Choose the correct option.
1. Annelida - Exhibit bilateral symmetry, metamerism and pseudocoelom.
2. Echinodermata - Exhibit tissue level organisation and radial symmetry.
3. Arthropoda - Exhibit complete digestive system and segmentation.
4. Notochord is present on ventral side in vertebrate.
Match the following columns. Mark the most appropriate match only.
|
Column-I |
Column-II |
||
|
A. |
Ctenophora |
I |
Blind sac plan |
|
B. |
Cnidaria |
II |
Comb plates |
|
C. |
Platyhelminthes |
III |
Spiny skinned |
|
D. |
Echinodermata |
IV |
Sting cells |
Codes:
A B C D
1. IV I II III
2. III II I IV
3. II IV I III
4. I III IV II
Match the columns.
|
|
Cloumn ‘A’ |
|
Column ‘B’ |
|
(a) |
Tight junctions |
(i) |
Macula adherens |
|
(b) |
Gap junctions |
(ii) |
Stop substances leaking in columnar epithelium |
|
(c) |
Inter digitations |
(iii) |
Common in transitional epithelium |
|
(d) |
Adhering junctions |
(iv) |
Rapid transfer of ions and molecules |
a b c d
1. i ii iii iv
2. ii iv iii i
3. ii iv i iii
4. ii i iii iv
Tick mark the statement which is not correct with respect to cockroach?
1. In cockroach the grinding of the food particles is performed by mandibles & gizzard.
2. Malpighian tabules help in the removal of nitrogenous wastes in the form of urea & ammonia.
3. Midgut gut of cockroach absorb food.
4. Main body cavity is called haemocoel.
Jurassic period of the Mesozoic era is characterised by-
1. Gymnosperms were dominant plants and age of dinosaur.
2. Radiation of reptiles and extinction of mammal like reptiles.
3. Archaeopteryx became extinct and angiosperms appeared.
4. Flowering plants and first reptile appeared.
Flood, drought and monsoon damage in India are the cause of:-
1. Water pollution
2. Air pollution
3. Green house effect
4. Ozone depletion
They may reduce the survival, growth and reproduction of the other organisms and reduce it's population density. They might render the organism more vulnerable to predation by making it physically weak. They are:-
1. Predator
2. Parasite
3. Ammensal
4. Commensal
Which type of interaction is shown by above graph between species A and B?
1. Mutualism
2. Commensalism
3. Competition
4. Amensalism
A bird introduced from another country become a serious pest due to:-
1. Better adaption to new area.
2. More sexual reproduction.
3. Better nesting habitats.
4. Absence of natural predator.
Consider the following four statement (a-d) and select the option which includes all the correct ones only
a. The menstrual flow results due to breakdown of myometrium lining of uterus and its blood
vessels.
b. The secretion of acrosome help the sperm enter into the cytoplasm of the ovum through the
zonapellucida and the plasma membrane.
c. Scientifically it is correct to say that the sex of the baby is determined by the mother and not by
the father.
d. The blastomeres in the blastocyst are arranged into an outer layer called trophoblast and it gets
differentiated as the embryo.
Option:-
1. Statements b, c and d.
2. Statements a, b.
3. Statements c, d.
4. Statement b only.
Find the correct statement:-
1. HIV is retrovirus having reverse transcriptase.
2. HIV has two copies of similar ssRNA.
3. Widal test is done for typhoid.
4. All the above.
Find out the correct match from the following match:-
|
Column-I |
Column-II |
Column-III |
|
|
i. |
Cry-du-chat syndrome |
Terminal deletion |
3rd chromosome |
|
ii. |
Cronic myeloid |
Reciprocal translocation |
22nd and 9th chromosome |
|
iii. |
Sickle cell anaemia |
Duplication |
X-chromosome |
1. i and ii
2. ii only
3. ii and iii
4. i only
In MOET technology:
1. Each genetic mother produces one egg per cycle.
2. Genetic mother is administered with FSH like hormones.
3. Fertilized eggs are recovered at 64-celled stage to transfer into surrogate mother.
4. New offsprings have the characters of surrogate mother.
Which one of the following pairs of the kind of cells and their secretion is correctly matched?
1. Oxyntic cells - Castle's intrinsic factor.
2. Alpha cells of Islets of Langerhans - glucagon that decreases blood sugar level.
3. Gastric glands - a digestive enzyme that hydrolyses starch.
4. Sebaceous glands - a secretion that evaporates for cooling.
How many of the hormones in the list given below interact with membrane bound receptors and regulate cellular metabolism:-
Calcitonin, Glucagon, Growth hormone, Cortisol, Thyroxin, Testosterone, Estradiol, Progesterone, Adrenalin
1. Four
2. Five
3. Six
4. Three
Consider the following statements.
(a) Occurs involuntarily and without conscious effort.
(b) At least no afferent and two efferent neuron required.
(c) Only motor neuron is involved in the action.
(d) Polysynaptic and conditional reflex.
(e) Regulated by the spinal cord.
Which of the above statements are correct for the reflex shown in the above figure?
