Botany - Section A
1. Consider the given two statements:
| I: |
Meiosis is responsible for maintaining the chromosome number across generations in sexually reproducing organisms. |
| II: |
Meiosis results in reduction of chromosome number by half in the daughter cells. |
| 1. |
Both I and II are correct and II explains I |
| 2. |
Both I and II are correct but II does not explain I |
| 3. |
I is correct but II is incorrect |
| 4. |
I is incorrect but II is correct |
2. Which of the following statement regarding the characteristic and defining features of life is true?
| 1. |
When it comes to multicellular organisms, reproduction is synonymous with growth |
| 2. |
Isolated metabolic reactions in vivo are not living things but living reactions |
| 3. |
Cellular organisation and consciousness are the defining features of living organism |
| 4. |
In majority of the higher animals and plants, growth and reproduction are mutually inclusive events |
3. Identify the incorrect statement:
| 1. |
In dicot root, initiation of lateral roots and vascular cambium during the secondary growth takes place in the pericycle |
| 2. |
In monocot roots xylem bundles are usually more than six [polyarch] |
| 3. |
In dicot stems vascular bundles are arranged in a ring |
| 4. |
Monocot stem has a parenchymatous hypodermis |
4. Which of the following algae exhibits the life cycle as shown in the diagram below?
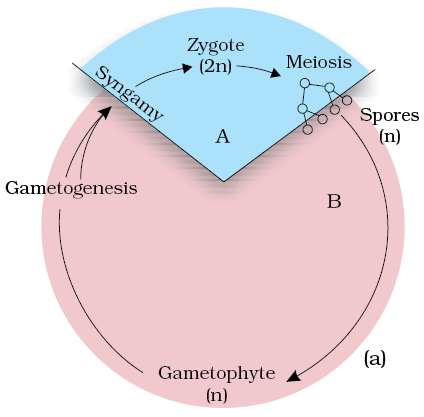
1.
Fucus
2.
Spirogyra
3.
Ectocarpus
4.
Polysiphonia
5. Which of the following amino acids is not an aromatic amino acid?
| 1. |
Tyrosine |
2. |
Tryptophan |
| 3. |
Threonine |
4. |
Phenylalanine |
6. What is incorrect regarding DNA replication?
| 1. |
For a cell to divide, it must first replicate its DNA. |
| 2. |
DNA replication is an all-or-none process; once replication begins, it proceeds to completion. |
| 3. |
DNA replication (DNA amplification) cannot be performed in vitro (artificially, outside a cell). |
| 4. |
Once replication is complete, it does not occur again in the same cell cycle. |
7. The female gametophyte in the angiosperms is:
| 1. |
7 celled and 8 nucleate |
| 2. |
8 celled and 7 nucleate |
| 3. |
7 celled and 7 nucleate |
| 4. |
8 celled and 8 nucleate |
8. The number of species known and described of algae is more than that of all the following except:
| 1. |
Ferns |
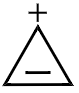
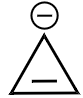
2. |
Lichens |
| 3. |
Mosses |
4. |
Fungi |
9. Which one of the following is essential for the process of imbibition to take place?
| I: |
Water potential gradient between the adsorbent and the liquid imbibed |
| II: |
Affinity between the absorbent and the liquid |
| III: |
Affinity between the adsorbent and the liquid |
| IV: |
Water potential gradient between the absorbent and the liquid imbibed |
| 1. |
Only III and IV |
2. |
Only I and II |
| 3. |
Only I and III |
4. |
Only II and IV |
10. In the mitochondrial electron transport system, ubiquinone receives reducing equivalents via:
1. Both Complex I and Complex II
2. Complex III
3. Complex IV
4. Complex V
11. Consider the two statements:
| I: |
The water-splitting complex is located on the inner side of the thylakoid membrane |
| II: |
The NADP reductase enzyme is located on the stromal side of the thylakoid membrane |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
12. Identify the correct statement:
| 1. |
Unlike facilitated transport, simple diffusion requires special membrane proteins |
| 2. |
Both simple diffusion and facilitated transport are highly selective |
| 3. |
Facilitated transport is an uphill transport and requires ATP energy |
| 4. |
Both facilitated transport and active transport are liable to saturate |
13. The following reaction will be catalyzed by an enzyme classified as a/an:
A–B + H2O → A–OH + B–H
| 1. |
Oxidoreductase |
2. |
Hydrolase |
| 3. |
Lyase |
4. |
Transferase |
14. Which essential element required by plants is used in the synthesis of cell wall, formation of mitotic spindle and accumulates in older leaves?
1. Phosphorus
2. Magnesium
3. Calcium
4. Iron
15. The number and linear sequence of amino acids in a protein are considered as the:
1. primary structure of a protein
2. secondary structure of a protein
3. tertiary structure of a protein
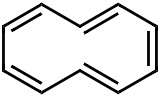
4. quaternary structure of a protein
16. Identify the incorrectly matched pair:
| 1. |
Brown rust of wheat |
Fungus |
| 2. |
Black rot of crucifers |
Bacterium |
| 3. |
Tobacco mosaic |
Virus |
| 4. |
Root knot of tobacco |
Annelid |
17. Specialized plastids like amyloplasts are differentiated forms of:
1. Chloroplasts
2. Chromoplasts
3. Leucoplasts
4. Gerontoplasts
18. In brinjal, stamens are:
1. Epipetalous
2. Epiphyllous
3. Monoadelphous
4. Six in number
19. In ‘double fertilization’ in angiosperms:
| I: |
Syngamy results in the formation of zygote. |
| II: |
Triple fusion results in the formation of the primary endosperm nucleus. |
| 1. |
Only I is correct. |
| 2. |
Only II is correct. |
| 3. |
Both I and II are correct. |
| 4. |
Both I and II are incorrect. |
20. Cattle and goats are not seen browsing on
Calotropis growing in abandoned fields because:
| 1. |
the plant has a large number of thorns. |
| 2. |
the plant produces toxic cardiac glycosides. |
| 3. |
the plant leaves secrete a lot of distasteful mucilage. |
| 4. |
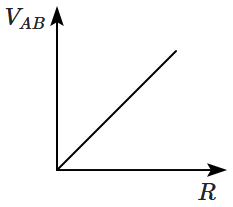
the plant produces secondary metabolite strychnine. |
21. If the margins of sepals or petals overlap one another but not in any particular direction, the type of aestivation is called:
1. Valvate and is seen in Calotropis
2. Twisted and is seen in Lady’s finger
3. Imbricate and is seen in Cassia
4. Vexillary and is seen in pea
22. Meiosis in sexually reproducing organisms creates new variations by shuffling the available genetic material and leads to the formation of a recombinant DNA which is created at:
| 1. |
Pachytene of Prophase I |
| 2. |
Diplotene of Prophase I |
| 3. |
Anaphase I of Meiosis I |
| 4. |
Anaphase II of Meiosis II |
23. The algae shown in the diagram below belongs to the class which has:

1. 2-8 equal and apical flagella
2. 2 unequal and lateral flagella
3. 2 unequal and apical flagella
4. 2-8 equal and lateral flagella
24. Which element is an activator for the enzymes RuBisCo and PEP carboxylase?
1. Zinc
2. Magnesium
3. Potassium
4. Molybdenum
25. RNA interference:
| I: |
involves silencing of a specific mRNA due to a complementary dsRNA molecule that binds to and prevents translation of the mRNA (silencing). |
| II: |
has been used to create transgenic tobacco plants resistant to nematode Meloidogyne incognita |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
26. Hyphae, that are continuous tubes filled with multinucleated cytoplasm, are seen in the members of:
1. Ascomycetes
2. Phycomycetes
3. Deuteromycetes
4. Basidiomycetes
27. Which of the following statements will be correct regarding restriction endonucleases used in constructing recombinant DNA molecules?
| 1. |
They are RNA molecules acting as enzymes. |
| 2. |
The enzymes used in such experiments cut dsDNA randomly at any site. |
| 3. |
They have been isolated from bacterial cells. |
| 4. |
All restriction enzymes are thermostable and hence can be used in PCR procedures. |
28. Consider the given two statements:
| I: |
Dicot roots do not undergo secondary growth. |
| II: |
In the dicot root, the vascular cambium is completely secondary in origin. |
| 1. |
Both I and II are correct and II explains I |
| 2. |
Both I and II are correct but II does not explain I |
| 3. |
I is correct but II is incorrect |
| 4. |
I is incorrect but II is correct |
29. A ‘continuous culture system’ method usually produces a larger biomass mainly because:
| 1. |
the environmental conditions for the growth of the microbes are very easily maintained in such a system |
| 2. |
such a system keeps most of the cells in their active/log exponential phase |
| 3. |
it is very cheap with respect to other systems and hence cost of production is low |
| 4. |
such a system uses larger sized bioreactors than other systems |
30. Consider the following statements about Mustard:
| I: |
Its ovary is one-chambered but becomes two-chambered due to formation of a false septum |
| II: |
There may be variation in the length of filaments within its flowers |
| III: |
Its flower is zygomorphic |
| IV: |
Its flowers are located at the rim of the thalamus almost at the same level |
| 1. |
Only I and II are correct |
| 2. |
All I, II, III and IV are correct |
| 3. |
Only I, III and IV are correct |
| 4. |
Only I, II and III are correct |
31. Xylem is associated with the translocation of:
| 1. |
a variety of organic and inorganic solutes |
| 2. |
water and a variety of organic and inorganic solutes |
| 3. |
water, mineral salts, some organic phosphorus and hormones |
| 4. |
water, mineral salts, some organic nitrogen and hormones |
32. The genetic code is:
| 1. |
triplet, degenerate, nearly universal and read in a contiguous fashion |
| 2. |
triplet, degenerate, nearly universal and read in an overlapping fashion |
| 3. |
doublet, degenerate, nearly universal and read in a contiguous fashion |
| 4. |
triplet, ambiguous, totally universal and read in a contiguous fashion |
33. An alga lacks flagellar insertions in its life cycle and stores food as floridean starch. This alga can be:
| 1. |
Chara |
2. |
Fucus |
| 3. |
Porphyra |
4. |
Ectocarpus |
34. Which part of the endomembranous system of a eukaryotic cell is rich in acid hydrolases?
1. Peroxisome
2. Lysosome
3. Vacuole
4. Golgi apparatus
35. Match the PGR in
Column-I with their chemical nature in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Auxin |
P. |
F. W. Went |
| B. |
Gibberellin |
Q. |
E. Kurosawa |
| C. |
Cytokinin |
R. |
F. Skoog and co-workers |
| D. |
Ethylene |
S. |
H. H. Cousins |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
Q |
R |
S |
P |
| 3. |
R |
S |
P |
Q |
| 4. |
S |
P |
Q |
R |
Botany - Section B
36. The Amazon Forest:
| I: |
is a largely tropical rainforest in South Africa |
| II: |
has the greatest biodiversity on Earth |
| III: |
produces about 20% of total oxygen in the earth’s atmosphere through photosynthesis |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
37. What would be true for the phosphorus cycle in an ecological ecosystem?
| 1. |
A considerable amount of phosphorus enters the biosphere through the primary producers. |
| 2. |
Fossil fuels represent the major reservoir of phosphorus. |
| 3. |
Phosphorus cycling occurs through the atmosphere, ocean, and through living and dead organisms. |
| 4. |
There is no respiratory release of phosphorus into the atmosphere. |
38. All living organisms - past, present and future are linked to each other. All organisms have arisen from a single distant ancestor. The strongest evidence for this will be:
| 1. |
All organisms have a triplet genetic code |
| 2. |
DNA is the genetic material of almost all living organisms |
| 3. |
The genetic code is degenerate but non-ambiguous |
| 4. |
The amino acids specified by particular triplets are almost always identical between any two organisms. |
39. Given below are two statements:
| Assertion (A): |
The reaction centres in both photosystems in plants is the same. |
| Reason (R): |
The reaction centres of both photosystems are formed by chlorophyll b molecules. |
| 1. |
Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. |
(A) is true but (R) is false. |
| 4. |
Both (A) and (R) are false. |
40. Identify the incorrect statement:
| 1. |
Mercury and DDT undergo biomagnifications in the terrestrial food chain. |
| 2. |
Nitrates and phosphates can cause rapid enrichment of nutrients in a lake. |
| 3. |
Recycling is the only solution for the treatment of E-waste. |
| 4. |
Without greenhouse effect the average temperature at surface of earth would have been minus 180C |
41. The numbers of FADH2, ATP, Carbon dioxide and NADH molecules produced in a single turn of citric acid cycle respectively are:
1. One, Two, Three and Four
2. One, One, Two and Three
3. One, Two, Two and Three
4. Two, One Two and Three
42. Microbodies in eukaryotic cells include all the following except:
| 1. |
Peroxisomes |
2. |
Glycosomes |
| 3. |
Lysosomes |
4. |
Glyoxysomes
|
43. Centromeres split leading to the separation of sister chromatids and the pulling of the separated sister chromatids to the opposite poles is seen in:
| I: |
Mitotic anaphase |
| II: |
Anaphase I |
| III: |
Anaphase II |
| 1. |
Only I |
2. |
Only I and II |
| 3. |
Only III |
4. |
Only I and III |
44. Identify the incorrect statement:
| 1. |
The process of separation and purification of expressed protein before marketing is called as downstream processing |
| 2. |
DNA precipitation out of a mixture of biomolecules can be achieved by treatment with a divalent cation such as calcium ion |
| 3. |
Stirred-tank bioreactors have been designed for availability of oxygen throughout the process |
| 4. |
Reporter genes can be used as markers for selecting successfully transformed cells |
45. Identify the incorrect statement:
| 1. |
All cellular life-forms use a primase to synthesize a short RNA primer with a free 3′ OH group which is subsequently elongated by a DNA polymerase. |
| 2. |
DNA is read by DNA polymerase in the 5′ to 3′ direction, meaning the new strand is synthesized in the 3' to 5' direction. |
| 3. |
The leading strand is the strand of new DNA that is synthesized in the same direction as the growing replication fork. |
| 4. |
The lagging strand is the strand of new DNA whose direction of synthesis is opposite to the direction of the growing replication fork. |
46. In most seeds, the ratio of which of the following pair of PGRs determines whether the embryo remains dormant or germinates?
| 1. | ABA and GA | 2. | Auxin and Cytokinin |
| 3. | Auxin and GA | 4. | ABA and Cytokinin |
47. In the cross-pollination experiments between red and white snapdragon plants, the resulting F1 offspring are all pink. What would be true?
| I: |
When the first filial (F1) generation consisting of all pink plants is allowed to self-pollinate, the resulting F2 generation phenotypic ratio is 1:2:1. |
| II: |
When the F1 generation is allowed to cross-pollinate with true breeding red plants, the resulting F2 phenotypic ratio is 1:1. |
| III: |
When the F1 generation is allowed to cross-pollinate with true breeding white plants, the resulting F2 plants' phenotypic ratio is 1:1. |
1. Only
I and
II
2. Only
I and
III
3. Only
II and
III
4.
I,
II and
III
48. When oxygen is not available to a cell, NADH formed during glycolysis:
| 1. |
does not undergo any change as there is no need for the cell to regenerate NAD+ |
| 2. |
passes electrons to the electron transport system |
| 3. |
passes hydrogen atoms to pyruvic acid |
| 4. |
passes electrons and hydrogen atoms to acetyl coA |
49. In primary hydrarch succession, the seral stage just before the climax forest is likely to be:
1. Submerged free floating plant stage
2. Scrub stage
3. Marsh meadow stage
4. Reed swamp stage
50. Plietesials are plants that grow for a number of years, flower gregariously (synchronously), set seeds, and then die. Such plants will include:
I. Neelakuranji
II. Bamboo
1. Only I
2. Only II
3. Both I and II
4. Neither I nor II
Zoology - Section A
51. The mucosa lining of the alimentary canal does not:
1. have goblet cells
2. form gastric glands
3. have Brunner’s glands
4. form crypts of Lieberkuhn
52. The cell shown in the given diagram:

| 1. |
is a bone marrow stem cells committed to produce RBCs |
| 2. |
is the megakaryocyte |
| 3. |
is the commonest WBC and is phagocytic |
| 4. |
is responsible for acquired immunity in humans |
53. Contraction of diaphragm and contraction of external intercostals:
| 1. |
decrease the intrapulmonary pressure and lead to inhalation |
| 2. |
increase the intrapulmonary pressure and lead to inhalation |
| 3. |
decrease the intrapulmonary pressure and lead to exhalation |
| 4. |
increase the intrapulmonary pressure and lead to exhalation |
54. Beginning with a single template DNA, a PCR reaction is allowed to continue for 30 cycles. The number of copies of the template DNA at the end is expected to be about:
| 1. |
256 |
2. |
256,000 |
| 3. |
1 million |
4. |
1 billion |
55. Which of the following joints is not expected to allow any movement?
1. Sutures in the skull
2. Joint between adjacent vertebra
3. Gliding joint
4. Joint between carpal and metacarpal of the thumb
56. Ahmed Khan of Bangalore is famous for his:
| 1. |
concept of organic farming |
| 2. |
contribution for developing ways of disposing radioactive wastes |
| 3. |
discovery of the process of incineration |
| 4. |
innovation that can be a remedy for plastic wastes |
57. To produce human recombinant insulin, Eli Lilly:
| 1. |
prepared two DNA sequences corresponding to A and B chains of human insulin and introduced them into plasmids of E. coli to produce insulin chains. |
| 2. |
prepared two DNA sequences corresponding to A and C chains of human insulin and introduced them in plasmids of Yeast to produce insulin chains. |
| 3. |
used reverse transcription of the hnRNA transcript of the insulin gene to make complementary DNA. |
| 4. |
used fragments created by restriction enzyme digestion to isolate and used the gene for insulin from pancreatic cells. |
58. Which of the following is the correct description of the relation between T cells and B cells?
| 1. |
Both are processed in bone marrow and respond to antigen from thymus |
| 2. |
T cells help B cells produce antibodies |
| 3. |
B cells increase the phagocytic capabilities of T cells |
| 4. |
While B cells are involved in first response to an antigen, T cells respond to subsequent encounters with the same antigen |
59. The increase of the level of which of the following in the sarcoplasm of a skeletal muscle fibre will lead to the contraction of a muscle?
1. Acetylcholine
2. ATP
3. Calcium ions
4. Troponin C
60. Identify a disease that is caused by an abnormality where the ability of the acquired immune system to distinguish between self and non-self is compromised:
| 1. |
Creutzfeldt Jakob Disease |
2. |
Rheumatoid arthritis |
| 3. |
Oculo-cutaneous albinism |
4. |
Diabetes insipidus |
61. Given below are two statements:
| Assertion (A): |
Selection of good breeds is the most important task when setting up a dairy farm for cattle. |
| Reason (R): |
Milk yield is primarily dependent on the quality of breeds in the farm. |
| 1. |
Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. |
(A) is true but (R) is false. |
| 4. |
Both (A) and (R) are false. |
62. Match the structures in Cockroach given in Column-I with their location in Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Testes |
P. |
6th abdominal segment |
| B. |
Ovaries |
Q. |
4th – 6th abdominal segments |
| C. |
Mushroom gland |
R. |
6th – 7th abdominal segments |
| D. |
Spermatheca |
S. |
2nd – 6th abdominal segments |
Codes:
|
A |
B |
C |
D |
| 1. |
S |
Q |
R |
P |
| 2. |
Q |
S |
R |
P |
| 3. |
P |
R |
Q |
S |
| 4. |
R |
S |
P |
Q |
63. In the Avery, Macleod, and McCarty experiment, the use of which of the following will affect bacterial transformation?
1. trypsin
2. chymotrypsin
3. ribonuclease
4. deoxyribonuclease
64. In traditional DNA fingerprinting [such as RFLP], the DNA that has been collected is first cut into smaller fragments by:
| 1. |
probe molecules |
2. |
restriction enzymes |
| 3. |
gel electrophoresis |
4. |
denaturation |
65. Which gene in the lac operon codes for an intracellular enzyme that cleaves the disaccharide lactose into glucose and galactose?
1. Lac A
2. Lac Y
3. Lac Z
4. Lac I
66. The rod-shaped bacteria belongs to which of the following category of bacteria?
1. Vibrio
2. Bacillus
3. Coccus
4. Spirillum
67. Erythroxylum coca, Atropa belladonna, and
Datura innoxia are plants:
| 1. |
that are critically endangered according to the IUCN list. |
| 2. |
which have become invasive in Indian ecosystems. |
| 3. |
with hallucinogenic properties. |
| 4. |
found only in the Himalayan region of the Indian subcontinent. |
68. For which of the following animals will thermoregulation be the most energetically expensive?
1. Elephant
2. Humming-bird
3. Rabbit
4. Horse
69. Identify the incorrectly matched pair:
1. LH surge: Ovulation
2. Corpus luteum: Ruptured Graafian follicle
3. hCG: Secreted by the ovary during pregnancy
4. Oxytocin: Milk ejection
70. Consider the given two statements:
| I: |
Sexually transmitted infections can lead to pelvic inflammatory disease. |
| II: |
Pelvic inflammatory disease increases the risk of ectopic pregnancy. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
71. A monovalent antibody molecule can be represented as:
72. Which of the following cofactors required by enzymes is often vitamins or made from vitamins?
| 1. | Apoenzymes | 2. | Prosthetic groups |
| 3. | Zymogens | 4. | Co-enzymes |
73. Identify the incorrectly matched pair:
| 1. |
Obelia |
Metagenesis |
| 2. |
Planaria |
Free living with high regeneration capacity |
| 3. |
Ancylostoma |
Acoelomate |
| 4. |
Nereis |
Parapodia |
74. Match each item in Column I with one in Column II and select the correct match from the codes given below:
|
Extinct animal |
|
Country/continent |
| A |
Stellar’s sea cow |
P |
Australia |
| B |
Thylacine |
Q |
Russia |
| C |
Quagga |
R |
Mauritius |
| D |
Dodo |
S |
Africa |
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
Q |
P |
S |
R |
| 3 |
R |
S |
P |
Q |
| 4. |
S |
R |
Q |
P |
75. Identify the part of the nephron that:
| I: |
is lined by cuboidal brush bordered epithelium |
