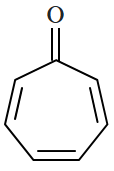
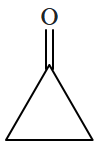
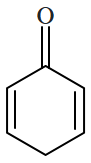
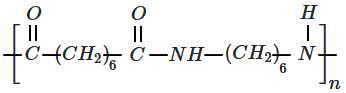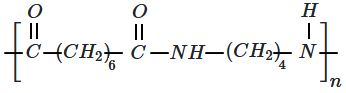Botany - Section A
1. Identify the incorrect statement regarding the secondary growth seen in plants:
| 1. |
Secondary growth does not occur in monocotyledons |
| 2. |
The cells of secondary cortex are parenchymatous |
| 3. |
Bark refers to all the tissues exterior to the vascular cambium, therefore it does not include secondary phloem |
| 4. |
In the dicot root, the vascular cambium originates from the tissue located just below the phloem bundles, a portion of pericycle tissue, above the protoxylem forming a complete and continuous wavy ring, which later becomes circular |
2. Identify the incorrectly matched pair:
| 1. |
Pericarp |
Wall of the fruit |
| 2. |
Perisperm |
Residual, persistent nucellus |
| 3. |
Perigynous |
A flower with superior ovary |
| 4. |
Perianth |
Floral structure comprised of calyx and corolla especially when the two whorls are fused |
3. Which of the following would not be true for facilitated diffusion?
1. Requires special membrane proteins
2. Highly selective
3. Transport saturates
4. Uphill transport and requires ATP energy
4. In cymose type of inflorescence:
| 1. |
the main axis terminates in a flower and flowers are borne in basipetal order |
| 2. |
the main axis continues to grow and the flowers are borne laterally in an acropetal order |
| 3. |
the main axis terminates in a flower and flowers are borne in an acropetal order |
| 4. |
the main axis continues to grow and the flowers are borne laterally in basipetal order |
5. Which of the following is not an anatomical feature of a dicot stem?
| 1. |
Hypodermis of collenchyma |
| 2. |
Endodermis, also called the starch sheath |
| 3. |
Pericycle semilunar patches of sclerenchyma |
| 4. |
Vascular bundle – conjoint and closed |
6. The oxygen evolving complex is associated with:
| 1. |
PS I |
2. |
PS II |
| 3. |
Both 1 and 2 |
4. |
Neither 1 nor 2 |
7. Amongst the ‘Evil Quartet’, which of the following is the most important cause that is driving plants and animals to extinction in contemporary times?
| 1. |
Habitat loss and fragmentation |
| 2. |
Over-exploitation |
| 3. |
Alien species invasion |
| 4. |
Co-extinctions |
8. Consider the given statements:
| I: |
Griffith discovered the process of bacterial transformation. |
| II: |
Hershey and Chase unequivocally proved that DNA is the genetic material. |
| III: |
Meselson and Stahl showed that DNA replicated semi-conservatively. |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
9. The endomembrane system is composed of the different membranes that are suspended in the cytoplasm within a eukaryotic cell. These membranes divide the cell into functional and structural compartments, or organelles. The system includes all the following except:
| 1. |
ER |
2. |
Golgi apparatus |
| 3. |
Chloroplasts |
4. |
Vacuoles |
10. Consider the given statements:
| I: |
Prop roots are seen in Banyan tree |
| II: |
Stilt roots are seen in maize and sugarcane |
| III: |
Pneumatophores are seen in Rhizophora |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
11. Consider the following statements about the different classes of algae:
| I: |
The members of Chlorophyceae have a rigid cell wall made of an inner layer of cellulose and an outer layer of pectose |
| II: |
The members of Rhodophyceae do not have a flagellum |
| III: |
Members of Phaeophyceae store food as floridean starch which is very similar to amylopectin and glycogen in structure |
| 1. |
Only I and III are correct |
| 2. |
Only I and II are correct |
| 3. |
Only II and III are correct |
| 4. |
All I, II and III are correct |
12. The increased growth per unit time is termed as growth rate. The exponential growth can be mathematically expressed as:
1. Lt = L0 +rt
2. Lt = L0 rt
3. W1 = W0ert
4. W1 = W0 + ert
13. Identify the incorrectly matched pair:
| 1. |
Golden rice |
Vitamin A enriched |
| 2. |
Parbhani kranti |
Resitance to yellow mosaic virus transferred from a wild species |
| 3. |
Jaya and Ratna |
Semi-dwarf rice varieties |
| 4. |
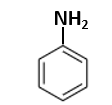
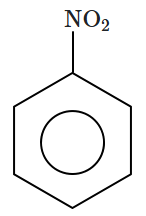
Sonalika and kalyan Sona |
Varieties of sugarcane grown in south India |
14. The shield-like cotyledon in grass [monocot] seeds is called:
1. Plumule
2. Scutellum
3. Perisperm
4. Coleoptile
15. What stage of prophase I of meiosis I:
| a. |
is characterized by the visibility of chiasmata |
| b. |
can last for months and years in some vertebrate oocytes |
1. Zygotene
2. Pachytene
3. Diplotene
4. Diakinesis
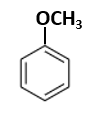
16. Normally, microinjection and biolistics [gene gun] are used, respectively, to transform:
1. plant cells and animal cells
2. animal cells and plant cells
3. animal cells and bacterial cells
4. plant cells and bacterial cells
17. Given below are two statements:
| Assertion (A): |
The respiratory pathway is an amphibolic pathway. |
| Reason (R): |
The respiratory pathway is involved in both anabolism and catabolism. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
(A) is False but (R) is True. |
18. While carrying out a dihybrid cross [a mating of two organisms heterozygous for two traits], a phenotype ratio of 9:3:3:1 in the offspring is expected when:
| 1. |
the genes are linked [located on the same chromosome |
| 2. |
each gene contains two mutations |
| 3. |
the gene pairs assort independently during meiosis |
| 4. |
the alleles of both genes show either co-dominance or incomplete dominance |
19. Match the mineral element in COLUMN I with their function in plants given in COLUMN II and select the correct match from the codes given:
|
COLUMN I |
|
COLUMN II |
| A |
Magnesium |
P |
Synthesis of chlorophyll |
| B |
Managanese |
Q |
Photolysis of water |
| C |
Molybdenum |
R |
Nitrogen metabolism |
| D |
Boron |
S |
Pollen germination |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
Q |
R |
S |
P |
| 3. |
R |
S |
P |
Q |
| 4. |
S |
P |
Q |
R |
20. During complete aerobic respiration of one molecule of glucose, there can be a net gain of:
| 1. |
24 ATP |
2. |
28 ATP |
| 3. |
33 ATP |
4. |
38 ATP |
21. Which of the following statements regarding ecological succession is/are true?
| I. |
The change in communities during ecological succession is orderly and sequential, parallel with the changes in the physical environment. |
| II. |
Secondary succession is faster than primary succession. |
| III. |
Usually, lichens are pioneer species during primary xerarch succession. |
| 1. |
Only I |
| 2. |
Only II |
| 3. |
Both I and III |
| 4. |
I, II and III |
22. Which of the following is an incorrect statement?
| 1. |
Carbon constitutes 51 percent of dry weight of organisms and is only next to water. |
| 2. |
During phosphorus cycle, there is no respiratory release of phosphorus into the atmosphere. |
| 3. |
Robert Constanza and his colleagues have tried to put price tags on nature’s life-supporting services. |
| 4. |
Out of the total cost of various ecosystem services, soil formation accounts for about 50 percent. |
23. Match each item in
Column-I with the one in
Column-II and select the correct match from the codes given:
|
Column-I
[Family] |
|
Column-II
[Floral character] |
| A. |
Brassicaceae |
P. |
Tetradynamous stamen |
| B. |
Fabaceae |
Q. |
Obliquely placed ovary |
| C. |
Solanaceae |
R. |
Epitepalous stamen |
| D. |
Liliaceae |
S. |
Vexillary aestivation |
Codes:
|
A |
B |
C |
D |
| 1. |
S |
R |
Q |
P |
| 2. |
R |
Q |
S |
P |
| 3. |
P |
Q |
S |
R |
| 4. |
P |
S |
Q |
R |
24. Which component of the endomembrane system of a eukaryotic cell is frequently observed in cells involved in protein synthesis and secretion?
| 1. |
Rough endoplasmic reticulum |
| 2. |
Golgi complex |
| 3. |
Smooth endoplasmic reticulum |
| 4. |
Chloroplasts |
25. Consider the following statements:
| I: |
Gymnosperms lack sieve tubes and companion cells |
| II: |
Phloem parenchyma is absent in most of the monocotyledons |
| III: |
Phloem fibres [bast fibres] are generally absent in the primary phloem |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
26. Identify the incorrect statement:
| 1. |
Typical angiosperm anther is bilobed and dithecous. |
| 2. |
Tapetum in the wall of the anther nourishes the developing pollen grains. |
| 3. |
In over 60 % of angiosperms, pollen grains are shed at the three-celled stage. |
| 4. |
Pollen exine is made up of sporopollenin. |
27. Consider the following statements regarding seed dormancy:
| I: |
Seed dormancy is the state in which a seed is unable to germinate, even under favorable growing conditions. |
| II: |
True dormancy or innate dormancy is caused by conditions within the seed that prevent germination under normally ideal conditions. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
28. DNA fragments created by the action of restriction enzymes can be separated by agar gel electrophoresis where:
| I: |
larger the fragment size, the farther it moves in the gel |
| II: |
the DNA fragments are forced to move towards anode under an electric field |
1. Only
I is correct
2. Only
II is correct
3. Both
I and
II are correct
4. Both
I and
II are incorrect
29. Which of the following is not an ex-situ method for conservation of biodiversity?
1. Botanical Gardens
2. Seed banks
3. Plant tissue culture
4. Biosphere Reserves
30. The mass of living material at a trophic level at a particular time is known as:
| 1. |
Standing state |
| 2. |
Primary Productivity |
| 3. |
Secondary Productivity |
| 4. |
Standing crop |
31. Viruses:
1. contain both DNA and RNA as genetic material
2. are facultative parasites
3. cannot be crystallized
4. are nucleoproteins
32. Which of the following secondary metabolites is an alkaloid and is clinically used as a very potent analgesic?
1. Morphine
2. Abrin
3. Lectin
4. Taxol
33. Identify the option where the names of scientists given in
Column-I are not correctly matched with their contribution to the science of genetics:
|
Column-I |
Column-II |
| 1. |
De Vries, Correns, Tschermak |
Disproved Mendel’s law of Independent Assortment |
| 2. |
Sutton and Boveri |
Gave the chromosomal theory of inheritance |
| 3. |
Alfred Sturtevant |
Developed the first genetic map |
| 4. |
T. H. Morgan |
Discovery of the white-eyed mutation in the fruit fly |
34. GEAC stands for:
| 1. |
Genetic Engineering Approval Committee |
| 2. |
Green Environment Action Committee |
| 3. |
Genetic Engineering Appraisal Council |
| 4. |
Genetic Ethics Approval Committee |
35. In transcription:
| 1. |
RNA complement formed includes the nucleotide uracil (U) in all instances where thymine (T) would have occurred in a DNA complement. |
| 2. |
The template strand of DNA is called the coding strand. |
| 3. |
RNA polymerase can only add nucleotides to the 5' end of the growing mRNA chain. |
| 4. |
The error rate is much lower than that in DNA replication. |
Botany - Section B
36. What type of argument for need to conserve biodiversity am I making when I say that we all must do our best to protect the biodiversity on the Earth as the nature provides invaluable services and intangible benefits like aesthetic pleasures of being in nature?
| 1. | Narrowly utilitarian | 2. | Broadly utilitarian |
| 3. | Ethical | 4. | Useless |
37. What could be considered as the most significant contribution of root pressure in the process of water transport?
| 1. |
It provides a push which plays a major role in movement of water up the trees |
| 2. |
It re-establishes the continuous chain of water molecules in the xylem which often breaks under the enormous tension created by transpiration |
| 3. |
It helps in active transport of various ions into the vascular tissues of the root from the soil |
| 4. |
It helps in loss of excess water in its liquid phase (guttation) |
38. The mode of replication of B-DNA can best be described as:
1. Semi-conservative and semi-discontinuous
2. Semi-conservative and discontinuous
3. Conservative and semi-discontinuous
4. Conservative and discontinuous
39. Identify the incorrect statement:
| 1. |
Transcription and translation can be coupled in prokaryotes |
| 2. |
DNA is a better genetic material than RNA |
| 3. |
It is the presence of structural gene in a transcription unit that defines the template and coding strands |
| 4. |
RNA polymerase III is responsible for transcription of tRNA, 5srRNA and snRNAs |
40. In the DNA double helix:
| I: |
At each step of ascent, the strand turns 34º. |
| II: |
The rise per base pair would be 0.36 nm. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
41. After cytokinesis in a plant cell, which of the following is formed first?
| 1. |
The primary cell wall |
| 2. |
The middle lamella |
| 3. |
The secondary cell wall |
| 4. |
There is no particular chronology |
42. A plasmid contains two genes for resistance to separate antibiotics. One of the two is insertionally inactivated due to insertion of alien DNA.
| I: |
The gene that was not inactivated helps in selecting the transformants. |
| II: |
The gene that was insertionally inactivated helps in selection of recombinants. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
I, II and III are correct |
43. Which of the following is not a correct difference between a C
3 and a C
4 plant?
|
Feature |
C3 Plant |
C4 Plant |
| 1. |
Cell type in which Calvin cycle takes place |
Mesophyll cells |
Bundle sheath cells |
| 2. |
Cell type in which the initial carboxylation reaction occurs |
Bundle sheath cells |
Mesophyll cells |
| 3. |
Primary carbon dioxide acceptor |
RuBP |
PEP |
| 4. |
Number of carbons in the primary carbon dioxide fixation product |
3 |
4 |
44. The substrate has to go through a higher energy state or transition state during its conversion to a product in:
| I: |
Exothermic spontaneous reactions |
| II: |
Endothermic reactions |
| III: |
Enzyme catalyzed reactions |
1. Only
I and
II
2. Only
I and
III
3. Only
II and
III
4.
I,
II, and
III
45. In algae with haplontic life cycle:
| I: |
there are no free-living sporophytes |
| II: |
mitosis in zygote results in diploid spores |
| III: |
gametophyte is the dominant photosynthetic phase |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
Only II and III are correct |
46. The branch of biology that deals with the relationships of different groups of organisms and examines their natural variation and relationships is known as:
| 1. |
Cladistics |
2. |
Systematics |
| 3. |
Taxonomy |
4. |
Identification |
47. If N is the population size, r is the intrinsic growth rate, K is the carrying capacity of the local environment, and dN/dt is the rate of change in population with time, then the Verhulst-Pearl Logistic growth equation is correctly written as:
| 1. |
dN/dt = rN[1- N/K] |
2. |
dN/dt = rN[1- K/N] |
| 3. |
dN/dt = rN + [1- N/K] |
4. |
dN/dt = rN + [1- K/N] |
48. What would be true for
Ophioglossum?
| I: |
It has the largest number of chromosomes in the known plant kingdom. |
| II: |
It has the largest number of chromosomes out of all known organisms. |
| 1. |
Both I and II |
| 2. |
Only I |
| 3. |
Only II |
| 4. |
Neither I nor II |
49. Consider the given two statements:
| I: |
In competitive enzyme inhibition, the substrate and inhibitor cannot bind to the enzyme at the same time. |
| II: |
The inhibitor always has a much higher affinity for the active site than the normal substrate. |
| 1. |
Both I and II are correct and II explains I |
| 2. |
Both I and II are correct but II does not explain I |
| 3. |
I is correct but II is incorrect |
| 4. |
I is incorrect but II is correct |
50. In Sorghum:
| 1. |
Calvin cycle takes place in Mesophyll cells |
| 2. |
Initial carboxylation reaction occurs in Bundle sheath cells |
| 3. |
Number of carbon atoms in the primary carbon dioxide acceptor is 3 |
| 4. |
PGA is the primary carbon dioxide fixation product |
Zoology - Section A
51. A functional mammary gland is a characteristic feature of:
1. all mammals
2. all female mammals
3. only primate female mammals
4. all female vertebrates
52. Mycoplasma:
| I: |
completely lack a cell wall |
| II: |
are the smallest living cells known |
| III: |
can survive without oxygen |
| 1. |
Only I and II are true |
| 2. |
Only I and III are true |
| 3. |
Only II and III are true |
| 4. |
All I, II and III are true |
53. If we consider the problem of environmental pollution, CNG is considered a better fuel for motor vehicles than diesel primarily because:
| 1. |
it burns more efficiently than diesel |
| 2. |
it cannot be adulterated |
| 3. |
it cannot be siphoned off by thieves |
| 4. |
setting up distribution points is very easy |
54. In accordance with their lifestyles, parasites evolved special adaptations that do not include:
| 1. |
the loss of unnecessary sense organs |
| 2. |
presence of adhesive organs or suckers to cling on to the host |
| 3. |
loss of digestive system |
| 4. |
low reproductive capacity. |
55. Saheli is:
| 1. |
a copper releasing intra-uterine device |
| 2. |
combination oral contraceptive pill |
| 3. |
non-steroidal oral contraceptive pill developed by CDRI |
| 4. |
is an assisted reproductive technology centre located in New Delhi |
56. What is incorrect regarding typhoid fever in human beings?
| 1. |
It is caused by a bacterium |
| 2. |
The pathogen is transmitted through contaminated food and water |
| 3. |
It can be confirmed by the Widal test |
| 4. |
No carrier state is seen in typhoid fever |
57. Identify the incorrect statement:
| 1. |
Morphine is a potent painkiller |
| 2. |
Excessive dose of cocaine can cause hallucinations |
| 3. |
Nicotine stimulates adrenal cortex to secrete mineralocorticoids |
| 4. |
Marijuana is derived from Cannabis sativa |
58. Which of the following can permanently cure ADA deficiency?
| 1. |
Bone marrow transplant |
| 2. |
Enzyme replacement therapy |
| 3. |
Introduction of the gene isolated from marrow cells producing ADA at early embryonic stages. |
| 4. |
Introduction of a functional ADA cDNA (using a retroviral vector) into peripheral lymphocytes. |
59. The mRNA codons GAG and GUG respectively code for:
1. Valine and Glutamic acid
2. Glutamic acid and Valine
3. Tryptophan and Methionine
4. Methionine and Tryptophan
60. Which of the following hormones is most important for supporting pregnancy in a human female?
1. LH
2. FSH
3. Progesterone
4. Estrogen
61. Identify the incorrectly matched pair:
| 1. |
Echinodermata |
Water vascular system |
| 2. |
Hemichordata |
Stomochord |
| 3. |
Cyclostomata |
Cartilaginous jaw |
| 4. |
Cephalochordata |
Notochord persistent throughout life |
62. At the end of the S-phase during the cell cycle in a typical eukaryotic animal cell [having 2N chromosome number and 2C DNA content at the G1 phase]:
| 1. |
The chromosome number and DNA content remain 2N and 2C respectively |
| 2. |
The chromosome number and DNA content are 4N and 2C respectively |
| 3. |
The chromosome number and DNA content are 2N and 4C respectively |
| 4. |
The chromosome number and DNA content are 4N and 4C respectively |
63. In bacteria:
| 1. |
translation can begin before the mRNA is fully transcribed |
| 2. |
removal of introns from hnRNA is carried out by ribozymes |
| 3. |
there are at least three different RNA polymerases to transcribe different RNA molecules |
| 4. |
the transcription of mRNA takes place in a template independent manner |
64. Match each item in Column I with one in Column II and choose the correct option from the codes given below:
|
Column I |
|
Column II |
| A. |
Porifera |
P |
Metagenesis |
| B. |
Cnidaria |
Q |
Flame cells |
| C. |
Ctenophora |
R |
Comb Plates |
| D. |
Platyhelminthes |
S |
Choanocyte |
|
A |
B |
C |
D |
| 1. |
R |
Q |
S |
P |
| 2. |
S |
P |
R |
Q |
| 3. |
P |
S |
Q |
R |
| 4. |
Q |
R |
P |
S |
65. Taq polymerase is:
| 1. |
is a ribozyme involved in RNA splicing in eukaryotes |
| 2. |
is a thermostable DNA polymerase used in PCR |
| 3. |
is a key reagent in ELISA |
| 4. |
an enzyme isolated from Thermus aquaticus, a fungus found in damp soils |
66. Match each item in Column I with one in COLUMN II and select the correct match from the codes given:
|
COLUMN I |
|
COLUMN II |
| A |
Saliva |
P |
Intrinsic factor |
| B |
Gastric secretion |
Q |
Nuclease |
| C |
Pancreatic juice |
R |
Lysozyme |
| D |
Succus entericus |
S |
Nucleosidase |
Codes:
|
A |
B |
C |
D |
| 1. |
R |
P |
Q |
S |
| 2. |
P |
Q |
S |
R |
| 3. |
Q |
S |
R |
P |
| 4 |
S |
R |
P |
Q |
67. Cardiac output can be increased by:
| I: |
Neural signals through sympathetic nerves |
| II: |
Neural signals through parasympathetic nerves |
| III: |
Adrenal medullary hormones |
| 1. |
Only I and II |
| 2. |
Only I and III |
| 3. |
Only II and III |
| 4. |
I, II and III |
68. You want to set up a dairy farm. Which of the following is the most important step?
1. Selection of good breeds
2. Cleanliness and hygiene
3. Processes should be mechanised
4. Employing a veterinary doctor
69. Match the items given in Column I with those in Column II and select the correct option given below:
|
Column I |
|
Column II |
| a. |
Eutrophication |
i. |
UV-B radiation |
| b. |
Enhanced greenhouse effect |
ii. |
Deforestation |
| c. |
Snow blindness |
iii. |
Nutrient enrichment |
| d. |
Slash and burn agriculture |
iv. |
Global warming |
| 1. |
a-ii b-i c-iii d-iv |
| 2. |
a-i b-iii c-iv d-ii |
| 3. |
a-iii b-iv c-i d-ii |
| 4. |
a-i b-ii c-iv d-iii |
70. What is not correct regarding malpighian tubules in cockroaches?
| 1. |
They are present at the junction of midgut and hindgut |
| 2. |
They are lined by glandular and ciliated cells |
| 3. |
They are 100-150 in number and arranged in a ring-like manner |
| 4. |
Though they are excretory structures, they do not have osmoregulatory functions |
71. HIV infection can cause life-threatening opportunistic infections in humans as it attacks and destroys:
1. Helper T cells
2. Cytotoxic T cells
3. Suppressor T cells
4. B cells
72. Which of the following is not a salient feature of the human genome?
| 1. |
Less than 2 percent of the genome codes for proteins |
| 2. |
Chromosome 1 has most genes [2968] and the Y has the fewest [231] |
| 3. |
The function of almost all the discovered genes is known |
| 4. |
Repeated sequences make up a very large portion of human genome |
73. Which of the following statements regarding epithelium is not correct?
| 1. |
Blood capillaries and alveoli are lined with simple sqamous epithelium |
| 2. |
Fallopian tubes are lined with ciliated epithelium |
| 3. |
Proximal convoluted tubule of the nephron is lined with compound cuboidal epithelium |
| 4. |
Columnar brush bordered epithelium [with microvilli] is found in the lining of small intestine |
74. All the following are examples of synovial joints except:
1. joint between carpal bones
2. joint between adjacent thoracic vertebrae
3. joint between atlas and axis vertebra
4. joint between the humerus and pectoral girdle
75. Work of which of the following scientists was the basis upon which the discipline of biotechnology was founded?
| 1. |
Cohen and Boyer |
| 2. |
Watson and Crick |
| 3. |
Hershey and Chase |
| 4. |
Avery, Macleod and McCarty |
76. Cells in the human body that secrete products like lipids and steroids are likely to have an abundance of:
| 1. |
Smooth endoplasmic reticulum |
| 2. |
Rough endoplasmic reticulum |
| 3. |
Lysosomes |
| 4. |
Mitochondria |
77. Identify the incorrect statement:
| 1. |
Bad ozone is the one that is formed in the troposphere |
| 2. |
Good ozone is the one that is found in the stratosphere |
| 3. |
In human eye, cornea absorbs UV-A and UV-C and not UV-B |
| 4. |
The thickness of the ozone layer is measured in Dobson units |
78. Thermoregulation is likely to be energetically most expensive for:
1. Blue whale
2. Humans
3. Humming birds
4. Elephant
79. When two genes in a dihybrid cross are situated on the same chromosome close to each other, the proportion of parental gene combinations is:
| 1. |
much higher than the non-parental type |
| 2. |
much lower than the non-parental type |
| 3. |
equal to the non-parental type |
| 4. |
not seen at all |
80. The ‘H-zone’ in a sarcomere:
| 1. |
is the central part of thick filaments not overlapped by thin filaments |
| 2. |
is the peripheral part of thick filaments not overlapped by thin filaments |
| 3. |
is the central part of thick filaments overlapped by thin filaments |
| 4. |
is the peripheral part of thick filaments overlapped by thin filaments |
81. The axonal membrane:
| 1. |
is relatively more permeable to potassium ions than to sodium ions during its resting stage |
| 2. |
is relatively more permeable to sodium ions than to potassium ions during its resting stage |
| 3. |
is equally permeable to potassium ions and to sodium ions during its resting stage |
| 4. |
is impermeable to potassium ions and to sodium ions during its resting stage |
82. Which of the following pulmonary capacities is not correctly matched with its constituent pulmonary volumes?
| 1. |
Inspiratory capacity |
Tidal volume + Inspiratory reserve volume |
| 2. |
Expiratory capacity |
Tidal volume + Expiratory reserve volume |
| 3. |
Functional residual capacity |
Residual volume + Inspiratory reserve volume |
| 4. |
Vital capacity |
Tidal volume + Inspiratory reserve volume + Expiratory reserve volume |
83. In human embryonic development, the first movements of the foetus and the appearance of hair on the head are usually observed:
| 1. |
By the end of the second month |
| 2. |
During the third month |
| 3. |
By the end of the fourth month |
| 4. |
During the fifth month |
84. Malignant tertian malaria is caused by:
| 1. |
Plasmodium vivax |
| 2. |
Plasmodium ovale |
| 3. |
Plasmodium falciparum |
| 4. |
Plasmodium malariae |
85. Identify the incorrect statement regarding bile:
1. It can activate lipases
2. It contains digestive enzymes working at acidic pH
3. It contains bile pigments
4. It helps in emulsification of fats
Zoology - Section B
86. Identify the incorrectly matched pair
| 1. |
Pineal |
Located on the ventral side of the forebrain |
| 2. |
Thymus |
Located on the ventral side of aorta between lungs |
| 3. |
Adrenal |
Located at the superior pole of each kidney |
| 4. |
Parathyroid |
Located on the back side of the thyroid gland |
87. Sickle cell anaemia is inherited as an autosomal recessive condition. What proportion of progeny will be normal as a result of mating between two individuals who are carriers for the trait but are themselves not affected?
| 1. |
25% |
2. |
100% |
| 3. |
50% |
4. |
75% |
88. In a healthy adult human being, on an average:
| 1. |
about 25 – 30 mg of urea is excreted out per day |
| 2. |
125 ml per minute urine is produced |
| 3. |
about 200 L carbon dioxide is removed by the lungs per minute |
| 4. |
1100-1200 ml of blood is filtered by the kidneys per minute |
89. Given below are two statements:
| Assertion (A): |
Some infections with gram-negative bacteria can lead to life-threatening septic shock. |
| Reason (R): |
Lipopolysaccharide layer in gram-negative bacteria can trigger an innate immune response. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
90. Which of the following does not involve a fungus?
1. Commercial production of ethanol
2. Commercial production of citric acid
3. Commercial production of statins
4. Commercial production of streptokinase
91. Consider the given two statements:
| I: |
Diabetes insipidus is caused by the deficiency of ADH and diabetes mellitus is caused by the deficiency of insulin. |
| II: |
Patients suffering from diabetes insipidus will have glycosuria but patients suffering from diabetes mellitus will not have glycosuria. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
92. Consider the given two statements:
| I: |
The type of antigen present on the RBCs of a person with blood group type A is A. |
| II: |
The type of antibodies present in the plasma of a person with blood group type A is anti-A. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
93. Consider the two statements:
| I: |
Haemoglobin in humans adapted to very high altitudes has a lower binding affinity for oxygen than that found in humans living at the sea level. |
| II: |
Lower affinity of haemoglobin to oxygen means more oxygen can bind to haemoglobin even at low partial pressures |
| 1. |
Both I and II are correct and II explains I |
| 2. |
Both I and II are correct but II does not explain I |
| 3. |
I is correct but II is incorrect |
| 4. |
Both I and II are incorrect |
94. Identify the incorrectly matched pair:
|
Bio-control agent |
Pest controlled |
| 1. |
Lady bird |
Aphids |
| 2. |
Dragon fly |
Mosquito |
| 3. |
Trichoderma |
Fungi |
| 4. |
Baculoviruses |
Nematodes |
95. In the human ear:
| 1. |
Tectorial membrane separates the scala vestibuli and scala media |
| 2. |
Reissner’s membrane separates the scala media and the scala tympani |
| 3. |
Basilar membrane lies over the hair cells of the organ of Corti |
| 4. |
Tympanic membrane separates the external ear and the middle ear |
96. Luteinizing hormone:
| I: |
acts at the Leydig cells and stimulates synthesis and secretion of androgens. |
| II: |
acts on the Sertoli cells and stimulates secretion of some factors which help in the process of spermiogenesis. |
| 1. |
Only I is correct |
2. |
Only II is correct |
| 3. |
Both I and II are correct |
4. |
Both I and II are incorrect |
97. Cr-Jacob disease (CJD) in humans is caused by:
| 1. |
An agent which causes mosaic formation in plants |
| 2. |
An agent having DNA |
| 3. |
An agent which consists of abnormally folded protein and is similar in size to viruses |
| 4. |
The same agent which causes potato spindle tuber disease |
98. Adrenaline, Angiotensin II and ADH can all:
| 1. |
cross target cell membrane and influence gene activity |
| 2. |
cause vasoconstriction |
| 3. |
be secreted by postganglionic sympathetic neurons |
| 4. |
increase blood glucose levels |
99. What would be the number of intercostal spaces in the human thoracic cage?
100. Which of the following Intrauterine devices are most likely to make the uterus unsuitable for implantation and the cervix hostile to the sperms?
1. Lippes loop
2. Multiload 375
3. Progestasert
4. Cu T
Chemistry - Section A
101. pH of acid rain is approximately
1. 5.6
2. 6.2
3. 6.7
4. 7
102. The most paramagnetic species is:
1. \(Ni^{2+}(Z = 28)\)
2. \(Fe^{2+}(Z = 26)\)
3. \(Mn^{2+}(Z = 25)\)
4. \(V^{3+}(Z = 23)\)
103. The correct order of the stability of ions is:
| 1. |
\(Ge^{2+} < Ge^{4+} \) |
2. |
\(Sn^{2+} < Sn^{4+} \) |
| 3. |
\(Pb^{2+} > Pb^{4+} \) |
4. |
All of the above |
104. The hybridised state of Cr & number of unpaired electrons in [Cr(NH3)6]Cl3, respectively are
1. sp3d2 & 3
2. d2sp3 & 3
3. d2sp3 & 2
4. sp3d2 & 2
105. In the preparation of
\(H_2O_2\), when 50%
\(H_2SO_4\) solution is electrolysed, the ion discharged at the anode is:
| 1. |
\(SO^{2-}_4\) |
2. |
\(OH^-\) |
| 3. |
\(HSO^-_4\) |
4. |
\(Cl^-\) |
106. In the metallurgy of copper, 'Matte' is formed in smelting process. The composition of 'Matte' is
1. FeS with small amount of Cu2S
2. Cu2S with small amount of FeS
3. Cu2S with small amount of FeO
4. FeS with small amount of Cu2O
107. An element (Atomic mass = 50 g/mol) having fcc structure has a unit cell edge length of 400 pm. The density of the element is-
1. 5.188 g/cc
2. 10.376 g/cc
3. 2.56 g/cc
4. 1.2 g/cc
108. Which among the following species has the highest ionization potential?
1. \(F^-\)
2. \(Cl^-\)
3. \(O^-\)
4. \(S^-\)
109. The most volatile halogen acid is:
| 1. |
HF |
2. |
HCl |
| 3. |
HBr |
4. |
HI |
110. The correct representation of Nylon-6, 6 is-
111. The correct order of reactivity of the following compounds towards electrophilic substitution reaction will be:
1. I > II > III > IV > V
2. I > II > IV > V > III
3. I > IV > II > V > III
4. I > II > IV > III > V
112. Which of the following situations results in a reduction of the system's entropy?
1. Boiling of egg
2. Combustion of benzene at 27oC
3. Combustion of benzene at 127oC
4. Dissolution of sugar in water
113. When fused form of saline hydride is electrolysed, then hydrogen gas is evolved at
1. anode
2. cathode
3. maybe at anode or at cathode
4. hydrogen gas is not formed
114. The correct order among the following is:
| 1. |
\(\mathrm{I^- > I >I^+}\) (Radii) |
| 2. |
\(\mathrm{I^- > I > I^+}\) (Ionisation energy) |
| 3. |
\(\mathrm{Li^+}\) (aqueous) < \(\mathrm{Na^+}\) (aqueous) < \(\mathrm{K^+}\) (aqueous) (Radii) |
| 4. |
\(\mathrm{F > Cl > Br > I}\) (Electron Affinity) |
115. Which pair of compounds are anomers to each other?
1. Glucose & Mannose
2. Glucose & Galactose
3. \(\alpha\)-D Glucose & \(\beta\)-D Glucose
4. Glucose & Fructose
116. What is the correct order of boiling points for the hydrides of 15th group ?
1. \(\mathrm{NH}_3>\mathrm{BiH}_3>\mathrm{SbH}_3>\mathrm{AsH}_3>\mathrm{PH}_3\)
2. \(\mathrm{BiH}_3>\mathrm{NH}_3>\mathrm{SbH}_3>\mathrm{AsH}_3>\mathrm{PH}_3\)
3. \(\mathrm{BiH}_3>\mathrm{SbH}_3>\mathrm{NH}_3>\mathrm{AsH}_3>\mathrm{PH}_3\)
4. \(\mathrm{BiH}_3>\mathrm{SbH}_3>\mathrm{AsH}_3>\mathrm{NH}_3>\mathrm{PH}_3\)
117. Given the following reaction:

The compound
\('A'\) in the above reaction is:
118. The most stable free radical, amongst the following, is
1. Allyl
2. Benzyl
3. Vinyl
4. Phenyl
119. If an optically active alkyl halide is reacted with nucleophile through SN1 mechanism then which of the following statement is correct ?
1. 100% racemisation takes place
2. Maximum racemisation with partial inversion takes place
3. Maximum inversion with partial racemisation takes place
4. 100% inversion takes place
120. For an exothermic reaction, the value of activation energy for forward reaction is
1. Less than \(\Delta\)H
2. More than \(\Delta\)H
3. Equal to \(\Delta\)H
4. All of the above are possible
121. Which compound among the following has the highest lattice energy?
1. LiF
2. MgO
3. NaF
4. CaO
122. The cationic detergent, among the following, is:
1. Sodium lauryl sulphate
2. Sodium Dodecylbenzene sulphate
3. Cetyl trimethyl ammonium bromide
4. None of these
123. A buffer solution is defined as a solution whose pH remains practically constant even when small amounts of an acid or a base are added to it.
Henderson's equation is used to determine pH of buffer mixtures of different types:
For acidic buffer, Henderson's equation is :
pH= pKa + log \([Salt] \over [Acid]\) (ka = ionisation constant of weak acid)
For basic buffer, Henderson's equation is :
POH = Pkb + log \([Salt] \over [Base]\) (kb = ionisation constant of weak base)
How many moles of HCl are required with 0.01 mole NaCN to prepare a buffer solution of pH =9?
[Given: Ka of HCN = \(1 \times 10^{-10}\)]
1. 0.009
2. 0.09
3. 0.9
4. Buffer solution cannot formed
124. When 50% \(H_2SO_4\) solution is electrolysed using inert electrodes, the products formed at anode and cathode, respectively, are
1. \(H_2 ~and ~O_2\)
2. \(O_2 ~and~H_2O_2\)
3. \(H_2O_2 ~and~ H_2\)
4. \(O_2 ~and ~H_2\)
125. Which, of the following, laws of chemical combination is followed by \(N_2O~, NO \) and \(N_2O_3\) ?
1. Law of conservation of mass
2. Law of multiple proportions
3. Law of constant proportions
4. Gay Lussac's law of constant volume
126. Two sets of quantum numbers with the same number of radial nodes are :
1. n = 3, l = 0, ml = 0 and n = 2, l = 0, ml = 0
2. n = 3, l = 2, ml = 0 and n = 2, l = 1, ml = 0
3. n = 3, l = 1, ml = –1 and n = 2, l = 1, ml = 0
4. n = 3, l = 1, ml = 1 and n = 2, l = 1, ml = 0
127. When FeCl3 solution is added in a freshly prepared precipitate of Fe(OH)3, it forms
1. positively charged colloid
2. positively charged precipitate
3. negatively charged colloid
4. negatively charged precipitate
128. When chromite ore is heated with \(Na_2CO_3 \) and \(O_2\) then the compound of chromium formed and its colour, respectively, are:
1. \(Na_2CrO_4 \) , yellow
2. \(Na_2Cr_2O_7 \) , yellow
3. \(Na_2CrO_4 \) , orange
4. \(Na_2Cr_2O_7 \) , orange
129. Carbonyl compounds reacts with ammonia derivatives through substitution reaction. In these reactions, what pH value needs to be maintained?
1. 2 to 3
2. 4 to 5
3. 6 to 7
4. 9 to 10
130. The minimum boiling azeotrope among the following mixtures is:
1. Chloroform and Acetone
2. Ethanol and \(H_2O \)
3. Benzene and Toluene
4. Acetic acid and \(H_2O \)
131. Incorrect order of solubility in water for the following compounds will be
\(\begin{aligned} &\text { 1. } \mathrm{Li}_{2} \mathrm{CO}_{3}<\mathrm{Na}_{2} \mathrm{CO}_{3}<\mathrm{K}_{2} \mathrm{CO}_{3}<\mathrm{Rb}_{2} \mathrm{CO}_{3} \\ &\text { 2. } \mathrm{Be}(\mathrm{OH})_{2}>\mathrm{Mg}(\mathrm{OH})_{2}>\mathrm{Ca}(\mathrm{OH})_{2}>\mathrm{Sr}(\mathrm{OH})_{2} \\ &\text { 3. } \mathrm{BeSO}_{4}>\mathrm{MgSO}_{4}>\mathrm{CaSO}_{4}>\mathrm{Sr} \mathrm{SO}_{4} \\ &\mathrm 4. {LiOH}>\mathrm{NaOH}>\mathrm{KOH}>\mathrm{CsOH} \end{aligned}\)
132. Which of the following acts as a lewis acid?
1. \(NH_3\)
2. \(SnCl_4\)
3. \(CCl_4\)
4. None of above
133. The boiling point of 1M Urea solution is 101°C. At the same conditions of temperature and pressure, the boiling point of 1M KCl solution will be (Assume that KCl is 100% dissociated)
1. 102°C
2. 202°C
3. 104°C
4. Data insufficient
134. Which compound does not give Lassaigne's test for nitrogen?
| 1. |
Urea |
2. |
\(\mathrm {NH_2OH}\) |
| 3. |
 |
4. |
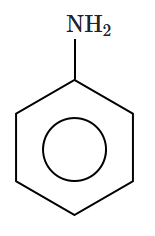 |
135. What will be the total volume of the mixture when 32 gm of oxygen and 3 gm of hydrogen are mixed and kept at 1 atm and
0 °C?
1. 22.4 litres
2. 44.8 litres
3. 56 litres
4. 67.2 litres
Chemistry - Section B
136. The molarity of
\(NaNO_3 \) solution (molecular weight of
\(NaNO_3\) = 85) is 1M. The density of the solution is 1.25 gm/ml. The molality of the solution, approximately, will be:
1. 0.80
2. 0.86
3. 0.96
4. 1
137.
| Assertion (A): |
Ortho nitrophenol is more volatile than para nitrophenol. |
| Reason (R): |
In ortho nitrophenol, intramolecular as well as intermolecular hydrogen bonding is present while in para nitrophenol only intermolecular hydrogen bonding is present. |
| 1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. |
(A) is True but (R) is False. |
| 4. |
Both (A) and (R) are False. |
138. When magnesium is burnt in the air, the product formed is
1. \(MgO\)
2. \(Mg_3N_2\)
3. \(MgO + Mg_3N_2\)
4. No reaction takes place
139. For the spontaneity of freezing of water, which condition must be imposed?
1. \(\Delta H>T\Delta S \)
2. \(\Delta H<T\Delta S \)
3. \(\Delta H=T\Delta S \)
4. Freezing of water is always non-spontaneous
140. Which of the following is not an ambidentate ligand?
| 1. |
\(CN^-\) |
2. |
\(NH_3\) |
| 3. |
\(SCN^-\) |
4. |
\(NO^-_2\) |
141. Given the equation
\(CH_3 - C \equiv CH + H_2O \xrightarrow [20\% ~H_2SO_4] {1\% ~HgSO_4} A\)
The product 'A' is:
1. \(CH_3 - CH_2 - CHO \)
2. \(CH_3 - CO - CH_3\)
3. \(CH_3 - CH_2 -CH_2 - OH\)
4. \( CH_3 - CH(OH) - CH_3\)
142. The hybridisation of Xe & shape of XeO
2F
2, respectively, are:
| 1. |
sp3d & see saw |
| 2. |
sp3d & T-shape |
| 3. |
sp3d2 & square planar |
| 4. |
sp3d & Linear |
143. Given below are some reactions. The reaction which is not feasible is
1. \(2KClO_3 + I_2 \rightarrow 2KIO_3 + Cl_2 \uparrow\)
2. \(2KCl + I_2 \rightarrow 2KI + Cl_2 \uparrow\)
3. Both 1 and 2 are not feasible
4. \(2KI + Cl_2 \rightarrow 2KCl + I_2\)
144. Which among the following is a primary alcohol?
| 1. |
\(CH_3- OH\) |
| 2. |
\(CH_3-CH_2 -OH\) |
| 3. |
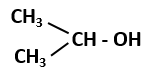 |
| 4. |
Both (1) and (2) |
145. The most basic oxide among the following is:
1. \(CrO\)
2. \(Cr_2O_3\)
3. \(CrO_3\)
4. \(Cr_2O_4\)
146. Consider the following reaction:
\(A(g) \rightarrow B(g) + C (g)~\)
The initial pressure is\(P^o\) and the pressure at 't' time is\(P_t (P_t>P^o)\). Assuming that the above reaction is of first order, the rate expression will be:
1. \(K = {2.303 \over t}log {P^o \over P_t}\)
2. \(K = {2.303 \over t}log {2P^o \over P^o - P_t}\)
3. \(K = {2.303 \over t}log {P^o \over 2P^o - P_t}\)
4. \(K = {2.303 \over t}log {2P^o \over 3P^o- P_t}\)
147. Given \(A(g) + 3B(g) \rightleftharpoons4C(g)\) and initial concentration of A is equal to that of B. The equilibrium concentration of A and C are equal. The value of \(K_C\) is :
1. 0.08
2. 0.8
3. 8
4. 80
148. Compounds with molecular formula
\(C_5H_{12}O\) cannot show :
| 1. |
Tautomerism |
2. |
Position isomerism |
| 3. |
Metamerism |
4. |
Functional isomerism |
149. Which of the following carbonyl oxygen forms a stronger bond with
\(H_2O \) molecule?
150. Which, of the following, compounds is most reactive towards electrophilic substitution reaction?
1. Phenol
2. Toluene
3. Aniline
4. Methoxy benzene
Physics - Section A
151. An ideal gas forms the working substance of a Carnot engine, and is taken around the Carnot cycle. We form the integral:
\(I=\int\dfrac{dQ}{T},\)
where
\(dQ\) is the heat supplied to the gas and
\(T\) is the temperature of the gas. The integral is evaluated over the entire cycle. The value of the integral
\(I\) is:
| 1. |
zero |
| 2. |
negative |
| 3. |
positive |
| 4. |
non-negative(positive or zero) |
152. The gravitational potential energy of a particle of mass
\(m\) increases by
\(mgh,\) when it is raised through a height
\(h\) in a uniform gravitational field "
\(g\)". If a particle of mass
\(m\) is raised through a height
\(h\) in the earth's gravitational field (
\(g\): the field on the earth's surface) and the increase in gravitational potential energy is
\(U\), then:
| 1. |
\(U > mgh\) |
| 2. |
\(U < mgh\) |
| 3. |
\(U = mgh\) |
| 4. |
any of the above may be true depending on the value of \(h,\) considered relative to the radius of the earth. |
153. The speed of electromagnetic waves is:
| 1. |
higher in vacuum compared to other media. |
| 2. |
lower in vacuum compared to other media. |
| 3. |
independent of the medium. |
| 4. |
lower in some media than in vacuum, and higher in others. |
154. Given below are two statements:
| Statement I: |
The centre-of-mass of a system of particles lying on a straight line must lie between the two extreme particles. |
| Statement II: |
The centre-of-mass of a system of bodies moving with different velocities, cannot be moving with constant velocity. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
155. Consider the circuit shown in the figure given below. The input voltage is
\(V_i\), the current through the diode is
\(I_z\), and the current through the load is
\(I_L\).

| 1. |
\(\text{If} ~{V_i}= 15~\text{V}~\text{and}~{R_L}= 40~\Omega, ~\text{and}~{I_z}= 0.375~\text{A}\) |
| 2. |
\(\text{If} ~{V_i}= 10~\text{V}~\text{and}~{R_L}= 5~\Omega, ~\text{and}~{I_L}=1~\text{A}\) |
| 3. |
\(\text{If} ~{V_i}= 20~\text{V}~\text{and}~{R_L}= 20~\Omega, ~\text{and}~{I_z}= 0.25~\text{A}\) |
| 4. |
\(\text{If} ~{V_i}= 20~\text{V}~\text{and}~{R_L}= 10~\Omega, ~\text{and}~{I_z}= 0.5~\text{A}\) |
156. A horizontal
force \(F\) is applied to a uniform solid sphere at rest, so that its line of action passes through the
mid-point (\(P\)) of the vertical radius
\(OA;O\) being the centre of the sphere
(mass : \(m\)). The acceleration of the uppermost
point \(A\) is:

| 1. |
equal to \(\dfrac{F}{m}.\) |
| 2. |
greater than \(\dfrac{F}{m}.\) |
| 3. |
less than \(\dfrac{F}{m}.\) |
| 4. |
unpredictable, and depends on the radius of the sphere. |
157. Two circuits:
\((1)\) an
\(L\text-R\) circuit and
\((2)\) an
\(R\text-C\) circuit are driven by the same alternating current. The phase difference between the current and the voltage is twice in the
\(1\)st case with respect to the
\(2\)nd case and both the angles add up to
\(90^\circ.\) The resistances are equal in both cases. The ratio of their reactances (first: second) is:
| 1. |
\(\sqrt3:1\) |
2. |
\(1:\sqrt3\) |
| 3. |
\(3:1\) |
4. |
\(2:1\) |
158. Two cars
\(A\) and
\(B,\) each
\(5~\text{m}\) long, are travelling along parallel lanes with speeds of
\(36~\text{km/hr}\) and
\(72~\text{km/hr}.\) The car
\(B\) is just behind the car
\(A,\) but is beginning to overtake. What will be the distance covered by the car
\(B\) by the time it is just ahead of the car
\(A?\)

1.
\(10~\text{m}\)
2.
\(15~\text{m}\)
3.
\(20~\text{m}\)
4.
\(25~\text{m}\)
159. The
\(\text{DC}\) time constant of an
\(L\)-\(R\) circuit is the same as that of an
\(R\)-\(C\) circuit where the inductor in the first circuit was replaced by a capacitor. The value of the resistance
\(R\) equals:
| 1. |
\(\dfrac{1}{2\pi}\sqrt{\dfrac{L}{C}}\) |
2. |
\(\sqrt{\dfrac{L}{C}}\) |
| 3. |
\(2\pi\sqrt{\dfrac{L}{C}}\) |
4. |
\(2\sqrt{\dfrac{L}{C}}\) |
160. A
\(20~\text V\) cell delivers the same power to a
\(2~\Omega\) resistor as it does to an
\(8~\Omega\) resistor. The internal resistance of the cell is:
| 1. |
\(2~\Omega\) |
2. |
\(4~\Omega\) |
| 3. |
\(6~\Omega\) |
4. |
\(1.6~\Omega\) |
161. A uniformly charged metallic sphere holds a total charge
\(Q_0\) and has a potential
\(V_0.\) The total potential energy stored is:
| 1. |
\(Q_0V_0\) |
2. |
\(\dfrac{Q_0V_0}{2}\) |
| 3. |
\(\dfrac{Q_0V_0}{4}\) |
4. |
\(-Q_0V_0\) |
162. A charged particle of charge
\(q,\) mass
\(m\) moves in a circular path under the action of a uniform magnetic field, whose flux through this path is
\(\phi.\) The magnetic moment due to the particle's motion is:
| 1. |
\(\dfrac{q^2\phi}{2m}\) |
2. |
\(\dfrac{q^2\phi}{2\pi m}\) |
| 3. |
\(\dfrac{q^2\phi}{m}\) |
4. |
\(\dfrac{q^2\phi}{\pi m}\) |
163. Given below are two statements:
| Statement I: |
If an ideal gas expands without any heat being supplied to it, then its temperature does not change. |
| Statement II: |
If an ideal gas expands freely without any heat being supplied to it, then its internal energy remains unchanged. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
164. Consider an electric field of the form:
\(\vec E=K(y\hat i+x\hat j)\)
where
\(K\) is a constant, and
\(x,y\) are the coordinates.
| Statement I: |
If a charged particle is taken along the \(x\)-axis, no work will be done by the electric field. |
| Statement II: |
This electric field is conservative in nature i.e. it can be derived from a potential: \(V(x,y)=C-Kxy\) |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
165. The breaking stress in two wires of different materials
\(A,B\) are in the
ratio: \(\dfrac{S_A}{S_B}=\dfrac12,\) while their radii are in the
ratio: \(\dfrac{r_A}{r_B}=\dfrac12.\) The tensions under which they break are
\(T_A\) and
\(T_B.\) Then
\(\dfrac{T_A}{T_B}=\)?
| 1. |
\(2\) |
2. |
\(\dfrac14\) |
| 3. |
\(\dfrac18\) |
4. |
\(\dfrac1{2\sqrt2}\) |
166. Consider a square carrom board \(ABCD\) of size \({3~ \text{ft}} \times 3~\text{ft}. \) A piece moves 'from' pocket \(A\) (close from a pocket), strikes side \(BC\) and then side \(AD\), and reaches pocket \(C\). If the piece is reflected perfectly from each side, then the ratio of the \(x,y\) components of velocity is given by \(\dfrac{v_x}{v_y}=\)
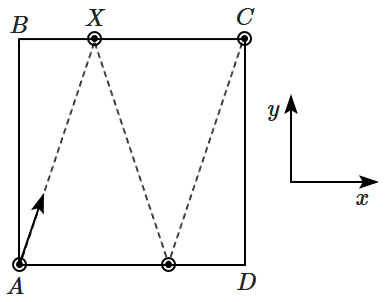
| 1. |
\(2\) |
2. |
\(\dfrac{1}{2}\) |
| 3. |
\(3\) |
4. |
\(\dfrac{1}{3}\) |
167. A straight wire
\(AB\) of length
\(L\) rotates about
\(A,\) with an angular speed
\(\omega.\) A constant magnetic field
\(\mathbf B\) acts into the plane, as shown.

| Assertion (A): |
The average induced electric field within the wire has a magnitude of \(\dfrac12B\omega L.\) |
| Reason (R): |
The induced electric field is the motional EMF per unit length, and the motional EMF is \(\dfrac12B\omega L^2.\) |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
168. A horizontal force
\(F\) acts on a
\(2~\text{kg}\) block placed on a smooth horizontal plane. It varies with time
\(t\) as shown in the figure. The block is initially at rest.

The work done by the force until
\(t=2~\text s,\) equals:
1.
\(6.25 ~\text J\)
2.
\(4.5 ~\text J\)
3.
\(2.25 ~\text J\)
4.
\(1.5~\text J\)
169. A small sphere of density \(\rho\) is completely submerged in a liquid of density \(3\rho\) and released. The magnitude of its acceleration just after release equals:
1. \(g\)
2. \(\dfrac{g}{3}\)
3. \(2g\)
4. \(3g\)
170. If two convex lenses of powers
\(P_1,P_2\) be placed close together, co-axially, the combination behaves as a lens of power:
| 1. |
\(P_1+P_2\) |
2. |
\(|P_1-P_2|\) |
| 3. |
\({\Large\frac{P^2_1}{P_2}}\) |
4. |
\({\Large\frac{P^2_2}{P_1}}\) |
171. Given below are two statements:
| Assertion (A): |
The average velocity of the molecules of an ideal gas increases when the temperature rises. |
| Reason (R): |
The internal energy of an ideal gas increases with temperature, and this internal energy is the random kinetic energy of molecular motion. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
172. A rod of mass \(M\) and length \(L\) is suspended vertically at its highest point. The rod is held so that it is horizontal and free to rotate about \({A}\) and then released. There is no friction anywhere.

The force exerted by the hinge at \({A},\) when the rod is at its lowest position is:
1. \(2~Mg\)
2. \(3~Mg\)
3. \(4~Mg\)
4. \(2.5~Mg\)
173. Two identical right-angled prisms are placed back to back as shown. A ray of light, incident on the first prism, making an angle
\(\theta\) with its surface – passes through the system and emerges parallel to itself. The refractive index of the material of the prism is:

1.
\(\sin\theta\)
2.
\(\cos\theta\)
3.
\(\tan\theta\)
4.
\(\cot\theta\)
174. The work done by a force \(F=2x^2\) (newton) in a displacement from \(x=0\) (origin) to \(x=3\) m is:
1. \(54\) J
2. \(27\) J
3. \(18\) J
4. \(9\) J
175. Light of wavelength \(4000~\mathring{A}\) is incident on a metal whose work function is \(2.0\text{ eV}.\) The fastest photo-electrons emitted have an energy of: (Take \(hc=12400\) eV-\(\mathring A\))
1. \(0.5\text{ eV}\)
2. \(3.1\text{ eV}\)
3. \(1.1\text{ eV}\)
4. \(2\text{ eV}\)
176. A wheel of radius \(R\) rolls on a horizontal flat surface, without slipping. The speed of its center is constant and equals \(v\). The acceleration of the point of contact, of the wheel, is:
1. zero
2. \(\frac{v^2}{R},\) upward
3. \(\frac{v^2}{R},\) downward
4. \(\frac{v^2}{2R},\) upward
177. Two particles move with the same uniform angular speed around two different circles of radii \(r,~ 2r.\) Their accelerations are in the ratio:
1. \(1:2\)
2. \(1:4\)
3. \(2:1\)
4. \(4:1\)
178. Assume that the magnetic field of the earth is due to a small magnetic dipole, placed at the centre of the earth
(radius: \(R\)). The magnetic field at the equator is
\(B_e.\) The dipole moment of the dipole is:
| 1. |
\(\dfrac{\pi}{\mu_0}\left(B_eR^3\right )\) |
2. |
\(\dfrac{2\pi}{\mu_0}\left(B_eR^3\right )\) |
| 3. |
\(\dfrac{4\pi}{\mu_0}\left(B_eR^3\right )\) |
4. |
\(\dfrac{2}{\mu_0}\left(B_eR^3\right )\) |
179. A source of sound of frequency \(160~\text{Hz},\) when moving with a speed \(v\) towards a fixed identical vibrating source, produces a beat frequency of \(10~\text{Hz}\) in the ground frame. The speed of sound in air is \(340~\text{m/s}.\) The speed \(v\) equals:
1. \(20~\text{m/s}\)
2. \({\dfrac{68}{3}}~\text{m/s}\)
3. \({\dfrac{85}{4}}~\text{m/s}\)
4. \(25~\text{m/s}\)
180. A particle moves in the x-y plane according to the equation
\(x = A \cos^2 \omega t\) and \(y = A \sin^2 \omega t\)
Then, the particle undergoes:
| 1. |
uniform motion along the line \(x + y = A\) |
| 2. |
uniform circular motion along \(x^2 + y^2 = A^2\) |
| 3. |
SHM along the line \(x + y = A\) |
| 4. |
SHM along the circle \(x^2 + y^2 = A^2\) |
181. Equal and opposite charges are placed at the two vertices of an equilateral triangle, giving a dipole moment
\(p.\) A small dipole
\(p'\) is placed at the third vertex, parallel to the previous dipole. If the electric fields due to both dipoles
\(p,~p',\) at the mid-point of the dipole
\(p,\) are equal, then
\(\dfrac{p'}{p}\) equals:
| 1. |
\(3\sqrt3\) |
2. |
\(\dfrac{3\sqrt3}{2}\) |
| 3. |
\(\sqrt3\) |
4. |
\(\dfrac{\sqrt3}{2}\) |
182. Light having the wavelength equal to the first line of the Lyman series is incident on a metal having a work function of \(6\text{ eV}.\) The energy of the fastest photo-electron emitted is:
1. \(7.6\text{ eV}\)
2. \(4.2\text{ eV}\)
3. \(2.1\text{ eV}\)
4. \(0.8\text{ eV}\)
183. By writing down the truth table or otherwise, determine the output
\(X\) of the circuit given below.

| 1. |
\(A\) |
2. |
\(B\) |
| 3. |
\(A~\text{or}~B\) |
4. |
NOT \((A)\) |
184. Match the units of some physical quantities in
List-I with their corresponding dimensions in
List-II.
| List-I |
List-II |
| (a) |
Ohm |
(i) |
\([ML^2T^{-2}A^{-1}]\) |
| (b) |
Farad |
(ii) |
\([ML^2T^{-2}A^{-2}]\) |
| (c) |
Henry |
(iii) |
\([M^{-1}L^{-2}T^4A^2]\) |
| (d) |
Weber |
(iv) |
\([ML^2T^{-3}A^{-2}]\) |
| 1. |
(a)-(i), (b)-(ii), (c)-(iv), (d)-(iii) |
| 2. |
(a)-(iv), (b)-(iii), (c)-(i), (d)-(ii) |
| 3. |
(a)-(ii), (b)-(iii), (c)-(i), (d)-(iv) |
| 4. |
(a)-(iv), (b)-(iii), (c)-(ii), (d)-(i) |
185. Of the radiations/particle beams mentioned below, which one causes maximum ionisation when passing through matter?
1. neutron beam
2. \(\beta\)-rays
3. \(\gamma \)-rays
4. infra-red light
Physics - Section B
186. Light of wavelength
\(\lambda\) is just able to cause the emission of photoelectrons from a metallic surface. If the metallic surface moves relative to the light source with a velocity
\(v,\) it will be able to cause photoemission if:
| 1. |
the surface moves away from the light source. |
| 2. |
the surface moves towards the light source. |
| 3. |
the light source moves away from the surface. |
| 4. |
both light source and surface move away from each other. |
187. The moment of inertia of a uniform right-angled triangular lamina
(mass: \(m\)) \(\Delta ABC \) about an axis passing through
\(C,\) perpendicular to its plane is:

| 1. |
\(m\left(\dfrac{a^2 +b^2}{3}\right ) \) |
2. |
\(m\left(\dfrac{a^2 +b^2}{6}\right) \) |
| 3. |
\(m\left(\dfrac{a^2 +b^2}{12}\right) \) |
4. |
\(m\left(\dfrac{a^2 +b^2}{2}\right) \) |
188. A ball thrown vertically upward rises to a maximum height of \(8\) m, then falls down and rebounds to a height of \(2\) m. After the next rebound, it will rise to:
1. \(1\) m
2. \(0.5\) m
3. \(0.25\) m
4. \(0.125\) m
189. Two concentric metallic spheres, surface areas
\(A_1,A_2\) and separation
\(d\), have a capacitance
\(C_0.\) If a parallel plate capacitor is built with the same separation
\(d,\) and has the same capacitance
\(C_0\) then its plate area will be:
| 1. |
\(\dfrac{A_1+A_2}{2}\) |
2. |
\(\sqrt{A_1A_2}\) |
| 3. |
\(\dfrac{2A_1A_2}{A_1+A_2}\) |
4. |
\(\dfrac{A_1^2A_2^2}{A_1+A_2}\) |
190. An ammeter having a resistance of
\(0.1~ \Omega\) can measure a maximum current of
\(2~\text{A}\). To convert it into a voltmeter that can measure upto
\(50~\text{V},\) one must add:
| 1. |
a resistance of \(25~\Omega\) in series |
| 2. |
a resistance of \(\dfrac1{25}~\Omega\) in series |
| 3. |
a resistance of \(25~\Omega\) in parallel |
| 4. |
a resistance of \(\dfrac1{25}~\Omega\) in parallel |
191. We compare npn and pnp transistors: let f represent the fraction of the total current carried by electrons within the transistors.
| 1. |
f (npn) > f (pnp) |
| 2. |
f (npn) < f (pnp) |
| 3. |
f (npn) = f (pnp) |
| 4. |
Any of the above may be true depending upon the temperature of the transistors. |
192. If the temperature (in
\(^\circ\text C\)) of a blackbody is increased
\(2\)-fold, then the rate of radiation from the body will become:
| 1. |
\(16\)-fold |
2. |
\(4\)-fold |
| 3. |
less than \(16\)-fold |
4. |
more than \(16\)-fold |
193. A simple pendulum is taken into an elevator that is moving up with an acceleration equal to \(g.\) If its time period was \(T\) before, the new time period will be:
1. \(2T\)
2. \(\sqrt2T\)
3. \(\dfrac{T}{2}\)
4. \(\dfrac{T}{\sqrt2}\)
194. A parallel beam of light of wavelength
\(\lambda\) is incident normally on a single slit of width
\(d,\) and a pattern of maxima and minima are observed on a screen placed far behind the slit. The first minimum (nearest to the central maximum) is formed at an angle
\(\theta,\) where
\(\sin\theta=\)
| 1. |
\(\dfrac{\lambda}{d}\) |
2. |
\(\dfrac{\lambda}{2d}\) |
| 3. |
\(\dfrac{2\lambda}{d}\) |
4. |
\(\dfrac{\lambda}{4d}\) |
195. When sound waves produced under water emerge into the air, then:
| 1. |
the frequency increases, and wavelength decreases. |
| 2. |
the frequency remains constant, but the wavelength decreases. |
| 3. |
the frequency decreases, wavelength remains constant. |
| 4. |
the frequency remains constant but the wavelength increases. |
196. The voltage drop across a silicon diode
\((D)\) which is in forward bias and conducting a small current is approximately
\(0.7\) V. Which of the following sources will deliver a voltage of
\(3\) V?
197. Water flows from a small hole at the bottom of a rectangular tank at \(10\) m/s after it had been filled for \(20\) min. For how much time should the empty tank be filled at the same rate so that the speed of efflux is doubled? (i.e. it becomes \(20\) m/s)
1. \(40\) min
2. \(80\) min
3. \(160\) min
4. \(320\) min
198. The circuit shown in the figure has ideal batteries wired up along with the resistances.
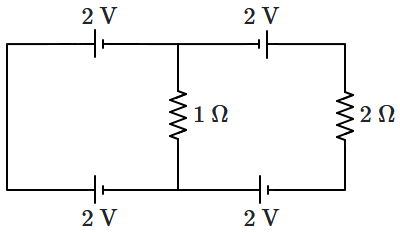
The power dissipated in the resistances in the circuit is:
1.
\(12~\text{J/s}\)
2.
\(9~\text{J/s}\)
3.
\(8~\text{J/s}\)
4.
\(5.5~\text{J/s}\)
199. Two smooth balls
\(A,B \) of radius
\(10\) cm each are placed in a cylinder of diameter
\(30\) cm, as shown. The upper ball
\((A)\) has a mass of
\(2\) kg, while the lower ball
\((B)\) has a mass of
\(4\) kg. The system is at rest. Let the force exerted on the ball
\(A\) by the cylinder be
\(F_1\) along the horizontal (as shown) and that on the ball
\(B,\) along the horizontal be
\(F_2\) (as shown). Then:

| 1. |
\(F_1 < F_2\) |
| 2. |
\(F_1 = F_2\) |
| 3. |
\(F_1 >F_2\) |
| 4. |
the relationship between \(F_1\) and \(F_2\) depends on the mass of the cylinder |
200. Two convex lenses \((L_1,~L_2) \) of focal lengths, \(20\text{ cm}\) \((L_1) \) and \(40\text{ cm}\) \((L_2),\) are placed co-axially at a separation of \(60\text{ cm}.\) If a parallel beam of light is incident on \(L_1, \) the emerging beam from \(L_2 :\)
1. is parallel but wider.
2. is parallel but narrower.
3. is convergent.
4. is divergent.
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course