1. a and e
2. b, c and d
3. a, b, d and e
4. a, b, c, d and e
Which of the following statement is true?
1. Cardiac output of an athlete is much lower than that of an ordinary man.
2. In each minute a single cardiac cycle is performed.
3. ECG are of no clinical diagnostic significances.
4. Cardiac cycle includes Auricular systole, ventricular systole and joint diastole/complete diastole.
Which option is correct with respect to blood urea content?
1. Hepatic portal vein - less, renal vein - much.
2. Pulmonary artery - less, hepatic vein - much.
3. Hepatic vein - less, renal artery - much.
4. Renal vein - less, hepatic vein - much.
Which of the following situations would result in the greatest degree of saturation for haemoglobin, assuming remains constant-
1. Increased levels, decreased temperature.
2. Increased levels, increased acidity.
3. Decreased levels, decreased acidity.
4. Increased levels, increased temperature.
The joint which play a significant role in locomotion:-
1. Synarthrosis
2. Synchondrosis
3. Diarthrosis
4. Both (1) and (3)
Macula maintains
1. Static equilibrium
2. Dynamic equilibrium
3. Balance & hearing
4. Hearing only
What is the correct sequence of events in DNA finger printing.
1. Hybridization of DNA fragments with a labelled probe sequence followed by separation by
electrophoresis and then transfer to a nylon membrane.
2. Separation of DNA fragments by electrophoresis followed by hybridization with labelled probe
sequence and then transfer to a nylon membrane.
3. Separation of DNA fragments by electrophoresis followed by transfer to a nylon membrane and
then hybridization with a labelled probe sequence.
4. Transfer of DNA fragments to a nylon membrane followed by separation by electrophoresis and
then hybridization with a labelled probe sequence.
The (I) provides the template, during translation and (II) brings amino acids and read the genetic code, and (III) play structural and catalytic role.
Choose the correct word for I, II and III blanks?
|
I |
II |
III |
|
|
1. |
r-RNA |
t-RNA |
m-RMA |
|
2. |
hn-RNA |
Sn-RNA |
t-RNA |
|
3. |
m-RNA |
t-RNA |
r-RNA |
|
4. |
m-RNA |
r-RNA |
t-RNA |
Given figure represents the maturation of proinsulin into insulin. Identify the product
1. Polypeptide chain A
2. Polypeptide chain B
3. Polypeptide chain C
4. Polypeptide chain D
Read the following four (A-D) statements:
1. When migration of a section of population to another place, gene frequencies changes.
2. If gene migration happens multiple times there would be a gene flow.
3. The sum total of genes in reproductive gametes of a population called as gene pool.
4. The existence within the population of disadvantagous allele in heterozygous genotypes is
known as genetic load.
How many of the above statements are true:-
1. Two
2. One
3. Four
4. Three
Paraquat:-
1. Inhibits flow of from PSII to SPI.
2. Inhibits flow of from PSI to ferredoxin.
3. Inhibits flow of from PSII to plastoquinone.
4. Promotes flow of from PSI to ferredoxin.
Glycolysis is a chain of (A) reaction which occur in the (B) of the cell.
1. (A)-Five, (B)-Mitochondria.
2. (A)-Ten, (B)-Mitochondria.
3. (A)-Eight, (B)-Cytoplasm.
4. (A)-Ten, (B)-Cytoplasm.
Inhibition of cell-division in plants is due to the deficiency of:-
1. N, S, Mn, Co
2. N, S, Mo, Si
3. Mo, S, Fe, Se
4. N, K, S, Mo
Which of the following group of enzymes catalyzes removal of groups from substrate without hydrolysis of and it leads to formation of double bond:-
1. Oxidoreductases
2. Lyases
3. Hydrolases
4. Isomerases
Match the column I with column-II :-
Column I Column II
(i) Dinoflagellates (a) Pellicle
(ii) Diatoms (b) Saprotrophic
(iii) Slime moulds (c) Cellulosic cell wall
(iv) Euglenoids (d) Cellulosic cell wall with silica
1. i-d, ii-c, iii-b, iv-a
2. i-c, ii-d, iii-b, iv-a
3. i-c, ii-d, iii-a, iv-b
4. i-d, ii-c, iii-a, iv-b
Read the following statements. Find out how many statements are correct?
(A) Key are based on the contrasting character generally in a pair called couplet.
(B) Each statement in the key is called lead.
(C) Separate taxonomic keys are required for each taxonomic category.
(D) Keys are generally analytical in nature.
