The power radiated by a black body is P and it radiates maximum energy at wavelength . If the temperature of the black body is now changed so that it radiates maximum energy , the power radiated by it becomes np. The value of n is
1. 3/4
2. 4/3
3. 81/256
4. 256/81
A spherical black body with a radius of 12 cm radiates 450-watt power at 500 K. If the radius 4power radiated in watt should be
1. 225
2. 450
3. 1000
4. 1800
Two rods A and B of different materials are welded together as shown in the figure. Their thermal conductivities are . The thermal conductivity of the composite rod will be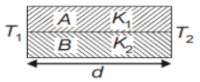
1.
2.
3.
4.
A body cools from a temperature 3T to 2T in 10 minutes. The room temperature is T. Assume that Newton's law of cooling is applicable. The temperature of the body at the end of the next 10 minutes will be
1.
2.
3.
4. T.
Coefficient of linear expansion of brass and steel rods are . Lengths of brass and steel rods are respectively. If () is maintained same at all temperatures, which one of the following holds good?
1.
2.
3.
4.
A piece of ice falls from a height h so that melts completely. Only one-quarter of the heat produced is absorbed by the ice and all energy of ice melts gets converted into heat during its fall. The value of h is
[Latent heat of ice is J/kg and g = 10N/kg]
1. 68 km
2. 34 km
3. 544 km
4. 136 km
A black body is at a temperature of 5760 K. The energy of radiation emitted by the body at wavelength 250 nm is , at wavelength 500 nm is , and that at 1000 nm is . Wein's constant, b = 2.88 x 106 nmK. Which of the following is correct?
1. >
2. = 0
3. = 0
4. >
The value of coefficient of volume expansion of glycerin is . The fractional change in hte density of glycerin for a rise of C in its temperature is
1. 0.010
2. 0.105
3. 0.020
4. 0.025
The two ends of a metal rod are maintained at temperatures 100°C and 110°C. The rate of heat flow in the rod is found to be 4.0 J/s. If the ends are maintained at temperatures 200°C and 210°C, the rate of heat flow will be
1. 4.0J/S
2. 44.0 J/s
3. 16.8 J/s
4. 8.0 J/s
On observing light from three different stars P, Q, and R, it was found that intensity of the violet color is maximum in the spectrum of P, the intensity of the green color is maximum in the spectrum of R and the intensity of the red color is maximum in the spectrum of Q. If are the respective absolute temperatures of P, Q and R then it can be concluded from the above observations that
1.
2.
3.
4.
Steam at is passed into 20g of water at . When acquires a temperature of , the mass of water present will be:
[Take specific heat of water = 1 cal and latent heat of steam = 540 cal ]
1. 24 g
2. 31.5 g
3. 42.5 g
4. 22.5 g
A certain quantity of water from 70C to 60C in the first 5 minutes and to 54C in the next 5 minutes. The temperature of the surroundings is
1. 45C
2. 20C
3. 42C
4. 10C
A piece of iron is heated in a flame. It first becomes dull red then becomes reddish yellow and finally turns to white-hot. The correct explanation for the above observation is possibly by using:
1. Wien's displacement law
2. Kirchoff's law
3. Newton's law of cooling
4. Stefan's law