An analysis of chromosomal DNA, using the Southern blot technique involves the following steps.
(I) Hybridisation
(II) Cleavage
(III) Autoradiography
(IV) Electrophoresis
(V) Blotting
Choose the option depicting the correct sequences of steps involved in technique.
1. II, IV, I, V, III
2. II, IV, V, I, III
3. IV, V, II, I III
4. I, IV, II, III, V
Choose the correct statements, regarding the gel electrophoresis technique.
(I) DNA migrates towards the negative electrode cathode.
(II) Smaller molecules migrate faster than larger molecules.
(III) Fragments get sorted according to their molecular weight.
(IV) Et Br can be used to visualise DNA
1. I, III and IV
2. III and IV
3. II and IV
4. I, II and III
Match the following columns I, II with III
Column I Column II Column III
A. Sickle-cell 1. Due to recessive (i) Arrangement of valine
anaemia PP genes in place of glutamic acid
B. Phenylketon 2. Due to absence (ii) Inborn error of
uria of homogenetic metabolism
oxidase enzyme
C. Alkaptonuria 3. Follows Medelian (iii) Urine turns black
Principles when exposed to air
D. Thalassaemia 4. Characters caused (iv) The required
by homozygous haemoglobin is not
recessive genes generated in the
blood
Codes
A B C D
1. 3-(iii) 1-(ii) 4-(i) 2-(iv)
2. 4-(i) 1-(ii) 2-(iii) 3-(iv)
3. 4-(i) 3-(iii) 1-(iv) 2-(iii)
4. 2-(iv) 3-(iii) 1-(ii) 4-(i)
Which one of the following combination of all three fatty acids are essential for human beings?
1. Oleic acid, linoleic acid and linolenic acid
2. Palmitic acid, linoleic acid and arachidonic acid
3. Oleic acid, linoleic acid and arachidonic acid
4. Linoleic acid, linolenic acid and arachidoonic acid
Antibodies produced by a group of identical B-cells against a single epitope of an antigen is called
1. Polyclonal antibodies
2. Monoclonal antibodies
3. Anti-hapten antibodies
4. Somaclonal antibodies
Which one of the following acts solely as an inhibitory neurotransmitter?
1. Norepinephrine
2. Gamma () aminobutyric acid
3. Acetylcholine
4. Dopamine
Which one of the following statements is incorrect?
1. Insects have one pair of antenna
2. Millipeds possess two pairs of appendages in each segment of the body
3. Prawns have two pairs of antenna
4. Animals belonging to the phylum-Porifera have nematocyst
Select the correct combination of statements for lymph
I. It helps to maintain fluid balance of the body
II. It is coontained in lymphatic vessels and lymphatic organs in mammals
III. It is derived from tissue fluid
IV. It contains less antibodies than plasma
V. Flows sin both directions
VI. It helps to conserve proteins and remove bacteria
1. I, II, III, V
2. II, III, IV, VI
3. I, IV, V, VI
4. III, IV, V, VI
Given below is a diagrammatic cross-section of a single loop of human cochlea. Which one of the following options correctly represents the name of three different parts?
1. A - Perilymph
B- Tectorial membrane
C- Endolymph
2. B-Tectorial membrane
C- Perilymph
D- Secretory cells
3. C-Endolymph
D- Sensory hair cells
A-Serum
4. D-Sensory hair cells
A- Endolymph
B-Tectorial membrane
1. B-Blind spot-has only a few rods and cones
2. C - Aqueous chamber - reflects the light which does not pass through the lens
3. D- Choroid- its anterior part forms ciliary body
4. A - Retina- contains photo receptors- rods and cones
Consider the diagram shown below
Carefully read the statements in relation to the above depicted mechanism. Mark the true and false statements
I. Reflex action requires the involvement of a part of the central nervous system
II. The afferent neuron receives signal from a sensory organ and transmits into the CNS
III. The efferent neuron carries signals from CNS to the effecctor
IV. The stimulus and response forms a reflex arc
Select the correct option
1. True True False True
2. True False True False
3. False False True True
4. False True False False
Convergent evolution is—
1. Species from different evolutionary branches may comes to resemble one anohter, if they have similar ecological roles
2. Similarity in characteristic resulting from common ancestry
3. Process of evolution of different species in a given geographical area starting from a point and
literally radiating to other areas
4. Phyletic transformation of a single species.
Which forms the largest portion of the coxal bone?
1. Ischium
2. Pubis
3. Ilium
4. Pelvic
Given a population that contains genetic variation, what is the correct sequence of the following events, under the influence of natural selection?
(1) Well adapted individuals leave more offspring than do poorly adapted individuals
(2) A change occurs in the einvironment
(3) Genetic frequencies within the population change
(4) Poorly adapted individuals have decreased survivorship
1. 4 2 3 1
2. 4 1 2 3
3. 4 2 1 3
4. 2 4 1 3
Which of the following statements about adaptive radiation is correct?
