| Assertion(A): | In aerobic respiration presence of O2 is necessary. |
| Reason(R): | O2 in aerobic respiration plays only one role i.e. terminal acceptor of the electron. |
| 1. | Both (A) & (R) are true and (R) is the correct explanation of (A) |
| 2. | Both (A) & (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is a true statement but (R) is false. |
| 4. | Both (A) and (R) are false statements. |
During glycolysis:
| 1. | ATP molecules are synthesized between fructose-6-phosphate and fructose 1, 6-diphosphate |
| 2. | Water molecules are released between PEP and pyruvate |
| 3. | is oxidized between PGAL and BPGA |
| 4. | ATP molecules are synthesized in the conversion of BPGA to PGA |

| Options | A | B | C |
| 1. | SDP | DNP | LDP |
| 2. | SDP | LDP | SDP |
| 3. | DNP | LDP | SDP |
| 4. | SDP | LDP | DNP |
| Column -I | Column-II | ||
| A. | Substitute of vernalisation | (i) | High temperature |
| B. | Critical photoperiod | (ii) | ABA |
| C. | Stress hormone | (iii) | Gibberellin |
| D. | Devernalisation | (iv) | SDP and LDP |
| 1. | A = (i) , B =(ii) , C= (iv) , D=(iii) |
| 2. | A = (i) , B =(iii) , C= (ii) , D=(iv) |
| 3. | A = (iii) , B =(iv) , C= (ii) , D=(i) |
| 4. | A = (ii) , B =(iv) , C= (i) , D=(iii) |
In the figure given below the correct match is: 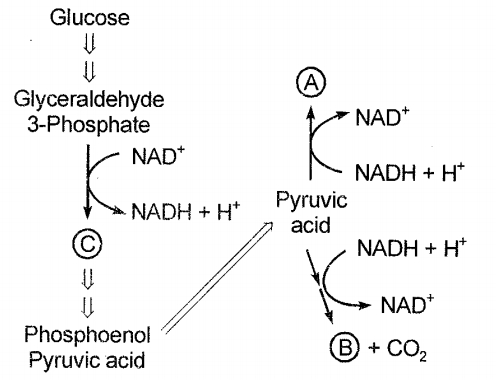
1. A-Ethanol, B-Lactic acid, C-Dihydroxyacetone phosphate
2. A-Lactic acid, B-Ethanol, C-Oxaloacetic acid
3. A-Lactic acid, B-Ethanol, C-3-Phosphoglyceric acid
4. A-Ethanol, B-Lactic acid, C-3-phosphoglyceric acid
During citrate synthesis in the Krebs cycle:
| 1. | A molecule of CoA is used. |
| 2. | There is a condensation of the acetyl group with oxalosuccinic acid. |
| 3. | There is a condensation of the acetyl group with oxaloacetic acid and water. |
| 4. | Succinyl CoA is oxidised in presence of citrate synthetase. |
The growth of a root elongating at a constant rate will be represented by:
1. A only
2. B only
3. Both A and B
4. Neither A nor B
Suppose there are three cells A, B, and C as shown in the figure. What is the direction of movement of water?

1.
2.
3.
4.
In which step of glycolysis does water form?
| 1. | Fructose 1,6 –bisphosphate | → | Glyceraldehyde-3-phosphate |
| 2. | 1,3 bisphosphoglyceric acid | → | 3-phosphoglyceric acid |
| 3. | 2-phosphoglycerate | → | Phosphoenolpyruvate |
| 4. | Phosphoenolpyrespruvate | → | Pyruvic acid |
| (i) | Causes horizontal growth of seedlings. |
| (ii) | It is highly effective in causing fruit ripening. |
| (iii) | Is responsible for respiratory climatic in fruits which is the enhanced respiration rate during fruit ripening. |
| (iv) | Ethylene breaks seed and bud dormancy, initiates germination in peanut seeds, and causes the sprouting of potatoes. |
| (v) | It is used to initiate flowering and for synchronising fruit set in pineapples. |
Which one of the following statement is correct?
| 1. | Under certain conditions organic acids can be used as a respiratory substrate. |
| 2. | Carbon skeleton produced during respiration is used as a precursor for the biosynthesis of other molecules. |
| 3. | During cellular respiration, energy is released in a series of slow stepwise reactions. |
| 4. | All of the above statements are correct. |
Which one of the following concerns photophosphorylation:
| 1. | \(\mathrm{ADP}+\mathrm{AMP} \xrightarrow{\text { Lightenergy }} \text { ATP }\) |
| 2. | \(\mathrm{ADP}+\text { Inorganic } \mathrm{PO}_4 \xrightarrow{\text { Lightenergy }} \text { ATP }\) |
| 3. | \(\text { ADP }+ \text { Inorganic } \mathrm{PO}_4 \rightarrow \text { ATP }\) |
| 4. | \(\mathrm{AMP}+\text { Inorganic } \mathrm{PO}_4 \xrightarrow{\text { Lightenergy }} \text { ATP }\) |
Which of the following is not a feature of the active transport of solutes in plants?
