2 \) depending upon whether the lift is going up or down. |
| 2. | \(t_1<t_2 \) |
| 3. | \(t_1>t_2 \) |
| 4. | \(t_1=t_2 \) |
A truck is stationary and has a bob suspended by a light string in a frame attached to the truck. The truck suddenly moves to the right with an acceleration of \(a.\) In the frame of the truck, the pendulum will tilt:
| 1. | to the left and the angle of inclination of the pendulum with the vertical is \(\text{sin}^{-1} \left( \dfrac{a}{g} \right )\) |
| 2. | to the left and the angle of inclination of the pendulum with the vertical is \(\text{cos}^{-1} \left ( \dfrac{a}{g} \right )\) |
| 3. | to the left and the angle of inclination of the pendulum with the vertical is \(\text{tan}^{-1} \left ( \dfrac{a}{g} \right )\) |
| 4. | to the left and the angle of inclination of the pendulum with the vertical is \(\text{tan}^{-1} \left ( \dfrac{g}{a} \right )\) |
In a U-tube, as shown in the figure, the water and oil are in the left side and right side of the tube respectively. The height of the water and oil columns are \(15~\text{cm}\) and \(20~\text{cm}\) respectively. The density of the oil is:
\(\left[\text{take}~\rho_{\text{water}}= 1000~\text{kg/m}^{3}\right]\)
| 1. | \(1200~\text{kg/m}^{3}\) | 2. | \(750~\text{kg/m}^{3}\) |
| 3. | \(1000~\text{kg/m}^{3}\) | 4. | \(1333~\text{kg/m}^{3}\) |
A deep rectangular pond of surface area \(A\), containing water (density = \(\rho,\) specific heat capacity = \(s\)), is located in a region where the outside air temperature is at a steady value of \(-26^{\circ}\text{C}\). The thickness of the ice layer in this pond at a certain instant is \(x\). Taking the thermal conductivity of ice as \(k\), and its specific latent heat of fusion as \(L\), the rate of increase of the thickness of the ice layer, at this instant, would be given by:
| 1. | \(\dfrac{26k}{x\rho L-4s}\) | 2. | \(\dfrac{26k}{x^2\rho L}\) |
| 3. | \(\dfrac{26k}{x\rho L}\) | 4. | \(\dfrac{26k}{x\rho L+4s}\) |
An LED is constructed from a \(\mathrm{p\text{-}n}\) junction diode using \(\mathrm{GaAsP}.\) The energy gap is \(1.9~\text{eV}.\) The wavelength of the light emitted will be equal to:
1. \(10.4 \times 10^{-26}~ \text{m}\)
2. \(654~ \text{nm}\)
3. \(654~ \text{m}\)
4. \(654\times 10^{-11}~\text{m}\)
The circuit diagram shown here corresponds to the logic gate:

1. \(\text{NOR}\)
2. \(\text{AND}\)
3. \(\text{OR}\)
4. \(\text{NAND}\)
The value \(\gamma = \frac{C_P}{C_V}\) for hydrogen, helium, and another ideal diatomic gas \(X\) (whose molecules are not rigid but have an additional vibrational mode), are respectively equal to:
| 1. | \(\dfrac{7}{5}, \dfrac{5}{3}, \dfrac{9}{7}\) | 2. | \(\dfrac{5}{3}, \dfrac{7}{5}, \dfrac{9}{7}\) |
| 3. | \(\dfrac{5}{3}, \dfrac{7}{5}, \dfrac{7}{5}\) | 4. | \(\dfrac{7}{5}, \dfrac{5}{3}, \dfrac{7}{5}\) |
A biconvex lens has power \(P.\) It is cut into two symmetrical halves by a plane containing the principal axis. The power of one part will be:
| 1. | \(0\) | 2. | \(\dfrac{P}{2}\) |
| 3. | \(\dfrac{P}{4}\) | 4. | \(P\) |
In Young's double-slit experiment, if there is no initial phase difference between the light from the two slits, a point on the screen corresponding to the fifth minimum has a path difference:
| 1. | \( \dfrac{5\lambda}{2} \) | 2. | \( \dfrac{10\lambda}{2} \) |
| 3. | \( \dfrac{9\lambda}{2} \) | 4. | \( \dfrac{11\lambda}{2} \) |
A double convex lens has a focal length of \(25\) cm. The radius of curvature of one of the surfaces is double of the other. What would be the radii if the refractive index of the material of the lens is \(1.5?\)
1. \(100\) cm, \(50\) cm
2. \(25\) cm, \(50\) cm
3. \(18.75\) cm, \(37.5\) cm
4. \(50\) cm, \(100\) cm
Two bullets are fired horizontally and simultaneously towards each other from the rooftops of two buildings (building being \(100~\text{m}\) apart and being of the same height of \(200~\text{m}\)) with the same velocity of \(25~\text{m/s}.\) When and where will the two bullets collide?