1. Two
2. Three
3. Four
4. One
Choose the incorrect statement:-
1. Gametophyte of Gymnosperm are always Dioecious
2. Sporophyte of Gymnosperm are always heterosporous.
3. Sporophyte of pteridophytes may be monoecious or dioecious.
4. Gametophyte of pteridophytes may be monoecious or dioecious.
Choose the incorrect match from following:-
1. Cycas-branched stem.
2. Salvinia-heterosporous sporophyte.
3. Marchantia-Elaters.
4. Chlorella-used by space travellers.
Important site for formation of glycoproteins and glycolipids is:-
1. Lysosome
2. Vacuole
3. Golgi apparatus
4. Plastid
Identify the following diagram :-
1. Transition to Metaphase (mitosis).
2. Transition to Anaphase II (meiosis).
3. Transition to Metaphase I (meiosis).
4. Transition to Anaphase (mitosis).
Macromolecule chitin is an example of:-
1. Simple polysaccharide.
2. Nitrogen containing polysaccharide.
3. Phosphorus containing polysaccharide.
4. Sulphur containing polysaccharide.
In some (A) , the leaf base may become swollen and is called as (B) .
A B
1. Monocots Sheathing leaf base
2. Legumes Pulvinus
3. Legumes Sheathing leaf base
4. Monocots Pulvinus
Select the correct statements from the following:-
(i) In dicotyledonous roots, the parenchymatous cells which lie between the xylem and phloem
are medullary rays.
(ii) Gymnosperms lack sieve tubes and companion cells.
(iii) Presence of vessels is characteristic feature of angiosperms.
(iv) The end walls of vessels are imperforated but pitted.
1. Only (i)
2. (i) & (iv)
3. (ii) & (iii)
4. All of the above
How many meiotic division are required to form 300 seeds of wheat?
1. 250
2. 350
3. 75
4. 375
Which of the following hormone is used for shoot differentiation in callus?
1. 2, 4-D.
2. Benzyl amino purine (BAP).
3. Deformylase.
4. Gibberellic acid.
Assertion: During green revolution, crop production is increased.
Reason: Deforestation is one of the method of green revolution to increases productivity.
Assertion: The levels of FSH and LH attain a peak level in the middle of the menstrual cycle.
Reason: The FSH and LH surge induces rupture of Graafian follicle that is called ovulation.
Assertion: The substances which produces allergy are called allergens.
Reason: Common allergens are mites in dust, pollen and animal dander.
Assertion: Continuous and close inbreeding among milker animals is discouraged by animal breeders.
Reason: Continuous and close inbreeding leads to inbreeding depression.
Assertion: In PCT all of the essential nutrient and 70-80 percent of electrolytes and water are reabsorbed.
Reason: PCT is lined by simple cuboidal brush border epithelium which decreases the surface, are for reabsorption.
Assertion: Right atrium and right ventricle of heart are known as pulmonary heart.
Reason: Both right atrium and ventricle pump the deoxygenated blood.
Assertion: Neural signals through the parasympathetic nerves can increase the cardiac output.
Reason: Parasympathetic neurons stimulation can increase the activities of SA node and strength
of ventricular contraction.
Assertion: Dissociation of from carbamino haemoglobin takes place at alveolar level.
Reason: High concentration at alveolar level promotes the dissociation of carbaminohaemoglobin.
Assertion: Paratharmone is a hypercalcemic hormone.
Reason: PTH increases level in blood.
Assertion: Ear is a stato Acoustic organ.
Reason: Ear is related to hearing & balance.
Assertion: Dominance is not always an autonomous feature of gene or its product.
Reason: Occasionally a single gene product may produce more than one effects.
Assertion: Many transgenic animals are designed to study how gene contribute to the development
of disease.
Reason: Transgenic animal models can be used to understand how genes are regulated and how they
affect normal functions of body by trans genic animals.
Assertion: Dark reaction occur only at night in the stroma of chloroplast.
Reason: fixation occur only during cycle.
Assertion: Diatomaceous earth is used in polishing, filtration of oil and syrups.
Reason: Diatomaceous earth is gritty due to presence of silica.
Assertion: Seeds are formed in Gymnosperms.
Reason: Female gametophyte is retained within megasporangium in Gymnosperms.
Assertion: In mitochondria, process of ATP formation with the help of electron transport system, is
called oxidative phosphorylation.
Reason: In mitochondria, energy for the creation of proton gradient is obtained from the splitting of
oxygen.
Assertion: Saturated fatty acids are non-essential fatty acids.
Reason: They can be synthesized in animal body.
Assertion: Strawberry is a pseudocarpic fruit.
Reason: The fruit develops from apocarpous ovary.
Assertion: Procambium is a type of primary meristem.
Reason: It develops at the early stage of plant life.
Assertion: Pollen-pistil interaction is an important step for post pollination germination of pollen grains
in flowering plants.
Reason: It is an interaction in between pollen and pistil leading to the recognition followed by acceptance
or rejection for compatible or incompatible pollen grains.
What is the shortest wavelength line in the Paschen series of ion?
1.
2.
3.
4.
Which of the following does not represent radius of the atom correctly?
(i) Simple cubic cell : Radius =
(ii) Face centred cubic cell : Radius =
(iii) Body centred cubic cell : Radius =
1. (i)
2. (iii)
3. (ii)
4. (i) and (ii)
In the adsorption of a gas on solid, Freundlich isotherm is obeyed. The slope of the plot is zero. Thus, the ectent of adsorption is
1. Directly proportional to the pressure of gas
2. Inversely proportional to the pressure of the gas.
3. Independent of the pressure of the gas.
4. Proportional to the square of the pressure of the gas.
Which of the following is not the correct difference between lyophobic and lyophilic sols?