1. Occurs against the concentration gradient
2. Non-selective
3. Occurs through membranes
4. Requires ATP
| Statement I: | In fermentation, there is a net gain of 12 molecules of ATP for each molecule of glucose degraded to pyruvic acid whereas only 2 molecules of ATP are generated under aerobic conditions. |
| Statement II: | Fermentation accounts for only a partial breakdown of glucose whereas in aerobic respiration it is completely degraded to CO2 and H2O. |
Which of the following statements regarding mitochondria is incorrect?
| 1. | Mitochondrial matrix contains single circular DNA molecule and ribosomes. |
| 2. | Outer membrane is permeable to monomers of carbohydrates, fats, and proteins. |
| 3. | Enzymes of electron transport are embedded in the outer membrane. |
| 4. | Inner membrane is convoluted with infoldings. |
Match the following columns.
|
Column I |
Column II |
|
A. Molecular oxygen B. Electron acceptor C. Pyruvate dehydrogenase D. Decarboxylation |
1. - ketoglutaric acid 2. Hydrogen acceptor 3. Cytochrome-c 4. Acetyl Co - A |
Codes
| Options: | A | B | C | D |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 3 | 4 | 2 | 1 |
| 3. | 2 | 1 | 3 | 4 |
| 4. | 4 | 3 | 1 | 2 |
Which of the following statements is incorrect?
| 1. | ATP is synthesized through complex V. |
| 2. | Oxidation-reduction reactions produce a proton gradient in respiration. |
| 3. | During aerobic respiration, the role of oxygen is limited to the terminal stage. |
| 4. | In ETC (Electron Transport Chain), one molecule of NADH+H+ gives rise to 2 ATP molecules, and one FADH2 gives rise to 3 ATP molecules. |
| Assertion(A): | 2,4-D is widely used as an herbicide. |
| Reason(R): | It is used to kill mature monocotyledonous plants. |
| 1. | Both (A) & (R) are true and (R) is the correct explanation of (A) |
| 2. | Both (A) & (R) are true but (R) is not the correct explanation of (A) |
| 3. | (A) is a true statement but (R) is false. |
| 4. | Both (A) and (R) are false statements. |
Find the wrong statement about the Electron Transport system:
| 1. | Release and utilize the energy stored in NADH and FADH2 |
| 2. | Oxidizes NADH and reduces FADH2 |
| 3. | Electrons are passed on to oxygen resulting in the formation of water |
| 4. | The metabolic pathway through which electron passes from one carrier to another |
| 1. | Enzymes of the TCA cycle are present in the mitochondrial matrix. |
| 2. | Glycolysis occurs in the cytosol. |
| 3. | Glycolysis operates as long as it is supplied with NAD that can pick up hydrogen atoms. |
| 4. | Oxidative phosphorylation takes place in the outer mitochondrial membrane. |
Select the incorrect statement regarding the physiological effects of auxins.
| 1. | Auxin promotes growth in roots |
| 2. | Auxin rapidly increases the extensibility of cell wall |
| 3. | Auxin has no effect on vascular differentiation |
| 4. | Phototropism is mediated by the lateral redistribution of auxin |

| 1. | A = Initial slow growth, B = Rapid growth, C = Phase of growth during limited nutrient supply |
| 2. | A = Rapid growth, B = Initial slow growth, C = Phase of growth during limited nutrient supply |
| 3. | A = Lag phase, B = Stationary phase, C = Exponential phase |
| 4. | A = Exponential phase, B = Stationary phase, C = Lag phase |
How many statements are correct with respect to ETS?
1. ETS is helpful in the restoration of NAD
2. is terminal e acceptor in electron transport system
3. Ubiquinone & FMN are hydrogen carriers and helpful in the movement of H from matrix to space.
4. Complex lV is also known as cytochrome C oxidase.
5. Movement of e in ETS is due to variable valency of metal ions present in carrier complexes.
1. 3
2. 4
3. 5
4. 2
Read the following statements (A-D):
| A. | Helps in the closing of stomata. |
| B. | Induces seed dormancy. |
| C. | Inhibits cambial activity. |
| D. | Seedling develops epicotyl hook. |
Select the correct option with respect to the physiological role of ABA (abscisic acid) by selecting True (T) and False (F).
| A | B | C | D | |
| 1. | F | T | T | T |
| 2. | T | F | F | F |
| 3. | T | T | T | F |
| 4. | F | T | T | F |
GTP is formed at which step of the Krebs cycle during aerobic respiration?
1. Conversion of isocitric acid into oxalosuccinic acid.
2. Oxidation of -Ketoglutaric acid into succinyl co-A.
3. Conversion of succinyl Co-A into succinic acid.
4. Oxidation of malic acid into oxaloacetic acid.
What is correct about the movement of substances across the membrane in facilitated diffusion?
1. It is an active transport.
2. It causes the transport of molecules from low concentration to high concentration.
3. It is insensitive to inhibitors.
4. It is a very specific transport.
Identify A, B, C, and D in the following diagram:
| 1. | A - Inner mitochondrial membrane; B - ; C - ; D - Matrix |
| 2. | A- Inner mitochondrial membrane; B - ; C - Matrix; D - |
| 3. | A - Matrix; B - ; C - Inner mitochondrial membrane; D - |
| 4. | A - Cytoplasm; B - ; C - Matrix; D - |
Decapitation is widely applied in tea plantations because it:
| 1. | Inhibits lateral bud formation nearby the tip |
| 2. | Induces lateral bud formation nearby the tip |
| 3. | Induces lateral bud growth |
| 4. | More than one option is correct |
| Assertion (A): | In coriander, the leaves of juvenile plants are different in shape from those in mature plants. |
| Reason (R): | It shows environmental plasticity. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true, but (R) is not the correct explanation of (A). |
| 3. | (A) is true, but (R) is false. |
| 4. | Both (A) and (R) are false. |
The common feature of aerobic respiration and the alcoholic fermentation process is:
1. ATP and metabolic as end products
2. NADH + oxidation
3. Oxygen use
4. Complete breakdown of glucose to &
| Assertion (A): | Development is the sum total of growth and differentiation. |
| Reason (R): | Development is controlled by extrinsic factors as well as intrinsic factors. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true, but (R) is not the correct explanation of (A). |
| 3. | (A) is true, but (R) is false. |
| 4. | Both (A) and (R) are false. |
| 1. | Carbohydrates are used as respiratory substrates |
| 2. | Fats are used as respiratory substrates |
| 3. | Proteins are used as respiratory substrates |
| 4. | Amino acids are used as respiratory substrates |
The effects of abscisic acid in plants include:
| I. | It induces stomatal closure, decreasing transpiration to prevent water loss. |
| II. | Stimulates fruit ripening. |
| III. | Responsible for seed dormancy by inhibiting cell growth—inhibits seed germination. |
| IV. | Stimulates the synthesis of Kinetin nucleotide. |
| V. | Regulates enzymes needed for photosynthesis. |
1. I, II, III, IV, V
2. I, III, V
3. I, II, III, IV
4. II, III, IV, V
Which of the following is an incorrect statement?
