Consider the following two statements:
I. In the single-celled organisms, we are not very clear about the usage of the two terms – growth and reproduction.
II. When it comes to unicellular organisms like bacteria, unicellular algae or amoeba, reproduction is synonymous with growth, i.e., increase in number of cells.
1. Both I and II are true and II explains I
2. Both I and II are true but II does not explain I
3. I is true but II is false
4. Both I and II are false
Consider the following two statements:
I. Cellular organization of the body is the defining feature of life forms.
II. Metabolic reactions cannot be demonstrated outside the body in cell-free systems.
1. Both I and II are true and II explains I
2. Both I and II are true but II does not explain I
3. I is true but II is false
4. Both I and II are false
The most obvious & complicated feature of all living organisms is:
(1) The ability to sense their surroundings or environment and respond to these environmental stimuli.
(2) Reproduction - sexual or asexual - for production of progeny of own kind.
(3) The ability to growth in size due to cell division.
(4) Presence of complex organs systems such as digestive and nervous system.
Identify the incorrect statement:
| 1. | Biological names are generally in Latin and written in italics. They are Latinised or derived from Latin irrespective of their origin. |
| 2. | The first word in a biological name represents the genus while the second component denotes the specific epithet. |
| 3. | Both the words in a biological name, when handwritten, are separately underlined, or printed in italics to indicate their vernacular origin. |
| 4. | The first word denoting the genus starts with a capital letter while the specific epithet starts with a small letter. |
The scientific term for biological classification categories is the:
| 1. | Species | 2. | Taxon |
| 3. | Phylum | 4. | Domain |
What is meant by the "species problem"?
(1) The exact definition of species is still controversial.
(2) The exact mechanisms of speciation is not known.
(3) The number of species present is much more than identified species.
(4) The extinction of species is taking place at an alarming rate.
Which of the following taxonomic categories are correctly matched to their standard termination of names with respect to biological classification of plants?
| I | Division: | - phyta |
| II | Class: | - opsida |
| III | Order: | - ales |
| IV | Family: | - idae |
1. I, II, IV
2. I, II, III
3. II, III, IV
4. I, II, III, IV
The Indian Botanical Garden and the National Botanical Research Institute are located respectively at:
(1) Pune and Howrah
(2) Howrah and Lucknow
(3) Darjeeling and Lucknow
(4) Shimla and Dehradun
Match each item in Column I with one in Column II and choose your answer from the codes given below:
|
COLUMN I |
|
COLUMN II |
|
| I. | Flora | 1. | Useful in providing information for identification of names of species found in an area |
| II. | Manuals | 2. | Contains the actual account of habitat and distribution of plants of a given area. |
| III. | Monographs | 3. | contain information on any one taxon |
Codes:
|
I |
II |
III | |
| (1) |
1 |
2 |
3 |
| (2) | 3 | 2 | 1 |
| (3) | 2 | 1 | 3 |
| (4) | 2 | 3 | 1 |
Consider the following regarding the reasons for the fact that now Cyanobacteria are kept in Monera and not in Plantae:
I. They are prokaryotes.
II. The cell wall of cyanobacteria has peptidoglycan.
III. They can fix atomspheric nitrogen
The correct explanations would be:
(1) I and II only
(2) I and III only
(3) II and III only
(4) I, II and III
Which of the following are valid pointers to the demerits of the two kingdom classification proposed by Linnaeus?
I. Euglena has features both of plants and animals.
II. Chlamydomanas is autotrophic but has flagella.
III. Slime molds resemble animals in one phase of their life cycle and plants in the other phase.
(1) I and II only
(2) I and III only
(3) II and III only
(4) I, II and III
Identify the group that is not matched correctly to all the characters shown:
| Group | Cell Type | Cell Wall | Nuclear Membrane | Body Organization | |
| 1. | Monera | Prokaryotic | Absent | Absent | Cellular |
| 2. | Protista | Eukaryotic | Present in some | Present | Cellular |
| 3. | Fungi | Eukaryotic | Present | Present | Multicellular/loose tissue |
| 4. | Plantae | Eukaryotic | Present | Present | Tissue/organ |
Math Column I with Column II and choose your answer from the codes given:
|
|
Column I (Bacterium) |
|
Column II (shape) |
|
I. |
Escherechia coli |
1. |
Spherical |
|
II. |
Streptococcus pyogenes |
2. |
Helically coiled |
|
III. |
Treponema pallidum |
3. |
Rod shaped |
|
|
I |
II |
III |
|
(1) |
1 |
2 |
3 |
|
(2) |
3 |
1 |
2 |
|
(3) |
3 |
2 |
1 |
|
(4) |
1 |
3 |
2 |
Identify the incorrect statement regarding bacteria:
| 1. | Bacteria are the most abundant microorganisms. |
| 2. | Bacteria live in extreme habitats where very few other life forms can survive. |
| 3. | Bacteria as a group show the most extensive metabolic diversity. |
| 4. | They are obligate intracellular parasites. |
Identify a character that is not unique to archaebacteria:
(1) Cell wall structure
(2) Cell membrane structure
(3) Flagellin protein structure
(4) Fatty acid synthetase present
Identify the incorrect statement regarding true bacteria?
