Corpus luteum contains cells of
(i) Theca externa
(ii) Theca interna
(iii) Follicular cells
(iv) Secondary oocyte.
1. (i), (ii), (iii) (iv)
2. (ii), (iii) only
3. (i), (ii) only
4. (iii) only
Mark the incorrect match
Structure Location
1. Tertiary follicle – Ovarian medulla
2. Secondary oocyte – Ampulla of fallopian tube
3. Primary follicle – Ovarian cortex
4. Morulla – Between isthmus and Ampulla
Surrogate mother headed during
1. ZIFT
2. IUT
3. GIFT
4. both (1) and (2)
The organs which differentiate from ectoderm are –
1. Primordial germ cells, urinary bladder, tongue
2. Spleen, cortical cells of adrenal gland, coelomic epithelium.
3. Lens, cornea and lateral line sense organ
4. Nasal epithelia, coelomic epithelium and lungs.
Dieter’s cells occar in
1. Retina of eyes
2. Organ of Corti.
3. Schneiderian membrane
4. Skin
Match the following
(i) Carboniferous (p) origin of reptiles
(ii) Triassic (q) origin of egg laying mammals
(iii) Jurassic (r) Rise of toothed birds
(iv) Permian (s) Rise of modern insects
1. (i – q); (ii – p); (iii – r); (iv – s)
2. (i – p); (ii – e); (iii – q); (iv – s)
3. (i – p); (ii – q); (iii – r); (iv – s)
4. (i – s); (ii – r); (iii – q); (iv – p)
Which of the following statements are incorrect related to Retina?
(i) Rods are more sensitive to red than blue light
(ii) Rods are more numerous in the central than in peripheral retina
(iii) Rods and cones are present in the innermost layer of the retina next to vitreous.
(iv) Amacrine cells synapses with each rods and cones.
1. (i), (ii), (iii), (iv)
2. (iii), (iv)
3. (i), (ii) only
4. (iv) only
During sleep
1. Body temperature Increases
2. Melatonin secretion decreases
3. Growth hormone secretion increases
4. cortisol secretion increases.
Which of the following is not an effect of parasympathetic stimulation?
1. Salivation
2. Gastric secretion
3. Pancreatic secretion
4. Contraction of sphincter of Oddi
Haldane effect explain –
1. Decrease in affinity of haemoglobin for oxygen due to presence of 2, 3 DGP
2. Increased ability of oxy-haemoglobin to take up more CO2 as bicarbonate
3. Increased ability of reduced haemoglobin to take up more CO2 as bicarbonate and cabamino form.
4. Dissociation of O2 from oxy-haemoglobin
Proximal tubular function includes all of the following except?
1. Reabsorption of sodium and water
2. Reabsorption of
3. Reabsorption of urea
4. Reabsorption of ammonia
After several days of starvation plasma is likely to have high concentration of
1. Glucose
2. Lactic acid
3. Ketone bodies
4. Insulin
Oxidation of 100 gm of which of the following produces maximum energy?
1. Glucose
2. Sucrose
3. Palmitic acid
4. Glycogen
Which of the following statement is incorrect regarding frog?
1. Digestive system without salivary gland
2. Respiratory system without intercostals muscles
3. Circulatory system without SA node.
4. Reproductive system without copulatory organs.
Which of the following statement is true about genetic drift?
1. If operates in large populations
2. If always eliminates favourable allele from a population.
3. If work in opposite to natural selection
4. If can not fix non-adaptive trait in a population
Which of the following character is not applicable for reptiles?
1. Mostly terrestrial
2. Body is covered with dry and cornfield skin
3. Paired limbs are present
4. Absence of tympanum.
Some characteristic of animals are given below.
i. Notochord is present in the embryonic as well as adult form
ii. Animals have closed or open circulatory system
iii. Animals show bilateral symmetry
iv. Animals are always triplobastic and coelomates
The characteristic that apply to chordates are
1. (i), (ii), (iii) and (iv)
2. (i), (ii) and (iii)
3. (ii), (iii) and (iv)
4. Only (iii) and (iv)
Which of the following is true about Stanley cohen and Herbet Boyer experiment ?
1. They used plasmid of salmonella typhimurium as a vector
2. They used plasmid of Escherichia coli to obtained antibiotic resistance gene
3. They used molecular scissor’s to obtained antibiotic resistance gene from the plasmid of salmonella typhimurium
4. They used DNA ligase to join antibiotic resistance gene obtained from Escherichia coli to the plasmid of salmonella
Which of the following would occur if an impulse from the SA node were blocked before it reaches the AV node?
1. The heart would not contract
2. Only the atria would contract
3. Only the ventricle would contract
4. Blood would travel only to the pulmonary system.
Match the following?
|
(i) |
Epilepsy |
(p) |
degeneration of neurons in the cerebral cortex |
|
(ii) |
Alzheimer’ disease |
(q) |
Irregular electric discharge in the neuron’s |
|
(iii) |
Parkinson’s disease |
(r) |
Decreased production of acetylcholine |
|
(iv) |
Huntington’s disease |
(s) |
Regeneration of dopamine releasing neurons. |
|
|
|
(t) |
Formation of blood clots in the brain. |
clots in the brain.
1. (i – t); (ii – s); (iii – r); (iv – p)
2. (i – q); (ii – r); (iii – p); (iv – s)
3. (i – q); (ii – r); (iii – s); (iv – p)
4. (i – q); (ii – s); (iii – r); (iv – p).
Tay-Sachs disease is a human genetic abnormality that results in cells accumulating and becoming clogged with very large and complex lipids. Which cellular organelle must be involved in this condition?
1. the endoplasmic reticulum
2. the Golgi apparatus
3. the lysosome
4. mitochondria
The chemical reactions involved in respiration are virtually identical between prokaryotic and eukaryotic cells. In eukaryotic cells, ATP is synthesized primarily on the inner membrane of the mitochondria. In light of the endosymbiont theory for the evolutionary origin of mitochondria, where is most ATP synthesis likely to occur in prokaryotic cells?
1. in the cytoplasm
2. on the inner mitochondrial membrane
3. on the endoplasmic reticulum
4. on the plasma membrane
When a potassium ion (K+) moves from the soil into the vacuole of a cell on the surface of a root, it must pass through several cellular structures. Which of the following correctly describes the order in which these structures will be encountered by the ion?
1. plasma membrane primary cell wall cytoplasm vacuole
2. secondary cell wall plasma membrane primary cell wall cytoplasm vacuole
3. primary cell wall plasma membrane cytoplasm vacuole
4. primary cell wall plasma membrane lysosome cytoplasm vacuole
Which of the following is a characteristic feature of a carrier protein in a plasma membrane?
1. It is a peripheral membrane protein.
2. It exhibits a specificity for a particular type of molecule.
3. It requires the expenditure of cellular energy to function.
4. It works against diffusion.
Thomas Hunt Morgan’s choice of Drosophila melanogaster has been proven to be useful even
today. Which of the following has/have continued to make it a most useful species?
I. its four pairs of chromosomes
II. a very large number of visible as well as biochemically mutant phenotypes
III. easy and inexpensive maintenance
IV. short generation time and large number of offspring
1. II and III only
2. I, II, and III only
3. II, III, and IV only
4. I, II, III, IV
Which of the following statements is true of linkage?
1. The closer two genes are on a chromosome, the lower the probability that a crossover will occur between them.
2. The observed frequency of recombination of two genes that are far apart from each other has a maximum value of 100%.
3. All of the traits that Mendel studied–seed color, pod shape, flower color, and others–are due to genes linked on the same chromosome.
4. Linked genes are found on different chromosomes
The “universal” genetic code is now known to have exceptions. Evidence for this can be found if which of the following is true?
1. If UGA, usually a stop codon, is found to code for an amino acid such as tryptophan (usually coded for by UGG only).
2. If one stop codon, such as UGA, is found to have a different effect on translation than another stop codon, such as UAA.
3. If prokaryotic organisms are able to translate a eukaryotic mRNA and produce the same polypeptide.
4. If several codons are found to translate to the same amino acid, such as serine
Which of these paired fungal structures are structurally and functionally most alike?
1. conidia and basidiocarps
2. sporangia and hyphae
3. soredia and gills
4. haustoria and arbuscules
What characteristic of short tandem repeat DNA makes it useful for DNA fingerprinting?
1. The number of repeats varies widely from person to person or animal to animal.
2. The sequence of DNA that is repeated varies significantly from individual to individual.
3. The sequence variation is acted upon differently by natural selection in different environments.
4. Every racial and ethnic group has inherited different short tandem repeats
Mendel accounted for the observation that traits which had disappeared in the generation reappeared in the generation by proposing that
1. new mutations were frequently generated in the progeny, “reinventing” traits that had been lost in the .
2. the mechanism controlling the appearance of traits was different between the and the plants.
3. traits can be dominant or recessive, and the recessive traits were obscured by the dominant ones in the .
4. the traits were lost in the due to dominance of the parental traits.
Which statement describes the functioning of photosystem II?
1. Light energy excites electrons in the thylakoid membrane electron transport chain.
2. Photons are passed along to a reaction-center chlorophyll.
3. The P680 chlorophyll donates a pair of protons to NADP+, which is thus converted to NADPH.
4. The electron vacancies in P680+ are filled by electrons derived from water
Which of the following statements best represents the relationships between the light reactions and the Calvin cycle?
1. The light reactions provide ATP and NADPH to the Calvin cycle, and the cycle returns ADP, Pi , and NADP+ to the light reactions.
2. The light reactions provide ATP and NADPH to the carbon fixation step of the Calvin cycle, and the cycle provides water and electrons to the light reactions.
3. The light reactions supply the Calvin cycle with CO2 to produce sugars, and the Calvin cycle supplies the light reactions with sugars to produce ATP.
4. The light reactions provide the Calvin cycle with oxygen for electron flow, and the Calvin cycle provides the light reactions with water to split.
Which of the following is incorrectly paired with its structure and function?
1. sclerenchyma supporting cells with thick secondary walls
2. periderm protective coat of woody stems and roots
3. pericycle waterproof ring of cells surrounding the central stele in roots
4. mesophyll parenchyma cells functioning in photosynthesis in leaves
Compared to a cell with few aquaporins in its membrane, a cell containing many aquaporins will
1. have a faster rate of osmosis.
2. have a lower water potential.
3. have a higher water potential.
4. have a faster rate of active transport
Which of the following is a correct sequence of processes that takes place when a flowering plant reproduces?
1. meiosis fertilization ovulation germination
2. fertilization meiosis nuclear fusion formation of embryo and endosperm
3. meiosis pollination nuclear fusion formation of embryo and endosperm
4. growth of pollen tube pollination germination fertilization
Exponential growth of a population is represented by
dN/dt =
1.
2. rN
3. rN(K+N)
4.
Which of the following groups is most threatened by global extinctions?
1. mammals
2. birds
3. fish
4. amphibians
The oxygen consumed during cellular respiration is involved directly in which process or event?
1. glycolysis
2. accepting electrons at the end of the electron transport chain
3. the citric acid cycle
4. the oxidation of pyruvate to acetyl CoA
The generative cell of male angiosperm gametophytes is haploid. This cell divides to produce two haploid sperm cells. What type of cell division does the generative cell undergo to produce these sperm cells?
1. binary fission
2. mitosis
3. meiosis
4. mitosis without subsequent cytokinesis
Match the following and choose correct option
Group-A Group-B
A. Aleurone layer i. without fertilization
B. Parthenocarpic fruit ii. Nutrition
C. Ovule iii. Double fertilization
D. Endosperm iv. Seed
Option
1. A-1,B-ii, C-iii, D-iv
2. A-ii, B-I, C-iv, D-iii
3. A-iv, B-ii, C-I, D-iii
4. A-ii, B-iv, C-I, D-iii
A: Second infection of the same pathogen is quickly eliminated.
R: Killer T cells becomes hyperactivated, and destroy extracellular pathogens
A: During acromegay, hands and feet becomes abnormally enlarged.
R: Due to increased GH secretion long and short bones of the body get elongated
A. Reptiles are similar to Aves in the term of development
R. Both are strictly oviparous and lay cleiodic eggs.
A: In nearly all animal tissues specialized junction provide both structural and functional links between its individual cells.
R: Cell junctions always help to stop substances from leaking across a tissue
A: Trypsin inhibitor protects the pancreas from autodigestion
R: Trypsin inhibitor prevents the subsequent activation of other pancreatic proteolytic enzymes
A: Lipids are essentially ester of fatty acids and glycerol.
R: Lipids are always either in the form of monogycerides, diglynerides, and triglycerides.
A: At birth a female ovary consist of large number of ovarian follicles with diploid oocytes.
R: During fetal development primary oocytes enter a prolonged diplotene stage of the first meiotic division and remain in this state until ovulation occurs at puberty.
A: Beer and alcohol are diuretic substances
R: They inhibit the release of ADH from the posterior lobe of pituitary
A: In ECG by counting the number of QRS complexes, that occur in a given time period, one can determine the heart beat rate of an individual.
R: The QRS complex represents the depolarization of the ventricles which initiate the ventricular contraction.
A: Even the cartilaginous ring is absent in respiratory bronchiole, it does not collapse during rapid
expiration.
R: Respiratory bronchiole is lined with few surfactant releasing cells
A: In a food chain members of successive higher levels are fewer in number.
R: Number of organisms at any trophic level depends upon the availability of organisms which serve as food at the lower level.
A: Long distance flow of photo assimilaties in plants occurs through sieve tubes
R: Mature sieve tubes have parietal cytoplasm and perforated sieve plates.
A: Water and mineral uptake by root hairs from the soil occurs through apoplast until it reaches endodermis.
R: Casparian strips in endodermis are suberized.
A: In plant tissue culture, somatic embryos can be induced from any plant cell.
R: Any viable plant cell can differentiate into somatic embryos.
A: The megaspore mother cell divide mitotically to produce four spores.
R: Megaspore mother cells are diploid and megaspore is haploid
A: Six molecules of CO2 and twelve molecules of NADPH + H+ and 18 ATP are used to form one hexose molecules.
R: Light reaction results in formation of ATP and NADPH2
A: Glycolysis occurs in cytoplasm.
R: Enzymes of glycolysis are found in cytoplasm. It is common in aerobic/anaerobic respiration.
A: Mosses are evolved from algae.
R: Protonema of mosses is similar to some green algae
A: TMV is a virus which causes mosaic disease.
R: TMV has RNA as genetic material.
A: Plasmids are extrachromosomal DNA.
R: Plasmids are found in bacteria and are useful in genetic engineering.
Which of the statement is incorrect for the following compound?
1. It contains four lone pair of electrons
2. It contains 19 sigma and 3 pi bonds
3. It contains sp3, sp2 and sp hybrid carbon atom
4. It contains only sp3, and sp2 hybrid carbon atom.
How many optical isomers are possible in this compound?
1. 3
2. 2
3. 4
4. zero
The correct order of basicity of the following compound is
(i)
(ii)
(iii)
(iv)
1. ii > i > iii > iv
2. iv > ii > iii > i
3. iii > ii > i > iv
4. i > iii > ii > iv
Ease of -dehydrobromination among these substrates under the treatment of strong base will be in the order as
1. I > II > III
2. III > II > I
3. II > I > III
4. II > III > I
In the following compounds, the order of basicity is:
(i)
(ii)
(iii)
(iv)
1. (i) > (iv) > (ii) > (iii)
2. (ii) > (i) > (iv) > (iii)
3. (iii) > (i) > (iv) > (ii)
4. (iv) > (i) > (iii) > (ii)
The compound B is
1.
2.
3.
4.
The addition of Br2 to Z-2 butene gives:
1. (R, R)-2, 3-dibromobutane only
2. (S, S)-2, 3-dibromobutane only
3. (R, S)-2, 3-dibromobutane only
4. a mixture of (R, R) and (S, S)-2, 3-dibromobutanes
In the following sequence of reactions, the compound ‘B’ is the compound (B) is
1.
2.
3.
4.
Phenol, when treated with bromine water gives a white precipitate due to the formation of
1. o-and p-bromophenol
2. 2, 4, 6-tribromophenol
3. 3, 5-dibromophenol
4. meta-bromophenol
When nitrobenzene is treated with bromine in presence of FeBr3 the major product formed is mononitrobenzene. The statement which is related to obtain m-isomer is
1. the intermediate carbonium ion formed after initial attack of Br+ at the meta position is least stabilised
2. The electron density on meta carbon is more than that on ortho and para positions
3. loss of aromaticity when Br+ attacks at the ortho and para positions and not at meta position
4. none of the three
Which one of the following pairs gives effervescence with aq. ?
(i)
(ii)
(iii)
(iv)
1. (i) and (iii)
2. (i) and (iv)
3. (ii) and (iii)
4. (i) and (ii)
Which of the following hexoses will form the same osazone when treated with excess phenyl hydrazine?
1. D-glucose, D-galactose and D-talose
2. D-fructose, D-mannose and D-galactose
3. D-glucose, D-fructose and D-galactose
4. D-glucose, D-fructose and D-mannose
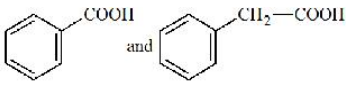
give positive test with
(i) CH3OH
(ii) Na Metal
(iii) NaOH
(iv) Sodalime decarboxylation followed by oxidation with chromyl chloride vapours followed by
treatment with Tollen’s reagent.
1. (i), (ii)
2. (ii), (iii)
3. (i), (ii), (iii)
4. (i), (iii), (iv)
The pH of 0.1 M solution of the following salts increases in the order
1. NaCl < NH4Cl < NaCN < HCl
2. HCl < NH4Cl < NaCl < NaCN
3. NaCN < NH4Cl < NaCl < HCl
4. HCl < NaCl < NaCN < NH4Cl
Equivalent weight of H3PO4 may be (M Molecular weight of H3PO4)
1. M/3
2. M/2
3. M/1
4. Any one of the above is possible
Metallic gold crystallises in FCC lattice with edge length 4.070 . Closest distance between gold atom is :-
1. 2.035
2. 8.140
3. 2.878
4. 1.357
and are example of :-
1. law of conservation of mass
2. law of definite proportion
3. law of multiple proportion
4. law of gaseous volumes
At for the dissociation reaction
atm. If the total pressure is 1 atm. The degree of dissociation of is:-
1. 87%
2. 13%
3. 17%
4. 29%
A precipitate of AgCl is formed when equal volumes of the following are mixed
1.
2.
3.
4.
45g ethylene glycol (C2H6O2) is mixed with 600g of water. The freezing point of solution is (Kf for water 1.86 K kg mol–1 and freezing pt of pure water is 273.15 K):-
1. 2.25 K
2. 270.9 K
3. 271.9 K
4. 270 K
Considering entropy(S) as a thermodynamic parameter for the criterion of the spontaneity of any process then
1.
2.
3.
4.
Partial pressure of A, B, C and D is gaseous system in equilibrium are A = 0·20, B = 0·10, C = 0·30 and D = 0·50 atm. The numerical value of equilibrium constant is :-
1. 11·25
2. 18·75
3. 5
4. 3·75
Calculate percentage by mass of solute in a solution obtained by mixing 300g of a 25% and 400g of a 40% same solution by mass of :-
1. 33.6%
2. 65%
3. 30%
4. 60%
In a process, 701 J of heat is absorbed by a system and 394 J of work is done by the system. What is the change in internal energy for the process :-
1. 307J
2. 1095J
3. –307J
4. –1095J
Lewis base is:-
1.
2.
3.
4.
Two liquids X and Y form an ideal solution. The mixture has a vapour pressure of 400 mm at 300K when mixed in the molar ratio of 1 : 1 and a vapour pressure of 350 mm when mixed in the molar ratio of 1 : 2 at the same temperature. The vapour pressure of two pure liquids X and Y respectively are :-
1. 250mm, 550mm
2. 350mm, 450mm
3. 550mm, 250mm
4. 350mm, 700mm
The enthalpy of vaporisation of a liquid is 30 kJ mol–1 and entropy of vaporisation is 75 J mol–1 k-1. The boiling point of the liquid at 1 atm is -
1. 250k
2. 400k
3. 450k
4. 600k
For the phase change
1.
2.
3.
4.
A white powder solid A forms a light green solution with water, which on treatment with potassium hexacyanoferrate (III) gives a blue precipitate. On being strongly heated, A leaves a brown residue and forms a mixture of two gaseous oxides, which turns a dichromate solution green and forms a white precipitate with a BaCl2 solution containing concentrated HCl. A is:
1. CuSO4
2. Fe2(SO4)3
3. FeSO4
4. Cr2(SO4)3
The incorrect order of given properties is :-
1. Vander waal radius - Ne < F < O
2. Electron gain enthalpy - O < Se < S
3. Electronegativity – Cl–< Cl < Cl+
4. Ionisation potential - P < Si < S
Which of the following compound is/are partially soluble or insoluble in NH4OH solution:
(i) Fe(OH)3
(ii) Ag2CrO4
(iii) Al(OH)3
(iv) Ag2CO3
(v) Ni(OH)2
1. i, iii, v
2. ii, iii, iv
3. i, iii
4. ii, iii, v
First ionisation enthalpy of oxygen is less than of nitrogen because of :-
1. More shielding of paired electron is 2p orbital of oxygen.
2. Half filled stable configuration of nitrogen.
3. More Z effective of oxygen.
4. Small atomic size of oxygen.
In which of the following process hybridization of central atoms of molecule does not change?
1.
2.
3.
4.
Which of the following species having square planer geometry with lone pair of electrons in the valence shell of central atom :-
1.
2.
3.
4.
Correct order of dipole moment is :-
1.
2.
3. Trans-1-fluoro propene < Cis-1-fluoro propene
4.
During the extraction of copper from chalcopyrites, iron is removed as
1.
2.
3.
4.
The complex used as anticancer agent is
1.
2.
3.
4.
Which of the following statement is false :-
1. Fullerene contain all the rings having six carbons
2. Graphite is thermodynamically the most stable allotrope of carbon
3. Melting point of diamond is higher than that of graphite
4. Graphite is slippery and hard therefore used as a dry lubricant in machines
KMnO4 is strong oxidising agent in acidic medium. To provide an acid medium H2SO4 is used instead of HCl.
This is because :-
1. H2SO4 is stronger acid than HCl
2. HCl is oxidised by KMnO4 to Cl2
3. H2SO4 is dibasic acid
4. rate is faster in presence of H2SO4
Which of the following compound is coloured due to charge transfer spectrum:-
1.
2.
3.
4.
Assertion: Enol form of cyclohexane –1, 3, 5 trione is more stable than its keto form.
Reason: Enol form contains –hydrogen atoms.
Assertion: Molecules that are not superimposable on their mirror images are chiral.
Reason: All chiral molecules have chiral centre.
Assertion: PH3 is stronger nucleophile than NH3.
Reason: PH3 is stronger base than NH3.
Assertion: is more stable than
Reason: In due to smaller size of F– atom P–P overlapping shifts lone towards Gatom and CF3
group is highly electron withdrawing also.
Assertion: Addition of HBr to 1-butene gives two optical isomers.
Reason: The product contains one chiral carbon atom.
Assertion: Addition of bromine to trans-2-butene yields meso-2, 3-dibromo butane.
Reason: Bromine addition to an alkene is an electrophilic addition.
Assertion: Bromobenzene upon reaction with Br2/Fe givers 1, 4-dibromobenzene as the major product.
Reason: In bromobenzene, the inductive effect of the bromo group is more dominant than the
mesomeric effect in directing the incoming electrophile.
Assertion: In strongly acidic solutions, aniline becomes more reactive towards electrophilic regents.
Reason: The amino group being completely protonated in strongly acidic solution, the lone pair of electrons on the nitrogen is not longer available for resonance.
Assertion: A : D-glucose and D-Mannose are C-2 epimers.
Reason: They only have different configuration at carbon number-2.
Assertion: Azeotropic mixtures are formed only by non-ideal solutions and they may have boiling points either greater than both the components or less than both the components.
Reason: The composition of the vapour phase is same as that of the liquid phase of an azeotropic mixutre.
Assertion: The presence of a large number of Schottky defects in NaCl lowers its density.
Reason: In NaCl, there are approximately 106 Schottky pairs per cm3 at room temperature.
Assertion: Greater the value of Vander Waal’s constant 'a ' greater is the liquification of gas.
Reason: 'a ' indirectly measures the magnitude of attractive forces between the molecules.
Assertion: There is no reaction known for which G is positive, yet it is spontaneous.
Reason: For photochemical reactions G is negative.
Assertion: Equivalent conductance of all electrolytes decreases with the increases in concentration.
Reason: Lesser number of ions are available per gram equivalent at higher concentration.
Assertion: The micelle formed by sodium stearate in water has groups at the surface.
Reason: Surface tension of water is reduced by the addition of stearate.
Assertion: Amongst the halogens fluorine can oxidise the elements to highest oxidation state.
Reason: Due to small size of fluoride ion, it is difficult to oxidise fluoride ion to fluorine. Hence
reverse reaction takes place more easily.
Assertion: The degree of complex formation in actinides decreases in the order
Reason: Actinides form complexes with -bonding ligands such as alkyl phosphines and thioethers.
Assertion: is coloured while is colourless.
Reason: d-d transition is not possible in .
Assertion: will not give any gas when treated with conc. .
Reason: can only be detected in dil. acid test.
Assertion: Gold is recovered from its solution containing aurocyanide complex by adding zinc dust.
Reason: Zinc is more electropositive than gold.
The dimension of Planck constant equals to that of
1. energy
2. momentum
3. angular momentum
4. power
The presence of gravitational field is required for the heat transfer by
1. radiation
2. conduction
3. stirring of liquids
4. natural convection
In a forward biased p-n junction diode, the potential barrier in the depletion region is of the form
1.
2.
3.
4.
Which of the following is not exhibited by X-rays?
1. Photoelectric effect
2. Polarisation
3. Interference
4. Deflection in electric field
In a given process on an ideal gas, dW = 0 and dQ < 0 . Then for the gas
1. the temperature will decrease
2. the volume will increase
3. the pressure will remain constant
4. the temperature will increase
Among which the magnetic susceptibility does not depend on the temperature?
1. dia-magnetism
2. para-magnetism
3. ferro-magnetism
4. ferrite
Potentiometer measures potential more accurately because
1. it measures potential in the closed circuit
2. it measures potential in the open circuit
3. it uses sensitive galvanometer for null detection
4. it uses high resistance potentiometer wire
Tropospheric scatter happens in
1. VHF
2. UHF
3. HF
4. SHF
The frequency used in TV and rader system lies in
1. VHF
2. UHF
3. EHF
4. VLF
The output of OR gate is 1
1. if either or both inputs are 1
2. only if both inputs are 1 or zero
3. if either input is zero
4. if both inputs are zero
The displacement of a particle is represented by the following equation where s is in metre and t in second. The acceleration of the particle at t = 1s is
1. zero
2.
3.
4.
The electrical circuit used to get smooth dc output from a rectifier circuit is called
1. logic gates
2. amplifier
3. filter
4. oscillator
The velocity of projection of an oblique projectile is . The speed of the projectile at the highest point of the trajectory is
1.
2.
3.
4. zero
A full wave rectifier circuit along with the input and output voltages is shown in the figure. The contribution to output voltage from diode-2 is
1. A, C
2. B, D
3. B, C
4. A, D
A particle is thrown vertically upward. Its velocity at half of the height is 10 m/s, then the maximum height attained by it (g=10 m/s2)
1. 8 m
2. 20 m
3. 10 m
4. 16 m
A ring of mass 10 kg and diameter 0.4 m is rotated about its axis. If it makes 2100 revolutions per minute, then its angular momentum will be
1.
2.
3.
4.
A solid cylinder and a hollow cylinder both of same mass and same external diameter are released from the same height at the same time on an inclined plane. Both roll down without slipping. Which one will reach the bottom first?
1. Hollow cylinder
2. Both together
3. Solid cylinder
4. One with higher density
A body weighs 72N on the surface of the earth. What is the gravitational force on it due to earth at a height equal to half the radius of the earth from the surface?
1. 72 N
2. 32 N
3. 28 N
4. 16 N
A reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by 62°C, the efficiency of the engine is doubled. The temperatures of the source and sink are
1. 90°C, 37°C
2. 99°C, 37°C
3. 80°C, 37°C
4. 95°C, 37°C
What causes the depletion layer across the p-n junction?
1. Drift of holes
2. Drift of electrons
3. Diffusion of holes and electrons
4. Diffusion of neither holes nor electrons
A cylinder rolls up an inclined plane, reaches some height, and then rolls down (without slipping throughout these motions). The directions of the frictional force acting on the cylinder are
1. up the incline while ascending and down the incline while descending
2. up the incline while ascending as well as descending
3. down the incline while ascending and up the incline while descending
4. down the incline while ascending as well as descending
-particles, -particles and -rays each having an energy of 10 MeV. The increasing order of penetrating powers of the radiations is
1. , ,
2. , ,
3. , ,
4. , =
A particle executes simple harmonic motion between and . The time taken for it to go from 0 to is and to go from to A is . Then
1.
2.
3.
4.
An ideal spring with spring-constant k is hung from the ceiling and a block of mass M is attached to its lower end. The mass is released with the spring initially unstretched. Then the maximum extension in the spring is
1. 4Mg / k
2. 2Mg / k
3. Mg / k
4. Mg / 2k
The equation of a wave is represented by then the velocity of wave will be
1. 100 m/s
2. 4 m/s
3. 1000 m/s
4. 0.00 m/s
Energy per unit volume for a capacitor having area A and separation d kept at potential difference V is given by
1.
2.
3.
4.
The resistance of each arm of the Wheatstone’s bridge is 10 ohm. A resistance of 10 ohm is connected in series with galvanometer, then the equivalent resistance across the battery will be
1. 10 ohm
2. 15 ohm
3. 20 ohm
4. 40 ohm
If specific resistance of a potentiometer wire is and current flow through it is 0.1 amp, cross-sectional area of wire is then potential gradient will be
1.
2.
3.
4.
A capacitor of capacity C and reactance X, if capacitance and frequency become double, then reactance will be
1. 4X
2. X/2
3. X/4
4. 2X
A capacitor is charged to store an energy U. The charging battery is disconnected. An identical capacitor is now connected to the first capacitor in parallel. The energy in each of the capacitor is
1. U/4
2. U/2
3. 3U/2
4. U
An inductance L having a resistance R is connected to an alternating source of angular frequency . The quality factor (Q) of the inductance is
1.
2.
3.
4.
A proton moving with a velocity enters a magnetic field of 0.3 Tesla at an angle of 30 with the field. The radius of curvature of its path will be (e/m for proton C/kg)
1. 1.25 cm
2. 2 cm
3. 0.5 cm
4. 0.02
In nuclear fission 0.1% mass is converted into energy. How much electrical energy can be generated by the fission of 1kg of fuel?
1. 1 kWh
2. 107 kWh
3. 2.5 kWh
4. 2.5107 kWh
The threshold frequency for a photosensitive metal is . When photons of frequency are incident on a photosensitive plate, the cut off potential is . What will be the cut off potential, when light of frequency 5 is incident on it?
1.
2. 2
3. 4
4. 5
De-Brogile wavelength of a body of mass 1 kg moving with velocity 2000 m/s is
1.
2.
3.
4. none of these
Three rods made of the same material and having the same cross-section have been joined as shown in the figure. Each rod is of the same length. The left and right ends are kept at and respectively. The temperature of the junction of the three rods will be
1.
2.
3.
4.
If the potential difference applied across X-ray tube is V volt, then minimum wavelength of the emitted X-rays will be approximately
1.
2.
3.
4.
If M be the mass of the nucleus and m be the sum of the individual masses of the nucleons forming the nucleus, then
1. M is slightly more than
2. M is slightly less than
3. M=m
4. M may be less or more than depending upon the nature of the nucleus.
A sonometer wire resonates with a given tuning fork forming standing waves with five antinodes between the two bridges when a mass of 9 kg is suspended from the wire. When this mass is replaced by a mass M, the wire resonates with the same tuning fork forming three antinodes for the same positions of the bridges. The value of M is
1. 25 kg
2. 5 kg
3. 12.5 kg
4. 1/25 kg
The effective resistance between points P and Q of the electrical circuit shown in figure.
1. 2Rr/(R + r)
2. 8R(R + r)/(3R + r)
3. 2r + 4R
4. 5R / 2 + 2r
Assertion: solid and hollow sphere of the same diameter and same material, when heated through the same temperature, will expand by the same amount.
Reason: The change in volume is independent of the original mass but depends on the original volume.
Assertion: At a point in space, the electric field points towards north. In the region, surrounding this point the rate of change of potential will be zero along the east and west.
Reason: Electric field due to a charge is the space around the charge.
Assertion: In everyday life the Doppler’s effect is observed readily for sound waves than light waves
Reason: Velocity of light is greater than the sound.
Assertion: Bending a wire does not effect electrical resistance.
Reason: Resistance of wire is proportional to resistivity of material.
Assertion: An empty test tube dipped into water in a beaker appears silvery, when viewed from a suitable direction.
Reason: Due to refraction of light the substance in water appears silvery.
Assertion: In all conductors, for studying the thermoelectric behaviour of metals, lead is taken as a reference metal.
Reason: In lead, the Thomson effect is negative.
Assertion: On reducing the volume of a gas at constant temperature, the pressure of the gas increases.
Reason: At constant temperature according to Boyle’s law, volume is inversely proportional to pressure
Assertion: For cooking in a microwave oven, food is always kept in metal containers
Reason: The energy of microwave is easily transferred to the food in metal container
Assertion: Two unequal springs of same material are loaded with same load. The longer one will have larger value of time period.
Reason: The concept will follow, if we made a experiment to measure.
Assertion: Within a glass slab, a double convex air bubble is formed. This air bubble behaves like a converging lens
Reason: Refractive index of air is more than the refractive index of glass.
Assertion: In a stationary wave, there is no transfer to energy.
Reason: There is no outward motion of the disturbance from one particle to adjoining particle in a stationary wave.
Assertion: Microwave communication is preferred over optical communication.
Reason: Microwaves provide large number of channels and bandwidths compared to optical signals.
Assertion: The graph of velocity versus displacement for a particle executing SHM is an ellipse.
Reason: The velocity and displacement are related as
Assertion: For the scattering of -particles at large angles, only the nucleus of the atom is responsible.
Reason: Nucleus is very heavy in comparison to electrons.
1. Both assertion and reason are true and reason is the correct explanation of assertion
2. Both assertion and reason are true but reason is not the correct explanation of assertion
3. Assertion is true but reason is false
4. Both assertion and reason are false
Assertion: The poles of magnet cannot be separated by breaking into two pieces.
Reason: The magnetic moment will be reduced to half when a magnet is broken into two equal pieces.
Assertion: For an electric lamp connected in series with a variable capacitor and ac source, its brightness increases with increase in capacitance.
Reason: Capacitive reactance decreases with increase in capacitance of capacitor.
Assertion: An aircraft flies along the meridian, the potential at the ends of its wings will be the same.
Reason: Whenever there is change in the magnetic flux e.m.f. induces.
Assertion: Light is produced in gases in the process of electric discharge through them at high pressure.
Reason: At high pressure electrons of gaseous atoms collide and reach and excited state.
Assertion: Number of air molecules in a room in winter is more than the number of molecules in the same room in summer.
Reason: At a given pressure and volume, the number of molecules of a given mass of a gas is directly proportional to the absolute temperature.
Assertion: Material used for making cooking utensils is the one having high specific heat and high conductivity.
Reason: Low conductivity means high specific heat.
India became a republic country on which of the following date ?
1. January 26, 1948
2. January 26, 1949
3. January 26, 1950
4. January 26, 1947
Konark Temple is siuated on the bank of
1. Arabian Sea
2.Indian Ocean
3. Bay of Bengal
4.Caspean Sea
Aravali range is situated in the
1. North-east region
2. North-west region
3. South-east region
4. South-west region
Primary sector refers to
1. Industry
2. Agriculture
3. Trade
4. Banks
When was song ‘Jana Gana Mana’ adopted as the National Anthem of India ?
1. 14th August 1947
2. 24th January 1950
3. 15th August 1947
4. 26th January 1950
Which gas is filled in the electric bulb is
1. Hydrogen
2. Nitrogen
3. Helium
4. Oxygen
At what temperature do both the Centigrade and Fahrenheit thermometers show the same reading
1. –20
2. –40
3. 42
4. 0
The human skeleton is divided into
1. Four parts
2. Two parts
3. Six parts
4. Three parts
‘Blogs’ means
1. Online journals where cyber-diarists unravel their stories
2. Black spots in somebody’s life
3. Big logs used to cross the river in hills
4. Big blocks of land used for grazing animals
A very much publicized treatment method “DOTS” is being adopted for the cure of Tuberculosis. What is the fullform of DOT ?
1. Direct Observation Treatment
2. Distant Observation Treatment
3. Domestic Oral Treatment
4. Duel Oriented Treatment
Arrange the words given below in a meaningful sequence.
1.Presentation 2.Recommendation 3. Arriva
4.Discussion 5.Introduction
1. 5, 3, 4, 1, 2
2. 3, 5, 4, 2, 1
3. 3, 5, 1, 4, 2
4. 5, 3, 1, 2, 4
What was the day on 15th August 1947?
1. Friday
2. Saturday
3. Sunday
4. Thursday
Today is Monday. After 61 days, it will be:
1. Tuesday
2. Monday
3. Sunday
4.Thursday
A bag contains 50 P, 25 P and 10 P coins in the ratio 5: 9: 4, amounting to Rs. 206. Find the number of coins of each type respectively.
1. 360, 160, 200
2.160, 360, 200
3. 200, 360, 160
4. 200, 160, 300
In an election between two candidates, one got 55% of the total valid votes, 20% of the votes were invalid. If the total number of votes was 7500, the number of valid votes that the other candidate got, was :
1. 2500
2. 2700
3. 2900
4. 3100
If each side of a square is increased by 25%, find the percentage change in its area?
1. 65.25
2. 56.25
3. 65
4. 56
Tea worth Rs. 126 per kg are mixed with a third variety in the ratio 1: 1 : 2. If the mixture is worth Rs. 153 per kg, the price of the third variety per kg will be
1. Rs. 169.50
2. Rs. 1700
3. Rs. 175.50
4. Rs. 180
If each edge of a cube is increased by 50%, find the percentage increase in Its surface area
1. 125%
2. 150%
3. 175%
4. 110%
A bag contains 6 white and 4 black balls .2 balls are drawn at random. Find the probability that they are of same colour.
1. 1/2
2. 7/15
3. 8/15
4. 1/9
There is a certain relation between two given words on one side of : : and one word is given on another side of : : while another word is to be found from the given alternatives, having the same relation with this word as the given pair has.
Select the best alternative. Seismography :
Earthquake :: Taseometer : ?
1. Landslides
2. Srains
3. Resistances
4. Volcanoes