1. An analysis of xylem exudate suggests that some of the nitrogen travels as inorganic ions and much of it is carried in the organic form as amino acids.
2. Small amount of P and S is also carried as inorganic compounds in xylem exudate.
3. There is exchange of material between the xylem and phloem.
4. Xylem transports inorganic nutrients while phloem transports organic materials, this observation was proved wrong by the analysis of xylem exudate.
Which of the following facilitates the opening of the stomatal aperture?
1. Contraction of the outer wall of guard cells
2. Decrease in turgidity in guard cells
3. Radial orientation of cellulose microfibrils in the cell wall of guard cells
4. Longitudinal orientation of cellulose microfibrils in the cell wall of guard cells
Match column l and column ll.
| Column I | Column II | ||
| (a) | (i) | catalase | |
| (b) | (ii) | Chlorophyll | |
| (c) | (iii) | Phosphorylation | |
| (d) | (iv) | Coenzyme A | |
| (a) | (b) | (c) | (d) | |
| 1. | (i) | (ii) | (iv) | (iii) |
| 2. | (ii) | (iii) | (iv) | (i) |
| 3. | (iv) | (iii) | (ii) | (i) |
| 4. | (iv) | (i) | (ii) | (iii) |
In muscles, during exercise, when oxygen is inadequate for cellular respiration:
| 1. | Pyruvic acid is oxidised to lactic acid |
| 2. | 45% of the energy in glucose is released in the form of ATP |
| 3. | NADH + is oxidised to NA |
| 4. | Pyruvic acid is reduced to ethanol and |
| 1. | Conversion of shoot apical meristem into cortex is a case of differentiation |
| 2. | The non-living differentiated cells can regain the capacity of division which is called dedifferentiation |
| 3. | The formation of interfascicular cambium and cork cambium can occur from the fully differentiated parenchyma |
| 4. | Secondary xylem and phellem are the products of dedifferentiation |
In which of the following locations of the plant cell, energy currency is formed?
(a) Nucleus
(b) Chloroplast
(c) Cytosol
(d) Mitochondrion
The correct one(s) is/are:
| 1. | All (a), (b), (c) & (d) | 2. | (b), (c) & (d) only |
| 3. | (d) only | 4. | (b) & (d) only |
The oxygenation activity of RuBisCo enzyme in photorespiration leads to the formation of:
| 1. | 1 molecule of the 3-C compound |
| 2. | 1 molecule of the 6-C compound |
| 3. | 1 molecule of a 4-C compound and 1 molecule of the 2-C compound |
| 4. | 2 molecules of the 3-C compound |
Crops such as tomatoes and bell pepper, allowed to grow in a carbon dioxide rich environment, show higher yields because:
| 1. | They show an increased rate of photosynthesis at higher carbon dioxide concentrations. |
| 2. | They can respond to high carbon dioxide conditions even in low light conditions. |
| 3. | They show C4 pathway for carbon fixation at high carbon dioxide is the limiting factor in such plants. |
| 4. | Only carbon dioxide is the limiting factor in such plants. |
| 1. | Light energy excites electrons in the thylakoid membrane electron transport chain. |
| 2. | Photons are passed along to a reaction-center chlorophyll. |
| 3. | The P680 chlorophyll donates a pair of protons to NADP+, which is thus converted to NADPH. |
| 4. | The electron vacancies in P680+ are filled by electrons derived from water. |
The photochemical phase of photosynthesis includes:
| 1. | Light absorption & formation of high-energy chemical intermediates. |
| 2. | Light absorption, water splitting, and synthesis of carbohydrates. |
| 3. | Light absorption, oxygen release, and formation of high-energy chemical intermediates. |
| 4. | Light absorption, water splitting, trapping of and formation of high-energy chemical intermediates. |
Select the incorrectly matched pair with respect to fixation:
1. Reductive amination - Transaminase
2. Nitrate reductase - Molybdoflavoprotein
3. Source of electrons - Ferredoxin
for nitrite reduction
4. Fixation of one - Requires 16 ATP
| A: | Glycolysis is the first common step of both aerobic as well as anaerobic respiration. |
| R: | 1 molecule of hexose sugar is converted into molecules of pyruvic acid in the absence or presence of oxygen in glycolysis. |
| 1. | If both (A) & (R) are true and the (R) is the correct explanation of the (A), then mark (1). |
| 2. | If both (A) & (R) are true but the (R) is not the correct explanation of the (A), then mark (2). |
| 3. | If (A) is a true statement but the (R) is false, then mark (3). |
| 4. | If both (A) and (R) are false statements, then mark (4). |
| 1. | When shoots are exposed to light, a chemical substance migrates towards the light. |
| 2. | Agar contains a chemical substance that mimics a plant hormone. |
| 3. | A chemical substance involved in shoot bending is produced in the shoot tips. |
| 4. | Once the shoot tip is cut, normal growth cannot be induced. |
Minerals known to be required in large amounts for plant growth include:
1. Magnesium, sulphur, iron, zinc
2. Phosphorus, potassium, sulphur, calcium
3. Calcium, magnesium, manganese, copper
4. Potassium, phosphorus, selenium, boron
| Column-I | Column-II | ||
| a. | Glucagon | (i) | Steroid |
| b. | Epinephrine | (ii) | Peptide |
| c. | Thyroxine | (iii) | Iodothyronine |
| d. | Estradiol | (iv) | Amino-acid derivative |
Match each item in Column I with one in Column II and choose your answer from the codes given below:
| Column I | Column II | ||
| (a) | Estrogen | (i) | Supports pregnancy |
| (b) | Progesterone | (ii) | Secondary sexual characters in females |
| (c) | FSH | (iii) | Regulates spermatogenesis |
| (d) | LH | (iv) | Ovulation |
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (i) | (iii) | (iv) |
| 2. | (i) | (iii) | (ii) | (iv) |
| 3. | (i) | (ii) | (iv) | (iii) |
| 4. | (i) | (ii) | (iii) | (iv) |
The structure marked as X in the following diagram showing a sagittal section of the human brain would be:
1. Corpora quadrigemina
2. Limbic system
3. Corpus callosum
4. Hippocampal gyrus
Human body temperature is regulated by the centre located in:
1. Cerebrum
2. Cerebellum
3. Medulla
4. Hypothalamus
The muscle can directly use which of the following as a source of energy?