Lyophobic sols Lyophilic sols
1. Require special methods for preparation Can be prepared by shaking with the solvent
2. Are reversible Are irreversible
3. Easily coagulated by electrolytes Cannot be easily coaguilated
4. Are less stable Are more stable
Given below are the critical temperatures of a few gases. When the gases are started cooling, which gas will liquify first and which will liquify in the end?
Gas Tc/K
N2 126.0
CO2 304.10
NH3 405.5
O2 154.3
1. will liquify first and at last
2. will liquify first and at last
3. will liquify first and at last
4. will liquify first and at last
Fe shows an oxidation state of +1 in :-
1.
2.
3.
4.
A first order reaction was started with a decimolar solution of the reactant, 8 minutes and 20 seconds later its concentration was found to be M/100. So the rate constant of the reaction is :-
1.
2.
3.
4.
The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is 1/100th of that of a strong acid (HX, 1M) at 25. The of HA is :-
1.
2.
3.
4.
How many moles of can be produced by reaction of 0.10 moles F, o.36 moles and 0.90 moles C according to the following reaction:-
1. 0.060
2. 0.030
3. 0.045
4. 0.075
Van't Hoffs equation shows the effect of temperature on equilibrium constants and .The varies with temperature according to the relation:-
1. log
2. log
3. log
4. None of these
In which of the following options, the heat of neutralisation per equivalent will be highest?
1. and
2. and
3. and
4. and
In the closest packing of atoms:
1. The size of tetrahedral void is greater than that of octahedral void.
2. The size of tetrahedral void is smaller than that of octahedral void.
3. The size of tetrahedral void is equal to that of octahedral void.
4. The size of tetrahedral void may be greater or smaller or equal to that of octahedral void depending
upon the size of atoms.
Two liquids A and B have and in the ratio of 1 : 3 and the ratio of number of moles of A and B in liquid phase are 1 : 3 then mole fraction of 'A' in vapour phase in equilibrium with the solution is equal to:
1. 0.1
2. 0.2
3. 0.5
4. 1.0
Oxidising product of the substance would be:-
1.
2.
3.
4.
Correct IUPAC name of the given compound
1. 5-amino-3-hydroxy benzene carbaldehyde.
2. 3-amino-5-formyl benzenol.
3. 3-amino-5-hydroxy benzaldehyde.
4. 3-formyl-5-hydroxy aniline.
Which of the following can have meso isomer?
1.
2.
3.
4.
The number of structural isomeric aldehyde with molecular formula is:-
1. 3
2. 4
3. 5
4. 6
Arrange the following in increasing order of basic strength:-
1.
2.
3.
4.
Which of the following resonating structure is most stable?
1.
2.
3.
4.
Give decreasing order of reactivity for ESR reaction:-
1. 2 > 3 > 4 > 1
2. 1 > 2 > 3 > 4
3. 4 > 2 > 3 > 1
4. 2 > 3 > 1 > 4
Phenol
. The product
is:
1.
2.
3.
4.
Intermediates formed in the reaction P and Q is :-
P.
Q.
1.
2.
3.
4.
In the following reaction,
the structure of the major product ‘X’ is
1. 
2. 
3. 
4. 
Which of the following shows mutarotation?
1. Sucrose.
2. Fructose.
3. Starch.
4. Cellulose.
Monomer unit of terylene (Dacron) polymer is:-
1. Ethylene glycol and terephthalic acid.
2. Ethylene glycol and phthalic acid.
3. Adipic acid and Hexamethylenediamine.
4. Caprolactum.
Example of Bactericidal antibiotic is/are:-
1. Penicillin.
2. Aminoglycosides.
3. Ofloxacin.
4. All.
Cyclohexene on ozonolysis followed by reaction with zinc dust and water gives compound E. Compound E on further treatment with aqueous KOH yields compound F. Compound F is
1. 
2. 
3. 
4. 
Maximum no. of electron in outermost shell of s, p, d and f block elements are respectively:-
1. 2, 8, 2, 2
2. 2, 6, 10, 14
3. 2, 8, 18, 32
4. 2, 8, 10, 8
No. of electron in penultimate shell of d-block elements :-
1. 9-18
2. 19-32
3. 1-10
4. 9-32
A Ans. Sol. tion of metal ion when treated with KI gives a red precipitate which disAns. Sol. es in excess KI to give a colourless Ans. Sol. tion. Moreover, the Ans. Sol. tion of metal ion on treatment with a Soltion of cobalt (II) thiocyanate gives rise to a deep blue crystalline precipitate. The metal ion is
1. Pb2+
2. Hg2+
3. Cu2+
4. Co2+
Among the following metal carbonyls, the bond order is lowest in:-
1.
2.
3.
4.
In which of the following pairs, the EAN of central metal atom is not same?
1.
2.
3.
4.