1. As a group, they exhibit maximum metabolic diversity
2. Chemosynthetic autotrophic bacteria play a great role in recycling nutrients like nitrogen, phosphorous, iron and sulphur.
3. Heterotrophic bacteria are the most abundant in nature.
4. Bacteria reproduce mainly by fission but sometimes under favourable conditions, they produce spores.
Consider the following statements regarding Mycoplasma:
I. They are a genus of bacteria that have a unique cell wall of their own.
II. In their genetic code UGA codes for tryptophan.
III. None of them is pathogenic to humans.
IV. They have a large genome.
V. They can be killed by penicillins.
VI. They can survive without oxygen
The number of correct statements is:
(1) 1
(2) 2
(3) 3
(4) 4
Diatoms have left behind large amounts of cell wall deposits in their habitat because:
1. They are most abundant in that habitat
2. Their life span is long
3. Their wall is indestructible
4. The statement is not true
Dinoflagellates:
I. Are mostly marine and photosynthetic.
II. Have a cell wall that has stiff cellulose plates on the outer surface.
III. Have two flagella
IV. Have chloroplasts bound by three membranes
The correct statements are:
(1) I, II and III
(2) I, III and IV
(3) II, III and IV
(4) I, II III and IV
Saprophytic protists are:
(1) Slime moulds
(2) Dinoflagellates
(3) Chrysophytes
(4) Protozoans
Identify the incorrectly matched pair:
Infection Causative Protozoan
1. Kala azar Leishmania donovani
2. Chagas disease Trypanosoma cruzi
3. Gastroenteritis Giardia lamblia
4. Malaria Female Anopheles
Which of the following infections is not caused by a flagellated protozoan?
1. Malaria
2. Kala azar
3. Giardiasis
4. Sleeping sickness
Consider the following characters:
I. Heterotroph organisms
II. A dikaryon stage
III. Cell wall made of chitin
IV. Undergo nuclear mitosis
Which of the above relate to fungi?
1. I, II and III
2. I, III and IV
3. II, III and IV
4. I, II, III and IV
How many of the following statements regarding fungi are true?
| I. | Asexual reproduction is common by the formation of spores. |
| II. | Their bodies consist of hyphae that may be interconnected to form mycelium. |
| III. | They secrete digestive enzymes onto organic matter and then absorb the products of the digestion. |
| IV. | Fungi can break down almost any carbon containing product. |
| V. | Fungi do not enter symbiotic relationships. |
1. 2
2. 3
3. 4
4. 5
Which of the following is not an edible mushroom?
(1) Agaricus bispora
(2) Pleurotus ostreatus
(3) Lentinula edodes
(4) Amantia phalloides
Consider the following characters:
I. Nuclear envelope
II. Membranes lacking sterols
III. 80s ribosomes
IV. split genes
The character not shared by fungi with other eukaryotes is/are:
(1) II alone
(2) IV alone
(3) II and IV only
(4) III and IV only
Fungi resemble plants in:
(1) Lack of chloroplasts and heterotrophic nutrition
(2) Reproduction by only sexual means
(3) Having a cell wall and vacuoles
(4) Exhibition of bioluminiscence
Identify the characteristic that is unique to fungi:
(1) Cell wall of glucans and chitin
(2) Lack of chloroplasts and heterotrophic nutrition
(3) Mannitol, trehalose and glycogen as storage products.
(4) Having haploid nuclei and production of spores
What is not true for Lichens?
(i) They are symbiotic relationship between a fungus and an alga only.
(ii) Ascomycetes are the fungal partners in most lichens
(iii) They are named for the algal partner.
(iv) They are generally absent in and around big cities
1. i and ii
2. iii and iv
3. i and iii
4. ii and iv
What is not true for Mycorrhizae?