1. Creatine phosphate
2. Glucose
3. ATP
4. Fatty acids
A motor unit consists of:
1. A motor neuron and all the muscle fibers innervated by it
2. All the motor neurons innervating one muscle fiber
3. All the muscle fibers in a fasciculus
4. All the muscle fibers in a skeletal muscle
| 1. | Exchange of NaCl between the ascending limb of Henle's loop and the descending limb of the vasa recta. |
| 2. | NaCl returns to the interstitium by the ascending portion vasa recta. |
| 3. | Small amount of urea enters the ascending limb of Henle's loop and transport back to the interstitium. |
| 4. | All of the above. |
A canal called the cerebral aqueduct passes through the:
1. Cerebral hemispheres
2. Diencephalon
3. Mid brain
4. Hind brain
A hormone that is antagonist to the parathyroid hormone is secreted from :
1. Thyroid
2. Adrenal cortex
3. Hypothalamus
4. Parathyroid itself
| 1. | Receiving of signal from a sensory organ |
| 2. | Transmission of impulsive into the central nervous system through effector organ |
| 3. | Carrying of signal from the central nervous system to the effector |
| 4. | Response to an involuntary peripheral nervous stimulation |
In the given diagram of the human skull, the bones represented by the letters A, B, and C are respectively:
1. Sphenoid, Ethmoid and Lacrimal
2. Ethmoid, Sphenoid and Lacrimal
3. Sphenoid, Lacrimal and Ethmoid
4. Ethmoid, Lacrimal and Sphenoid
| 1. | a, b, and c | 2. | b, c, and e |
| 3. | a, b, and d | 4. | c, d, and e |
In the human eye, there are three types of cones – red, blue, and green. When these cones are stimulated equally, a sensation for which of the following colors would be produced?
1. White
2. Black
3. Orange
4. Yellow
Low intelligence quotient abnormal skin and deafmutism are related to:
1. Low secretion of growth hormone
2. Hypothyroidism
3. Hyperparathyroidism
4. Hyposecretion of adrenal cortex hormone
Glucocorticoids do not:
1. Stimulate gluconeogenesis
2. Cause lipolysis
3. Cause proteolysis
4. Stimulate cellular uptake and utilization of amino acids
The amino acid derivative among the following hormone is:-
1. Insulin
2. Epinephrine
3. Estradiol
4. Testosterone
| a. | Sarcoplasmic reticulum of the muscle fibres is the storehouse of calcium ions. |
| b. | The type of muscles present in our thigh are striated and voluntary. |
| c. | The H-zone in the skeletal muscle fibre is due to the central gap between myosin filaments in the A-band. |
| d. | Production of body heat and storage of minerals are the functions of the skeletal system. |
Mark the correct statement:
| 1. | Electrical synapses are more common in our neural system than chemical synapses. |
| 2. | The new potential in the post-synaptic neurons may be either excitatory or inhibitory. |
| 3. | Hypothalamus is the major coordination centre for sensory and motor signaling. |
| 4. | The tracts of nerve fibres that connect two cerebral hemispheres are called corpora bigemina. |
Scapula is a large triangular flat bone situated in the dorsal part of the thorax between:
1. the second and fifth ribs
2. the second and seventh ribs
3. the third and sixth ribs
4. the third and eighth ribs
The normal value of GFR is approximately:
1. 650 ml/min
2. 180 ml/min
3. 180 ml/day
4.125 ml/min
The sensory neuron enters the spinal cord through:
| 1. | The dorsal root of the spinal nerve |
| 2. | The ventral root of the spinal nerve |
| 3. | Either dorsal or ventral root depending on the point of origin |
| 4. | Both dorsal and ventral roots of the spinal nerves |
A person passes a lot of dilute urine and drinks a lot of water but does not have glycosuria. He is most likely suffering from :
1. Type 1 diabetes mellitus
2. Type 2 diabetes mellitus
3. Pituitary diabetes
4. Diabetes insipidus
Which area actually secretes renin into the blood?