Which of the following is correct?
1. shows amphoteric behaviour.
2. White phosphorus has 6 P-P bond.
3. Thermodynamic stability of diamond < thermodynamic stability of graphite.
4. All of the above are correct.
Moles of hydrocarbon produced by hydrolysis of 1 mole (magnesium carbide) is :-
1. 1
2. 3
3. 4
4. 2
Among the total number of substance which produce gas when react with cold water:-
1. 3
2. 4
3. 5
4. 6
(Ammonium dichromate) is used in fire works. The green coloured powder blown in air is:-
1.
2.
3.
4.
Geometry of is :-
1. distorted tetrahedral
2. V-shape
3. T-shape
4. K-shape
The sum of total Number of sigma and pie bonds present in:-
1. 10
2. 12
3. 13
4. 15
has hybridization respectively:-
1.
2.
3.
4.
Which of the following is not incorrect?
1. 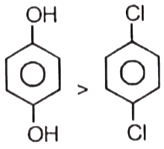
2. (solubility order)
3. (Lattice energy)
4. All are correct
Assertion: An orbital cannot have more than two electrons and they must have opposite spins.
Reason: No two electrons in an atom can have same set of all the four quantum numbers as per
Pauli's exclusion principle.
Assertion: If the activation energy of a reaction is zero, temperature will have no effect on the rate constant.
Reason: Lower the activation energy, faster is the reaction.
Assertion: Equivalent mass of a base
Reason: Acidity is the number of replaceable hydrogen atoms in one molecule of the base.
Assertion: Change in internal energy is zero for all gases at constant temperature.
Reason: Internal energy is function of temperature.
Assertion: An increase in surface area increases the rate of evaporation.
Reason: Stronger the inter-molecular attraction forces, faster is the rate of evaporation at a given temperature.
Assertion: is a disproportionation reaction.
Reason: Oxidation number can be zero, negative, positive or fractional.
Assertion: Boiling point of 
Reason: Molecules of primary amines are associated due to intermolecular hydrogen bonding.
Assertion: 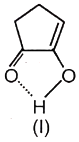
Reason: All enol forms are more stable than keto form.
Assertion: Allyl halides and benzyl halides give goof yield of product in reaction.
Reason: In case of allyl and benzyl halides, transition state is resonance stabilized.
Assertion: Aromatic primary amines cannot be prepared by Gabriel phthalimide synthesis.
Reason: Aryl halides do not undergo nucleophilic substitution reactions.
Assertion: Bromine water test can be used to distinguish 1-Butene and 1-Butyne
Reason: unsaturated hydrocarbons decolourise bromine water.
Assertion: Rate of nitration of benzene and hexa deutero benzene are different
Reason: C-H bond is stronger than C-D bond
Assertion: On free radical monochlorination of four monochloro structure
isomers are possible.
Reason: In there are four different radicals formed.
Assertion: bond is stronger than bond
Reason: Rotation around bond is possible
Assertion: bond length of is shorter than
Reason: is an ionic compound.
Assertion: has least tendency among halides of nitrogen to act as donor.
Reason: of participates in back bonding.
Assertion: molecule has two bond.
Reason: 4 'e' are present in & bonding M.O. while antibonding M.O.'s are empty.
Assertion: First ionisation energy of Al is less than Mg.
Reason: Ionic radius of Al smaller than Mg.
Assertion: is an acidic oxide while is basic.
Reason: Nonmetal oxides are acidic while oxides of metals are basic.
Assertion: Metallic character increases down the group in lA.
Reason: Down the group ionisation energy decreases.
If y represents pressure and x represents velocity gradient then the dimension of are:-
1.
2.
3.
4.
A physical quantity P is given by the relation P=, here t is time then dimension of are:-
1. [T]
2.
3.
4.
Time taken by the projectile to reach from A to B is t. The distance AB is equal to:
1.
2.
3.
4. 2 ut
At a metro station, a girl walks up a stationary escalator in time . If she remains stationary on the escalator, then the escalator take her up in time . The time taken by her to walk up on the moving escalator will be:
1.
2.
3.
4.
A uniform rope of length L and mass M is placed on a smooth fixed wedge as shown. Both ends of rope are at same horizontal level. The rope is initially released from rest, then the magnitude of initial acceleration of rope is:
1. zero
2.
3.
4. None of these.
A 3 kg block by a force which is inclined at 37 to the horizontal table. The friction coefficient between the table and block is 1/3, for what minimum value of this force, will the block start sliding?
1. 5 N
2. 10 N
3. 20 N
4. 25 N
A particle describes a horizontal circle on smooth inner surface of a conical funnel as shown. If the height of the plane of the circle above the vertex is 10 cm, then find the speed of the particle. (g=10 )
1. 1 m/s
2. m/s
3. 2 m/s
4. m/s
A force of N acts on a 5 kg body. The body is at a position of m and is travelling at 4 m/s. The force acts on the body until it is at the position m. The final speed of the body is:
1. 5 m/s
2. 10 m/s
3.
4.
The figure shows how the potential energy \((U)\) of a particle varies with its position \(x.\) The particle moves under the influence of a conservative force along the \(x\)-axis.
| Statement I: | If the particle is released from rest at the origin \((x=0),\) it will start moving in the negative \(x\)-direction. |
| Statement II: | The positions \(x = 5~\text{m}\) and \(x =- 5~\text{m}\) are both stable equilibrium points of the particle. |
| 1. | Statement I is correct only. |
| 2. | Statement II is correct only. |
| 3. | Both statements are correct. |
| 4. | Both statements are incorrect. |
The given figure shows the electric lines of force emerging from a charged body. If the electric fields at A and B are respectively and if the displacement between A and B is r, then:
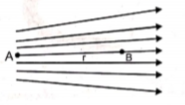
1.
2.
3.
4.
A charged particle of mass m and charge q is released from rest in an electric field of constant magnitude E. The kinetic energy of the particle after time t is:
1.
2.
3.
4.
A hollow charged metal sphere has a radius r. If the potential difference between its surface and a point at distance 3r from the centreis V, then electric intensity at distance 3r from the centre is:
1.
2.
3.
4.
A capacitor of 10 charged upto 250 volt is connected in parallel with another capacitor of 5 chrged upto 100 volt. The common potential is:
1. 500 V
2. 400 V
3. 300 V
4. 200 V
Power dissipated in 5 resistance is:
1. 512 W
2. 480 W
3. 120 W
4. 605 W
A 6V cell of negligible internal resistance is connected across a uniform wire AB of length 100cm. The positive terminal of another cell of 4V and internal resistance 1 is joined at point A as shown in figure. Take potential at B tobe zero. If potential at point D of wire AB is equal to potential of point C, then
1. AD = 60cm
2. AD = 50cm
3. AD = 40cm
4. AD = cm
For given figure consider the following statements:
(i) i= 0.25 A
(ii)
(iii)
(iv) equivalent resistance = 7
Correct statements are-
1. (i),(ii)
2. (i),(ii),(iii)
3. (i),(ii),(iv)
4. (i),(ii),(iii) and (iv)
A straight conductor of mass m and carrying a current i is hinged at one end and placed in a plane perpendicular to the magnetic field of intensity B as shown in the figure. At t=any moment if the conductor is let free, then the angular acceleration of the conductor will be (neglect gravity):-
1.
2.
3.
4.
Adjoining figure shows a rectangular loop of conductor carrying a current i. The length and breadth of the loop are respectively a and b. The magnetic field at the centre of loop is:-
1.
2.
3.
4.
As shown in the figure, P and Q are two coaxial conducting loops seperated by some distance. WHen the switch S is closed, a clockwise current flows in P (as seen by E) and an induces current flows in Q. The switch remains closed for a long time. Now S is opened, a current flows in Q. Then the directions of and (as sheen by E) are:-
1. Respectively clockwise and anticlockwise.
2. Both clockwise
3. Both anticlockwise
4. Respectively anticlockwise and clockwise.
For an electromagnetic wave the elctric and magnetic field vectors are along +y and +z directions respectively. Then the propogation of the EM wave should be along:-
1. +y direction
2. -y direction
3. +x direction
4. +z direction
In an oscillating LC circuit, the maximum charge on the capaciter is Q. The charge on the capacitor when the energy is stored equally between the electric and magnetic fields is:-
1.
2.
3.
4. Q
A ray of light enters into the prism normally at face 1 and comes out from the face 3. The value of prism angle A may be :-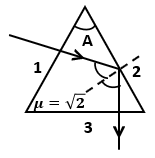
1. 30
2. 35
3. 42
4. 47
A telescope consists of two thin lenses of focal lengths 0.3 m and 3 cm, respectively. It is focussed on Moon which subtends an angle of 0.5 at the objective. Then, the angle subtended at the eye by the final image will be :
1. 5
2. 0.25
3. 0.5
4. 0.35
In the Frounhufer diffraction pattern of a single slit under polychromatic illumination, the first minimum with the wavelength is found to be coincides with the third maximum of wavelength then
1. 3=0.3
2. 3=0.3
3. = 3.5
4. 0.3= 3
If the ratio of amplitude of two interfering waves is 4 : 3, then the ratio of maximum and minimum intensity is
1. 16 : 8
2. 18 : 16
3. 49 : 1
4. 94 : 1
A satellite is moving round the earth with a speed of v in a circular orbit of radius r. If the orbit's radius is reduced by 1% its speed will:-
1. Increase by 1%
2. Increase by 0.5%
3. Decrease by 1%
4. Decrease by 0.5%
When Lithium is bombarded by a proton, two alpha particles are produced. The masses of are 7.016004 u, 1.007825 u and 4.002603 u respectively. The reaction energy is nearly:
1. 17 eV
2. 17 keV
3. 17 MeV
4. 170 MeV
When photons of wavelength are incident on an isolated metal sphere suspended by an insulated thread, the corresponding stoppig potential is found to be V. When photons of wavelength are used, the corresponding stopping potential was thrice the above value. If light of wavelength is used, the stopping potential for this case will be
1.
2.
3.
4.
A carrier frequency of 1 MHz ans peak value of 10V is amplitude modulated with a singal frequency of 10 kHz with peak value of 0.5 V. Then the modulation index and the side band frequencies respectively are
1. 0.05 and 10.010 MHz
2. 0.5 and 10.010 MHz
3. 0.05 and 10.005 MHz
4. 0.5 and 10.005 MHz
In the circuit shown in figure the maximum output voltage is
1. 0 V
2. 5 V
3. 10 V
4.
Two identical stringed instruments have frequency 100 Hz. If tension in one of them is increased by 4% and they are sounded together then the number of beats in one second is
1. 1
2. 8
3. 4
4. 2
Two cars are moving on two perpendicular roads towards a acrossing with uniform speeds of 72 km/hr and 36 km/hr. If first car blows horn of frequency 280 Hz, then the frequency of horn heard by the driver of second car when line joining the cars make 45 angle with the roads; will be
1. 321 Hz
2. 298 Hz
3. 300 Hz
4. 280 Hz
A horizontal force F acts on a solid sphere of mass M at its centre as shown. Coefficient of friction between the ground and the sphere is . What is maximum value of F, for which there is no slipping?
1.
2.
3.
4.
A long cylindrical vessel has a small hole of diameter D at its bottom. This vessel can be lowered vertically in water to a depth h without any water entering the vessel. Given A= surface tension, B= dnsity of liquid, C= acceleration due to gravity. The value of h is:-
1.
2.
3.
4.
The excess pressure due to surface tension inside a spherical drop is 6 unit. If eight such drops combine then the excess pressure due to surface tension inside the larger drop is:-
1. 3 unit
2. 6 unit
3. 12 unit
4. 48 unit
The value of for a gas in state A and in another state B. If denote the pressure and denote the temperatures in the two states, then:
| 1. | \(P_A=P_B ; T_A>T_B\) |
| 2. | \(P_A>P_B ; T_A=T_B\) |
| 3. | \(P_A<P_B ; T_A>T_B\) |
| 4. | \(P_A=P_B ; T_A<T_B\) |
If the P-V diagrams of two thermodynamics devices working in a cyclic process are as shown in the figure, then:-
1. A is a heat engine, B is a heat piump or refrigerator.
2. B is a heat engine, A is a heat pump or refrigerator.
3. Both A and B are heat engines.
4. Both A and B are heat pumps or refrigerator.
The density of material \(A\) is \(1500 ~\text{kg/m}^ 3\) and that of another material \(B\) is \(2000 ~\text{kg/m}^ 3.\) It is found that the heat capacity of \(8\) volumes of \(A\) is equal to the heat capacity of \(12\) volumes of \(B.\) The ratio of specific heats of \(A\) and \(B\) will be:
1. \(1 : 2\)
2. \(3 : 1\)
3. \(3 : 2\)
4. \(2 : 1\)
The plot of velocity (v) versus displacement (x) of a particle executing simple harmonic motion is shown in the figure. The time period of oscillation of the particle is:
1.
2.
3.
3.
When a block of mass m is suspended separately by two different springs have time period . If same mass is connected to parallel combination of bth springs, then its time period is given by :-
1.
2.
3.
4.
Assertion: An exact number has infinite number of significant digits.
Reason: A number which is not a measured value has infinite number of significant digits.
Assertion: There is no appreciable change in the position of the body during the action of the
impulsive force.
Reason: In case of impulsive force the time of action of the force is very short.
Assertion: Displacement of a body may be zero when distance travelled by it is not zero.
Reason: The displacement is the longest distance between initial and final position.
Assertion: A body can have energy without having momentum.
Reason: A body can have momentum without having mechanical energy.
Assertion: In uniform circular motion speed of particle must be constant.
Reason: In uniform circular motion tangential force on particle is always zero.
Assertion: When a charge is placed at a corner of a square, the flux passing through the square
will be zero.
Reason: When electric field and area vector are perpendicular to each other at every point, then
the flux passing through that surface will be zero.
Assertion: When a positive point charge is placed near a neutral conductor then the conductor
becomes negatively charged.
Reason: Induction is possible in conductors only.
Assertion: The drift speed of electrons is small and change of electrons is also very small yet we
can get a large current in a metal.
Reason: At room temperature thermal speed of electrons is very low.
Assertion: A wire carrying current has non zero electric field around it.
Reason: Electric field is produced due to drift of free electrons.
Assertion: The sensitivity of a moving coil galvanometer is increased by placing a suitable magnetic
material as a core inside the coil.
Reason: Soft iron has a high magnetic permeability and cannot be easily magnetized or demagnetized.
Assertion: Self-inductance is called the inertia of electricity.
Reason: In a LR circuit, the inductor opposes any change in current.
Assertion: Power loss in Ideal choke coil is zero.
Reason: Ideal choke coil has zero resistance.
Assertion: For observing traffic at our back, we prefer to use a convex mirror.