| II: |
reabsorbs 70-80 percent of electrolytes and water |
| 1. |
Proximal convoluted tubule |
| 2. |
Loop of Henle |
| 3. |
Distal convoluted tubule |
| 4. |
Collecting duct |
76. Often described as infoldings of bacterial cell membrane, which of the following structures help in cell wall formation and DNA replication?
1. Inclusion bodies
2. Chromatophores
3. Fimbriae
4. Mesosomes
77. In Cardiac muscle tissue:
| I: |
Cell junctions fuse the plasma membranes of cardiac muscle cells |
| II: |
Communication junctions allow the cell to contract as a unit |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
78. Which of the following hormones does not increase blood glucose level?
1. Glucagon
2. Cortisol
3. Insulin
4. Growth hormone
79. What is the mechanism of sex determination in honey bees?
| 1. |
XX / XY male heterogamety |
| 2. |
ZZ / ZW female heterogamety |
| 3. |
Haplo-diploidy |
| 4. |
XX / XO male heterogamety |
80. The allele of a gene locus that is least frequent, in a population, has a frequency greater than 0.01. Such a gene locus will be called:
| 1. |
Redundant |
2. |
Degenerate |
| 3. |
Polymorphic |
4. |
Hot spot |
81. The secondary oocyte completes the second meiotic division:
| 1. |
At the time of insemination |
| 2. |
At puberty |
| 3. |
At the time of fusion of a sperm with an ovum |
| 4. |
Just before birth |
82. What is incorrect regarding the ovary in humans?
| 1. |
The surface is covered with simple cuboidal epithelium, called the germinal epithelium. |
| 2. |
Follicles in various phases of development are seen in the ovarian cortex |
| 3. |
It is connected to the uterus and pelvic floor by tendons |
| 4. |
It is a primary sex organ |
83. A female is a "carrier" for the trait red color blindness [remember this trait is X-linked and colour blindness is sex-linked recessive condition], marries a normal male. What proportion of their female progeny will be affected by the disorder?
| 1. |
3/4 |
2. |
1/2 |
| 3. |
1/4 |
4. |
0 |
84. Clearing of bottled fruit juices is achieved by treatment with:
| 1. |
Lipases and Proteases |
2. |
DNase and RNase |
| 3. |
Proteases and Pectinases |
4. |
Cellulase and Chitinase |
85. Identify the set of hormones secreted by placenta that are essential to maintain pregnancy:
1. hCG, hPL, progesterone, prolactin
2. hCG, hPL, estrogens, relaxin, oxytocin
3. hCG, hPL, progesterone, estrogens
4. hCG, progesterone, estrogens, glucocorticoids
Zoology - Section B
86. Kangaroo rat:
| I: |
has a marsupium to nurse the young ones |
| II: |
obtain enough water from the metabolic oxidation of the seeds they eat to survive and do not need to drink water at all |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
87. During secondary treatment of sewage water:
| I: |
vigorous growth of useful aerobic microbes into flocs is allowed |
| II: |
aerobic microbes consume the major part of the organic matter in the effluent. |
| III: |
the BOD of the water sample gets significantly increased |
| 1. |
Only I and II are correct |
| 2. |
Only I is correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
88. Consider the given two statements:
| I: |
SA node is the natural pacemaker of the human heart. |
| II: |
SA node is the only part of that nodal musculature that is auto-excitable. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
89. What restores the resting membrane potential of the axonal membrane at the site of excitation?
1. Influx of sodium ions
2. Efflux of sodium ions
3. Influx of potassium ions
4. Efflux of potassium ions
90. Given below are two statements:
| Assertion (A): |
Vertebrochondral ribs are called true ribs. |
| Reason (R): |
Vertebraochondral ribs are attached to the thoracic vertebrae dorsally and are ventrally connected to the sternum. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
91. All the following features are present in bony fishes except:
| 1. |
operculum |
2. |
air bladder |
| 3. |
placoid scales |
4. |
terminal mouth |
92. What differentiates kwashiorkor from marasmus?
| I: |
There is sufficient calorie intake in kwashiorkor |
| II: |
There is oedema in Kwashiorkor |
1. Only I
2. Only II
3. Both I and II
4. Neither I nor II
93. An abnormal increase in the secretion of aldosterone will lead to:
| 1. |
An increased concentration of sodium in the blood and a reduced blood pressure |
| 2. |
An increased concentration of potassium in the blood and a reduced blood pressure |
| 3. |
A decreased concentration of sodium in the blood and an elevated blood pressure |
| 4. |
A decreased concentration of potassium in the blood and an elevated blood pressure |
94. Arrange the following acts in their chronological sequence in which they were passed in India [earliest to latest]:
| I: |
Air [Prevention and Control of Pollution] Act |
| II: |
Environment [Protection] Act |
| III: |
Water [Prevention and Control of Pollution] Act |
1. II, III, I
2. III, I, II
3. II, I, III
4. I, II, III
95. An infertile couple has been suggested Artificial Insemination into the uterus [IUI]. This should be done:
| 1. |
just after the completion of the menstrual flow |
| 2. |
near the time of ovulation |
| 3. |
in the middle of the follicular phase |
| 4. |
after corpus luteum starts the synthesis of progesterone |
96. Detritus food chain:
| 1. |
begins with primary producers like grass |
| 2. |
is the major conduit for energy flow in an aquatic ecosystem |
| 3. |
is not made up of any decomposers |
| 4. |
may be connected with the grazing food chain at some levels |
97. Our lungs remove large amounts of carbon dioxide [approximately 200 mL/minute]. This translates to about:
1. 156 L/day
2. 288 L/day
3. 412 L/day
4. 504 L/day
98. What would not be true regarding Down’s Syndrome?
| 1. |
It is also known as trisomy 21 |
| 2. |
The parents of the affected individual are usually affected from the disorder. |
| 3. |
There is no cure for Down syndrome. |
| 4. |
The extra chromosome 21 material may also occur due to a Robertsonian translocation in 2–4% of cases. |
99. Given below are two population pyramids, both of India, one for year 2000 and another for year 2020:

| 1. |
Pyramid A is of 2020 as it has a broader base |
| 2. |
Pyramid B is of 2000 as it has more females in the younger age groups |
| 3. |
Pyramid B is of 2020 as it has more females in the younger age groups |
| 4. |
Pyramid A is of 2000 as it has a broader base |
100. What is the sensory part of the utricle and saccule in the internal ear?
1. Macula
2. Cupula
3. Crista
4. Organ of Corti
Chemistry - Section A
101. Which of the following is/are correct about isothermal expansion of work?
1. \(w_{irreversible} =w_{reversible}\)
2. \(w_{irreversible} > w_{reversible}\)
3. \(w_{irreversible} < w_{reversible}\)
4. All of the above
102. From the given pairs below, the pair in which both elements have the same electronegativity is:
| 1. |
O and Cl |
2. |
N and Cl |
| 3. |
N and Br |
4. |
C and Br |
103. What is the number of optically active isomers among all the isomers of \( (C_6H_{13}Br)\)?
1. 7
2. 8
3. 9
4. 17
104. The formal charge of \(S\) in \(SO_3\) will be:
1. Zero
2. +1
3. +2
4. +6
105. Determine the molality of the solution, if the mole fraction of a solute in an aqueous solution is 0.1:
| 1. |
3.085 |
2. |
6.17 |
| 3. |
12.34 |
4. |
24.68 |
106. When hydrated barium peroxide is reacted with ice cold \(H_2SO_4\) then \(H_2O_2\) is formed. In hydrated barium peroxide, the number of water of crystallisation is
1. 2
2. 4
3. 6
4. 8
107. Which of the following molecules has a non-zero dipole moment?
1. \(PCl_3Br_2\)
2. \(PCl_3F_2\)
3. \(PCl_3(CH_3)_2\)
4. \(PCl_5\)
108. Analgesics are used to treat
1. vomiting in pregnancy
2. pain in sports injuries
3. rash on body due to allergies
4. fever in typhoid infection
109. Consider the given statements:
| Statement I: |
\(Eu^{2+}\) acts as a reducing agent. |
| Statement II: |
The +3 oxidation state is more common among lanthanoids. |
| 1. |
Statement I is correct; Statement II is correct. |
| 2. |
Statement I is correct; Statement II is incorrect. |
| 3. |
Statement I is incorrect; Statement II is correct. |
| 4. |
Statement I is incorrect; Statement II is incorrect. |
110. Which of the following is the main constituent of cement?
1. Dicalcium silicate
2. Tricalcium silicate
3. Tricalcium aluminate
4. Tetracalcium aluminoferrite
111. From the polymers given below, which one is a chain growth polymer?
1. Bakelite
2. Neoprene
3. PHBV
4. Nylon-6, 6
112. Which of the following alkanes has the highest melting point?
1. n-pentane
2. Isopentane
3. Neopentane
4. Butane
113. What can be the active mass of CaCO3 if 10 grams of CaCO3 are taken in a one-liter container?
[Given: Molecular weight of CaCO3 = 100]
1. 0.1
2. 1
3. 0.01
4. 10
114. Which of the following is not regarded as a pollutant?
1. \(CO_2\)
2. \(O_3\)
3. \(NO_2 \)
4. Hydrocarbons
115. What is the reason behind the color of KMnO4?
1. d-d transition
2. Charge transfer spectra
3. Both (1) and (2)
4. Polarisation of anion
116. Consider the given reaction:
a K2Cr2O7 + b HCl \(\rightarrow\) c KCl + d CrCl3 + e H2O + f Cl2
The value of a,d & f, respectively, are:
| 1. |
2, 4, 8 |
2. |
1, 2, 3 |
| 3. |
2, 2, 3 |
4. |
1, 2, 4 |
117.
In which of the following transitions of hydrogen atom, radiation of minimum frequency is emitted ?
1. n2 = \(\infty\) to n1 = 1
2. n2 = 2 to n1 = 1
3. n2 = 3 to n1 = 2
4. n2 = \(\infty\) to n1 = 3
118. Which solution acts as an antiseptic and not as a disinfectant?
1. 0.2 to 0.4 ppm chlorine in aqueous solution
2. 2-3% I2 solution in alcohol and water
3. 1% solution of phenol
4. Sulphur dioxide in very low concentration
119. From the given species, the lowest critical temperature will be for
1. \(He\)
2. \(NH_3\)
3. \(CO_2\)
4. \(N_2\)
120. Dichromate ion shows the following equilibrium in an aqueous solution
\(Cr_{2}O_{7}^{2-} + H_{2}O\)  \(2CrO_{4}^{2-} + 2H^{+} \)
\(2CrO_{4}^{2-} + 2H^{+} \)
The value of X and Y, respectively, are:
1. 4 and 8
2. 8 and 4
3. 7 and 7
4. 4 and 4
121. The most unsymmetrical and symmetrical systems, respectively, are
1. Tetragonal and cubic
2. Rhombohedral and triclinic
3. Triclinic and cubic
4. None of the above
122. The water-soluble vitamin is:
1. A
2. C
3. D
4. E
123.
| Assertion (A): |
For an exothermic reaction, the value of Kc decreases on increasing the temperature. |
| Reason (R): |
Rate constant for a backward reaction increases more than the increase in the rate constant for a forward reaction. |
1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
124. Ethane is reacted with Cl2 in presence of sunlight. Number of halogenated alkanes possible out of this reaction are:
1. 6
2. 7
3. 8
4. 9
125. The most effective electrolyte to cause the flocculation of a positively charged ferric hydroxide colloid is
1. AlCl3
2. K4[Fe(CN)6]
3. BaSO4
4. Na3PO4
126. Which of the following compounds can show geometrical isomerism?
1. \([Pt(NH_3)_2 Cl_2]\)
2. \([Zn(NH_3)_2 Cl_2]\)
3. \([Pt(NH_3)_3 Cl]\)
4. Both (1) and (2)
127. The boiling point of an aqueous solution is . The freezing point of this solution will be
1.
2.
3.
4.
128. For an exothermic reaction, the value of \(\Delta H\) will be
1. More than \(E_a\)
2. Less than \(E_a\)
3. Equal to \(E_a\)
4. All of the above
129. 
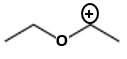
The compound "A' is-
130. The feasible reaction, amongst the following, is
1. \(2KCl + Br_2 \rightarrow 2KBr + Cl_2\)
2. \(2KF + I_2 \rightarrow 2KI + F_2\)
3. \(2 KClO_3 + I_2 \rightarrow 2KIO_3 + Cl_2\)
4. \(2KIO_3 + Cl_2 \rightarrow 2KClO_3 + I_2\)
131. 1 mole NaCl is mixed with 6 mole of H2O. As a result, relative lowering in vapour pressure is observed to be 0.2. In these conditions, percentage ionisation of NaCl will be
1. 25
2. 50
3. 75
4. 100
132. Which of the following species has the highest bond energy?
1. N2
2. CO
3. CO+
4. \(N^+_2\)
133. All elements of 14th group form MX4 (Tetrahalides) except PbI4 because:
1. Pb2+ is more stable than Pb4+
2. I- ion is a better reducing agent
3. Pb4+ is most stable
4. I- ion is an oxidising agent
134. Which of the following alkali metal has the lowest density?
1. Na
2. K
3. Rb
4. Cs
135. In Ellingham diagram, which conversion has a negative slope when a graph is plotted in between \(\Delta G\) and T?
1. \(C \rightarrow CO_2\)
2. \(Mg \rightarrow MgO\)
3. \(C \rightarrow CO \)
4. \(Al \rightarrow Al_2O_3\)
Chemistry - Section B
136. Which one of the following has the greatest number of moles at NTP?
1.
\(50 ~ml~CH_4\)
2.
\(150 ~ml~N_2\)
3.
\(10 ~ml~H_2O\)
4.
\(200 ~ml~CO_2\)
137. Which compound can react with Na as well as NaOH but not with \(\mathrm{N a H C O_{3}}\) ?
1. Ethanol
2. Picric acid
3. Carbolic acid
4. Ethyne
138. Which of the following aqueous solutions has the minimum freezing point?
1. 4% solution of \(NaCl\)
2. 10% solution of sugar
3. 6% solution of Urea
4. 2% solution of \(MgCl_2\)
139. How many electrons are there in a copper atom \((_{29}Cu)\), whose (n+l) = 4?
1. 6
2. 7
3. 8
4. 4
140. Identify the nonaromatic species among the following options.
141. Consider the following carbocations
The correct order of stability of the above-listed carbocations is
1. III > I >II > IV
2. II > III > I > IV
3. II > III > IV > I
4. III > I > IV > II
142. For a reaction,
\(\small{{\mathrm{A}(\mathrm{~g}) \rightarrow 2 \mathrm{~B}(\mathrm{~g})+\mathrm{C}(\mathrm{~g}) ; \Delta \mathrm{H}=4 \mathrm{~kJ}~ \&~ \Delta \mathrm{~S}=10 ~\mathrm{JK}^{-1}}}\).
At what temperature will the reaction be spontaneous?
1. Above 400 K
2. Below 400 K
3. Exactly at 400 K
4. At 27°C
143. Solubilities of AgCl in 0.01M NaCl, 0.01M CaCl2, 0.05M AgNO3 , pure water and in 0.01M NH4OH are S1 , S2 , S3 , S4 and S5 respectively. The correct order of these solubilities is:
1. S1 > S2 > S3 > S4 > S5
2. S3 > S2 > S1 > S5 > S4
3. S5 > S4> S1 > S2 > S3
4. S5 > S4 > S3 > S2 > S1
144. \(C_4H_6\) may have:
1. One triple bond
2. Two double bonds
3. Two cycles
4. All of the above
145. The time required for 40% completion of first-order reaction is 10 minutes. Time required for completion of 90% of this reaction will be
1. 22.5 minutes
2. 45 minutes
3. 90 minutes
4.180 minutes
146. In the adduct of NH3 and BF3, the hybridised state of N and B, respectively, will be
1. sp3 and sp3
2. sp3 and sp2
3. sp2 and sp2
4. sp2 and sp3
147. When 2 faradays of electricity is passed through \(AlCl_3\) solution, then the weight of aluminium deposited will be:
1. 27 gram
2. 18 gram
3. 9 gram
4. 54 gram
148. The correct order of the boiling point of the hydrides of 15th group is:
1. NH3>BiH3>SbH3>AsH3>PH3
2. BiH3>NH3>SbH3>AsH3>PH3
3. BiH3>SbH3>NH3>AsH3>PH3
4. BiH3>SbH3>AsH3>PH3>NH3
149. Identify the endothermic process among the following reactions:
1. \(F \rightarrow F^-\)
2. \(O^- \rightarrow O^{2-}\)
3. \(Cl \rightarrow Cl^-\)
4. \(O \rightarrow O^-\)
150. In which of the following complexes, the central atom has \(sp^3\) hybridisation?
1. \([PtCl_4]^{2-}\)
2. \([Ni(CN)_4]^{2-}\)
3. \([Cu(NH_3)_4]^{2+}\)
4. None of the above.
Physics - Section A
151. When a parallel beam of light of wavelength
\(\lambda\) falls onto a convex lens of diameter
\(D,\) focal length
\(f\) parallel to its principal axis, the image has a bright spot at the focal plane. As one moves away from the central point, the intensity becomes zero at a distance
\(r_1.\) Then
\(r_1=\)
1.
\(\frac{f\lambda}{D}\)
2.
\(\frac{f\lambda}{2D}\)
3.
\(\frac{0.61f\lambda}{D}\)
4.
\(\frac{1.22f\lambda}{D}\)
152. Two long straight wires carrying currents
\(i_1, i_2\) are placed as shown in the figure, just avoiding contact. The separation between the wires is negligible, and the wires are aligned along
\(x\) &
\(y\) axes respectively.

The wire along the
\(x\text-\)axis experiences:
| 1. |
a force along \(+y\) axis only. |
| 2. |
a force along \(-y\) axis. |
| 3. |
zero force, but a torque. |
| 4. |
no force and no torque. |
153. The radius of a nucleus \((r)\) is directly proportional to the cube-root of the number of nucleons \((N).\) Assume that the mass of a nucleon is fixed \((m_n).\) The density \((\rho)\) of a nucleus varies as:
1. \(N^3\)
2. \(N^1\)
3. \(N^0\)
4. \(N^{-1}\)
154. Which, of the following, does not suffer from chromatic aberration?
| 1. |
convex lens |
2. |
plano-convex lens |
| 3. |
concave lens |
4. |
concave mirror |
155. The system of gates shown in the adjacent diagram represents

1. AND gate
2. OR gate
3. XOR gate
4. null gate (always zero)
156. If the rate of rotation of the earth were to increase, the apparent acceleration due to gravity on the equator would:
1. increase
2. decrease
3. remain unchanged
4. change in direction away from the cente
157. A source of sound moves along a straight line undergoing SHM given by:
\(x=A~sin\omega t \)
The frequency of the source is
\(f_0\) and the speed of waves in the medium
\(v_s.\) The maximum and minimum frequencies of the sound emitted are
| 1. |
\(f_0\Big(1+{\large\frac{\omega A}{v_s}}\Big)\) and \(f_0\Big(1-{\large\frac{\omega A}{v_s}}\Big)\) |
| 2. |
\(f_0\Big(1-{\large\frac{\omega A}{v_s}}\Big)^{-1}\) and \(f_0\Big(1+{\large\frac{\omega A}{v_s}}\Big)^{-1}\) |
| 3. |
\(f_0\sqrt{1+{\large\frac{\omega A}{v_s}}} \) and \(f_0\sqrt{1-{\large\frac{\omega A}{v_s}}} \) |
| 4. |
\(f_0\Big(1+{\large\frac{\omega^2 A^2}{v_s^2}}\Big)\) and \(f_0\Big(1-{\large\frac{\omega^2 A^2}{v_s^2}}\Big)\) |
158. A block is suspended from a spring and causes an extension of \(2\) cm. It is now imparted a kinetic energy \(E\) so that the block rises up by exactly \(2\) cm. If the block were to be given the same kinetic energy upward, without being attached to a spring, it would rise up by:
1. \(1\) cm
2. \(2\) cm
3. \(4\) cm
4. \(8\) cm
159. Given below are two statements:
| Assertion (A): |
If the sun were to 'suddenly' be removed, then the earth would continue to move around in its orbit. |
| Reason (R): |
Angular momentum of a system of particles is conserved when there is no external torque. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
160. The average electric field associated with the plane electromagnetic wave
\(\vec E = E_0 \hat {i} \sin (wt - kz)\) is:
| 1. |
\(E_0 \hat i\) |
2. |
\(\dfrac {E_0} { \sqrt 2}\) \(\hat i \) |
| 3. |
\(\sqrt 2E_0 \hat i\) |
4. |
zero |
161. A uniform rod of length
\(L\) is standing upright, pivoted at its lower end. The rod can freely rotate about the pivot. If it is slightly disturbed so that it falls to the ground, the speed of the highest point, when it strikes the ground will be:

| 1. |
\(\sqrt{2gL}\) |
2. |
\(\sqrt{3gL}\) |
| 3. |
\(\sqrt{6gL}\) |
4. |
\(\sqrt{gL}\) |
162. The de-Broglie wavelength of an electron in the ground state of a
\(\mathrm H\text -\)atom is
\(\lambda_1\) and that on the
\(\mathrm{He}^{+}\text -\)ion is
\(\lambda_2.\) The kinetic energies of the electrons in the
\(\mathrm H\text -\)atom and the
\(\mathrm{He}^{+}\text -\)ion-having the same de-Broglie wavelength are
\(E_1\) and
\(E_2.\) The ratio
\(\dfrac{\lambda_1}{\lambda_2}\) is:
| 1. |
\(4\) |
2. |
\(2\) |
| 3. |
\(\dfrac12\) |
4. |
\(\dfrac14\) |
163. As temperature increases, the resistivity of silicon:
1. increases
2. decreases
3. remains unchanged
4. first increases and then decreases
164. Two stones
\(A\) and
\(B\) are projected simultaneously with the same speed: one
\((A)\) vertically up and the other
\((B)\) horizontally from the same point. Stone
\(A\) rises up to a maximum height
\(H\) and falls down. The separation between
\(A\) and
\(B,\) when
\(A\) reaches its maximum height:
| 1. |
is less than \(H\) |
| 2. |
is greater than \(H\) |
| 3. |
is equal to \(H\) |
| 4. |
cannot be related with \(H\) |
165. During a certain atmospheric process, a pocket of air near the earth's surface rises upwards very rapidly into the upper regions of the atmosphere. As a result, the temperature of this air:
| 1. |
increases. |
| 2. |
decreases. |
| 3. |
remains constant. |
| 4. |
first increases, then decreases. |
166. The circuit shown in the diagram given below, is set up with all the capacitors initially uncharged.

The potential difference between
\(A\) and
\(B,\) after
\(A\) is connected to
\(Y\) and
\(B\) to
\(X,\) is:
1.
\(2.6\) V
2.
\(2.4\) V
3.
\(6\) V
4. zero
167. An elevator moving up is decelerating at \(1\) m/s2. Then, the apparent weight of a \(50\) kg man, on a weighing machine, will be: (take \(g=\) \(10\) m/s2)

| 1. |
\(55\) kg |
2. |
\(45\) kg |
| 3. |
\(40\) kg |
4. |
none of the above |
168. A uniform ladder of mass
\(10\) kg is placed at an angle against a frictionless vertical wall, as shown in the figure, by applying a horizontal
force \(F\) at the
bottom \((B)\) of the ladder, towards the wall.
(Take \(g=10\) m/s2).

Assume that the ground is frictionless. The force
\(F\) equals:
| 1. |
\(100\sqrt3\) N |
2. |
\(50\sqrt3\) N |
| 3. |
\(\dfrac{100}{\sqrt3}\) N |
4. |
\(\dfrac{50}{\sqrt3}\) N |
169. A viscous liquid is flowing through a straight tube under the action of a pressure difference across the ends of the tube. The tube is aligned with the
\(x\)-axis. The force exerted on the tube by the liquid has the form:
\((a,b>0)\)

| 1. |
\(a\hat i\) |
2. |
\(-a\hat i\) |
| 3. |
\(a\hat i+b\hat j\) |
4. |
\(-a\hat i+b\hat j\) |
170. A particle of mass
\(m\) collides with another particle of mass
\(m',\) which is at rest and the combined mass moves with
\(10\text{%}\) reduction in velocity. The ratio of the masses is:
| 1. |
\(\dfrac{m'}{m}=\dfrac{1}{10}\) |
2. |
\(\dfrac{m'}{m}=\dfrac{1}{9}\) |
| 3. |
\(\dfrac{m'}{m}=\dfrac{1}{8}\) |
4. |
\(\dfrac{m'}{m}=\dfrac{1}{2}\) |
171. An alternating emf (AC) is applied across the circuit shown in the figure. Let
\(V_{AX}\) be the voltage across the inductor
\(L,\) and
\(V_{XY}\) be the voltage across the resistor
\(R.\) Let the angular frequency be
\(\omega.\) The phase difference between
\(V_{XY}\) and
\(V_{AX}\):

| 1. |
depends on the ratio \(\dfrac{\omega L}{R}\) |
| 2. |
depends on the quantity \(\sqrt{(\omega L)^2+R^2}\) |
| 3. |
depends on \(L\) and \(R,\) but not on \(\omega\) |
| 4. |
is independent of \(L,R,\omega\) |
172. A heavy uniform rope is suspended from the ceiling, and a block equal to
\(3\) times the mass of the rope is suspended from the bottom. The ratio of the speed of a transverse pulse at the top
\((v_t)\) to its speed at the bottom
\((v_b)\) of the rope,
\(\dfrac{v_t}{v_b}=\)
| 1. |
\(\dfrac43\) |
2. |
\(\sqrt{\dfrac43}\) |
| 3. |
\(\dfrac34\) |
4. |
\(\sqrt{\dfrac34}\) |
173. A boat, when rowed perpendicular to the flow of a
\(500\) m wide river, reaches its opposite bank in
\(10\) min. If the boat is rowed downstream at an angle of
\(30^\circ\) with the flow, it will cross in:
| 1. |
\(10\) min |
2. |
\(5\sqrt3\) min |
| 3. |
\(20\) min |
4. |
\(\dfrac{10}{\sqrt3}\) min |
174. A ray is incident normally onto the surface
\(AB\) of the prism
\((\angle A=30^\circ,\angle B=90^\circ).\) The refractive index of the material of the prism is
\(\sqrt2.\) The deviation of this ray is:

1.
\(30^\circ\) downward
2.
\(15^\circ\) downward
3.
\(30^\circ\) upward
4.
\(15^\circ\) upward
175. A piece of iron is heated until it is red hot. It is placed in a solenoid. As the temperature of the iron decreases, the magnetic field inside it:
1. first increases, then decreases.
2. first decreases, then increases.
3. increases.
4. decreases.
176. A straight horizontal wire
\(\mathrm{AB}\) of length
\(l\) falls from rest under gravity. A uniform horizontal magnetic field
\(B\) acts perpendicular to the plane of motion of
\(\mathrm{AB},\) as shown in the figure. The induced emf across
\(\mathrm{AB},\) \(E,\) is proportional to:

1.
\(B\)
2.
\(l\)
3. time,
\(t\)
4. all of the above
177. Assume that an electron orbits around a nucleus in a circular orbit, and Newton's laws are valid. What is the ratio of the kinetic energy \((KE)\) of the electron to its potential energy \((PE)\) in orbit (in magnitude)?
1. \(1:2\)
2. \(2:1\)
3. \(1:1\)
4. none of the above
178. The left plate
\(A\) of an air capacitor is connected to the positive terminal while the right plate
\(B\) is connected to the negative terminal of a cell of voltage
\(V_0.\) Assume that the plate area is
\(A,\) and the plate separation is
\(d.\) If a slab of dielectric constant
\(K\) is inserted into the space between the plates, the electric field in the dielectric will be: (compared to the air capacitor)

| 1. |
more. |
| 2. |
less. |
| 3. |
equal. |
| 4. |
more or less or equal depending on the value of \(K\). |
179. Two cells are balanced on a potentiometer and give balance length readings of \(20~\text{cm}\) and \(30~\text{cm}\) respectively. If the cells are connected in parallel (with their like terminals connected together), the balance length of the combination is found to be \(28~\text{cm}\). Their resistances are in the ratio:
1. \(\dfrac{1}{3}\)
2. \(\dfrac{3}{1}\)
3. \(\dfrac{1}{4}\)
4. \(\dfrac{4}{1}\)
180. If the volume of an ideal gas is doubled, isothermally, the final pressure is \(1\) atm. If the process was adiabatic, the pressure would have been:
1. \(1\) atm
2. \(0.5\) atm
3. less than \(1\) atm
4. greater than \(1\) atm
181. Let I
E, I
B, I
C be the magnitudes of the emitter, base, and collector currents in a transistor. Further, let V
BE and V
CE be the base-emitter and collector-emitter voltages (in magnitude) respectively. Then, in the common-emitter (CE) mode:
| 1. |
VCE - small, IB - large, when the transistor is in saturation |
| 2. |
VCE - large, IB - small, when the transistor is in saturation |
| 3. |
VBE - large, IC - small, when the transistor is in cutoff |
| 4. |
VBE - large, IE - small, when the transistor is in cutoff |
182. The effective spring constant in calculating the time period of SHM of the system of springs and the block is:

1.
\((k_1+k_2) \)
2.
\(|k_1-k_2| \)
3.
\(\Big(\dfrac{1}{k_1}+\dfrac{1}{k_2}\Big)^{-1} \)
4.
\(\Big|\dfrac{1}{k_1}-\dfrac{1}{k_2}\Big|^{-1} \)
183. Given below are two statements:
| Statement I: |
Gauss's law for electric fields is a consequence of the conservation of energy. |
| Statement II: |
Coulomb's law for electric charges leads to a conservative electric field. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
184. A simple pendulum is made with a thin wire
(length: \(l,\) area: \(A,\) Young's modulus: \(Y\)) attached to a heavy bob of mass
\(M.\) The pendulum is released from the rest with the bob at the same level as the point of suspension and swings down in a circular arc. The elongation in the wire when the bob reaches the lowest point is:
| 1. |
\(\dfrac{3 M g l}{A Y}\) |
2. |
\(\dfrac{2 M g l}{A Y}\) |
| 3. |
\(\dfrac{3 M g l}{2 A Y}\) |
4. |
\(\dfrac{M g l}{A Y}\) |
185. Find the dimension of the quantity
\(\varepsilon_0hc\):
(where
\(\varepsilon_0=\) permittivity of free space,
\(h=\) Planck's constant, and
\(c=\) speed of light in vacuum)
| 1. |
\(\text{(charge)}\times\text{(energy)}\) |
2. |
\(\text{(energy)}^2\) |
| 3. |
\(\text{(charge)}^2\) |
4. |
\(\dfrac{\text{charge}}{\text{energy}}\) |
Physics - Section B
186. The potential difference
\(V_{AB}\) (in volts) is plotted as a function of the resistance
\(R.\) The graph is given by:

187. The electric field associated with a light wave is given by
\(E = E_0~ (\sin \omega_1 t)~ (\sin \omega_2 t)\).
This light wave falls on a metal having a threshold frequency,
\(\nu_0.\) The maximum kinetic energy of the emitted photoelectrons will be: (
\(h\) is Planck's constant)
| 1. |
Either \(\dfrac{h \omega_{1}}{2 \pi}\) or \(\dfrac{h \omega_{2}}{2 \pi}\) |
| 2. |
Either \(\left(\dfrac{h \omega_{1}}{2 \pi}-h \nu_{0}\right)\) or \(\left(\dfrac{h \omega}{2 \pi}-h \nu_{0}\right)\) |
| 3. |
\(\dfrac{h\left(\omega_{1}+\omega_{2}\right)}{2 \pi}-h \nu_{0}\) |
| 4. |
Both \(\dfrac{h\left(\omega_{1}+\omega_{2}\right)}{2 \pi}-h \nu_{0}\) and \(\dfrac{h\left |\omega_{1}-\omega_{2}\right|}{2 \pi}-h \nu_{0}\) |
188. The magnitude of the integral of the quantity
\(\int\vec B\cdot d\vec{ l}\) around the loop
\(PQR\) of the equilateral triangle is
\(K.\) The field at the centre of the long solenoid is:

| 1. |
\(\dfrac{K}{a}\) |
2. |
\(\dfrac{K}{b}\) |
| 3. |
\(\dfrac{K}{a-b}\) |
4. |
\(\dfrac{K}{a+b}\) |
189. An elastic ball is projected vertically upward with a
speed \(u,\) and it returns to the ground and rebounds, the motion is periodic with a
period \(T.\) A simple pendulum, having a length equal to maximum altitude attained by this ball, would have a time period of:
| 1. |
\(T\) |
2. |
\(\pi T\) |
| 3. |
\(\pi\sqrt2T\) |
4. |
\(\dfrac{\pi}{\sqrt 2}T\) |
190. An astronaut, in a space shuttle, orbiting close to the earth's surface (take \(g= 10~\text{m/s}^2\)), suddenly fires his engines so as to give him a forward acceleration of \(\dfrac{3g}{4}\) along the direction of his motion. At that instant, his apparent weight is:
| 1. |
\(25\%\) more than his real weight. |
| 2. |
\(25\%\) less than his real weight. |
| 3. |
\(75\%\) more than his real weight. |
| 4. |
\(75\%\) less than his real weight. |
191. The input to a Zener diode circuit is an AC-voltage along with a DC:
\(V_i=(20~\text V)\sin\omega t+(20~\text V)~\)
Current flows through the diode:

| 1. |
continuously, in both directions |
| 2. |
continuously, only in one direction |
| 3. |
intermittently, in both directions |
| 4. |
intermittently, only in one direction |
192. A particle moves in a circle of radius \(R\) with a constant speed \(v.\) The average acceleration of the particle in during \(\left(\dfrac16\right)^{\mathrm{th}}\) revolution is:
| 1. |
\(\dfrac{v^2}{R}\) |
2. |
\(\dfrac{2\pi v^2}{6R}\) |
| 3. |
\(\dfrac{\pi v^2}{6 R}\) |
4. |
\(\dfrac{3v^2}{\pi R}\) |
193. As light traverses through an optical medium, it is observed that its wavelength decreases.
| Statement I: |
In this case, the frequency of light increases. |
| Statement II: |
In the above case, the refractive index of the medium increases. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
194. The moment of inertia of a uniform semi-circular disc of
mass \(m\) and
radius \(r,\) about its open diameter is:
| 1. |
\(\dfrac{1}{2}mr^2\) |
| 2. |
\(\dfrac{1}{4}mr^2\) |
| 3. |
greater than \(\dfrac{1}{4}mr^2\) but less than \(\dfrac{1}{2}mr^2\) |
| 4. |
less than \(\dfrac{1}{4}mr^2\) |
195. A rod of mass M and length L is suspended vertically at its highest point. The rod is held so that it is horizontal and free to rotate about A and then released. There is no friction anywhere.
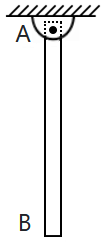
The angular speed of the road, when it reaches its lowest position, is \(\omega\). Then \(\omega^2\)=
| 1. |
\(\dfrac gL\) |
2. |
\(\dfrac {2g}L\) |
| 3. |
\(\dfrac {3g}L\) |
4. |
\(\dfrac {12g}L\) |
196. The quantity of heat required to take a system from \(\mathrm{A}\) to \(\mathrm{C}\) through the process \(\mathrm{ABC}\) is \(20\) cal. The quantity of heat required to go from \(\mathrm{A}\) to \(\mathrm{C}\) directly is:

1. \(20\) cal
2. \(24.2\) cal
3. \(21\) cal
4. \(23\) cal
197. A submarine having a fixed volume is floating submerged underwater. Air, at high pressure, is pumped into the submarine using pipes. Due to this, the submarine will:
| 1. |
rise. |
| 2. |
sink. |
| 3. |
float in the same position. |
| 4. |
either rise or sink depending on the air pressure. |
198. The unit of coefficient of volume expansion of metal is:
1. \((^\circ\text C)^ {-1}\)
2. \(\text{m}^3/^\circ\text C\)
3. \(^\circ\text C\)
4. \(^\circ\text C/\text{m}^3\)
199. Three charges are placed at the three corners of an equilateral triangle as shown in the figure. The potential at the mid-point of a side with opposite charges is
\(2~\text V.\) The potential at the centre of the triangle is:

| 1. |
\(2~\text V\) |
2. |
\(3~\text V\) |
| 3. |
\(2\sqrt3~\text V\) |
4. |
\(\dfrac{2}{\sqrt3}~\text V\) |
200. Match the quantities in
List-I with their appropriate units in
List-II.
| List-I |
List-II |
| (A) |
inductance \(\times\) current |
(I) |
V |
| (B) |
frequency \(\times\) capacitance |
(II) |
Wb |
| (C) |
frequency \(\times\) magnetic flux |
(III) |
\(\Omega^{-1}\) |
| (D) |
electric flux |
(IV) |
V-m |
| 1. |
\(\mathrm{A\text-I, B\text{-}IV, C\text-II, D\text- III}\) |
| 2. |
\(\mathrm{A\text-II, B\text{-}III, C\text-I, D\text- IV}\) |
| 3. |
\(\mathrm{A\text-III, B\text{-}I, C\text-II, D\text- IV}\) |
| 4. |
\(\mathrm{A\text-III, B\text{-}IV, C\text-II, D\text- I}\) |
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course




 \(2CrO_{4}^{2-} + 2H^{+} \)
\(2CrO_{4}^{2-} + 2H^{+} \)