1. macula densa
2. juxtaglomerular apparatus
3. juxtaglomerular cells
4. cortical nephron
| A. | Increase in Ca++ concentration level leads to the binding of calcium with a subunit of troponin on actin filaments. and thereby removes the masking of active sites for myosin. |
| B. | During the shortening of the muscle, A-bands get reduced, whereas, the I-bands retain the length. |
| C. | A muscle fibre is a syncytium. |
| D. | Isotropic band of a myofibril consists of myosin. |
Juxtaglomerular apparatus is a special sensitive region formed by cellular modifications in the:
1. DCT and Efferent arteriole at the point of their contact
2. DCT and the Afferent arteriole at the point of their contact
3. PCT and Afferent arteriole at the point of their contact
4. PCT and Efferent arteriole at the point of their contact
Consider the following statements:
| I: | The resting axonal membrane is nearly impermeable to sodium ions. |
| II: | Depolarization of the axonal membrane is due to the influx of sodium ions. |
| III: | The size of the action potential, if produced, does not depend on the strength of the stimulus. |
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II and III
The brain stem does not include :
1. Mid-brain
2. Medulla oblongata
3. Pons Varolii
4. Cerebellum
Adenohypophysis is an endocrine gland that secretes all of the following except:
| 1. | corticotropin | 2. | luteinizing hormone |
| 3. | somatostatin | 4. | somatotropin |
Extracellular receptors must be required for the action of:
I. Oxytocin
II. Thyroxine
III. Epinephrine
IV. Glucagon
1. I, II, III & IV
2. I, IV only
3. I, III, IV
4. III & IV only
| Statement A: | Excitable cells called neuroglial cells make up more than one-half the volume of neural tissue in our body. |
| Statement B: | The greatest control over the body’s responsiveness to changing conditions is exerted by neural tissue. |
In cochlea, the auditory receptors are:
1. Organ of Corti
2. Ampulla
3. Macula
4. Hair cells
| 1. | Kidneys are reddish brown, round-shaped structures situated between the levels of the last thoracic and third lumbar vertebra. |
| 2. | Kidneys are close to the dorsal inner wall of the abdominal cavity. |
| 3. | Each kidney of adult human measures 0.5 - 0.7 m in width, 0.2 – 0.3 m in thickness with an average weight of 120 – 150 g. |
| 4. | Towards the centre of the inner convex surface of the kidney is a notch called the hilum through which the ureter, blood vessels, and nerves pass. |
During the concentration of urine by the human kidneys, NaCl is returned to the medullary interstitium by the:
1. Descending limb of the loop of Henle
2. Ascending limb of the loop of Henle
3. Descending limb of the vasa recta
4. Ascending limb of the vasa recta
Which hormone stimulates exocrine pancreas to secrete water and bicarbonate ions?
1. Gastrin
2. Cholecystokinin
3. Secretin
4. Gastric inhibitory peptide
The globular head of the myosin filament has active site/s for binding to:
I. ATP
II. the binding site for actin filament
III. Calcium
1. Only I
2. Only I and II
3. Only II and III
4. I, II, and III
Which statement about the glomerular filtration rate (GFR) is false?
| 1. | The GFR averages approximately 180 L per day. |
| 2. | The GFR will increase during fight-or-flight situations due to the vasodilation of afferent arterioles. |
| 3. | A decrease in GFR results in a decrease in the total urine output. |
| 4. | Renal autoregulation maintains the GFR at a relatively constant rate despite fluctuations in the mean arterial blood pressure. |
Cardiac muscles can be best defined as:
| 1. | Many cardiac muscle cells assemble in a branching pattern. |
| 2. | Single cardiac muscle fibre enclosed by connective tissue layer. |
| 3. | Group of multinucleated muscle fibres enclosed by connective tissue layer. |
| 4. | Group of unstriated muscle fibres enclosed by endocardium. |
What is correct for the hormone whose mechanism of action is shown in the diagram?
| 1. | This hormone will most likely be synthesized by the neural cells of the hypothalamus |
| 2. | This hormone cannot be thyroxin or triiodothyronine |
| 3. | This hormone can be a derivative of cholesterol |
| 4. | This hormone will have an almost instantaneous onset of action |
Consider the following statements:
| I. | Myasthenia gravis is an autoimmune disorder affecting the neuromuscular junction |
| II. | Muscular dystrophy is a progressive degeneration of skeletal muscles mostly due to a genetic disorder |
| III. | Tetany is rapid spasms in muscles due to high Ca++ in body fluid. |
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II and III
Thermal power plants can lead to-
1. Eutrophication
2. Acid rain
3. Blue baby syndrome
4. Ozone layer depletion
Dihydrogen of high purity (> 99.95%) is obtained through
| 1. | The electrolysis of warm Ba(OH)2 solution using Ni electrodes. |
| 2. | The reaction of Zn with dilute HCl. |
| 3. | The electrolysis of brine solution. |
| 4. | The electrolysis of acidified water using Pt electrodes. |
Choose the correct set of elements X, Y, and Z, which belong to groups 13, 14, and 15, respectively, from the options below.
Among the following statements about water, the FALSE statement is-
| 1. | Water is oxidized to oxygen during photosynthesis |
| 2. | Water can act both as an acid and as a base |
| 3. | There is extensive intramolecular hydrogen bonding in the condensed phase |
| 4. | Ice formed by heavy water sinks in normal water |



| Statement-I: | Hydrogenation of vegetable oils using nickel as a catalyst gives Vanaspati ghee. |
| Statement-II: | Hydroformylation of ethene gives propanal. |
Match the species given in Column I with the properties given in Column II.
|
Column I |
Column II |
|
A. Diborane |
(i). Used as a flux for soldering metals |
|
B. Gallium |
(ii). Banana bonds |
|
C. Borax |
(iii). Low melting, high boiling, useful for measuring high temperatures |
|
D. Aluminosilicate |
(iv). Used as a catalyst in petrochemical industries |
The correct match is-
1. A-(ii), B-(i), C-(iii), D-(iv)
2. A-(ii), B-(i), C-(iv), D-(iii)
3. A-(ii), B-(iii), C-(i), D-(iv)
4. A-(ii), B-(iii), C-(iv), D-(i)
Which of the following is incorrect?