I. Nearly 30% of vascular plants have this symbiotic relationship with fungi.
II. The fungus primarily makes nitrogen available to plants.
III. Fungi get organic carbon from plants.
(1) I alone
(2) I and II only
(3) II and III only
(4) I, II and III
Identify the group of fungi that is not correctly matched with all the characters given below:
| 1. | Phycomycetes | Mycelium – aseptate and coenocytic / Asexual reproduction by motile zoospores or by nonmotile aplanospores / spores – endogenously produced in the sporangium |
| 2. | Ascomycetes | Mycelium – unbranched and septate / Asexual spores are conidia / Conidia produced endogenously on conidiophores |
| 3. | Basidiomycetes | Mycelium–branched and septate / Asexual spores are generally not found |
| 4. | Deuteromycetes | Only the asexual or vegetative phase of these fungi are known |
Match the names of the scientists with contributions and choose your answer from the codes given below:
|
Scientists |
Contribution |
||
|
I. |
Beijerinck |
1. |
Isolated HIV |
|
II. |
Luc Montagnier |
2. |
Gave the term ‘Contagium vivum fluidum’ |
|
III. |
Twort |
3. |
Discovered bacteriophages |
|
IV. |
Stanley |
4. |
Crystallized TMV |
Codes:
|
I |
II |
III |
IV |
||
|
(1) |
2 |
1 |
3 |
4 |
|
|
(2) |
2 |
3 |
1 |
5 |
|
|
(3) |
4 |
5 |
1 |
3 |
|
|
(4) |
4 |
3 |
1 |
5 |
Identify the incorrect statement:
| 1. | Virus has a protein coat called capsid made of small subunits called capsomeres. |
| 2. | The core has the genetic material - either RNA or DNA or both. |
| 3. | The capsomeres are arranged in helical or icosahedral forms. |
| 4. | Viruses can infect any type of organism. |
Consider the following statements regarding lichens:
(i) The largest number of lichenized fungi belongs to basidiomycota.
(ii) The photosynthetic partner is either a green alga or a cyanobacterium normally
(iii) Lichens are poikilohydric
(iv) Both the lichen and the fungal partner bear the same name.
The correct statements are
1. i ,ii and iii
2. ii , iii and iv
3. i, iii and iv
4. i, ii , iii and iv
Identify the incorrect statement:
| 1. | The earliest systems of classification used only gross superficial morphological characters. |
| 2. | Linnaeus classification of plants was a natural classification as it was based on the androecium structure. |
| 3. | Artificial systems gave equal weightage to vegetative and sexual characteristics. |
| 4. | Natural classification systems are based on natural affinities among the organisms |
Identify the incorrect statement:
| 1. | Numerical Taxonomy is based on all observable characteristics. |
| 2. | More weightage is given to reproductive characters and all other characters are given equal importance in numerical taxonomy. |
| 3. | Cytotaxonomy is based on cytological information like chromosome number, structure, behaviour |
| 4. | Chemotaxonomy uses the chemical constituents of the plant |
Match each term with its definition and choose your answer from the codes given:
|
I. |
Isogamy |
1. |
The union of two gametes |
|
II. |
Syngamy |
2. |
Two gametes, one larger than the other |
|
III. |
Heterogamy |
3. |
Two kinds of gametes, one motile and other non motile |
|
IV. |
Anisogamy |
4. |
Two kinds of morphologically distinct gametes |
|
V. |
Oogamy |
5. |
Both gametes morphologically indistinguishable |
Codes:
|
|
I |
II |
III |
IV |
V |
|
1. |
1 |
2 |
3 |
4 |
5 |
|
2. |
4 |
1 |
2 |
3 |
4 |
|
3. |
5 |
1 |
2 |
4 |
3 |
|
4. |
3 |
1 |
2 |
4 |
5 |
Identify the incorrectly matched pair:
| 1. | Chlamydomonas: | Microscopic unicellular algae |
| 2. | Volvox: | Colonial algae |
| 3. | Ulothrix: | Filamentous algae |
| 4. | Fucus: | Isogamous algae |
Identify the incorrect statement regarding algae:
| 1. | At least a half of the total carbon dioxide fixation on earth is carried out by algae through photosynthesis. |
| 2. | Around 70 species of freshwater algae can be used as food. |
| 3. | Certain marine brown and red algae produce large amounts of hydrocolloids (water holding substances), e.g., algin (brown algae) and carrageen (red algae) which are used commercially. |
| 4. | Agar, obtained from Gelidium and Gracilaria are used to grow microbes and in preparations of ice-creams and jellies. |
Identify the incorrect statement regarding green algae?
| 1. | They contain chl a, chl b as well as carotenoids. |
| 2. | The chloroplasts may be discoid, plate-like, reticulate, cup-shaped, spiral or ribbon-shaped in different species. |
| 3. | Most members have one or more storage bodies called pyrenoids located outside the chloroplasts. |
| 4. | They usually have a rigid cell wall made of an inner layer of cellulose and an outer layer of pectose. |
Consider the following statements regarding brown algae:
| I. | The pigments are chl a, c and xanthophylls, fucoxanthin |
| II. | Storage food is laminarin and mannitol |
| III. | The cellulosic cell wall is covered with algin |
| IV. | The protoplast have a centrally located vacuole |
| V. | Their photosynthetic organs are called as fronds - leaf like structures |
| VI. | They have pear shaped biflagellate zoospores |
| VII. | They have two unequal laterally attached flagella. |
The number of correct statements is
1. 5
2. 6
3. 7
4. 4
Identify the incorrect statement regarding mosses:
| 1. | Leaves are one cell thick except at the midrib and lack stomata. |
| 2. | The first gametophyte stage is protonema, that is creeping and green. |
| 3. | The second stage is the leafy stage, which develops from the secondary protonema as a lateral bud. |
| 4. | Rhizoids are elongated and unicellular. |
Evolutionarily, the first terrestrial plants to possess vascular tissues are:
1. Bryophytes
2. Pteridophytes
3. Cycades
4. Gnetales
Which of the following statements regarding gymnosperms are correct?
| I. | Ovules are naked - not enclosed by any ovary wall before fertilisation but seeds that develop after fertilisation are covered. |
| II. | In all living Gymnosperms, sporophyte is dominant over the gametophyte. |
| III. | They are heterosporous. |
| IV. | The male and female gametophytes do not have an independent existence. |
1. I, II and III
2. I, III and IV
3. II, III and IV
4. I, II, III and IV
Which human worm infestation are we talking about?
| I: | In socio-economic impact on the human race, it is second only to Malaria. |
| II: | The worm larva enters the human body by penetrating the skin |
| III: | The worm requires snails as an intermediate host. |
| IV: | In developing countries, it is a common cause of anaemia in children. |
1. Elephantiasis
2. Bilharziasis
3. Cysticercosis
4. Plague
Consider the following characteristics of Porifers. Which of these can be regarded as synapomorphies of Porifera?
| 1. | Collared cells called as choanocytes |
| 2. | Canal system with pores (ostia) in body wall |
| 3. | Mineral spicules |
| 4. | High cellular mobility and totipotency |
Identify the incorrect statement:
1. Medusa of Scyphozoans are called as jelly fishes.
2. Ctenophores are the largest animals that move with the help of cilia.
3. Flat worms that have a digestive cavity, have an incomplete gut.
4. Nematodes are covered with thick cuticle that does not moult.
Match Nematode with their common names and choose answer from codes given below:
|
|
Nematode |
|
Common names |
|
I. |
Ancylostoma duodenale |
1. |
Pin worm |
|
II. |
Enterobius vermicularis |
2. |
Filaria worm |
|
III. |
Trichuris trichiura |
3. |
Loa loa |
|
IV. |
Dracunculus medinensis |
4. |
Whip worm |
|
V. |
Onchocerca volvulus |
5. |
Hook worm |
|
VI. |
Wuchereria bancrofti |
6. |
Guinea worm |
Codes:
|
|
I. |
II. |
III. |
IV. |
V. |
VI. |
|
1. |
5 |
1 |
4 |
6 |
3 |
2 |
|
2. |
5 |
4 |
3 |
6 |
1 |
2 |
|
3. |
5 |
3 |
4 |
1 |
6 |
2 |
|
4. |
5 |
6 |
4 |
3 |
1 |
2 |
What is the most important reason for the immense success of arthropods?
1. Chitinous exoskeleton
2. Uricotelic nature
3. Compound eye
4. Multipurpose appendages
What is not true for a generalized mollusc?
| 1. | Body segmented into head, visceral mass and foot. |
| 2. | A rasping tongue like organ – radula present. |
| 3. | Mantle cavity with gills |
| 4. | Excretory structures – nephridia |
How many of the characters given below are true for echinoderms?
| I. | An endoskeleton of calcareous ossicles |
| II. | The adult echinoderms are radially symmetrical but larvae are bilaterally symmetrical. |
| III. | They are triploblastic and coelomate animals. |
| IV. | Digestive system is complete. |
| V. | Water vascular system |
| VI. | Sexual reproduction, internal fertilization, and direct development. |
1. 3
2. 4
3. 5
4. 6
Which of the following is not seen in hemichordates?
1. A tripartite body organization
2. Stomochord
3. Rudimentary notochord
4. Proboscis gland
Consider the following features:
I. Ampulla of Lorenzini
II. Operculum or gill cover
III. Ureotelism
IV. Viviparity
V. Swim bladder
VI. Lateral line system
The features that can be attributed to cartilaginous fishes are:
1. I, III and IV only
2. II, V and VI only
3. I, II, III and IV
4. All
Key features that appeared for the first time in amphibians do not include:
1. Legs
2. Nutritional deficiencies
3. Lungs
4. Pulmonary veins
Consider the following characters:
| I. | Wish bone furculum |
| II. | Pneumatic bones |
| III. | Feathers |
| IV. | Endothermy |
| V. | Left aortic arch |
| VI. | Four chambered heart |
Which of these are unique to birds?
| 1. | I, II and III only |
| 2. | I, II, III and IV only |
| 3. | I, II, III, IV and V only |
| 4. | All |
Identify the incorrectly matched pair:
|
1. |
Ornithorhynchus: |
Oviparous mammal |
|
2. |
Macropus: |
Marsupial mammal |
|
3. |
Balaenoptera: |
Largest land mammal |
|
4. |
Pteropus: |
Flying mammal |
Match each item in Column I with one item in Column II and chose your answer from the codes given below:
|
Column I Feature |
Column II Phylum |
|
(a) Lateral line system (b) Water vascular system (c) Radula (d) Comb plates (e) Parapodia |
(i) Ctenophora (ii) Mollusca (iii) Echinodermata (iv) Osteichthyes (v) Annelida |
Codes
(a) (b) (c) (d) (e)
1. (i) (ii) (iii) (iv) (v)
2. (iv) (iii) (ii) (i) (v)
3. (iv) (iii) (i) (v) (ii)
4. (iii) (ii) (i) (v) (iv)
The type aestivation of calyx in members of fabaceae is:
1. Imbricate
2. Vexillary
3. Twisted
4. Valvate
In marginal placentation, the placenta forms a ridge along the:
1. Ventral suture of the ovary
2. Dorsal suture of the ovary
3. Anterior suture of the ovary
4. Posterior suture of the ovary
A lateral branch with short internodes and each node bearing a rosette of leaves and a tuft of roots is found in:
1. Chrysanthemum
2. Pistia
3. Jasmine
4. Pineapple
Zygomorphic flower is not found in:
1. Cassia
2. Canna
3. Gulmohur
4. Bean
The following diagram can represent the position of the ovary in:
1. Brinjal
2. Guava
3. Rose
4. Mustard
Ovary is one-chambered but becomes two-chambered due to the formation of the false septum in:
1. Argemone
2. Salvia
3. Cassia
4. Primrose
Which of the following is not a correct floral character of the members of family Solanaceae?
1. Flower : Zygomorphic
2. Calyx : Valvate aestivation
3. Stamens : Epipetalous
4. Fruit : Berry or capsule
Pneumatophores are seen in:
1. Rhizophora
2. Banyan tree
3. Carrot
4. Turnip
Stem tendrils are not seen in:
1. Grapevine
2. Watermelon
3. Citrus
4. Cucumber
When a shoot tip transforms into a flower:
1. It is always solitary
2. The flower is always short-lived
3. It is never solitary
4. The flower is always long-lived
Some foliar roots also produce adventitious roots as in:
1. Oxalis
2. Coleus
3. Tapioca
4. Bryophyllum
Chief characteristics of stem do not include:
1. Develops from the plumule
2. Positive phototropic nature
3. Presence of nodes and internodes
4. Endogenously developed multicellular hair
Match aerial modifications of stem given in column I with their descriptions in column II and choose your answer from the codes given below:
|
A |
Phylloclade |
P |
Green, flattened or cylindrical branches of unlimited growth |
|
B |
Cladode |
Q |
Branches of limited growth that become green and flat like a leaf |
|
C |
Bulbil |
R |
Modified vegetative bud with stored food meant for reproduction |
Codes:
|
|
A |
B |
C |
|
1. |
P |
Q |
R |
|
2. |
P |
R |
Q |
|
3. |
R |
P |
Q |
|
4 . |
Q |
R |
P |
In racemose type of branching:
1. Branches continue to grow indefinitely and give off further branches laterally in a basipetal order
2. Branches continue to grow indefinitely and give off further branches laterally in an acropetal order
3. Branches stop growing after some time and give off further branches laterally in a basipetal order
4. Branches stop growing after some time and give off further branches laterally in an acropetal order
When secretary granules leave cell by exocytosis with no loss of other cellular material, the glands are called
1. Holocrine
2. Merocrine
3. Apocrine
4. Autocrine
Large amoeboid cells, that are a part of our innate immune system, found in the areolar tissue are called as:
1. Macrophages
2. Mast cells
3. Fibroblasts
4. Adipocytes
With respect to blood, the composition of lymph can be expressed as:
1. No RBc, No Plateletes, Less proteins, Less calcium and phosphorus, Predominant corpuscles lymphocytes.
2. No RBc, No Plateletes, Less proteins, More calcium and phosphorus, Predominant corpuscles lymphocytes.
3. No RBc, Less or no Plateletes, Less proteins, Less calcium and phosphorus, Predominant corpuscles macrophages.
4. No RBc, less Plateletes, Less proteins, Less calcium and phosphorus, Predominant corpuscles monocytes.
The ATPase activity of the myosin head is dependent on:
1. Magnesium ions
2. Manganese ions
3. Calcium ions
4. Ferric ions
In cockroach, the formation of ootheca is by the secretions of:
1. Prothoracic glands of male
2. Prothoracic glands of female
3. Colleterial glands of male
4. Colleterial glands of female
Bipolar neurons are found in humans in:
1. Brain
2. Spinal cord
3. Embryonic stages
4. Olfactory epithelium
The myelin sheath in the CNS is synthesized by:
1. Oligodendrocytes
2. Astrocytess
3. Schwann cells
4. Microglia
A sample of CaCO3 is 50% pure. On heating 1.12 litres of CO2 (at STP) is obtained. The weight of residues left (assuming non-volatile impurity) is:
(Ca = 40, C= 12, O=16)
| 1. | 7.8 g | 2. | 5 g |
| 3. | 3.8 g | 4. | 2.8 g |
Consider the given reaction, 2SO2 + O2 2SO3
6.4 g SO2 and 3.2 g O2 to form SO3 . The mass of SO3 formed is:
1. 32 g
2. 16 g
3. 8 g
4. 4 g
The molality of a 20% (by mass) CaCO3 solution is :
(Given: Density of solution is 1.2 gm/ml)
| 1. | 1.25 m | 2. | 2.5 m |
| 3. | 2.08 m | 4. | 1.5 m |
1 gm butane (C4H10) is burnt with excess of O2 to form CO2. The approximate mass of CO2 produced is
1. 1gm
2. 2gm
3. 3gm
4. 4gm
If 0.5 moles of BaCl2 is reacted with 0.2 moles of Na3PO4 then the maximum moles of Ba3(PO4)2 formed is:
| 1. | 0.33 | 2. | 0.25 |
| 3. | 0.10 | 4. | 0.52 |
2 gm Iron pyrite (FeS2) is burnt with O2 to form Fe2O3 and SO2. The mass of SO2 produced is (Fe=56, S=32, O=16)
1. 2 gm
2. 2.13 gm
3. 4 gm
4. 4.26 gm
Mg(OH)2 + HCl Mg(OH)Cl + H2O2, the equivalent weight of Mg(OH)2 will be (M= Mol. wt. of Mg(OH)2)
1. zero
2. M
3. M/2
4. M/4
A metallic oxide contains 20% oxygen. The equivalent weight of metal is
1. 12
2. 16
3. 32
4. 64
1.575 gm crystalline oxalic acid C₂H₂O₄· XH₂O
1. Zero
2. 1
3. 2
4. 5
An isotone of 32Ge76 is:
(i) 32Ge77
(ii) 33As77
(iii) 34Se77
(iv) 34Se78
1. only (i) & (ii)
2. only (ii) & (iii)
3. only (ii) & (iv)
4. (ii) (iii) & (iv)
Which of the following has greater number of electrons than neutrons?
1. Al3+
2. O2-
3. F-
4. C
The relationship between the energy \(E_1\) of radiation with a wavelength of \(8000\) Å and the energy \(E_2\) of radiation with a wavelength of \(16000\) Å is:
1. \(E_1=6E_2\)
2. \(E_1=2E_2\)
3. \(E_1=4E_2\)
4. \(E_1=1 / 2 E_2\)
The transition of electrons in H atom that will emit maximum energy is:
1. n3 n2
2. n4 n3
3. n5 n4
4. n6 n5
If the speed of electron in Bohr's first orbit of hydrogen atom be x, then speed of the electron in 3rd orbit is:
1. x/9
2. x/3
3. 3x
4. 9x
In Bohr's model of the hydrogen atom the ratio between the period of revolution of an electron in the orbit n=1 to the period of revolution of the electron in the orbit n=2 is
1. 1:2
2. 2:1
3. 1:4
4. 1:8
A cricket ball of 0.5 kg is moving with a velocity of 100 ms-1. The wavelength associated with its motion is:
1. 1/100cm
2. 6.6 x 10-34m
3. 1.32 x 10-35m
4. 6.6 x 10-28m
If uncertainties in the measurement of position and momentum are equal, what is the uncertainty in the measurement of velocity?
1.
2.
3.
4. None of the above.
The First ionisation enthalpies of Na,Mg,Al and Si are in the order
1. Na<Mg>Al<Si
2. Na>Mg>Al>Si
3. Na<Mg<Al<Si
4. Na>Mg>Al<Si
The electronic configuration of chalcogens in their outermost shell is :
1.
2.
3.
4.
Among the following species, the smallest ion is :
| 1. | Na+ | 2. | F- |
| 3. | O2- | 4. | N3- |
The correct order for electron affinity of halogens is :
1. Br > F
2. F > Cl
3. Br > Cl
4. F > I
Which one of the elements has the highest ionization energy?
1. [Ne]3s23p1
2. [Ne]3s23p2
3. [Ne]3s23p3
4. [Ar]3d104s24p2
Element/Ion that has the highest electron affinity is:
| 1. | F- | 2. | O- |
| 3. | O | 4. | Na |
The correct order of second ionisation energies of B, C, N, & O is
1. O > N > C > B
2. O > C > N > B
3. O > B > C > N
4. O > N >B > C
An atom of the element A has 3 electrons in its outermost shell while another atom X has 6 electrons in its outermost shell. The formula of the compound between A and X could be.
1. A2X3
2. AX3
3. A3X2
4. A2X
Among the following iso-structural compounds, the one that has the highest lattice energy is:
1. LiCl
2. MgO
3. NaCl
4. LiF
A sbonded molecule MX3 is T-shaped. The number of non-bonding pairs of electrons is :
1. 0
2. 2
3. 1
4. Can be predicted only if atomic number of M is known,
In a chemical change from , the hybrid state of P changes from:
1. sp2 to sp3
2. sp3 to sp2
3. sp3 to sp3d
4. sp3 to dsp2
The geometrical arrangement and shape of are respectively
1. Trigonal bipyramidal geometry, linear shape
2. Hexagonal structure, linear shape
3. Triangular planar geometry, triangular shape
4. Tetrahedral geometry, pyramidal shape
Which molecules/ions are most paramagnetic?
1. B2
2. C2
3.
4.
Most basic oxide is
1.
2.
3.
4.
The correct statement is:
1. When is converted into then bond distance increases
2. When is converted into then bond distance increases
3. When CO is converted into then bond distance increases
4. All of these
An ion that has the largest size among the following is :
1. Li+(aq)
2. Cs+(aq)
3. Li+(g)
4. K+(aq)
In which case observed dipole moment is greater than theoretical dipole moment?
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Which has linear shape but not sp hibridisation ?
1.
2.
3.
4. Both 1 and 2
A particle is moving along the path y = from x = 0 m to x = 2 m. Then the distance traveled by the particle is:
1. 4 m
2.
3.
4.
Six particles situated at the corners of a regular hexagon of side \(a\) move at constant speed \(v\). Each particle maintains a direction towards the particle at the next. The time which the particles will take to meet each other is:
1. \(\frac{2 a}{v}~\text{sec}\)
2. \(\frac{a}{v}~\text{sec}\)
3. \(\frac{2 a}{3v}~\text{sec}\)
4. \(\frac{3 a}{v}~\text{sec}\)
The value of \(M\) of the hanging block is in the figure, which will prevent the smaller block (\(m\)\(=\)\(1\) kg) from slipping over the triangular block. All the surfaces are smooth and string and pulley are ideal. (Given: \(M'\)\(=4\) kg and \(\theta\) \(=37^\circ\))
1. \(12\) kg
2. \(15\) kg
3. \(10\) kg
4. \(4\) kg
A block of mass m = 25 kg on a smooth horizontal surface with a velocity =3 meets the spring of spring constant k = 100 N/m fixed at one end as shown in the figure. The maximum compression of the spring and velocity of the block as it returns to the original position respectively are:

1. 1.5 m, -3
2. 1.5 m, 0
3. 1.0 m, 3
4. 0.5 m, 2
A lighter body and heavier body both are moving with same momentum and applied same retarding force. Their stoping distances are respectively. Then the correct relation between -
1.
2.
3.
4. None of these
The velocity, given to the block of mass (m), is to rotate it in a circle of radius l. Calculate the height (h) where the block leaves the circle.
1.
2.
3.
4. None of these
A body is projected with velocity m/s with an angle of projection 60 with horizontal. Calculate velocity on that point where body makes an angle 30 with the horizontal.
1. 20 m/s
2.
3.
4. 10 m/s
| 1. | \(2~\text{kg}\) | 2. | \(3~\text{kg}\) |
| 3. | \(4~\text{kg}\) | 4. | \(5~\text{kg}\) |

If length of string is
1. 10 m/s
2. 20 m/s
3.
4.
A simple pendulum (bob of mass \(m\)) oscillates in a vertical plane. Let \(\theta\) be the angular displacement from the vertical. The tension in the string is equal to \(mg \mathrm{cos\theta}\) under which condition?
1. Always
2. never
3. At the extreme position
4. At the mean position
In a uniform circular motion, which of the following quantity is not constant
1. Angular momentum
2. Speed
3. Kinetic energy
4. Momentum
A metal ring of mass m and radius R is placed on smooth horizontal table and is set rotating about its own axis in such a way that each part of the ring moves with a speed v. Tension in the ring is:
1.
2.
3.
4. None of these
Tension on the string at point \(P\) is \(T\). The graph for tension \((T)\) versus \(x\) is shown in the figure. Then the string is:

| 1. | massless |
| 2. | massfull |
| 3. | tension on every point on the string is same when the string is having finite mass \(4\) |
| 4. | None of these |
The relation between velocity (v) and time (t) is , then which one of the following quantity is constant?
1. Force
2. Power
3. Momentum
4. Kinetic Energy
A particle is moving on the circular path of the radius (R) with centripetal acceleration . Then the correct relation showing power (P) delivered by net force versus time (t) is

1. 1
2. 2
3. 3
4. 4
While walking on ice one should take small steps to avoid slipping. This is because smaller steps ensure
1. Larger friction
2. Smaller friction
3. Larger normal force
4. Smaller normal force
A particle is moving with veocity ; where k is constant. The general equation for the path is:
1.
2.
3.
4. xy=constant
Two forces, \(1\) N and \(2\) N, act along with the lines \(x=0\) and \(y=0\). The equation of the line along which the resultant lies is given by:
1. \(y-2x =0\)
2. \(2y-x =0\)
3. \(y+x =0\)
4. \(y-x =0\)
If the velocity of light (c), gravitational constant (G) and Planck's constant (h) are chosen as fundamental units, then which of the following represents the dimensions of the mass?
1.
2.
3.
4.
A block is placed on a rough horizontal plane. A time dependent horizontal force F=kt acts on the block. The acceleration time graph of the block is :
| 1. |  |
2. |  |
| 3. |  |
4. |  |
A projectile of mass 50 kg is shot vertically upwards with an initial velocity of 100ms-1. After 5 seconds it explodes into two fragments, one of which having mass 20 kg, travels vertically up with a velocity of 150 ms-1. The velocity of the other fragment at that instant is: [Take g= 9.8 m/]
1. 100 ms-1
2. 150 ms-1
3. -150 ms-1
4. -15 ms-1
The displacement \(x\) of a particle moving in one dimension under the action of a constant force is related to time \(t\) by the equation \(t=\sqrt{x}+3,\) where \(x\) is in meters and \(t\) is in seconds. What is the displacement of the particle from \(t=0~\text s\) to \(t = 6~\text s?\)
1. \(0\)
2. \(12~\text m\)
3. \(6~\text m\)
4. \(18~\text m\)
A body thrown vertically so as to reach its maximum height in t second. The total time from the time of projection to reach a point at half of its maximum height while returning (in second) is:
1.
2.
3.
4.
A stone falls freely from rest from a height h and it travels a distance in the last second. The value of h is:
1. 145 m
2. 100 m
3. 125 m
4. 200 ms
Component of perpendicular to and in the same plane as that of is:
1.
2.
3.
4.
Given that \(\vec {C}= \vec{A}+\vec {B}~\text{and}~\vec{C}\) makes an angle \(\alpha\)
1. \(\alpha \) cannot be less than \(\beta\)
2. \(\alpha <\beta, ~\text{if}~A<B\)
3. \(\alpha <\beta, ~\text{if}~A>B\)
4. \(\alpha <\beta, ~\text{if}~A=B\)
If the angle between the vector is , the value of the product is equal to:
1.
2.
3.
4. zero
A steel wire can withstand a load up to 2940 N. A load of 150 kg is suspended from a rigid support. The maximum angle through which the wire can be displaced from the mean position, so that the wire does not break when the load passes through the position of equilibrium, is (2008 E)
1. 30
2. 60
3. 80
4. 85
A particle is projected with a velocity u making an angle with the horizontal. At any instant, its velocity v is at right angles to its initial velocity u; then v is:
1. ucos
2. utan
3. ucot
4. usec
A projectile is given an initial velocity of . The cartesian equation of its path is (g = 10 )
1.
2.
3.
4.
A ship A is moving westwards with a speed of 10 km and a ship B, 100 km south of A is moving northwards with a speed of 10 km . The time after which the distance between them becomes the shortest, is:
1. 5 hr
2. hr
3. hr
4. 0 hr
Time taken by the projectile to reach from A to B is t. Then the distance AB is equal to :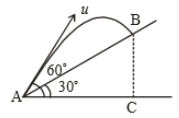

1.
2.
3.
4. 2ut
A sphere of mass m moving with constant velocity hits another sphere of the same mass at rest. If e is the coefficient of restitution. The ratio of their velocities after the collision is
1. 1 + e
2.
3.
4.
A particle is moving in a vertical circle. The tension in the string when passing through two positions at angle of 30 and 60 from vertical (the lowest position) are respectively, then:
1.
2.
3.
4. Tension in the string always remains the same.
A body is thrown vertically up with a certain initial velocity. The potential and the kinetic energy of the body are equal at a point P in its path. If the same body is thrown with double the velocity upwards, the ratio of the potential and the kinetic energies of the body when it crosses at the same point will be:
1. 1:1
2. 1:4
3. 1:7
4. 1:8
A motorcycle is going on an overbridge of radius R. The driver maintains a constant speed. As the motorcycle is ascending on the overbridge, the normal force on it:
1. Increases
2. decreases
3. remains the same
4. fluctuates erratically
A rod of length L and mass m is acted on by two unequal forces and as shown in the following figure

The tension in the rod at a distance y from the end A is given by :
1.
2.
3.
4. None of the above
In a FCC lattice, atom A occupies the corner positions and atom B occupies the face centred positions. If one atom of B is missing from one of the face centred points, the formula of the compound is
1. A2B
2. AB2
3. AB
4. A2B5
Mark the incorrect order for the bond angle:
1. NH3 > NF3
2. NF3 < PF3
3. NH3 > PH3
4. NH3 > H2O