Reason: A convex mirror has a much larger field of view than a plane mirror or a concave mirror.
| Assertion (A): | Radio waves can be polarised. |
| Reason (R): | Sound waves in air are longitudinal in nature. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Assertion: Light described by equation having work function . The maximum kinetic energy of
the photoelectron is
Reason: Maximum kinetic energy of photoelectron depends on the maximum frequency present in
incident light according to Einstein's photoelectric effect equation.
Assertion: In transistor common emitter mode as an amplifier is preferred over common base mode.
Reason: In common emitter mode the input signal is connected in series with the voltage applied to the
base emitter junction.
Assertion: The change in air pressure effect the speed of sound.
Reason: The speed of sound in a gas is proportional to square root of pressure.
Assertion: Stress is the internal force per unit area of a body
Reason: Rubber is more elastic than steel
Assertion: When a gas is compressed suddenly, its temperature increases.
Reason: Work done in compression of gas increases internal energy of gas in adiabatic process.
Assertion: When a simple pendulum is made to oscillate on the surface of moon, its time period increase.
Reason: Moon is much smaller compared to earth.
Which state has declared the Indus river Dolphins as the state aquatic animal?
1. Haryana
2. Punjab
3. Delhi
4. Rajasthan
Who won women's single title in Australian Open 2019?
1. Simona Halep
2. Petra Kvitova
3. Naomi Osaka
4. Caroline Wozniacki
Who has been chosen for 2019 Bharat Ratna award?
1. Bhupen Hazarika
2. Nanaji Deshmukh
3. Pranab Mukherjee
4. All of the above
According to World Steel Association, which country has replaced Japan as world's second largest steel producing country?
1. India
2. China
3. Russia
4. USA
Which world is listed as "Hindi World of the Year 2018" by Oxford Dictionaries?
1. Stree India
2. Stree Shakti
3. Yuva
4. Nari Shakti
Where is the headquarters of India Council of Agricultural Research?
1. Lucknow
2. Mumbai
3. Chennai
4. New Delhi
Name of the first Indian athlete who is nominated for the prestigious "Laureus World Comeback of the Year" Award for 2019.
1. P.V. Sindhu
2. Vinesh Phogat
3. Saina Nehwal
4. Abhinav Bindra
Which city is named as the World Capital of Architecture for 2020 by UNESCO?
1. Brasilia
2. Salvador
3. Rio de Janeiro
4. Fortaleza
Name the mountaineer who became the first Indian and youngest man to climb the 7 highest peaks and volcanic summits across all continents
1. Kuntal Joisher
2. Krushnaa Patil
3. Mingma Sherpa
4. Satyarup Siddhanta
Who has been chosen as the "Emerging Player of the Year 2018" by the International Cricket Council (ICC)?
1. Ross Taylor
2. Mustafizur Rahman
3. Rohit Sharma
4. Rishabh Pant
In the following question, select the world which cannot be formed using the letters of 'OPTICIAN' the given word.
1. PAINT
2. PAIN
3. COIN
4. TAIL
In the following question below are given some statements followed by some conclusions. Taking the given statements to be true even if they seem to be at variance from commonly known facts, read all the conclusions and then decide which of the given conclusion logically follows the given statements.
Statements:
I. All pens are chairs.
II. Some cups are pens.
Conclusions:
I. All pens are chairs.
II. Some cups are pens.
1. Only conclusion (i) follows
2. Only conclusion (II) follows
3. Both conclusions follow
4. Neither conclusion (I) nor (II) follows
In a certain code language, "SLOPE" is written as "54973" and "CRAMP" is written as "18627". How is "PROMO" written in that code language?
1. 76828
2. 96838
3. 76939
4. 78929
If 'c' means '', 'd' means '+', 'e' means '' and 'f' means '', then 24 c 3 f 4 d 6=?
1. 32
2. 30
3. 38
4. 28
After their day's work two village women are returning home. The first woman walks 1.5 km West, then turns to her right and walks 3 km to reach her house. In the meanwhile the second woman walks 1 km North, then she turns East and walks 3.5 km, then she turns to her left and walks 2 km to reach her house. Where is the first woman's house with respect to the second woman's house?
1. 2 km West
2. 5 km East
3. 5 km West
4. 2 km East
In an examination Raj got more marks than Modi but not as many as Meena. Meena got more marks than Ganesh and Rupali. Ganesh got less marks than Modi but his marks are not the lowest in the group. Who is second in the descending order of marks?
1. Meena
2. Rupali
3. Raj
4. Ganesh
Select the missing number from the given alternatives.
|
? |
120 |
150 |
|
110 |
100 |
90 |
|
80 |
60 |
40 |
1. 60
2. 70
3. 80
4. 90
Which answer figure will complete the pattern in the question figure?
1.
2.
3.
4.
From the given answer figures, select the one in which the question figure is hidden/embedded.
1.
2.
3.
4.
A piece of paper is folded and cut as shown below in the question figures. From the given answer figures, indicate how it will appear when opened.
1.
2.
3.
4.