1. Slaked lime → Ca(OH)2
2. Bleaching powder → Ca(OCl)2
3. Plaster of Paris → CaSO4· ½ H2O
4. Quick lime Ca(OH)2
Green chemistry deals with the:
1. Production of green vegetables
2. Reduction in the production of pollutants
3. Study of the ozone hole
4. Waste management
Among the following, the correct statement is-
| 1. | Beryllium exhibits a coordination number of six. |
| 2. | Chlorides of both beryllium and aluminum have bridged chloride structures in solid phase. |
| 3. | B2H6.2NH3 is known as 'inorganic benzene'. |
| 4. | Boric acid is a protonic acid. |
Both lithium and magnesium display several similar properties due to the diagonal relationship. However, the one which is incorrect is:
| 1. | Both form nitrides |
| 2. | Nitrates of both Li and Mg yield NO2 and O2 on heating |
| 3. | Both form basic carbonates |
| 4. | Both form soluble bicarbonates |
Sodium metal dissolves in liquid ammonia and forms a deep blue solution. The colour is due to the absorption of light by:
1. Sodium ions
2. Ammoniated electrons
3. Free electrons
4. Ammoniated sodium ions
A hydrated solid X on heating initially gives a monohydrated compound Y. Y upon heating above 373 K leads to an anhydrous white powder Z. X and Z, respectively, are:
1. Baking soda and dead burnt plaster.
2. Washing soda and soda ash.
3. Washing soda and dead burnt plaster.
4. Baking soda and soda ash
The pair of compounds that do not exist in solution together are:
1. NaHCO3 and NaOH
2. NaHCO3 and H2O
3. NaHCO3 and Na2CO3
4. Na2CO3 and NaOH
The correct statements among (a) to (d) are:
| (a) | Saline hydrides produce H2 gas when reacted with H2O. |
| (b) | Reaction of LiAlH4 with BF3 leads to B2H6. |
| (c) | PH3 and CH4 are electron-rich and electron-precise hydrides, respectively. |
| (d) | HF and CH4 are called molecular hydrides. |
1. (a), (b), (c) and (d)
2. (a), (c), and (d) only
3. (c) and (d) only
4. (a), (b) and (c) only
Sodium carbonate is manufactured by the Solvay process. The products that are recycled are:
1. CO2 and NH3
2. CO2 and NH4Cl
3. NaCl and CaO
4. CaCl2 and CaO
| Assertion (A): | SiF62- is known but SiCl62- is not. |
| Reason (R): | The size of fluorine is small and the interaction between the lone pair of chloride ions and Si4+ is not very strong. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
The O - O - H bond angles in the solid and gas phases of H2O2 are-
1. 90.2 and 111.5
2. 101.9 and 94.8
3. 90.2 and 94.8
4. 101.9 and 111.5
The correct statements among I to III regarding group 13 element oxides are:
(I) Boron trioxide or diboron trioxide is acidic.
(II) Oxides of aluminium and gallium are amphoteric.
(III) Oxides of indium and thallium are basic.
1. (I) and (II) only
2. (I) and (III) only
3. (II) and (III) only
4. (I), (II), and (III) only
The volume strength of 8.9 M H2O2 solution calculated at 273 K and 1 atm is-
(R = 0.0821 L atm K–1 mol–1) rounded off to the nearest integer)
1. 100
2. 80
3. 120
4. 60
| Statement I: | Excess nitrate in drinking water can cause diseases such as methemoglobinemia (‘blue baby’ syndrome). |
| Statement II: | Sodium chlorate (NaClO3), and sodium arsenite (Na3AsO3) are examples of herbicides. |
| 1. | Both Statement-I & Statement-II are true. |
| 2. | Both Statement-I & Statement-II are false. |
| 3. | Statement-I is true while Statement-II is false. |
| 4. | Statement-I is false while Statement-II is true. |
| Assertion (A): | Internuclear distance in D2 is equal to H2. |
| Reason (R): | Enthalpy of dissociation of D2 is more than H2. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
In view of the signs of for the following reactions:
The oxidation states more characteristic for lead and tin are respectively:
1. For lead +2, for tin +2
2. For lead +4, for tin +4
3. For lead +2, for tin +4
4. For lead +4, for tin +2
The structure of Al2Cl6 contains:
1. Four 2c-2e bonds and two 3c-2e bonds
2. Four 2c-2e bonds and four 3c-3e bonds
3. Four 2c-2e bonds and two 3c-4e bonds
4. Two 2c-2e bonds and four 3c-4e bonds
CO2 is passed through lime water. Initially, the solution turns milky and then becomes clear upon continued bubbling of CO2. The clear solution is due to the formation of-
1. CaCO3
2. CaO
3. Ca(OH)2
4. Ca(HCO3)2
(Me)2SiCl2 on hydrolysis, followed by condensation polymerization yields-
| 1. | (Me)2Si(OH)2 |
| 2. | (Me)2Si=O |
| 3. | 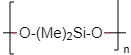 |
| 4. | Me2SiCl(OH) |
The alloy used in the construction of aircrafts is :
1. Mg-Zn
2. Mg-Al
3. Mg-Sn
4. Mg-Mn
The gas released during anaerobic degradation of vegetation may lead to:
1. Ozone hole
2. Acid rain
3. Corrosion of metals
4. Global warming and cancer
Some statements about heavy water are given below:
(i) Heavy water is used as a moderator in nuclear reactors.
(ii) Heavy water is more associated than ordinary water.
(iii) Heavy water is a more effective solvent than ordinary water.
Which of the above statements are correct?
1. (i) and (ii)
2. (i), (ii) and (iii)
3. (ii) and (iii)
4. (i) and (iii)
| 1. | Borox | 2. | Orthoboric acid |
| 3. | Boric oxide | 4. | Diborane |
Incorrect statement among the following regarding ozone is-
1. In the stratosphere, it forms a protective shield against UV radiation.
2. It is a toxic gas and its reaction with NO gives NO2.
3. In the stratosphere, CFCs release chlorine-free radicals which react with O3 to give chlorine dioxide radicals.
4. In the atmosphere, it is depleted by CFCs.
Among the following, the metal that can be used as an electrode in a photoelectric cell is-
1. Li
2. Na
3. Rb
4. Cs
The correct order of first ionization energy of group-14 elements is:
1.