1. Adaptive radiation occurs in the presence of competition
2. Adaptive radiation occurs very slowly over time
3. Adaptive radiation occurs within a single lineage
4. Adaptive radiation occurs in species that live in one habitat
Match the following columns
|
Column I |
Column II |
||
|
A. |
These possess electric organs |
1. |
Trygon |
|
B. |
Animals of this class are poikilothermous |
2. |
Cyclostomata |
|
C. |
These possess poison sting |
3. |
Torpedo |
|
D. |
These migrate for spawning to freshwater |
4. |
Chondrichthyes |
|
5. |
Lamprey |
||
|
6. |
exocoetus |
Codes
A B C D
1. 6 1 3 2
2. 1 4 6 5
3. 3 4 1 5
4. 3 4 6 2
Which one of the following statements is incorrect?
1. A competitive inhibitor reacts reversibly with the enzyme too form an enzyme-inhibitor
2. In competitive inhibition, the inhibitor molecule is not chemically changed by the enzyme
3. The competitive inhibitor does not affect the rate of breakdown of the enzyme-substrate complex
4. The presence of the competitive inhibitor decreases the km of the enzyme for the substrate
Match the following columns
|
|
Column I |
|
Column II |
|
A. |
Hepatic lobule |
1. |
Sub-mucosal glands |
|
B. |
Brunner’s glands |
2. |
Base of villi |
|
C. |
Crypts of Leiberkuhn |
3. |
Glisson’s capsule |
|
D. |
Sphineter of Oddi |
4. |
Hepato-pancreatic duct |
Codes
A B C D
1. 3 1 2 4
2. 4 3 1 2
3. 3 1 4 2
4. 1 3 4 2
If the amount of reabsorbed solute from the nephric
filtrate is equal to the amount of solute secreted in
the nephric tubule is than the
1. No urine will be formed
2. Urine will be isotonic to blood plasma
3. Urine will be hypertonic to blood plasma
4. Urine will be hypotonic to blood plasma
The ability of a pregnant woman not to reject her “foreign” fetus may be due to
1. the fact that fetal and maternal blood never mix
2. the suppression of her immune response to the paternal chemical markers on fetal tissue
3. the protection of the fetus within the trophoblast that is made of maternal tissue.
4. the production of human chorionic gonadotropin that maintains the pregnancy.
Which of the following are incorrectly paired?
1. Variable Region – determines antibody specificity for an epitope
2. Immunioglobulins – glycoproteins that form epitopes
3. Constant Region – determines class and function of antibody
4. IgG – most abundant circulating antibodies, confer passive immunity to fetus
Which of the following represents a difference between viruses and viroids?
1. Viruses infect many types of cells, whereas viroids infect only prokaryotic cells
2. Viruses have capsids composed of protein, whereas viroids have no capsids
3. Viruses contain introns, whereas viroids have only exons
4. Viruses always have genomes composed of DNA, whereas viroids always have genomes
composed of RNA
Given its composition and location, the phragmoplast
should be directly involved in the
1. segregation of daughter chromosomes during
anaphase
2. poleward migration of centrosomes during
prophase
3. synthesis of sporopollenin during G1 and G2
phases
4. construction of the cell plate during cytokinesis
The functional role of sporopollenin is primarily to
1. comprise spore surface structures that catch the wind and assist in spore dispersal
2. reduce dehydration
3. make spores less dense and able to disperse more readily
4. repel toxic chemicals
Which of the following is true of the life cycle of mosses?
1. The haploid generation grows on the sporophyte generation
2. Spores are primarily distributed by water currents
3. Antheridia and archegonia are produced by gametophytes
4. The sporophyte generation is dominant
All of the following cellular structures are functionally important in cells of the gametophytes of both angiosperms and gymnosperms except
1. haploid nuclei
2. mitochondria
3. cell walls
4. chloroplasts
The seeds of orchids are among the smallest known,
with virtually no endosperm and with miniscule seed
leaves. Consequently, what should one expect to be
true of such seeds?
1. They require extensive periods of dormancy
during which the embryo develops
2. They are surrounded by brightly colored, sweet
fruit
3. They germinate very soon after being released
from the ovary
4. The developing embryo within is dependent upon
the gametophyte for nutrition
During glycolysis, when each molecule of glucose is =catabolized to two molecules of pyruvate, most of the potential energy contained in glucose is
1. transferred to ADP, forming ATP
2. transferred directly to ATP
3. retained in the two pyruvates
4. stored in the NADH produced
A mutation that inactivates the regulatory gene of a repressible operon in an E. coli cell would result in
1. continuous transcription of the structural gene controlled by that regulator
2. complete inhibition of transcription of the structural gene controlled by that regulator
3. irreversible binding of the repressor to the operator
4. inactivation of RNA polymerase by alteration of its active site
The most serious consequence of a decrease in global biodiversity would be the
1. increase in global warming and thinning of the ozone layer
2. potential loss of ecosystem services on which people depend
3. increase in the abundance and diversity of edgeadapted species
4. loss of source of genetic diversity to preserve endangered species
Introduced species can have deleterious effects on biological communities by
1. preying on native species
2. competing with native species for food or light
3. displacing native species
4. All of the options are correct
Which of the following is true of detrivores?