\((g = 10~\text{m/s}^2)\)
| 1. | After \(2~\text{s}\) at a height of \(180~\text{m}\) |
| 2. | After \(2~\text{s}\) at a height of \(20~\text{m}\) |
| 3. | After \(4~\text{s}\) at a height of \(120~\text{m}\) |
| 4. | They will not collide. |
The stress-strain curves are drawn for two different materials \(X\) and \(Y.\) It is observed that the ultimate strength point and the fracture point are close to each other for material \(X\) but are far apart for material \(Y.\) We can say that the materials \(X\) and \(Y\) are likely to be (respectively):
| 1. | ductile and brittle |
| 2. | brittle and ductile |
| 3. | brittle and plastic |
| 4. | plastic and ductile |
A body of mass \(m\) is kept on a rough horizontal surface (coefficient of friction = \(\mu).\) A horizontal force is applied to the body, but it does not move. The resultant of normal reaction and the frictional force acting on the object is given by \(\vec {F}\) where:
1. \(|{\vec {F}}| = mg+\mu mg\)
2. \(|\vec {F}| =\mu mg\)
3. \(|\vec {F}| \le mg\sqrt{1+\mu^2}\)
4. \(|\vec{F}| = mg\)
A particle of mass \(5m\) at rest suddenly breaks on its own into three fragments. Two fragments of mass \(m\) each move along mutually perpendicular directions with speed \(v\) each. The energy released during the process is:
| 1. | \(\dfrac{3}{5}mv^2\) | 2. | \(\dfrac{5}{3}mv^2\) |
| 3. | \(\dfrac{3}{2}mv^2\) | 4. | \(\dfrac{4}{3}mv^2\) |
An object of mass \(500~\text g\) initially at rest is acted upon by a variable force whose \(x\)-component varies with \(x\) in the manner shown. The velocities of the object at the points \(x=8~\text m\) and \(x=12~\text m\) would have the respective values of nearly:

| 1. | \(18~\text {m/s}\) and \(22.4~\text {m/s}\) | 2. | \(23~\text {m/s}\) and \(22.4~\text {m/s}\) |
| 3. | \(23~\text {m/s}\) and \(20.6~\text {m/s}\) | 4. | \(18~\text {m/s}\) and \(20.6~\text {m/s}\) |
A solid cylinder of mass 2 kg and radius 50 cm rolls up an inclined plane of angle of inclination . The centre of mass of the cylinder has a speed of 4 m/s. The distance travelled by the cylinder on the inclined surface will be
| 1. | 2.2 m | 2. | 1.6 m |
| 3. | 1.2 m | 4. | 2.4 m |
Two toroids \(1\) and \(2\) have total no. of turns \(200\) and \(100\) respectively with average radii \(40~\text{cm}\) and \(20~\text{cm}\) respectively. If they carry the same current \(i,\) what will be the ratio of the magnetic fields along the two loops?
1. \(1:1\)
2. \(4:1\)
3. \(2:1\)
4. \(1:2\)
| 1. | zero | 2. | \(\dfrac{3\mu_0 i}{32R},~\text{inward}\) |
| 3. | \(\dfrac{3\mu_0 i}{32R},~\text{outward}\) | 4. | \(\dfrac{\mu_0 i}{2R},~\text{inward}\) |
The variation of EMF with time for four types of generators is shown in the figures. Which amongst them can be called AC voltage?
 |
 |
| (a) | (b) |
 |
 |
| (c) | (d) |
| 1. | (a) and (d) |
| 2. | (a), (b), (c), and (d) |
| 3. | (a) and (b) |
| 4. | only (a) |
The radius of the first permitted Bohr orbit for the electron in a hydrogen atom is 0.5 Å, and its ground state energy is \(-13.6~\text{eV}.\) If the electron in the hydrogen atom is replaced by a muon \((\mu^{-}),\) which has the same charge as the electron but is \(207\) times more massive, what will be the new values for the first Bohr radius and ground state energy?
| 1. | \(0.53\times10^{-13}~\text{m}, ~-3.6~\text{eV}\) |
| 2. | \(25.6\times10^{-13}~\text{m}, ~-2.8~\text{eV}\) |
| 3. | \(2.56\times10^{-13}~\text{m}, ~-2.8~\text{keV}\) |
| 4. | \(2.56\times10^{-13}~\text{m}, ~-13.6~\text{eV}\) |
The reading of an ideal voltmeter in the circuit shown is:

| 1. | \(0.6~\text V\) | 2. | \(0~\text V\) |
| 3. | \(0.5~\text V\) | 4. | \(0.4~\text V\) |
The metre bridge shown is in a balanced position with \(\frac{P}{Q} = \frac{l_1}{l_2}\). If we now interchange the position of the galvanometer and the cell, will the bridge work? If yes, what will be the balanced condition?
| 1. | Yes, \(\frac{P}{Q}=\frac{l_1-l_2}{l_1+l_2}\) | 2. | No, no null point |
| 3. | Yes, \(\frac{P}{Q}= \frac{l_2}{l_1}\) | 4. | Yes, \(\frac{P}{Q}= \frac{l_1}{l_2}\) |
The relation amongst the three elements of Earth's magnetic field, namely horizontal component H, vertical component V and dip angle is: (=total magnetic field):
1. V=tan, H=
2. V=sin, H=cos
3. V=cos, H=sin
4. V=, H=tan
The rate of radioactive disintegration at an instant for a radioactive sample of half-life 2.2 . The number of radioactive atoms in that sample at that instant is:
1. 3.7
2. 3.17
3. 3.17
4. 3.17
| 1. | \(6\sqrt{2}~\text{hr}\) | 2. | \(12\sqrt{2}~\text{hr}\) |
| 3. | \(\frac{24}{2.5}~\text{hr}\) | 4. | \(\frac{12}{2.5}~\text{hr}\) |
A circuit when connected to an AC source of \(12~\text{V}\) gives a current of \(0.2~\text{A}\). The same circuit when connected to a DC source of \(12~\text{V}\), gives a current of \(0.4~\text{A}\). The circuit is:
| 1. | series \({LR}\) | 2. | series \({RC}\) |
| 3. | series \({LC}\) | 4. | series \({LCR}\) |
A cycle wheel of radius \(0.5\) m is rotated with a constant angular velocity of \(10\) rad/s in a region of a magnetic field of \(0.1\) T which is perpendicular to the plane of the wheel. The EMF generated between its centre and the rim is:
| 1. | \(0.25\) V | 2. | \(0.125\) V |
| 3. | \(0.5\) V | 4. | zero |
| 1. | \(2.5\times 10^{8}~\text{m/s}\) | 2. | \(3\times 10^{8}~\text{m/s}\) |
| 3. | \(2.08\times 10^{8}~\text{m/s}\) | 4. | \(4.32\times 10^{8}~\text{m/s}\) |
A sphere encloses an electric dipole with charges \(\pm3\times10^{-6}~\text C.\) What is the total electric flux through the sphere?
1. \(-3\times10^{-6}~\text{N-m}^2/\text C\)
2. zero
3. \(3\times10^{-6}~\text{N-m}^2/\text C\)
4. \(6\times10^{-6}~\text{N-m}^2/\text C\)
Two identical capacitors \(C_{1}\) and \(C_{2}\) of equal capacitance are connected as shown in the circuit. Terminals \(a\) and \(b\) of the key \(k\) are connected to charge capacitor \(C_{1}\) using a battery of emf \(V\) volt. Now disconnecting \(a\) and \(b\) terminals, terminals \(b\) and \(c\) are connected. Due to this, what will be the percentage loss of energy?
1. \(75\%\)
2. \(0\%\)
3. \(50\%\)
4. \(25\%\)
The main scale of a vernier calliper has \(n\) divisions/cm. \(n\) divisions of the vernier scale coincide with \((n-1)\) divisions of the main scale. The least count of the vernier calliper is:
| 1. | \(\dfrac{1}{(n+1)(n-1)}\) cm | 2. | \(\dfrac{1}{n}\) cm |
| 3. | \(\dfrac{1}{n^{2}}\) cm | 4. | \(\dfrac{1}{(n)(n+1)}\) cm |
| 1. | \(\dfrac{1}{v} = \dfrac{1}{v_1}+\dfrac{1}{v_2}\) | 2. | \(\dfrac{2}{v} = \dfrac{1}{v_1}+\dfrac{1}{v_2}\) |
| 3. | \(\dfrac{v}{2} = \dfrac{v_1+v_2}{2}\) | 4. | \(v = \sqrt{v_1v_2}\) |
Assuming that the gravitational potential energy of an object at infinity is zero, the change in potential energy (final - initial) of an object of mass \(m\) when taken to a height \(h\) from the surface of the earth (of radius \(R\) and mass \(M\)), is given by:
| 1. | \(-\frac{GMm}{R+h}\) | 2. | \(\frac{GMmh}{R(R+h)}\) |
| 3. | \(mgh\) | 4. | \(\frac{GMm}{R+h}\) |
\(1~\text g\) of water of volume \(1~\text{cm}^3\) at \(100^\circ \text{C}\) is converted into steam at the same temperature under normal atmospheric pressure \(\approx 1\times10^{5}~\text{Pa}.\) The volume of steam formed equals \(1671~\text{cm}^3.\) If the specific latent heat of vaporization of water is \(2256~\text{J/g},\) the change in internal energy is:
| 1. | \(2423~\text J\) | 2. | \(2089~\text J\) |
| 3. | \(167~\text J\) | 4. | \(2256~\text J\) |
The angular width of the central maximum in the Fraunhofer diffraction for \(\lambda=6000~{\mathring{A}}\) is \(\theta_0.\) When the same slit is illuminated by another monochromatic light, the angular width decreases by \(30\%.\) The wavelength of this light is:
1. \(1800~{\mathring{A}}\)
2. \(4200~{\mathring{A}}\)
3. \(420~{\mathring{A}}\)
4. \(6000~{\mathring{A}}\)
The work function of the photosensitive material is \(4.0~\text{eV}\). The longest wavelength of light that can cause photoelectric emission from the substance is (approximately):
1. \(3100~\text{nm}\)
2. \(966~\text{nm}\)
3. \(31~\text{nm}\)
4. \(310~\text{nm}\)
A proton and an \(\alpha\text{-}\)particle are accelerated from rest to the same energy. The de-Broglie wavelength \(\lambda_p\) and \(\lambda_\alpha\) are in the ratio:
1. \(2:1\)
2. \(1:1\)
3. \(\sqrt{2}:1\)
4. \(4:1\)