2.
3.
4.
| List I | List II |
| Hydrides | Examples |
| I. Saline hydrides | A. HF |
| II. Electron-deficient molecular hydrides | B. B2H6 |
| III. Electron-rich molecular hydrides | C. CrH |
| IV. Interstitial hydrides | D. BeH2 |
| Statement I: | At atmospheric pressure, ice crystallises in the cubic form, but at very low temperatures it condenses to a hexagonal form. |
| Statement II: | Hydrogen bonding gives ice a rather open type structure with wide holes. |
The metal that gives hydrogen gas upon treatment with both acids, as well as the base, is:
1. Fe
2. Mg
3. Zn
4. Cd
A container with rigid walls is covered with a perfectly insulating material. The container is divided into two parts by a partition. One part contains a gas while the other is fully evacuated (vacuum). The partition is suddenly removed. The gas rushes to fill the entire volume and comes to an equilibrium after a little time. If the gas is not ideal, which of the following statements is correct?
| 1. | The initial internal energy of the gas equals its final internal energy. |
| 2. | The initial temperature of the gas is not equal to its final temperature. |
| 3. | The initial pressure of the gas equals its final pressure. |
| 4. | The initial entropy of the gas equals its final entropy. |
The time period of the given spring-mass system is:
| 1. | \(2\pi \sqrt{\dfrac{m}{k}}\) | 2. | \(2\pi \sqrt{\dfrac{m}{2k}}\) |
| 3. | \(2\pi \sqrt{\dfrac{2m}{\sqrt{3}k}}\) | 4. | \(\pi \sqrt{\dfrac{m}{k}}\) |
| (A) | Simple harmonic motion |
| (B) | Non-periodic motion |
| (C) | Oscillatory motion |
| (D) | Periodic motion |
| 1. | (A), (B), (C) and (D) |
| 2. | (B), (C) and (D) only |
| 3. | (C) and (D) only |
| 4. | only (D) |
| Assertion (A): | A group of soldiers is asked to break steps while crossing the bridge. |
| Reason (R): | The frequency of matching may be equal to the natural frequency of the bridge and may lead to resonance which can break the bridge. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
| 1. | \(\dfrac{M v^2}{7 R} \) | 2. | \(\dfrac{M v^2}{5 R} \) |
| 3. | \(\dfrac{2M v^2}{7 R} \) | 4. | \(\dfrac{7M v^2}{5 R} \) |
| 1. | \(1\text{%}\) | 2. | \(5\text{%}\) |
| 3. | \(10\text{%}\) | 4. | \(20\text{%}\) |
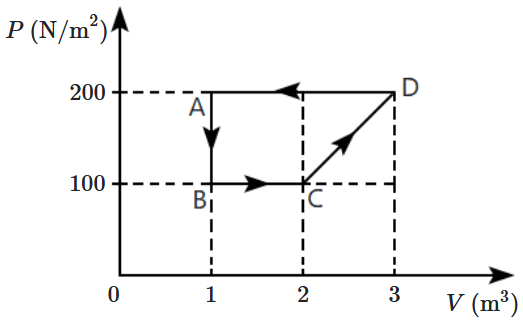
| 1. | \(RT~ \ln\left ( \dfrac{P_{1}}{P_{2}} \right ) \) |
| 2. | \(\dfrac{3}{2}RT+RT~ \ln\left ( \dfrac{P_{1}}{P_{2}} \right ) \) |
| 3. | \(\dfrac{5}{2}RT+RT~ \ln\left ( \dfrac{P_{1}}{P_{2}} \right ) \) |
| 4. | \(RT ~\ln\left ( \dfrac{V_{1}}{V_{2}} \right ) \) |
A graph shows the distribution of kinetic energy \(E_{k}\) among the constituent molecules of a gas at a given temperature. \(N\) is the number of molecules each having energy in a small energy band as shown. Choose the correct statement.
| 1. | the value of \(E_{k}\) at which the peak of the curve occurs increases when the temperature rises. |
| 2. | kinetic energy possessed by the maximum number of molecules is also the greatest kinetic energy of any of the molecules. |
| 3. | the total kinetic energy of the total gas molecules is independent of the temperature of the gas. |
| 4. | the average velocity of the molecules in the gas sample depends upon the temperature as per the graph. |
| 1. | \(\dfrac \pi 2\) | 2. | \(\pi \) |
| 3. | \(\dfrac {3\pi} 2\) | 4. | \(2\pi \) |
Which of the following can not determine the state of a thermodynamic system?