1. They recycle chemical elements directly back to primary consumers
2. They synthesize organic molecules that are used by primary producers
3. They convert organic materials from all trophic levels to inorganic compounds usable by primary
producers
4. They secrete enzymes that convert the organic molecules of detritus into CO2 and H2O
Why is net primary production (NPP) a more useful measurement to an ecosystem ecologist than gross primary production (GPP)?
1. NPP can be expressed in energy/unit of area unit of time
2. NPP can be expressed in terms of carbon fixed by photosynthesis for an entire ecosystem
3. NPP represents the stored chemical energy that is available to consumers in the ecosystem
4. NPP is the same as the standing crop
Which of the following best describes resource partitioning?
1. competitive exclusion that results in the success of the superior species
2. slight variations in niche that allow similar species to coexist
3. two species that can coevolve to share identical niches
4. differential resource utilization that results in a decrease in community species diversity
Logistic growth of a population is represented by dN/dt =
1.
2. rN
3. rN(K+N)
4.
Plants growing in a partially dark environment will grow toward light in a response called phototropism. Which of the following statements is true regarding phototropism?
1. It is caused by an electrical signal.
2. One chemical involved is ethylene.
3. Auxin causes a growth increase on one side of the stem.
4. Auxin causes a decrease in growth on the side of the stem exposed to light
Which of the following statements applies to plant growth regulators?
1. They only act by altering gene expression
2. They often have a multiplicity of effects
3. They function independently of other hormones.
4. They directly control plant protein synthesis and assembly.
Recent research has shown that pollination requires that carpels recognize pollen grains as “self or nonself.” For self-incompatibility, the system requires
1. the rejection of nonself cells
2. the rejection of self cells.
3. carpel incompatibility with the egg cells.
4. that the flowers be incomplete
Which of the following elements is incorrectly paired with its function in a plant?
1. nitrogen component of nucleic acids, proteins, hormones, coenzymes
2. magnesium component of chlorophyll; activates many enzymes
3. phosphorus component of nucleic acids, phospholipids, ATP, several coenzymes
4. sulfur component of DNA; activates some enzymes
All of the following have an effect on water potential(Ψ) in plants except
1. physical pressure
2. water-attracting matrices
3. dissolved solutes
4. DNA structure
A: Amphibians cannot survive in sea water.
R: Amphibians have lungs for breathing on land. These would collaps under the water pressure
of the sea.
A : The passage starting with the external nostrils upto the terminal bronchiole constitute the respiratory part.
R : The respiratory part transport the air to the alveoli, clears it from the foreign material, humidified and brings the air to body temperature
A: Atrial systole results in 30% passive filling of ventricles
R: During atrial systole ventricles are in diastolic pahse to take blood froom atria
A: Epinephrine, Glucoagon, and glucocorticoides are synergistic hormone for carbohydrate metabolism
R: All promote glycogenolysis, and hence increases blood glucose level
A: Rods and cones get hyperpolarised to form action potential after absorption of light energy
R: Rods and contains opsin protein, which get denatured after absorption of light
A: All the secondary follicle can not be converted into tertiary follicle during follicular phase of menstrual cycle.
R: Fertiliz in - Antifertiz in reaction is required for the formation of tertiary follicle
A: Use of oral pills are very effective contraceptive method
R: Oral pills contains, estrogen or estrogen progesterone both prevent ovolation by controlling the release of FSH and LH.
A: Not all the copulations lead to fertilization and pregnancy.
R: Fertilization can only occur if the ovum and sperms are transported simultaneously to the ampullary-isthmus junction.
A: Retroviruses are used to transform the human cell during gene therapy.
R: Retroviruses have the ability to perform process of Reverse genetics.
A: Pre-existing advantageous mutations when selected will result in observation of new phenotype.
R: In small populations natural selection has less role to play in fixing a mutated gene or a gene combination.
A: Progesterone is the most important hormone of post-ovulationary phase
R: Progesterone maintain the thickening of endometrium.
A: Both the solute and solvent can pass through a emipermeable memrane.
R: A semipermeable memberance is always living.
A: Peroxisomes participate in photorespiration in C4 plants.
R: Catalase enzyme is present in high concentration in peroxisomes
A: For evolution of O2 in photosynthesis, PS-II is required
R: PS II is reduced by pulling electrons from water due to which photolysis of H2O occurs
A: Glycolysis is the first common step of both aerobic as well as anaerobic respiration.
R: 1 molecule of hexose sugar is converted into 2 mol ecules of pyruvic acid in absence or presence of oxygen in glycolysis
A: Meiosis leads to recombinations of genes.
R: Crossing over occurs in pachytene sub-stage of prophase I of meiosis
A: Tendency of parental combinations to remain
together is called crossing over.
R: Crossing over provides a proof in favour of linear
arrangement of genes on chromosomes.
A: Organisms lacking one complete chromosome are called monosomics.
R: Down’s syndrome is an example of monosomy
A: Histones are basic proteins of major importance in packaging of eukaryotic DNA. DNA and histones comprise chromatin, forming the bulk of eukaryotic chromosome.
R: Histones are of five major types H1, H2A, H2B and H3 and H4
.
A: Gram-negative bacteria do not retain the stain when washed with alcohol.
R: The outer face of the outer membrance of Grainnegative bacteria contains lipopolysaccharides, a part of which is integrated into the membrance lipids.
If % abundance of C12 and C14 are 90% and 10% respectively then number of carbon-12 in 12 gram sample
1.
2.
3.
4.
Which of the following statements is not true?
1. Cosmic rays have highest frequency
2. Radius of nth orbit
3. Velocity of e- in orbit
4. Bohr model cannot explain spectrum of multi electron species
Which of the following will have +ve value of electron gain enthalpy?
1. N
2. Be
3. He
4. All of these
Which of the following has strongest - bonding?
1.
2.
3.
4.
Which of the following is regarded is true order about dipole moment?
1.
2.
3. (p-nitro chloro benzene) > (o-nitro chloro benzene)
4. None of these
Vapour density of an oxide is 22, the rate of diffusion of this compared to He is
1.
2.
3.
4.
The graph of compressibility factor Z vs P is given. The gas C cannot be
1.
2.
3.
4.
Which of the following is not true statement?
1. In an isolated system the value of
2.
3. For a spontaneous process entropy of system must increase
4. for spontaneous change
Solubility of AgCl is maximum in
1.
2. 0.1 M KCl
3. 0.05 M
4. 0.1 M
Which of the following description is incorrect for CsCl type crystal?
1. Coordination number 8 : 8
2. 2nd Co-ordination is 8 for Cs
3. Cs+ occupies cubical void
4. Distance between adjacent opposite ion
A+BProducts. Kinetic data are following for this process
[A] [B] Rate of reaction
mol L–1 mol L–1 mol L–1 s–1
0.01 0.01 0.001
0.02 0.01 0.002
0.04 0.02 0.006
Which of the following conclusion holds good for presented datas?
1. Order with respect to A = 1
2. Order with respect to B = 1
3. Unit of rate constant = mol–1 L–1
4. All of these
Which of the following is correct about H2?
1. Under very high pressure hydrogen is expected to behave like a metal
2. H+ is hydrated in tetrahedral manner
3. Much fraction of H2 is produced from petrochemical
4. All of these
Which of the following comparison is not proper for alkali metal and alkali earth metal?
Alkali metal Alkali earth metal
1. Solubility of Hydroxides increases downward increases downward
2. Valence orbit configuration ns1 ns2
3. Solubility of sulphate decreases downward decreases downward
4. Flame colouration All metals impart Except Be, Mg other given
Which of the following order is mismatched?
1.
2.
3.
4.
Which of the following set is mismatched according to given shape?
1. Linear
2. Bent
3. Pyramidal
4. All of these
Which of the following has zero magnetic moment?
1.
2.
3.
4. Both 2 and 3
Acidified KMnO4 does not produce following conversion
1.
2.
3.
4.
Which of the following is not true about ferrocene?
1. Coordination number of iron is 10
2. Iron III forms this complex
3. Cyclopentadienyl carbocation form this complex
4. Both 2 & 3
Which of the following is true anhydride?
1.
2.
3.
4. None of the above is true anhydride
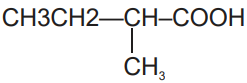
1. 2-chloro-2-methyl butanoic acid
2. 1-chloro-2-methyl butanoic acid
3. 2-chloro-2-methyl butanoyl chloride
4. Both (1) & (3)
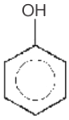
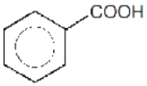
Which of the following sequence will produce meta-nitrotoluene?
1. 
2. 
3. 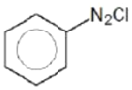
4. Both (1) and (3)
Which of the following could react with Lucas reagent in a quick manner even at room temperature?
1.
2.
3.
4. All of these
Which of the following method does not work for differentiation?
1. 1º, 2º amine (carbylamine reaction)
2. Propanol-1 and propanol-2 (iodoform reaction)
3. HCOOH and HCHO (Tollen’s reagent test)
4. Glucose and 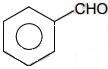
Which of the following is an unmatched fact?
1. SBR (Addition polymer)
2. PHBV (Biodegradable polymer)
3. Amylose (Linear polymer of -D glucose)
4. Alanine (Optical active -amino acid)
Product B is
1.
2.
3.
4. Both (2) and (3)
Product E is
1. 3-methyl But-2-eneal
2. Crotonaldehyde
3. Mesityl oxide
4. 3-hydroxy butanal
Which of the following result is not proper?
1.
2.
3.
4.
Which of the following comparison is incorrect?
1. 
2. 
3. 
4. Both 1 and 2
the major alkene product of this process
1.
2.
3.
4.
Which of the following is incorrect order?
1. Stability,
2. Acidity
3. Basicity in aq. solution
4. Order of reactivity for pathway
For precipitation of cations of Group IV in the salt analysis, the medium is made alkaline before passing H2S gas. The purpose of alkaline solution is
1. to suppress the ionization of H2S
2. to increase the ionization of H2S
3. to increase the ionization of metal salt
4. to decrease the ionization of metal salt
Depression is freezing point of 0.1M aqueous solutions of HBr, ZnSO4 and Na2SO4 are in the ratio,
1. 1 : 1 : 1
2. 1 : 2 : 3
3. 1 : 1 : 1.5
4. 2 : 4 : 3
Which of the following is not an organometallic compound?
1.
2.
3.
4. all are organometallic compounds
Which of the following is not optically active?
1.
2.
3.
4.
Any metal chloride on heating with K2Cr2O7 and conc H2SO4 produces orange-red vaopurs which can oxidize toluene into benzaldehyde. The vapours contain
1. CrO2Cl2
2. Cr2OCl2
3. Cr2O2Cl2
4. Cr2O2Cl
Electric current is passed in the molten mass of hydrolith (CaH2). The gas or metal deposited at the anode is
1. Ca
2. H2
3. O2
4. Both (1) & (2)
Nitrobenzene can be prepared from benzene by using mixture of concentrated HNO3 and concentrated H2SO4. In the nitrating mixture, HNO3 acts as a
1. Base
2. Acid
3. Reducing agent
4. Catalyst
Maximum bond angle is not present in
1.
2.
3.
4.
The reaction takes place in the following steps.
the rate equation of this reaction is given by
1. rate = k [NO2Cl]
2. rate = k[NO2Cl]2
3. rate = k[NO2Cl]0
4. rate = k[NO2Cl][Cl]
The value of n = 2 will be given by the slope of which line in the figure.
1. OA
2. OB
3. OC
4. OD
Assertion: Both ethyl acetoacetate and acetylacetone exhibit keto-enol tautomerism but the amount of emlic form is much higher in acetylacetone than in acetoacetic ester.
Reason: Keto group is a much better electron-withdrawing group than an ester group.
Assertion: The dipole moment of SF4 molecule is zero.
Reason: Dipole moment vectors cancel each other as shape of SF4 molecule is symmetrical.
Assertion: The first ionisation energy of Gallium is more than that of aluminium.
Reason: Gallium because of poor screening effect has smaller size.
Assertion: If is added to the solution of then pH of the solution increases.
Reason: Because of common-ion effect concentration of ion decreases and pH increases.
Assertion: The shape of trisilylamine N(SiH3)3 molecule is pyramidal.
Reason: Nitrogen contains 3-bond pairs and one lone pair.
Assertion: Alcohol can be protonated by a strong halogen acid to form an oxonium ion.
Reason: Water is a good leaving group, it can leave spontaneously when the alcohol is tertiary to
form a stable carbocation.
Assertion: Basicity of is in order.
I>II>III
Reason: Electron-donating groups (such as alkyl group) increase the basicity of amines, and
electron withdrawing groups (such as aryl group) decrease the basicity of amines.
Assertion: Benzoic acid stronger than acetic acid.
Reason: Benzoic acid has carboxyl group attached to sp2- hybridised carbon while acetic acid has carboxyl group attached to sp3-hybridised carbon.
Assertion: Major elimination product of the following is
Reason: Due to elimination, conjugation increases hence, stability is increased.
Assertion: Covalent linkage that maintains a higher order protein structure, is the disulphide bond.
Reason: These interaction are week and easily disrupted by thermal motion.
Assertion: The relative stabilities of cycloalkanes can be determined by comparing the heat of combustion.
Reason: Cycloalkanes have different amount of ring strain.
Assertion: The relative lowering of vapour pressure of solute in a solution containing a nonvolatile solute is directly proportional to the mole fraction of solvent in the solution.
Reason: The vapour pressure of a solution containing a nonvolatile solute is only due to solvent molecules.
Assertion: Graphite is thermodynamically less stable than diamond.
Reason: The free energy of formation of graphite is zero while that of diamond has a negative value.
Assertion: Disubstituted cycloalkanes can display geometric isomerism.
Reason: The geometric isomerism is due to rigid structure of cycloalkanes.
Assertion: Brown ring test of cannot be performed satisfactorily if the solution also contains Br–/I– ions.
Reason: Halogen (Br2 or I2) is released from halides Br– or I– when treated with concentrated H2SO4.
Assertion: The addition of an inert gas at constant volume does not affect the equilibrium.
Reason: Addition of NH3 at constant volume increases the pressure of the gaseous mixture of a reaction at equilibrium.
Assertion: The value of a real gas is independent of its nature.
Reason: As , a real gas behave as if there exists no molecular attractions and the volume occupied by gaseous molecules is negligible in comparison to its volume.
Assertion: In photoelectric effect, the number of electrons emitted is proportional to the frequency of incident radiation.
Reason: Largest the frequency of incident photon, larger the energy of photon.
Assertion: and orbitals in free ion have the same energy but in , the energy of is larger than orbital.
Reason: The incoming ligands in octahedral crystal field cause more electron-electron repulsion in as compared to orbital.
Assertion: An adiabatic expansion of a gas in accompanied with decrease in its internal energy.
Reason: The decreases of energy is due to the transfer of heat from the system to its surroundings.
Work done in carrying a charge ‘Q’ from ‘A’ to ‘B’ (as shown in figure) on a circle of radius ‘r’ with a charge ‘Q’ at the centre is
1.
2.
3. zero
4.
Two protons are placed 1 apart. If they are released, what will be the kinetic energy of each proton when they are at large separation?
1. 2.56 × 10–19 joule
2. 11.52 × 10–19 joule
3. 23.04 × 10–19 joule
4. 2.56 × 10–28 joule
‘N’ batteries each of emf E and internal resistance ‘r’ are first connected in series and then in parallel to an external resistance ‘R’. If current through ‘R’ is same in both cases then
1.
2.
3.
4.
The potential difference between points A and B in the figure is? if and i = 3 is
1. 3 V
2. 15 V
3. 5.1 V
4. +5.1 V
Maximum number of 60 W bulbs that can be used in a 220 V supply line protected by a 10A fuse is
1. 40
2. 36
3. 10
4. 60
A dielectric material of dielectric constant ‘K’ is introduced in half the length of a parallel plate capacitor as shown in the figure. If the area of the plates of the capacitor is ‘A’ and the plate separation is ’d’. Then the capacitance of the arrangement CAB is
1.
2.
3.
4.
If the reading of ammeter ‘A2’ in the figure shown is 2A, the ammeter ‘A3’ will read
1. 1A
2. 2A
3. 3A
4. 5A
Two rectangular loops enter into uniform magnetic field ‘B’ with same velocity ‘v’ as shown in the figure. ‘VA’ and ‘VB’ are induced emf in two loops respectively, then
1.
2.
3.
4. none
A proton projected with some velocity in a magnetic field experiences a magnetic force , then 'x' is equal to
1. 1
2. 4
3. 2
4. 3
If \(M\) be the mass of the charged particle having a charge \(q_0\) which enters with a velocity \(v\) normal to the magnetic field \(B,\) it will revolve with angular speed given by:
| 1. | \( \dfrac{B}{q_0 M} \) | 2. | \( \dfrac{q_0 B}{M} \) |
| 3. | \( \dfrac{q_0 M}{B} \) | 4. | \( q_0 M B\) |
An electric bulb of (100 W, 300 V) is connected with an AC supply of 500 V and (150/)Hz. The required inductance to save the electric bulb is
1.
2.
3.
4.
Out of the two slits of a Young’s double slit experiment, one of the slit is painted to make its intensity less than that of the other slit. Then
1. the fringes will disappear (uniform illumination)
2. screen will become dark
3. contrast between fringes will increase
4. contrast between fringes will decrease
First diffraction minimum due to single slit diffraction is at ‘ = 30o’ for a light of wavelength 5000. The width of the slit is
1. 5 ×10–5 cm
2. 10×10–5 cm
3. 2.5 × 10–5 cm
4. 1.25 × 10–5 cm
A combination of two thin lenses with focal lengths f1 and f2 respectively forms an image of a distant object at distance 60cm when lenses are in contact. The position of the image shifts by 30cm towards the combination when lenses are separated by 10cm. The value of f1 and f2 will be
1. 30cm, –60cm
2. 20cm, –30cm
3. 15cm, –12cm
4. 12cm, –15cm
A telescope has an objective lens of 10cm diameter and is situated at a distance of one kilometer from two objects. The minimum distance between these two objects, which can be resolved by the telescope, when the mean wavelength of light is 5000, is of the order of
1. 6.1 m
2. 0.61 m
3. 6.1 mm
4. 6.1 cm
The magnifying power of an astronomical telescope is 8 and the distance between the two lenses is 54cm. The focal lengths of eye lens and objective lens will be respectively
1. 6cm and 48 cm
2. 48cm and 6cm
3. 8cm and 64cm
4. 64cm and 8cm
On increasing the number of electrons striking the anode of an X-ray tube, which one of the following parameters of the resulting X-rays would increase?
1. Penetration power
2. Frequency
3. Wavelength
4. Intensity
Light of two different frequencies whose photons have energies 1eV and 2.5eV respectively, successively illuminate a metal whose work function is 0.5 eV. The ratio of the maximum speeds of the emitted electrons will be
1. 1:5
2. 1:4
3. 1:2
4. 1:1
If represent the frequencies of X-ray lines of a given material, then
1.
2.
3.
4.
When n-type semiconductor is heated
1. number of electrons increases while that of holes decreases
2. number of holes increases while that of electrons decreases
3. number of electrons and holes remain same
4. number of electrons and holes increases equally
How many NAND gates are used to form AND gate?
1. 1
2. 2
3. 3
4. 4
The mobility of conduction electrons is greater than that of holes since electrons
1. are lighter
2. are negatively charged
3. require smaller energy for moving through crystal lattice
4. undergo smaller number of collisions
The velocity \(v\) of a particle at time \(t\) is given by \({v}={at}+\frac{{b}}{{t}+{c}}.\) The dimensions of \({a}\), \({b}\), and \({c}\) are respectively:
1. \( {\left[{LT}^{-2}\right],[{L}],[{T}]} \)
2. \( {\left[{L}^2\right],[{T}] \text { and }\left[{LT}^2\right]} \)
3. \( {\left[{LT}^2\right],[{LT}] \text { and }[{L}]} \)
4. \( {[{L}],[{LT}], \text { and }\left[{T}^2\right]}\)
Two resistors of resistance and connected in parallel. The equivalent resistance is
1.
2.
3.
4.
A particle is moving in a circle of radius r with constant speed v. The change in velocity in moving from P to Q is
1. 2v cos 40º
2. 2v sin 40º
3. 2v cos 20º
4. 2v sin 20º
What is the value of linear velocity, if and ?
1.
2.
3.
4.
An object of mass 3 kg is at rest. Now a force of is applied on the object then velocity of object at t=3 sec is
1.
2.
3.
4.
A particle starts from origin at t = 0 along x-axis. Th acceleration time graph is as shown. The corresponding displacement time graph is
1.
2.
3.
4.
The friction force on the block is
1.
2.
3.
4. None of these
A heavy uniform chain lies on a horizontal table top. If the coefficient of friction between the chain and the table surface is 0.25, then the maximum percentage of the length of the chain that can hang off the edge of the table is
1. 20%
2. 25%
3. 35%
4. 45%
A moving steel ball of mass m collides elastically with another steel ball of mass 2m at rest. If the kinetic energy of the moving ball before collision is 100 J then after the collision it will have a kinetic energy of
1.
2.
3.
4.
Two masses m1 and m2 are hanging on the sides of smooth & light pulley. The system is released to move. If m1 > m2 then acceleration of the centre of mass of the system constituted by m1 and m2 is
1.
2.
3.
4. zero
In case of rifle shooting, the kick will be maximum when
1. a light rifle is held loosely against shoulder
2. a light rifle is held tightly against shoulder
3. a heavy rifle is held loosely against shoulder
4. a heavy rifle is held tightly against shoulder
A bomb of mass 9 kg, initially at rest, explodes into two pieces of masses 3 kg ad 6 kg. The velocity of mass 3 kg is 16 m/s. The kinetic energy of mass 6 kg in joule is
1. 46
2. 384
3. 192
4. 768
Arod of length '2m' and mass 0.5 kg is pivoted at one end as shown in figure. The increase in potential energy when is
1. 1.25 J
2. 2.5 J
3. 5 J
4. 10 J
A bullet of mass 5g strikes a wooden plank of thickness 5 cm at 350 m/s and emerges out at 150 m/s. The average frictional force acting on the bullet when it was inside the plank is
1. 100 N
2. 200 N
3. 1000 N
4. 2000 N
Two particles are executing SHM of same amplitude and frequency along the same straight line path. They pass each other when moving in opposite directions each time their displacement is half their amplitude. The phase difference between them is
1.
2.
3.
4.
A block of wood floats in a liquid with four-fifths of its volume submerged. If the relative density of wood is 0.8, what is the density of the liquid in units of kg/m3.
1. 750
2. 1000
3. 1250
4. 1500
A boat carrying a number of large stones is floating in a water tank. What will happen to the water level if the stones are unloaded into the water? The water level
1. remains unchanged
2. falls
3. rises
4. rises till half the number of stones are unloaded and then begins to fall
Water is flowing through a tube of radius ‘r’ with a speed ‘v’. If this tube is joined to another tube of radius ‘r/2’, what is the speed of water in the second tube?
1.
2.
3.
4.
Assertion: If an electric fan be switched on in a closed room, the air of the room will be cooled.
Reason: Fan air decrease the temperature of the room.
Assertion: The molecules of 0º C ice and 0º C water will have same potential energy.
Reason: Potential energy depends only on temperature of the system.
Assertion: A block of wood floats in a bucket of water in a lift. The block sink more if the lift starts accelerating up.
Reason: When lift is accelerating upward then weight is more than force of buoyancy.
Assertion: We can distinguish between two notes of the same frequency, one produced by a violin and the other by a guitar.
Reason: The quality of a Guitar note and a Violin note, both of the same frequency is different.
Assertion: If amplitude of simple pendulum increases, its time period also increases.
Reason: The simple pendulum covers equal distance in each vibration when its amplitude is large.
| Assertion (A): | If two charged bodies placed near each other attract each other then they must be oppositely charged. |
| Reason (R): | Unlike charges repel each other and like charges attract each other. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
| Assertion (A): | While applying Gauss’s law, the axis of the Gaussian cylinder should coincide with the line. |
| Reason (R): | In the case of a Gaussian cylinder, the Gaussian surface should be drawn in such a manner that it encloses the charge whose electric field is to be found. |
In the light of the above statements choose the correct answer from the options given below:
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Assertion: The capacitance of a solid copper sphere is greater than the capacitance of a hollow copper sphere of the same radius.
Reason: The mass of a solid copper sphere is greater than the mass of a hollow copper sphere of the same radius.
A steady current Iin flows through the wire that connects to a resistor. A steady current Iout flows
through the wire that is connected to the other end. the potential at the first end is Vin and the
potential at the second end is Vout.
Assertion: Iin is greater than Iout
Reason: Vin is greater than Vout
| Assertion (A): | To measure the current in a circuit element, the ammeter should be connected in series with that element. |
| Reason (R): | Connecting the ammeter in series with a circuit element causes at least a small reduction of the current in that element. |
In light of the above statements choose the correct answer from the options given below:
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Given below are two statements:
| Assertion (A): | At most of the points in northern hemisphere, the magnetic field is inclined to an angle below horizontal, whereas in the southern hemisphere, the magnetic field is inclined to an angle above horizontal. |
| Reason (R): | The magnetic south pole is near the geographic north pole and vice versa. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
The point form of Ampere law is given by
1. Curl(B) = I
2. Curl(D) = J
3. Curl(V) = I
4. Curl(H) = J
Given below are two statements:
| Assertion (A): | The number of field lines drawn from a charge is proportional to the magnitude of the charge. |
| Reason (R): | The electric field at any point is proportional to the magnitude of the source charge. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
A series LCR circuit is operated at resonance.
Assertion: The power transferred to the resistor is maximum
Reason: The current in the circuit is maximum
Assertion: The formula connecting u, v and f for a spherical mirror is valid only for mirrors whose sizes are very small compared to their radii of curvature.
Reason: Laws of reflection are strictly valid for plane surfaces, but not for large spherical surfaces.
Assertion: The object and image both lie on the opposite side of focus for a spherical lens.
Reason: By Newton’s formula, x0xi = – f(2)
Assertion: By de Broglie hypothesis momentum of an electron, p = h/
Reason: Energy of an electron is given by E = hc/
Assertion: Balmer’s series is not observed in absorption experiments done on earth, but is observed in sunlight coming to earth.
Reason: The temperature on the earth surface is much lesser than on the sun.
1. Both assertion and reason are true and reason is the correct explanation of assertion
2. Both assertion and reason are true but reason is not the correct explanation of assertion
3. Assertion is true but reason is false
4. Both assertion and reason are false
Assertion: A beam of visible light and a beam of monochromatic x-ray have the same wavelength in vacuum.
Reason: A beam of visible light and a beam of monochromatic x-ray have the same velocity in vacuum.
Assertion: If there is no net charge inside a Gaussian surface, the net flux through the Gaussian surface is zero.
Reason: If there is no net charge inside a Gaussian surface, the electric field at every point of the Gaussian surface is zero.
A clock is set right at 8 a.m. The clock gains 10 minutes in 24 hours will be the true time when the clock indicates 1 p.m. on the following day?
1. 48 min. past 12
2. 46 min. past 12
3. 45 min. past 12
4. 47 min. past 12
If 20% of a=b, then b% of 20 is the same as:
1. 4% of a
2. 6% of a
3. 8% of a
4. 10% of a
Fresh fruit contains 68% water and dry fruit contains
20% water. How much dry fruit can be obtained
from 100 kg of fresh fruits ?
1. 20
2. 30
3. 40
4. 50
The last day of a century cannot be
1. Monday
2. Wednesday
3. Tuesday
4. Friday
A is B’s sister. C is B’s mother. D is C’s father. E is
D’s mother. Then, how is A related to D?
1. Grandfather
2. Grandmother
3. Daughter
4. Granddaughter
. A milkman purchases the milk at Rs. x per litre and
sells it at Rs. 2x per litre still he mixes 2 litres water
with every 6 litres of pure milk. What is the profit
percentage?
1. 116%
2. 166.66%
3. 60%
4. 100%
Find the number of triangles in the given figure
1. 28
2. 32
3. 36
4. 40
In an examination, a student scores 4 marks for every correct answer and loses 1 mark for every wrong answer. If he attempts all 60 questions and secures 130 marks, the no of questions he attempts correctly is :
1. 35
2. 38
3. 40
4. 42
What was the day of the week on, 16th July, 1776?
1. Tuesday
2. Wednesday
3. Monday
4. Saturday
How many such pairs of digits are there in the number 421579368 each of which has as many digits between them in the number as when they are arranged in ascending order?
1. None
2. One
3. Two
4. Three
Test-2Grand Central Terminal, Park Avenue, New York is the world’s
1. largest railway station
2. highest railway station
3. longest railway station
4. None of the above
Eritrea, which became the 182nd member of the UN in 1993, is in the continent of
1. Asia
2. Africa
3. Europe
4. Australia
Garampani sanctuary is located at
1. Junagarh, Gujarat
2. Diphu, Assam
3. Kohima, Nagaland
4. Gangtok, Sikkim
For which of the following disciplines is Nobel Prize awarded?
1. Physics and Chemistry
2. Physiology or Medicine
3. Literature, Peace and Economics
4. All of the above
Hitler party which came into power in 1933 is known as
1. Labour Party
2. Nazi Party
3. Ku-Klux-Klan
4. Democratic Party
FFC stands for
1. Foreign Finance Corporation
2. Film Finance Corporation
3. Federation of Football Council
4. None of the above
First human heart transplant operation conducted by Dr. Christiaan Barnard on Louis Washkansky, was conducted in
1. 1967
2. 1968
3. 1958
4. 1922
Galileo was an Italian astronomer who
1. developed the telescope
2. discovered four satellites of Jupiter
3. discovered that the movement of pendulum produces a regular time measurement
4. All of the above
Golf player Vijay Singh belongs to which country?
1. USA
2. Fiji
3. India
4. UK
Famous sculptures depicting art of love built some
time in 950 AD – 1050 AD are
1. Khajuraho temples
2. Jama Masjid
3. Sun temple
4. Mahabalipuram temples