The length and breadth of a rectangular strip are measured to be 750.0 cm and 2.0 cm. The area of the strip in cm2 will be reported as
1. 1500.0
2. 1.500 × 103
3. 1.50 × 103
4. 1.5 × 103
If velocity (v) varies with displacement (x) as shown in the graph, then the acceleration
1. Is constant.
2. Increases with time.
3. Decreases with time.
4. Is zero at x = 0.
A particle is projected vertically up with speed v. The particle covers same distance during 6th second and 7th second. Then (g = 10 m/s2) v is
1. 50 m/s
2. 60 m/s
3. 80 m/s
4. 120 m/s
A particle is projected at angle with the horizontal. At a certain instant the velocity is and gravitational force on the particle is . The value of is
1. Positive during upward journey.
2. Negative during downward journey.
3. Zero at the highest point.
4. All of these.
What is the frictional force acting on the block B?
1. Zero
2. 10 N
3. 50 N
4. 60 N
What is the value of tension in the string connecting the blocks A and B? (g = 10 m/s2)
1. 48 N
2. 24 N
3. 12 N
4. Zero
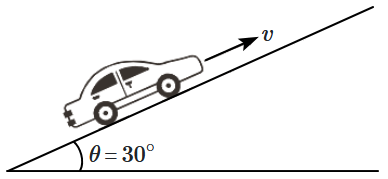
1. \(60~\text{kW}\)
2. \(50~\text{kW}\)
3. \(30~\text{kW}\)
4. \(10~\text{kW}\)
A wooden block of mass \(M\) is suspended from a fixed support by a light thread as shown. A bullet of mass \(M\) moving horizontally with speed \(v\) collides and get embedded into the block. If the block and bullet system rises up till the thread becomes horizontal, the minimum value of \(v\) is:
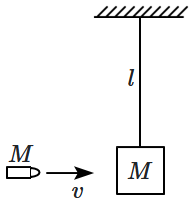
1. \(\sqrt{16gl}\)
2. \(\sqrt{8gl}\)
3. \(\sqrt{5gl}\)
4. \(\sqrt{gl}\)
What is the moment of inertia of the solid sphere of mass M and radius ‘a’ about z-axis?
1.
2.
3.
4.
Two particles of mass m1 and m2 start to move towards each other due to mutual forces of attraction. At a certain instant the speed of the two particles are v1 and v2 ; then speed of centre of mass is
1.
2.
3.
4. Zero
A sphere rolls down without slipping on an inclined plane from a point at vertical height h. The speed of the centre of mass of the sphere when it reaches the bottom is:
1.
2.
3.
4.
A horizontal force acts on the disc shown in the figure, kept on a rough horizontal surface, then
1. Friction is in the left side.
2. Friction is in the right side.
3. Friction is zero.
4. Any of the above depending on the value of F.
At a certain point of the circular orbit of a satellite, if some mass is dropped from the satellite, then:
1. It will revolve in the same orbit with the same orbital velocity in the opposite direction of the satellite.
2. Revolve in the same orbit with the same orbital velocity in the same direction of the satellite.
3. Mass drop to earth.
4. Escape from orbit linearly.
If a particle is dropped from a height \(h = 3R\) from the Earth's surface, the speed with which the particle will strike the ground is:
1. \(\sqrt{3gR}\)
2. \(\sqrt{2gR}\)
3. \(\sqrt{1.5gR}\)
4. \(\sqrt{gR}\)
Heat is flowing in steady state in an insulated rod as shown. If then which of the following is incorrect?
1. Temperature is same at all points of the rod.
2. Temperature decreases from A to B.
3. At a given point temperature does not change with time.
4. No part of the rod absorbs heat energy.
The temperature of a body falls from 50°C to 40°C in ten minutes. After the next ten minutes, the temperature change will become
1. > 10°
2. < 10°
3. > 20°
4. = 10°
Select the correct statement for the processes ‘1’ and ‘2’ as shown in the diagram. [The symbols stand for their usual meanings]
1.
2.
3.
4. All of these
The ratio of temperature T1 and T2 in the given P-V curve is-
1.
2.
2.
3.
What is the number of the average degrees of freedom for the mixture containing two moles of helium & 3 moles of hydrogen at room temperature?
1. 3.8
2. 4.0
3. 4.2
4. 4.5
The time-period of a simple harmonic oscillator is 12 s and its amplitude is 14 m. If initially the oscillator is at mean position then the average speed during oscillation will be
1. 1 m/s
2. 2 m/s
3. 5 m/s
4. 14 m/s
In a stationary lift a simple pendulum oscillates with time-period T and when the lift moves up with constant velocity, the time-period become T', then
1. T' = T
2. T' < T
3. T' > T
4. Any of the above depending on the value of velocity.
Which of the following graphs may represent the potential energy (U) of a simple harmonic oscillation with displacement (x)?
1.
2.
3.
4.
The equation of a travelling wave is given as , where x, t are in metres and in seconds respectively. The minimum distance between two particles oscillating in same phase is
1. 40 m
2. 20 m
3. 10 m
4. 5 m
Two identical sonometer wires oscillate in unison. When tension in one wire increased by 1%, 4 beats per second are heard. The initial frequency of the wires is
1. 800 Hz
2. 600 Hz
3. 400 Hz
4. 200 Hz
A car moves with speed v0 towards a vertical cliff, the apparent frequency of sound coming after reflection from the cliff is found 10% higher. If speed of sound in air is 336 m/s, the value of v0
1. 12 m/s
2. 16 m/s
3. 18 m/s
4. 33.6 m/s
A point charge +q is kept outside a conducting shell as shown. Electric flux associated with the Gaussian surface S (shown dotted) is
1. Positive.
2. Negative.
3. Zero.
4. Data is insuffient to predict.
What is the value of electric field at point P if a point charge q is kept inside a conducting spherical shell as shown?
1.
2.
3.
4. Zero
The equivalent capacitance between the points A and B is
1.
2.
3.
4.
Two charged capacitors are shown in the diagram. If a dielectric slab is introduced between the plates of capacitor x, then potential of
1. Both the capacitors will decrease.
2. x will decrease and y will increase.
3. Both the capacitors will increase.
4. x will decrease and that of y will remain same.
In the diagram shown current increases at the rate of 2 A/s. At an instant when value of current is 5 A, the potential at point ‘a’ is
1. 41 V
2. 29 V
3. 24 V
4. 15 V
If readings of the voltmeters \(V_1\) and \(V_2\) are the same, the reading of the ammeter is:
1. \(10\) A
2. \(10\sqrt{2}\) A
3. \(5\) A
4. \(20\) A
What is the value of refractive index at the first medium in the diagram shown below?
1. 1.5
2. 1.6
3. 1.4
4. 1.8
Which of the following is not correct about the nature of the image of the object O formed by the mirror in the diagram?
1. Image is inverted.
2. Image is enlarged.
3. Image is virtual.
4. Image beyond the centre of curvature.
Angular magnification in normal adjustment is 39 for an astronomical telescope. The focal length of eye piece is 5 cm. The distance between the objective and eye piece is
1. 205 cm
2. 200 cm
3. 195 cm
4. 180 cm
In two positions of the lens in displacement method, the heights of the image of an object are 9 mm and 16 mm. The height of the object is
1. 25 mm
2. 12 mm
3. 10 mm
4. 5 mm
Which compound cannot decolourise KMnO4 solution?
1. CO3-2
2. NO2-
3. S2-
4. Cl-
Match the Column-I with Column-II :-
(A) Poling (P) Titanium
(B) Cupellation (Q) Copper
(C) Electrorefining (R) Silver
(D) Van Arkel Method (S) Tin
1. A P, B R, C S, D Q
2. A R, B S, C Q, D P
3. A Q, B R, C S, D P
4. A S, B Q, C R, D P
Which of the following statement is not correct?
1. All bonds in are equal but not in .
2. All bonds in are equal but not in .
3. bond length in is smaller than bond length in .
4. bond length in and bond length in are equal.
Which of the following atom has the highest first ionisation energy?
1. Rb
2. Na
3. K
4. Sc
The complete combustion region of Bunsen flame in the figure below is :-
1. region-1
2. region-2
3. region-3
4. region-4
Paramagnetic oxides are :-
1.
2.
3.
4.
Which of the following can exhibit geometrical isomerism?
1.
2.
3.
4.
4f14 configuration is observed in :-
1. Dy & Pm
2. Ln & La
3. Yb & Lu
4. Tm & Lu
LiF and CsI less soluble in water. What factors are responsible for this?
1. LiF : High ionic character
CsI : High covalent character
2. LiF : Strong reductant
CsI : Strong reductant
3. LiF : High Hydration
CsI : Less Hydration
4. LiF : High Lattice energy
CsI : Low hydration energy
Oxyacid with maximum P–H bond is :-
1. hypophosphorous acid.
2. cyclotrimeta phosphoric acid.
3. hypophosphoric acid.
4. orthophosphorous acid.
Ozone can be quantitatively estimated by :-
1.
2.
3.
4.
In Ion, number of bond pairs and lone pairs of electrons on nitrogen atom are :-
1. 2, 2
2. 3, 1
3. 1, 3
4. 4, 0
Select the incorrect statement about the complex formed in the brown ring test for nitrates is ?
1. Colour change in due to charge transfer.
2. It has iron in +1 oxidation state & nitrosyl as NO+.
3. It has magnetic moment of 3.87 B.M. confirming three unpaired electrons in Fe.
4. Colour change is due to d-d-transition.
Predict the product of reaction of I2 with H2O2 in basic medium.
1.
2.
3.
4.
Choose the wrong set :-
1. XeOF2 – ‘T’ shape –sp3d
2. XeOF4 – Square pyramidal –sp3d2
3. XeO2F2 = distorted trigonal bipyramidal –sp3d
4. XeO3F2= octahedral –sp3d
Which of the following is most acidic?
1.
2.
3.
4.
1.
2.
3.
4.
Hyperconjugation is not observed in :-
1.
2.
3.
4.
Which is least reactive towards grignard’s reagent.
1.
2.
3.
4.
Which of the following is not formed as product :-
1.
2.
3.
4.
Which of the following is reducing sugar?
1. Sucrose.
2. Lactose.
3. Starch.
4. Cellulose.
Which is condensation homopolymer?
1. Terylene.
2. Buna-S.
3. PAN.
4. Nylon-6.
Number of isomeric phenols possible with the molecular formula is :-
1. 3
2. 4
3. 5
4. 6
1.
2.
3.
4. Both 2 and 3
Z is
1.
2.
3.
4.
Which of the following statement is/are correct regarding to basic strength of pyridine?
1. Pyridine is more basic than aniline because it has localised lone pair of electron but aniline has delocalising lone pair of electron.
2. Pyridine is less basic than pyrrole because latter is aromatic.
3. Pyridine is more basic than all of other alkyl amine because it is aromatic.
4. All of these.
Which of the following can show haloform reaction?
1.
2.
3.
4.
Which characteristic feature is required for a substrate to undergo haloform reaction when treated with halogen and alkali?
1. All the aldehyde excepting formaldehyde have acidic -H, thus undergoes haloform reaction.
2. All the carbonyl compounds and alcohols which oxidised to ketone undergoes haloform reaction.
3. All the carbonyl compounds and alcohols which oxidised to aldehyde or ketone undergoes
haloform reaction.
4. All the carbonyl compound having 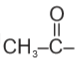
compound having methyl ketonic group undergoes haloform reaction.
IUPAC name of given compound is :-
1. But-1-en-4-ol
2. But-4-en-1-ol
3. But-3-en-1-ol
4. Pent-4-en-1-ol
The necessary and sufficient condition for a compound to show optical activity is :-
1. It should having one or more chiral center.
2. The molecules should be chiral.
3. It should have more than one chiral center along with a plane of symmetry within.
4. The compound may or may not having chiral center but having symmetry.
An atomic solid A crystallizes in a body centre lattice and the inner surface of the atoms at the adjacent corner are separated by 60.3 pm. If the atomic weight of A is 48, then density of the solid is nearly :-
1. 2.7 g/cc
2. 5.07 g/cc
3. 3.5 g/cc
4. 1.75 g/cc
When an atom is placed in a magnetic field the possible number of orientations for orbitals of azimuthal quantum number 3 is:-
1. 3
2. 2
3. 5
4. 7
In an adsorption experiment, a graph between log(x/m) versus log P was found to be linear with a slope of 45. The intercept on the y-axis was found to be 0.3.
What will be the value of x/m at a pressure of 3 atm for the Freundlich adsorption isotherm when 1/n remains constant?
[Antilog 0.3 = 2]
1. 2
2. 4
3. 6
4. 5
For the reaction initially concentrations of a and b are equal and at equilibrium the concentration of d will be twice of that of a. What will be the equilibrium constant for reaction :-
1. 2
2. 9
3. 4
4. 3
Which is correct for catalyst?
1. A catalyst can initiate a reaction.
2. An inhibitor can decrease the activation energy.
3. A catalyst can't change the enthalpy of reaction.
4. A catalyst can increases the rate of forward reaction and decreases that of the backward reaction [For a reversible reaction].
Solubility product of radium sulphate is . What will be the solubility of in 0.1 M :-
1.
2.
3.
4.
The edge length of NaCl unit cell is 508 pm. If radius of cation is 110 pm. The radius of anion is :-
1. 110 pm
2. 220 pm
3. 285 pm
4. 144 pm
converts into and according to following equation :-
x% of is converted into product. Rate of diffusion of gaseous mixture is double than that of uner identical conditions. Calculate x.
1. 98.23%
2. 31.25%
3. 25.27%
4. 6.25%
Half-life period of a first-order reaction is 10 minute, what % of the reaction will be completed in 100 minute:-
1. 25
2. 50
3. 99.9
4. 75
Which of the following is not a correct match?
1. Butter o/w type emulsion.
2. Vanishing cream o/w type emulsion.
3. Milk o/w type emulsion.
4. Cream w/o type emulsion.
Classification of organisms on the basis of gene sequencing is
1. Cytotaxonomy
2. Karyotaxonomy
3. Chemotaxonomy
4. Classical taxonomy
Asexual spored in brown algae are
1. Non-motile sporangiospore.
2. Biflagellated zoospores with two equal flagella.
3. Quadriflagellated zoospores with thick cell walls.
4. Biflagellated zoospores with two unequal lateral flagella.
Select the group of animals belonging to different phyla?
| 1. | Hirudinaria, Limulus, Laccifer. |
| 2. | Nereis, Dentalium, Aplysia. |
| 3. | Pheretima, Chaetopleura, Ophiura. |
| 4. | Cucumaria, Antedon, Loligo. |
For a reaction . The value of is -30 kJ/mol and is -100 J/mol. At what temperature, the reaction will be at equilibrium :-
1.
2.
3.
4.
A solution of a non-volatile solute in water has a boiling point of 375.3 K. The vapour pressure of water above this solution at 338 K is :- [Given (water)=0.2467 atm at 338 K and for water=0.52 K kg mol-1]
1. 0.18 atm
2. 0.23 atm
3. 0.34 atm
4. 0.42 atm
Which of the following changes decrease the vapour pressure of water kept in a sealed vessel?
1. Decreasing the quantity of water.
2. Increasing the quantity of water.
3. Decreasing the volume of the vessel to one-half.
4. Decreasing the temperature of water.
for the reaction is
1. 11.44 kJ
2. -180.8 kJ
3. -150.5 kJ
4. 28.5 kJ
At 20C, the vapour pressure of 0.1 M solution of urea is 0.0311 mm less than that of water and the vapour pressure of 0.1 M solution of KCl is 0.0574 mm less than that of water. The apparent degree of dissociation of KCl at this dilution is :-
1. 92.1%
2. 84.6%
3. 68.4%
4. 54.1%
Facilitated transport differs from active transport as the former
1. Transports saturates.
2. Response to protein inhibitors.
3. Requires special membrane proteins.
4. Shows downhill transport.
Initial carboxylation reaction in and plants occurs respectively in
plants plants
1. Mesophyll cells - Mesophyll cells.
2. Mesophyll cells - Bundle sheath cells.
3. Bundle sheath cells - Mesophyll cells.
4. Bundle sheath cells - Bundle sheath cells.
Identify the mismatched pair with respect to respiratory quotient (RQ)
| Substrates | R.Q. | ||
| 1. | Carbohydrates | 1 | |
| 2. | Oxalic acid | 4 | |
| 3. | Proteins | >1 | |
| 4. | Malic acid | 1.33 | |
Consider the following statements and select the option that correctly fill in the blanks.
| A. | _________ is used to induce the dormancy of buds and storage organs. |
| B. | _________ speed up the malting process in brewing industry. |
| C. | Senescence is prevented by __________. |
| A | B | C | |
| 1. | Gibberellin | Cytokinin | GAs |
| 2. | Ethylene | ABA | Cytokinin |
| 3. | Cytokinin | Auxins | ABA |
| 4. | ABA | GAs | Cytokinin |
Identify the incorrect statement with respect to lac operon.
1. Regulation of lac operon by repressor is referred to as negative control.
2. Promote gene possesses site for RNA polymerase attachment.
3. Lac z gene codes for -galactosidase.
4. Regulator gene interacts with protein molecule which prevents the transcription of structural genes.
Mark the incorrect statement with respect to semi-autonomous organelles.
| 1. | They are capable of self duplication. |
| 2. | They have circular ds DNA. |
| 3. | They can synthesize some of their proteins. |
| 4. | They have low GC content. |
DNA having labelled thymidine is allowed to replicate in medium having non-radioactive thymidine. After three duplications, the DNA having labelled thymidine shall be
1. one molecule
2. two molecules
3. four molecules
4. eight molecules
Select the odd one with respect to redifferentiated tissues.
1. Cork cambium
2. Secondary phloem
3. Secondary xylem
4. Secondary cortex
Assertion : Dragonflies are useful to get rid of mosquitoes
Reason : Baculoviruses are pathogens that attack insects and other arthropods.
Which of the following statements is incorrect?
| 1. | Microfilaments are long, thin and branch structures of dynein protein. |
| 2. | 80S ribosomes are attached to RER through ribophorins. |
| 3. | Chloroplasts and mitochondria are believed to be bacterial endosymbionts. |
| 4. | Fungal cell wall is made up of polymer of N-acetyl glucosamine units. |
Growth and reproduction are mutually exclusive events in
1. Bacteria and protista.
2. Protista and unicellular fungi.
3. Yeast and Paramoecium.
4. Higher plants and animals.
Initiation of the assembly of mitotic spindle and the complete disintegration of the nuclear envelope can be observed in respectively
| 1. | Prophase and anaphase I. |
| 2. | Metaphase and telophase. |
| 3. | Prophase and metaphase. |
| 4. | Metaphase and metaphase-I. |
Which of the following is not correct for slime moulds?
1. Somatic phase is plasmodium.
2. They are saprophytic.
3. Spore is with wall.
4. They are pathogenic.
Which of the following pair of plants reproduce by sub-aerial vegetative propagules?
1. Potato and strawberry.
2. Pineapple and strawberry.
3. Adiantum and Agave.
4. Lily and garlic.
The total energy available to the third trophic level organisms if ecosystem is supported by 10 lac J light energy
1. 10 J
2. 100 J
3. 1000 J
4. 10,000 J
Natural ageing of a lake by biological enrichment of its water is called
1. Biomagnification
2. Eutrophication
3. Algal blooms
4. Toxification
Catalytic converter of automobiles converts unburnt hydrocarbons into
1.
2.
3.
4.
Pioneer communities in ecological succession are
(i) Phytoplanktons (ii) Scrubs
(iii) Floating plants (iv) Lichens
(v) Herbs
1. (i), (ii), (iii), (iv)
2. (i), (iv)
3. (i), (iv), (v)
4. (i), (ii), (v)
Which one of the following trait is only expressed in the presence of identical allele?
1. Yellow seed
2. Inflated pod
3. Green pod
4. Green seed
Blood-cholesterol lowering agent is and produced by
1. Statins, Monascus purpureus
2. Statins, Trichoderma polysporum
3. Cyclosporin A, Streptococcus
4. Streptokinase, Streptococcus
Which one of the following is incorrect statement?
| 1. | Phosphoenol pyruvic acid is most efficient CO2 acceptor in C4 plants. |
| 2. | Leaves of C4 plants show kranz anatomy. |
| 3. | In CAM plants, acidification (formation of malic acid) occurs in presence of light. |
| 4. | Chlorophyll consists of a porphyrin head & a phytol tail. |
Select the correct match with respect to mineral as activator/components of enzymes.
a. Nitrogenase (i) Zn
b. PEPcase (ii) Fe
c. Alcohol dehydrogenase (iii) Mo
d. Catalase (iv) Mg
1. a (iii), b (iv), c (i), d (ii)
2. a (iii), b (iv), c (ii), d (i)
3. a (iv), b (iii), c (i), d (ii)
4. a (iii), b (ii), c (i), d (iv)
DNA replication is semiconservative because
1. Daughter DNA molecules have both parental strands.
2. Daughter DNA molecules have one parental and one newly synthesized strand.
3. Daughter DNA molecules have both newly synthesized strands.
4. Both 1 & 3.
Bacterial cells containing F-plasmid
1. Are designated as F+ cells.
2. Functions as DNA donor during conjugation.
3. Function as DNA recipients during conjugation.
4. Both 1 & 2.
Oxygen evolving photoautotrophic monerans are
1. Cyanobacteria
2. Actinomycetes
3. Archaea
4. Photolithotrophs
On analyzing the number of different samples of DNA which, result would be constant with the base pairing rules?
1. A = G
2. A = C
3. A G = C T
4. G = T
Which of the following statements is correct?
1. In Fabaceae, stamens show tetradynamous condition.
2. Superior ovary with oblique septa are found in members of Solanaceae.
3. Gynoecium is tricarpellalry and apocarpous in members of Liliaceae.
4. In Smilax, root tip is tendrillar.
How many plants in the list given below have basal placentation?
Pea, China rose, Tomato, Sunflower, Wheat, Bean, Argemone, Marigold, Mustard, Lemon, Maize
1. Eight
2. Four
3. Two
4. Six
Which of the following is absent in monocot stem?
| 1. | Ground tissue and hypodermis. |
| 2. | Phloem parenchyma and water containing cavities. |
| 3. | Endodermis and phloem parenchyma. |
| 4. | Bundle sheath and pericycle. |
Which of the following is correct statement?
1. Tyloses are outgrowths of vessels in xylem fibres.
2. Complementary cells are thin-walled sclerenchyma cells produced in bark.
3. Tyloses are produced when sapwood is converted to heartwood.
4. Vessels are present only in latewood.
If a colourblind woman marries a man who has normal colour vision, which of the following is possible with respect to their children?
1. All sons and daughters are colourblind.
2. All sons and daughters are normal.
3. 100% sons are colourblind.
4. 25% daughters are colourblind.
Continuous source of variations in sexually reproducing organisms is
1. Disjunction of chromosomes.
2. Crossing over between sister chromatids.
3. Crossing over between non-sister chromatids.
4. Mutation.
In maize, which of the following leads to resistance to maize stem borers?
1. High aspartic acid and low sugar content.
2. Solid stem and smooth leaves.
3. Nectarless flowers.
4. Solid stem and high sugar contents.
Genetically identical plants produced by micropropagation are called
1. Explants
2. Somaclones
3. Somatic hybrids
4. Cybrids
During secondary treatment, major part of activated sludge is pumped into tanks called
1. Aerobic sludge digesters.
2. Anaerobic sludge digesters.
3. Primary sludge digesters.
4. Settling tanks.
Population density will increase if
1. Number of births and number of deaths increase.
2. Number of births and number of immigrants is high.
3. Number of immigrants and number of deaths is high.
4. Number of births and number of immigrants is low.
The number of linkage groups in pea is
1. 14
2. 7
3. 9
4. 24
How many type(s) of gametes is/are produced by a double recessive parent?
1. One
2. Two
3. Four
4. Six
Painful skeletal deformity due to cadmium is
1. Minimata
2. Uremia
3. Itai-itai
4. Black foot disease
Hotspots are the area of
1. High endemism.
2. High level of species richness.
3. High noise pollution.
4. Both 1 and 2.
Isoclinic lines on a magnetic map are formed by joining the points where
1. Dip angle is same.
2. Declination is same.
3. Vertical component of earth’s magnetic field is the same.
4. Horizontal component of earth’s magnetic field is the same.
At room temperature (27°C) magnetic susceptibility of a paramagnetic substance is 0.004. At 600 K, the value of the magnetic susceptibility will become/remains
1. 0.004
2. 0.002
3. 0.008
4. Zero
If the threshold wavelength for a photosensitive plate is \(4000\) Å, what will be the value of saturation current if radiation of wavelength \(5000\) Å is incident?
1. zero
2. \(1\) mA
3. \(3\) mA
4. data is insufficient to predict
The de Broglie wavelength of -particle is and that of proton is '. If the two particles are accelerated through same potential , the ratio will be
1.
2.
3.
4.
If potential energy in first excited state in H-atom is taken to be zero then kinetic energy in this orbit will be
1. 3.4 eV
2. 6.8 eV
3. 10.2 eV
4. 13.6 eV
Similarity between protochordata and hemichordata is
| 1. | Presence of notochord. |
| 2. | Exclusively marine. |
| 3. | Dorsal hollow and single central nervous system. |
| 4. | Exclusively parasitic. |
According to the discovery made in 1980's, RNA can act like enzyme to assemble new RNA molecules in RNA template. Which of the following statements is not proved by this theory?
| 1. | Coacervates may not have been the first step in the evolution of life. |
| 2. | Perhaps the first macromolecule was RNA. |
| 3. | Coacervates were the basis for the first cell. |
| 4. | After the formation of RNA, the stability of molecule proved by surrounding RNA within a coacervate. |
Mark the incorrect match related to the cockroach:
| 1. | Gizzard | → | Grinding the food particle. |
| 2. | Malpighian tubule | → | Absorb uric acid from haemolymph and excreted out through the hindgut. |
| 3. | Tegmina | → | Opaque, dark and leathery structure and cover the hind wing when at rest. |
| 4. | Collateral gland | → | Formation of ootheca after fertilization in the female genital pouch. |
Nucleotide is made up of
1. Heterocyclic compound, Monosaccharide, Phosphoric acid.
2. Nitrogenous base, hexose sugar, phosphate.
3. Heterocyclic compound and pentose sugar only.
4. Heterocyclic compound and nucleoside.
NAD and NADP are consider as
| 1. | Apoenzyme and cofactor respectively. |
| 2. | Coenzyme and co-factor respectively. |
| 3. | Both as co-enzyme. |
| 4. | Apoenzyme and holoenzyme respectively. |
Adipose tissue and areolar tissue are consider as
| 1. | Loose connective tissue and dense connective tissue respectively. |
| 2. | Dense regular and dense irregular tissue respectively. |
| 3. | Both are specialised connective tissue. |
| 4. | Both are loose connective tissue. |
| Assertion (A): | Compound epithelium is always made up of more than one layer of cells. |
| Reason (R): | Its main function is to provide protection against chemical and mechanical stresses. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true, but (R) is not the correct explanation of (A). |
| 3. | (A) is true, but (R)) is false. |
| 4. | Both (A) and (R) are false. |
Common bile duct open into
1. Gall bladder.
2. Duodenum.
3. Hepato-pancreatic duct.
4. Jejunum.
Mucus neck cell is a part of
1. Gastric gland.
2. Intestinal gland.
3. Salivary gland.
4. Both 1 and 2.
According to Bohr's effect
1. Hemoglobin oxygen binding affinity is inversely related to acidity.
2. Hemoglobin oxygen binding affinity is inversely related to concentration of .
3. Hemoglobin oxygen binding affinity is directly related to acidity.
4. Both 1 and 2.
Chloride shift occurs when
1. Blood gives up oxygen and receives from tissues.
2. Blood receive oxygen from lungs.
3. When the hemoglobin saturation is minimum in the blood.
4. When the blood pH is maximum.
The evolutionary process giving rise to new species adapted to new habitats and ways of life is called adaptive radiation. For example,
1. Darwin's finches in Galapagos Islands.
2. Australian marsupials that radiated to form new species.
3. Wolf and Tasmanian wolf.
4. Both 1 and 2.
'Erythroblastosis Foetails' occurs when:
I. Mother is Rh+ and foetus is Rh-.
II. Mother is Rh- and foetus is Rh+.
III. Mother and foetus both are Rh+.
IV. Mother and foetus both are Rh-.
1. I & II
2. Only I
3. Only II
4. II, III & IV
In ECG the ventricular contraction occurs
1. Just after P wave and before Q wave.
2. Just after Q wave but before T wave.
3. In between S-T segment.
4. After the end of T wave.
Which of the following can not be expected in the filtrate. When it arrives to descending limb of Henle's loop?
I. Albumin II. Glucose III. Urea IV. Amino acid
1. I, II, III & IV
2. I only
3. II, IV only
4. I, II & IV
Gill clefts and notochord appear in the embryonic development of all vertebrates from fishes to mammals. This supports the theory of
1. Recapitulation
2. Metamorphosis
3. Biogenesis
4. Abiogenesis
Blood present in afferent arteriole and efferent arteriole will differ in
I. Amount of protein.
II. Number if blood cells.
III. Amount of glucose.
IV. Amount of urea.
1. I, II, III & IV
2. III & IV only
3. IV only
4. III only
Cardiac muscles can be best defined as:
| 1. | Many cardiac muscle cells assemble in a branching pattern. |
| 2. | Single cardiac muscle fibre enclosed by connective tissue layer. |
| 3. | Group of multinucleated muscle fibres enclosed by connective tissue layer. |
| 4. | Group of unstriated muscle fibres enclosed by endocardium. |
Which of the following is required for the breaking of cross-bridge during muscle contraction?
1. ATP and Ca++
2. ADP and Ca++
3. Only Ca++
4. ATP only
Zygomatic and sphenoid bone is an example of
1. Cranial bones.
2. Cranial bone and facial bone respectively.
3. Facial bone and cranial bone respectively.
4. Facial bone.
Mark the incorrect match?
| 1. | Cerebral aqueduct | A canal passes through the midbrain, connecting 3rd ventricle to 4th ventricle of the brain. | |
| 2. | Corpus callosum | Thick band of nerve fibres that divides the cerebral cortex lobes into left and right hemispheres. | |
| 3. | Limbic system | The inner part of forebrain involved in the regulation of sexual behaviour. | |
| 4. | Pons | Consist of fibre tracts that interconnect different regions of the brain. |
The vestibular apparatus is composed of:
I. Semi-circular canals
II. Otolith organs
III. Organ of Corti
IV. Crista and macula
1. I, II, III & IV
2. I, II only
3. IV only
4. II only
'Retinal' is present in:
1. Rods
2. Cones
3. Both 1 and 2
4. All the retinal layer
Low intelligence quotient abnormal skin and deafmutism are related to:
1. Low secretion of growth hormone
2. Hypothyroidism
3. Hyperparathyroidism
4. Hyposecretion of adrenal cortex hormone
Extracellular receptors must be required for the action of:
I. Oxytocin
II. Thyroxine
III. Epinephrine
IV. Glucagon
1. I, II, III & IV
2. I, IV only
3. I, III, IV
4. III & IV only
Mark the correct statement for pBR322 as a plasmid vector
| I: | Contains relaxed origin of replication. |
| II: | Two genes coding for resistance to antibiotics have been introduced. |
| III: | There are single recognition sites for a number of restriction enzymes at various points. |
| IV: | Insertional inactivation is a useful selection method for identifying recombination vectors with insert. |
1. I, II, III & IV
2. I, II only
3. II, III only
4. II only
If two enzymes A and B are used for restriction mapping of 9 kb molecule, cleavage with A gives fragments of 2 and 7 kb and B gives fragments and 3 and 6 kb. If A and B cut at opposite end of molecule what will be the length of fragment after double digestion?
1. 2 kb, 3 kb, 4 kb.
2. 1 kb, 2 kb, 6 kb.
3. 3 kb, 3 kb, 3 kb.
4. 2 kb, 2 kb, 5 kb.
Which of the following is very effective sedative and pain killer?
1. Cocaine.
2. Heroin.
3. Morphine.
4. Cannabis extract.
Cellular oncogenes can be best explained as
1. Mutated gene.
2. Present in normal cells.
3. Present only in the cells of benign tumour.
4. Genes due to which cells loose the property of contact inhibition.
___A___ and ___B___ provide micro-environments for the development and maturatiion of T-lymphocytes. Here A and B is :-
1. Spleen and thymus gland.
2. Thymus gland and MALT.
3. Bone marrow and thymus gland.
4. Spleen and MALT.
Recombination DNA technology has allowed the production of __A__ of pathogen in bacteria or yeast. Here A is :-
| 1. | Pro-toxin. |
| 2. | Anti-bodies. |
| 3. | Antigenic polypeptide. |
| 4. | Antigenic polysaccharide. |
Vaccines are available for the disease like
I. Polio.
II. Diphtheria.
III. Pneumonia.
IV. Tetanus.
1. I, II, III & IV
2. I and IV only
3. I only
4. I, II & IV only
Odd one out?
1. PMNL-neutrophils.
2. Monocytes.
3. Natural killer cells.
4. Interferons.
Gene pool of a population tends to remain stable if the population is large, without large-scale mutations, without migration and with
1. Random mating.
2. Moderate environmental changes.
3. Natural selection.
4. Reduction in predators.
There are two opposing views about the original of modern man. According to one view, Homo erectus in Asia were the ancestors of modern man. A study of variation in DNA, however, suggested African origin of modern man. What kind of observation of DNA variation could suggest this?
1. Greater variation in Asia than in Africa.
2. Greater variation in Africa than in Asia.
3. Similar variation in Africa and Asia.
3. Variation only in Asia and no variation in Africa.
Complexes formed during immune complex mediated hypersensitivity are removed by
1. Eosinophils and T-cells.
2. Monocytes and B-cells.
3. Eosinophils and monocytes.
4. Eosinophils and basophils.
At which stage of HIV infection does one usually show symptoms of AIDS?
1. When the infecting retrovirus enters host cells.
2. When viral DNA is produced by reverse transcriptase.
3. When HIV replicates rapidly in helper T-lymphocytes and damages large number of these.
4. Within 15 days of sexual contact with an infected person.
How does a bacterial cell protect its own DNA from restriction enzymes?
1. By adding methyl groups to adenines and cytosines.
2. By reinforcing bacterial DNA structure with covalent phosphodiester bonds.
3. By adding histones to protect the double-stranded DNA.
4. By forming "sticky ends" of bacterial DNA to prevent the enzyme from attaching.
DNA fragments separated by gel electrophoresis are shown. Mark the correct statement:
1. Band 3 contains more positively charged DNA molecules than bond 1.
2. Band 3 indicates more charge density than bands 1 and 2.
3. Band 1 has longer DNA fragment than bands 2 and 3.
4. All bands have equal length and charges but differ in base composition.
Level of estrogen and progesterone are minimum at the time of
1. Follicular phase.
2. Ovulation.
3. Secretory phase.
4. Onset of menstrual phase.
Fertilization membrane is formed to
1. Faciliate entry of sperm into egg.
2. Provide stability to egg.
3. Prevent ectopic pregnancy.
4. Prevent polyspermy.
Seminal plasma of humans is rich in
1. Fructose and certain enzymes, poor in .
2. Fructose, and certain enzymes.
3. Fructose, but no enzymes.
4. Glucose, certain enzymes but no .
Which one of the following is not the function of placenta?
1. Facilitates removal of carbon dioxide and waste material from embryo.
2. Secrete oxytocin during parturition.
3. Facilitates supply of oxygen and nutrients to embryo.
4. Secretes estrogen.
Diaphragms, cervical caps and vaults prevent conception by
| 1. | Increasing phagocytosis of sperms within uterus. |
| 2. | Suppressing sperm motility. |
| 3. | Inhibiting ovulation and implantation. |
| 4. | Blocking the entry of sperms through the cervix. |
How many meiotic divisions are required to produce 480 seeds?
1. 479
2. 600
3. 70
4. 650
Identify the statements that are true for anemophilous flowers
a. Pollen grains are light and non-sticky.
b. They possess well exposed stamens.
c. They have large coloured flowers.
d. They have single ovule in each ovary.
e. Flowers produce nectar and scent.
1. a, b and e
2. a, b and d
3. b, c and e
4. c, d and e
Both chasmogamous and cleistogamous flowers, are produced by
1. Pea and Zostera.
2. Bean and Vallisneria.
3. Maize and bamboo.
4. Oxalis and Commelina.
When current flows in steady state in the wire whose area of cross-section decreases as shown. Which of the following is incorrect?
1. Drift speed increases in the direction of current.
2. Electric field decreases in the direction of current.
3. Electric Potential decreases in the direction of current.
4. Current density increases in the direction of current.
For what value of the resistance X, the reading of ammeter will be zero?
1.
2.
3.
4.
What is the ratio of the readings of the voltmeter, V1 and V2 in the network shown below?
1. 1 : 2
2. 2 : 3
3. 3 : 1
4. 1 : 1
A wire of length l and carrying current i is folded to form double circular loop. The magnetic field at the centre is
1.
2.
3.
4.
A proton and an -particle enter with same momentum in a transverse uniform magnetic field. The ratio of the radii of curvature of their circular path will be
1. 1 : 2
2. 2 : 1
3. 3 : 2
4. 1 : 1