| 1. | pressure and volume |
| 2. | volume and temperature |
| 3. | temperature and pressure |
| 4. | any one of pressure, volume, or temperature |
| 1. | transverse waves cause the medium to move parallel to the direction of the wave. |
| 2. | the sound waves in the air are transverse while the light waves are longitudinal. |
| 3. | both light and sound waves in the water are transverse. |
| 4. | the sound waves in the air are longitudinal while the light waves are transverse. |
A sound wave with a frequency of \(100\) kHz is propagating through the air and encounters a water surface. The transmitted sound wave in the water will have:
| 1. | the same speed but a different frequency |
| 2. | a different speed and a different frequency |
| 3. | the same speed but a different wavelength |
| 4. | a different speed and a different wavelength |
On the basis of the kinetic theory of gases, one compares \(1~\text{gm}\) of hydrogen with \(1~\text{gm}\) of argon both at \(0^\circ \text{C}.\) Then:
| 1. | the same temperature implies that the average kinetic energy of the molecules is the same in both cases. |
| 2. | the same temperature implies that the average potential energy of the molecules is the same in both cases. |
| 3. | the internal energies in both cases are equal. |
| 4. | when both the samples are heated by \(1^\circ \text{C},\) the total energy added to both of them is the same. |
| 1. | no process is possible whose sole result is the transfer of heat from a colder object to a hotter object. |
| 2. | no process is possible whose sole result is the absorption of heat from a reservoir and the complete conversion of the heat into work. |
| 3. | the heat released to the cold reservoir can never be made zero. |
| 4. | none of the above. |
| 1. | \(500\) | 2. | \(600\) |
| 3. | \(700\) | 4. | \(800\) |
| 1. | \(4~\text m\) | 2. | \(2~\text m\) |
| 3. | \(1~\text m\) | 4. | \(0.5~\text m\) |
| Statement I: | If the acceleration of a particle is directed towards a fixed point, and proportional to the distance from that point – the motion is SHM. |
| Statement II: | During SHM, the kinetic energy of the particle oscillates at twice the frequency of the SHM. |
| 1. | Statement I is incorrect and Statement II is correct. |
| 2. | Both Statement I and Statement II are correct. |
| 3. | Both Statement I and Statement II are incorrect. |
| 4. | Statement I is correct and Statement II is incorrect. |
| 1. | \(1.5~\text{cm}\) | 2. | \(3~\text{cm}\) |
| 3. | \(6~\text{cm}\) | 4. | \(4~\text{cm}\) |
Given below are two statements:
| Statement I: | A pressure wave can be generated from a displacement wave. |
| Statement II: | Displacement and pressure waves have a phase difference of \(\dfrac{\pi }{2}.\) |
| 1. | Statement I is incorrect and Statement II is correct. |
| 2. | Both Statement I and Statement II are correct. |
| 3. | Both Statement I and Statement II are incorrect. |
| 4. | Statement I is correct and Statement II is incorrect. |
| List-I (\(x \text{-}y\) graphs) |
List-II (Situations) |
||
| (a) |  |
(i) | Total mechanical energy is conserved |
| (b) |  |
(ii) | Bob of a pendulum is oscillating under negligible air friction |
| (c) |  |
(iii) | Restoring force of a spring |
| (d) |  |
(iv) | Bob of a pendulum is oscillating along with air friction |
| (a) | (b) | (c) | (d) | |
| 1. | (iv) | (ii) | (iii) | (i) |
| 2. | (iv) | (iii) | (ii) | (i) |
| 3. | (i) | (iv) | (iii) | (ii) |
| 4. | (iii) | (ii) | (i) | (iv) |
| 1. | \(7A\) | 2. | \(A\) |
| 3. | \(3.5A\) | 4. | \(5A\) |
If a simple pendulum is brought deep inside a mine from the earth's surface, its time period of oscillation will:
| 1. | increase |
| 2. | decrease |
| 3. | remain the same |
| 4. | be any of the above, depending on the length of the pendulum |
The temperature at which the RMS speed of atoms in neon gas is equal to the RMS speed of hydrogen molecules at \(15^{\circ} \text{C}\) is:
(the atomic mass of neon \(=20.2~\text u,\) molecular mass of hydrogen \(=2~\text u\))
1. \(2.9\times10^{3}~\text K\)
2. \(2.9~\text K\)
3. \(0.15\times10^{3}~\text K\)
4. \(0.29\times10^{3}~\text K\)
The internal energy of a gas is given by \(U=2pV.\) The gas expands from \(100\) cc to \(200\) cc against a constant pressure of \(10^{5}\) Pa. The heat absorbed by the gas is:
| 1. | \(10\) J | 2. | \(20\) J |
| 3. | \(30\) J | 4. | \(40\) J |
If the vertical spring-mass system is dipped in a non-viscous liquid, then:
| 1. | Only the mean position changes. |
| 2. | Only the time period changes. |
| 3. | Both the time period and mean position change. |
| 4. | Both the time period and mean position remain the same. |
If the pressure in a closed vessel is reduced by removing some of the gas, how is the mean free path between two gas molecules affected?
| 1. | It increases. |
| 2. | It decreases. |
| 3. | It remains unchanged. |
| 4. | It increases or decreases depending on the nature of the gas. |

| 1. | maximum at \(A,\) minimum at \(O\) |
| 2. | minimum at \(A,\) maximum at \(O\) |
| 3. | uniform |
| 4. | minimum at \(A\) and \(O,\) maximum in the middle |
| 1. | \(2T\) | 2. | \(4T\) |
| 3. | \(\dfrac T 2\) | 4. | \(\dfrac T 4\) |
A spring-block system shown in the figure oscillates with a certain time period. If charge \(q\) is given to the block and a uniform field \(E\) is switched on, then its time period of oscillation is:
1. increases
2. decreases
3. may increase or decrease
4. remains the same

| 1. | \(W_1<W_2<W_3\) | 2. | \(W_2<W_1=W_3\) |
| 3. | \(W_2<W_1<W_3\) | 4. | \(W_1>W_2>W_3\) |
| 1. |  |
2. | 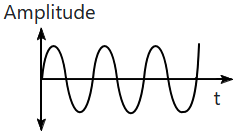 |
| 3. | 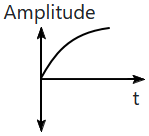 |
4. | 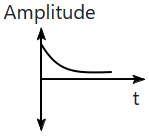 |
| 1. | \(\dfrac{\pi}{\omega}\) | 2. | \(\dfrac{2\pi}{\omega}\) |
| 3. | \(\dfrac{1}{\omega}\) | 4. | \(\dfrac{\omega}{2\pi}\) |
| 1. | \(\dfrac{3}{2} kT\) | 2. | \(\dfrac{kT}{2}\) |
| 3. | \(\dfrac{3}{4} kT\) | 4. | \(kT\) |
| Assertion (A): | The molar heat capacity of the gas can have any value from \(-\infty\) to \(\infty\). |
| Reason (R): | The molar heat capacity of the gas for the isothermal process is \(\infty\). |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |